Abstract
Purpose of review
Over half a million Americans die each year from coronary heart disease (CHD), 26 million suffer from chronic kidney disease (CKD), and a large proportion have periodontal disease (PD), a chronic infection of the tissues surrounding teeth. Chronic inflammation contributes to CHD and CKD occurrence and progression, and PD contributes to the cumulated chronic systemic inflammatory burden. This review examines recent evidence regarding the role of PD in CHD and CKD.
Recent findings
Periodontal pathogens cause both local infection and bacteremia, eliciting local and systemic inflammatory responses. PD is associated with the systemic inflammatory reactant CRP, a major risk factor for both CHD and CKD. Non-surgical PD treatment is shown to improve periodontal health, endothelial function and levels of CRP and other inflammatory markers. Evidence for the association of PD with CKD consists of a small body of literature represented mainly by cross-sectional studies. No definitive randomized-controlled trials exist with either CHD or CKD as primary endpoints.
Summary
Recent evidence links PD with CHD and CKD. Adding oral health self-care and referral for professional periodontal assessment and therapy to the repertoire of medical care recommendations is prudent to improve patients’ oral health and possibly reduce CHD and CKD risk.
Keywords: chronic kidney disease, coronary heart disease, inflammation, periodontal disease
Introduction
Coronary heart disease (CHD) and chronic kidney disease (CKD) are important public health concerns. CHD is the leading cause of death [1], and 26 million Americans with CKD are more likely to progress to CHD and premature death due to CHD than progress to kidney failure [2]. Multiple studies support the role of PD in contributing to a chronic systemic inflammatory burden in both CHD and CKD. A consensus report simultaneously published in the Journal of Periodontology and American Journal of Cardiology recommended periodontists inform patients with periodontal disease (PD) they may have increased risk for cardiovascular disease and that cardiologists recommend patients have oral health checkups [3**]. Chronic inflammation also contributes to the pathogenesis of hypertension and diabetes, both major risk factors for both CHD and CKD [4]. Health care personnel advise patients to reduce CHD and CKD risk through smoking cessation, diet modification, exercise, and antihypertensive drug therapy, as well as good glycemic control for individuals with diabetes. This review examines the evidence for the role of PD in CHD and CKD to assist health care providers as they consider adding home self-care and referrals for periodontal assessment and therapy to their repertoire of recommendations to improve patient’s oral health and thereby possibly reduce risks for CHD and CKD.
Definitions
First, we will define the key terms central for this review.
Periodontal Disease
The term “periodontal diseases” has been used for years to include all diseases of the soft and hard tissue surrounding the teeth (i.e. the periodontium), including gingivitis, which is a reversible infection of the soft tissue without permanent loss of periodontal support. Chronic periodontitis, henceforth referred to as PD, is a chronic bacterial infection that causes a persistent host inflammatory response with destruction of the bone and soft tissue surrounding teeth, leading to tooth mobility and ultimately to loss of teeth. In adults, PD is a major cause of tooth loss, hence edentulism can be regarded as a marker of past PD [5]. Observational and interventional studies use various combinations of clinical and/or radiographic measures of extent and/or severity of gingival bleeding, probing pocket depth, attachment loss, and radiographic bone loss in specifying case definitions for PD [6, 7]. When studying the possible role of PD in systemic conditions, we believe it is the inflammatory component of PD that is important, thus a useful clinical measure of active PD is one that incorporates bleeding on probing as a measure of active inflammation in combination with clinical attachment loss [8, 9**, 10] and/or probing pocket depth [11]. Additional definitions of PD that may be important to studies of the systemic effect of PD are based on serological antibody titers to periodontal pathogens [12*, 13, 14] or clinical measures shown to correlate well with periodontal pathogen antibodies and inflammatory markers [11].
Coronary Heart Disease
CHD, as used in this review, refers to atherosclerosis of the coronary arteries leading to insufficiency of the myocardial blood supply due to reduction of blood flow through one or more of the coronary arteries or its branches [15]. Myocardial infarction, angina pectoris, and ischemic heart disease will be included as CHD in this report [1].
Chronic Kidney Disease
CKD is currently defined by the National Kidney Foundation’s Kidney Disease Outcome Quality Initiative’s (KDOQI) classification of five stages of increasing severity of CKD based on kidney damage (microalbuminuria) and decreasing kidney function measured as glomerular filtration rate [16].
Risk Marker
A risk marker is an attribute or exposure that is associated with an increased probability of disease but not necessary a causal factor [17]. Thus, a risk marker can be determined based on cross-sectional data. Because most studies of the association between PD and CHD and CKD are of cross-sectional design, we refer to these factors as “risk markers”.
Conceptual Model Linking PD with CHD and CKD
The conceptual model shown in the Figure is a simplified framework linking PD with CHD and CKD. Chronic inflammation elicits the acute-phase response and other inflammatory mediators that are common risk markers for CHD and CKD [4] and has a fundamental ongoing role in atherosclerosis [18]. As a chronic infection, PD contributes to the cumulative chronic inflammatory burden through the systemic dissemination of inflammatory mediators produced during the local tissue destructive immune-inflammatory response to periodontal pathogens as well as the systemic dissemination of the periodontal pathogens and their products (e.g., lipopolysaccharides) [19].
The link between PD and CKD may be bi-directional, as evidence is emerging to suggest uremia may contribute to increased gingival inflammation in patients on dialysis [20*]. Parenthetically, this bi-directional association is similar to the bi-directional relationship between PD and diabetes [21]. Kidney disease, by virtue of uremia or other mechanisms, may predispose to PD. The systemic inflammatory burden of PD may contribute to endothelial injury and atherogenesis [22*, 23]. The link between CHD and CKD may also be bi-directional. CKD is a well-established risk factor for CHD [2, 4, 16, 24] and clinical atherosclerosis and heart failure have recently been shown to be independently associated with rapid kidney function decline [25*]. It is noteworthy that adults with CKD are more likely to progress to cardiovascular events including CHD and heart failure, than to kidney failure [24].
