Abstract
The ribosomal rDNA gene array is an epigenetically-regulated repeated gene locus. While rDNA copy number varies widely between and within species, the functional consequences of subtle copy number polymorphisms have been largely unknown. Deletions in the Drosophila Y-linked rDNA modifies heterochromatin-induced position effect variegation (PEV), but it has been unknown if the euchromatic component of the genome is affected by rDNA copy number. Polymorphisms of naturally occurring Y chromosomes affect both euchromatin and heterochromatin, although the elements responsible for these effects are unknown. Here we show that copy number of the Y-linked rDNA array is a source of genome-wide variation in gene expression. Induced deletions in the rDNA affect the expression of hundreds to thousands of euchromatic genes throughout the genome of males and females. Although the affected genes are not physically clustered, we observed functional enrichments for genes whose protein products are located in the mitochondria and are involved in electron transport. The affected genes significantly overlap with genes affected by natural polymorphisms on Y chromosomes, suggesting that polymorphic rDNA copy number is an important determinant of gene expression diversity in natural populations. Altogether, our results indicate that subtle changes to rDNA copy number between individuals may contribute to biologically relevant phenotypic variation.
Author Summary
The repeated rDNA array gives rise to the nucleolus, which is one of the first described intracellular structures and is known to be involved in key cellular processes such as stress response, cell cycle regulation, RNA modification, and production of more than 90% of all cellular RNAs (the ribosomal RNAs). The rDNA exists in excess; and, although many copies are inactivated through epigenetic mechanisms, the biological significance of inactive copies has been a matter of debate. We present a system that allows for the identification of global gene expression effects stemming from differences in rDNA copy number. We have discovered that deletions in the rDNA locus result in the differential expression of hundreds to thousands of genes. This raises the expectation that important phenotypic variation affecting health and disease might be traced to polymorphic variation in rDNA copy number. Furthermore, the manifold effects of rDNA copy number indicate that considering polymorphisms in the rDNA might bring new light to studies of epigenetic inheritance and its contribution to the heritability of complex traits.
Introduction
The ribosomal rDNA gene array is an epigenetically-regulated repeated locus responsible for producing over 90% of cellular RNAs. The large (35S) rDNA array gives origin to the Nucleolus Organizer Region (NOR) and the 35S rRNA genes are present from fewer than 50 to more than 25,000 copies among eukaryotes [1]. However, even though copy number varies widely between and within species [2], [3], the functional consequences of copy number polymorphisms have been largely unknown, and often assumed to have negligible functional consequence. This is in part because redundant rDNA arrays are found in eukaryotic genomes. In humans, for example, rDNA arrays containing the 35S rRNA genes are found on chromosomes 13, 14, 15, 21, and 22. Similarly, redundant rDNA genes within each array can compensate for copy number polymorphisms to maintain a sufficient supply of rRNAs [4]–[6]; indeed, only a fraction of the rDNA units in a particular array are transcribed at any given time [7], and flexibility in the transcriptional elongation rate allows for control of rRNA output in conditions of rapid cell division and growth. Hence, even though rDNA copy number and rRNA production is regulated by the cell, the relevance of supernumerary or inactive rDNA repeat units has remained elusive. Recent work has investigated whether inactive rDNA copies are necessary for genome stability [8].
In Drosophila melanogaster, rDNA arrays exist as a pair of functionally-redundant loci on the X and Y chromosomes [5], [6], which have further suggested that variable copy number in the Y-linked rDNA array might have little, if any, functional significance. Indeed, even though X0 males are sterile due to loss of Y-linked fertility genes, they appear morphologically normal despite having no Y-linked rDNA [9]. Nevertheless, Paredes and Maggert [10] have recently shown that induced variation in the Y-linked rDNA copy number modifies heterochromatin-induced position effect variegation (PEV), and natural changes in rDNA copy number through development correlate with PEV. Taken together, these findings suggested that polymorphisms in rDNA copy number might be relevant to the maintenance of genome-wide chromatin structure.
Polymorphic naturally occurring Y chromosomes induce Y-linked Regulatory Variation (YRV), which affects the expression of autosomal and X-linked genes [11]. In Drosophila melanogaster, YRV is observed in males differing only in the origin of their Y chromosomes and it is manifested as the differential expression of hundreds of non-Y-linked genes [11]. The source of YRV cannot be simply ascribed to polymorphisms in protein-coding genes [12], [13], nor is it easily mapped to sub-regions of the Y chromosome because of the lack of recombination along the Y-chromosome and the difficulty in manipulating large segments of heterochromatin. The similar contributions of induced deletions in the Y-linked rDNA array and naturally occurring Y chromosome variation to PEV [10]–[13], suggested that naturally occurring rDNA copy number polymorphism might be a significant source of regulatory variation. The hypothesized association between Y-linked regulatory variation and rDNA copy number might provide a molecular framework for the contribution of the Drosophila melanogaster Y chromosome to adaptive phenotypic variation and fitness.
Here we tested the hypothesis that induced deletions in the Y-linked rDNA in an otherwise isogenic background might modulate variation in genome-wide gene expression. Our results indentified hundreds to thousands of genes whose expression is sensitive to deletions in the rDNA. Importantly, many of the induced changes in expression could be replicated in both male and female genotypes. Further, we show significant associations between genome-wide gene expression variation arising from Y chromosomes with targeted deletions in the rDNA loci and variation arising from naturally occurring polymorphic Y chromosomes, which harbor polymorphisms in rDNA copy number. This suggests that variation in rDNA copy number may be an underappreciated source of natural variation with important consequences to the modulation of gene expression across the genome.
Results
To address the relevance of rDNA copy number to genome-wide gene expression we used Y chromosomes containing targeted reductions of rDNA copy number. These chromosomes are otherwise expected to remain structurally unchanged. Although it is possible that these chromosome differ in ways that we cannot detect, four lines of evidence suggest that alterations of the rDNA copy number are the sole difference between these chromosomes: (1) I-CreI-mediated chromosome alterations were genetically and cytologically limited to the rDNA [14], (2) no other identifiable I-CreI consensus site has been cloned from Drosophila, (3) the position effect variegation phenotype arising form these alterations were reverted concomitant with rDNA magnification [10], and (4) Drosophila rDNA is known to be comprised solely of rDNA and two retroelements [15], [16]. Although we cannot formally rule out the possibility that heretofore uncharacterized non-rDNA sequences are in the rDNA loci, for ease of discussion we consider that deletions in the rDNA are the major source of variation between these chromosomes.
