Abstract
This study examined the association between maternal cocaine use and maternal behavior and tested a conceptual model predicting maternal insensitivity during mother–infant interactions. Participants included 130 mother–infant dyads (68 cocaine-exposed and 62 noncocaine-exposed) who were recruited after birth and assessed at 4–8 weeks of infant age. Results of model testing indicated that when the effects of prenatal cocaine use were examined in the context of polydrug use, maternal psychopathology, maternal childhood history, and infant birth weight, only postnatal cocaine use and maternal depression/anxiety were unique predictors of maternal insensitivity during mother–infant interactions.
Keywords: cocaine, depression, maternal behavior, substance use
The teratological impact of prenatal cocaine exposure on specific aspects of development is still under study. However, there is a growing recognition that one pathway to later developmental problems among cocaine-exposed infants may be through problematic maternal behavior or parenting difficulties. For instance, cocaine-using mothers have been reported to be more disengaged and passive during mother–infant interactions in the neonatal period (Gottwald & Thurman, 1994; Molitor, Mayes, & Ward, 2003), to have lower responsiveness and enthusiasm in later infancy (Burns, Chethik, Burns, & Clark, 1991, 1997), to be less positively reinforcing and use more threats of physical discipline in the toddler/preschool period (Bauman & Dougherty, 1983), and to be more hostile and intrusive and less supportive in a structured teaching situation at 3 years of age (Johnson et al., 2002). Others have specifically measured behavior during feeding, a naturalistic context for interactions in infancy. These results indicate that cocaine-using mothers are less flexible and engaged during feeding interactions with their 1-month-old infants compared with the control group (LaGasse et al., 2003) and have higher dyadic conflict with their infants (Eiden, 2001). However, the literature is by no means unequivocal. Several studies have noted no association between maternal cocaine use and parenting difficulties (M. Black, Schuler, & Nair, 1993; Johnson & Rosen, 1990; Neuspiel, Hamel, Hochberg, Greene, & Campbell, 1991). These disparate findings may be explained not only by differences in timing of measurement and varying sample sizes but also by the large within-group heterogeneity on variables such as placement in foster care and other maternal and infant risk characteristics. Notable among these risk characteristics are comorbid maternal psychopathology, use of other substances, continued postnatal substance abuse, and compromised infant growth outcomes.
Several studies have noted the association between maternal cocaine use and higher postpartum psychological distress (Singer et al., 1995), higher depression and anxiety (Espinosa, Beckwith, Howard, Tyler, & Swanson, 2001; Hawley, Halle, Drasin, & Thomas, 1995), paranoia (Espinosa et al., 2001), and antisocial personality disorder (Eiden, 2001; Espinosa et al., 2001). There is now a large body of literature indicating that depressed mothers are more likely to display flatter affect during mother–child interactions, provide less stimulation, and be less responsive toward their infants (Cohn & Campbell, 1992; Jameson, Gelfand, Kulcsar, & Teti, 1997). Maternal anger/hostility and antisocial behavior have also been found to exacerbate negative maternal behavior in substance-using populations (Eiden, Chavez, & Leonard, 1999; Hans, Bernstein, & Henson, 1999). Thus, comorbid maternal psychopathology may mediate or moderate the association between maternal cocaine use and maternal behavior during feeding.
In addition, maternal cocaine use is a polydrug issue. The majority of mothers using cocaine also use other substances, such as alcohol and marijuana. Often, cocaine is smoked in combination with marijuana and nicotine. Cocaine use is also often accompanied by heavy drinking during pregnancy. Several studies have reported that mothers who continue to drink during pregnancy are less responsive and stimulating toward their infants in the first year of life (O'Connor, Sigman, & Kasari, 1993). Thus, the potential effects of cocaine use need to be considered in the context of other substance use during pregnancy. Moreover, continued maternal substance abuse in the postnatal period is likely to have a significant impact on the quality of parenting. As noted by other researchers (e.g., Schuler, Nair, & Black, 2002), women who continue to use drugs after the infant's birth are less likely to be able to provide a stable, nurturing environment for their children. The combination of cocaine and other substance use during prenatal and postnatal periods is likely to increase the risk for negative maternal behavior after delivery.