Empirical Evidence to Support the Conceptual Model
Next, we will briefly describe the scientific evidence supporting each specific component of our conceptual model.
Bacteremia
The periodontal pathogenic bacteria and their byproducts (e.g., lipopolysaccharides) translocate via bacteremia after they easily penetrate the inflamed, ulcerated gingival tissue surrounding the teeth and enter the bloodstream, by which they are dispersed systemically and elicit host responses in distal tissues and organs. Researchers have found periodontal pathogenic bacteria in atherosclerotic plaque [26, 27] and in the arterial walls [28, 29]. Periodontal pathogens are associated with endothelial dysfunction [30].
Periodontopathogen-Specific Immune Response
Elevated Porphyromonas gingivalis (Pg) antibody titers have been found to be independently associated with PD, and high Eubacterium nodatum titers were associated with periodontal health [12*]. Two other studies also found those with PD had higher Pg antibody titer levels, as well as those with CHD [31*, 32]. Periodontopathogen antibody titers are shown to be associated with subclinical atherosclerosis as measured by carotid intima-medial thickness (IMT) [19] and with CHD and CHD death [33], whereas higher P. intermedia antibody levels have been found in acute coronary syndrome and coronary artery disease (CAD), suggesting that infection of an atheroma by periodontopathogens may trigger the acute coronary syndrome [10]. Interestingly, antibody titers to Aggregatibacter actinomycetemcomitans, another periodontal pathogen, have been reported to have an independent negative [5] and positive association [34] with CKD. The current evidence from these few reports remains inconclusive but support conducting further investigations to determine the value of measuring antibody titers to periodontal pathogenic bacteria in studying the role of PD in the pathogenesis of CHD and CKD.
Diabetes Duration, Glycemic Control, and Insulin Resistance
Diabetes must be considered in an evaluation of the relationships between PD and CHD and CKD because these three diseases are recognized as complications of diabetes. Further, rigorous management of glycemic control reduces the risk of CHD and CKD in people with diabetes [35], and evidence is emerging that non-surgical periodontal therapy significantly contributes to improvement in glycemic control [35, 36**]. As indicated in the conceptual model (see Figure), there are both direct and indirect effects of diabetes on CHD and/or CKD, with the indirect effects occurring through hypertension [37] and atherosclerosis [38]. In addition, insulin resistance has also been discussed as the metabolic link for PD contributing to poorer glycemic control in diabetes [39].
Figure.
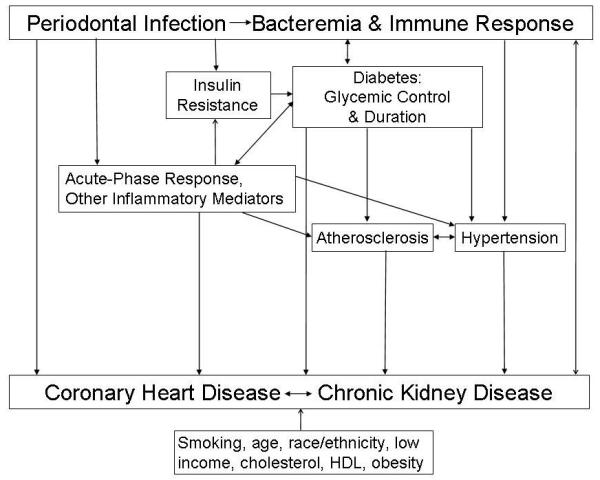
Conceptual Model Linking Periodontal Disease with Coronary Heart Disease and Chronic Kidney Disease
Hypertension
It is well established that hypertension increases the risk for CHD and CKD occurrence, progression, and mortality [40*, 4], as does lower blood pressure in dialysis patients. Relevant to this review, in cross-sectional studies of moderate-to-severe CKD (Stage 3-4) [5, 9**, 41], and kidney transplantation [42], those with hypertension were twice as likely to have CKD than those without hypertension after simultaneously taking into account other statistically significant risk markers, including PD. A large cross-sectional study provides the first population-based evidence for a direct relationship between the amounts of pathogenic subgingival periodontal bacteria and systolic and diastolic blood pressure, respectively, as well as prevalent hypertension [43**].
Atherosclerosis
Endothelial dysfunction
The inability of arteries and arterioles to dilate (endothelial dysfunction) is a key event in the development of atherosclerosis and precedes clinically obvious vascular pathology by many years. Endothelial dysfunction predicts myocardial dysfunction. Brachial artery flow-mediated dilation is a non-invasive measurement of endothelial dysfunction. A randomized controlled trial (RCT) of intensive periodontal therapy showed significant improvement of flow-mediated dilation [30], suggesting a causal link between PD and endothelial dysfunction as an indicator of early atherosclerosis in otherwise healthy individuals affected by severe PD. In order to elucidate the mechanisms involved, another study investigated the association between PD and circulating endothelial progenitor cells as markers for endothelial dysfunction [44*]. This study showed, for the first time, that moderate-to-severe PD was associated with an increased level of circulating endothelial progenitor cells which were also associated with CRP.