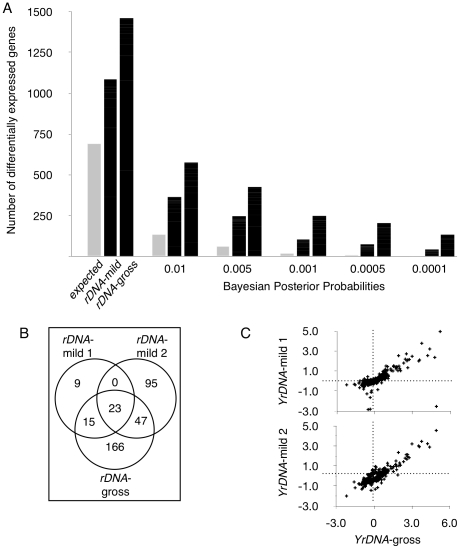
Four Y chromosomes that differ in the copy number of rDNA repeats were introgressed into an isogenic background of autosomes and X chromosome (Figure S1); genome-wide expression was interrogated with microarrays (Figure S2). We compared adult male flies bearing a wild-type Y chromosome to three strains carrying mutant derivative Y chromosomes with reduced rDNA repeats – two mildly (YrDNA-mild, with rDNA copy numbers approximately 87% and 85% of wild-type) and one grossly (YrDNA-gross, 46% of wild type) reduced array. We observed substantial gene expression variation among these strains as compared to the random expectation across a range of P-values (Figure 1A). We expected that laboratory induced deletions in the rDNA array would result in gene expression modulation that would not only be replicated in each independently generated mutant, but would also be more pronounced in mutants lacking a larger proportion of the original locus. Our data are in agreement with both these expectations, and further suggest that rDNA copy number may be the major source of variation between these chromosomes. Accordingly, the numbers of differentially expressed genes were positively correlated with the rDNA deletion size. We observed that the Y chromosome with the lowest rDNA copy number (YrDNA-gross) induced the highest number of expression changes, whereas the two Y chromosomes with mildly-deleted arrays resulted in smaller numbers of differentially expressed genes. This finding is consistent with evidence that chromosomes with fewer rDNA copies have a stronger effect on position effect variegation [10]. Furthermore, the genes identified as differentially expressed in the mild deletions were a subset of those induced in the strain with grossly deleted rDNA (Figure 1B). Accordingly, 42–71% (P<0.001, FDR <0.05) of the genes identified with chromosomes containing mild rDNA deletions were also identified by the Y chromosome with the smallest rDNA copy number. In support of the reproducibility of the gene expression modulation that was observed in PEV across the lines [10], 24% of differentially expressed genes were shared by at least two chromosomes with reduced rDNA arrays, whereas fewer than 0.2% were expected to be shared by chance in randomly permuted datasets (P<0.001). In addition, the direction and magnitudes of changes in expression were significantly correlated (ρ = 0.78–0.84, P<10E−16) between Y chromosomes harboring rDNA deletions (Figure 1C), suggesting a similar responsiveness of the affected genes regardless the size of the rDNA deletion. These data support our contention that a common lesion – namely rDNA copy number variation – underlies this phenotype, and points to the relevance of rDNA copy number variation to modulation of genome-wide gene expression.
Figure 1. Induced deletions in the rDNA locus result in the differential expression of hundreds of genes.
(A) Number of differentially expressed genes for Y chromosomes bearing deletions within the ribosomal DNA (rDNA). Data are given at P<0.05 (first set of data) and other indicated Bayesian Posterior Probabilities. Expected values are calculated from permuted datasets and shown in light gray. rDNA-mild are average numbers for two chromosomes with 87% and 85% wild-type copy number of rDNA, and rDNA-gross is a chromosome with 46% of wild-type rDNA (Figure 6). (B) Venn diagram showing number of differentially expressed genes in each rDNA deletion line relative to the wild-type chromosome (at P<0.001, FDR<0.05). (C) Correlation between the magnitude of change in gene expression (log-fold-changes) for YrDNA-gross (abscissa) and either YrDNA-mild-1 (ordinate – Top panel) or YrDNA-mild-2 (ordinate – Bottom panel). ρ = 0.84 and 0.78, respectively. Fold-changes are for contrasts between each rDNA deletion line and the wild-type chromosome.
Natural polymorphisms and induced deletions of the rDNA show no overt dominant phenotype [3], [17]. Even X0 males, which other than being sterile due to loss of Y-linked fertility genes, appear morphologically normal despite having no Y-linked rDNA [9]. Therefore, we expected that induced rDNA deletion would have impacts on individual gene expression that were generally small. Indeed, we found that 85% of genes whose expression differed significantly from wild-type had changes in expression level of no more than 50% (Figure S3A). It was unexpected and unlikely that the number of genes showing only a 10% change in relative expression would be less than those showing a 20% change. Instead this result is likely an underestimation of genes exhibiting small changes caused by limitation of statistical power to detect them. We projected a linear regression of the number of genes whose differential expression was altered by 10–20%, 20–30%, 30–40%, and 40–50% in an attempt to correct the first decile (Figure S3B–S3D). From that extrapolation, we estimate that as many as 1200–1700 differentially expressed genes at P<0.05 and 360–435 at P<0.01 might escape statistical detection despite our high level of replication (Figure S2). Since we analyzed 8073 unique gene IDs from our microarrays, these estimates suggest that as much as 40% of the genome might be subtly affected by partial rDNA deletion.
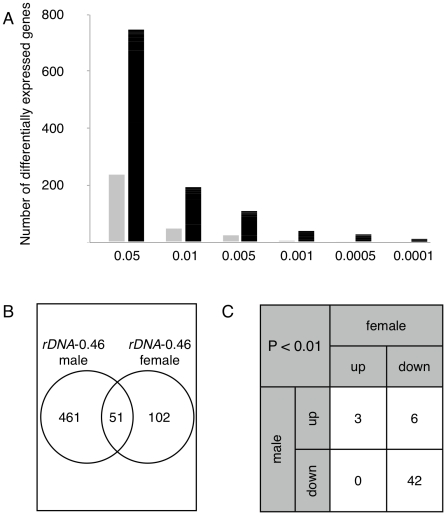
To determine the generality of rDNA induced expression changes, we investigated the differential expression between the rDNA-deleted YrDNA-gross and its ancestral wild-type Y chromosome in females with XXY karyotype. We observed hundreds of gene expression differences between XXYrDNA-gross and isogenic XXY females bearing the wild-type Y chromosome. The number of differentially expressed genes was fewer in XXY females than in males (Figure 2A), possibly due to the smaller fractional rDNA size difference in the female karyotype which contains two wild-type X-linked arrays. Nevertheless, we observed a significant enrichment of differentially expressed genes commonly shared between males and females harboring the same Y chromosome; at P<0.01, 51 genes were shared between the sexes whereas only a single gene was expected to be shared by chance alone. As before, expected numbers were calculated from randomly permuted datasets (Figure 2B, cf. Figure S4A). While the number of affected genes differed between the sexes, we observed a significant association between down-regulated genes (Figure 2C, Figure S5), and a significant correlation in fold-changes between sexes (ρ = 0.45, P<10E–16). A similar trend of up- and down-regulation and commonly shared genes remained across a range of P-values used for ascertaining differential expression (Figure S4B, S4C). Together, our data show that rDNA copy number variation commonly affects male and female transcription and identifies a similar set of “rDNA-sensitive” genes. These data suggest that the response of a gene to rDNA deletion is an attribute of the gene structure and/or its regulation, rather than a sex-dependent effect.
Figure 2. Differentially expressed genes are shared in males and females.
