Abstract
OBJECTIVES
Age is an important risk factor for morbidity after lung resection. This study was performed to identify specific risk factors for complications after lung resection in octogenarians.
METHODS
A prospective database containing patients aged 80 years or older who underwent lung resection at a single institution between January 2000 and June 2009 was reviewed. Preoperative, histopathologic, perioperative, and outcome variables were assessed. Morbidity was measured as a patient having any perioperative event as defined by the Society of Thoracic Surgeons General Thoracic Surgery Database. A multivariable risk model for morbidity was developed using a panel of established preoperative and operative variables. Survival was calculated using the Kaplan-Meier method.
RESULTS
During the study period, 193 patients aged 80 years or older (median age 82) underwent lung resection: wedge resection in 77, segmentectomy in 13, lobectomy in 96, bilobectomy in 4, and pneumonectomy in 3. Resection was accomplished via thoracoscopy in 149 patients (77%). Operative mortality was 3.6% (7 patients) and morbidity was 46% (89 patients). Patient discharge disposition was directly to home in 181 patients (94%). Postoperative events included atrial arrhythmia in 38 patients (20%), prolonged air leak in 24 patients (12%), postoperative transfusion in 22 patients (11%), delirium in 16 patients (8%), need for bronchoscopy in 14 patients (7%), and pneumonia in 10 patients (5%). Significant predictors of morbidity by multivariable analysis included resection greater than wedge (odds ratio 2.98, p=0.0064), thoracotomy as operative approach (odds ratio 2.64, p=0.034), and % predicted Forced Expiratory Volume in 1 second (odds ratio 1.28 for each 10% decrement, p=0.01).
CONCLUSIONS
Octogenarians can undergo lung resection with low mortality. Extent of resection, use of a thoracotomy, and impaired lung function increase the risk of complications. Careful evaluation is necessary to select the most appropriate approach in octogenarians being considered for lung resection.
Keywords: Lung Surgery, Outcomes, Octogenarians
Introduction
The evalution of octogenarians for pulmonary resection is a critical process, as advanced age is an important risk factor for postoperative morbidity and mortality [1]. Thoracic surgeons increasingly consider lung resection for octogenarians, as the percentage of patients that are found to have lung cancer who are 80 years or older is 7.5 to 17% and increasing with time [2-6]. Likely because of the increased risks associated with age, patients with non-small-cell lung cancer over 80 years of age are less likely to undergo potentially curative surgery [2-7]. However, older patients are more likely to have local disease at diagnosis, have similar post-resection survival when compared to younger patients, and have better survival with resection than with nonsurgical therapies [2-7]. When older patients are treated surgically for lung cancer, they are more likely than younger patients to undergo more limited resections than a lobectomy [8-10].
Lung cancer is the major source of mortality in even the oldest patients that do not undergo resection, suggesting that aggressive surgical management should be carefully considered for elderly patients with resectable lung cancer [4]. Determination of which patients are appropriate for surgery as well as planning the operative approach and extent of resection is especially important in this patient population, as the operative mortality after lung resection for octogenarians ranges from 0 to 15% [11-16]. Published risk factors for poor short and long-term outcomes after lung surgery in octogenarians include more than one co-morbid condition and poor performance status [6,15]. However, more detailed risk models examining patient characteristics as well as the impact of operative approach and extent of resection have not been developed. In particular, the patient selection criteria may be modified with utilization of a thoracoscopic approach, which lessens complications in elderly patients undergoing lobectomy [17,18]. The purpose of this study was to examine our experience with lung resection in octogenarians and develop a risk model for complications after lung resection in a series of octogenarians in the thoracoscopy era in order to improve patient selection, procedure selection, and risk stratification.
Materials and Methods
After local Institutional Review Board approval was granted, including waiver of the need for patient consent, the Duke University Medical Center Data Center was queried for Current Procedural Terminology codes linked with pulmonary resection for patients aged 80 years or older at the time of surgery between January 2000 and June 2009. Retrospective review of a prospective institutional database maintained on all thoracic surgery patients identified demographics, preoperative functional status, the use of induction therapy, smoking history, significant comorbidities, the histology and stage of disease, intraoperative details, and postoperative course. Chart review was utilized as necessary to complete data collection. Preoperative performance status was based on Zubrod, or Eastern Cooperative Oncology Group, scores that were included in the prospective database [19]. Any postoperative event prolonging or otherwise altering the postoperative course was recorded. Operative mortality included deaths that occurred within 30 days after operation or those that occurred later but during the same hospitalization. Deaths were captured both by chart review and use of the Social Security Death Index Database. The definitions of postoperative events were based on the Society of Thoracic Surgeons General Thoracic Surgery Database ( http://www.sts.org/sections/stsnationaldatabase, accessed March 5, 2009). Morbidity was defined as the occurrence of at least one postoperative event.
