Abstract
Objective
The variant alleles in the mannose binding lectin-2 (MBL-2) gene have been associated with MBL deficiency and increased susceptibility to sepsis. We postulate that the variant MBL-2 genotypes are associated with increased susceptibility to and mortality in acute respiratory distress syndrome (ARDS).
Design
Nested case-control study.
Setting
Tertiary academic medical center.
Patients
Two hundred and twelve Caucasians with ARDS and 442 controls genotyped for the variant X, D, B, and C alleles of codon -221, 52, 54, and 57, respectively.
Interventions
None.
Measurements and Main Results
Patients homozygous for the variant codon 54B allele (54BB) had worse severity of illness on admission (p = .007), greater likelihood of septic shock (p = .04), and increased odds of ARDS (adjusted odds ratio, 6.7; 95% confidence interval, 1.5-31) when compared with heterozygotes and homozygotes for the wild-type allele. This association with ARDS was especially strong among the 311 patients with septic shock (adjusted odds ratio, 12.0; 95% confidence interval, 1.9-74). Among the patients with ARDS, the 54BB genotype was associated with more daily organ dysfunction (p = .01) and higher mortality (adjusted hazard rate, 4.0; 95% confidence interval, 1.6-10). Development of ARDS and outcomes in ARDS did not vary significantly with variant alleles of codon -221, 52, and 57, but the power to detect an effect was limited secondary to the low allele frequencies.
Conclusions
The MBL-2 codon 54BB genotype may be important in ARDS susceptibility and outcome. Additional studies are needed to confirm these findings in other populations.
Keywords: acute respiratory failure, genetic susceptibility, acute lung injury, molecular epidemiology
Although clinical predictors for the development to acute respiratory distress syndrome (ARDS) are well recognized, a minority of patients with these risks develop ARDS (1). Genetic susceptibility to acute lung injury may explain the observed interindividual differences in risk and in outcomes (2-4).
Mannose binding lectin (MBL) is a member of the collectin family important in the initiation of the lectin pathway of complement activation and opsonin-induced phagocytosis (5-7). The MBL protein is encoded by the mannose binding lectin-2 (MBL-2) gene on chromosome 10. It is now known that circulating MBL levels are due largely to three single nucleotide polymorphisms (SNPs) in codon 52 (db SNP ID rs5030737), 54 (db SNP ID rs1800450), and 57 (db SNP ID rs1800451) in exon 1 and a promoter SNP at codon -221 (MBLXY; db SNP ID rs7096206) (7). The variant exon 1 alleles are known as D, B, and C, respectively, whereas the wild-type alleles are known collectively as the A allele. The variant alleles in exon 1 and the X allele in the MBLXY polymorphism have been found to be associated with serum MBL deficiency especially in individuals homozygous for the variant alleles (8, 9).
In clinical studies, variant MBL-2 alleles have been associated with increased susceptibility to meningococcemia (10), invasive pneumococcal infection (11), hepatitis B (12, 13), severe acute respiratory syndrome (14), and other infections (8, 9, 15-17). In critical illnesses, two small studies have found an association between the variant MBL-2 alleles and increased incidence of systemic inflammatory syndrome from both infectious and noninfectious causes, increased severity of sepsis, and/or increased mortality in sepsis (18, 19). In both studies, the variant alleles were associated with serum MBL deficiency with the lowest levels found among those patients who were homozygous for the variant alleles.
Genes that are important in sepsis are likely to be relevant in ARDS because of the many common links between sepsis and ARDS. Sepsis is the leading cause of ARDS (1). Most patients who die from ARDS die of refractory infection and sepsis, not from respiratory failure (20-22).
We describe a nested case-control study of patients at risk for ARDS. We hypothesized that the X allele of the MBLXY polymorphism and the variant D, B, and C alleles of the codon 52, 54, and 57 in the MBL-2 gene are associated with increased susceptibility to and increased mortality in ARDS.
MATERIALS AND METHODS
Study Population and Design
A schematic summary of the study design is illustrated in Figure 1. Details of the study have been described previously (23). Briefly, all admissions to the intensive care units (ICU) of the Massachusetts General Hospital (Boston, MA) were screened daily for study-defined clinical risk factor for ARDS as detailed in Table 1 (2, 23). Exclusion criteria included age <18, diffuse alveolar hemorrhage, chronic lung diseases, directive to withhold intubation, immunosuppression except if secondary to corticosteroid, and treatment with granulocyte colony-stimulating factor. ICU admissions with one or more defined risks for ARDS and no exclusion criteria were eligible for the study. Enrolled patients were screened daily for the primary outcome of ARDS as defined by respiratory failure requiring intubation and fulfillment of American-European Consensus Conference criteria for ARDS as follows (23, 24): a) presence of hypoxemia as evidenced by PaO2/FIO2 ≤200 mm Hg; b) presence of bilateral infiltrates on chest radiographs; and c) absence of left atrial hypertension as evidenced by pulmonary arterial occlusion pressure ≤18 mm Hg or lack of notation for congestive heart failure as a problem in the progress note.
Figure 1.
Flow diagram of study design and patient selection for case-control study. ICU, intensive care unit; ARDS, acute respiratory distress syndrome.
Table 1.
