Abstract
Autism, like intellectual disability, represents the severe end of a continuous distribution of developmental impairments that occur in nature, that are highly inherited, and that are orthogonally related to other parameters of development. A paradigm shift in understanding the core social abnormality of autism as a quantitative trait rather than as a categorically-defined condition has key implications for diagnostic classification, the measurement of change over time, the search for underlying genetic and neurobiologic mechanisms, and public health efforts to identify and support affected children. Here a recent body of research in genetics and epidemiology is presented to examine a dimensional reconceptualization of autistic social impairment—as manifested in clinical autistic syndromes, the broader autism phenotype, and normal variation in the general population. It illustrates how traditional categorical approaches to diagnosis may lead to misclassification of subjects (especially girls and mildly affected boys in multiple-incidence autism families), which can be particularly damaging to biological studies, and proposes continued efforts to derive a standardized quantitative system by which to characterize this family of conditions.
Introduction
Research in child development over the past several decades has revealed that clinical neuropsychiatric syndromes often represent the severe end of continuous distributions of core competencies and/or deficiencies that occur in nature. This concept was seminally applied to child psychopathology by Achenbach and colleagues (1) who developed and validated an empirically-based dimensional system of measurement; the implementation of that system in studies involving hundreds of thousands of children has revolutionized the way in which disorders of behavior and development are understood. Although on the surface, the difference between categorical (“all-or-nothing”) and dimensional classification systems may appear trivial, a paradigm shift between the two can have profound implications for the exploration of neural mechanisms underlying behavior, the enhancement of statistical power in biological research, the monitoring of effects of intervention, and helping parents understand (and accept) the nature of a psychiatric condition in a child. In recent years, the conceptualization of autistic syndromes as part of a continuum of social variation in nature has garnered considerable attention in science.
The fact that a variety of rare single-gene disorders now account for some 10-15 per cent of autistic syndromes (at the time of this writing) might seem to reinforce a categorical concept of autism; that the “autisms” actually represent a collection of such discrete disorders of social cognition (2). Just as is true for intellectual disability, however, the identification of such discrete syndromes of deficiency does not necessarily contradict the notion of a continuum, rather that the continuum as a whole (the “bell curve” for the example of intelligence) may be comprised, in part, of an array of clusters, each engendered by its own cause—independent, partially overlapping, or fully overlapping with the underlying causes of other clusters—and varying in range of severity. In addition, a discrete genetic cause may result in varying phenotypic expression (variable penetrance has been directly observed for specific structural chromosomal variations associated with autism (3) presumably on the basis of the manner in which that discrete genetic cause interacts with other attributes of the genotype or life experience of the individual. Alternatively, quantitative variation in an inherited trait may arise from the additive or interactive effects of multiple genetic influences of modest effect—whether rare or common, inherited or de novo—as well as environmental influences, the totality of which relate to severity in a stochastic fashion.
For the 85% of autism cases in which specific molecular or structural genetic variations are not yet appreciable, it remains unknown whether such multigenic influences are at play, however it has become increasingly clear that in some 25% of families affected by autism, multiple family members are affected by clinical or sub clinical autistic traits; and that within this sub set of families the distribution of autistic traits and symptoms appears highly quantitative, in a way that makes it extremely difficult to establish a clear demarcation between those affected versus unaffected (4, 5).
Multigenic mechanisms of causation have been identified for other complex conditions (diabetes, obesity, hypertension) and specific diseases (see below), in which common susceptibility alleles of relatively minor effect exert joint or interactive effects on the condition of interest, and some of the features of these medical conditions bear striking similarities to what is observed in autism spectrum conditions. For example, the short-segment variety of Hirschsprung's Disease—a disorder of neuronal migration to the large intestine—affects boys 5 times more commonly than girls, and exhibits both sporadic and familial patterns of intergenerational transmission. In the familial form of the disease, length of the gut that is affected (a straightforward quantitative index measured in centimeters) is associated with the number of recessive susceptibility alleles possessed by the patient, and is predictive of sibling recurrence risk (6, 7). This multifactorial genetic mechanism contrasts with the rarer and more severe long-segment Hirschsprung's Disease which follows a dominant pattern of autosomal transmission of single gene mutations of major effect. In schizophrenia, a neurodevelopmental disorder in which the normal process of synaptic pruning in adolescence is believed to be dramatically accelerated (in contrast to the disruption in synapse formation that is believed to occur in autism), sporadic cases have been associated with an elevated occurrence of de novo copy number variations (CNV's), some of which are the same as those observed in excess among sporadic autism (8).
Quantitative Variation in Autistic Symptomatology in Affected Families
Numerous studies, using various methods of measurement, have documented the aggregation of autistic syndromes, symptoms, or traits in the close relatives of children with autism. Such observations of familial aggregation have ranged from a full diagnosis of autistic disorder (for which siblings of children with autism have a relative risk of 20 or higher (9)), to milder autistic syndromes (Asperger Syndrome, Pervasive Developmental Disorder, Not Otherwise Specified (PDD-NOS)) (10), to sub clinical behavioral features of the autistic syndrome (4, 5, 11-13), to the “broader autism phenotype”, including personality traits that are akin to autistic symptoms (14-19); to developmental impairments or delays that more specifically involve language (15, 16, 18, 19).
Lauritsen and colleagues (9) additionally observed that the siblings of children with Asperger Syndrome in the Danish National Register had a 13 times higher-than-general-population risk for the development of full-blown autism, which constitutes some of the strongest evidence to date that the two disorders share common underlying genetic susceptibility factors. Studies that have carefully differentiated simplex autism (single family member affected, also referred to as sporadic autism) from multiplex autism (two or more family members affected, also referred to as familial autism) have suggested that familial aggregation of sub clinical autistic traits may occur only in multiplex autism (2, 5, 14) (Figure 1), providing further evidence for differentiation of mechanisms of genetic transmission for sporadic versus familial forms of the disorder.
Figure 1.