Unpaired student’s t tests were used to compare continuous data, Fisher’s exact tests for dichotomous data, and χ2 for categoric variables. A two-tailed p value of less than 0.05 was considered significant. Univariate analyses were performed relating operative morbidity to the following 15 patient characteristics: age, the presence of diabetes, the use of induction chemotherapy, the use of induction radiation, previous thoracic surgery, history of tobacco use, peripheral vascular disease, cerebrovascular disease, coronary artery disease, smoking within two weeks of surgery, preoperative percent predicted Forced Expiratory Volume in one second (FEV1), cancer stage, history of chronic obstructive pulmonary disease, extent of resection (wedge resection versus more extensive), and surgical approach (thoracotomy versus thoracoscopy). The variables that were significant at P < .20 were entered into a multivariable logistic regression with morbidity as the dependent variable and significance set at the 0.05 level. Data are presented as mean ± standard deviation unless otherwise noted. Survival was modeled using the Kaplan-Meier method, with the log rank test used to compare survival curves. The SAS 9.0 statistical package (SAS Institute, Cary, North Carolina) was used for statistical analyses.
Results
During the study period, 193 patients aged 80 years or older (median age 82 years, range 80 to >95) underwent lung resection. Demographic, baseline characteristics, comorbid conditions, and indications for surgery are shown in Table 1. 35 patients had previous thoracic surgery: 16 patients had previously undergone lung resection, 13 had previously undergone sternotomy for heart surgery, 2 patients had previously undergone thoracic aortic surgery, 1 patient had previously undergone an esophagectomy, and 3 patients had unknown previous thoracic surgery. The procedures performed were wedge resection in 77, segmentectomy in 13, lobectomy in 96, bilobectomy in 4, and pneumonectomy in 3 patients. There was not a statistically significant difference in percent predicted FEV1 between patients who had wedge resections and patients having more extensive resections (71±21 for wedge resections versus 77±21 for more extensive resections, p=0.06). History of congestive heart failure was the only variable of the demographics, baseline characteristics, and co-morbid conditions listed in table 1 that differed between patients with wedge resections and more extensive resections (12% for wedge versus 3% for more extensive resections, p=0.04). Resection was accomplished via thoracoscopy in 149 patients (77%). Percent predicted FEV1 was stastically similar for patients undergoing both thoracoscopy and thoracotomy (76±21 for thoracoscopy versus 71±18 for thoractomy, p=0.19).
Table 1.
Demographics, Baseline Characteristics, Comorbid Conditions, and indications for surgery.
| Characteristic | All (n=193) | Thoracoscopy (n=149) |
Thoracotomy (n=44) |
p- value |
|---|---|---|---|---|
| Age | 82.6±2.6 | 82.8±2.7 | 81.7±1.8 | 0.008 |
| Forced Expiratory Volume in one second (% predicted) |
75±21 | 76±21 | 71±18 | 0.19 |
| Diffusing Capacity of the Lung for Carbon Monoxide (% predicted) |
73±20 | 72±20 | 78±19 | 0.17 |
| American Society of Anesthesiology Score |
2.4±0.9 | 2.5±0.8 | 2.1±1.0 | 0.01 |
| Zubrod Status 0 or 1 | 176 (91%) | 134 (90%) | 42 (95%) | 0.4 |