(A) Number of differentially expressed genes in XX/YrDNA-gross females (relative to the wild-type Y chromosome in XX/Y females). Data are presented as in Figure 1A. (B) Venn diagram showing number of differentially expressed genes that are unique or common to X/YrDNA-gross males and XX/YrDNA-gross females (at P<0.001). (C) Breakdown of overlapping genes from (B), separately categorizing genes whose expression was increased (up) or decreased (down) relative to the wild-type Y chromosome in the same genetic background.
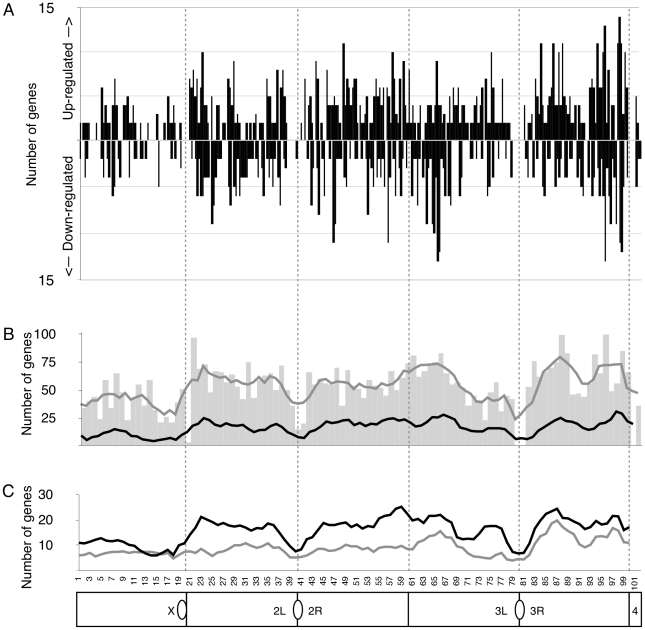
One mechanism for the wide-ranging effects of rDNA copy number on gene expression might arise if deletions of the rDNA compromised the spread of heterochromatin structure to euchromatin from centric heterochromatic regions [18], [19]. Indeed, this simple model is suggested by the observation that three heterochromatin-induced variegating alleles were affected by rDNA deletion [10]. However, studies have established that the spreading of heterochromatin-associated factors is neither long-ranged, processive, or simple [20], [21]. Nonetheless, to address the issue we tested two strong predictions of a simple heterochromatin spreading model: that (i) genes residing in the proximity of heterochromatin will be more strongly affected by changes in rDNA copy number, and that (ii) the majority of gene expression changes will be seen as increase in expression as repressive heterochromatin is reduced. We therefore tested for an enrichment of differentially expressed genes according to their cytological location by plotting the number of differentially expressed genes from all three chromosomes (Figure 3A black bars, Figure 3B black line). We compared the number of differentially expressed genes to the number of analyzed genes from the microarray (Figure 3B gray bars and line), which corrected for uneven gene density across the genome and for any bias in the representation of the genome on our microarrays.
Figure 3. rDNA–responsive genes are found throughout the genome.
(A) Number of differentially expressed genes either up-regulated or down-regulated as a function of cytological location. Each cytological division shows grouped data for all three Y chromosomes (YrDNA-mild-1, YrDNA-mild-2, YrDNA-gross) relative to the wild-type Y chromosome (at P<0.01). (B) Distribution of microarray spots yielding high quality data (gray bars) with scanning 5-division average (gray line). Overlaid scanning 5-division average (black line) of the number of differentially expressed genes. For each window we show the number of differentially expressed genes grouped for all three chromosomes (YrDNA-mild-1, YrDNA-mild-2, and YrDNA-gross) relative to the wild-type Y chromosome (P<0.01). (C) Scanning 5-division average of number of differentially expressed genes only from males (black) and females (gray) bearing YrDNA-gross (at P<0.01). Cytological divisions are aligned across entire figure (dotted vertical lines). Stylized chromosome map represents euchromatic regions of the genome and location of centromeres and centric heterochromatin (ovals).
We found that while the number of differentially expressed genes appears to drop near the cytological bands juxtaposed to centric heterochromatic blocks (bands 20, 40/41, and 80/81), this is indistinguishable from our expectation based on lower representation of genes from these regions. Moreover, we could discern no general trend for genes near the telomeres (bands 1, 21, and 60/61). Furthermore, while X-linked loci were less affected than were chromosome 2- and 3-linked genes (43–47% the frequency), these trends were not statistically different from that expected given the distribution of represented genes (Figure S6A). Next, we analyzed the genomic region at the transition between the X-linked 20 megabases of centric heterochromatin and adjacent euchromatin. None of the first 20 genes were differentially expressed. The same held true for the telomere of the long arm of the X chromosome. Finally, we assessed the density of genes responding to rDNA copy number along all the chromosome arms in a sliding 2 megabase window at 1 megabase increments. Significance was determined empirically by analyzing 1000 sets of randomly sampled genes taken from the entire genome. Each set of random data had a sample size equal to the number of differentially expressed genes. No significant overrepresentation of differentially expressed genes was detected in any increment. Thus, our analysis indicated that the genes affected by rDNA deletion are randomly distributed in the genome, and are neither influenced by proximity to heterochromatin nor chromosome linkage. There was also no preponderant increase or decrease of differential expression. Corroborating our conclusion that rDNA deletion affects the same set of genes in males and females, we saw that the distribution of affected genes in males and females was coincident (Figure 3C, Figure S6B). Taken together these results argue against a simple heterochromatin spreading model but instead suggest that loci with sensitivity to rDNA copy number variation are scattered through the genome.
An intriguing possibility is that polymorphisms of the rDNA copy number in naturally occurring Y chromosomes could account in part for the differential genome-wide modulation of gene expression exerted by these chromosomes. One indication that such rDNA-driven Y-linked variation is relevant came from categorizing the differential expressed genes by Gene Ontogeny (GO) category. These analyses pointed to five categories that overlapped with those discovered by comparing differential gene expression due to natural Y chromosome polymorphisms (Table 1) [11]. Importantly, energy metabolism and rDNA activity are closely associated [22], [23]. Our data suggest that rDNA copy number itself might play a role regulating energy metabolism through modulation of gene expression of genes responsible for energy management or membrane metabolism. Accordingly, we observed that in both males and females “rDNA sensitive” genes are highly enriched for proteins localized to the mitochondrial membrane (46 genes in males, P<2.1E–10; 38 genes in females P<7.2E–12) and involved in electron transport (44 genes in males, P<2.3E–05; 38 genes in females, P<1.4E–09). These enrichments are significant in both males and females, but collectively account for 16% and 9% respectively of the differentially expressed genes (at P<0.05). Although energy and lipid metabolism may be influenced by rDNA copy number, they are not the sole targets.
