Abstract
Several subtypes of interneurons in the feedback circuit in stratum oriens of the hippocampus exhibit NMDA receptor-independent long-term potentiation (LTP) at glutamatergic synapses made by local pyramidal neurons. LTP has been reported with both “Hebbian” and “anti-Hebbian” induction protocols, where high-frequency presynaptic stimulation is paired with either postsynaptic depolarization or hyperpolarization. Do these phenomena represent distinct forms of plasticity, dependent on group I metabotropic receptors (mGluRs) and rectifying Ca2+-permeable AMPA receptors, respectively? Blockade of either mGluR1 or mGluR5 prevented anti-Hebbian LTP induction in stratum oriens interneurons in rat hippocampal slices. Exogenous activation of group I mGluRs by the selective agonist (S)-3,5-dihydroxyphenylglycine (DHPG) was unable to induce LTP on its own, and instead depressed excitatory transmission. However, when paired with postsynaptic hyperpolarization, DHPG or the group I metabotropic receptor (mGluR5)-selective agonist (R,S)-2-chloro-5-hydroxyphenylglycine (CHPG) elicited a delayed long-lasting potentiation, which was accompanied by a decrease in paired-pulse facilitation. Anti-Hebbian LTP occluded the effect of DHPG paired with hyperpolarization, implying that the induction cascades triggered by both conjunctions of stimuli converge on common expression mechanisms.
Introduction
Several subtypes of hippocampal interneurons exhibit NMDA receptor-independent long-term potentiation (LTP) at glutamatergic synapses made by local pyramidal neurons. One induction protocol relies on 5–100 Hz presynaptic stimulation paired with postsynaptic hyperpolarization (Lamsa et al., 2007; Oren et al., 2009; Nissen et al., 2010). This phenomenon, which mainly occurs in the feedback inhibitory circuit, has been termed “anti-Hebbian” to distinguish it from associative NMDA receptor-dependent LTP in principal cells, whose induction requirements are reminiscent of Hebb's postulate (Kullmann and Lamsa, 2007). The induction mechanisms of NMDA receptor-independent LTP in the feedback circuit are incompletely understood. It occurs at synapses equipped with strongly rectifying AMPA receptors, and is prevented by selective blockers of AMPA receptors (Oren et al., 2009). Indeed, LTP dependent on Ca2+-permeable AMPA receptors has been reported in several other interneurons (Mahanty and Sah, 1998; Polepalli et al., 2010; Sambandan et al., 2010). However, a “Hebbian” form of LTP has also been reported in stratum oriens interneurons, induced by pairing theta-burst presynaptic stimulation with postsynaptic stimulation (Perez et al., 2001; Pelletier and Lacaille, 2008). This form of LTP requires the mGluR1 subtype of group I mGluRs, because it is prevented by pharmacological blockade (Perez et al., 2001) or genetic ablation (Lapointe et al., 2004) of this receptor. mGluR1-dependent LTP has also been reported at synapses made by mossy fibers on interneurons in stratum lacunosum/moleculare (Galván et al., 2008). Interestingly, at synapses made by pyramidal neurons on fast-spiking interneurons in layer 2/3 of the visual cortex, LTP depends not on mGluR1 but on the other group I mGluR subtype, mGluR5 (Sarihi et al., 2008).
Although many of the studies listed above have examined different synapses and are therefore not directly comparable (see also Cowan et al., 1998; Alle et al., 2001; Laezza and Dingledine, 2004; Pelkey et al., 2005), the reports of Hebbian and anti-Hebbian NMDA receptor-independent LTP in stratum oriens have focused on overlapping, if not congruent, populations of interneurons innervated by local pyramidal neurons (Perez et al., 2001; Lapointe et al., 2004; Lamsa et al., 2007; Oren et al., 2009; Croce et al., 2010; Nissen et al., 2010), which include oriens-lacunosum/moleculare (O-LM) cells (Lacaille et al., 1987; Blasco-Ibáñez and Freund, 1995). Are the LTP dependent on Ca2+-permeable AMPA receptors and the LTP dependent on group I mGluRs two sides of the same coin? Both forms of LTP appear to be expressed presynaptically, as witnessed by changes in failure rates, trial-to-trial variability, paired-pulse facilitation, and sensitivity to use-dependent AMPA receptor blockers (Perez et al., 2001; Lamsa et al., 2007; Croce et al., 2010). Indeed, because LTP has been studied with either whole-cell (Perez et al., 2001; Lapointe et al., 2004; Croce et al., 2010), cell-attached (Croce et al., 2010), or perforated-patch pipettes (Lamsa et al., 2007; Oren et al., 2009; Nissen et al., 2010), subtle differences in recording methods could potentially reconcile the different reports.
Here, we show that mGluR1 and mGluR5 are necessary for induction of anti-Hebbian LTP. Although exogenous activation of group I mGluRs with (S)-3,5-dihydroxyphenylglycine (DHPG) fails to elicit LTP on its own, when paired with postsynaptic hyperpolarization, a slowly evolving but long-lasting potentiation is evoked. This is associated with a decrease in paired-pulse facilitation and is occluded by anti-Hebbian LTP. The results imply that a group I mGluR-dependent cascade contributes, together with Ca2+-permeable AMPA receptors, to the induction of anti-Hebbian LTP.