| Tobacco abuse | 146 (76%) | 113 (76%) | 33 (75%) | 1 |
| Pack years | 44.4±31.7 | 44.2±30.8 | 44.9±34.8 | 0.9 |
| Smoking within 14 days | 11 (6%) | 9 (6%) | 2 (4.5%) | 1 |
| Hypertension | 110 (57%) | 85 (57%) | 25 (57%) | 1 |
| Coronary Artery Disease | 47 (24%) | 36 (24%) | 11 (25%) | 1 |
| Chronic Obstructive Pulmonary Disease |
44 (23%) | 35 (23%) | 9 (20%) | 0.8 |
| Previous Thoracic Surgery | 35 (18%) | 20 (13%) | 15 (34%) | 0.003 |
| Diabetes | 26 (13%) | 22 (15%) | 4 (9%) | 0.4 |
| Renal Insufficiency | 19 (10%) | 16 (11%) | 3 (7%) | 0.6 |
| Cerebrovascular Disease | 18 (9%) | 14 (9%) | 4 (9%) | 1 |
| Peripheral Vascular Disease | 17 (9%) | 14 (9%) | 3 (7%) | 0.8 |
| Congestive Heart Failure | 13 (7%) | 11 (7%) | 2 (4.5%) | 0.7 |
| Preoperative Weight Loss | 12 (6%) | 8 (5%) | 4 (9%) | 0.5 |
| Pre-operative Chemotherapy | 8 (4%) | 5 (3%) | 3 (7%) | 0.4 |
| Pre-operative Radiation | 8 (4%) | 5 (3%) | 3 (7%) | 0.4 |
| Stroke | 4 (2%) | 3 (2%) | 1 (2%) | 1 |
| Steroid Use | 2 (1%) | 0 | 2 (4.5%) | 0.05 |
| Indications for Surgery | ||||
| Primary Lung Cancer | ||||
| Pathologic Stage 1 | 109 (56%) | 88 (59%) | 21 (48%) | 0.23 |
| Pathologic Stage 2 | 19 (10%) | 9 (6%) | 10 (23%) | 0.003 |
| Pathologic Stage 3 | 18 (9%) | 11 (7%) | 7 (16%) | 0.13 |
| Pathologic Stage 4 | 5 (3%) | 4 (3%) | 1 (2%) | 1 |
| Benign Etiology | 26 (13%) | 23 (15%) | 3 (7%) | 0.2 |
| Metastatic Lesion | 15 (8%) | 14 (9%) | 1 (2%) | 0.2 |
| Mesothelioma | 1 (0.5%) | 0 | 1 (2%) | 0.2 |
The median hospitalization was 4 days (range 1 to 27 days). Operative mortality was 3.6% (7 patients) and morbidity was 46% (89 patients). Patient discharge disposition was directly to home in 181 patients (94%), while one patient (0.5%) was discharged to an acute rehabiliation and three patients (1.5%) were discharged to a skilled nursing facililty. Five patients (2.6%) died in the hospital, one patient (0.5%) was discharged to hospice, and the discharge disposition could not be determined in two patients (1%). Specific postoperative events for all patients are listed in Table 2. The most common events were atrial arrhythmia in 38 patients (20%), prolonged air leak in 24 patients (12%), postoperative transfusion in 22 patients (11%), delirium in 16 patients (8%), need for bronchoscopy in 14 patients (7%), and pneumonia in 10 patients (5%). The 1-year and 3-year overall survival of 109 patients who underwent resection for pathologic stage I lung cancer was 85% and 56% (Figure 1A).
Table 2.
Postoperative Events.
| Event | Number of Patients (%) |
|---|---|
| Mortality (Thirty-day) | 7 (3.6 %) |
| Morbidity (Thirty-day) | 89 (46%) |
| Hospital Stay (days) | 4.9±3.5 |
| Atrial arrhythmia | 38 (20%) |
| Need for chest tube > 5 days | 24 (12%) |
| Post-operative transfusion | 22 (11%) |
| Delirium/Mental Status Changes | 16 (8%) |
| Post-operative bronchoscopy | 14 (7%) |
| Pneumonia | 10 (5%) |
| Need for New Chest Tube | 9 (5%) |
| Urinary Tract Infection | 9 (5%) |
| Reintubation | 6 (3%) |
| Need for enteral nutrition tube | 3 (2%) |
| New Renal Failure | 3 (2%) |
| Need for tracheostomy | 3 (2%) |
| Phlebitis | 2 (1%) |
| New Neurological Event | 2 (1 %) |
| Fall | 2 (1%) |
| Pulmonary embolism/Deep Venous Thrombosis | 2 (1%) |
| Vocal cord paralysis | 1 (0.5%) |
| Myocardial infarction | 1 (0.5%) |
| Empyema | 1 (0.5%) |
| Clostridium Difficile Colitis | 1 (0.5%) |
Figure 1.