Table 1. rDNA copy number affects genes related to energy metabolism.
| Category | Description | N | P-value | |
| male | GO: 0031966 | Mitochondrial membrane | 46 | 2.1E−10 |
| GO: 0006118 | Electron transport | 44 | 2.3E−05 | |
| GO: 0016298 | Lipase activity | 16 | 1.1E−03 | |
| GO: 0006629 | Lipid metabolic process | 53 | 1.4E−03 | |
| GO: 0006631 | Fatty acid metabolism | 9 | 1.8E−02 | |
| female | GO: 0031966 | Mitochondrial membrane | 38 | 7.2E−12 |
| GO: 0006118 | Electron transport | 38 | 1.4E−09 |
Gene Ontogeny (GO) categories whose genes are disproportionally represented among those with expression significantly altered in males (top) and females (bottom). P-values are corrected for multiple testing using the modified Bonferroni correction of the statistical software package GeneMerge.
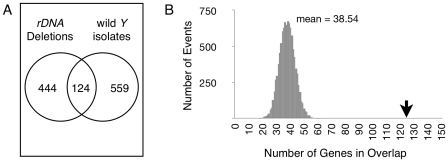
To address the relevance of rDNA copy number in natural populations, we integrated data from gene expression variation due to natural Y-linked polymorphisms and gene expression variation due to deletions in the rDNA array. We found an unexpected level of overlap in the identity of differentially expressed genes when compared to the level expected by chance alone (Figure 4A, Figure S7). We found 124 genes with evidence for differential expression (P<0.005) in comparisons between chromosomes with induced changes in rDNA copy number and in comparisons between naturally-occurring chromosomes. We assessed statistical significance of this overlap in two ways. First, the same procedure for identifying differential expression was applied to datasets in which the expression data were randomly permuted. These analyses showed that only five genes were identified as differentially expressed in both permuted datasets. Second, we generated random samples of size 568 (the observed number of differentially expressed genes shared between the induced rDNA chromosomes) and 683 (the observed number of differentially expressed genes shared between the natural chromosomes), and counted the number of genes that appeared in the overlap of both sets in each of 10,000 replicated of this experiment. The analysis indicates that the observed overlap of 124 genes is significantly enriched compared to random datasets (P<0.0001, Figure 4B). This enrichment shows that a gene affected by YRV is predictive of being affected by rDNA copy number.
Figure 4. Differentially expressed genes are shared between chromosomes with induced rDNA deletions and naturally occurring Y chromosomes.
(A) Venn diagram showing number of differentially expressed genes unique to Y chromosomes with induced rDNA copy number changes (“rDNA Deletions”) or natural Y chromosomes (“wild Y isolates”), and overlap of genes common to both groups (at P<0.005). (B) Event histogram showing that 10,000 randomly-generated datasets produces an average of 38.54 genes shared between rDNA Deletions and wild Y isolates. Arrow shows the observed value of 124 (from (A)).
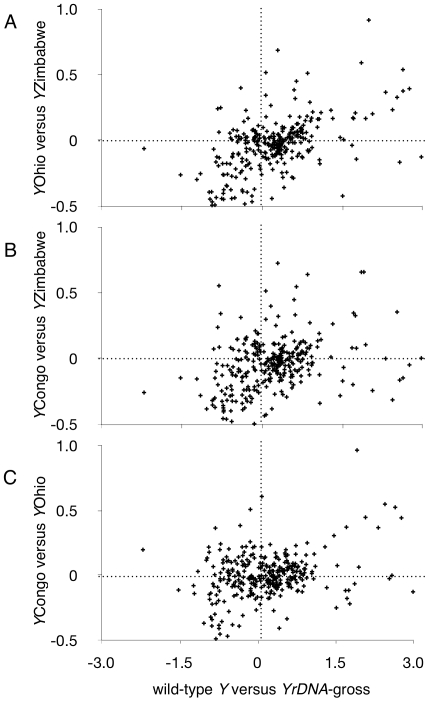
Finally, we analyzed the set of genes whose expression is affected by rDNA deletion, and found that absolute fold changes due to these deletions is significantly correlated with expression variation arising due to naturally-occurring Y chromosome polymorphisms (ρ = 0.25–0.55, P<1.0E–12, Figure 5). These correlation coefficients indicate that approximately 5–30% of gene expression variation detected on natural Y chromosomes might be due to polymorphisms in the rDNA loci. Using real-time PCR we confirmed that the natural Y chromosomes possessed polymorphisms in rDNA copy number, the range of which included the two mild rDNA deletions used in this study (Figure 6). Moreover, the effect of naturally occurring Y chromosomes from YOhio and YZimbabwe on heterochromatin-induced position effect variegation were consistent with effects seen for induced deletions: lower rDNA copy number correlated with increased suppression of variegation [10], [12].
Figure 5. Correlations between absolute fold changes in rDNA responsive genes identified by induced rDNA deletions and absolute fold changes arising from natural Y chromosome polymorphisms.
(A) Correlation of absolute log-fold-changes comparing differentially expressed genes between YrDNA-gross and wild-type Y (abscissa) to those differentially expressed between YZimbabwe (YZimb.) and YOhio (ordinate); ρ = 0.55, P<10E−16. (B) Correlation of absolute log-fold-changes for YrDNA-gross versus wild-type Y compared to YCongo versus YZimbabwe; ρ = 0.38, P<10E−12. (C) Correlation of absolute log-fold-changes for YrDNA-gross versus wild-type Y compared to YCongo versus YOhio; ρ = 0.25, P<10E−6.
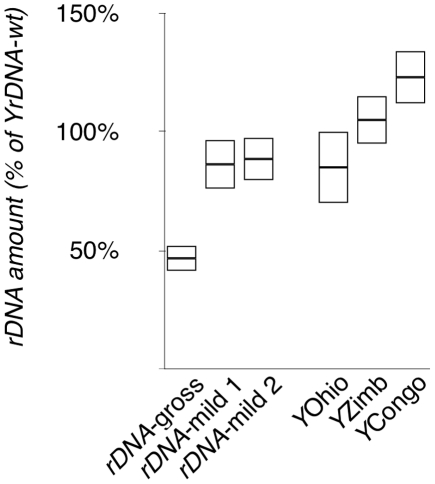
Figure 6. Quantification of rDNA copy number of the chromosomes in this study.
Quantification of rDNA copy number determined by Real Time Polymerase Chain Reaction, presented as percentage of a common wild-type Y chromosome (the progenitor of YrDNA-mild-1, YrDNA-mild-2, and YrDNA-gross). Plots show average ± 1 S.D.
