This work is of importance, not only because it explores the physiological role of kynurenines in controlling immune responses, but also because it opens the possibility of using kynurenine-like molecules as immunosuppressive therapy.
Abstract
Purpose.
IDO (indoleamine 2,3-dioxygenase) modulates the immune response by depletion of the essential amino acid tryptophan, and IDO overexpression has been shown to prolong corneal allograft survival. This study was conducted to examine the effect of kynurenines, the products of tryptophan breakdown and known to act directly on T lymphocytes, on corneal graft survival.
Methods.
The effects of kynurenines on T-cell proliferation and death, T-regulatory-cell development, and dendritic cell function, phenotype, and viability were analyzed in vitro. The effect of topical and systemic administration of 3-hydroxykynurenine (3HK) on orthotopic murine corneal allograft survival was examined.
Results.
T-lymphocyte proliferation was inhibited by two of the four different kynurenines: 3HK and 3-hydroxyanthranilic acid (3HAA). This effect was accompanied by significant T-cell death. Neither 3HK nor 3HAA altered dendritic cell function, nor did they induce apoptosis or pathogenicity to corneal endothelial cells. Administration of systemic and topical 3HK to mice receiving a fully mismatched corneal graft resulted in significant prolongation of graft survival (median survival of control grafts, 12 days; of treated, 19 and 15 days, respectively; P < 0.0003). While systemic administration of 3HK was associated with a significant depletion of CD4+ T, CD8+ T, and B lymphocytes in peripheral blood, no depletion was found after topical administration.
Conclusions.
The production of kynurenines, in particular 3HK and 3HAA, may be one mechanism (in addition to tryptophan depletion) by which IDO prolongs graft survival. These molecules have potential as specific agents for preventing allograft rejection in patients at high rejection risk.
Indoleamine 2,3-dioxygenase (IDO) is the rate-limiting enzyme in the catabolism of tryptophan and is mainly expressed by antigen-presenting cells and in the placenta.1,2 It is present at low levels in healthy individuals, but production markedly increases during infection or inflammation, being induced by cytokines, lipopolysaccharide (LPS), or other agents.3–6 Early literature documents the ability of IDO to inhibit the proliferation of microbes and tumor cells in vitro through consumption of the essential amino acid tryptophan.7 In 1998, Munn et al.8 proposed a further role for IDO, suggesting that IDO-dependent suppression of T-cell responses might function as a natural immunoregulatory mechanism, based on data showing that IDO regulates maternal T-cell immunity during pregnancy. So far, physiological IDO activity has been implicated as an effector mechanism for the immunosuppressive reagent CTLA4-Ig fusion protein,9 in T-cell tolerance to tumors,2,10–12 in dysfunctional self-tolerance in nonobese diabetic (NOD) mice,13 as a protective negative regulator in autoimmune disorders,14–16 and as an inhibitor in an induced model of asthma.17
While it is clear that IDO suppresses T-cell responses, the exact mechanism has not been fully elucidated. T cells are particularly sensitive to tryptophan deprivation.18 At low tryptophan concentrations, cell cycle progression is arrested at mid-G1 phase. Restoration of tryptophan to arrested cells, along with a second round of T-cell receptor signaling, reverses the state of nonreactivity and induces cell cycle progression. These and other observations led to the hypothesis that the main mechanism by which IDO inhibits T-cell proliferation is the depletion of tryptophan. However, it is also known that the kynurenines resulting from tryptophan catabolism, which include l-kynurenine (Kyn), 3-hydroxykynurenine (3HK), 3-hydroxyanthranilic acid (3HAA), and quinolinic acid (QA), can themselves inhibit T-cell activation and proliferation.19,20 Kynurenines have proapoptotic properties particularly for activated cells and Th1 lymphocytes,21,22 and the molecular mechanisms of apoptosis have been characterized in murine thymocytes.21 In monocyte/macrophage cell lines, 3HAA can also induce apoptosis by production of hydrogen peroxide.23
The combined effect of tryptophan degradation and increasing concentration of kynurenines has been shown to be responsible for GCN2 kinase–mediated downregulation of the TCRζ-chain in CD8+ cells, reducing their cytotoxic effector function.24 In addition, long-term tryptophan depletion with increased production of tryptophan metabolites promotes conversion of naïve CD4+CD25− T cells into a regulatory phenotype.24 There has only been one report on the effect of local administration of 3HAA and Kyn in a model of skin transplantation, in which prolongation of graft survival by 2 days was found.25
Having previously shown that IDO can be expressed in the cornea during inflammation, including allograft rejection and that overexpression of IDO in murine corneas prolongs allograft survival,26 we examined whether kynurenines can modulate the allogeneic response to a corneal transplant. In this study, we show that systemic 3HK administration results in prolonged corneal graft survival and is associated with a depletion of the circulating lymphocyte count in peripheral blood.
The kynurenine molecule (208 Da) is well below the size of molecule that can penetrate to the corneal stroma.27 Therefore, using these agents as a topical treatment may be a simpler, more effective approach to preventing graft rejection than gene-based therapy. In this study, we demonstrate that topical 3HK administration also results in prolonged corneal graft survival and is associated with moderate depletion of the lymphocyte count in the draining lymph node (DLN).
Materials and Methods
Murine Corneal Endothelial Cells
A murine corneal endothelial cell (MCEC) line, a kind gift from Jerry Niederkorn, (University of Texas Southwestern Medical Center, Dallas, TX), was maintained in DMEM supplemented with 100 U/mL penicillin-streptomycin, nonessential amino acids (Invitrogen-Gibco, Paisley, UK) and 10% heat-inactivated fetal bovine serum (Invitrogen-Gibco).
Generation and Culture of Murine Bone Marrow–Derived Dendritic Cells
The hind limbs, tibia, and femur, were collected in PBS and the BM cavities flushed out to obtain a single cell suspension. The cells were pelleted and red blood cells were lysed with red blood cell (RBC) lysis solution (Flowgen, Nottingham, UK). The cells were pelleted and put in RPMI 1640 medium supplemented with 10% FCS, 100 U/mL penicillin, 100 μg/mL streptomycin, 2 mM l-glutamine, and 50 μM β-mercaptoethanol (Invitrogen) in the presence of a 1:30 dilution of culture medium obtained from a GM-CSF-producing hybridoma.28 DCs were cultured at 37°C with 5% CO2 until day 7. The DC culture medium was replaced with fresh GM-CSF-containing medium on day 3, and half of the culture medium was replaced with fresh GM-CSF-containing medium on day 6. To generate mature DCs, the cultures were stimulated for 16 hours with 1 μg/mL LPS (Sigma-Aldrich, Dorset, UK) on day 7.