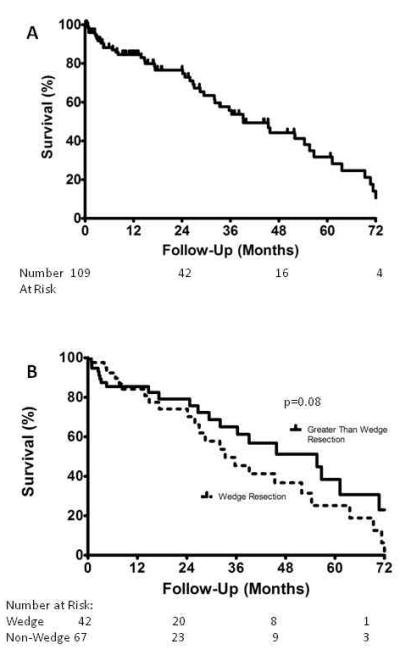
A) Kaplan-Meier analysis of overall survival after resection of pathologic stage I lung cancer. B) Kaplan-Meier analysis of overall survival after resection of pathologic stage I lung cancer for patients undergoing wedge resection and greater than wedge resection.
On univariate analysis using the predictors listed in Table 3, resection greater than wedge, use of a thoracotomy, and FEV1were significantly associated with morbidity. These predictors remained significant in the multivariable logistic model [resection greater than wedge (odds ratio 2.98, p=0.00), use of a thoracotomy (odds ratio 2.6, p=0.03), and FEV1 (odds ratio 1.28 for each 10% decrement, p=0.01)] (Table 4). Table 5 stratifies morbidity for several ranges of FEV1 for all patients and by operative approach and extent of resection. Table 5 stratifies morbidity for several ranges of FEV1 for all patients and by operative approach and extent of resection.
Table 3.
Univariate analysis of risk factors for morbidity after lung resection in octogenarians.
| Variable | Patients with Complications (n=89) |
Patients without Complications (n=104) |
p |
|---|---|---|---|
| Forced Expiratory Volume in one second (% predicted) per 10% decrease |
69.5±18.9 | 78.9±21.5 | 0.006 |
| Resection Greater Than Wedge | 67 (75%) | 49 (47%) | <0.001 |
| Surgical Approach (Thoracotomy versus Thoracoscopy) |
32 (36%) | 12 (12%) | 0.0001 |
| History of Diabetes | 8 (9%) | 18 (17%) | 0.1 |
| Age | 82.3±2.1 | 82.8±2.9 | 0.17 |
| History of Coronary Artery Disease | 24 (27%) | 23 (22%) | 0.43 |
| Smoking Within 2 Weeks of Surgery | 5 (6%) | 6 (6%) | 0.96 |
| History of Cerebrovascular Disease | 8 (9%) | 10 (10%) | 0.9 |
| History of Peripheral Vascular Disease | 7 (8%) | 10 (10%) | 0.67 |
| Use of Induction Chemotherapy | 5 (6%) | 3 (3%) | 0.35 |
| Use of Induction Radiation | 5 (6%) | 3 (3%) | 0.35 |
| History of Tobacco Use | 73 (82%) | 73 (70%) | 0.37 |
| Previous Thoracic Surgery | 18 (20%) | 17 (16%) | 0.49 |
| History of Chronic Obstructive Pulmonary Disease |
25 (28%) | 19 (18%) | 0.11 |
| Cancer Stage | 0.25 |
Table 4.
Logistic regression model of risk factors for morbidity after lung resection in octogenarians.
| Variable | Odds Ratio | 95% CI | P |
|---|---|---|---|
| Forced Expiratory Volume in one second (% predicted) per 10% decrease |
1.28 | 1.05-1.54 | 0.01 |
| Resection Greater Than Wedge | 2.98 | 1.36-6.51 | 0.006 |
| Surgical Approach (Thoracotomy versus Thoracoscopy) |
2.61 | 1.08-6.30 | 0.03 |
| History of Diabetes | 0.35 | 0.11-1.08 | 0.07 |
| Age | 1.01 | 0.88-1.17 | 0.87 |
| History of Chronic Obstructive Pulmonary Disease |
2.23 | 0.92-5.43 | 0.08 |
Table 5.