Study required risk factors for acute respiratory distress on admission to intensive care unit
| Sepsis: As defined by the Society of Critical Care Medicine (42) to be a known or suspected source of systemic infection and at least two of the following: a) temperature >38°C or <36°C; b) heart rate >90 beats/min; c) respiratory rate >20 breaths/min or PaCO2 <32 mm Hg; d) WBC count >12,000/mm3, <4000/mm3, or >10% bands |
| Septic shock: Fulfill requirements for sepsis and one of the following: a) SBP <90 mm Hg or reduction of ≥40 mm Hg from baseline for ≥30 mins unresponsive to 500 mL of fluid resuscitation; b) need for vasopressors to maintain SBP ≥90 mm Hg or within 40 mm Hg of baseline |
| Pneumonia: Fulfill two or more of the following: a) new infiltrate on chest radiograph; b) temperature >38.3°C or <36.0°C or WBC >12,000 or <4000 or >10% bandemia; c) positive microbiologic culture |
| Trauma: Defined as multiple fractures and/or pulmonary contusions. Multiple fractures are defined as a fracture of two long bones, an unstable pelvic fracture, or one long bone and a pelvic fracture. Pulmonary contusion is defined as infiltrates on chest radiographs within 8 hrs of admission to the emergency room and evidence of blunt trauma to the chest such as fractured ribs or ecchymosis overlying the infiltrate. |
| Multiple transfusions: Defined as receiving ≤8 units of packed red blood cells within 24 hrs |
| Aspiration: Defined as witnessed or documented aspiration event or the retrieval of gastric contents from the oropharynx, endotracheal tube, or bronchial tree |
WBC, white blood cell; SBP, systolic blood pressure.
Nested within the prospective cohort, a case-control study was designed (Fig. 1). All patients who did not develop ARDS during their hospitalization with no prior history of ARDS or prior enrollment into the study were selected as controls. The Human Subjects Committees approved the study, and informed written consent was obtained from all subjects or their appropriate surrogates.
Baseline clinical information was collected for all subjects on admission to ICU. Vital signs and laboratory variables in the first 24 hrs after ICU admission were collected for calculation of Acute Physiology and Chronic Health Evaluation (APACHE) III. ARDS patients were followed for the secondary outcomes of all-cause 60-day mortality and daily multiple organ dysfunction score (MODS) for 28 days as defined according to Brussels Organ Dysfunction Score (23, 25). Respiratory failure was not included in the calculation of the organ dysfunction score in the ARDS patients.
Recollection and re-entry of the clinical data from 89 (13%) subjects selected at random revealed a data-entry error rate of 1% and a data collection error rate of 2.8%. Missing physiology data occurred in <13% of patients. Similar to the APACHE III study, missing physiology values were assumed to be within the normal range. All other missing data were coded as missing. All data were collected onto clinical data forms and entered into an ACCESS data-base at the Harvard School of Public Health.
Genotyping Methods
Blood (10 mL) was collected for DNA extraction and polymerase chain reaction amplification. Genotyping was done on the Sequenom MassARRAY for MBLXY and codon 57 and by polymerase chain reaction/restriction fragment length polymorphism for codon 54 (9) and Taqman for codon 52. The genotyping for a random 5% of samples was repeated using alternative methods for quality control. All genotyping results were interpreted by two separate research staff. Research personnel were blinded to the case-control status or genotype of the subjects.
Haplotypes were generated from the genotype results using the Partition Ligation-Expectation Maximization (PL-EM) version 1.0 (26), as in other association studies (27) that reconstruct individual probabilities for individual phasing accuracy based on unphased genotype data. Only those 589 patients with complete genotyping data for the four MBL2 polymorphisms were used to infer haplotypes.
Statistical Analysis
Conformity to Hardy-Weinberg equilibrium and Lewontin’s D’ for linkage disequilibrium was determined using SAS/Genetics (version 9.0) (28). Univariate analysis was performed using Fisher’s exact test, analysis of variance, or Wilcoxon rank-sum tests. Kaplan-Meier curves for 60-day ARDS survival were compared using the log-rank test. ARDS patients who were lost to follow-up, who were discharged home alive before 60 day, or who survived to 60 days were censored at discharge or at 60 days if known to be alive.
Multivariate analyses included logistic regression for development of ARDS and Cox proportional hazard models for ARDS survival. Backward selection algorithms were used to determine possible confounders. The final multivariate model include the gene effect, results from backward elimination, and clinically relevant variables such as APACHE III score. A Hosmer-Lemeshow test was used to evaluate logistic regression model fit (29). Interactions with the MBL-2 genotypes were tested with interaction terms, and no significant interactions were found. Only APACHE III scores deviated from proportional hazard assumption as indicated by time varying covariates (p = .04), so the final model for ARDS survival was stratified by APACHE III score by quartiles.
Daily MODS after development of ARDS was compared using linear mixed effects models assuming an unstructured covariance matrix using the Proc Mixed procedure in SAS. The fixed factors included the polymorphisms and potential confounders such as age, trauma, baseline APACHE III scores, treatment with corticosteroids before admission, liver failure, transfusion, and septic shock. The number of days after development of ARDS was considered to be a random factor. To evaluate whether the MODS score varied significantly by genotype during the course of ARDS, an interaction term between genotype and time was included in the model. For patients discharged from the hospital within 28 days of ARDS, their last available MODS score before discharge was assigned to all subsequent days. For ARDS patients who died within 28 days of ARDS, the maximal score was assigned to all days subsequent to death. Ventilator-free days in ARDS were calculated as previously described (30). All analyses were conducted using SAS version 9. A p value of .05 was considered statistically significant.
Assuming an α-error of .05, 80% power, and genotype frequencies of 24% for the MBLXY X allele and 12%, 26%, and 3% for variant 52D, 54B, and 57C, respectively (9, 31), a study with 212 cases and 442 controls would have a minimum detectable odds ratios of 1.6 for MBLXY and 1.9, 1.7, and 2.9 for codon 52, 54, and 57, respectively.
RESULTS
Patient Population
Between September 9, 1999, and October 15, 2002, 752 patients were enrolled including 237 ARDS patients and 477 controls selected and genotyped for the MBL2 polymorphisms in the case-control study (Fig. 1). All subsequent analyses were restricted to the 212 Caucasian cases and 442 Caucasian controls.
Clinical risk factors for ARDS and baseline characteristics on admission to the ICU are shown in Tables 2 and 3. Variables in the final model for development of ARDS included direct pulmonary injury (p < .001), trauma (p < .001), age (p = .002), female gender (p = .04), diabetes (p = .003), platelets ≤80,000/mm (p = .003), blood transfusion (p < .001), septic shock (p = .1), and APACHE III score (p = .6). The Hosmer-Lemeshow goodness-of-fit statistic for the final model was 9.3 with 8 degrees of freedom (p = .3), indicating no significant lack of fit of the model.