Intima Media Thickness (IMT)
IMT is a measure of subclinical atherosclerosis. A small study of 35 people with mild-to-moderate PD, found non-surgical periodontal therapy reduced levels of inflammatory markers and IMT [22*]. Evidence of a direct relationship between PD and IMT was first demonstrated among 657 dentate subjects when individuals with greater quantities of periodontopathogens had higher mean IMT than individuals with lower quantities. This relationship was independent of CRP, and was specific for pathogenic periodontal bacteria [45].
General Inflammatory Markers
An acute-phase inflammatory response, indicated by elevated CRP levels, is associated with many chronic diseases and is included in the Framingham risk score used to predict coronary death or myocardial infarction [40*]. The U.S. Preventive Services Task Force [46*] concluded from systematic reviews there is strong evidence that CRP is associated with CHD events [47*], but there is insufficient evidence to support reducing CRP levels prevents CHD. An important PD-related RCT is the multicenter Periodontitis And Vascular Events (PAVE) study. This pilot study evaluated the effect of periodontal treatment in preventing a secondary cardiac event in 303 individuals with CHD history by assessing periodontal status and CRP levels [48**]. No change in CRP level was found after treating obese individuals, but significantly fewer non-obese participants had CRP ≥3 mg/l following periodontal treatment. Periodontal therapy did not decrease CRP levels among individuals with CRP levels <3 mg/l at baseline. The authors suggested obesity increased CRP levels and nullified any periodontal treatment effects on CRP reduction.
In addition to CHD, higher CRP levels are independently associated with more rapid loss of kidney function [49]. African Americans are more likely to have CKD than non-Hispanic whites. (Race/ethnicity is another risk marker in the conceptual model depicted in the Figure). Only limited data exist for this racial disparity, which is not fully explained by the higher rates of hypertension, diabetes and obesity. Hence, an important study is the large cross-sectional study of 4,320 African Americans in the Jackson Heart Study that investigated the role of inflammation in CKD and found CRP was independently associated with CKD, but not significantly associated with albuminuria [50*]. This finding adds to the researchers earlier finding that CRP level is heritable after adjusting for age, gender and body mass index [51]. A meta-analysis reported strong evidence for elevated CRP in people with PD compared to those without PD and modest evidence for the effect of non-surgical periodontal therapy decreasing CRP levels [52]. A small RCT showed for the first time that non-surgical periodontal treatment caused a simultaneous reduction in plasma levels of CRP, interleukin(IL)-6, and fibrinogen in subjects with severe periodontitis and severe non-responsive arterial hypertension under medical treatment [53*]. A large population-based study of 4,830 Scottish adults found those who never or rarely brushed their teeth had an increased concentration of both CRP and fibrinogen [54*]. A recent case-control study of 68 cases with severe PD and 48 periodontally healthy controls found PD was associated with increased levels of CRP, glucose, fibrinogen, and IL-18, and with decreased levels of IL-4 [55*]. However, a study of dialysis patients found no association between moderate-to-severe PD and CRP or with serum albumin [56**], even though those with moderate-to-severe PD were five times more likely to die from cardiovascular events than those with no or mild PD. An earlier study by these researchers found severe PD was associated with serum albumin, but not with CRP [57]. An RCT found no significant difference in levels of CRP, IL-6, and plasminogen activator inhibitor-1 between an intensive periodontal treatment group and the community-based periodontal care control groups at 2- and 6-months follow-up examinations [30]. In another study, provision of comprehensive therapy to 30 patients with severe PD resulted in alterations in several inflammatory biomarkers that varied greatly and inconsistently among individuals. These changes were unrelated to severity of the periodontal infection and seemed to depend more on the inflammatory host response. The authors point to atherosclerotic status as a reasonable candidate to investigate as a confounder [58**].
Other Risk Markers
Additional risk markers common to CHD and CKD include smoking, age, race/ethnicity, low income/education, cholesterol, high-density lipoprotein and obesity [16, 40*]. However, the traditional factors explain only about half the deaths from CHD [59], and almost half of all heart attacks occur in patients without the classic Framingham study risk factors [60]. A candidate-gene association study demonstrated that CHD and PD are genetically related by at least one susceptibility locus, and this shared genetic susceptibility locus did not appear to be modified by the common risk factors for CHD and PD. Hence, the authors conclude the association between these common inflammatory complex diseases could be partially due to a shared genetic cause, thus providing new insight into the underlying partially shared pathogenic mechanisms of these complex common diseases [61**].
Empirical Evidence of the Role of PD in CHD
The effects of PD and treatment of PD are described under the respective headings of the main risk marker measured. A growing body of epidemiologic evidence supports an association between clinically diagnosed PD and CHD [62], and recently, the strength of the evidence for this biologically plausible association was found to be insufficient, but suggestive, of a causal relationship [63**]. Meta-analysis of 22 case-control and cross-sectional studies and 12 cohort studies concluded the risk for ischemic cardiovascular disease was significantly higher among individuals with PD [64**]. A large cross-sectional study of women [65] and a large prospective study of men and women with 12 years follow-up [66*] did not find a statistically significant relationship between probing pocket depth or PD, respectively and CHD, but found a significant association between tooth loss and increased risk for CHD and CHD mortality.
A U.S. cohort study with an average of 2.9 years follow-up of men and women with a history of myocardial infarction showed that loss of periodontal attachment level independently predicts recurrent cardiovascular events among analyses restricted to never-smokers [67**]. Similarly, a longitudinal study of patients with acute coronary syndrome reported PD was strongly associated with both first events and acute coronary syndrome recurrence [68*]. A case-control study of CAD and non-CAD found PD was independently associated with CAD [69].