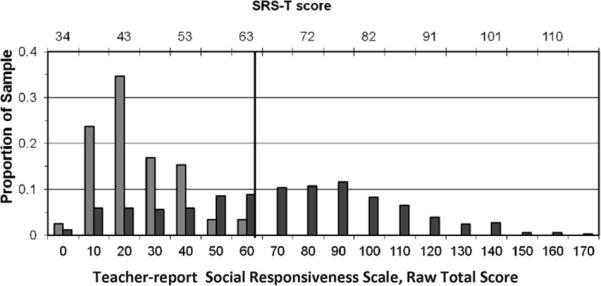
Distribution of Teacher-Report Social Responsiveness Scale scores (raw scores on x-axis, corresponding T-scores across the top of the figure) for male siblings of children with autism in single-incidence ( ) and multiple-incidence (■) families.
) and multiple-incidence (■) families.
In a recent large-scale study (n=1235 families) of the siblings of children affected by autism spectrum disorder (ASD) (4), 10 per cent were clinically-affected, but an additional 16 per cent were non-diagnosed siblings who nevertheless exhibited substantial elevations in social and/or communicative impairment occurring in these family members with a frequency approximately one order of magnitude more commonly than observed in the general population. Furthermore, when “recurrence” in this sample was operationalized using quantitative indices standardized by gender rather than absolute categorical definitions of case status, the typically-cited 3:1 sex ratio of males to females narrowed to 3:2. In essence, girls in families affected by autism were twice as likely to fall above a first percentile cutoff for females than they were to acquire a clinical diagnosis, and over ten times more likely to exceed the first percentile for autistic social impairment than girls in the general population.
Thus what is observed among clinically- and sub-clinically-affected individuals in families affected by autism is a very wide, and in essence, continuous distribution of autistic symptoms and traits (4, 5) comprising the so-called autism spectrum (as depicted for boys in multiple-incidence families in Figure 1). The question of how quantitative variation among clinically-affected individuals relates to the variation observed for autistic-like traits in the general population (i.e. generally less severe variations in social communication that are measurable using the same methods as employed in the studies above) remains another open and important question. Given the fact that even very subtle levels of autistic symptomatology have been shown to aggregate in some of the family members of children affected by ASD, it is possible that variation in the general population is influenced by the same sets of genetic and environmental factors that influence autism itself (this is discussed in further detail below. We have recently shown that when parents (4) and classroom teachers (5) rate the severity of ASD-diagnosed versus undiagnosed populations, the point of greatest differentiation of the respective distributions occurs at a point that is at approximately 1.5 standard deviations above the general population mean (thus not at an extreme tail), suggesting that there is substantial overlap between the general population distribution and the continuum of affectation that encompasses clinically-diagnosed patients.
The Factor Structure of Quantitative Autistic Traits
A key aspect of quantitative variation in autistic symptomatology involves the questions of whether (or to what extent) the three components of the autistic syndrome (social deficits, communicative deficits, and stereotypic behavior/restricted interests) represent independent symptom clusters (i.e., each with its own quantitative or qualitative architecture), or rather covary (“travel” together) in nature. In this review we focus on autistic social impairment as a core feature of autistic syndromes but recognize that language impairments and stereotypic behaviors may exhibit distributions and patterns of aggregation that diverge from the pattern described here for autistic social impairment. Here we summarize briefly what is understood about the associations between the three symptom domains of the autism triad in order to best contextualize the implications of a quantitative architecture for autistic social impairment.
Although some large-scale general population studies have suggested that the inherited influences on the three domains of symptoms are substantially (20) or partially (21) independent of one another, a caveat is that such studies can be confounded by measurement methods that do not ascertain autistic symptoms and traits with enough specificity. For example if children with non-autism-related disorders (such as specific language impairment or conduct disorder) are contributing to elevations in “autistic trait” scores ascertained in large populations, it can lead to overestimates of the extent to which separate lines of inheritance are responsible for the symptoms, since many of these syndromes are likely to have their own causal mechanisms independent from autism.
In contrast, factor, cluster, and latent class analyses of autistic symptoms in family studies have revealed substantial overlap in the three criterion domains for autism delineated in the Diagnostic and Statistical Manual of Mental Disorders 4th edition (DSM-IV). For example, Spiker and colleagues (22) studied sibling pairs affected by autism, and found that empirically-derived clusters of symptoms within families differed not by specific symptom sets, but by the degree of impairment that existed (mild, moderate or severe) across all three DSM-IV criterion domains for autism. Their findings were most consistent with a model of autistic symptomatology arising from a single, heritable, continuously distributed deficit which might influence dysfunction in all three symptom domains.
Similarly, Sung, Dawson and colleagues (12) examined features of the broader autism phenotype in the relatives of autistic probands and found evidence for the primary aggregation of highly heritable social deficits that explained variation in symptomatology across other domains of the autistic syndrome. Constantino and colleagues (23-25) applied factor, cluster, and latent class analysis to diagnostic interview and quantitative trait data (using the Social Responsiveness Scale, SRS) from children representing the entire range of autistic social impairment, from minimally affected to severe, and consistently observed a unitary factor structure across data sources and methods of analysis, reinforcing the syndromic nature of autistic impairment. A recent principal components factor analysis of an accumulated Washington University sample of 1,799 boys from 1,799 separate families, including over 300 clinically affected by autism, yields the factor structure depicted in the scree plot in Figure 3, indicating that a primary factor accounts for greater than 30% of the variance with all other possible factors contributing only minor components of variance. It is important to note that in this observation and in previous studies, the symptoms which load most strongly onto the principal factor represent all three of the DSM-IV criterion domains for autism.
Figure 3.