Materials and Methods
Hippocampal slices.
All procedures followed the Animals (Scientific Procedures) Act, 1986. Transverse 350 μm hippocampal slices were prepared from postnatal day 20–24 male Sprague Dawley rats. Animals were decapitated under terminal pentobarbital anesthesia (140 mg/kg). Slices were cut with a vibratome (VT1000S, Leica) in an ice-cold solution containing (in mm): 70 sucrose, 80 NaCl, 2.5 KCl, 1.25 NaH2PO4, 25 NaHCO3, 7 MgCl2, 0.5 CaCl2, 25 glucose (pH 7.3, 315 mOsm), bubbled with 95% O2/5% CO2. Slices were allowed to recover at room temperature in an interface chamber, in carbogen-bubbled NaCl (119 mm), KCl (2.5 mm), NaHCO3 (26 mm), NaH2PO4 (1 mm), MgCl2 (3 mm), CaCl2 (2 mm), glucose (15 mm) (pH 7.3, 297 mOsm), and then transferred to the recording chamber of an upright microscope (BX50WI, Olympus). The perfusion solution (3 ml/min, 30–32°C) was the same as the storage solution except that CaCl2 was increased to 2.5 mm, and Mg2+ was reduced to 1.3 mm and was supplemented with picrotoxin (100 μm) and 3-[[(3,4-dichlorophenyl)methyl]amino]propyl]diethoxymethyl)phosphinic acid (CGP 52432) (1 μm) to block GABA receptors, and with dl-2-amino-5-phosphonovalerate (dl-APV) (50 μm) to block NMDA receptors. A cut was made between CA3 and CA1.
Electrophysiology and analysis.
Interneurons in stratum oriens of the CA1 subfield with dendrites running parallel to stratum pyramidale were patch-clamped under infrared differential interference contrast microscopy, using a Multiclamp 700B amplifier (Molecular Devices). Records were filtered at 5 kHz, digitized at 10 kHz, and recorded using programs written in Labview (National Instruments). For perforated-patch current-clamp recordings, gramicidin (100 mg · ml−1; Sigma-Aldrich), prepared daily in DMSO, was added (1:1000) to a pipette solution containing (in mm): 130 K-gluconate, 8 NaCl, 20 HEPES, 0.2 EGTA, and 5 QX-314 Br [2-(triethylamino)-N-(2,6-dimethylphenyl)acetamine bromide (pH 7.2, 295 mOsm)]. The pipette tip was filled with gramicidin-free solution. Recordings were started when the series resistance was <150 MΩ, and the experiment was discontinued if depolarizing pulses failed to evoke action potentials. The membrane potential was maintained between −75 and −65 mV with current injections where necessary. For whole-cell current-clamp recordings, the pipette solution contained (in mm) 117.5 K-gluconate, 17.5 KCl, 10 CsOH HEPES, 0.2 EGTA, 8 NaCl, 2 MgATP, 0.3 Na3GTP, 5 QX-314 Br (pH 7.2, 296 mOsm). The pipette resistance was 4–5 MΩ. Neurons were voltage-clamped at −60 mV. Cells were rejected if either the series resistance or the holding current varied by >25%.
Two bipolar stainless steel electrodes were positioned in the alveus/stratum oriens border on either side of the interneuron (distance 100–500 μm), to stimulate axon collaterals of pyramidal neurons. Either single or paired stimuli (50–100 μs duration, 50 ms interpulse interval; Digitimer) were alternately delivered via each electrode, with a 15 s duty cycle. Paired-pulse ratios (PPRs) were calculated from averages of 20 cycles. LTP was induced in one pathway by stimulating at 100 Hz for 1 s, twice with a 20 s interval. The postsynaptic cell was voltage-clamped between −90 and −100 mV during each train to prevent it from spiking.
To control for nonspecific drift in recording conditions, we expressed the pathway-specific potentiation as 100 × (EPSPtest/EPSPcontrol − 1)%, where the EPSP initial slopes in test and control pathways (EPSPtest and EPSPcontrol, respectively) were first normalized by their average values before pairing. LTP was estimated from a 5 min period between 20 and 25 min after induction. Data were analyzed with Student's paired t test, and significance was taken as p < 0.05.
Drugs.
The following drugs were obtained from Tocris Bioscience: CGP 52432, dl-APV, DHPG, LY 367385 [(S)-(+)-α-amino-4-carboxy-2-methylbenzeneacetic acid], and MPEP [2-methyl-6-(phenylethynyl)pyridine hydrochloride]. Picrotoxin was obtained from Sigma. QX314 Br and CHPG were obtained from Ascent Scientific.