Empirical Evidence of the Role of PD in CKD
The evidence for the role of PD in CKD prevalence and progression stems mainly from population-based cross-sectional studies, and case-control and smaller clinical studies. The effects of PD have been described under the respective headings of the main risk marker measured. In the context of studies on the relationship between PD and CKD, a population-based cross-sectional study found PD or its severe consequence, edentulism was independently associated with CKD after adjusting for 11 other traditional and nontraditional risk factors [5]. This study is noteworthy because it provided evidence for the importance of simultaneously taking into account other risk factors rather than identifying high-risk subgroups based on a single risk factor. In addition, the dose response for periodontal status was reported such that the odds of CKD increased as the severity or extent of PD increased. This finding is similar to an earlier longitudinal study of 529 Gila River Indian Community adults with type 2 diabetes that found a dose-response between PD (none/mild PD, moderate PD, severe PD, and edentulous) and incidence of macroalbuminuria and end-stage renal disease [70]. Another key finding is a prediction model for CKD in which PD or edentulism was a significant predictor, after controlling for the effect of diabetes (duration), in addition to controlling for high CRP level and eight other commonly recognized risk markers for CKD [9**]. Most importantly, the beta coefficients and formula provided in this report provide the opportunity to estimate anyone’s probability of CKD based on their specific characteristics as expressed by the presence or absence of 12 risk markers.
A highly relevant study of dialysis patients investigated the role of PD in CHD (thus incorporating all three diseases under review) using a well-designed retrospective 18 month follow-up study of 168 dentate adults. The important finding was that patients on dialysis with moderate-to-severe PD were five times more likely to die from cardiovascular disease than those with no or mild PD [56**].
Limitations to Causal Inferences
The major limitation to making unequivocal statements regarding the causal role of PD in the pathogenesis or progression of CHD and CKD is the inability to establish causality due to the cross-sectional study design of most of the large, population-based reports. Other limitations are the small number and lack of consistent findings from well-designed prospective studies, including clinical trials that control for important confounders and effect modifiers, such as smoking, diabetes, hypertension, and obesity.
Conclusion
While the evidence linking PD with CHD and CKD is not yet conclusive, recent evidence strengthens support of the biologically plausible role for PD in the occurrence and progression of CHD and CKD. To better understand the complex relationships between PD, CHD, and CKD, additional well-designed prospective cohort and controlled intervention studies are needed. Nevertheless, adding personal oral hygiene and referral for professional periodontal assessment and therapy to the repertoire of medical care recommendations is prudent to improve patients’ oral health and possibly reduce CHD and CKD risk.
Acknowledgements
This work was supported in part by grant NIH DE016031 (MAF).
Supported in part by NIH grant DE016031 to Dr. Monica A. Fisher
Footnotes
The authors report no conflicts of interest related to this report.
This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References and Recommended Reading
- 1.Centers for Disease Control and Prevention [Accessed 07/04/2010];Heart disease facts and statistics: deaths: final data for 2006. http://www.cdc.gov/heartdisease/statistics.htm.
- 2.National Kidney Foundation [Accessed 06/05/2010];Chronic Kidney Disease (CKD). The Facts About Chronic Kidney Disease (CKD) http://www.kidney.org/kidneydisease/ckd/index.cfm.
- 3**.Friedewald VE, Kornman KS, Beck JD, et al. The American Journal of Cardiology and Journal of Periodontology editors’ consensus: periodontitis and atherosclerotic cardiovascular disease. doi: 10.1902/jop.2009.097001. J Periodontol. 2009;80:1021–1032. doi: 10.1902/jop.2009.097001. Am J Cardiol. 2009;104:59–68. The guidelines in this unique consensus report by expert periodontists and cardiologists, published simultaneously in their journals, are paradigm-changing in their recommendation that medical and dental health care providers should closely collaborate in managing their mutual patients. Each recommendation is for a specific patient category and comes with scores of levels of confidence and evidence type, respectively.
- 4.Tonelli M, Pfeffer MA. Kidney disease and cardiovascular risk. Annu Rev Med. 2007;58:123–139. doi: 10.1146/annurev.med.58.071105.111123. [DOI] [PubMed] [Google Scholar]
- 5.Fisher MA, Taylor GW, Papapanou PN, et al. Clinical and serologic markers of periodontal infection and chronic kidney disease. J Periodontol. 2008;79:1670–1678. doi: 10.1902/jop.2008.070569. [DOI] [PubMed] [Google Scholar]
- 6.Borrell LN, Papapanou PN. Analytical epidemiology of periodontitis. J Clin Periodontol. 2005;32(Suppl 6):132–158. doi: 10.1111/j.1600-051X.2005.00799.x. [DOI] [PubMed] [Google Scholar]
- 7.Page RC, Eke PI. Case definitions for use in population-based surveillance of periodontitis. J Periodontol. 2007;78(7 Suppl):1387–1399. doi: 10.1902/jop.2007.060264. [DOI] [PubMed] [Google Scholar]
- 8.Fisher MA, Taylor GW, Tilashalski KR. Smokeless tobacco and severe active periodontal disease, NHANES III. J Dent Res. 2005;84:705–710. doi: 10.1177/154405910508400804. [DOI] [PubMed] [Google Scholar]
- 9**.Fisher MA, Taylor GW. A prediction model for chronic kidney disease includes periodontal disease. J Periodontol. 2009;80:16–23. doi: 10.1902/jop.2009.080226. With an estimated 75% of 7 million Americans with moderate-to-severe CKD undiagnosed, this U.S. population-based study of 11,955 adults is important because the prediction model identified high-risk subgroups for CKD. The most interesting aspects of this model are that 1) PD is included, and 2) it allows individuals or health care providers to estimate anyone’s probability of CKD by using the provided formula and beta-coefficients for each of the 12 risk markers: age, race/ethnicity, periodontal status, smoking status, duration of diabetes, and presence/absence of hypertension, macroalbuminuria, high cholesterol, low high-density lipoprotein, high CRP, lower income, and whether they were hospitalized in the past year.