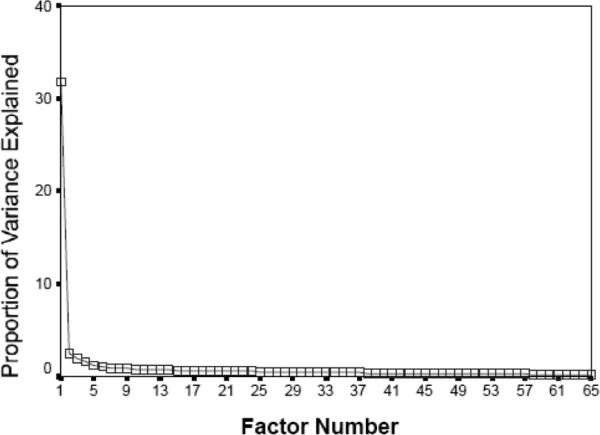
Scree plot of principal components analysis of SRS sample of 1,799 boys from 1,799 separate families, representing the full range of variation in autistic severity from unaffected to severely affected by clinical autistic syndromes,
Subsequent to the original reports summarized above, Gotham, Lord, and colleagues (26, 27) factor analyzed data from thousands of structured diagnostic observations of children with autism and concluded that the symptoms encompassing social deficiency and communicative deficiency comprised a single empirically-derived factor. Even at face value, a review of typical autistic traits reveals aspects of overlap across symptom domains that provide insight into key unifying constructs. The tendency for the social ability of individuals with ASD to be compromised by an over-focus on details (“missing the forest for the trees”) is highly reminiscent of the preoccupation with detail that characterizes young affected children's unusual (stereotypic) play with toys and older children's restriction in range of interests. It has been hypothesized that an underlying deficit in the assignment of salience (28) and the consequent absorption with details could underlie the social, stereotypic/behavioral, and even the communicative deficits (e.g., sentence comprehension compromised in the context of preserved decoding of individual words) of the autistic syndrome.
These observations provide a new framework for understanding quantitative variation in autistic symptomatology and challenge the 3-criterion taxonomy for differentiating specific pervasive developmental disorders (PDDs) in DSM-IV, by raising the possibility that each of the common disorders (Autistic Disorder, Asperger Syndrome, PDD-NOS) lie along a continuous distribution of impairment with a more parsimonious underlying factor structure. In such a reconceptualization, variation in autistic severity interacts with variation in other domains of development (general cognition, temperament, proneness to anxiety) to produce specific profiles of individual adaptation. For example, Asperger Syndrome, which is described in DSM-IV as a separate disorder (defined as autistic impairment without substantive language delay, and generally characterized by average to above average intellectual functioning), might be viewed as an autistic syndrome that is compensated by a level of intellectual functioning that is adequate to sustain normal language development, even in the presence of a level of social impairment that is otherwise typically associated with language impairment. In that sense it is the preservation of a relatively high level of general cognition—not the existence of a separate disorder—that uncouples the usual association between social and communicative impairment observed in autistic syndromes.
Molecular genetic studies are now just beginning to add to our knowledge of the factoral structure of autistic syndromes. While many disparate mutations have been associated with the same triad of symptoms observed in autism, quantitative trail loci (QTL) analyses have indicated that common autism susceptibility alleles may preferentially confer risk among specific subsets of autistic patients, for example those with versus without severe language delays (29). It has also been observed that a given large chromosomal rearrangement or allelic variation associated with autism (i.e. one that occurs more common in ASD than in the general population) might have highly variable phenotypic expression, as observed for 16p11 deletions which are found in patients with autism, patients with mental retardation without autism, and in relatively-typically-developing individuals within autism-affected families (3). Recognizing that a wide range of quantitative autistic traits may be present in the siblings of children with autism, it is possible to explore whether the association between phenotype and underlying genetic or neurobiologic mechanisms might be more appreciable when considering quantitative autistic trait information from all siblings in affected families rather than restricting analysis to the transmission of categorical disease states. Molecular genetic analyses utilizing quantitative trait information from all children of participating families (not just the fully affected subjects) have indicated that specific linkage signals in two independent multiplex family registries were substantially enhanced when adopting this approach (30, 31).
Finally, it is well recognized that some syndromes of specific language impairment (SLI) are inherited independently from autism, however, as discussed above, several studies have reported that language disorders with distinct autistic qualities (pronoun reversal, socially inappropriate phrases) and accompanied by mild-to-moderate levels of social deficiency also aggregate in the unaffected siblings of autistic probands (4, 15, 16, 18, 19). Thus, although the relationships between the symptom domains of the autism triad remain a subject of active investigation, there is accumulating evidence that they represent correlated behavioral manifestations of an underlying quantitative neurodevelopmental impairment. Ultimately molecular genetic studies and neuroimaging studies are poised to make major contributions to our understanding of how (and in what combinations) the various clinical manifestations of autism arise, and this in turn will inform strategies for intervention for specific sub-groups. It is highly conceivable that the severity distribution that constitutes the autism spectrum disorders is fully continuous with the distribution of sub clinical autistic symptomatology that is appreciable in the general population (as reviewed below). It has recently been demonstrated for autism (32) and for other complex diseases such as hypertension (33) that variation within the normal phenotypic range can be caused by variation in the same genes that are responsible for clinical disease states. From an evolutionary standpoint, it is possible that inherited factors that are actually adaptive when phenotypic expression is mild (as may well be the case for autistic traits) may be highly preserved in the population and result in clinical disease states only when severely expressed or in interaction with other genetic or environmental factors.
Quantitative Variation in Autistic Symptomatology in the General Population
Several large general population studies involving symptom counts and/or quantitative ratings of the severity of autistic social impairment, using disparate measurement methods (34-36) have consistently shown that the distribution of autistic symptoms in epidemiologic samples is continuous; this is shown for SRS total scores in a general population twin sample in the histogram in Figure 2. The SRS is a quantitative measure that was designed for use in public health settings and large research samples. It was intended to avoid ascertainment of specific impairments of speech and language, rather to focus on core deficiencies that characterize autism spectrum conditions, and therefore elicits severity ratings for impairment in reciprocal social behavior, social communication, and abnormalities in the domain of stereotypic behavior/restricted interests. Since the SRS exhibits a unitary factor structure, total scores are used as an index of autistic severity. A limitation of the scale by design is that impairments in language ascertained by the SRS are limited to social communication deficits in order to avoid confounds with Specific Language Impairment (SLI) and deficits in general cognition.
Figure 2.
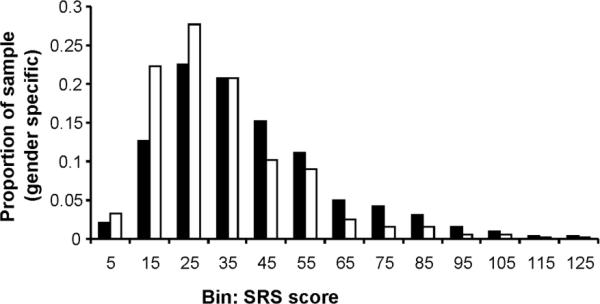
Reciprocal Social Behavior in the General Population: Distribution of score for reciprocal social deficits, parent-report Social Responsiveness Scale. males (■); females (□). Adapted from Constantino JN and Todd RD Arch Gen Psychiatry 60:524-530; Copyright © 2003 American Medical Association, with permission.