Table 2.
Clinical risk factors for acute respiratory distress syndrome (ARDS) between cases and controls and survivors and nonsurvivors in ARDS
| Development of ARDS No. (%) |
Mortality in ARDS No. (%) |
|||||
|---|---|---|---|---|---|---|
| Risk for ARDSa | Controls (n = 442) |
Cases (n = 212) |
p Value | Survivors (n = 114) |
Nonsurvivors n = 98) |
p Value |
| Sepsis syndrome | 168 (38) | 67 (32) | .1 | 44 (39) | 23 (23) | .03 |
| Pneumonia source | 87 (20) | 52 (25) | <.001 | 33 (29) | 19 (19) | .6 |
| Extrapulmonary source | 81 (18) | 15 (7) | 11 (10) | 4 (4) | ||
| Septic shock | 196 (44) | 115 (54) | .02 | 52 (46) | 63 (64) | .009 |
| Pneumonia source | 89 (20) | 79 (37) | <.001 | 36 (32) | 43 (44) | >.9 |
| Extrapulmonary source | 107 (24) | 36 (17) | 16 (14) | 20 (20) | ||
| Trauma | 40 (9) | 9 (4) | .04 | 8 (7) | 1 (1) | .04 |
| Multiple transfusions | 51 (12) | 26 (12) | .8 | 13 (11) | 13 (13) | .7 |
| Aspiration | 34 (8) | 24 (11) | .1 | 13 (11) | 11 (11) | >.9 |
| >1 Risk for ARDS | 48 (11) | 29 (14) | .3 | 16 (14) | 13 (13) | >.9 |
| Direct pulmonary injuryb | 211 (48) | 144 (68) | <.001 | 79 (69) | 65 (66) | .7 |
| Indirect pulmonary injuryc | 231 (52) | 68 (32) | 35 (31) | 33 (34) | ||
Numbers of controls and cases with each risk add up to >654 patients because of multiple risks in 75 patients
pneumonia, aspiration, or pulmonary contusions were categorized as direct pulmonary injury
sepsis from an extrapulmonary source, trauma without pulmonary contusions, and multiple transfusions were categorized as indirect pulmonary injury. Patients (n = 69) with both direct and indirect pulmonary injuries were considered to have direct pulmonary injury.
Table 3.
Baseline characteristics between patients with acute respiratory distress syndrome (ARDS) and controls and survivors and nonsurvivors in ARDS
| Development of ARDS |
Mortality in ARDS |
|||||
|---|---|---|---|---|---|---|
| Controls (n = 442) |
Cases (n = 212) |
p Value | Survivors (n = 114) |
Nonsurvivors (n = 98) |
p Value | |
| Females, n (%) | 179 (41) | 101 (48) | .09 | 50 (44) | 51 (52) | .3 |
| Age, median (range) | 69 (18-94) | 65 (18-97) | .05 | 57 (18-89) | 73 (22-97) | <.001 |
| APACHE III median (range)a | 64 (14-130) | 68 (8-136) | .08 | 69 (8-115) | 88 (29-150) | <.001 |
| Diabetes, n (%)b | 116 (26) | 34 (16) | .004 | 20 (18) | 14 (14) | .6 |
| History of alcohol abuse, n (%) | 42 (10) | 27 (13) | .2 | 10 (9) | 17 (17) | .07 |
| Tobacco abuse, n (%)c | 217 (49) | 105 (50) | .4 | 57 (50) | 48 (49) | .7 |
| Chronic liver disease, n (%)b | 18 (4) | 12 (6) | .4 | 4 (4) | 8 (8) | .2 |
| End-stage renal disease, n (%) | 23 (5) | 6 (3) | .2 | 2 (2) | 4 (4) | .4 |
| History of steroid use, n (%) | 37 (8) | 20 (9) | .7 | 6 (5) | 14 (14) | .03 |
| Transfusion of PRBC, n (%) | 218 (49) | 133 (63) | .001 | 64 (56) | 69 (70) | .03 |
| Number of PRBC transfused, median (range) | 0 (0-74) | 2 (0-63) | .005 | 1 (0-31) | 2 (0-63) | .01 |
| Systolic BP <90 mm Hg, n (%) | 305 (69) | 163 (77) | .04 | 85 (75) | 78 (80) | .4 |
| Creatinine <2.0 mg/L, n (%) | 150 (34) | 65 (31) | 0.4 | 28 (25) | 37 (38) | .05 |
| Bilirubin <2.0 mg/dL, n (%) | 53 (12) | 39 (18) | .03 | 14 (12) | 25 (26) | .02 |
| Hematologic failure (platelets ≤80,000/mm), n (%) | 60 (14) | 47 (22) | .007 | 20 (18) | 27 (28) | .1 |
APACHE, Acute Physiology and Chronic Health Evaluation; PRBC, packed red blood cells; BP, blood pressure.
For development of ARDS, APACHE III scores for patients and controls were calculated without the PaO2/FIO2 component; for survivors and nonsurvivors in ARDS, APACHE III scores were calculated with all components
chronic health information was missing on one patient and two controls
tobacco history was missing in 56 (26%) patients and 97 (22%) controls.