Discussion
Our work identified the consequences of induced rDNA deletions to the expression of hundreds to thousands of genes scattered through the genome of both males and females. The data are reproducible between independently-generated chromosomes derived from a common ancestor, with lines containing smaller deletions showing smaller effects on gene expression. Although we cannot exclude the possibility that some undetected Y-linked differences other than rDNA copy number might confound some of the effects we have seen, we interpret the changes in gene expression to be, at least in part, a consequence of induced deletions in the rDNA locus. Previous studies lend credence to this interpretation [10], [14], [17]. Furthermore, the results herein reported are well in line with the expectation that the measured rDNA deletions partly underlie the expression phenotypes we report. Accordingly, we observed that the Y chromosome with the lowest rDNA copy number (YrDNA-gross) induced the highest number of expression changes, whereas the two Y chromosomes with mildly-deleted arrays resulted in smaller numbers of differentially expressed genes. This finding is consistent with evidence that chromosomes with fewer rDNA copies have a stronger effect on position effect variegation [10]. Furthermore, the genes identified as differentially expressed in the mild deletions were a subset of those induced in the strain with grossly deleted rDNA. Furthermore, we showed that such rDNA-responsive genes are not physically clustered or near heterochromatin; instead, affected genes are distributed across the genome and show functional enrichments for genes encoding proteins localized to the mitochondria and involved in electron transfer. This finding is reminiscent of previous observations for a close link between rDNA activity and energy metabolism. Furthermore, our analyses suggested that naturally occurring copy number polymorphism in the rDNA array might be an important and underappreciated source of regulatory variation in natural populations.
Epigenetic regulation of chromatin structure is necessary for the stability of the rDNA array and the nucleolus. Indeed, destabilization of rDNA copy number and nucleolar structure are markers of aging and cancer progression [24], [25] and our findings raise the possibility that rDNA alterations might underlie some of these pleiotropic phenotypes through its consequences on genome-wide gene expression. Mechanistically, the transcriptional activity of the rDNA locus is regulated through histone modifications and allows for nucleolus formation from even a single rDNA repeat unit [8], [26]–[28]. Furthermore, many chromatin components known to regulate rDNA activity and stability are dosage sensitive. For instance, the Sir2-family proteins are required for the silencing of genes inserted into the rDNA repeated array, rDNA stability, and telomeric silencing [29], [30]. In Drosophila, Sir2 has an essential role in both euchromatic gene repression and heterochromatic silencing [29]. Hence, it is conceivable that rDNA expression and rDNA copy number are related, and perturbations to either might yield heritable alteration in genome structure with consequences on gene expression of unlinked genes. Accordingly, we propose that short and long rDNA arrays may result in the differential recruitment of nuclear proteins for rRNA expression/processing and nucleolar structure. In such a model, variation of protein dosages due to either the release of proteins required for silencing of non-expressed rDNA repeats (which might be more abundant in the larger array) or due to the recruitment of proteins required for the higher expression of a smaller number of expressed rDNA repeats (as might be the case in the shorter rDNA arrays) might be expected. Indeed, Michel and colleagues showed that spontaneous rDNA deletions in yeast caused decreased cellular abundance of Sir2 mRNA and protein [31]. Hence, the widespread effects of rDNA copy number on genome-wide expression might arise from imbalances in the nuclear protein pool, which includes chromatin and transcription factors, that might result from the requirements for silencing and expression of variable numbers of rDNA repeats or structural elements within nucleoli. Alternatively, Paredes and Maggert showed alterations in nucleolus stability as a consequence of reduced rDNA copy number [10]. The dynamic composition of the nucleolus [32] may differ in nucleoli coalesced around long or short rDNA arrays.
Paredes and Maggert showed that rDNA copy number altered the balance of euchromatin and heterochromatin in Drosophila models of position effect variegation [10]. Those observations suggested a global effect of rDNA copy number on chromatin structure. Significantly, multiple studies show a complex regulation of heterochromatin spreading at natural euchromatin-heterochromatin transition zones and chromosome rearrangement breakpoint junctions. Yasuhara and Wakimoto showed that increased levels of HP1 association at variegating euchromatic-heterochromatic junctions is not homogenous across the chromosome [20], and both Talbert and Henikoff, and Ahmad and Henikoff showed that heterochromatin spreading does not translate into uniform responses for closely-linked genes [33], [34]. Most recently, Vogel and colleagues showed that while a variegating white + gene was responsive to changes in chromatin environment, the majority of linked genes remained unresponsive to the spreading of HP1 from nearby heterochromatin [21]. Hence, it was not surprising that genes responding to rDNA copy number were distributed across the genome. More important, this observation does not contradict the view that rDNA copy number impacts global chromatin structure. To the contrary, recent studies suggested that heterochromtain-induced PEV [12], [13] and euchromatic gene expression might be related [11], and rDNA deletions affect heterochromatin-induced PEV [10]: the data we present here supports the unity of these observations by showing that deletions within the rDNA affect euchromatic gene expression, uncover a significantly overlapping subset of genes, and account for a fraction of naturally occurring YRV.
Although rDNA copy number polymorphism may only account for a small fraction of YRV, our data indicate a relevant contribution with significant enrichments in the number of differentially expressed genes that are shared by both rDNA deletions lines and naturally occurring Y chromosomes. This is unexpected because rDNA copy number is typically thought to be without genetic consequence. The remainder of YRV may map to other heterochromatin of the Y chromosome, either simple repeats or transposable elements. Similarly, the remainder of rDNA-linked variation may be ameliorated or epistatically masked by compensatory polymorphisms elsewhere in the heterochromatin. Finally, our results raise the issue of how widespread these effects may be in other chromosomes and organisms containing highly variable rDNA loci. Indeed, the large and varied number of rDNA copies is a common characteristic of eukaryotic genomes [2]. The human diploid karyotype has ten 35S and two 5S arrays, each highly variable with regard to size and activity [35]. In plants such as Arabidopsis, the 5S rDNA arrays contain heritable regulatory chromatin structure [36], and in flax, heritable induced phenotypic variation is mapped to rDNA copy number [37], [38]. Although rDNA copy number in Drosophila may vary over an order of magnitude, the upper limits remain consistent across many studies [1]. What limits the expansion of rDNA is yet to be established, but our results suggest that superfluous rDNA repeats – inactive or active – have genetic consequence to gene expression, which might impose a heretofore underappreciated pressure against excessive expansion or contraction. We envision this genetic consequence to be heritable as a result of copy number polymorphism, suggesting that rDNA copy number is an important consideration in understanding the evolution of a genome. Indeed, the rDNA array has a uniquely dynamic biology of copy number loss and gain that occurs meiotically and somatically through development [5], [39]–[41]. Hence, the ability of the rDNA to change in copy number and activity might provide a novel mechanism for adaptation to environmental changes by maintaining a euchromatin/heterochromatin balance that is most conducive for fitness.
