Abstract
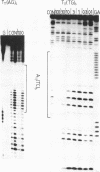
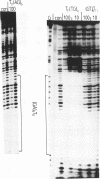
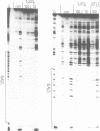
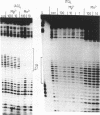
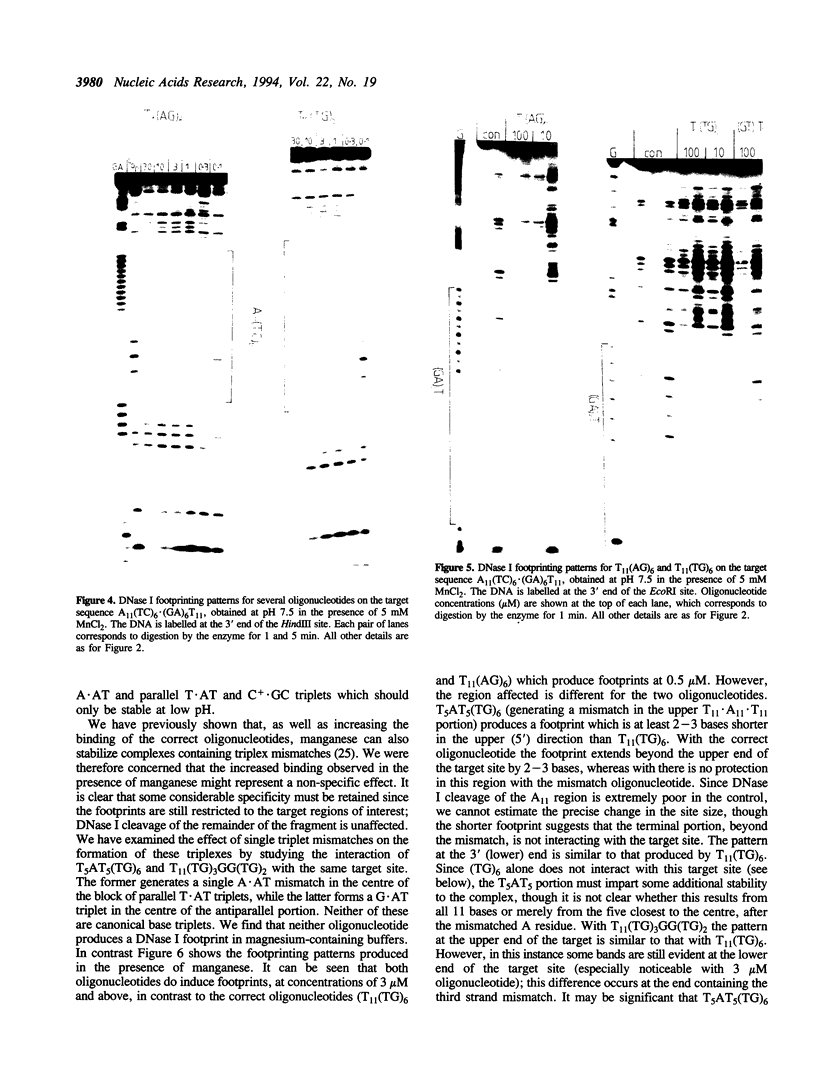
We have examined the formation of alternate strand triple-helices at the target sequence A11(TC)6.(GA)6T11 using the oligonucleotides T11(AG)6 and T11(TG)6, by DNase I footprinting. These third strands were designed so as to form parallel T.AT triplets together with antiparallel G.GC and A.AT or T.AT triplets. We find that, although both oligonucleotides yield clear footprints at similar concentrations (0.3 microM) in the presence of manganese, only T11(TG)6 forms a stable complex in magnesium-containing buffers, albeit at a higher concentration (10-30 microM). Examination of the interaction of (AG)6 and (TG)6 with half the target site confirmed that the complex containing A.AT triplets was only stable in the presence of manganese. In contrast no binding of (TG)6 was detected in the presence of either metal ion, suggesting that the reverse-Hoogsteen T.AT triplet is less stable that G.GC. We suggest that, within the context of G.GC triplets, the rank order of antiparallel triplet stability is A.AT (Mn2+) > T.AT (Mn2+) > T.AT (Mg2+) > A.AT (Mg2+). Third strands containing a single base substitution in the centre of either the parallel or antiparallel portion showed a (10-fold) weaker interaction in manganese-containing buffers, and no interaction in the presence of magnesium.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arnott S., Selsing E. Structures for the polynucleotide complexes poly(dA) with poly (dT) and poly(dT) with poly(dA) with poly (dT). J Mol Biol. 1974 Sep 15;88(2):509–521. doi: 10.1016/0022-2836(74)90498-7. [DOI] [PubMed] [Google Scholar]
- Beal P. A., Dervan P. B. Second structural motif for recognition of DNA by oligonucleotide-directed triple-helix formation. Science. 1991 Mar 15;251(4999):1360–1363. doi: 10.1126/science.2003222. [DOI] [PubMed] [Google Scholar]
- Beal P. A., Dervan P. B. The influence of single base triplet changes on the stability of a pur.pur.pyr triple helix determined by affinity cleaving. Nucleic Acids Res. 1992 Jun 11;20(11):2773–2776. doi: 10.1093/nar/20.11.2773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broitman S. L., Im D. D., Fresco J. R. Formation of the triple-stranded polynucleotide helix, poly(A.A.U). Proc Natl Acad Sci U S A. 1987 Aug;84(15):5120–5124. doi: 10.1073/pnas.84.15.5120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chandler S. P., Fox K. R. Triple helix formation at A8XA8.T8YT8. FEBS Lett. 1993 Oct 11;332(1-2):189–192. doi: 10.1016/0014-5793(93)80510-2. [DOI] [PubMed] [Google Scholar]