Results
Both mGluR1 and mGluR5 contribute to anti-Hebbian LTP induction
We focused on interneurons with dendrites oriented parallel to stratum pyramidale, most of which correspond to O-LM cells. These interneurons abundantly express an isoform of mGluR1 (van Hooft et al., 2000; Ferraguti et al., 2004), and, together with parvalbumin-positive basket and axoaxonic cells, exhibit anti-Hebbian LTP (Lamsa et al., 2007; Oren et al., 2009; Nissen et al., 2010). Because this form of LTP is rapidly disrupted with whole-cell recordings (Lamsa et al., 2007), we used the perforated-patch method throughout this study unless otherwise indicated.
Pairing high-frequency stimulation of one pathway (100 Hz, 1 s, twice) with postsynaptic hyperpolarization elicited 54 ± 10% potentiation, measured at 20 min, in 40 interneurons (Fig. 1A). When applied in the continued presence of the mGluR1 blocker LY 367385 (100 μm), the same pairing protocol only induced post-tetanic potentiation, which rapidly decayed to baseline. However, the paired pathway subsequently diverged from the control pathway, such that, after 20 min, the EPSP slope reached a maximum 35 ± 13% increase relative to the control pathway (n = 10, p = 0.02; Fig. 1B). We also interleaved experiments in which the pairing was applied in the presence of the mGluR5 blocker MPEP (25 μm). This also failed to elicit LTP: the mean EPSP slope after 20 min was 88 ± 7% of baseline (n = 9), and was not significantly different from the control pathway (p = 0.33; Fig. 1C).
Figure 1.
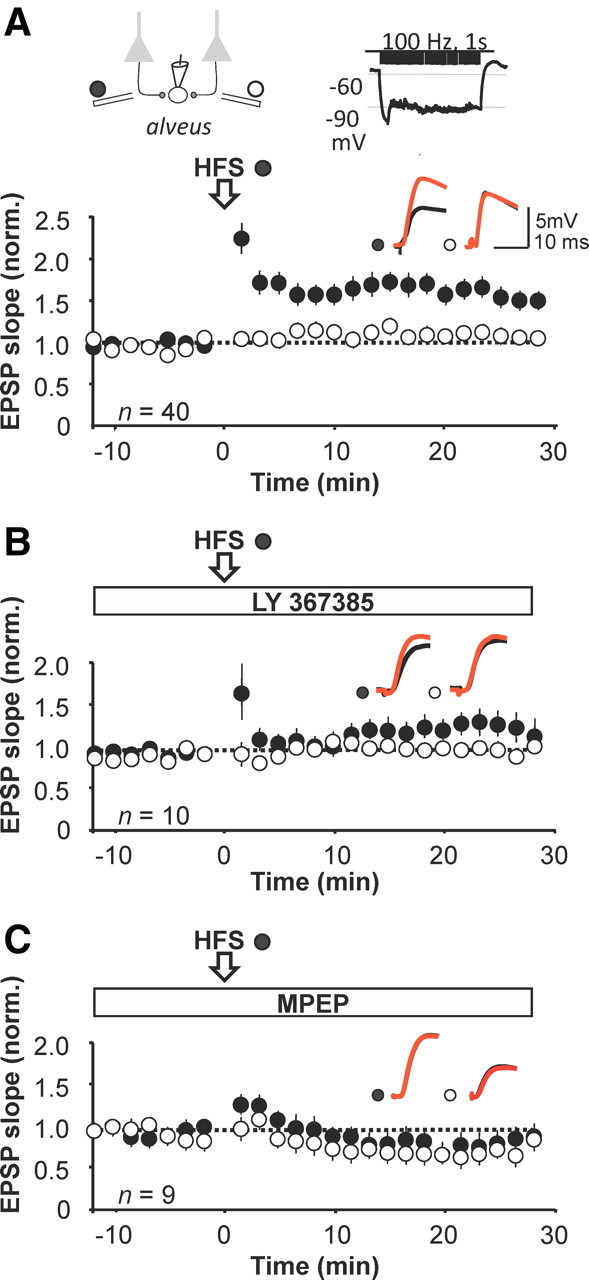
Blockade of either mGluR1 or mGluR5 prevents anti-Hebbian LTP induction. A, Baseline-normalized EPSP slope, showing the effect of pairing presynaptic high-frequency stimulation (HFS) of one pathway (filled symbols) with postsynaptic hyperpolarization (open arrow). Open symbols, Control pathway. Error bars show SEM. Top, Schematic showing the arrangement of stimulating and recording electrodes and the LTP induction protocol. Inset, Representative sample traces from a single neuron before (black) and after (red) pairing in the two pathways. B, LTP protocol repeated during perfusion of the selective mGluR1 blocker LY 367385 (100 μm). C, LTP protocol repeated during perfusion of the mGluR5 blocker MPEP (25 μm).