Materials and Methods
Drosophila stocks and crosses
Drosophila melanogaster Y-chromosomes containing targeted deletions in the rDNA locus were generated as previously described [17]. These chromosomes were derived from a common single male ancestor in the laboratory that was subjected to targeted deletions within the rDNA locus, and thus created an allelic series of Y chromosomes virtually identical with the exception of variation in rDNA copy number. These Y chromosomes were introgressed into an isogenic (X chromosome, autosomes, and mitochondrial genome) laboratory stock [11] by crossing XY males to females homozygous for the markers yellow, brown, ebony, cubitus interruptus, and eyeless (y/y; bw/bw; e/e; ci ey/ci ey) for two generations (Figure S1). This isogenic stock is expected to contain very little genetic variation, and upon receipt was subjected to no fewer than eight additional generations of brother-sister mating to reinforce homozygosity of the genetic background. Four Y chromosomes were analyzed: The original Y chromosome that contains a wild type rDNA array (100%), two derived chromosomes with mild deletions 87% (YrDNA-0.87) and 85% (YrDNA-0.85) of wild-type, and one grossly reduced derived chromosome that contains 46% (YrDNA-0.46) of wild-type. Flies were grown under 24 h light at constant temperature (25°C) and humidity (80%). XXY female flies were obtained by crossing males from the isogenic Y chromosome substitution lines described above to females from a laboratory stock containing a compound (attached) X chromosome, C(1)M4, y.
rDNA copy number analyses
rDNA copy number in Y-chromosome substitution strains containing the rDNA deletions were checked with a random subset of flies used for gene expression analysis. rDNA copy number was measured as described in Paredes and Maggert [17]. For each stock whose Y-linked rDNA array was measured males were crossed with C(1)DX, y 1 f 1 bb 0, which lacks all X-linked rDNA. The resulting C(1)DX/Y females contain only Y-linked rDNA, whose copy number was determined with quantitative Real-time PCR using conserved rDNA primers that amplified 18S subunit rRNA gene sequence. For Real-time PCR analyses, five biological replicates consisting of three experimental replicates from individual flies were sampled for each genotype. Real-time PCR analyses were carried out with the Power Sybr Green Master Mix (Applied Biosystems, Foster City, California), and amplification profiles were obtained with an ABI Step-One Real-time PCR (Applied Biosystems, Foster City, California). Reactions were quality-confirmed for the absence of dimers or nonspecific amplification by analyzing melting curve kinetics and electrophoretic analysis of products.
Gene expression analyses
Microarrays were approximately 18,000-feature cDNA arrays spotted with Drosophila melanogaster cDNA PCR products. For RNA extraction, newly emerged male flies were collected and aged for three days at 25°C, after which they were flash frozen in liquid nitrogen and stored at −80°C. When females were analyzed, they were collected within 7 hours of eclosion to assure they were unmated prior to aging under the same conditions as were males. Total RNA was extracted from whole flies using TRIZOL (Gibco-BRL, Life Technologies, Gaithersburg, Maryland). cDNA synthesis, labeling with fluorescent dyes (Cy3 and Cy5) and hybridization reactions were carried out using 3DNA protocols and reagents (Genisphere Inc., Hatfield, Pennsylvania). Slides were scanned using AXON 4000B scanner (Axon Instruments, Foster City, California) and the GenePix Pro 6.0 software. Stringent quality-control criteria were used to ensure reliability of foreground intensity reads for both Cy3 and Cy5 channels. Foreground fluorescence of dye intensities was normalized by the Loess method in the library Limma [42], [43] of the software R. Significance of variation in gene expression due to Y chromosome origin was assessed with linear models and empirical Bayes moderated F statistics in Limma [42], [43]. P values were adjusted for multiple testing by using the method of Benjamini and Hochberg to control the false discovery rate [44]. Test results were considered to be significant if the adjusted P values were less than 0.05, nominally controlling the expected false discovery rate to no more than 5%. Differential expression was also assessed using the Bayesian Analysis of Gene Expression Levels (BAGEL) model [45]. Results were robust to the choice of linear models in Limma or BAGEL. False discovery rates were estimated based on the variation observed when randomized versions of the original dataset were analyzed. Similarly, expected values for the overlap between independent datasets were estimated by applying the same statistical procedure (Limma or BAGEL) to permuted versions of the datasets. Significance of the overlap was further assessed by generating samples of sizes that matched the number of differentially expressed genes in separate datasets (induced rDNA deletions and natural Y chromosomes) and determining the number of genes found in both random samples; the mean number was 38.54, and the maximum was 62 genes in 10,000 trials. Enrichment in gene ontology categories was assessed using a modified Bonferroni correction with GeneMerge [46]. Microarray gene expression data can be obtained at the GEO database (GSE27695).
Supporting Information
(A) Crossing scheme to introgress Y chromosomes to a common and isogenic genetic background. y (yellow), bw (brown), e (ebony), ci (cubitus interruptus), ey (eyeless) were used as recessive genetic markers. (B) Crossing scheme to generate XX/Y aneuploid females. Circle represents common centromere linking compound-X chromosome arms.
(0.30 MB TIF)
(A) Array design for comparing males. rDNA sizes were determined at the onset of the experiments. rDNA-mild-1 is approximately 87% the wild-type size, rDNA-mild-2 is 85% the wild-type size, and rDNA-gross is 46% the wild-type size (Figure 6). Lines are direct comparisons and indicate number of replicates. (B) Array design for comparing females.
(0.24 MB TIF)
(A) Number of differentially expressed genes (at P<0.01), broken down by decile “fold-changes” along the abscissa for the YrDNA-gross deleted chromosome, presented as absolute counts (solid lines and ordinal values) and cumulative percentage (dotted lines at 20% increments). (B) Data from (A) graphed as separate deciles to show quality of estimation. Log scale inset shows fold-changes 1.1 to 1.5. (C) Data from Figure 1A (P<0.05) graphed as separate deciles to show quality of estimation. Log scale inset shows fold-changes 1.1 to 1.5. (D) Number of differentially expressed genes (data from (B) and (C)) with estimated number of genes whose expression was modulated by less than 10% and missed due to limited statistical power (white). Data were generated from linear regression of subsequent four deciles. Projections are shown for P<0.05 (left) and P<0.01 (right).
(0.36 MB TIF)
(A) Expectation of overlap at P<0.01 based on chance alone. cf. Figure 2B. (B) Data from Figure 2A at P<0.005 versus values expected by chance alone. (B) Data from Figure 2A at P<0.005 versus values expected by chance alone. (C) For P<0.001. cf. Figure 2B, 2C.
(0.47 MB TIF)
Correlation of log-fold-changes comparing differentially expressed genes between YrDNA-gross and the wild-type Y in males (abscissa) to those differentially expressed between YrDNA-gross and the wild-type Y in females (ordinate); ρ = 0.45, P<0.0001.
(0.22 MB TIF)
(A) Ratio of number of differentially expressed genes to total number of genes on the microarray (data from Figure 3B). Solid horizontal line shows the average, dotted lines show two standard deviations. (B) Cumulative counts of differentially expressed genes between YrDNA-gross and wild-type Y (at P<0.01) in males (black), females (gray), and females multiplied by a correction (dotted gray) to more easily compare trends (data from Figure 3C).
(0.29 MB TIF)
(A) Observed number of shared differentially expressed genes across pairwise chromosome comparisons. P<0.05 data are shown above the diagonal, P<0.005 are shown below the diagonal, and total number (shared plus unique) of differentially expressed genes (P<0.05/P<0.005) are shown on the diagonal (bold, gray background). (B) Expected numbers of genes shared between induced rDNA deletion Y chromosomes and natural Y chromosomes, calculated from randomized datasets (N for individual pairwise comparisons are on diagonal) at P<0.05 (above diagonal) and P<0.005 (below diagonal).