- Chen F. M. Intramolecular triplex formation of the purine.purine.pyrimidine type. Biochemistry. 1991 May 7;30(18):4472–4479. doi: 10.1021/bi00232a014. [DOI] [PubMed] [Google Scholar]
- Chubb J. M., Hogan M. E. Human therapeutics based on triple helix technology. Trends Biotechnol. 1992 Apr;10(4):132–136. doi: 10.1016/0167-7799(92)90195-2. [DOI] [PubMed] [Google Scholar]
- Durland R. H., Kessler D. J., Gunnell S., Duvic M., Pettitt B. M., Hogan M. E. Binding of triple helix forming oligonucleotides to sites in gene promoters. Biochemistry. 1991 Sep 24;30(38):9246–9255. doi: 10.1021/bi00102a017. [DOI] [PubMed] [Google Scholar]
- FELSENFELD G., RICH A. Studies on the formation of two- and three-stranded polyribonucleotides. Biochim Biophys Acta. 1957 Dec;26(3):457–468. doi: 10.1016/0006-3002(57)90091-4. [DOI] [PubMed] [Google Scholar]
- Fox K. R. Formation of DNA triple helices incorporating blocks of G.GC and T.AT triplets using short acridine-linked oligonucleotides. Nucleic Acids Res. 1994 Jun 11;22(11):2016–2021. doi: 10.1093/nar/22.11.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giovannangéli C., Rougée M., Garestier T., Thuong N. T., Hélène C. Triple-helix formation by oligonucleotides containing the three bases thymine, cytosine, and guanine. Proc Natl Acad Sci U S A. 1992 Sep 15;89(18):8631–8635. doi: 10.1073/pnas.89.18.8631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffin L. C., Dervan P. B. Recognition of thymine adenine.base pairs by guanine in a pyrimidine triple helix motif. Science. 1989 Sep 1;245(4921):967–971. doi: 10.1126/science.2549639. [DOI] [PubMed] [Google Scholar]
- Hélène C. Sequence-selective recognition and cleavage of double-helical DNA. Curr Opin Biotechnol. 1993 Feb;4(1):29–36. doi: 10.1016/0958-1669(93)90028-u. [DOI] [PubMed] [Google Scholar]
- Jayasena S. D., Johnston B. H. Intramolecular triple-helix formation at (PunPyn).(PunPyn) tracts: recognition of alternate strands via Pu.PuPy and Py.PuPy base triplets. Biochemistry. 1992 Jan 21;31(2):320–327. doi: 10.1021/bi00117a002. [DOI] [PubMed] [Google Scholar]
- Jayasena S. D., Johnston B. H. Oligonucleotide-directed triple helix formation at adjacent oligopurine and oligopyrimidine DNA tracts by alternate strand recognition. Nucleic Acids Res. 1992 Oct 25;20(20):5279–5288. doi: 10.1093/nar/20.20.5279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones G. B., Davey C. L., Jenkins T. C., Kamal A., Kneale G. G., Neidle S., Webster G. D., Thurston D. E. The non-covalent interaction of pyrrolo[2, 1-c] [1, 4]benzodiazepine-5, 11-diones with DNA. Anticancer Drug Des. 1990 Aug;5(3):249–264. [PubMed] [Google Scholar]