Although these results suggest that both mGluR1 and mGluR5 are involved in anti-Hebbian LTP induction, a potential pitfall is that it is not possible to know whether LTP would have been induced without the blockers. We took advantage of the finding that sequentially pairing two homologous pathways results in the same pattern of plasticity at each population of synapses (Lamsa et al., 2005, 2007). We first asked whether the pathway-specific potentiation was sustained after pairing in the absence of mGluR blockers. In 10 interneurons showing LTP, we monitored the test and control pathways for up to 60 min after pairing. The average potentiation thus measured in the test pathway was 60 ± 14%, 40 min post-pairing (n = 5, p < 0.001; Fig. 2A). We next verified that sequential pairing of two pathways elicited pathway-specific LTP. In 7 of 7 cases in which the first pairing elicited LTP, pairing the second pathway also resulted in a pathway-specific potentiation, albeit with a smaller magnitude (measured 10 min after pairing, first pathway: 54 ± 21%, p < 0.01; second pathway: 28 ± 9%, p < 0.01; Fig. 2B). (Attention was restricted to the first 10 min after pairing, because it proved difficult to maintain a stable recording for over an hour.)
Figure 2.
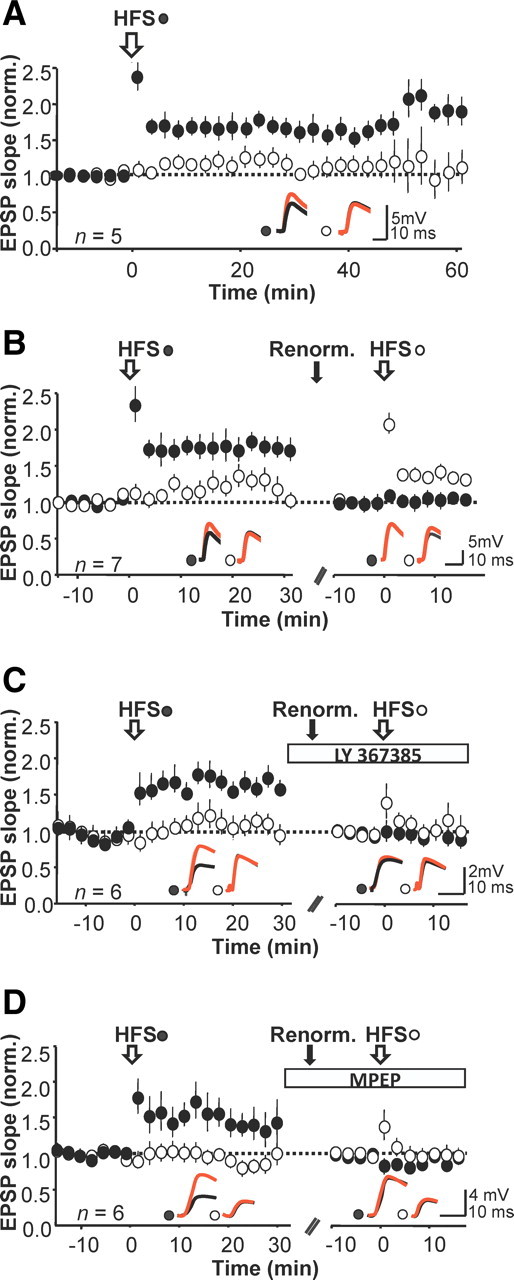
Consecutive pairing of two pathways reveals roles of mGluR1 and mGluR5. A, Anti-Hebbian LTP persists for at least 60 min. EPSP slope (mean ± SEM) normalized to average baseline amplitude before induction of LTP (n = 5). Insets, Representative sample traces from a single neuron before (black) and after (red) pairing. B, Potentiation could be induced with an anti-Hebbian protocol in the second pathway. LTP was first induced in one pathway (filled symbols), and the same protocol was subsequently applied to the other pathway (open symbols). The EPSP slopes were renormalized before the second pairing (n = 7). C, Blocking mGluR1 with LY 367385 prevented potentiation when washed in before applying an anti-Hebbian protocol to the second pathway. D, Data plotted in the same way in a separate set of experiments in which mGluR5 receptors were blocked with MPEP before the second pairing.
We applied the sequential pairing protocol with either LY 367385 or MPEP applied after LTP was elicited in the first pathway. Pairing the second pathway in the presence of LY 367385 failed to evoke a significant potentiation (relative potentiation 10 ± 10%, p = 0.14, n = 6; Fig. 2C). We repeated the experiment with bath application of MPEP after the first pairing. The pairing protocol delivered to the second pathway again failed to elicit a significant potentiation (12 ± 6%, p = 0.09, n = 6; Fig. 2D). These results confirm that preventing either mGluR1 or mGluR5 receptor activation blocks anti-Hebbian LTP induction, at least over the first 10 min.
Biphasic modulation of EPSPs by group I mGluRs
Is group I mGluR activation sufficient to induce LTP on its own? We applied the group I mGluR agonist DHPG and monitored the EPSP initial slope in perforated-patch mode. Direct current injection was used to keep the membrane potential within 5 mV of baseline. DHPG (5 μm, applied for 10 min) reversibly depressed EPSPs to 77 ± 6% of baseline (n = 18, p < 0.05; Fig. 3A). The depression was accompanied by a decrease in 1/CV2 and a 27 ± 6% increase in PPR (p = 0.001) (data not shown). Higher concentrations of DHPG led to larger and more prolonged depression when recording either in perforated-patch or in whole-cell mode (Le Duigou et al., 2011).