- 10.Soejima H, Oe Y, Nakayama H, et al. Periodontal status and Prevotella intermedia antibody in acute coronary syndrome. Int J Cardiol. 2009;137:304–306. doi: 10.1016/j.ijcard.2008.05.057. [DOI] [PubMed] [Google Scholar]
- 11.Offenbacher S, Barros SP, Singer RE, et al. Periodontal disease at the biofilm-gingival interface. J Periodontol. 2007;78:1911–1925. doi: 10.1902/jop.2007.060465. [DOI] [PubMed] [Google Scholar]
- 12*.Dye BA, Herrera-Abreu M, Lerche-Sehm J, et al. Serum antibodies to periodontal bacteria as diagnostic markers of periodontitis. J Periodontol. 2009;80:634–647. doi: 10.1902/jop.2009.080474. This is an important, large, U.S. population-based study reporting on periodontal antibody titers as diagnostic markers of PD.
- 13.Colombo AP, Sakellari D, Haffajee AD, et al. Serum antibodies reacting with subgingival species in refractory periodontitis subjects. J Clin Periodontol. 1998;25:596–604. doi: 10.1111/j.1600-051x.1998.tb02493.x. [DOI] [PubMed] [Google Scholar]
- 14.Dye BA, Choudhary K, Shea S, Papapanou PN. Serum antibodies to periodontal pathogens and markers of systemic inflammation. J Clin Periodontol. 2005;32:1189–1199. doi: 10.1111/j.1600-051X.2005.00856.x. [DOI] [PubMed] [Google Scholar]
- 15.Labarthe D. Epidemiology and prevention of cardiovascular disease: a global challenge. Aspen Publishers; Gaithersburg, MD: 1998. [Google Scholar]
- 16.National Kidney Foundation K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am J Kidney Dis. 2002;39(2 Suppl 1):S1–S266. [PubMed] [Google Scholar]
- 17.Last J. A dictionary of epidemiology. 4th ed. Oxford University Press; New York: 2001. [Google Scholar]
- 18.Libby P, Ridker PM, Maseri A. Inflammation and atherosclerosis. Circulation. 2002;105:1135–1143. doi: 10.1161/hc0902.104353. [DOI] [PubMed] [Google Scholar]
- 19.Beck JD, Eke P, Lin D, et al. Association between IgG antibody to oral organisms and carotid intima-medial thickness in community-dwelling adults. Atherosclerosis. 2005;183:342–348. doi: 10.1016/j.atherosclerosis.2005.03.017. [DOI] [PubMed] [Google Scholar]
- 20*.Cengiz MI, Sumer P, Cengiz S, Yavuz U. The effect of the duration of the dialysis in hemodialysis patients on dental and periodontal findings. Oral Dis. 2009;15:336–341. doi: 10.1111/j.1601-0825.2009.01530.x. This study is important in its suggestion that uremia may contribute to increased gingival inflammation in patients on dialysis so that the link between PD and CKD may be bi-directional. It was not until having been on dialysis for ten years that participants had greater loss of periodontal bony support as measured by attachment loss.
- 21.Taylor GW, Borgnakke WS. Periodontal disease: associations with diabetes, glycemic control and complications. Oral Dis. 2008;14:191–203. doi: 10.1111/j.1601-0825.2008.01442.x. [DOI] [PubMed] [Google Scholar]
- 22*.Piconi S, Trabattoni D, Luraghi C, et al. Treatment of periodontal disease results in improvements in endothelial dysfunction and reduction of the carotid intima-media thickness. FASEB Journal. 2009;23:1196–1204. doi: 10.1096/fj.08-119578. A small, but potentially important, study of 35 people with mild-to-moderate PD investigated a secondary end-point of CHD and found non-surgical periodontal therapy reduced levels of inflammatory markers and IMT. However, these findings may be related to factors other than the periodontal therapy because this study did not include a control group.
- 23.Basile DP. The endothelial cell in ischemic acute kidney injury: implications for acute and chronic function. Kidney Int. 2007;72:151–156. doi: 10.1038/sj.ki.5002312. [DOI] [PubMed] [Google Scholar]
- 24.Go AS, Chertow GM, Fan D, et al. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004;351:1296–1305. doi: 10.1056/NEJMoa041031. [DOI] [PubMed] [Google Scholar]
- 25*.Shlipak MG, Katz R, Kestenbaum B, et al. Clinical and subclinical cardiovascular disease and kidney function decline in the elderly. Atherosclerosis. 2009;204:298–303. doi: 10.1016/j.atherosclerosis.2008.08.016. This important study provides evidence for a bidirectional relationship between CKD and CHD by finding an independent association between rapidly progressing decline in kidney function and clinical atherosclerosis and heart failure and carotid IMT.