Morbidity stratified by pulmonary function, operative approach, and extent of resection.
| % predicted FEV1 | |||
|---|---|---|---|
| <45 | 45-60 | >60 | |
| All Patients | 64% | 52% | 43% |
| Operative Approach | |||
| Thoracoscopy | 60% | 41% | 36% |
| Thoracotomy | 100% | 64% | 71% |
| Extent of Resection | |||
| Wedge | 57% | 36% | 26% |
| Greater than Wedge | 75% | 65% | 52% |
Because the multivariable analysis identified resection extent and operative approach as independent significant predictors of morbidity, complications are also stratified by resection extent and operative approach for patients with stage 1 lung cancer, which is probably the most common clinical situation when a surgeon considers a resection less than a lobectomy for lung cancer (Table 6). The majority of octogenarians who underwent a thoracotomy for treatment of pathologic stage 1 lung cancer had a complication (16 of 21 patients, 76%). The morbidity after resections more extensive than a wedge was much less in patients who underwent thoracoscopy compared to thoracotomy [48% versus 88%, p=0.004]. There were no statistically significant differences in mortality (p=0.63), morbidity (p=0.08), and hospital stay (p=0.29) between wedge resections and more extensive resections in the patients who had thoracoscopy. The difference in long-term survival in patients treated for pathologic stage 1 lung cancer by wedge resection and more extensive resection was not statistically significant (Figure 1B).
Table 6.
Postoperative events for patients with pathologic stage I lung cancer stratified by approach and extent of resection.
| Event | All (n=109) | Thoracotomy (n=21) | Thoracoscopy (n=88) | ||
|---|---|---|---|---|---|
| Greater than wedge (n=17) |
Wedge (n=4) |
Greater than wedge (n=50) |
Wedge (n=38) |
||
| Mortality (Thirty-day) | 4 (3.7%) | 0 (0%) | 0 (0%) | 3 (6%) | 1 (2.6%) |
| Morbidity (Thirty-day) | 51 (47%) | 15 (88%) | 1 (25%) | 24 (48%) | 11 (29%) |
| Hospital Stay | 4.9±3.8 | 6.6±3.3 | 4.8±1.7 | 5.0±4.5 | 4.1±2.7 |
| Atrial arrhythmia | 19 (17%) | 8 (47%) | 0 | 8 (16%) | 3 (8%) |
| Need for chest tube > 5 days | 16 (15%) | 4 (24%) | 1 (25%) | 5 (10%) | 6 (16%) |
| Delirium/Mental Status Changes | 12 (11%) | 5 (29%) | 0 | 4 (8%) | 3 (8%) |
| Post-operative transfusion | 11 (10%) | 2 (12%) | 0 | 6 (12%) | 3 (8%) |
| Post-operative bronchoscopy | 7 (6%) | 2 (12%) | 1 (25%) | 3 (6%) | 1 (3%) |
| Pneumonia | 7 (6%) | 4 (24%) | 0 | 2 (4%) | 1 (3%) |
| Need for New Chest Tube | 7 (6%) | 3 (18%) | 0 | 2 (4%) | 2 (5%) |
| Urinary Tract Infection | 6 (6%) | 2 (12%) | 0 | 4 (8%) | 0 |
| Reintubation | 4 (4%) | 1 (6%) | 0 | 2 (4%) | 1 (3%) |
| Need for tracheostomy | 3 (3%) | 0 | 0 | 2 (4%) | 1 (3%) |
| Need for enteral nutrition tube | 2 (2%) | 0 | 0 | 1 (2%) | 1 (3%) |
| New Renal Failure | 2 (2%) | 1 (6%) | 0 | 0 | 1 (3%) |
| Fall | 2 (2%) | 0 | 0 | 1 (2%) | 1 (3%) |
| Phlebitis | 1 (1%) | 0 | 0 | 1 (2%) | 0 |
| New Neurological Event | 1 (1%) | 0 | 0 | 1 (2%) | 0 |
| Vocal cord paralysis | 1 (1%) | 0 | 0 | 1 (2%) | 0 |
| Pulmonary embolism/Deep Venous Thrombosis |
1 (1%) | 0 | 0 | 1 (2%) | 0 |
| Myocardial infarction | 1 (1%) | 0 | 0 | 1 (2%) | 0 |
| Clostridium Difficile Colitis | 1 (1%) | 0 | 0 | 1 (2%) | 0 |
Discussion
This study demonstrates that lung resections can be performed in patients over the age of 80 with acceptable overall morbidity and mortality (3.6% and 46%, respectively). The majority of octogenarians in this series did not experience a postoperative complication despite advanced age and significant comorbidities. In addition, most of the complications that did occur were not life-threatening and almost all of the patients were discharged to directly home after surgery. These results compare favorably to those published from other series of lung resection in octogenarians (Table 7) [11-16,20]. Age should not be the sole determinant when considering surgery as a treatment option for lung cancer or other pulmonary diseases in these elderly patients.
Table 7.