Using this measurement method quantitative variations measured throughout the course of childhood and adolescence have exhibited an extraordinary level of preservation of inter-individual differences (intra class coefficients on the order of 0.80) over years of time (37), demonstrating a level of stability that is similar to that observed in clinical autistic syndromes. Based on the distribution depicted in Figure 2, it appears highly arbitrary where distinctions would be drawn between the designation of affected versus unaffected status, and ASD can be viewed as the extreme end of a normative distribution for reciprocal social behavior (and its associated traits) that occurs in nature. The average child with a current designation of Pervasive Developmental Disorder – Not Otherwise Specified (PDD-NOS) scores at about the 99th percentile of the severity distribution for males. As is depicted in Figure 3, the female distribution is shifted toward the non-pathological end across the entire distribution; this results in sex ratios being exaggerated when extreme cutoffs using non-standardized scores are implemented as criteria for diagnosis, which is the case for all traditional autism diagnostic methods. Females who score at the 99th percentile for their standardized distribution are only half as likely as males to acquire a clinical diagnosis of ASD using current community standards for diagnosis (4), and this may explain a substantial share of what is universally-observed as a 3:1 sex ratio for categorical ASD diagnoses.
In order to ascertain whether gender-specific genetic effects account for the non-pathological shift in the distribution for females compared to males, opposite sex dizygotic twins have been studied (34). Opposite-sex twin pairs represent a gender-comparison condition within the classic twin design, in which common environmental influences can be modeled and controlled for. The results of structural equation modeling applied to the data in Figure 2 indicated that the genes influencing autistic traits appeared to be the same for males and females in the general population. Lower prevalence (and severity) of autistic traits in females was found not to be a function of sex-linked genetic influences, rather a possible result of increased sensitivity to early environmental influences (in females), which operate to reduce the phenotypic expression of genetic susceptibility factors and promote social competency (34). This is highly consistent with findings on sibling recurrence in clinically-affected families (4): given similar levels of genetic susceptibility, girls appear relatively protected (compared to boys) from severe phenotypic expression of this liability.
It is important to note that variation in the general population is highly heritable, at the same level of genetic influence that autism itself is believed to be inherited (34, 35, 38). This appears to be true throughout the distribution, i.e. both for social competency and social deficiency. This does not necessarily imply that social competency and social deficiency are controlled by the same genetic factors, but that possibility exists, and could conceivably be explored in very large epidemiologic twin samples in which the numbers of clinically-affected subjects are large enough to estimate whether genetic influences on a) quantitative trait scores and b) categorical designations of affectation status (at a clinical level cutoff) are determined by the same set of genetic influences—as of this writing, no such analysis has ever been published.
What is known about genetic influences on “sub threshold” autistic traits is that they overlap at least partially with those that influence some forms of autism: they preferentially aggregate in the unaffected family members of many children with ASD (4, 5, 11), particularly those for whom other members of the family are also fully affected by ASD (so-called multiple-incidence or “multiplex” ASD). Very recently, St. Pourcain and colleagues (32) showed that a common variation in genetic variant on 5p14.1 (rs4307059), a replicated susceptibility allele for autism spectrum disorder, is also associated with social communication spectrum phenotypes in the general population. Notably the association in the general population was explained neither by single-trait associations nor by overall behavioral adjustment problems but by a joint effect of multiple subthreshold social, communicative, and cognitive impairments—this is highly consistent with findings supporting a unitary factor structure for the autistic syndrome, discussed above.
The question also arises whether inherited influences or quantitative autistic traits are the same as or different from those influences involved in other dimensional domains of psychopathology. Twin designs are capable of answering such questions about genetic overlap, as long as the various traits of interest are all measured in the subjects in a given genetically informative sample. A series of studies in the general population have indicated that a) scores for internalizing behavior and externalizing behavior explained only a minority of the variance in quantitative autistic trait scores (39); b) there is moderate phenotypic and/or genetic overlap between attention problem syndromes and quantitative autistic traits (34); c) elevation in quantitative autistic trait scores may exacerbate co-occurring psychopathologic syndromes (25); and d) youths with severe mood or anxiety disorders exhibit substantially higher autistic trait scores than healthy controls (40) and the level of impairment incurred by these and other variations in personality and behavior may be predicted in part by co-occurrence of subclinical autistic symptomatology (41). Recently, Lichtenstein et al. (42) analyzed data from one of the largest twin studies ever to ascertain data on symptoms of neurodevelopmental impairment in children (n= 10,895 twin pairs). It revealed that while about 80% of the variation in liability for autism spectrum disorders was due to genetic effects, a large share of that genetic variance was shared respectively with ADHD, developmental coordination disorder, tic disorders and learning disorder, when respective bivariate analyses were applied to the data . Similarly, in a sample of 674 young adult Australian twins, Reiersen and colleagues (43) reported significant overlap of additive genetic influences for self-reported autistic traits and ADHD symptoms. Taken together these findings suggest that the inherited liabilities that predispose to ADHD and other neurodevelopmental conditions may exacerbate or contribute to the development of autistic syndromes.
The Clinical Ascertainment of Autistic Severity
A variety of established and emerging rating scales for social impairment in autism are capable of reliably quantifying its severity—these include checklists or questionnaires of current functioning completed by adult-informants (usually parents, teachers, or both) who have observed a child routinely in his/her naturalistic social contexts, assessments of developmental history as provided by parents, and direct observations of current social communicative behavior in response to structured or semi-structured social prompts. Each domain of observation can provide a unique and valuable vantage point for assessing the severity of autistic social impairment (as well as its relationship to other domains of development and psychopathology) in a given child.