- 26.Haraszthy VI, Zambon JJ, Trevisan M, et al. Identification of periodontal pathogens in atheromatous plaques. J Periodontol. 2000;71:1554–1560. doi: 10.1902/jop.2000.71.10.1554. [DOI] [PubMed] [Google Scholar]
- 27.Kozarov EV, Dorn BR, Shelburne CE, et al. Human atherosclerotic plaque contains viable invasive Actinobacillus actinomycetemcomitans and Porphyromonas gingivalis. Arterioscl Thromb Vasc Biol. 2005;25:e17–e18. doi: 10.1161/01.ATV.0000155018.67835.1a. [DOI] [PubMed] [Google Scholar]
- 28.Deshpande RG, Khan MB, Genco CA. Invasion of aortic and heart endothelial cells by Porphyromonas gingivalis. Infect Immun. 1998;66:5337–5343. doi: 10.1128/iai.66.11.5337-5343.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Dorn BR, Dunn WA, Jr., Progulske-Fox A. Invasion of human coronary artery cells by periodontal pathogens. Infect Immun. 1999;67:5792–5798. doi: 10.1128/iai.67.11.5792-5798.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tonetti MS, D’Aiuto F, Nibali L, et al. Treatment of periodontitis and endothelial function. N Engl J Med. 2007;356:911–920. doi: 10.1056/NEJMoa063186. [DOI] [PubMed] [Google Scholar]
- 31*.Bohnstedt S, Cullinan MP, Ford PJ, et al. High Antibody Levels to P.gingivalis in Cardiovascular Disease. J Dent Res. 2010 doi: 10.1177/0022034510370817. DOI: 10.1177/0022034510370817. By suggesting the immune response to Pg may play a role in CHD -- or serve as a marker for periodontal status in studies of CHD - this case-control study provides support for the role of the immune response to P. gingivalis in the relationship between periodontal disease and CHD (myocardial infarction or unstable angina).
- 32.Holmlund A, Hedin M, Pussinen PJ, et al. Porphyromonas gingivalis (Pg) a possible link between impaired oral health and acute myocardial infarction. Int J Cardiol. 2009 doi: 10.1016/j.ijcard.2009.10.034. DOI: 10.1016/j.ijcard.2009.10.034. [DOI] [PubMed] [Google Scholar]
- 33.Pussinen PJ, Nyyssönen K, Alfthan G, et al. Serum antibody levels to Actinobacillus actinomycetemcomitans predict the risk for coronary heart disease. Arterioscl Thromb Vasc Biol. 2005;25:833–838. doi: 10.1161/01.ATV.0000157982.69663.59. [DOI] [PubMed] [Google Scholar]
- 34.Kshirsagar AV, Offenbacher S, Moss KL, et al. Antibodies to periodontal organisms are associated with decreased kidney function; the dental atherosclerosis risk in communities study. Blood Purif. 2007;25:125–132. doi: 10.1159/000096411. [DOI] [PubMed] [Google Scholar]
- 35.Taylor GW, Borgnakke WS. Treatment of established complications: periodontal disease. In: Herman WH, Kinmouth AL, Wareham NJ, Williams R, editors. The evidence base in diabetes care. 2nd ed. John Wiley & Sons; Singapore: 2010. pp. 291–316. [Google Scholar]
- 36**.Simpson TC, Needleman I, Wild SH, et al. Treatment of periodontal disease for glycaemic control in people with diabetes. Cochrane Database of Systematic Reviews. 2010;(5) doi: 10.1002/14651858.CD004714.pub2. CD004714. This excellent recent Cochrane meta-analysis contributes to further evidence for the potentially positive effect of non-surgical periodontal therapy on improving glycemic control for individuals with diabetes. This is relevant in supporting the conceptual model developed for this review of the inter-relationship of three diabetes-related diseases.
- 37.Franjic B, Marwick TH. The diabetic, hypertensive heart: epidemiology and mechanisms of a very high-risk situation. J Hum Hypertens. 2009;23:709–717. doi: 10.1038/jhh.2009.43. [DOI] [PubMed] [Google Scholar]
- 38.D’Souza A, Hussain M, Howarth FC, et al. Pathogenesis and pathophysiology of accelerated atherosclerosis in the diabetic heart. Mol Cell Biochem. 2009;331:89–116. doi: 10.1007/s11010-009-0148-8. [DOI] [PubMed] [Google Scholar]
- 39.Donahue RP, Wu T. Insulin resistance and periodontal disease: An epidemiologic overview of research needs and future directions. Ann Periodontol. 2001;6:119–124. doi: 10.1902/annals.2001.6.1.119. [DOI] [PubMed] [Google Scholar]
- 40*.Framingham Heart Study [Accessed 07/04/2010];a project of the National Heart, Lung, and Blood Institute and Boston University: general cardiovascular disease risk score profile (10-year risk) http://www.framinghamheartstudy.org/risk/coronary.html. An excellent website of the Framingham Heart Study’s 10-year risk prediction for cardiovascular disease.
- 41.Fisher MA, Taylor GW, Shelton BJ, et al. Periodontal disease and other non-traditional risk factors for CKD. Am J Kidney Dis. 2008;51:45–52. doi: 10.1053/j.ajkd.2007.09.018. [DOI] [PubMed] [Google Scholar]
- 42.Ioannidou E, Shaqman M, Burleson J, Dongari-Bagtzoglou A. Periodontitis case definition affects the association with renal function in kidney transplant recipients. Oral Dis. 2010 doi: 10.1111/j.1601-0825.2010.01665.x. DOI: 10.1111/j.1601-0825.2010.01665.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43**.Desvarieux M, Demmer RT, Jacobs DR, et al. Periodontal bacteria and hypertension: the oral infections and vascular disease epidemiology study (INVEST) J Hypertens. 2010;28:1413–1421. doi: 10.1097/HJH.0b013e328338cd36. This report of 653 dentate adults is the first population-based study relating actual periodontal pathogenic bacteria to hypertension and therefore provides the first direct microbiological evidence of a possible contributory role for periodontal infections in hypertension etiology. These findings strengthen the hypothesis that periodontal infections may contribute to clinical CHD along with the finding that periodontopathogenic bacteria were not associated with hypertension.