Results of studies reviewing complication rates and mortality in octogenarians undergoing pulmonary resections.
| Study | Years | Number of patients |
Morbidity | Mortality |
|---|---|---|---|---|
| Naunheim et al [14] | 1981-1991 | 40 | 45% overall, 30% major |
15% |
| Brock et al [13] | 1980-2002 | 68 | 83% overall | 8.8% |
| Port et al [12] | 1990-2003 | 61 | 38% overall, 13% major |
1.6% |
| Voltolini et al [20] | 1990-2005 | 96 | 18% major | 9.4% |
| Hope et al [11] | 1999-2004 | 20 | 45% overall, 20% major |
10% |
| Mun et al [24] | 1999-2006 | 55 | 25.6% overall | 3.6% |
| Igai et al [16] | 1982-2008 | 95 | 21% cardiopulmonary | 0 |
Thorough evaluation and planning must be undertaken prior to performing lung resection in an octogenarian. However, this analysis showed that worsening pulmonary function was the only pre-operative patient variable that independently predicted the occurrence of complications in this age group. This finding is not surprising, as pulmonary function has consistently been identified as important in stratifying the risks of lung resection in all patients [21,22]. Additionally, this analysis found that previous thoracic surgery, which was relatively common in the series and likely due to both the patients’ advanced age and presence of risk factors for cardiothoracic disease in general, did not increase risk of lung resection in this series. Interestingly, the other significant risk factors identified in our model are related to technical surgical factors. Although appropriate patient selection is clearly critical to optimizing outcomes when considering octogenarians for lung resection, operative planning and performance also clearly play an important role.
The use of thoracoscopy independently predicted improved outcomes in our study. This result is consistent with results from previous studies that have shown that a VATS approach to lobectomy in patients over the age of 70 with lung cancer reduces morbidity and hospital stay compared with thoracotomy [17,18]. These results suggest that thoracoscopy should be strongly considered as the approach of choice when technically possible in all elderly patients.
This study also found that performing a resection more extensive than a wedge was a risk for increased complications. A previous study has demonstrated that extended procedures predict mortality in patients older than 70 [10]. Balancing the potential oncologic benefits of an anatomic lung resection against the potential increased risks of more extensive surgery in octogenarians is difficult. However, the results of our study show that short term outcomes are equivalent for wedge resections and more extensive resections when thoracoscopy is used for stage 1 lung cancer, which is probably the most typical clinical situation when a surgeon might consider a wedge resection as treatment for primary lung cancer. These results suggest that a lobectomy should strongly be considered over a wedge resection for resection of stage 1 lung cancer in octogenarians if a thoracoscopic approach can be utilized. Of course, it must be acknowledged that some lesions may not be amenable to wedge resection either due to location or size and anatomic resection must be weighed against no resection when planning surgery.
The analysis did not show a statistically significant improvement in long-term survival for patients who had a more extensive resection than a wedge for stage 1 lung cancer, but the study was likely underpowered to detect a difference. Other studies have shown that the survival benefit conferred by lobectomy in the surgical treatment of lung cancer is a function of age, with lung cancer patients over 75 undergoing either lobectomy or lesser resection having no difference in overall survival [9]. Patients with extensive comorbidities and limited life expectancy might not realize the benefit of lobectomy over limited resection due to an increased rate of non-cancer deaths [23]. Statistical modeling is helpful in guiding decisions, but the final decision on how much surgery is most appropriate for a particular elderly patient comes down to clinical judgement while considering patient preferences.
The main strengths of this study compared to other series of lung resections in octogenarians are the relatively large number of patients in the series and that a significant number underwent a minimally invasive approach, allowing assessment of its effect on outcomes. The main limitation of this study is the retrospective nature. While we attempted to control for all known contributors, it is impossible to completely control selection bias in a retrospective study. In particular, only patients that had already been deemed appropriate surgical candidates and undergone surgery were contained in the study. The study therefore only allows for fine-tuning of risk assessment, as the risk model does not contain the entire population of octogenarians who may have had a potential indication for lung surgery. A significant proportion of patients who would be considered particularly high-risk may have been screened out of the study. In addition, the heterogenity of the study cohort may limit the ability to directly translate these study results to an octogenarian undergoing lung resection for a specific indication. The procedures in this report were also performed by several different surgeons with variable experiences with thoracoscopic approaches, which introduces potential confounders such as different selection criteria for surgery and planned extent of resection, surgical techniques, and postoperative management. Finally, the positive effects of approach on outcome in this study cannot necessarily be immediately generalized to all thoracic surgeons, considering the extensive experience of the surgeons in this study with thoracoscopic surgery.