Prenatal cocaine exposure is also associated with poorer infant growth outcomes. Both norepinephrine- and serotonin-related effects of cocaine on vascular tone have been reported, resulting in decreased placental blood flow and fetal vasoconstriction (Institute of Medicine, 1996). This reduced blood flow has been implicated as a causal mechanism for the effects of prenatal cocaine exposure on poor fetal growth (Handler, Kistin, Davis, & Ferre, 1991; Oro & Dixon, 1987). Further, maternal cocaine use is also associated with poor maternal nutrition and lack of prenatal care, thus exacerbating the likelihood of poor fetal growth (e.g., low birth weight, being small for gestational age, intrauterine growth retardation, and prematurity) in this group of children (Amaro, Zuckerman, & Cabral, 1989). Studies have noted the association between growth outcomes such as low birth weight and more negative maternal behavior among cocaine-using mothers (Singer, Hawkins, Huang, Davillier, & Baley, 2001).
Finally, several studies have demonstrated the potential etiological association between childhood experiences of abuse and later substance abuse among women (Bulik, Prescott, & Kendler, 2001; Schuck & Widom, 2001). Childhood experience of abuse has been associated with both later substance abuse and other psychopathology, such as depression and anxiety, in retrospective studies (e.g., Schuck & Widom, 2001). In addition, models of child development have noted the importance of parents' own childhood experiences in predicting both parental personality and parenting behavior (Belsky, 1984; Snyder & Huntley, 1990). Thus, maternal reports of child abuse may be causally linked to both maternal psychopathology and substance abuse, in addition to having direct associations with parenting behavior.
The purpose of this study was to examine the association between prenatal and postnatal cocaine use and maternal behavior in a feeding context between 4 and 8 weeks of infant age. We hypothesized that cocaine-using mothers would display lower warmth and higher insensitivity during feeding. Next, we tested a conceptual model in which we examined the hypothesis that current levels of maternal depression and anger and lower infant birth weight would help explain this association. That is, cocaine-using mothers would have higher depression and anger, and these variables would in turn be associated with lower maternal warmth and higher insensitivity toward their infants. Similarly, we hypothesized that infants exposed to cocaine prenatally would have lower birth weight, and infants with lower birth weight would elicit lower maternal warmth and higher insensitivity. This model (see Figure 1) included associations between cocaine and other substance use, pregnancy and postnatal substance use, and paths from these variables to maternal behavior. Finally, we included maternal reports of child abuse as a predictor of maternal psychopathology and substance abuse, in addition to direct associations with mothers' behavior toward their infants.
Figure 1.
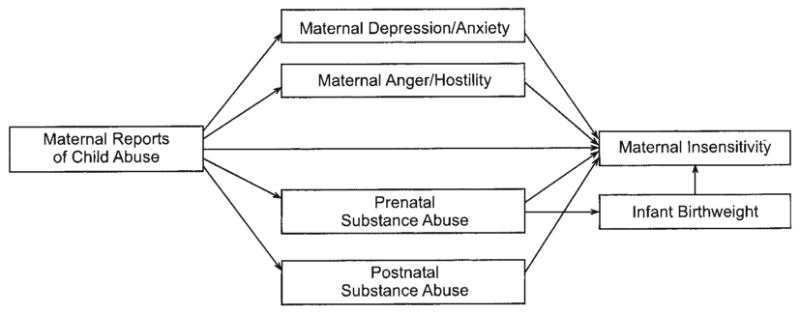
Conceptual model. Not depicted in this model but included in model estimation are associations among maternal depression/anxiety, anger/hostility, prenatal substance abuse, and postnatal substance abuse.