Genotype Analyses and Development of ARDS
The variant allele frequency in the study was 8% for codon 52, 15% for codon 54, 1.5% for codon 57, and 17% for MBLXY, which is similar to prior studies (8, 10, 11, 32). Genotype failure for codon 52, 54, 57, and MBLXY occurred in 38 (6%), 0 (0%), 15 (2%), and 13 (2%) patients, respectively. Genotype frequencies among cases and controls are shown in Table 4. Among the 212 patients with ARDS, the observed frequency for the codon 54BB genotype was higher than that predicted by Hardy-Weinberg equilibrium. However, this was not statistically significant (p = .4), given the smaller number of patients with ARDS. Among the 442 controls, the genotype frequency for codon 54 deviated from that predicted by Hardy-Weinberg equilibrium (p = .03). This is unlikely to be due to genotyping error as there was no discrepancy on repeat genotyping. However, controls in this study were not healthy, and patients homozygous for the codon 54B genotype were more likely to have septic shock on ICU admission as a predisposing injury for ARDS (eight of ten [80%] vs. 303 of 643 [47%]; p = .04). An association between the codon 54B genotype and the underlying condition among the controls may explain the deviation from Hardy Weinberg equilibrium.
Table 4.
Genotype frequencies for the mannose binding lectin-2 (MBL2) codon 52, 54, 57, and MBLXY polymorphisms among patients and controls and among survivors and nonsurvivors in acute respiratory distress syndrome (ARDS)
| Percentage of Patients (No.) |
||||||
|---|---|---|---|---|---|---|
| Polymorphism | Controls (n = 442) |
Cases (n = 212) |
p Value | Survivors in ARDS (n = 144) |
Nonsurvivors in ARDS (n = 98) |
p Value |
| Codon 52 | ||||||
| 52AA | 82 (345) | 86 (170) | 90 (97) | 82 (73) | ||
| 52AD | 17 (72) | 14 (27) | .4 | 10 (11) | 18 (16) | .1 |
| 52DD | 0.5 (2) | 0 (0) | 0 | 0 | ||
| Codon 54 | ||||||
| 54AA | 72 (319) | 71 (151) | 68 (78) | 74 (73) | ||
| 54AB | 27 (120) | 25 (54) | <.05 | 31 (35) | 19 (19) | .03 |
| 54BB | 0.7 (3) | 3.3 (7) | 1 (1) | 6 (6) | ||
| Codon 57 | ||||||
| 57AA | 97 (418) | 98 (205) | 98 (111) | 97 (94) | ||
| 57AC | 3 (12) | 2 (5) | >.9 | 2 (2) | 3 (3) | .5 |
| 57CC | 0.2 (1) | 0 (0) | 0 | 0 | ||
| MBLXY | ||||||
| YY | 71 (306) | 65 (136) | 67 (75) | 64 (61) | ||
| XY | 26 (111) | 29 (60) | .2 | 26 (29) | 32 (31) | .5 |
| XX | 3 (15) | 6 (12) | 7 (8) | 4 (4) | ||
Fisher’s exact test p values are reported.
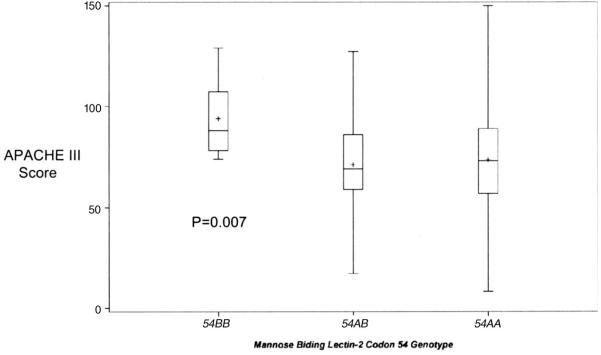
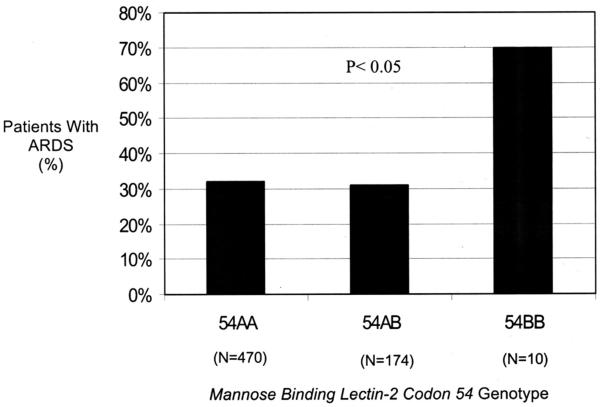
Among cases and controls, patients homozygous for the MBL-2 codon 54 variant B allele had significantly greater severity of illness, as indicated by APACHE III score, than patients homozygous or heterozygous for the wild-type A allele (p = .007) (Fig. 2). Development of ARDS varied with the codon 54 genotype (p < .05), with patients who were homozygous for the variant codon 54B allele having an increased odds of developing ARDS compared with homozygotes or heterozygotes for the wild-type A allele (odds ratio [OR], 5.0, 95% confidence interval [CI], 1.3-20; p = .02) (Fig. 3). On multivariate analyses after adjustment for age, gender, APACHE III, septic shock, trauma, direct pulmonary injury, hematologic and hepatic failure, and transfusion, the codon 54B genotype was still significantly associated with development of ARDS (ORadj, 6.7; 95% CI, 1.5-31; p = .01). When the analysis was restricted to the 311 patients admitted to the ICU with septic shock, the association between the 54BB genotype and ARDS was even stronger (ORadj, 12.0; 95% CI, 1.9 -74; p = .008).
Figure 2.
Acute Physiology and Chronic Health Evaluation (APACHE) III score on admission to the intensive care unit by genotype for the mannose binding lectin-2 codon 54 polymorphism among all 654 cases and controls. The box denotes the interquartile range (25-75%), the horizontal line in the box indicates the median, + designates the mean, and error bars indicate the 95% confidence intervals.
Figure 3.
The mannose binding lectin-2 codon 54 genotype and the percentage of patients with acute respiratory distress syndrome (ARDS) with each genotype. The p value was calculated from Fisher’s exact test.