DC and MCEC Phenotyping
Immature DCs taken at day 7, mature DCs at day 8, and MCECs were incubated with the individual kynurenines at a concentration of 100 μM for 24 hours. Kyn, 3HK, and 3HAA (Sigma-Aldrich) were dissolved in 1 M HCl as a carrier and QA (Sigma-Aldrich) dissolved in 1 M NaOH. After the cells were washed, they were incubated with 1 μg/μL unconjugated rat anti-mouse and with MHC class II (BD Biosciences-Pharmingen, Oxford, UK), CD80, and CD86 (Invitrogen-Caltag) for DCs; MHC class II, ICOS-L, PDL-1, and ICAM-1 (all BD-Biosciences) for MCECs; and rat anti-mouse IgG2a (R&D Systems, Abingdon, UK) as an isotype control for both, for 30 minutes. After three washes, the cells were incubated with goat anti-rat PE-conjugated secondary antibody (BD Biosciences-Pharmingen) for 30 minutes. Staining was analyzed on a flow cytometer (FACSCalibur; BD Biosciences).
T-Cell Proliferation Assays
Splenocytes from BALB/c mice were treated with a mixture of anti-CD45R/B220, anti-CD8, and anti-MHC class II supernatants (RA3–3A1, M5/114, 53.6.7, and 2.4G2) for 30 minutes. After antibody treatment, the cells were washed and incubated with goat anti-mouse IgG and goat anti-rat IgG-coated beads (Dynal, Bromborough, UK) for 30 minutes, and bound cells were removed with a magnet (CD4 T-cell purity 85%–90%). Responder CD4+ T cells (1 × 105 cells/well) were stimulated with anti-mouse CD3 and CD28 beads (mouse CD3/CD28 T-cell expander; 1 bead/cell; Dynabeads; Dynal) or allogeneic BM-derived C3H (H-2k) DCs at a ratio of 10:1 (1 × 105 CD4+ T cells: 1 × 104 DCs) in the presence of tryptophan metabolites in 96-well plates for 3 days. Proliferation was measured by a 16-hour pulse with [3H]thymidine (Amersham, Buckinghamshire, UK).
Tryptophan Metabolites
The main metabolites resulting from IDO-induced degradation of tryptophan are generally known as kynurenines and comprise N-formyl-kynurenine, kynurenine (Kyn), 3-hydroxykynurenine (3HK), 3-hydroxyanthranilic acid (3HAA), and quinolinic acid (QA). To determine the inhibitory concentration, we tested the tryptophan metabolites in a titration series. Kyn, 3HK, 3HAA, and QA (Sigma-Aldrich) with a purity ≥98% were dissolved in RPMI 1640 medium (Invitrogen-Gibco) and added to the cell cultures in different concentrations ranging from 0 to 100 μM (pH of final cell culture medium, 7.5–8.5).
Detection of Cell Death
CD4+ T cells (1 × 105 cells/well) were stimulated with CD3/CD28 beads (1 bead/cell) in the presence of Kyn, 3HK, 3HAA, or QA (100 μM concentration) individually for 3 days and MCECs for 24 hours. The Cells were then stained with FITC-labeled annexin V and 7-AAD (BD Biosciences), according to the manufacturer's instructions.
Detection of Regulatory T-Cells
CD4+ T cells were activated by CD3/CD28 beads, in low tryptophan (5 μM)–containing medium in combination with 3HK, 3HAA, Kyn, or QA (0–100 μM) for 3 to 7 days. The cells were stained with an APC anti-mouse/rat FoxP3 staining kit (eBioscience, Gloucestershire, UK) after permeabilization and analyzed by flow cytometry.
Calculation of Absolute Cell Counts from Spleen, Peripheral Blood, and DLN
Splenocytes, PBMCs, and lymphocytes (from the DLN) taken from mice after systemic and topical 3HK administration (or controls) were stained with rat anti-mouse CD4-FITC, CD8-peridinin chlorophyll protein (PerCP), and CD19-phycoerythrin (PE) (AbCam, Cambridge, UK). For absolute cell counts, cell counting beads (Perfect-count Microspheres; Cytognos SL, Salamanca, Spain) were used according to the manufacturer's instructions.
RNA Extraction, Reverse Transcription, and Quantitative PCR
Corneal tissue was ground using the beads and reagent supplied in a kit (Fast RNA Pro Green Kit; QBiogene, Carlsbad, CA). RNA extraction and quantification were performed as previously described.29 Quantitative PCR was performed as previously described,26 and FoxP3 mRNA was performed with the paired primers 5′-CACCCAGGAAAGACAGCAACC-3′ (forward) and 5′-GCAAGAGCTCTTGTCCATTGA-3′ (reverse) spanning 314 bp of the FoxP3 gene. Transcripts were normalized to levels of hypoxanthine phosphoribosyl transferase (HPRT) mRNA. Quantification of HPRT mRNA was performed using the paired primers 5′-GTAATGATCCAGTCAACGGGGGAC-3′ (forward) and 5′-CCAGCAAGCTTGCAACCTTAACCA-3′ (reverse).
Corneal Transplantation
Orthotopic corneal transplantation of fully mismatched C3H strain or syngeneic BALB/c donor corneas was undertaken in inbred BALB/c (6–10-week-old) mouse recipients (Harlan Olac UK, Bicester, UK). The animals were maintained in a specific pathogen-free facility and treated in accordance with the U.K. government regulations for care of experimental animals and with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research. Transplantation was performed in the right eye of all animals as originally described,30 with minor modifications.31 Corneal graft transparency was graded on a scale of 0 to 4 as described.31 Rejection was diagnosed when the opacity score reached or exceeded 3 in a previously transparent cornea.
Kynurenine Treatment of Graft Recipients
Stock 3HK was dissolved in PBS and administered intraperitoneally (IP) 560 mg/kg on a daily basis on days 1 to 7, days 7 to 14, or days 1 to 14 after surgery. Control animals were given the same volume of vehicle from days 1 to 14. For topical application, a total dose of 3HK of 0.165 mg was dissolved in 30 μL 0.9% NaCl eye drops (final pH 2.5–3.0) and administered twice daily on days 3 to 14 after surgery after removal of the protective eyelid suture. This was the highest tolerated topical dose, having been tested on nonsurgical eyes of BALB/c mice for 5 days. This dose is a molar equivalent and is based on one third of the total dose used in systemic administration. Control animals were given the same volume of vehicle.