In conclusion, lung resections can be performed safely in patients 80 years of age and older. Optimal patient selection and operative planning and performance are critical to minimizing morbidity. As with other age groups, pulmonary function tests are the most important patient factors that allow risk stratification. A thoracoscopic approach reduces the morbidity, as does performing more limited resections. Careful clinical judgement is needed to plan the most appropriate surgery for each individual patient.
Footnotes
Presented at the 18th European Conference on General Thoracic Surgery, Valladolid, Spain May 31, 2010
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Harpole DH, Jr., DeCamp MM, Jr., Daley J, Hur K, Oprian CA, Henderson WG, Khuri SF. Prognostic models of thirty-day mortality and morbidity after major pulmonary resection. J Thorac Cardiovasc Surg. 1999;117:969–979. doi: 10.1016/S0022-5223(99)70378-8. [DOI] [PubMed] [Google Scholar]
- 2.Brokx HA, Visser O, Postmus PE, Paul MA. Surgical treatment for octogenarians with lung cancer: results from a population-based series of 124 patients. J Thorac Oncol. 2007;2:1013–1017. doi: 10.1097/JTO.0b013e3181559fdf. [DOI] [PubMed] [Google Scholar]
- 3.Owonikoko TK, Ragin CC, Belani CP, Oton AB, Gooding WE, Taioli E, Ramalingam SS. Lung cancer in elderly patients: an analysis of the surveillance, epidemiology, and end results database. J Clin Oncol. 2007;25:5570–5577. doi: 10.1200/JCO.2007.12.5435. [DOI] [PubMed] [Google Scholar]
- 4.Sigel K, Bonomi M, Packer S, Wisnivesky J. Effect of age on survival of clinical stage I non-small-cell lung cancer. Ann Surg Oncol. 2009;16:1912–1917. doi: 10.1245/s10434-009-0475-8. [DOI] [PubMed] [Google Scholar]
- 5.Dillman RO, Zusman DR, McClure SE. Surgical resection and long-term survival for octogenarians who undergo surgery for non-small-cell lung cancer. Clin Lung Cancer. 2009;10:130–134. doi: 10.3816/CLC.2009.n.017. [DOI] [PubMed] [Google Scholar]
- 6.Satoh H, Kurishima K, Nakamura R, Ishikawa H, Kagohashi K, Ohara G, Ishikawa S, Ohtsuka M, Sekizawa K, Hizawa N. Lung cancer in patients aged 80 years and over. Lung Cancer. 2009;65:112–118. doi: 10.1016/j.lungcan.2008.10.020. [DOI] [PubMed] [Google Scholar]
- 7.Suemitsu R, Yamaguchi M, Takeo S, Ondo K, Ueda H, Yoshino I, Maehara Y. Favorable surgical results for patients with nonsmall cell lung cancer over 80 years old: a multicenter survey. Ann Thorac Cardiovasc Surg. 2008;14:154–160. [PubMed] [Google Scholar]
- 8.Hanna N, Brooks JA, Fyffe J, Kesler K. A retrospective analysis comparing patients 70 years or older to patients younger than 70 years with non-small-cell lung cancer treated with surgery at Indiana university: 1989-1999. Clin Lung Cancer. 2002;3:200–204. doi: 10.3816/clc.2002.n.003. [DOI] [PubMed] [Google Scholar]
- 9.Mery CM, Pappas AN, Bueno R, Colson YL, Linden P, Sugarbaker DJ, Jaklitsch MT. Similar long-term survival of elderly patients with non-small cell lung cancer treated with lobectomy or wedge resection within the surveillance, epidemiology, and end results database. Chest. 2005;128:237–245. doi: 10.1378/chest.128.1.237. [DOI] [PubMed] [Google Scholar]
- 10.Sirbu H, Schreiner W, Dalichau H, Busch T. Surgery for non-small cell carcinoma in geriatric patients: 15-year experience. Asian Cardiovasc Thorac Ann. 2005;13:330–336. doi: 10.1177/021849230501300408. [DOI] [PubMed] [Google Scholar]