Because of the linkage between the MBLXY Y allele and the codon 54 variant B allele, patients homozygous for the MBL Y allele had higher APACHE III scores on admission to the ICU compared with patients who were homozygous or heterozygous for the X allele (mean 83 [SD 25] vs. 73 [25]; p = .04). However, the frequency of the variant alleles of MBL XY, codon 52, or codon 57 on the MBL-2 gene did not vary significantly with ARDS (Table 4). When the variant alleles of codon 52, 54, and 57 were examined together as the variant O allele, carriage of the O allele was not associated with ARDS (ORadj, 0.96; 95% CI, 0.66-1.4)
The four polymorphisms are in complete linkage disequilibrium (D’ = 1.0). Consistent with other reports (14, 18, 33), the variant codon 54 B allele occurred only with the Y allele of the MBLXY polymorphism. Five haplotypes were identified with probability >99%. Haplotype analyses are similar to the preceding results. Only the ABAY haplotype was associated with the development of ARDS (Table 5).
Table 5.
Haplotype frequencies for the mannose binding lectin-2 (MBL-2) codon 52, 54, and 57 and MBLXY polymorphisms and development of acute respiratory distress syndrome (ARDS) and 60-day mortality in ARDS
| Development of ARDS |
Mortality in ARDS |
||||||||
|---|---|---|---|---|---|---|---|---|---|
| Patients With ARDS with Genotype/Total No. of Patients with Genotype (%) |
Nonsurvivors with Genotype/All ARDS Patients with Genotype (%) |
||||||||
| No. of Copies of Haplotype |
No. of Copies of Haplotype |
||||||||
| Haplotypea | Haplotype Frequencyb | 0 | 1 | 2 | p Value | 0 | 1 | 2 | p Value |
| AAAY | 0.59 | 42/107 (39) | 88/285 (30) | 62/197 (31) | .2 | 20/42 (48) | 41/86 (48) | 24/62 (39) | .5 |
| AAAX | 0.16 | 124/406 (31) | 55/158 (35) | 11/25 (44) | .3 | 54/124 (44) | 27/55 (49) | 4/11 (36) | .7 |
| DAAY | 0.09 | 165/496 (33) | 25/91 (27) | 0/2 (0) | .4 | 70/165 (42) | 15/25 (60) | 0 | .1 |
| ABAY | 0.14 | 134/422 (32) | 49/157 (31) | 7/10 (70) | <.05 | 63/134 (47) | 16/49 (33) | 6/7 (86) | .02 |
| AACY | 0.02 | 185/571 (32) | 5/17 (29) | 0/1 (0) | >.9 | 82/185 (44) | 3/5 (60) | 0 | .7 |
Haplotypes consist of codon 52, codon 54, codon 57, and MBLXY. The variant alleles for codon 52, 54, and 57 are D, B, and C, respectively. The wild-type allele is known collectively as the A allele
haplotype frequency among controls only. The p values from Fisher’s exact test for each haplotype are presented. Only the ABAY haplotype was associated with development of ARDS (p < .05) and mortality in ARDS (p = .02).
Outcomes in ARDS
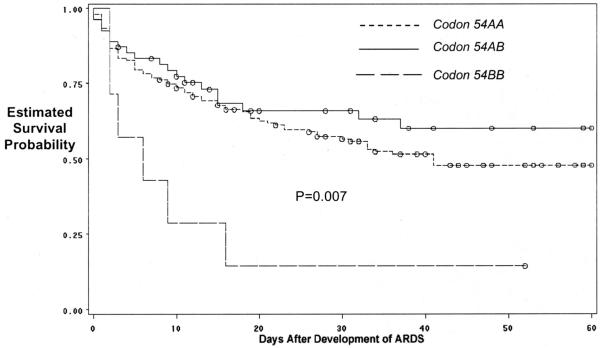
The 60-day mortality was 46% (98 of 211) for ARDS patients. Clinical risks for ARDS and baseline characteristics between survivors and non-survivors of ARDS are shown in Tables 2 and 3. Consistent with prior reports, survivors and nonsurvivors did not differ in Pao2/FIo2 (p = .4), compliance (p = .4), or Lung Injury Score (p = .5). However, non-survivors had significantly fewer ventilator-free days than survivors (median 0 days [25-75% 0-0] vs. 7 days [25-75% 2-14]; p < .001). The 60-day mortality in ARDS varied significantly with the codon 54 genotype (p = .03) (Table 4) and the ABAY haplotype (Table 5). ARDS patients homozygous for the variant B allele had decreased survival compared with ARDS patients who were homozygous or heterozygous for the wild-type allele (hazard ratio [HR], 3.1; 95% CI, 1.4 -7.2; p = .007) (Fig. 4). Among controls, the codon 54BB genotype was not significantly associated with ICU mortality (HRadj, 7.4; 95% CI, 0.90-61.3), although the power to determine an association was limited given that there were only three patients with the BB genotype who did develop ARDS. ARDS patients homozygous for the variant codon 54B allele had a nonsignificant trend to greater severity of illness on admission (p = .08) and significantly greater daily multiple organ dysfunction score after development of ARDS even after adjustment for potentially important variables (Fig. 5). On multivariate analysis, the codon 54BB genotype remained associated with increased mortality compared with ARDS patients who were homozygous for the wild-type A allele (HRadj, 4.0; 95% CI, 1.6-10; p = .003).
Figure 4.
Estimated survival probability in the 212 patients with acute respiratory distress syndrome (ARDS) by the mannose binding lectin-2 codon 54 genotype.
Figure 5.
Daily Brussels multiple organ dysfunction score for each day after development of acute respiratory distress syndrome (ARDS) for the 212 patients with ARDS after adjustment for potentially important variables such as age, trauma as a risk factor for ARDS, Acute Physiology and Chronic Health Evaluation III scores, history of treatment with corticosteroids, liver failure, transfusion, and septic shock.
The 60-day mortality in ARDS did not vary significantly with the MBL-2 polymorphisms at MBLXY, codon 52, or codon 57 (Table 4) or when examined together as the variant O allele (HRadj, 1.0; 95% CI, 0.68-1.6).