Statistical Analyses
The mean of triplicate values and associated standard deviations were calculated for all T-cell proliferation data. Statistically significant differences were calculated with a two-tailed t-test. Differences in graft survival were analyzed with a log rank test and plotted according to the Kaplan-Meier method. P ≤ 0.05 was regarded as statistically significant.
Results
The Tryptophan Metabolites 3HK and 3HAA Inhibit Murine T-Cell Proliferation In Vitro
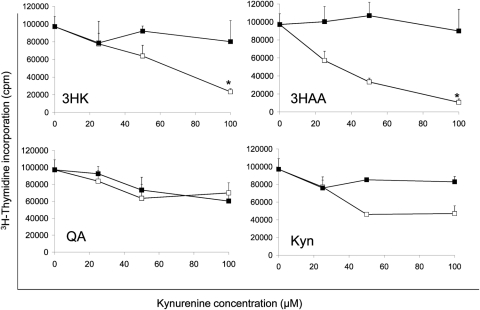
To confirm the effect of tryptophan metabolites on murine allogeneic T-cell responses, we added the metabolites at various concentrations to purified CD4+ T cells that were stimulated with anti-CD3/CD28 beads and proliferation determined using 3H thymidine incorporation. CD4+ T cells were examined as corneal graft rejection is mediated predominantly by this subset.32 As shown in Figure 1, addition of 3HK and 3HAA significantly inhibited CD4+ T-cell proliferation in a dose-dependent manner with IC50 of approximately 70 and 30 μM, respectively, when compared with T cells incubated in vehicle alone. A minimal effect with Kyn was observed, and no effect was seen with QA. Similar data were obtained when the T cells were stimulated with allogeneic dendritic cells in a mixed lymphocyte response (Supplementary Fig. S1, http://www.iovs.org/lookup/suppl/doi:10.1167/iovs.10-5793/-/DCSupplemental).
Figure 1.
Effect of kynurenines on T-cell proliferation. CD3/CD28 bead–stimulated CD4+ T cells were incubated for 72 hours in the presence of kynurenines (3HK, 3HAA, Kyn, and QA) at concentrations varying from 0 to 100 μM. Proliferation was measured by 3H-thymidine incorporation. (■) Vehicle control; (□) kynurenine. The data represent the mean ± SD of triplicate cultures. Results shown are representative of four experiments. *P < 0.005, versus the control.
3HK and 3HAA Induce CD4+ T-Cell Death
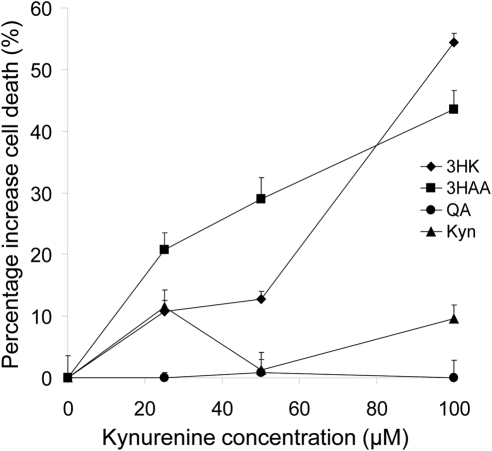
There are several possible mechanisms by which 3HK and 3HAA may influence T-cell responses. These include the induction of T-cell death or the induction of T regulatory cells (Tregs). To investigate cell death, we stimulated CD4+ T cells with anti-CD3/CD28 beads in the presence of the kynurenines (0–100 μM) for 24 hours and cell death assessed by flow cytometry after staining with 7-AAD and annexin V. Both 3HK and 3HAA induced significant CD4+ T-cell-mediated cell death in a dose-dependent manner, whereas neither QA nor Kyn did (Fig. 2). Furthermore, as previously reported,21 we found 3HAA to be more potent than 3HK; inducing apoptosis at concentrations as low as 25 μM compared with 100 μM for 3HK.
Figure 2.
Effect of kynurenines on T-cell death. CD3/CD28 bead activated CD4+ T cells were incubated with or without kynurenines for 72 hours. Cytotoxicity was evaluated by flow cytometry after vital staining with 7AAD. Data represent the mean ± SD of triplicate samples. Results shown are representative of three experiments.
Tryptophan Metabolites Induce a T-Regulatory-Cell Phenotype from the Whole CD4+ T-Cell Population
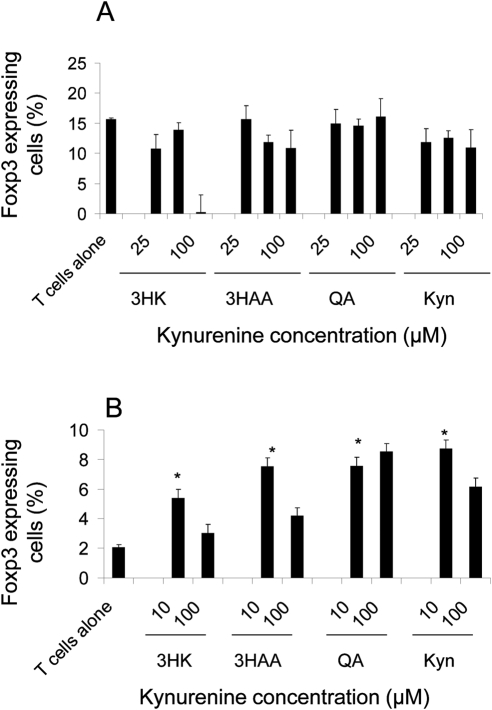
Expression of FoxP3 correlates with suppressor activity in CD4+ T cells.33 Fallarino et al.34 demonstrated that naïve CD4+ FoxP3− T cells, which are resistant to kynurenine-induced apoptosis, can convert into CD4+FoxP3+ cells by a GCN2-dependent mechanism after incubation with kynurenines.34 We looked at the effect of the kynurenines on the number of FoxP3+ cells from the whole CD4+ T-cell population. CD4+ T cells were stimulated with anti-CD3/CD28 beads in the presence of the kynurenines (10–100 μM) for 3 and 7 days. As shown in Figure 3A, there was no difference in the proportion of CD4+ cells expressing FoxP3 in the presence of any of the kynurenines after 3 days. However, after 7 days (Fig. 3B), there was a significant increase in the proportion of CD4+ cells expressing FoxP3 in the presence of all the kynurenines at the 10-μM concentration, flow histograms of 3HK and 3HAA data are also shown (Supplementary Fig. S2, http://www.iovs.org/lookup/suppl/doi:10.1167/iovs.10-5793/-/DCSupplemental). This suggests that lower concentrations of kynurenines are necessary to induce T-regulatory cells (Tregs), in keeping with earlier reports.34 Of note, at the 100-μM concentration, there was a reduction in the percentage of FoxP3-expressing cells after both 3 and 7 days in the presence of 3HK and 3HAA, but not QA or Kyn. The possible reasons for this are discussed below.