There are many ways in which multi-informant characterization of a developing child can help avoid misclassification that would otherwise arise from single-informant or single-context assessment (44). Young children with milder forms of ASD who might be rapidly identified by an experienced day care teacher could go unrecognized if observed exclusively at home, since young parents are often inexperienced with respect to normative trajectories of social development. Moreover, such children can be reasonably competent in one-on-one social interactions with caring adults who can support and scaffold their interpersonal behavior, but have a great deal of difficulty in less structured social contexts in the company of larger numbers of close-aged peers. This extends into childhood and adolescence as well—there are many children who respond reasonably appropriately to a clinician-examiner in the context of a structured diagnostic assessment, but may be observed by teachers to display floridly inappropriate social behavior when in an unstructured context at school (lunch, gym, recess, bathroom breaks between classes). For these reasons it is important to acquire information from multiple sources (ideally direct clinical observation, parent-report, and teacher-report, see Constantino and colleagues (24) in the clinical evaluation of social impairment in a child suspected of having an ASD.
Endophenotypes and Quantitative Mapping of Childhood Development
In addition, a quantitative conceptualization of autism motivates the search for endophenotypes, which are inherited, quantitative phenotypic components of a syndrome. Endophenotypes, if they exist for autism, should be appreciable in some or many of the unaffected family members of clinically-affected individuals, and might more closely map to specific genetic, neurobiologic, or psychologic factors that contribute to (but are not by themselves sufficient to cause) the phenotypic expression of the syndrome itself. Endophenotypes are not the same as associated symptoms of a condition (e.g. self-injurious behavior in Lesch-Nyan syndrome), rather core components of the syndrome which appear in sub clinical forms in unaffected relatives. Sub clinical autistic traits exhibiting a unitary factor structure (as described earlier in this review) appear to constitute a social endophenotype for males in multiple-incidence (multiplex) autistic families, and the search for more precise endophenotypic components of the autistic syndrome (e.g. electrophysiologic, neuroimaging, or developmental trajectory markers) is under way.
As an example of the way in which two candidate endophenotypes might interact to confer susceptibility to autistic social impairment, Reiersen, Constantino & Todd (45) analyzed data on inattention and motor coordination deficits (separately-inherited domains of quantitative neurodevelopmental deficiency) in over 800 children in the general population and showed that their co-occurrence is associated with the development of autistic social impairment, as shown in Figure 4. These associations are reminiscent of a syndrome of “Deficits in Attention, Motor Coordination, and Perception” (DAMP) that has been observed and previously described in the child psychiatric literature (46).
Figure 4.
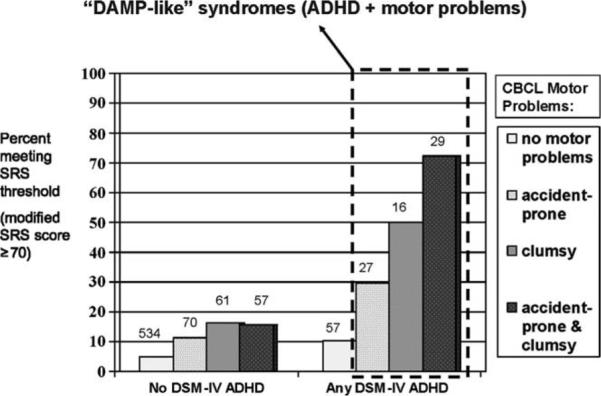
Percentage of subjects with clinically significant autistic traits, stratified by presence or absence of DSM-IV attention-deficit/hyperactivity disorder (ADHD) and by endorsement of Child Behavior Checklist (CBCL) motor problem items. N = 851. Number of subjects in each group is shown above each bar. DAMP = deficits in attention, motor control, and perception. Reprinted from Reiersen AM et al. J Am Acad Child Adolesc Psychiatry 47:662-672; Copyright © 2008 Elsevier, Ltd., with permission.
The current list of proposed candidate endophenotypes for autism is long, but the series of investigations required to establish them as true endophenotypes (through studies confirming heritability, patterns of familial aggregation, trait-like stability, and specific association with autism) is long as well. Endophenotypic candidates being actively pursued in autism research at the time of this writing include head circumference trajectory in infancy (47), laterality (handedness), electrophysiologic and brain activation responses to socially-relevant auditory and visual stimuli, sensory dysfunction, electroencephalogram and event related potential abnormality, newly-discovered neuroimaging phenotypes (48, 49), an array of neuropsychologic deficits (theory of mind, self-other representation, social motivation, capacity for shared intentionality, abnormalities in gaze cueing and/or joint attention), a variety of language endophenotypes (prosodic deficits, sentence comprehension abnormalities, timing of developmental milestones such as age of first word or first phrase), motor coordination problems, involuntary/repetitive motor movements, insistence on sameness, perseveration, and inattention. Each of these might be uniquely influenced by specific brain networks that could become targets of preventive or therapeutic interventions.
Finally, it is important to consider the implications of the interaction between: a) quantitative distributions of deficiency in reciprocal social behavior (accompanied by relative deficiency in communication and the presence of stereotypic behaviors and/or restricted interests as occurs in the autistic syndrome); and b) quantitative variation in general cognition, which is an independent developmental construct that can be differentiated from autistic social impairment within the normal distribution of IQ in the population, especially when using current measurement methods. Even if largely uncorrelated within the general population, clinical syndromes of severe deficiency in any developmental domain can exert expectable consequences on other developmental domains and many cases of ASD are complicated by co-occurring intellectual deficiency. Autism can occur in the context of low IQ, high IQ, or anything in between, and it is often difficult to know whether syndromes of combined social and cognitive impairment represent a cognitive disorder with secondary social impairment, the reverse, or some combination of two independent conditions.
Related to this is the confusion that can arise in clinically disentangling such deficits in young children with developmental delay. An example is the extremely common clinical scenario of the toddler who presents with language delay; it is perplexing (yet potentially relevant to the choice of intervention strategy) to determine whether that delay is most attributable to a primary cognitive deficit, an autistic syndrome, a specific language impairment, or some combination of dimensional variants of these conditions. A goal for the next decade is to map quantitative variation in social competency to quantitative variation in other fundamental (orthogonal) domains of childhood development.