- Le Doan T., Perrouault L., Praseuth D., Habhoub N., Decout J. L., Thuong N. T., Lhomme J., Hélène C. Sequence-specific recognition, photocrosslinking and cleavage of the DNA double helix by an oligo-[alpha]-thymidylate covalently linked to an azidoproflavine derivative. Nucleic Acids Res. 1987 Oct 12;15(19):7749–7760. doi: 10.1093/nar/15.19.7749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malkov V. A., Voloshin O. N., Soyfer V. N., Frank-Kamenetskii M. D. Cation and sequence effects on stability of intermolecular pyrimidine-purine-purine triplex. Nucleic Acids Res. 1993 Feb 11;21(3):585–591. doi: 10.1093/nar/21.3.585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moffat A. S. Triplex DNA finally comes of age. Science. 1991 Jun 7;252(5011):1374–1375. doi: 10.1126/science.2047850. [DOI] [PubMed] [Google Scholar]
- Moser H. E., Dervan P. B. Sequence-specific cleavage of double helical DNA by triple helix formation. Science. 1987 Oct 30;238(4827):645–650. doi: 10.1126/science.3118463. [DOI] [PubMed] [Google Scholar]
- Pilch D. S., Levenson C., Shafer R. H. Structure, stability, and thermodynamics of a short intermolecular purine-purine-pyrimidine triple helix. Biochemistry. 1991 Jun 25;30(25):6081–6088. doi: 10.1021/bi00239a001. [DOI] [PubMed] [Google Scholar]
- Radhakrishnan I., Patel D. J. Solution structure of a purine.purine.pyrimidine DNA triplex containing G.GC and T.AT triples. Structure. 1993 Oct 15;1(2):135–152. doi: 10.1016/0969-2126(93)90028-f. [DOI] [PubMed] [Google Scholar]
- Radhakrishnan I., Patel D. J. Solution structure of a pyrimidine.purine.pyrimidine DNA triplex containing T.AT, C+.GC and G.TA triples. Structure. 1994 Jan 15;2(1):17–32. doi: 10.1016/s0969-2126(00)00005-8. [DOI] [PubMed] [Google Scholar]
- Radhakrishnan I., de los Santos C., Patel D. J. Nuclear magnetic resonance structural studies of A.AT base triple alignments in intramolecular purine.purine.pyrimidine DNA triplexes in solution. J Mol Biol. 1993 Nov 5;234(1):188–197. doi: 10.1006/jmbi.1993.1573. [DOI] [PubMed] [Google Scholar]
- Wang E., Malek S., Feigon J. Structure of a G.T.A triplet in an intramolecular DNA triplex. Biochemistry. 1992 May 26;31(20):4838–4846. doi: 10.1021/bi00135a015. [DOI] [PubMed] [Google Scholar]
- Washbrook E., Fox K. R. Alternate-strand DNA triple-helix formation using short acridine-linked oligonucleotides. Biochem J. 1994 Jul 15;301(Pt 2):569–575. doi: 10.1042/bj3010569. [DOI] [PMC free article] [PubMed] [Google Scholar]