Method
Participants
Participants consisted of 84 cocaine-exposed and 63 comparison mother–infant dyads recruited into an ongoing longitudinal study of maternal substance use and child development. For the purposes of this study, 17 infants who were placed in foster care were excluded from data analysis. This resulted in 68 cocaine-exposed and 62 comparison mother–infant dyads. An outreach worker on the project staff recruited all participants from two local area hospitals after the infants' delivery. Mothers ranged in age from 18 to 42 years (M = 30.52, SD = 5.45). The majority of the mothers were African American (72%), were receiving Temporary Assistance for Needy Families (70%) at the time of their first laboratory visit (Years 2001–2004), and were single (60%). Once a mother–child dyad was recruited into the cocaine group, the closest matching noncocaine-using mother was recruited from the same hospital on the basis of maternal education, maternal age, maternal race/ethnicity, and infant gender. Fortyseven percent of the infants were male. Eighty-six percent of the cocaine-exposed infants and 97% of the comparison infants were full term (≥37 weeks gestational age). The study received approval from the institutional review boards of the hospitals as well as the primary institutions with which the authors were affiliated. In addition, informed written consent was obtained from all recruited participants, and Health Insurance Portability and Accountability Act authorization was obtained from all participants after April 2003. Participants received $35 in monetary incentives in the form of gift certificates, checks, and baby toys at the 4–8-week visit.
Procedure
All mothers at two local hospitals were screened after delivery for initial eligibility and matching criteria. Interested and eligible mothers were given detailed information about the study and asked to sign consent forms. About 2 weeks after delivery, mothers were contacted and scheduled for their first laboratory visit, which took place at the time that their infant was approximately 4–8 weeks old. This visit consisted of a maternal interview, a 10-min feeding session, and infant and mother physiological assessments. Because of concerns about maternal literacy, all measures were administered in an interview format.
Of the 4,619 women screened at delivery, 293 were eligible for participation. Of these, 35% either declined participation or were not enrolled in the study because they expressed initial interest but later withdrew. Mothers who participated were more likely to be between 18 and 25 years of age compared with those who were eligible but not enrolled in the study, χ2(2, N = 130) = 19.15, p < .001, and were more likely to have a high school or below education, χ2(2, N = 130) = 45.79, p < .001. Mothers who participated were also more likely to be in the cocaine group (90% of the mothers in the cocaine group participated) compared with those who were eligible but not enrolled, χ2(1, N = 130) = 50.26, p < .001. The majority of the mothers in the cocaine group who were eligible but not enrolled in the study had children who were placed in nonmaternal care. There were no other differences on any of the demographic variables between those who participated and those who were eligible but not enrolled.
Assessment of Growth and Risk Status
Three measures of growth were used in this study: birth weight (in grams), birth length (in centimeters), and head circumference (in centimeters). All measurements were taken by obstetrical nurses in the delivery room and recorded in the infant's medical chart. Research staff recorded this information from the charts after recruiting the mother–infant dyad. Medical chart review at the time of recruitment was also used to complete the Obstetrical Complication Scale (Littman & Parmelee, 1978), a scale designed to assess the number of perinatal risk factors experienced by the infant. Higher numbers on this scale indicate lower risk status.
Identification of Substance Use
Prenatal cocaine use was determined by a combination of maternal report, chart review, and maternal hair analysis. Urine toxicologies were routinely conducted at the first prenatal visit on maternal urine and/or at delivery (for those mothers who tested positive prenatally, obtained prenatal care elsewhere, or did not receive any prenatal care) on infant and maternal urine by participating hospitals. Thus, toxicology results were available through chart review for 129 (99%) of the mothers. Hair analysis was available for 101 (78%) of the mothers. Eleven percent of the sample refused to give a hair sample, and the remaining mothers had hair that was too short for analysis. Mothers were included in the cocaine group if self-reports were positive, regardless of urine toxicology or hair sample results. Similarly, mothers who reported that they did not use cocaine but had positive urine toxicology or hair samples were included in the cocaine group.