DISCUSSION
In a study of prospectively enrolled ICU patients with clearly defined ARDS risk factors, we found significant associations between the BB genotype of the MBL-2 codon 54 polymorphism and increased severity of illness on admission, increased development of ARDS, more multiple organ failures after development of ARDS, and increased mortality in ARDS. This study has a number of strengths. The prospective determination of ARDS using the American-European Consensus Conference definition helps minimize phenotype misclassification. In addition, clearly defined at-risk controls were used in this study. Using critically ill controls who have the opportunity to develop the outcome is more clinically relevant than using healthy individuals. Although this may bias the study toward the null, using at-risk controls also reduces the confounding from any possible association between the gene and the risk condition such as sepsis or pneumonia.
Several other studies have found the variant O allele (codon 52D, codon 54B, or codon 57C allele) to be associated with infections, systemic inflammatory responses, or other disease states in a codominant (AO and OO vs. AA genotype) (8, 15, 18, 34, 35) or recessive model (OO vs. AO and AA genotype) (11, 10, 9, 36). We did not find any associations between ARDS and the MBLXY X allele or the variant alleles for codon 52 and 57. However, it is important to note that, given the low variant allele frequency, our power to detect an association for the codon 52 and 57 was limited. Nevertheless, codon 54 may be very significant in the MBL-2 gene. The variant codon 54B allele is more prevalent than codon 52 and codon 57 in the Caucasian populations. Individuals with the codon 54B allele demonstrate even lower MBL protein concentration than individuals with the 52D allele (33), and what MBL protein is produced was found to be incapable of activating the classic complement pathway (37). Of the four polymorphisms, codon 54B has been independently found to be associated with increased susceptibility to infection (12, 14, 16, 17, 38).
It is not clear why patients with the codon 54 BB genotype may be at increased risk of developing and dying of ARDS in this study. Homozygotes for the variant MBL-2 alleles have been associated with a low circulating MBL (9, 18). It is possible that patients with low MBL may be more susceptible to multilobar pneumonia or more severe septic shock.
Alternatively, it is possible that MBL may influence the inflammatory response to the initial injury in critical illnesses (7). MBL has been shown to decrease tumor necrosis factor release (39) and stimulate the production of anti-inflammatory cytokines such as interleukin-10 (40), but MBL modulation of TNF may be dose dependent. At low doses of MBL, increasing MBL concentration increases production of proinflammatory cytokines. But at higher concentrations, increasing MBL suppresses these inflammatory cytokines (41). It is possible that in critically ill patients with the low or defective MBL-producing genotypes, circulating MBL may not be sufficiently high or effective enough to switch from enhancement to suppression of the proinflammatory cytokine response. Unfortunately, the functional significance of the MBL-2 polymorphisms and the inflammatory response of these patients in this study could not be examined. Additional studies are clearly needed to investigate the possible mechanisms by which MBL-2 genotypes may be important in acute lung injury.
Among the controls in this study, the genotype frequency of the codon 54 polymorphism deviated from that predicted by Hardy-Weinberg equilibrium. This is unlikely to be due to genotyping error. The genotype frequencies found here compare well with those reported in other populations. In addition, repeat genotyping in a random subset of patients revealed no discrepancy in genotype results. It is more likely that deviation from Hardy-Weinberg occurred because of a possible association between the codon 54 polymorphism and the underlying condition requiring ICU admission such as sepsis. Indeed, patients in this study who were homozygous for the variant codon 54B allele were more likely to have septic shock on admission to the ICU. The association found between the codon 54 MBL-2 polymorphism and ARDS cannot be attributed to this association between 54B and septic shock, a risk factor for ARDS. The association between the 54BB genotype and ARDS was actually strengthened when the analyses were restricted to those patients with septic shock.
Given the low allele frequency for codon 54, only ten patients were homozygous for the variant B allele in the study and no adjustment for multiple comparisons were made. However, the findings in this report are unlikely to be due to type I error. The polymorphism was chosen a priori as a candidate in ARDS based on previous studies supporting its role in infection and critical illnesses. The results are consistent with our hypothesis and with previous reports on infection and sepsis. Additionally, the findings are supported by results of secondary out-comes such as septic shock, severity of illness, and organ failure after development of ARDS. Nevertheless, as is true of all genetic association studies, our findings will need to be confirmed in other populations.
We recognize some other limitations to our study. The functional significance of the MBL-2 polymorphisms was not evaluated in this study. Because of the study design, the results may not be generalizable to the community setting, to immunocompromised hosts or patients without risk factors for ARDS, or with different clinical risks for ARDS. In addition, the analyses were restricted to Caucasians, which reduces the possibility of confounding from ethnicity (42) but does not permit extrapolation of the results to other ethnic groups.
CONCLUSIONS
We report an association between the MBL-2 codon 54 genotype and severity of illness, septic shock, and development of ARDS. The MBL-2 codon 54 genotype may also be associated with multiple organ dysfunction and mortality in ARDS. Additional studies are needed to confirm these findings in other populations with other risk factors.
ACKNOWLEDGMENTS
We thank Wei-Ling Zhang, Kelly McCoy, Thomas McCabe, Christopher Schwartzenburg, Barbara Bean, and Linda Lineback for patient recruitment; Andrea Solomon, Andrea Shafer, and Lia Shimada for research support; Li Su for laboratory expertise; Janna Frelich, Marcia Chertok, Sal Mucci, and Richard Rivera for data management; and Paul D. Boyce MD for his assistance in radiologic interpretation.
Supported, in part, by research grant RO1 HL60710 from NHLBI (Boston, MA). Dr. Gong is suported by K23 HL67197 and RO1 HL084060 from NHLBI (New York, NY). Dr. Zhou is supported by Flight Attendants Medical Research Institute Young Clinical Scientist Award (Boston, MA).
Footnotes
See also p. 293.
Dr. Christiani was a paid scientific advisor to Gentra Corp from 2001 to 2005. The remaining authors have not disclosed any potential conflicts of interest.