Figure 3.
FoxP3 expression in CD4+ T cells cultured with kynurenines. CD4+ T cells were activated with CD3/CD28 beads, with or without kynurenines. At 3 (A) and 7 (B) days, the cells were recovered and the percentage of CD4+ T cells that were FoxP3+ was quantified by intracellular flow cytometry. Data represent the mean ± SD of triplicate samples. *P ≤ 0.005, experimental versus T cells alone.
Tryptophan Metabolites Do Not Alter DC Function or Phenotype
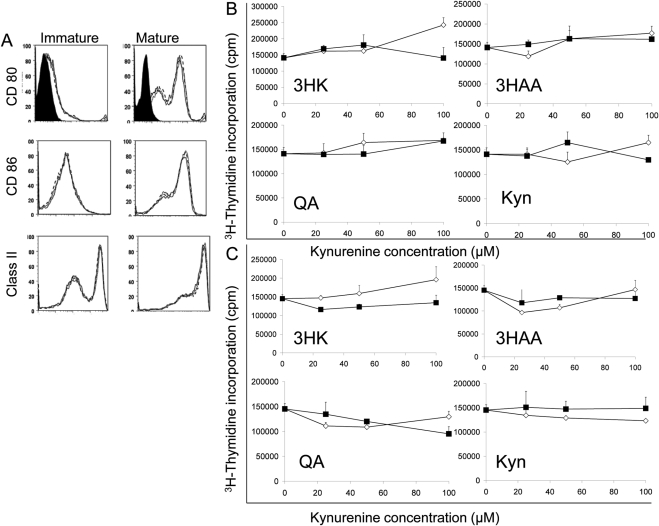
We speculated that kynurenines could modify DC biology in an autocrine manner. We assessed the effect of the individual kynurenines on both immature and mature DCs by co-incubation for 24 hours, followed by washing. The DCs were then used in mixed lymphocyte reactions to investigate their ability to stimulate CD4+ T cells; stained for CD80, CD86, and class II cell surface markers to identify phenotype; and assessed for viability by flow cytometry after vital staining with 7-AAD. We showed that incubation with kynurenines did not alter the phenotype of either immature or mature DCs (data shown for 3HK only; Fig. 4A), did not alter their ability to stimulate CD4+ T cells (Figs. 4B, 4C), and had no effect on their viability (data not shown). Similarly, kynurenines were not pathogenic to corneal endothelial cells and did not alter phenotype or viability (Supplementary Figs. S3, S4, http://www.iovs.org/lookup/suppl/doi:10.1167/iovs.10-5793/-/DCSupplemental).
Figure 4.
Effect of kynurenines on function and phenotype of DCs. (A) Mature and immature bone marrow–derived DCs were incubated with 3HK at a concentration of 100 μM for 24 hours, and expression of CD80, CD86, and MHC class II expression was quantified by flow cytometry (solid line: 3HK; dashed line: vehicle control; dotted line: DCs alone). The isotype control is represented by the solid profile. Mature (B) and immature (C) bone marrow-derived DCs were incubated with the individual kynurenines (□) as well as their vehicle controls (■) at concentrations ranging from 0 to 100 μM for 24 hours, subsequently washed, and then used to stimulate allogeneic CD4+ T cells in a mixed lymphocyte reaction and proliferation measured by [3H]thymidine incorporation. Data represent the mean ± SD of triplicate cultures. Results shown are representative of three experiments.
Systemic Administration of 3HK Prolongs Murine Corneal Allograft Survival
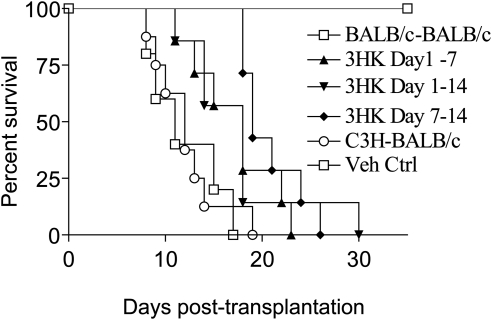
Given the ability of 3HK and 3HAA to suppress allogeneic T-cell responses in vitro, we went on to determine whether administration of 3HK resulted in prolonged C3H corneal allograft survival in BALB/c recipients, in which strain combination there is full MHC and multiple minor mismatches. 3HK was administered at 560 mg/kg IP on a daily basis on days 1 to 7, days 7 to 14, or days 1 to 14. This dose was chosen, as in preliminary experiments higher doses resulted in an observed lack of activity in treated animals. The early (days 1–7) and late (days 7–14) administration were chosen, to compare the effect of 3HK on the induction and effector phases of the allogeneic response. Control animals were given the same volume of vehicle from days 1 to 14. Median survival time (MST) of allogeneic C3H corneas was 12 days in untreated BALB/c recipients, whereas syngeneic corneas survived indefinitely and administration of the vehicle had no effect on survival (MST, 11 days). 3HK administration resulted in significant prolongation of graft survival whether administered between days 1 to 7 (MST, 18 days; P < 0.036), days 7 to 14 (MST 19 days; P < 0.0003), or days 1 to 14 (MST 18 days; P < 0.0037). There was no significant difference between the 3HK-treated groups (Fig. 5).
Figure 5.
