The expression “seesaw effect” was originally used by Sieradzki and Tomasz (7), and subsequently by others, to denote a frequently observed inverse relationship between evolving glycopeptide and β-lactam MICs in Staphylococcus aureus. The same expression has recently been revived in two interesting studies by Yang et al. (9) and Lee et al. (4) to signify a similar phenomenon, again seen in S. aureus, involving daptomycin instead of glycopeptides. On the other hand, it is well known that S. aureus strains progressively acquiring daptomycin nonsusceptibility during daptomycin exposure also exhibit progressively increasing vancomycin MICs (3, 6).
Among coagulase-negative staphylococci, Staphylococcus haemolyticus, second only to Staphylococcus epidermidis in the frequency of its association with human infections (1), is unique in being predisposed to developing glycopeptide resistance and was the first Gram-positive pathogen to acquire such resistance in the 1980s (2).
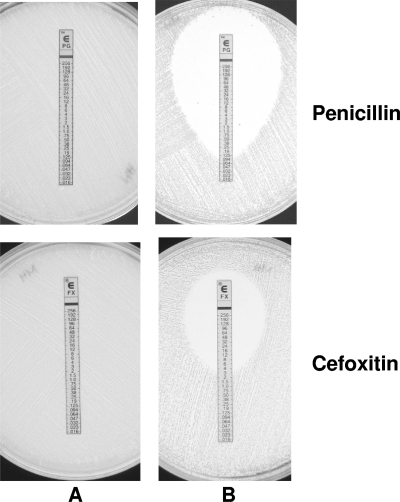
After exposure to increasing daptomycin concentrations, by a procedure successfully used with glycopeptides in previous studies in our laboratory (8), a stable clone with a daptomycin MIC of 4 μg/ml was obtained from a daptomycin-susceptible (MIC, 0.5 μg/ml) clinical isolate of S. haemolyticus. Vancomycin and teicoplanin MICs also increased from 4 and 8 μg/ml in the parent to 8 and 32 μg/ml in the laboratory derivative, respectively. The parent strain was both penicillin and methicillin resistant: penicillin and cefoxitin MICs were >256 μg/ml; molecular analysis disclosed a type I SCCmec cassette and regular mec and bla operons; β-lactamase production was confirmed by the nitrocefin test. The seesaw effect was striking (Fig. 1): in the daptomycin-nonsusceptible derivative, the penicillin MIC dropped to 0.125 μg/ml, in spite of persistent detection of the bla operon and of β-lactamase activity, and the cefoxitin MIC dropped to 2 μg/ml, despite persistent detection of the mecA gene and the mec operon in a type I SCCmec.
Fig. 1.
Diffusion tests using Etest strips (penicillin and cefoxitin). (A) Clinical isolate of daptomycin-susceptible S. haemolyticus (parent strain). (B) Daptomycin-nonsusceptible laboratory derivative.
Although a number of theories have been advanced to account for the vancomycin/β-lactam and the daptomycin/β-lactam seesaw effect (7, 9), the underlying mechanisms remain poorly understood. Moreover, while previous hypotheses were essentially aimed at explaining a fall in methicillin resistance, thus largely pointing to some modulation of mecA expression, this case also involves a plunge in penicillin resistance, despite apparently normal—phenotypically and genotypically—β-lactamase production in the laboratory derivative. In other words, here the seesaw effect needs to be explained in light not only of the mec operon/PBP2a-mediated β-lactam resistance system but also of the bla operon/β-lactamase-mediated one. It is worth noting that, in a reported case of decreased susceptibility to daptomycin and vancomycin in S. aureus during prolonged therapy, a decreased penicillin MIC was found to be associated with the apparent loss of β-lactamase activity (5).
REFERENCES
- 1. Bannerman T. L., Peacock S. J. 2007. Staphylococcus, Micrococcus, and other catalase-positive cocci, p. 390–411 In Murray P. R., Baron E. J., Jorgensen J. H., Landry M. L., Pfaller M. A. (ed.), Manual of clinical microbiology, 9th ed. ASM Press, Washington, DC [Google Scholar]
- 2. Biavasco F., Vignaroli C., Varaldo P. E. 2000. Glycopeptide resistance in coagulase-negative staphylococci. Eur. J. Clin. Microbiol. Infect. Dis. 19:403–417 [DOI] [PubMed] [Google Scholar]
- 3. Camargo I. L., Neoh H. M., Cui L., Hiramatsu K. 2008. Serial daptomycin selection generates daptomycin-nonsusceptible Staphylococcus aureus strains with a heterogeneous vancomycin-intermediate phenotype. Antimicrob. Agents Chemother. 52:4289–4299 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Lee C. H., Wang M. C., Huang I. W., Chen F. J., Lauderdale T. L. 2010. Development of daptomycin nonsusceptibility with heterogeneous vancomycin-intermediate resistance and oxacillin susceptibility in methicillin-resistant Staphylococcus aureus during high-dose daptomycin treatment. Antimicrob. Agents Chemother. 54:4038–4040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Mariani P. G., Sader H. S., Jones R. N. 2006. Development of decreased susceptibility to daptomycin and vancomycin in a Staphylococcus aureus strain during prolonged therapy. J. Antimicrob. Chemother. 58:481–483 [DOI] [PubMed] [Google Scholar]
- 6. Mishra N. N., et al. 2009. Analysis of cell membrane characteristics of in vitro-selected daptomycin-resistant strains of methicillin-resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 53:2312–2318 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Sieradzki K., Tomasz A. 1997. Inhibition of cell wall turnover and autolysis by vancomycin in a highly vancomycin-resistant mutant of Staphylococcus aureus. J. Bacteriol. 179:2557–2566 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Vignaroli C., Biavasco F., Varaldo P. E. 2006. Interactions between glycopeptides and β-lactams against isogenic pairs of teicoplanin-susceptible and -resistant strains of Staphylococcus haemolyticus. Antimicrob. Agents Chemother. 50:2577–2582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Yang S. J., et al. 2010. Daptomycin-oxacillin combinations in treatment of experimental endocarditis caused by daptomycin-nonsusceptible strains of methicillin-resistant Staphylococcus aureus with evolving oxacillin susceptibility (the “seesaw effect”). Antimicrob. Agents Chemother. 54:3161–3169 [DOI] [PMC free article] [PubMed] [Google Scholar]