Urine toxicologies consisted of standard urine screening for drug level or metabolites of cocaine, opiates, benzodiazepines, and tetrahydrocannabinol. Urine was rated positive if the quantity of drug or metabolite was above 300 g/mL. All mothers recruited into the study were negative for opiates and benzodiazepines. Hair samples were collected from the mothers at the first laboratory visit and sent to Psychemedics Corporation (Acton, MA) for radioimmunoanalyses (RIAH). Drugs and their metabolites are absorbed into the hair and can be extracted and measured. As hair grows at an average rate of half an inch per month, it can record a pattern of drug consumption related to the amount and frequency of use (see Baumgartner, Hill, & Blahd, 1989). Thus, a 2-in. length of hair could contain a record of approximately 4 months of use, and given adequate hair length (∼4–5 in.), use per trimester may be recorded. Drugs become detectable in hair about 3–4 days after use, a time when cocaine is rendered undetectable by urinalysis. RIAH is the most well established hair analysis technique, and it has been replicated by independent laboratories across the world (see Magura, Freeman, Siddiqi, & Lipton, 1992). Gas chromatography/mass spectrometry confirmations of RIAH have not revealed any false positives because of testing errors (see Magura et al., 1992).
Approximately 32% of mothers in the study (55% of the mothers in the cocaine group) had positive urine toxicologies at delivery, and 25% of mothers (79% of the mothers in the cocaine group) had hair samples that tested positive for cocaine during pregnancy. The remainder of mothers in the cocaine group admitted having used cocaine in the brief, self-report screening instrument administered after delivery. Mothers in the comparison group reported not having used any illicit substances other than marijuana. Additional exclusionary criteria for all mothers consisted of the following: maternal age of 18 or younger, use of illicit substances other than cocaine or marijuana, and significant medical problems for the infant (e.g., genetic disorders, major perinatal complications, baby in critical care for over 48 hr).
Continuous Measures of Substance Use
To assess maternal substance use before, during, and after pregnancy, participants completed the Timeline Follow-Back Interview (TLFB; Sobell, Sobell, Klajner, Pavan, & Basian, 1986). Participants were provided a calendar and asked to identify events of personal interest (holidays, birthdays, vacations, etc.) as anchor points to aid recall. This technique has been established as a reliable and valid method of obtaining longitudinal data on substance use patterns; it has good test–retest reliability and is highly correlated with other intensive self-report measures (Brandon, Copeland, & Saper, 1995; Brown, Bakeman, Coles, Sexson, & Demi, 1998). The TLFB yielded data about the number of days of cocaine use, number of cigarettes smoked, number of marijuana joints smoked, and alcohol binges (five or more standard drinks) for each trimester and in the postnatal period. Thus, all continuous measures of prenatal and postnatal cocaine use used in model testing were based on self-report alone.
Maternal Depression/Anxiety
The Brief Symptom Inventory (Derogatis, 1993), a brief form of the Symptom Checklist 90—Revised, is a widely used mental health screening measure in a variety of clinical and research settings. It consists of 53 items rated on a 5-point scale. The items are grouped into nine scales: Anxiety, Hostility, Somatization, Obsessive–Compulsive, Interpersonal Sensitivity, Depression, Phobic Anxiety, Paranoid Ideation, and Psychoticism. Because we were specifically interested in the association between maternal depression/anxiety and maternal behavior, we used the average of the Depression, Anxiety, and Phobic Anxiety scales as a measure of maternal depression/anxiety. The internal consistency of this scale was quite high (coefficient α = .82). High scores on this scale indicate high depression/anxiety.
Maternal Anger/Hostility
The Buss and Perry Aggression Questionnaire (Buss & Perry, 1992) was used to measure maternal anger and hostility. This questionnaire consists of four scales: Physical Aggression, Verbal Aggression, Anger, and Hostility. For this study, a total score was derived by averaging the four scales along with the Hostility scale from the Brief Symptom Inventory (see above). The internal consistency of this scale was .81. High scores on this scale indicate higher anger/hostility.
Childhood History of Abuse
The Childhood Trauma Questionnaire (Fink, Bernstein, Handelsman, Foote, & Lovejoy, 1995) was used to assess childhood history of abuse. This measure consists of five scales that assess childhood emotional, physical, and sexual abuse and emotional and physical neglect. A composite score reflecting child abuse was created by taking the average of the emotional, physical, and sexual abuse scales. The internal consistency of this scale was high (coefficient α = .84).