Effect of systemic administration of 3HK on murine corneal allograft survival. BALB/c (H2d) mice received unilateral C3H (H2k) donor corneal allografts, and IP injections of 3HK were administered at a dose of 560 mg/kg/d according to three different treatment regimens: on days 1 to 7, 1 to 14, and 7 to 14 after transplantation. There was significant prolongation of graft survival in all treatment groups; n = 7 (MST, 18, 18, and 19; P < 0.036, P < 0.0003, and P < 0.0037, respectively) in comparison to the controls (MST, 11 days). Indefinite survival was observed in the untreated syngeneic grafts (BALB/c-BALB/c).
Systemic Administration of 3HK Depletes Both Splenic and Peripheral Blood Lymphocytes
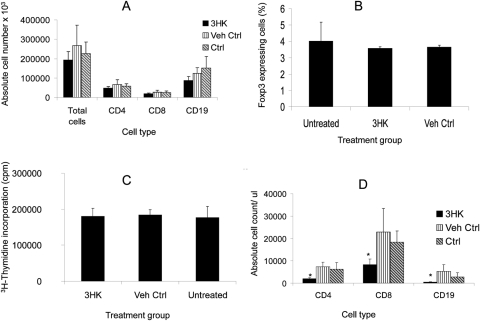
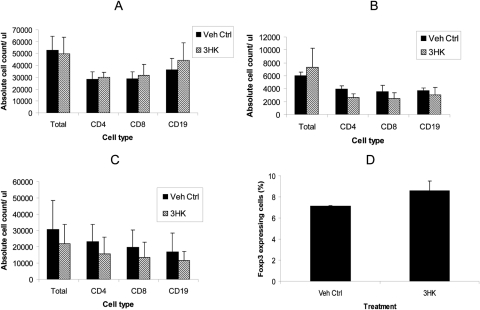
To determine whether 3HK alters the number of lymphocytes, we administered 3HK for 14 days (according to the same regimen) to animals that did not receive a graft. This treatment did not result in any significant change in the weight of the spleen (data not shown), but did result in a statistically insignificant reduction in the number of splenic CD4+ and CD8+ T cells and CD19+ B cells (Fig. 6A); this was a consistent observation in repeated experiments. There was no alteration in the proportion of splenic CD4+ cells expressing FoxP3 (Fig. 6B). Furthermore, it did not alter the proliferative capacity of CD4+ T cells after stimulation with CD3/CD28 beads (Fig. 6C). There was a significant reduction in the number of CD4+ and CD8+ T cells and CD19+ B cells in peripheral blood (Fig. 6D).
Figure 6.
Splenic and peripheral blood analysis after daily IP administration of 3HK. Animals (n = 3) received systemic IP 3HK 560 mg/kg/d for 14 days consecutively, after which the spleens were removed and (A) absolute counts of CD4, CD8, and CD19 assessed by using counting beads. (B) The cells were stained intracellularly for Fox P3. (C) CD4+ T cells isolated and subsequently stimulated with CD3/CD28 beads and proliferation assessed by 3H-thymidine incorporation. (D) Peripheral blood was taken from the tail vein, and the absolute number of CD4, CD8, and CD19 cells was assessed by using counting beads. *P ≤ 0.05, experimental versus vehicle control. Data represent the mean ± SD of triplicate counts. Results shown are representative of two experiments.
Topical Administration of 3HK Results in Prolongation of Corneal Allograft Survival
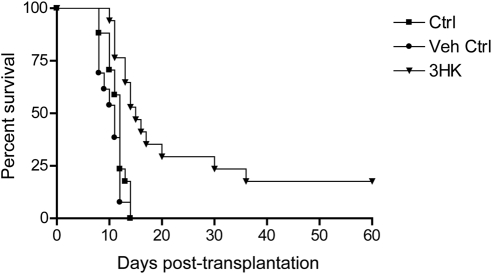
We went on to determine whether topical administration might prolong graft survival. 3HK (0.165 mg) was administered twice daily on days 3 to 14 after transplantation. Control animals were given the same volume of vehicle. Treated animals had a significant prolongation in graft survival (MST, 15 days; P < 0.0001) compared with untreated or vehicle alone (MST, 12; 11 days). In addition, a minority of animals (3/15) showed long-term survival (>50 days; Fig. 7).
Figure 7.
Effect of daily topical administration of 3HK on murine corneal allograft survival. BALB/c (H2d) recipients of unilateral C3H (H2k) donor corneal allografts were treated with topical 3HK or vehicle in the surgical eye twice daily on days 3 to 14 after surgery. There was a significant prolongation of graft survival in the 3HK treated group; n = 15 (MST, 15 days; P < 0.0001) in comparison to controls (MST, 11 days).
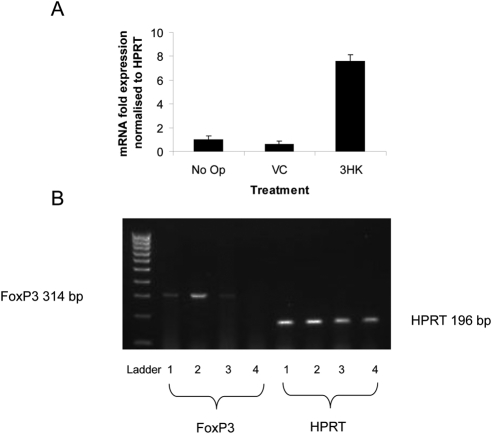
The corneal grafts of mice that had received topical 3HK treatment, as well as those of the controls, were excised after the onset of rejection (n = 8/group), and RNA was extracted. Corneal FoxP3 mRNA expression was subsequently assessed by quantitative real-time RT-PCR. There was a marked upregulation of FoxP3 expression in 3HK versus vehicle control–treated corneas (Fig. 8).
Figure 8.
FoxP3 mRNA expression in rejected corneal grafts. The corneal grafts of mice that were not operated on (No Op) and those that had received an allograft together with topical vehicle control (VC) or 3HK treatment were excised after the onset of rejection (n = 8/group) and RNA extracted. Corneal FoxP3 mRNA expression was subsequently assessed by quantitative, real-time RT-PCR. (A) Bar graph showing corneal FoxP3 mRNA expression normalized to HPRT, as assessed using qRT-PCR. Data represents mean ± SD of triplicate samples. (B) Separation of PCR products on 1.5% agarose gel. (1, VC; 2, 3HK; 3, Op-no treatment; 4, No Op).