If each neurodevelopmental domain is fundamentally quantitative in nature, it will ultimately require established maps of the expectable relations between the variables (analogous to the height versus weight norms used in pediatric practice) to accurately ascertain their relative contributions in a given case of developmental delay (50). We eagerly await a next generation of developmental-epidemiologic studies (some of which are currently under way) that will map the ways in which quantitative variations in fundamental domains of development (especially cognition, capacity for reciprocal social behavior, language, emotional regulation, sensorimotor function and interpersonal experience) interact with one another over the course of development from infancy to adulthood. It is possible that such studies will pave the way for a new system of characterizing all syndromes of developmental delay, along measurable quantitative axes, each of which might allow more precise associations with contributing biological mechanisms (37).
Acknowledgments
Statement of Financial Support:
J.N.C. is supported by a grant from the National Institute of Child Health and Human Development (HD42541).
Abbreviations
- ADHD
Attention-Deficit Hyperactivity Disorder
- ASD
Autism Spectrum Disorder
- DSM-IV
Diagnostic and Statistical Manual of Mental Disorders 4th edition
- PDD-NOS
Pervasive Developmental Disorder Not Otherwise Specified
Footnotes
Publisher's Disclaimer: Pediatric Research Articles Ahead of Print contains articles in unedited manuscript form that have been peer-reviewed and accepted for publication. As a service to our readers, we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting and review of the resulting proof before it is published in its final definitive form. Please note that during the production process errors may be discovered, which could affect the content, and all legal disclaimers that apply to the journal pertain.
Disclosure:
J.N.C. received royalties from Western Psychological Services for the commercial distribution of the Social Responsiveness Scale, which was originally developed to measure quantitative variation in autistic symptomatology in large research population, and for possible application in clinical, educational and public health settings.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- 1.Achenbach TM, Ruffle TM. The Child Behavior Checklist and related forms for assessing behavioral/emotional problems and competencies. Pediatr Rev. 2000;21:265–271. doi: 10.1542/pir.21-8-265. [DOI] [PubMed] [Google Scholar]
- 2.Frazier TW, Youngstrom EA, Sinclair L, Kubu CS, Law P, Rezai A, Constantino JN, Eng C. Autism spectrum disorders as a qualitatively distinct category from typical behavior in a large, clinically ascertained sample. Assessment. 2010;17:308–320. doi: 10.1177/1073191109356534. [DOI] [PubMed] [Google Scholar]
- 3.Bijlsma EK, Gijsbers AC, Schuurs-Hoeijmakers JH, van Haeringen A, Fransen van de Putte DE, Anderlid BM, Lundin J, Lapunzina P, Perez Jurado LA, Delle Chiaie B, Loeys B, Menten B, Oostra A, Verhelst H, Amor DJ, Bruno DL, van Essen AJ, Hordijk R, Sikkema-Raddatz B, Verbruggen KT, Jongmans MC, Pfundt R, Reeser HM, Breuning MH, Ruivenkamp CA. Extending the phenotype of recurrent rearrangements of 16p11.2: deletions in mentally retarded patients without autism and in normal individuals. Eur J Med Genet. 2009;52:77–87. doi: 10.1016/j.ejmg.2009.03.006. [DOI] [PubMed] [Google Scholar]
- 4.Constantino JN, Zhang Y, Frazier T, Abbacchi AM, Law P. Sibling Recurrence and the Genetic Epidemiology of Autism. Am J Psychiatry. 2010;167:1349–1356. doi: 10.1176/appi.ajp.2010.09101470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Virkud YV, Todd RD, Abbacchi AM, Zhang Y, Constantino JN. Familial aggregation of quantitative autistic traits in multiplex versus simplex autism. Am J Med Genet B Neuropsychiatr Genet. 2009;150B:328–334. doi: 10.1002/ajmg.b.30810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Amiel J, Sproat-Emison E, Garcia-Barcelo M, Lantieri F, Burzynski G, Borrego S, Pelet A, Arnold S, Miao X, Griseri P, Brooks AS, Antinolo G, de Pontual L, Clement-Ziza M, Munnich A, Kashuk C, West K, Wong KK, Lyonnet S, Chakravarti A, Tam PK, Ceccherini I, Hofstra RM, Fernandez R. Hirschsprung disease, associated syndromes and genetics: a review. J Med Genet. 2008;45:1–14. doi: 10.1136/jmg.2007.053959. [DOI] [PubMed] [Google Scholar]
- 7.Emison ES, Garcia-Barcelo M, Grice EA, Lantieri F, Amiel J, Burzynski G, Fernandez RM, Hao L, Kashuk C, West K, Miao X, Tam PK, Griseri P, Ceccherini I, Pelet A, Jannot AS, de Pontual L, Henrion-Caude A, Lyonnet S, Verheij JB, Hofstra RM, Antinolo G, Borrego S, McCallion AS, Chakravarti A. Differential contributions of rare and common, coding and noncoding Ret mutations to multifactorial Hirschsprung disease liability. Am J Hum Genet. 2010;87:60–74. doi: 10.1016/j.ajhg.2010.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Guilmatre A, Dubourg C, Mosca AL, Legallic S, Goldenberg A, Drouin-Garraud V, Layet V, Rosier A, Briault S, Bonnet-Brilhault F, Laumonnier F, Odent S, Le Vacon G, Joly-Helas G, David V, Bendavid C, Pinoit JM, Henry C, Impallomeni C, Germano E, Tortorella G, Di Rosa G, Barthelemy C, Andres C, Faivre L, Frebourg T, Saugier Veber P, Campion D. Recurrent rearrangements in synaptic and neurodevelopmental genes and shared biologic pathways in schizophrenia, autism, and mental retardation. Arch Gen Psychiatry. 2009;66:947–956. doi: 10.1001/archgenpsychiatry.2009.80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lauritsen MB, Pedersen CB, Mortensen PB. Effects of familial risk factors and place of birth on the risk of autism: a nationwide register-based study. J Child Psychol Psychiatry. 2005;46:963–971. doi: 10.1111/j.1469-7610.2004.00391.x. [DOI] [PubMed] [Google Scholar]