Maternal Insensitivity and Maternal Warmth
The 4–8-week laboratory visit was scheduled around a time when the infant was likely to be hungry. Mothers were asked to feed their infants in a comfortable, living room type setting as they normally would at home and were videotaped. The first 10 min of these interactions were coded by two research assistants blind to group status, using the Mother–Infant Feeding Scale (Chatoor, 1986). For the purposes of this study, 18 of the original 26 maternal items were coded at the 4–8-week visit. The remaining items were not included owing to low variability at 4–8 weeks of infant age. For these items, the scale requires attention to affect (body posture, facial expression), sensitivity to infant cues, nonverbal communication (positioning the infant for eye contact), verbal communication, and feeding behaviors. This scale has been used by several researchers to measure mother–infant feeding interactions among children with eating disorders (Chatoor & Egan, 1986; Chatoor, Schaefer, Dickson, & Egan, 1984) and in previous studies of mother–infant feeding interactions among substance abusing mothers (Eiden, 2001). However, little information is available about the factor structure of the scales in high-risk samples not identified as having feeding problems.
Two coders blind to group status were trained to code mother–infant interactions by Rina D. Eiden until interrater reliability criterion was reached (agreement of 90% or above). Subsequent interrater reliability was established on 22% of the tapes, with both coders viewing these tapes separately and comparing codes subsequently. If the coders disagreed by more than 1 point on any dimension, they arrived at a final code by viewing the tape again and resolving discrepancies by mutual agreement. Interrater reliability on individual items ranged from Pearson r = .71 (Cohen's κ = .53) to r = .98 (Cohen's κ = .95).
To reduce the number of observational variables for parenting, and following previous studies (e.g., Eiden et al., 1999), we conducted a principal-components analysis with varimax rotation using the individual maternal behavior items at 4–8 weeks. This initial analysis indicated high eigenvalues for the first two factors (>3) and lower eigenvalues (∼1) on five other factors. Thus, this analysis was repeated with a two-factor solution. The factor loadings for the individual items are displayed in Table 1. The first factor, labeled Maternal Insensitivity, had an eigenvalue of 4.57 and explained 24.03% of the variance. The second factor, labeled Maternal Warmth, had an eigenvalue of 3.36 and explained 17.69% of the variance.
Table 1. Factor Loadings for Maternal Behavior During Feeding.
| Variable | Factor 1 (Maternal Insensitivity) |
Factor 2 (Maternal Warmth) |
|---|---|---|
| Talks to infant | .85 | |
| Positive remarks | .69 | |
| Positive statements about food | .53 | |
| Pleasure toward infant | .80 | |
| Appears cheerful | .76 | |
| Appears sad | −.50 | |
| Appears detached | −.65 | |
| Position without support | .73 | |
| Holds stiffly | .68 | |
| Restricts movements | .64 | |
| Handles excessively | .78 | |
| Misses infant cues | .46 | |
| Makes critical remarks | .56 | |
| Negative statements about food | .55 | |
| Appears angry | .47 | |
| Controls feeding, overriding infant cues | .45 | |
| Concerned about messiness | .59 | |
| Handles roughly | .80 |
Results
Sample Characteristics
Descriptive statistics for the demographic and risk variables for both mothers and infants in the cocaine and comparison groups are presented in Tables 2 and 3. Results of multivariate analyses of variance (MANOVAs) with group status as the independent variable and biological mothers' age, education, socioeconomic status, parity, and number of prenatal care visits as the dependent variables yielded a significant multivariate effect of group, F(5, 122) = 8.94, p < .001. Step-down analyses were conducted with maternal education, age, socioeconomic status, parity, and number of prenatal visits, in that order, with alpha set at p < .01. These analyses indicated that mothers who used cocaine during pregnancy were significantly older and had more education compared with non-cocaine-using women (see Table 2). Results of MANOVAs with group status as the independent variable and infant risk variables as the dependent variables yielded a significant multivariate effect of group, F(5, 122) = 5.64, p < .001. Step-down analyses were conducted with infant gestational age, birth weight, head circumference, birth length, and obstetrical complications, in that order, with alpha set at p < .01. These analyses indicated that infants of women in the cocaine group had lower gestational age, lower birth weight, and a higher number of obstetrical complications compared with non-cocaine-using women. Self-reported cocaine use among women in the cocaine-using group ranged from 0 to 7 days a week during the month before conception and during each trimester of pregnancy, and from 0 to 3 days per week in the month after delivery (see Table 2). About 85% of mothers in the cocaine group abstained during pregnancy according to self-report on the TLFB, and 15% continued cocaine use in the month after delivery. It is important to note that cocaine-using mothers with children in foster care were excluded from this sample, and thus, this does not represent the continuation of cocaine use in the postnatal period in the sample as a whole. These data are also based on self-report alone.