- 44*.Li X, Tse HF, Yiu KH, et al. Increased levels of circulating endothelial progenitor cells in subjects with moderate to severe chronic periodontitis. J Clin Periodontol. 2009;36:933–939. doi: 10.1111/j.1600-051X.2009.01481.x. This is the first time that moderate to severe PD has been associated with an increased level of circulating endothelial progenitor cells, a subtype of bone marrow-derived stem cells that can express surface antigens of both hematopoietic stem cells and endothelial cells and participate in endothelial repair of vasculature and maintenance of vascular integrity.
- 45.Desvarieux M, Demmer RT, Rundek T, et al. Periodontal microbiota and carotid intima-media thickness: the oral infections and vascular disease epidemiology study (INVEST) Circulation. 2005;111:576–582. doi: 10.1161/01.CIR.0000154582.37101.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46*.Helfand M, Buckley DI, Freeman M, et al. Emerging risk factors for coronary heart disease: a summary of systematic reviews conducted for the U.S. Preventive Services Task Force. Ann Intern Med. 2009;151:496–507. doi: 10.7326/0003-4819-151-7-200910060-00010. This is an excellent recent review of the clinical usefulness of novel risk factors for CHD.
- 47*.Buckley DI, Fu R, Freeman M, et al. C-reactive protein as a risk factor for coronary heart disease: a systematic review and meta-analyses for the U.S. Preventive Services Task Force. Ann Intern Med. 2009;151:483–495. doi: 10.7326/0003-4819-151-7-200910060-00009. This systematic review and meta-analysis of studies of CRP as a risk factor for CHD concludes that strong evidence supports the association between CRP and CHD events, and moderate evidence suggests including CRP in risk prediction models to improve risk stratification. However, there is insufficient evidence that reducing CRP levels prevents CHD events.
- 48**.Offenbacher S, Beck JD, Moss K, et al. Results from the Periodontitis and Vascular Events (PAVE) Study: a pilot multicentered, RCT to study effects of periodontal therapy in a secondary prevention model of cardiovascular disease. J Periodontol. 2009;80:190–201. doi: 10.1902/jop.2009.080007. The interesting finding in this multicenter RCT is that periodontal treatment only seemed to have an effect on reduction of CRP levels for non-obese individuals. The authors suggest obesity increased CRP levels and nullified periodontal treatment effects on reducing CRP levels. This report also provides important insights into designing future RCTs to assess the role of periodontal treatment in reducing CHD risk.
- 49.Tonelli M, Sacks F, Pfeffer M, et al. Biomarkers of inflammation and progression of chronic kidney disease. Kidney Int. 2005;68:237–245. doi: 10.1111/j.1523-1755.2005.00398.x. [DOI] [PubMed] [Google Scholar]
- 50*.Fox ER, Benjamin EJ, Sarpong DF, et al. [Accessed 07/04/2010];The relation of C--reactive protein to chronic kidney disease in African Americans: the Jackson Heart Study. BMC Nephrol. 2010 11:1. doi: 10.1186/1471-2369-11-1. http://www.biomedcentral.com/1471-2369/11/1. This large cross-sectional study provides much needed data on the racial disparity in which African Americans are more likely to have CKD than non-Hispanic whites. This racial disparity is not fully explained by higher rates of hypertension, diabetes, and obesity, and thus, additional data -- such as this finding that CRP was independently associated with CKD, but not with albuminuria -- raise questions for further studies.
- 51.Fox ER, Benjamin EJ, Sarpong DF, et al. Epidemiology, Heritability, and Genetic Linkage of C-Reactive Protein in African Americans (from the Jackson Heart Study) Am J Cardiol. 2008;102:835–841. doi: 10.1016/j.amjcard.2008.05.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Paraskevas S, Huizinga JD, Loos BG. A systematic review and meta-analyses on C-reactive protein in relation to periodontitis. J Clin Periodontol. 2008;35:277–290. doi: 10.1111/j.1600-051X.2007.01173.x. [DOI] [PubMed] [Google Scholar]
- 53*.Vidal F, Figueredo CM, Cordovil I, Fischer RG. Periodontal therapy reduces plasma levels of interleukin-6, C-reactive protein, and fibrinogen in patients with severe periodontitis and refractory arterial hypertension. Journal of Periodontology. 2009;80:786–791. doi: 10.1902/jop.2009.080471. This small study of 11 adults in the treatment group and 11 non-treated controls found non-surgical periodontal treatment improves periodontal status among patients with severe PD and severe refractory hypertension under medical treatment over five years. Periodontal treatment reduced CRP, IL-6, and fibrinogen at the same time.
- 54*.de Oliveira C, Watt R, Hamer M. [Accessed 07/04/2010];Toothbrushing, inflammation, and risk of cardiovascular disease: results from Scottish Health Survey. BMJ. 2010 340:c2451. doi: 10.1136/bmj.c2451. http://www.bmj.com/cgi/content/full/340/may27_1/c2451. This is the first study to show an association between self-reported toothbrushing and incident cardiovascular disease -- of which 74% was CHD in a large representative sample of adults without overt cardiovascular disease. The causal nature of this association has yet to be determined.
- 55*.Buhlin K, Hultin M, Norderyd O, et al. Risk factors for atherosclerosis in cases with severe periodontitis. J Clin Periodontol. 2009;36:541–549. doi: 10.1111/j.1600-051X.2009.01430.x. This is the first report on the plasma pro-inflammatory cytokine IL-18 levels in PD, although increased levels of IL-18 have been shown to be present in inflamed periodontal tissues and in gingival crevicular tissue destruction.