- 10.Pickles A, Starr E, Kazak S, Bolton P, Papanikolaou K, Bailey A, Goodman R, Rutter M. Variable expression of the autism broader phenotype: findings from extended pedigrees. J Child Psychol Psychiatry. 2000;41:491–502. [PubMed] [Google Scholar]
- 11.Constantino JN, Lajonchere C, Lutz M, Gray T, Abbacchi A, McKenna K, Singh D, Todd RD. Autistic social impairment in the siblings of children with pervasive developmental disorders. Am J Psychiatry. 2006;163:294–296. doi: 10.1176/appi.ajp.163.2.294. [DOI] [PubMed] [Google Scholar]
- 12.Sung YJ, Dawson G, Munson J, Estes A, Schellenberg GD, Wijsman EM. Genetic investigation of quantitative traits related to autism: use of multivariate polygenic models with ascertainment adjustment. Am J Hum Genet. 2005;76:68–81. doi: 10.1086/426951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Szatmari P, Bryson SE, Streiner DL, Wilson F, Archer L, Ryerse C. Two-year outcome of preschool children with autism or Asperger's syndrome. Am J Psychiatry. 2000;157:1980–1987. doi: 10.1176/appi.ajp.157.12.1980. [DOI] [PubMed] [Google Scholar]
- 14.Losh M, Childress D, Lam K, Piven J. Defining key features of the broad autism phenotype: a comparison across parents of multiple- and single-incidence autism families. Am J Med Genet B Neuropsychiatr Genet. 2008;147B:424–433. doi: 10.1002/ajmg.b.30612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kjelgaard MM, Tager-Flusberg H. An Investigation of Language Impairment in Autism: Implications for Genetic Subgroups. Lang Cogn Process. 2001;16:287–308. doi: 10.1080/01690960042000058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lindgren KA, Folstein SE, Tomblin JB, Tager-Flusberg H. Language and reading abilities of children with autism spectrum disorders and specific language impairment and their first-degree relatives. Autism Res. 2009;2:22–38. doi: 10.1002/aur.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Piven J, Palmer P, Jacobi D, Childress D, Arndt S. Broader autism phenotype: evidence from a family history study of multiple-incidence autism families. Am J Psychiatry. 1997;154:185–190. doi: 10.1176/ajp.154.2.185. [DOI] [PubMed] [Google Scholar]
- 18.Toth K, Dawson G, Meltzoff AN, Greenson J, Fein D. Early social, imitation, play, and language abilities of young non-autistic siblings of children with autism. J Autism Dev Disord. 2007;37:145–157. doi: 10.1007/s10803-006-0336-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Whitehouse AJ, Barry JG, Bishop DV. The broader language phenotype of autism: a comparison with specific language impairment. J Child Psychol Psychiatry. 2007;48:822–830. doi: 10.1111/j.1469-7610.2007.01765.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Happé F, Ronald A. The ‘fractionable autism triad’: a review of evidence from behavioural, genetic, cognitive and neural research. Neuropsychol Rev. 2008;18:287–304. doi: 10.1007/s11065-008-9076-8. [DOI] [PubMed] [Google Scholar]
- 21.Ronald A, Larsson H, Anckarsäter H, Lichtenstein P. A twin study of autism symptoms in Sweden. Mol Psychiatry. 2010 doi: 10.1038/mp.2010.82. in press, doi:10.1038/mp.2010.82. [DOI] [PubMed] [Google Scholar]
- 22.Spiker D, Lotspeich LJ, Dimiceli S, Myers RM, Risch N. Behavioral phenotypic variation in autism multiplex families: evidence for a continuous severity gradient. Am J Med Genet. 2002;114:129–136. doi: 10.1002/ajmg.10188. [DOI] [PubMed] [Google Scholar]
- 23.Constantino JN, Gruber CP, Davis S, Hayes S, Passanante N, Przybeck T. The factor structure of autistic traits. J Child Psychol Psychiatry. 2004;45:719–726. doi: 10.1111/j.1469-7610.2004.00266.x. [DOI] [PubMed] [Google Scholar]
- 24.Constantino JN, Lavesser PD, Zhang Y, Abbacchi AM, Gray T, Todd RD. Rapid quantitative assessment of autistic social impairment by classroom teachers. J Am Acad Child Adolesc Psychiatry. 2007;46:1668–1676. doi: 10.1097/chi.0b013e318157cb23. [DOI] [PubMed] [Google Scholar]
- 25.Constantino JN, Todd RD. Genetic structure of reciprocal social behavior. Am J Psychiatry. 2000;157:2043–2045. doi: 10.1176/appi.ajp.157.12.2043. [DOI] [PubMed] [Google Scholar]
- 26.Gotham K, Risi S, Pickles A, Lord C. The Autism Diagnostic Observation Schedule: revised algorithms for improved diagnostic validity. J Autism Dev Disord. 2007;37:613–627. doi: 10.1007/s10803-006-0280-1. [DOI] [PubMed] [Google Scholar]
- 27.Lord C, Risi S, DiLavore PS, Shulman C, Thurm A, Pickles A. Autism from 2 to 9 years of age. Arch Gen Psychiatry. 2006;63:694–701. doi: 10.1001/archpsyc.63.6.694. [DOI] [PubMed] [Google Scholar]
- 28.Klin A, Jones W, Schultz R, Volkmar F, Cohen D. Visual fixation patterns during viewing of naturalistic social situations as predictors of social competence in individuals with autism. Arch Gen Psychiatry. 2002;59:809–816. doi: 10.1001/archpsyc.59.9.809. [DOI] [PubMed] [Google Scholar]
- 29.Alarcón M, Yonan AL, Gilliam TC, Cantor RM, Geschwind DH. Quantitative genome scan and Ordered-Subsets Analysis of autism endophenotypes support language QTLs. Mol Psychiatry. 2005;10:747–757. doi: 10.1038/sj.mp.4001666. [DOI] [PubMed] [Google Scholar]
- 30.Duvall JA, Lu A, Cantor RM, Todd RD, Constantino JN, Geschwind DH. A quantitative trait locus analysis of social responsiveness in multiplex autism families. Am J Psychiatry. 2007;164:656–662. doi: 10.1176/ajp.2007.164.4.656. [DOI] [PubMed] [Google Scholar]