REFERENCES
- 1.Hudson LD, Milberg JA, Anardi D, et al. Clinical risks for development of the acute respiratory distress syndrome. Am J Respir Crit Care Med. 1995;151:293–301. doi: 10.1164/ajrccm.151.2.7842182. [DOI] [PubMed] [Google Scholar]
- 2.Gong MN, Wei Z, Xu LL, et al. Polymorphism in the surfactant protein-B gene, gender, and the risk of direct pulmonary injury and ARDS. Chest. 2004;125:203–211. doi: 10.1378/chest.125.1.203. [DOI] [PubMed] [Google Scholar]
- 3.Marshall RP, Webb S, Bellingan GJ, et al. Angiotensin converting enzyme insertion/deletion polymorphism is associated with susceptibility and outcome in acute respiratory distress syndrome. Am J Respir Crit Care Med. 2002;166:646–650. doi: 10.1164/rccm.2108086. [DOI] [PubMed] [Google Scholar]
- 4.Quasney MW, Waterer GW, Dahmer MK, et al. Association between surfactant protein B + 1580 polymorphism and the risk of respiratory failure in adults with community-acquired pneumonia. Crit Care Med. 2004;32:1115–1119. doi: 10.1097/01.ccm.0000124872.55243.5a. [DOI] [PubMed] [Google Scholar]
- 5.Gadjeva M, Takahashi K, Thiel S. Mannan-binding lectin—A soluble pattern recognition molecule. Mol Immunol. 2004;41:113–121. doi: 10.1016/j.molimm.2004.03.015. [DOI] [PubMed] [Google Scholar]
- 6.Klein NJ. Mannose-binding lectin: Do we need it? Mol Immunol. 2005;42:919–924. doi: 10.1016/j.molimm.2004.12.006. [DOI] [PubMed] [Google Scholar]
- 7.Turner MW. The role of mannose-binding lectin in health and disease. Mol Immunol. 2003;40:423–429. doi: 10.1016/s0161-5890(03)00155-x. [DOI] [PubMed] [Google Scholar]
- 8.Garred P, Pressler T, Madsen HO, et al. Association of mannose-binding lectin gene heterogeneity with severity of lung disease and survival in cystic fibrosis. J Clin Invest. 1999;104:431–437. doi: 10.1172/JCI6861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Garred P, Madsen HO, Halberg P, et al. Mannose-binding lectin polymorphisms and susceptibility to infection in systemic lupus erythematosus. Arthritis Rheum. 1999;42:2145–2152. doi: 10.1002/1529-0131(199910)42:10<2145::AID-ANR15>3.0.CO;2-#. [DOI] [PubMed] [Google Scholar]
- 10.Hibberd ML, Sumiya M, Summerfield JA, et al. Meningococcal Research Group Association of variants of the gene for mannose-binding lectin with susceptibility to meningococcal disease. Lancet. 1999;353:1049–1053. doi: 10.1016/s0140-6736(98)08350-0. [DOI] [PubMed] [Google Scholar]
- 11.Roy S, Knox K, Segal S, et al. MBL genotype and risk of invasive pneumococcal disease: A case-control study. Lancet. 2002;359:1569–1573. doi: 10.1016/S0140-6736(02)08516-1. [DOI] [PubMed] [Google Scholar]
- 12.Song le H, Binh VQ, Duy DN, et al. Mannose-binding lectin gene polymorphisms and hepatitis B virus infection in Vietnamese patients. Mutat Res. 2003;522:119–125. doi: 10.1016/s0027-5107(02)00284-1. [DOI] [PubMed] [Google Scholar]
- 13.Thomas HC, Foster GR, Sumiya M, et al. Mutation of gene of mannose-binding protein associated with chronic hepatitis B viral infection. Lancet. 1996;348:1417–1419. doi: 10.1016/s0140-6736(96)05409-8. [DOI] [PubMed] [Google Scholar]
- 14.Ip WK, Chan KH, Law HK, et al. Mannose-binding lectin in severe acute respiratory syndrome coronavirus infection. J Infect Dis. 2005;191:1697–1704. doi: 10.1086/429631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gabolde M, Guilloud-Bataille M, Feingold J, et al. Association of variant alleles of mannose binding lectin with severity of pulmonary disease in cystic fibrosis: cohort study. BMJ. 1999;319:1166–1167. doi: 10.1136/bmj.319.7218.1166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Takahashi R, Tsutsumi A, Ohtani K, et al. Association of mannose binding lectin (MBL) gene polymorphism and serum MBL concentration with characteristics and progression of systemic lupus erythematosus. Ann Rheum Dis. 2005;64:311–314. doi: 10.1136/ard.2003.020172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yang IA, Seeney SL, Wolter JM, et al. Mannose-binding lectin gene polymorphism predicts hospital admissions for COPD infections. Genes Immun. 2003;4:269–274. doi: 10.1038/sj.gene.6363961. [DOI] [PubMed] [Google Scholar]
- 18.Garred P, Strom J, Quist L, et al. Association of mannose-binding lectin polymorphisms with sepsis and fatal outcome, in patients with systemic inflammatory response syndrome. J Infect Dis. 2003;188:1394–1403. doi: 10.1086/379044. [DOI] [PubMed] [Google Scholar]
- 19.Fidler KJ, Wilson P, Davies JC, et al. Increased incidence and severity of the systemic inflammatory response syndrome in patients deficient in mannose-binding lectin. Intensive Care Med. 2004;30:1438–1445. doi: 10.1007/s00134-004-2303-8. [DOI] [PubMed] [Google Scholar]
- 20.Bell RC, Coalson JJ, Smith JD, et al. Multiple organ system failure and infection in adult respiratory distress syndrome. Ann Intern Med. 1983;99:293–298. doi: 10.7326/0003-4819-99-3-293. [DOI] [PubMed] [Google Scholar]