(0.27 MB TIF)
Footnotes
References
- 1.Long EO, Dawid IB. Repeated genes in eukaryotes. Annu Rev Biochem. 1980;49:727–764. doi: 10.1146/annurev.bi.49.070180.003455. [DOI] [PubMed] [Google Scholar]
- 2.Prokopowich CD, Gregory TR, Crease TJ. The correlation between rDNA copy number and genome size in eukaryotes. Genome. 2003;46:48–50. doi: 10.1139/g02-103. [DOI] [PubMed] [Google Scholar]
- 3.Lyckegaard EM, Clark AG. Ribosomal DNA and Stellate gene copy number variation on the Y chromosome of Drosophila melanogaster. Proc Natl Acad Sci U S A. 1989;86:1944–1948. doi: 10.1073/pnas.86.6.1944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Karpen GH, Schaefer JE, Laird CD. A Drosophila rRNA gene located in euchromatin is active in transcription and nucleolus formation. Genes Dev. 1988;2:1745–1763. doi: 10.1101/gad.2.12b.1745. [DOI] [PubMed] [Google Scholar]
- 5.Tartof KD. Regulation of ribosomal RNA gene multiplicity in Drosophila melanogaster. Genetics. 1973;73:57–71. doi: 10.1093/genetics/73.1.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Terracol R, Prud'homme N. 26S and 18S rRNA synthesis in bobbed mutants of Drosophila melanogaster. Biochimie. 1981;63:451–455. doi: 10.1016/s0300-9084(81)80020-x. [DOI] [PubMed] [Google Scholar]
- 7.French SL, Osheim YN, Cioci F, Nomura M, Beyer AL. In exponentially growing Saccharomyces cerevisiae cells, rRNA synthesis is determined by the summed RNA polymerase I loading rate rather than by the number of active genes. Mol Cell Biol. 2003;23:1558–1568. doi: 10.1128/MCB.23.5.1558-1568.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ide S, Miyazaki T, Maki H, Kobayashi T. Abundance of ribosomal RNA gene copies maintains genome integrity. Science. 2010;327:693–696. doi: 10.1126/science.1179044. [DOI] [PubMed] [Google Scholar]
- 9.Bridges CB. Non-Disjunction as Proof of the Chromosome Theory of Heredity (Concluded). Genetics. 1916;1:107–163. doi: 10.1093/genetics/1.2.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Paredes S, Maggert KA. Ribosomal DNA contributes to global chromatin regulation. Proc Natl Acad Sci U S A. 2009;106:17829–17834. doi: 10.1073/pnas.0906811106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lemos B, Araripe LO, Hartl DL. Polymorphic Y chromosomes harbor cryptic variation with manifold functional consequences. Science. 2008;319:91–93. doi: 10.1126/science.1148861. [DOI] [PubMed] [Google Scholar]
- 12.Lemos B, Branco AT, Hartl LD. Epigenetic modulation of gene expression by polymorphic Y chromosomes: Sexual conflict, chromatin components, and immune response. Proc Natl Acad Sci U S A in press. 2010 doi: 10.1073/pnas.1010383107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jiang PP, Hartl DL, Lemos B. Y Not a Dead End: Epistatic Interactions Between Y-linked Regulatory Polymorphisms and Genetic Background Affect Global Gene Expression in Drosophila melanogaster. Genetics. 2010 doi: 10.1534/genetics.110.118109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Maggert KA, Golic KG. Highly efficient sex chromosome interchanges produced by I-CreI expression in Drosophila. Genetics. 2005;171:1103–1114. doi: 10.1534/genetics.104.040071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pellegrini M, Manning J, Davidson N. Sequence arrangement of the rDNA of Drosophila melanogaster. Cell. 1977;10:213–214. doi: 10.1016/0092-8674(77)90215-x. [DOI] [PubMed] [Google Scholar]
- 16.Wellauer PK, Dawid IB. The structural organization of ribosomal DNA in Drosophila melanogaster. Cell. 1977;10:193–212. doi: 10.1016/0092-8674(77)90214-8. [DOI] [PubMed] [Google Scholar]
- 17.Paredes S, Maggert KA. Expression of I-CreI endonuclease generates deletions within the rDNA of Drosophila. Genetics. 2009;181:1661–1671. doi: 10.1534/genetics.108.099093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rudolph T, Yonezawa M, Lein S, Heidrich K, Kubicek S, et al. Heterochromatin formation in Drosophila is initiated through active removal of H3K4 methylation by the LSD1 homolog SU(VAR)3-3. Mol Cell. 2007;26:103–115. doi: 10.1016/j.molcel.2007.02.025. [DOI] [PubMed] [Google Scholar]
- 19.Talbert PB, Henikoff S. Spreading of silent chromatin: inaction at a distance. Nat Rev Genet. 2006;7:793–803. doi: 10.1038/nrg1920. [DOI] [PubMed] [Google Scholar]
- 20.Yasuhara JC, Wakimoto BT. Molecular landscape of modified histones in Drosophila heterochromatic genes and euchromatin-heterochromatin transition zones. PLoS Genet. 2008;4:e16. doi: 10.1371/journal.pgen.0040016. doi: 10.1371/journal.pgen.0040016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vogel MJ, Pagie L, Talhout W, Nieuwland M, Kerkhoven RM, et al. High-resolution mapping of heterochromatin redistribution in a Drosophila position-effect variegation model. Epigenetics Chromatin. 2009;2:1. doi: 10.1186/1756-8935-2-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Guarente L. Sir2 links chromatin silencing, metabolism, and aging. Genes Dev. 2000;14:1021–1026. [PubMed] [Google Scholar]
- 23.Murayama A, Ohmori K, Fujimura A, Minami H, Yasuzawa-Tanaka K, et al. Epigenetic control of rDNA loci in response to intracellular energy status. Cell. 2008;133:627–639. doi: 10.1016/j.cell.2008.03.030. [DOI] [PubMed] [Google Scholar]
- 24.Johnson FB, Sinclair DA, Guarente L. Molecular biology of aging. Cell. 1999;96:291–302. doi: 10.1016/s0092-8674(00)80567-x. [DOI] [PubMed] [Google Scholar]
- 25.Drygin D, Rice WG, Grummt I. The RNA polymerase I transcription machinery: an emerging target for the treatment of cancer. Annu Rev Pharmacol Toxicol. 2010;50:131–156. doi: 10.1146/annurev.pharmtox.010909.105844. [DOI] [PubMed] [Google Scholar]
- 26.Peng JC, Karpen GH. H3K9 methylation and RNA interference regulate nucleolar organization and repeated DNA stability. Nat Cell Biol. 2007;9:25–35. doi: 10.1038/ncb1514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Peng JC, Karpen GH. Heterochromatic genome stability requires regulators of histone H3 K9 methylation. PLoS Genet. 2009;5:e1000435. doi: 10.1371/journal.pgen.1000435. doi: 10.1371/journal.pgen.1000435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kobayashi T. A new role of the rDNA and nucleolus in the nucleus--rDNA instability maintains genome integrity. Bioessays. 2008;30:267–272. doi: 10.1002/bies.20723. [DOI] [PubMed] [Google Scholar]
- 29.Rosenberg MI, Parkhurst SM. Drosophila Sir2 is required for heterochromatic silencing and by euchromatic Hairy/E(Spl) bHLH repressors in segmentation and sex determination. Cell. 2002;109:447–458. doi: 10.1016/s0092-8674(02)00732-8. [DOI] [PubMed] [Google Scholar]