In mice that did not receive a graft, topical administration of 3HK did not alter the splenic or peripheral blood lymphocyte count (Figs. 9A, 9B), but did result in a moderate, albeit insignificant generalized reduction in the lymphocyte count of the DLNs (Fig. 9C). The DLNs assessed were the deep cervical and submandibular nodes, which have been shown to be the ocular DLNs.35 There was minimal difference in the proportion of CD4+ cells expressing Fox P3 in the DLNs (Fig. 9D).
Figure 9.
Splenic, peripheral blood, and draining lymph node analysis after daily topical administration of 3HK. Animals (n = 3) received topical 3HK for 12 consecutive days, after which the spleens were removed (A), peripheral blood was taken (B), and lymphocytes from the DLNs collected and absolute counts of CD4, CD8, and CD19 assessed by using counting beads (C). Lymphocytes from the DLNs were also stained intracellularly for FoxP3 (D). Data represent the mean ± SD of triplicate counts.*P = 0.01, treatment versus vehicle control.
Discussion
Ever since it was reported that IDO can prevent rejection of the fetus during pregnancy in mice,8 there has been considerable interest in elucidating the role of this enzyme in immunoregulation. In the context of transplantation, tryptophan catabolism has been shown to suppress alloimmune responses. IDO-expressing DCs suppress allogeneic T-cell proliferation in vitro via tryptophan metabolites,19 and the latter inhibit allogeneic T-cell responses both in vitro and in vivo.25 There has been no investigation, however, as to how tryptophan metabolites exert their in vivo effect. In this study, we have investigated whether the tryptophan metabolites given either locally or systemically can modulate corneal allograft survival.
We confirmed that 3HK and 3HAA suppressed CD4+ T-cell proliferation in vitro, related to death of the T cells rather than Treg development. Previously, Fallarino et al.24,34 showed a large increase in the proportion of naïve CD4+ FoxP3 expressing cells in the presence of the kynurenines (70% vs. 2% controls), we showed only a moderate increase (8% vs. 2% controls) in the whole CD4+ T-cell population. This result may relate to differences in the populations studied. It is recognized that naïve T cells are more resistant to kynurenine-induced apoptosis than are effector CD4+ T cells.21 We found a reduction in FoxP3 expression in CD4+ T cells at the higher concentrations of 3HK and 3HAA after 3 and 7 days of incubation. This decrease could be because Tregs are more sensitive or it may be an indirect effect as, for example, it has been shown that CD4+CD25+ FoxP3+ T cells are extremely sensitive to two common cell metabolites that are released from necrotic cells: NAD and ATP.36 However, given the kinetics of Treg development, they will not contribute significantly to the inhibition of T-cell proliferation seen in vitro, where the major mechanism of action is the death of the responding cells.
Consistent with previous reports, we demonstrated that DCs are resistant to kynurenine-induced apoptosis.19,21 Taking this further, we showed that the function and phenotype of DCs is not altered in the presence of kynurenines. It is interesting to speculate why DCs are resistant to kynurenines. It may be the result of cell-specific uptake mechanisms or differential expression of enzymatic breakdown pathways.20,37 It is known that DCs metabolize tryptophan through the kynurenine pathway, as demonstrated by immunohistochemical staining of the presence of quinolinic acid, the penultimate product of the kynurenine pathway.38 Furthermore, it has been shown that 3HAA-induced T-cell death is as a result of inhibition of NF-κB activation on T-cell receptor engagement by specifically targeting PDK1.39 The same investigators showed that the induction of apoptosis is specific to CD4+ T cells, as 3HAA did not inhibit NF-κB activation or induce cell death on Toll-like receptor-4 stimulation in dendritic cells.39 We also demonstrated that there were no toxic effects of either IDO or kynurenines on corneal endothelial cells, in keeping with findings demonstrating IDO to be part of the ocular immune privilege.26,40 Furthermore, it has been recently demonstrated that a tryptophan/kynurenine transporter protein exchange mechanism (LAT1) is upregulated by inflammatory cytokines in human corneal endothelial cells. This transporter would allow rapid delivery of the cytotoxic tryptophan catabolites to the local corneal environment, dampening T-cell responses,41 again suggesting a probable role for kynurenines in corneal immune privilege.
Earlier reports on the effect of SC administration of 3HAA and Kyn in a model of skin transplantation indicated a short prolongation of graft survival.25 For the first time we demonstrate that both systemic and topical administration of 3HK resulted in prolongation of graft survival. In the systemic group, we administered 3HK IP in three different regimens. Although we observed a longer median graft survival in the day 7- to 14-treated group, there was no significant difference between the three treatment regimens. Despite concern about administration of 3HK in vivo owing to its neurotoxic potential,42,43 we did not observe any major effect at the doses we administered. Furthermore, the systemic doses administered (60 mg/L) are only 20 times higher than the serum concentration of Kyn in rats or humans (3 mg/L).44 The longest survival of individual grafts (>50 days) was observed in the topical group in comparison to 30 days in the systemic group. Direct application of drops to the graft-recipient eye offer a more targeted treatment, reducing possible systemic adverse effects.
To determine the effect of 3HK on lymphocyte counts we examined mice that had had systemic administration of 3HK for 14 days, finding a moderate although not statistically significant reduction in the CD4+, CD8+ and B-lymphocyte population of the spleen but a significant reduction in peripheral blood. This loss of lymphocytes could be as a result of cell death, consistent with our in vitro findings, or due to a redistribution of circulating lymphocytes to the bone marrow and/or transient arrest of recirculation, as has been observed with corticosteroid treatment.45,46
While there is a small decrease in the number of cells, it is not clear that such a small decrease would be responsible for the prolongation of graft survival. However, the topical administration of 3HK resulted in a significant upregulation of corneal FoxP3 mRNA expression in comparison to controls, suggesting that regulatory pathways may therefore be involved. Although significant prolongation in graft survival was observed in both the topically and systemically treated groups after a treatment duration of 14 days after transplantation, longer duration treatment might have resulted in longer term acceptance of grafts, as in other reported treatment studies in this mouse model.47
In conclusion, our data show that 3HK and 3HAA induce mouse T-cell suppression in vitro, primarily as a result of death of the T cells, and that 3HK prolongs corneal allograft survival whether administered by topical or systemic routes. These data show that these molecules may be responsible in part for the effect of IDO in preventing allograft rejection (although they cannot determine the relative importance of kynurenine production compared with tryptophan reduction). In addition, we have demonstrated the potential for using kynurenine or kynurenine-like molecules to prevent graft rejection.