- 56**.Kshirsagar AV, Craig RG, Moss KL, et al. Periodontal disease adversely affects the survival of patients with end-stage renal disease. Kidney Int. 2009;75:746–751. doi: 10.1038/ki.2008.660. This retrospective cohort study is important due to the link found between PD and the very serious outcome, namely that those with moderate-to-severe PD were over 5 times more likely to die from cardiovascular disease than those with no or mild PD.
- 57.Kshirsagar AV, Craig RG, Beck JD, et al. Severe periodontitis is associated with low serum albumin among patients on maintenance hemodialysis therapy. Clin J Am Soc Nephrol. 2007;2:239–244. doi: 10.2215/CJN.02420706. [DOI] [PubMed] [Google Scholar]
- 58**.Behle JH, Sedaghatfar MH, Demmer RT, et al. Heterogeneity of systemic inflammatory responses to periodontal therapy. J Clin Periodontol. 2009;36:287–294. doi: 10.1111/j.1600-051X.2009.01382.x. The key finding is the large inter-patient variability in pre-and post-intervention concentrations of inflammatory biomarkers, seemingly unrelated to the severity of periodontal infection. Such variance in host response we believe will become a main focus of investigations in the future.
- 59.National Center for Health Statistics [Accessed 06/09/2010];Deaths and Mortality (Data are for the U.S.): Number of deaths for leading causes of death; Source: Deaths: Final Data for 2006, tables B, D, 7, 30. 2009 http://www.cdc.gov/nchs/fastats/deaths.htm.
- 60.Smith SC., Jr Current and future directions of cardiovascular risk prediction. Am J Cardiol. 2006;97(2 Supplement 1):28–32. doi: 10.1016/j.amjcard.2005.11.013. [DOI] [PubMed] [Google Scholar]
- 61**.Schaefer AS, Richter GM, Groessner-Schreiber B, et al. Identification of a shared genetic susceptibility locus for coronary heart disease and periodontitis. PLoS Genetics. 2009;5:e1000378. doi: 10.1371/journal.pgen.1000378. This report provides new insight into the underlying partially shared risk factors for PD and CHD, such as smoking and diabetes, and involvement of a chronic inflammatory process. Interestingly, the finding of a shared genetic susceptibility locus did not appear to be modified by the common environmental and behavioral risk factors known to increase the susceptibility for CHD and/or PD.
- 62.Humphrey LL, Fu R, Buckley DI, et al. Periodontal disease and coronary heart disease incidence: a systematic review and meta-analysis. J Gen Intern Med. 2008;23:2079–2086. doi: 10.1007/s11606-008-0787-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63**.Joshipura K, Zevallos JC, Ritchie CS. Strength of evidence relating periodontal disease and atherosclerotic disease. Compend Contin Educ Dent. 2009;30:430–439. This report assessed the evidence supporting causal criteria (specificity, strength of association, dose-response, time sequence, biologic plausibility, consistency, and independence from confounding) relating PD and atherosclerotic disease, including ischemic heart disease.
- 64**.Blaizot A, Vergnes JN, Nuwwareh S, et al. Periodontal diseases and cardiovascular events: meta-analysis of observational studies. Int Dent J. 2009;59:197–209. Recent and comprehensive meta-analyses of 34 observational studies relating PD to ischemic heart disease due to cardiac atherosclerosis. The paradoxical finding that PD was more strongly associated with an acute cardiac event (i.e. acute myocardial infarction) than with a chronic cardiac condition (i.e. CHD) needs to be further investigated in future studies.
- 65.Stenman U, Wennstrom A, Ahlqwist M, et al. Association between periodontal disease and ischemic heart disease among Swedish women; a cross-sectional study. Acta Odontol Scand. 2009;67:193–199. doi: 10.1080/00016350902776716. [DOI] [PubMed] [Google Scholar]
- 66*.Holmlund A, Holm G, Lind L. Number of teeth as a predictor of cardiovascular mortality in a cohort of 7,674 subjects followed for 12 years. J Periodontol. 2010;81:870–876. doi: 10.1902/jop.2010.090680. 2010. A dose-dependent relationship between number of teeth and both all-cause and cardiovascular disease mortality is reported, indicating a possible link between oral health and cardiovascular disease.
- 67**.Dorn JM, Genco RJ, Grossi SG, et al. Periodontal Disease and Recurrent Cardiovascular Events in Survivors of Myocardial Infarction (MI): The Western New York Acute MI Study. J Periodontol. 2010;814:502–505. doi: 10.1902/jop.2009.090499. When studying smoking-related diseases, it is difficult to fully control for the effect of smoking when investigating the role of other factors. Thus, this longitudinal study is important because the researchers removed the effect of smoking by restricting subgroup analyses to never smokers, and in doing so demonstrated PD was associated with recurrent cardiovascular events. Also, this is one of few studies of a direct, primary outcome as opposed to indirect measures of CHD.
- 68*.Renvert S, Ohlsson O, Pettersson T, Persson GR. Periodontitis: a future risk for acute coronary syndrome? a follow up study over 3 years. J Periodontol. 2010;81:992–1000. doi: 10.1902/jop.2010.090105. An important Swedish longitudinal study of the role of PD and serum creatinine in both initial and recurrent acute coronary syndrome.
- 69.Oe Y, Soejima H, Nakayama H, et al. Significant association between score of periodontal disease and coronary artery disease. Heart & Vessels. 2009;24:103–107. doi: 10.1007/s00380-008-1096-z. [DOI] [PubMed] [Google Scholar]
- 70.Shultis WA, Weil EJ, Looker HC, et al. Effect of periodontitis on overt nephropathy and end-stage renal disease in type 2 diabetes. Diabetes Care. 2007;30:306–311. doi: 10.2337/dc06-1184. [DOI] [PubMed] [Google Scholar]