- 31.Coon H, Villalobos ME, Robison RJ, Camp NJ, Cannon DS, Allen-Brady K, Miller JS, McMahon WM. Genome-wide linkage using the Social Responsiveness Scale in Utah autism pedigrees. Mol Autism. 2010;1:8. doi: 10.1186/2040-2392-1-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.St Pourcain B, Wang K, Glessner JT, Golding J, Steer C, Ring SM, Skuse DH, Grant SF, Hakonarson H, Smith GD. Association between a high-risk autism locus on 5p14 and social communication spectrum phenotypes in the general population. Am J Psychiatry. 2010;167:1364–1372. doi: 10.1176/appi.ajp.2010.09121789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ji W, Foo JN, O'Roak BJ, Zhao H, Larson MG, Simon DB, Newton-Cheh C, State MW, Levy D, Lifton RP. Rare independent mutations in renal salt handling genes contribute to blood pressure variation. Nat Genet. 2008;40:592–599. doi: 10.1038/ng.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Constantino JN, Todd RD. Autistic traits in the general population: a twin study. Arch Gen Psychiatry. 2003;60:524–530. doi: 10.1001/archpsyc.60.5.524. [DOI] [PubMed] [Google Scholar]
- 35.Ronald A, Happe F, Price TS, Baron-Cohen S, Plomin R. Phenotypic and genetic overlap between autistic traits at the extremes of the general population. J Am Acad Child Adolesc Psychiatry. 2006;45:1206–1214. doi: 10.1097/01.chi.0000230165.54117.41. [DOI] [PubMed] [Google Scholar]
- 36.Skuse DH, Mandy W, Steer C, Miller LL, Goodman R, Lawrence K, Emond A, Golding J. Social communication competence and functional adaptation in a general population of children: preliminary evidence for sex-by-verbal IQ differential risk. J Am Acad Child Adolesc Psychiatry. 2009;48:128–137. doi: 10.1097/CHI.0b013e31819176b8. [DOI] [PubMed] [Google Scholar]
- 37.Constantino JN, Abbacchi AM, Lavesser PD, Reed H, Givens L, Chiang L, Gray T, Gross M, Zhang Y, Todd RD. Developmental course of autistic social impairment in males. Dev Psychopathol. 2009;21:127–138. doi: 10.1017/S095457940900008X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Constantino JN, Todd RD. Intergenerational transmission of subthreshold autistic traits in the general population. Biol Psychiatry. 2005;57:655–660. doi: 10.1016/j.biopsych.2004.12.014. [DOI] [PubMed] [Google Scholar]
- 39.Constantino JN, Hudziak JJ, Todd RD. Deficits in reciprocal social behavior in male twins: evidence for a genetically independent domain of psychopathology. J Am Acad Child Adolesc Psychiatry. 2003;42:458–467. doi: 10.1097/01.CHI.0000046811.95464.21. [DOI] [PubMed] [Google Scholar]
- 40.Pine DS, Guyer AE, Goldwin M, Towbin KA, Leibenluft E. Autism spectrum disorder scale scores in pediatric mood and anxiety disorders. J Am Acad Child Adolesc Psychiatry. 2008;47:652–661. doi: 10.1097/CHI.0b013e31816bffa5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kanne SM, Christ SE, Reiersen AM. Psychiatric symptoms and psychosocial difficulties in young adults with autistic traits. J Autism Dev Disord. 2009;39:827–833. doi: 10.1007/s10803-008-0688-x. [DOI] [PubMed] [Google Scholar]
- 42.Lichtenstein P, Carlstrom E, Rastam M, Gillberg C, Anckarsater H. The genetics of autism spectrum disorders and related neurospychaitric disorders in childhood. Am J Psychiatry. 2010;167:1357–1363. doi: 10.1176/appi.ajp.2010.10020223. [DOI] [PubMed] [Google Scholar]
- 43.Reiersen AM, Constantino JN, Grimmer M, Martin NG, Todd RD. Evidence for shared genetic influences on self-reported ADHD and autistic symptoms in young adult Australian twins. Twin Res Hum Genet. 2008;11:579–585. doi: 10.1375/twin.11.6.579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kanne SM, Abbacchi AM, Constantino JN. Multi-informant ratings of psychiatric symptom severity in children with autism spectrum disorders: the importance of environmental context. J Autism Dev Disord. 2009;39:856–864. doi: 10.1007/s10803-009-0694-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Reiersen AM, Constantino JN, Todd RD. Co-occurrence of motor problems and autistic symptoms in attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2008;47:662–672. doi: 10.1097/CHI.0b013e31816bff88. [DOI] [PubMed] [Google Scholar]
- 46.Landgren M, Pettersson R, Kjellman B, Gillberg C. ADHD, DAMP and other neurodevelopmental/psychiatric disorders in 6-year-old children: epidemiology and co-morbidity. Dev Med Child Neurol. 1996;38:891–906. doi: 10.1111/j.1469-8749.1996.tb15046.x. [DOI] [PubMed] [Google Scholar]
- 47.Constantino JN, Majmudar P, Bottini A, Arvin M, Virkud Y, Simons P, Spitznagel E. Infant head growth in male siblings of children with and without autism spectrum disorders. J Neurodev Disord. 2010;2:39–46. doi: 10.1007/s11689-009-9036-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kaiser MD, Hudac CM, Shultz S, Lee SM, Cheung C, Berken AM, Deen B, Pitskel NB, Sugrue DR, Voos AC, Saulnier CA, Ventola P, Wolf JM, Klin A, Vander Wyk BC, Pelphrey KA. Neural signatures of autism. Proc Natl Acad Sci USA. 2010;107:21223–21228. doi: 10.1073/pnas.1010412107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Scott-Van Zeeland AA, Abrahams BS, Alvarez-Retuerto AI, Sonnenblick LI, Rudie JD, Ghahremani D, Mumford JA, Poldrack RA, Dapretto M, Geschwind DH, Bookheimer SY. Altered functional connectivity in frontal lobe circuits is associated with variation in the autism risk gene CNTNAP2. Sci Transl Med. 2010;2:56ra80. doi: 10.1126/scitranslmed.3001344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Constantino JN. How continua converge in nature: cognition, social competence, and autistic syndromes. J Am Acad Child Adolesc Psychiatry. 2009;48:97–98. doi: 10.1097/CHI.0b013e318193069e. [DOI] [PubMed] [Google Scholar]