Supplementary Material
Footnotes
Supported by a project grant and research training fellowship (SSZ) from Fight for Sight, and by Wellcome Trust Project Grant 078732/Z/05/Z.
Disclosure: S.S. Zaher, None; C. Germain, None; H. Fu, None; D.F.P. Larkin, None; A.J.T. George, None
References
- 1. Yoshida R, Nukiwa T, Watanabe Y, Fujiwara M, Hirata F, Hayaishi O. Regulation of indoleamine 2,3-dioxygenase activity in the small intestine and the epididymis of mice. Arch Biochem Biophys. 1980;203(1):343–351 [DOI] [PubMed] [Google Scholar]
- 2. Fallarino F, Vacca C, Orabona C, et al. Functional expression of indoleamine 2,3-dioxygenase by murine CD8 alpha(+) dendritic cells. Int Immunol. 2002;14(1):65–68 [DOI] [PubMed] [Google Scholar]
- 3. Yoshida R, Urade Y, Tokuda M, Hayaishi O. Induction of indoleamine 2,3-dioxygenase in mouse lung during virus infection. Proc Natl Acad Sci U S A. 1979;76(8):4084–4086 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Yoshida R, Hayaishi O. Induction of pulmonary indoleamine 2,3-dioxygenase by intraperitoneal injection of bacterial lipopolysaccharide. Proc Natl Acad Sci U S A. 1978;75(8):3998–4000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Carlin JM, Borden EC, Sondel PM, Byrne GI. Biologic-response-modifier-induced indoleamine 2,3-dioxygenase activity in human peripheral blood mononuclear cell cultures. J Immunol. 1987;139(7):2414–2418 [PubMed] [Google Scholar]
- 6. Bianchi M, Bertini R, Ghezzi P. Induction of indoleamine dioxygenase by interferon in mice: a study with different recombinant interferons and various cytokines. Biochem Biophys Res Commun. 1988;152(1):237–242 [DOI] [PubMed] [Google Scholar]
- 7. Taylor MW, Feng GS. Relationship between interferon-gamma, indoleamine 2,3-dioxygenase, and tryptophan catabolism. FASEB J. 1991;5(11):2516–2522 [PubMed] [Google Scholar]
- 8. Munn DH, Zhou M, Attwood JT, et al. Prevention of allogeneic fetal rejection by tryptophan catabolism. Science. 1998;281(5380):1191–1193 [DOI] [PubMed] [Google Scholar]
- 9. Grohmann U, Orabona C, Fallarino F, et al. CTLA-4-Ig regulates tryptophan catabolism in vivo. Nat Immunol. 2002;3(11):1097–1101 [DOI] [PubMed] [Google Scholar]
- 10. Friberg M, Jennings R, Alsarraj M, et al. Indoleamine 2,3-dioxygenase contributes to tumor cell evasion of T cell-mediated rejection. Int J Cancer. 2002;101(2):151–155 [DOI] [PubMed] [Google Scholar]
- 11. Uyttenhove C, Pilotte L, Theate I, et al. Evidence for a tumoral immune resistance mechanism based on tryptophan degradation by indoleamine 2,3-dioxygenase. Nat Med. 2003;9(10):1269–1274 [DOI] [PubMed] [Google Scholar]
- 12. Munn DH, Sharma MD, Hou D, et al. Expression of indoleamine 2,3-dioxygenase by plasmacytoid dendritic cells in tumor-draining lymph nodes. J Clin Invest. 2004;114(2):280–290 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Grohmann U, Fallarino F, Bianchi R, et al. A defect in tryptophan catabolism impairs tolerance in nonobese diabetic mice. J Exp Med. 2003;198(1):153–160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Sakurai K, Zou JP, Tschetter JR, Ward JM, Shearer GM. Effect of indoleamine 2,3-dioxygenase on induction of experimental autoimmune encephalomyelitis. J Neuroimmunol. 2002;129(1–2):186–196 [DOI] [PubMed] [Google Scholar]
- 15. Gurtner GJ, Newberry RD, Schloemann SR, McDonald KG, Stenson WF. Inhibition of indoleamine 2,3-dioxygenase augments trinitrobenzene sulfonic acid colitis in mice. Gastroenterology. 2003;125(6):1762–1773 [DOI] [PubMed] [Google Scholar]
- 16. Swanson KA, Zheng Y, Heidler KM, Mizobuchi T, Wilkes DS. CDllc+ cells modulate pulmonary immune responses by production of indoleamine 2,3-dioxygenase. Am J Respir Cell Mol Biol. 2004;30(3):311–318 [DOI] [PubMed] [Google Scholar]
- 17. Hayashi T, Beck L, Rossetto C, et al. Inhibition of experimental asthma by indoleamine 2,3-dioxygenase. J Clin Invest. 2004;114(2):270–279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Munn DH, Shafizadeh E, Attwood JT, Bondarev I, Pashine A, Mellor AL. Inhibition of T cell proliferation by macrophage tryptophan catabolism. J Exp Med. 1999;189(9):1363–1372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Terness P, Bauer TM, Rose L, et al. Inhibition of allogeneic T cell proliferation by indoleamine 2,3-dioxygenase-expressing dendritic cells: mediation of suppression by tryptophan metabolites. J Exp Med. 2002;196(4):447–457 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Frumento G, Rotondo R, Tonetti M, Damonte G, Benatti U, Ferrara GB. Tryptophan-derived catabolites are responsible for inhibition of T and natural killer cell proliferation induced by indoleamine 2,3-dioxygenase. J Exp Med. 2002;196(4):459–468 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Fallarino F, Grohmann U, Vacca C, et al. T cell apoptosis by tryptophan catabolism. Cell Death Differ. 2002;9(10):1069–1077 [DOI] [PubMed] [Google Scholar]
- 22. Fallarino F, Grohmann U, Vacca C, et al. T cell apoptosis by kynurenines. Adv Exp Med Biol. 2003;527:183–190 [DOI] [PubMed] [Google Scholar]
- 23. Morita T, Saito K, Takemura M, et al. 3-Hydroxyanthranilic acid, an L-tryptophan metabolite, induces apoptosis in monocyte-derived cells stimulated by interferon-gamma. Ann Clin Biochem. 2001;38:242–251 [DOI] [PubMed] [Google Scholar]
- 24. Fallarino F, Grohmann U, You S, et al. The combined effects of tryptophan starvation and tryptophan catabolites down-regulate T cell receptor zeta-chain and induce a regulatory phenotype in naive T cells. J Immunol. 2006;176(11):6752–6761 [DOI] [PubMed] [Google Scholar]