- 11.Hope WW, Bolton WD, Kalbaugh CA, Blackhurst DW, Stephenson JE, Taylor SM. Lung cancer resection in octogenarians: a reasonable approach for our aging population. Am Surg. 2007;73:22–24. [PubMed] [Google Scholar]
- 12.Port JL, Kent M, Korst RJ, Lee PC, Levin MA, Flieder D, Altorki NK. Surgical resection for lung cancer in the octogenarian. Chest. 2004;126:733–738. doi: 10.1378/chest.126.3.733. [DOI] [PubMed] [Google Scholar]
- 13.Brock MV, Kim MP, Hooker CM, Alberg AJ, Jordan MM, Roig CM, Xu L, Yang SC. Pulmonary resection in octogenarians with stage I nonsmall cell lung cancer: a 22-year experience. Ann Thorac Surg. 2004;77:271–277. doi: 10.1016/s0003-4975(03)01470-x. [DOI] [PubMed] [Google Scholar]
- 14.Naunheim KS, Kesler KA, D’Orazio SA, Fiore AC, Judd DR. Lung cancer surgery in the octogenarian. Eur J Cardiothorac Surg. 1994;8:453–456. doi: 10.1016/1010-7940(94)90013-2. [DOI] [PubMed] [Google Scholar]
- 15.Finlayson E, Fan Z, Birkmeyer JD. Outcomes in octogenarians undergoing high-risk cancer operation: a national study. J Am Coll Surg. 2007;205:729–734. doi: 10.1016/j.jamcollsurg.2007.06.307. [DOI] [PubMed] [Google Scholar]
- 16.Igai H, Takahashi M, Ohata K, Yamashina A, Matsuoka T, Kameyama K, Nakagawa T, Okumura N. Surgical treatment for non-small cell lung cancer in octogenarians--the usefulness of video-assisted thoracic surgery. Interact Cardiovasc Thorac Surg. 2009;9:274–277. doi: 10.1510/icvts.2008.199455. [DOI] [PubMed] [Google Scholar]
- 17.Berry MF, Hanna J, Tong BC, Burfeind WR, Jr, Harpole DH, D’Amico TA, Onaitis MW. Risk factors for morbidity after lobectomy for lung cancer in elderly patients. Ann Thorac Surg. 2009;88:1093–1099. doi: 10.1016/j.athoracsur.2009.06.012. [DOI] [PubMed] [Google Scholar]
- 18.Cattaneo SM, Park BJ, Wilton AS, Seshan VE, Bains MS, Downey RJ, Flores RM, Rizk N, Rusch VW. Use of video-assisted thoracic surgery for lobectomy in the elderly results in fewer complications. Ann Thorac Surg. 2008;85:231–235. doi: 10.1016/j.athoracsur.2007.07.080. [DOI] [PubMed] [Google Scholar]
- 19.Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, Carbone PP. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol. 1982;5:649–655. [PubMed] [Google Scholar]
- 20.Voltolini L, Rapicetta C, Ligabue T, Luzzi L, Scala V, Gotti G. Short- and long-term results of lung resection for cancer in octogenarians. Asian Cardiovasc Thorac Ann. 2009;17:147–152. doi: 10.1177/0218492309103295. [DOI] [PubMed] [Google Scholar]
- 21.Kearney DJ, Lee TH, Reilly JJ, DeCamp MM, Sugarbaker DJ. Assessment of operative risk in patients undergoing lung resection. Importance of predicted pulmonary function. Chest. 1994;105:753–759. doi: 10.1378/chest.105.3.753. [DOI] [PubMed] [Google Scholar]
- 22.Ferguson MK. Preoperative assessment of pulmonary risk. Chest. 1999;115:58S–63S. doi: 10.1378/chest.115.suppl_2.58s. [DOI] [PubMed] [Google Scholar]
- 23.Landreneau RJ, Sugarbaker DJ, Mack MJ, Hazelrigg SR, Luketich JD, Fetterman L, Liptay MJ, Bartley S, Boley TM, Keenan RJ, Ferson PF, Weyant RJ, Naunheim KS. Wedge resection versus lobectomy for stage I (T1 N0 M0) non-small-cell lung cancer. J Thorac Cardiovasc Surg. 1997;113:691–698. doi: 10.1016/S0022-5223(97)70226-5. [DOI] [PubMed] [Google Scholar]
- 24.Mun M, Kohno T. Video-assisted thoracic surgery for clinical stage I lung cancer in octogenarians. Ann Thorac Surg. 2008;85:406–411. doi: 10.1016/j.athoracsur.2007.10.057. [DOI] [PubMed] [Google Scholar]