Figure 3.
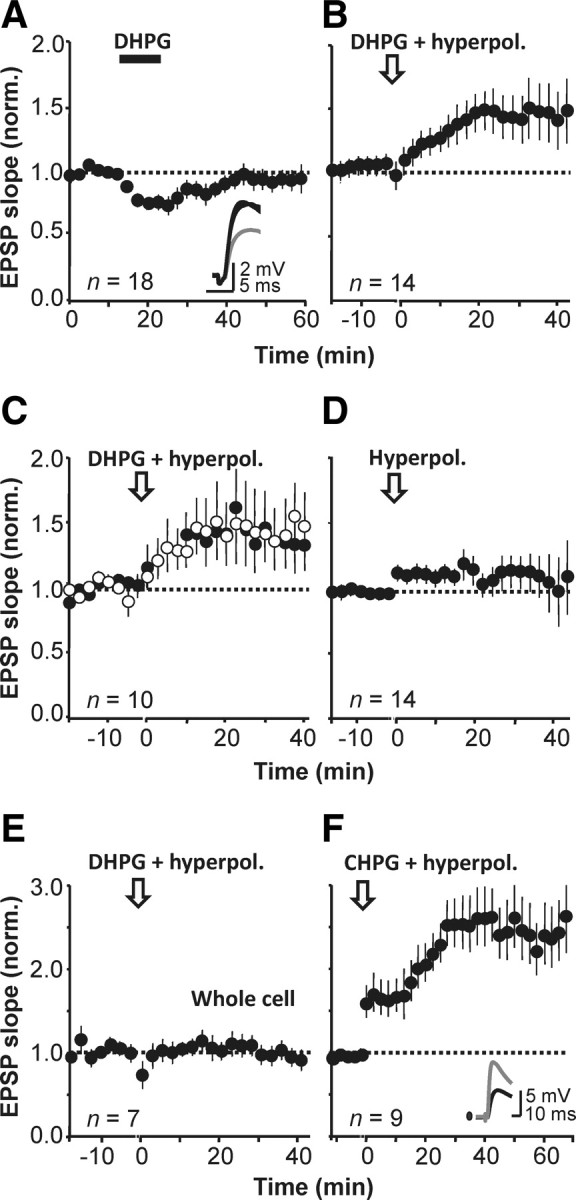
Bidirectional modulation of EPSPs by group I mGluRs. A, The group I mGluR agonist DHPG (5 μm) reversibly depressed monosynaptic EPSPs in CA1 stratum oriens interneurons. Inset, Representative sample traces before, during (gray), and after DHPG application (averages of 20 sweeps). B, DHPG evoked a delayed potentiation when paired with hyperpolarization. C, Interrupting stimulation of one pathway (open symbols) did not prevent the delayed potentiation induced by DHPG application together with hyperpolarization. D, Hyperpolarization alone had no long-lasting effect on transmission. E, DHPG paired with hyperpolarization had no effect on transmission when recording in whole-cell mode. F, The selective mGluR5 agonist CHPG (500 μm) induced a potentiation when paired with hyperpolarization (perforated patch). Inset, Representative traces before (black) and after (gray) CHPG .
We asked whether the DHPG-evoked depression could be converted to a potentiation by pairing application of the mGluR agonist with postsynaptic hyperpolarization to between −90 mV and −100 mV (similar to the LTP induction protocol) for 10 min. Pairing DHPG with hyperpolarization led to a slow increase in EPSP slope after DHPG washout in 10 of 14 interneurons (Fig. 3B). On average the EPSP slope increased to 155 ± 19% of baseline (n = 14, p < 0.01). The delayed potentiation did not require synaptic activity during the DHPG application, because it was no smaller in a pathway whose stimulation was interrupted and then resumed after washout (stimulated pathway: 41 ± 19% increase, p < 0.01 relative to baseline; unstimulated pathway: 39 ± 22% increase, p < 0.01; n = 10; between-pathway comparison, NS; Fig. 3C). However, we cannot rule out the possibility that spontaneous glutamate release occurred during the application of DHPG and hyperpolarization.
In contrast, hyperpolarization delivered on its own without DHPG application was ineffective (EPSP slope 113 ± 7% of baseline, p = 0.19, n = 14; Fig. 3D). DHPG application also failed to evoke a robust potentiation when the recording was performed in whole-cell mode (97 ± 8%, n = 7; Fig. 3E) even if paired with hyperpolarization, consistent with previous evidence that LTP induction in interneurons is highly sensitive to dialysis of the cytoplasm (Lamsa et al., 2005, 2007).