- 25. Bauer TM, Jiga LP, Chuang JJ, Randazzo M, Opelz G, Terness P. Studying the immunosuppressive role of indoleamine 2,3-dioxygenase: tryptophan metabolites suppress rat allogeneic T-cell responses in vitro and in vivo. Transpl Int. 2005;18(1):95–100 [DOI] [PubMed] [Google Scholar]
- 26. Beutelspacher SC, Pillai R, Watson MP, et al. Function of indoleamine 2,3-dioxygenase in corneal allograft rejection and prolongation of allograft survival by over-expression. Eur J Immunol. 2006;36(3):690–700 [DOI] [PubMed] [Google Scholar]
- 27. Brereton HM, Taylor SD, Farrall A, et al. Influence of format on in vitro penetration of antibody fragments through porcine cornea. Br J Ophthalmol. 2005;89(9):1205–1219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Karasuyama H, Kudo A, Melchers F. The proteins encoded by the VpreB and lambda 5 pre-B cell-specific genes can associate with each other and with mu heavy chain. J Exp Med. 1990;172(3):969–972 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. King WJ, Comer RM, Hudde T, Larkin DF, George AJ. Cytokine and chemokine expression kinetics after corneal transplantation. Transplantation. 2000;70(8):1225–1233 [DOI] [PubMed] [Google Scholar]
- 30. Zhang EP, Schrunder S, Hoffmann F. Orthotopic corneal transplantation in the mouse: a new surgical technique with minimal endothelial cell loss. Graefes Arch Clin Exp Ophthalmol. 1996;234(11):714–719 [DOI] [PubMed] [Google Scholar]
- 31. Ardjomand N, Berghold A, Komericki P, Reich ME. The significance of tissue storage time for success after corneal transplantation (in German). Ophthalmologe. 1997;94(4):285–289 [DOI] [PubMed] [Google Scholar]
- 32. Niederkorn JY. Mechanisms of corneal graft rejection: the sixth annual Thygeson Lecture, presented at the Ocular Microbiology and Immunology Group meeting, October 21, 2000. Cornea. 2001;20(7):675–679 [DOI] [PubMed] [Google Scholar]
- 33. Fontenot JD, Rasmussen JP, Williams LM, Dooley JL, Farr AG, Rudensky AY. Regulatory T cell lineage specification by the forkhead transcription factor foxp3. Immunity. 2005;22(3):329–341 [DOI] [PubMed] [Google Scholar]
- 34. Fallarino F, Grohmann U, You S, et al. Tryptophan catabolism generates autoimmune-preventive regulatory T cells. Transpl Immunol. 2006;17(1):58–60 [DOI] [PubMed] [Google Scholar]
- 35. Plskova J, Holan V, Filipec M, Forrester JV. Lymph node removal enhances corneal graft survival in mice at high risk of rejection. BMC Ophthalmol. 2004;4:3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Aswad F, Kawamura H, Dennert G. High sensitivity of CD4+CD25+ regulatory T cells to extracellular metabolites nicotinamide adenine dinucleotide and ATP: a role for P2X7 receptors. J Immunol. 2005;175(5):3075–3083 [DOI] [PubMed] [Google Scholar]
- 37. Moffett JR, Namboodiri MA. Tryptophan and the immune response. Immunol Cell Biol. 2003;81(4):247–265 [DOI] [PubMed] [Google Scholar]
- 38. Hill M, Tanguy-Royer S, Royer P, et al. IDO expands human CD4+CD25high regulatory T cells by promoting maturation of LPS-treated dendritic cells. Eur J Immunol. 2007;37(11):3054–362 [DOI] [PubMed] [Google Scholar]
- 39. Hayashi T, Mo JH, Gong X, et al. 3-Hydroxyanthranilic acid inhibits PDK1 activation and suppresses experimental asthma by inducing T cell apoptosis. Proc Natl Acad Sci U S A. 2007;104(47):18619–18624 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Chen X, Liu L, Yang P, et al. Indoleamine 2,3-dioxygenase (IDO) is involved in promoting the development of anterior chamber-associated immune deviation. Immunol Lett. 2006;107(2):140–147 [DOI] [PubMed] [Google Scholar]
- 41. Serbecic N, Lahdou I, Scheuerle A, Hoftberger R, Aboul-Enein F. Function of the tryptophan metabolite, L-kynurenine, in human corneal endothelial cells. Mol Vis. 2009;15:1312–1324 [PMC free article] [PubMed] [Google Scholar]
- 42. Freese A, Swartz KJ, During MJ, Martin JB. Kynurenine metabolites of tryptophan: implications for neurologic diseases. Neurology. 1990;40(4):691–695 [DOI] [PubMed] [Google Scholar]
- 43. Moroni F. Tryptophan metabolism and brain function: focus on kynurenine and other indole metabolites. Eur J Pharmacol. 1999;375(1–3):87–100 [DOI] [PubMed] [Google Scholar]
- 44. Chavele KM, Shukla D, Keteepe-Arachi T, et al. Regulation of MPO-specific T cell responses during disease remission in ANCA-associated vasculitis: the role of Tregs, and tryptophan degradation. Arthritis Rheum. 2010;62(5):1539–1548 [DOI] [PubMed] [Google Scholar]
- 45. Yu DT, Clements PJ, Paulus HE, Peter JB, Levy J, Barnett EV. Human lymphocyte subpopulations: effect of corticosteroids. J Clin Invest. 1974;53(2):565–571 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Fauci AS. Mechanisms of corticosteroid action on lymphocyte subpopulations, I: redistribution of circulating T and b lymphocytes to the bone marrow. Immunology. 1975;28(4):669–680 [PMC free article] [PubMed] [Google Scholar]
- 47. Zhang EP, Schulte F, Bulfone-Paus S, Hoffmann F. The effect of corticosteroid and cyclosporin A on murine corneal allograft rejection. Graefes Arch Clin Exp Ophthalmol. 2000;238(6):525–530 [DOI] [PubMed] [Google Scholar]
- 48. Sagoo P, Chan G, Larkin DF, George AJ. Inflammatory cytokines induce apoptosis of corneal endothelium through nitric oxide. Invest Ophthalmol Vis Sci. 2004;45(11):3964–3973 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.