- 21.Ferring M, Vincent JL. Is outcome from ARDS related to the severity of respiratory failure? Eur Respir J. 1997;10:1297–1300. doi: 10.1183/09031936.97.10061297. [DOI] [PubMed] [Google Scholar]
- 22.Montgomery AB, Stager MA, Carrico CJ, et al. Causes of mortality in patients with the adult respiratory distress syndrome. Am Rev Respir Dis. 1985;132:485–489. doi: 10.1164/arrd.1985.132.3.485. [DOI] [PubMed] [Google Scholar]
- 23.Gong MN, Thompson BT, Williams P, et al. Clinical predictors of and mortality in acute respiratory distress syndrome: Potential role of red cell transfusion. Crit Care Med. 2005;33:1191–1198. doi: 10.1097/01.ccm.0000165566.82925.14. [DOI] [PubMed] [Google Scholar]
- 24.Bernard GR, Artigas A, Brigham KL, et al. The American-European Consensus Conference on ARDS Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med. 1994;149:818–824. doi: 10.1164/ajrccm.149.3.7509706. [DOI] [PubMed] [Google Scholar]
- 25.Bernard GR, Doig G, Hudson L. Quantification of organ failure for clinical trials and clinical practice. Am J Respir Crit Care Med. 1995;151:A323. [Google Scholar]
- 26.Qin ZS, Niu T, Liu JS. Partition-ligation-expectation-maximization algorithm for haplotype inference with single-nucleotide polymorphisms. Am J Hum Genet. 2002;71:1242–1247. doi: 10.1086/344207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Han J, Hankinson SE, De Vivo I, et al. A prospective study of XRCC1 haplotypes and their interaction with plasma carotenoids on breast cancer risk. Cancer Res. 2003;63:8536–8541. [PubMed] [Google Scholar]
- 28.SAS . An Introduction to Genetic Data Analyses Using SAS. Customer Support Center; [Accessed October 10, 2005]. http://support.sas.com/rnd/papers/sugi27/genetics.pdf. [Google Scholar]
- 29.Hosmer D, Lemeshow S. Assessing the Fit of Model. 2nd ed. Wiley-Interscience; New York: 2000. [Google Scholar]
- 30.Schoenfeld DA, Bernard GR. Statistical evaluation of ventilator-free days as an efficacy measure in clinical trials of treatments for acute respiratory distress syndrome. Crit Care Med. 2002;30:1772–1777. doi: 10.1097/00003246-200208000-00016. [DOI] [PubMed] [Google Scholar]
- 31.Mead R, Jack D, Pembrey M, et al. The ALSPAC Study Team. Avon Longitudinal Study of Pregnancy and Childhood Mannose-binding lectin alleles in a prospectively recruited UK population. Lancet. 1997;349:1669–1670. doi: 10.1016/s0140-6736(05)62635-9. [DOI] [PubMed] [Google Scholar]
- 32.Meade MO, Cook RJ, Guyatt GH, et al. Interobserver variation in interpreting chest radiographs for the diagnosis of acute respiratory distress syndrome. Am J Respir Crit Care Med. 2000;161:85–90. doi: 10.1164/ajrccm.161.1.9809003. [DOI] [PubMed] [Google Scholar]
- 33.Madsen HO, Garred P, Thiel S, et al. Inter-play between promoter and structural gene variants control basal serum level of mannan-binding protein. J Immunol. 1995;155:3013–3020. [PubMed] [Google Scholar]
- 34.Maas J, de Roda Husman AM, Brouwer M, et al. Amsterdam Cohort Study Presence of the variant mannose-binding lectin alleles associated with slower progression to AIDS. Aids. 1998;12:2275–2280. doi: 10.1097/00002030-199817000-00008. [DOI] [PubMed] [Google Scholar]
- 35.Sutherland AM, Walley KR, Russell JA. Polymorphisms in CD14, mannose-binding lectin, and Toll-like receptor-2 are associated with increased prevalence of infection in critically ill adults. Crit Care Med. 2005;33:638–644. doi: 10.1097/01.ccm.0000156242.44356.c5. [DOI] [PubMed] [Google Scholar]
- 36.Davies JC, Turner MW, Klein N. Impaired pulmonary status in cystic fibrosis adults with two mutated MBL-2 alleles. Eur Respir J. 2004;24:798–804. doi: 10.1183/09031936.04.00055404. [DOI] [PubMed] [Google Scholar]
- 37.Super M, Gillies SD, Foley S, et al. Distinct and overlapping functions of allelic forms of human mannose binding protein. Nat Genet. 1992;2:50–55. doi: 10.1038/ng0992-50. [DOI] [PubMed] [Google Scholar]
- 38.Gomi K, Tokue Y, Kobayashi T, et al. Mannose-binding lectin gene polymorphism is a modulating factor in repeated respiratory infections. Chest. 2004;126:95–99. doi: 10.1378/chest.126.1.95. [DOI] [PubMed] [Google Scholar]
- 39.Chaka W, Verheul AF, Vaishnav VV, et al. Induction of TNF-alpha in human peripheral blood mononuclear cells by the mannoprotein of Cryptococcus neoformans involves human mannose binding protein. J Immunol. 1997;159:2979–2985. [PubMed] [Google Scholar]
- 40.Sprong T, Jack DL, Klein NJ, et al. Mannose binding lectin enhances IL-1beta and IL-10 induction by non-lipopolysaccharide (LPS) components of Neisseria meningitidis. Cytokine. 2004;28:59–66. doi: 10.1016/j.cyto.2004.06.007. [DOI] [PubMed] [Google Scholar]
- 41.Jack DL, Read RC, Tenner AJ, et al. Mannose-binding lectin regulates the inflammatory response of human professional phagocytes to Neisseria meningitidis serogroup B. J Infect Dis. 2001;184:1152–1162. doi: 10.1086/323803. [DOI] [PubMed] [Google Scholar]
- 42.Bone RC, Balk RA, Cerra FB, et al. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Chest. 1992;101:1644–1655. doi: 10.1378/chest.101.6.1644. [DOI] [PubMed] [Google Scholar]