- 30.Furuyama T, Banerjee R, Breen TR, Harte PJ. SIR2 is required for polycomb silencing and is associated with an E(Z) histone methyltransferase complex. Curr Biol. 2004;14:1812–1821. doi: 10.1016/j.cub.2004.09.060. [DOI] [PubMed] [Google Scholar]
- 31.Michel AH, Kornmann B, Dubrana K, Shore D. Spontaneous rDNA copy number variation modulates Sir2 levels and epigenetic gene silencing. Genes Dev. 2005;19:1199–1210. doi: 10.1101/gad.340205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Boisvert FM, van Koningsbruggen S, Navascues J, Lamond AI. The multifunctional nucleolus. Nat Rev Mol Cell Biol. 2007;8:574–585. doi: 10.1038/nrm2184. [DOI] [PubMed] [Google Scholar]
- 33.Talbert PB, Henikoff S. A reexamination of spreading of position-effect variegation in the white-roughest region of Drosophila melanogaster. Genetics. 2000;154:259–272. doi: 10.1093/genetics/154.1.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ahmad K, Henikoff S. Modulation of a transcription factor counteracts heterochromatic gene silencing in Drosophila. Cell. 2001;104:839–847. doi: 10.1016/s0092-8674(01)00281-1. [DOI] [PubMed] [Google Scholar]
- 35.Stults DM, Killen MW, Pierce HH, Pierce AJ. Genomic architecture and inheritance of human ribosomal RNA gene clusters. Genome Res. 2008;18:13–18. doi: 10.1101/gr.6858507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Riddle NC, Richards EJ. The control of natural variation in cytosine methylation in Arabidopsis. Genetics. 2002;162:355–363. doi: 10.1093/genetics/162.1.355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cullis CA. Mechanisms and control of rapid genomic changes in flax. Ann Bot. 2005;95:201–206. doi: 10.1093/aob/mci013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Schneeberger RG, Cullis CA. Specific DNA alterations associated with the environmental induction of heritable changes in flax. Genetics. 1991;128:619–630. doi: 10.1093/genetics/128.3.619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hawley RS, Tartof KD. A two-stage model for the control of rDNA magnification. Genetics. 1985;109:691–700. doi: 10.1093/genetics/109.4.691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tartof KD. Unequal mitotic sister chromatin exchange as the mechanism of ribosomal RNA gene magnification. Proc Natl Acad Sci U S A. 1974;71:1272–1276. doi: 10.1073/pnas.71.4.1272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cohen S, Yacobi K, Segal D. Extrachromosomal circular DNA of tandemly repeated genomic sequences in Drosophila. Genome Res. 2003;13:1133–1145. doi: 10.1101/gr.907603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Smyth GK. Linear models and empirical bayes methods for assessing differential expression in microarray experiments. Stat Appl Genet Mol Biol. 2004;3:Article3. doi: 10.2202/1544-6115.1027. [DOI] [PubMed] [Google Scholar]
- 43.Gentleman R. New York: Springer; 2005. Bioinformatics and Computational Biology Solutions Using R and Bioconductor. [Google Scholar]
- 44.Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. Journal of the Royal Statistical Society. 1995;57:289–300. [Google Scholar]
- 45.Townsend JP, Hartl DL. Bayesian analysis of gene expression levels: statistical quantification of relative mRNA level across multiple strains or treatments. Genome Biol. 2002;3:RESEARCH0071. doi: 10.1186/gb-2002-3-12-research0071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Castillo-Davis CI, Hartl DL. GeneMerge--post-genomic analysis, data mining, and hypothesis testing. Bioinformatics. 2003;19:891–892. doi: 10.1093/bioinformatics/btg114. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(A) Crossing scheme to introgress Y chromosomes to a common and isogenic genetic background. y (yellow), bw (brown), e (ebony), ci (cubitus interruptus), ey (eyeless) were used as recessive genetic markers. (B) Crossing scheme to generate XX/Y aneuploid females. Circle represents common centromere linking compound-X chromosome arms.
(0.30 MB TIF)
(A) Array design for comparing males. rDNA sizes were determined at the onset of the experiments. rDNA-mild-1 is approximately 87% the wild-type size, rDNA-mild-2 is 85% the wild-type size, and rDNA-gross is 46% the wild-type size (Figure 6). Lines are direct comparisons and indicate number of replicates. (B) Array design for comparing females.
(0.24 MB TIF)
(A) Number of differentially expressed genes (at P<0.01), broken down by decile “fold-changes” along the abscissa for the YrDNA-gross deleted chromosome, presented as absolute counts (solid lines and ordinal values) and cumulative percentage (dotted lines at 20% increments). (B) Data from (A) graphed as separate deciles to show quality of estimation. Log scale inset shows fold-changes 1.1 to 1.5. (C) Data from Figure 1A (P<0.05) graphed as separate deciles to show quality of estimation. Log scale inset shows fold-changes 1.1 to 1.5. (D) Number of differentially expressed genes (data from (B) and (C)) with estimated number of genes whose expression was modulated by less than 10% and missed due to limited statistical power (white). Data were generated from linear regression of subsequent four deciles. Projections are shown for P<0.05 (left) and P<0.01 (right).
(0.36 MB TIF)
(A) Expectation of overlap at P<0.01 based on chance alone. cf. Figure 2B. (B) Data from Figure 2A at P<0.005 versus values expected by chance alone. (B) Data from Figure 2A at P<0.005 versus values expected by chance alone. (C) For P<0.001. cf. Figure 2B, 2C.
(0.47 MB TIF)
Correlation of log-fold-changes comparing differentially expressed genes between YrDNA-gross and the wild-type Y in males (abscissa) to those differentially expressed between YrDNA-gross and the wild-type Y in females (ordinate); ρ = 0.45, P<0.0001.
(0.22 MB TIF)
(A) Ratio of number of differentially expressed genes to total number of genes on the microarray (data from Figure 3B). Solid horizontal line shows the average, dotted lines show two standard deviations. (B) Cumulative counts of differentially expressed genes between YrDNA-gross and wild-type Y (at P<0.01) in males (black), females (gray), and females multiplied by a correction (dotted gray) to more easily compare trends (data from Figure 3C).
(0.29 MB TIF)
(A) Observed number of shared differentially expressed genes across pairwise chromosome comparisons. P<0.05 data are shown above the diagonal, P<0.005 are shown below the diagonal, and total number (shared plus unique) of differentially expressed genes (P<0.05/P<0.005) are shown on the diagonal (bold, gray background). (B) Expected numbers of genes shared between induced rDNA deletion Y chromosomes and natural Y chromosomes, calculated from randomized datasets (N for individual pairwise comparisons are on diagonal) at P<0.05 (above diagonal) and P<0.005 (below diagonal).
(0.27 MB TIF)