Exogenous activation of group I mGluRs with hyperpolarization occludes LTP
Although, as pointed out above, activation of Ca2+-permeable AMPA receptors was not completely prevented during DHPG application, an alternative explanation for the interaction of group I mGluR activation with hyperpolarization is that this triggers Ca2+ influx via voltage-gated Ca2+ or TRP channels, which are differentially coupled to mGluR1 and mGluR5 (Topolnik et al., 2006, 2009). We asked whether selective activation of mGluR5 could trigger the same potentiation. The specific mGluR5 agonist CHPG (500 μm) induced a delayed potentiation of 151 ± 7% when paired with postsynaptic hyperpolarization (p ≪ 0.01, n = 9; Fig. 3F), which was again associated with a decrease in PPR (data not shown).
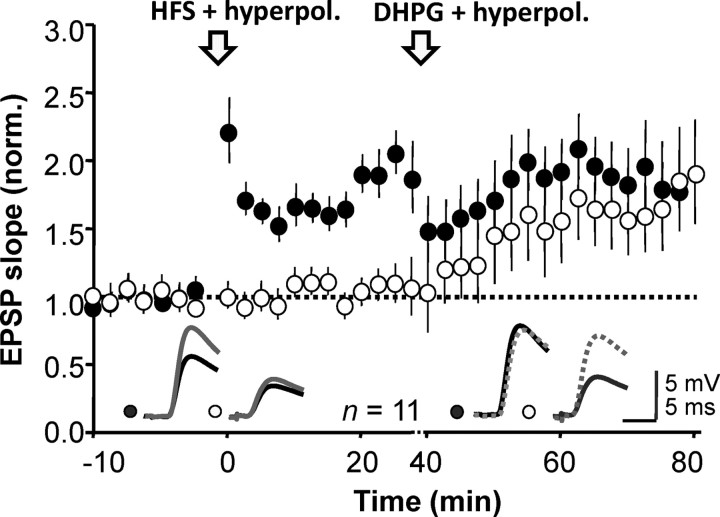
Finally, we looked for occlusion between the potentiation evoked by hyperpolarization with mGluR activation and anti-Hebbian LTP. We induced LTP by pairing high-frequency stimulation with hyperpolarization in one pathway (average potentiation 74 ± 3%, n = 11, p ≪ 0.01; Fig. 4) and then applied DHPG (5 μm) together with hyperpolarization. DHPG with hyperpolarization induced a 70 ± 3% increase in the naive pathway (p ≪ 0.01) but no change in the previously potentiated pathway. Both forms of potentiation were associated with similar changes in PPR (data not shown). Occlusion of the DHPG effect by prior induction of anti-Hebbian LTP thus argues that they converge on a common expression mechanism.
Figure 4.
Anti-Hebbian LTP occludes the potentiation induced by DHPG paired with hyperpolarization. Anti-Hebbian LTP was induced in one pathway (filled symbols) and DHPG perfusion was subsequently paired with a hyperpolarization, leading to delayed potentiation that was restricted to the control pathway (open symbols). Inset, Representative sample traces before (black) and after (gray) LTP induction, and before and after DHPG application (dashed).
Discussion
The present results argue that both mGluR1 and mGluR5 contribute to the induction of anti-Hebbian LTP at excitatory synapses on interneurons in stratum oriens. These receptors are abundantly expressed in many neurons, and tend to be located in the postsynaptic membrane, in a perisynaptic annulus surrounding glutamatergic synapses (Lujan et al., 1996). They are also enriched in several types of interneurons (van Hooft et al., 2000; Ferraguti et al., 2004). Although they are implicated in long-term depression at synapses made by Schaffer collaterals on several targets (Anwyl, 2009) including interneurons in stratum radiatum (Gibson et al., 2008), several previous studies show that they contribute to LTP in interneurons in stratum oriens (Perez et al., 2001; Lapointe et al., 2004). Demonstrating that an induction cascade is sufficient to lead to any form of long-term synaptic plasticity is notoriously difficult (Sanes and Lichtman, 1999). Nevertheless, the involvement of group I mGluRs in LTP is reinforced by the finding that exogenous application can lead to a delayed potentiation (Le Vasseur et al., 2008). In that study, depolarization was necessary to uncover the potentiation. The present study differs in that perforated-patch recordings were used to minimize cytoplasmic disruption. With this recording method, LTP occurs at synapses with strongly rectifying (and by implication, Ca2+-permeable) AMPA receptors (Lamsa et al., 2007; Nissen et al., 2010), and reversible blockade of these receptors prevents its induction (Oren et al., 2009). This evidence taken in isolation implies an instructive role for Ca2+ influx via AMPA receptors (Mahanty and Sah, 1998). However, we now show that exogenous activation of group I mGluRs with DHPG can itself induce a delayed potentiation, but only if paired with hyperpolarization. Importantly, this is occluded by prior induction of anti-Hebbian LTP, implying that the two phenomena indeed converge on a common cascade.
mGluR1 and mGluR5 exhibit distinct interactions with intracellular stores and transient receptor potential channels (Topolnik et al., 2006). Although selective activation of mGluR5 by pairing CHPG application with hyperpolarization was sufficient to evoke a long-lasting potentiation, it is difficult to argue that this receptor is preferentially linked to LTP. Indeed, we found that blockade of either mGluR1 or mGluR5 was sufficient to prevent anti-Hebbian LTP induction. A tentative model that reconciles these findings is that all three essential components (Ca2+-permeable AMPA receptors, mGluR1, and mGluR5) contribute cumulatively to generate a local postsynaptic signal required to induce LTP. This signal may be a local increase in Ca2+, and with exogenous activation of group I mGluRs may even obviate the requirement for AMPA receptors, explaining why DHPG application with depolarization yielded a potentiation that did not differ between the stimulated and unstimulated pathways. However, when synaptic stimulation is paired with hyperpolarization, removal of any one of these components may render this signal insufficient for LTP induction and may instead uncover a depression of transmission. Indeed, in a previous study using an anti-Hebbian induction protocol, we observed a small but significant depression of the conditioned pathway when AMPA receptors were temporarily blocked (Oren et al., 2009).
Footnotes
This work was supported by the Medical Research Council, the Wellcome Trust, and the European Research Council. We are grateful to K. Lamsa, I. Oren, and E. Nicholson for helpful comments.
References
- Alle H, Jonas P, Geiger JR. PTP and LTP at a hippocampal mossy fiber-interneuron synapse. Proc Natl Acad Sci U S A. 2001;98:14708–14713. doi: 10.1073/pnas.251610898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anwyl R. Metabotropic glutamate receptor-dependent long-term potentiation. Neuropharmacology. 2009;56:735–740. doi: 10.1016/j.neuropharm.2009.01.002. [DOI] [PubMed] [Google Scholar]
- Blasco-Ibáñez JM, Freund TF. Synaptic input of horizontal interneurons in stratum oriens of the hippocampal CA1 subfield: structural basis of feed-back activation. Eur J Neurosci. 1995;7:2170–2180. doi: 10.1111/j.1460-9568.1995.tb00638.x. [DOI] [PubMed] [Google Scholar]
- Cowan AI, Stricker C, Reece LJ, Redman SJ. Long-term plasticity at excitatory synapses on aspinous interneurons in area CA1 lacks synaptic specificity. J Neurophysiol. 1998;79:13–20. doi: 10.1152/jn.1998.79.1.13. [DOI] [PubMed] [Google Scholar]
- Croce A, Pelletier JG, Tartas M, Lacaille JC. Afferent-specific properties of interneuron synapses underlie selective long-term regulation of feedback inhibitory circuits in CA1 hippocampus. J Physiol. 2010;588:2091–2107. doi: 10.1113/jphysiol.2010.189316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferraguti F, Cobden P, Pollard M, Cope D, Shigemoto R, Watanabe M, Somogyi P. Immunolocalization of metabotropic glutamate receptor 1alpha (mGluR1alpha) in distinct classes of interneuron in the CA1 region of the rat hippocampus. Hippocampus. 2004;14:193–215. doi: 10.1002/hipo.10163. [DOI] [PubMed] [Google Scholar]
- Galván EJ, Calixto E, Barrionuevo G. Bidirectional Hebbian plasticity at hippocampal mossy fiber synapses on CA3 interneurons. J Neurosci. 2008;28:14042–14055. doi: 10.1523/JNEUROSCI.4848-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson HE, Edwards JG, Page RS, Van Hook MJ, Kauer JA. TRPV1 channels mediate long-term depression at synapses on hippocampal interneurons. Neuron. 2008;57:746–759. doi: 10.1016/j.neuron.2007.12.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kullmann DM, Lamsa KP. Long-term synaptic plasticity in hippocampal interneurons. Nat Rev Neurosci. 2007;8:687–699. doi: 10.1038/nrn2207. [DOI] [PubMed] [Google Scholar]
- Lacaille JC, Mueller AL, Kunkel DD, Schwartzkroin PA. Local circuit interactions between oriens/alveus interneurons and CA1 pyramidal cells in hippocampal slices: electrophysiology and morphology. J Neurosci. 1987;7:1979–1993. doi: 10.1523/JNEUROSCI.07-07-01979.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laezza F, Dingledine R. Voltage-controlled plasticity at GluR2-deficient synapses onto hippocampal interneurons. J Neurophysiol. 2004;92:3575–3581. doi: 10.1152/jn.00425.2004. [DOI] [PubMed] [Google Scholar]
- Lamsa K, Heeroma JH, Kullmann DM. Hebbian LTP in feed-forward inhibitory interneurons and the temporal fidelity of input discrimination. Nat Neurosci. 2005;8:916–924. doi: 10.1038/nn1486. [DOI] [PubMed] [Google Scholar]
- Lamsa KP, Heeroma JH, Somogyi P, Rusakov DA, Kullmann DM. Anti-Hebbian long-term potentiation in the hippocampal feedback inhibitory circuit. Science. 2007;315:1262–1266. doi: 10.1126/science.1137450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lapointe V, Morin F, Ratté S, Croce A, Conquet F, Lacaille JC. Synapse-specific mGluR1-dependent long-term potentiation in interneurones regulates mouse hippocampal inhibition. J Physiol. 2004;555:125–135. doi: 10.1113/jphysiol.2003.053603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Duigou C, Holden T, Kullmann DM. Short- and long-term depression at glutamatergic synapses on hippocampal interneurons by group I mGluR activation. Neuropharmacology. 2011;60:748–756. doi: 10.1016/j.neuropharm.2010.12.015. [DOI] [PubMed] [Google Scholar]
- Le Vasseur M, Ran I, Lacaille JC. Selective induction of metabotropic glutamate receptor 1- and metabotropic glutamate receptor 5-dependent chemical long-term potentiation at oriens/alveus interneuron synapses of mouse hippocampus. Neuroscience. 2008;151:28–42. doi: 10.1016/j.neuroscience.2007.09.071. [DOI] [PubMed] [Google Scholar]
- Lujan R, Nusser Z, Roberts JD, Shigemoto R, Somogyi P. Perisynaptic location of metabotropic glutamate receptors mGluR1 and mGluR5 on dendrites and dendritic spines in the rat hippocampus. Eur. J Neurosci. 1996;8:1488–1500. doi: 10.1111/j.1460-9568.1996.tb01611.x. [DOI] [PubMed] [Google Scholar]
- Mahanty NK, Sah P. Calcium-permeable AMPA receptors mediate long-term potentiation in interneurons in the amygdala. Nature. 1998;394:683–687. doi: 10.1038/29312. [DOI] [PubMed] [Google Scholar]
- Nissen W, Szabo A, Somogyi J, Somogyi P, Lamsa KP. Cell type-specific long-term plasticity at glutamatergic synapses onto hippocampal interneurons expressing either parvalbumin or CB1 cannabinoid receptor. J Neurosci. 2010;30:1337–1347. doi: 10.1523/JNEUROSCI.3481-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oren I, Nissen W, Kullmann DM, Somogyi P, Lamsa KP. Role of ionotropic glutamate receptors in long-term potentiation in rat hippocampal CA1 oriens-lacunosum moleculare interneurons. J Neurosci. 2009;29:939–950. doi: 10.1523/JNEUROSCI.3251-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelkey KA, Lavezzari G, Racca C, Roche KW, McBain CJ. mGluR7 is a metaplastic switch controlling bidirectional plasticity of feedforward inhibition. Neuron. 2005;46:89–102. doi: 10.1016/j.neuron.2005.02.011. [DOI] [PubMed] [Google Scholar]
- Pelletier JG, Lacaille JC. Long-term synaptic plasticity in hippocampal feedback inhibitory networks. Prog Brain Res. 2008;169:241–250. doi: 10.1016/S0079-6123(07)00014-3. [DOI] [PubMed] [Google Scholar]
- Perez Y, Morin F, Lacaille JC. A hebbian form of long-term potentiation dependent on mGluR1a in hippocampal inhibitory interneurons. Proc Natl Acad Sci U S A. 2001;98:9401–9406. doi: 10.1073/pnas.161493498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polepalli JS, Sullivan RK, Yanagawa Y, Sah P. A specific class of interneuron mediates inhibitory plasticity in the lateral amygdala. J Neurosci. 2010;30:14619–14629. doi: 10.1523/JNEUROSCI.3252-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sambandan S, Sauer JF, Vida I, Bartos M. Associative plasticity at excitatory synapses facilitates recruitment of fast-spiking interneurons in the dentate gyrus. J Neurosci. 2010;30:11826–11837. doi: 10.1523/JNEUROSCI.2012-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanes JR, Lichtman JW. Can molecules explain long-term potentiation? Nat Neurosci. 1999;2:597–604. doi: 10.1038/10154. [DOI] [PubMed] [Google Scholar]
- Sarihi A, Jiang B, Komaki A, Sohya K, Yanagawa Y, Tsumoto T. Metabotropic glutamate receptor type 5-dependent long-term potentiation of excitatory synapses on fast-spiking GABAergic neurons in mouse visual cortex. J Neurosci. 2008;28:1224–1235. doi: 10.1523/JNEUROSCI.4928-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Topolnik L, Azzi M, Morin F, Kougioumoutzakis A, Lacaille JC. mGluR1/5 subtype-specific calcium signalling and induction of long-term potentiation in rat hippocampal oriens/alveus interneurones. J Physiol. 2006;575:115–131. doi: 10.1113/jphysiol.2006.112896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Topolnik L, Chamberland S, Pelletier JG, Ran I, Lacaille JC. Activity-dependent compartmentalized regulation of dendritic Ca2+ signaling in hippocampal interneurons. J Neurosci. 2009;29:4658–4663. doi: 10.1523/JNEUROSCI.0493-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Hooft JA, Giuffrida R, Blatow M, Monyer H. Differential expression of group I metabotropic glutamate receptors in functionally distinct hippocampal interneurons. J Neurosci. 2000;20:3544–3551. doi: 10.1523/JNEUROSCI.20-10-03544.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]