Table 2. Maternal and Infant Characteristics by Group Status.
| Variable | Noncocaine-using | Cocaine-using | Effect size | ||
|---|---|---|---|---|---|
| M | SD | M | SD | ||
| Maternal characteristics | |||||
| Age (years) | 29.30 | 5.18 | 31.66 | 5.59 | −.44 |
| Education (years completed) | 12.31 | 1.84 | 11.25 | 1.75 | .59 |
| Socioeconomic status (Hollingshead, 1975) | 1.41 | 0.50 | 1.56 | 0.50 | −.30 |
| Parity | 3.91 | 1.74 | 3.63 | 2.17 | .14 |
| Prenatal care (no. of visits) | 14.99 | 7.66 | 11.64 | 8.16 | .42 |
| Days per week cocaine used | |||||
| Before conception | 0 | 0 | 1.46 | 2.61 | −.77 |
| 1st trimester | 0 | 0 | 1.30 | 2.32 | −.77 |
| 2nd trimester | 0 | 0 | 0.92 | 2.02 | −.63 |
| 3rd trimester | 0 | 0 | 0.60 | 1.32 | −.63 |
| After delivery | 0 | 0 | 0.10 | 0.40 | −.35 |
|
| |||||
| Infant characteristics | |||||
| Gestational age (weeks) | 39.3 | 1.34 | 38.6 | 1.93 | .45 |
| Birth weight (g) | 3,267.51 | 558.60 | 3,078.77 | 546.98 | .34 |
| Head circumference (cm) | 33.30 | 1.46 | 33.62 | 2.37 | −.16 |
| Birth length (cm) | 48.67 | 3.29 | 48.93 | 3.22 | .08 |
| Infant gender (% male) | 45 | 46 | |||
| Obstetrical complications | 92.41 | 13.18 | 85.11 | 14.52 | .53 |
|
| |||||
| Race/ethnicity | |||||
| % Black | 66 | 66 | |||
| % White | 21 | 10 | |||
| % other | 13 | 14 | |||
Note. The measure of effect size is Cohen's d. Measures of cocaine use reported in this table are based on self-report alone.
Table 3. Maternal Substance Use, Psychological Functioning, and Maternal Behavior During Feeding.
| Variable | Noncocaine-using | Cocaine-using | Effect sizea | Effect sizeb | ||
|---|---|---|---|---|---|---|
| M | SD | M | SD | |||
| Pregnancy substance use | ||||||
| Number of cigarettes | 454.67 | 966.82 | 1537.25 | 1674.69 | −.78 | |
| Number of standard drinks | 10.46 | 39.58 | 193.69 | 514.00 | −.49 | |
| Number of days binged | 1.05 | 4.02 | 16.64 | 50.64 | −.42 | |
| Number of joints | 92.40 | 384.99 | 45.41 | 137.27 | .17 | |
| Postnatal substance use | ||||||
| Number of cigarettes | 58.24 | 110.80 | 185.25 | 247.86 | −.65 | |
| Number of standard drinks | 1.84 | 6.00 | 9.04 | 37.65 | −.26 | |
| Number of days binged | 0.06 | 0.31 | 0.28 | 1.43 | −.21 | |
| Number of joints | 4.69 | 21.05 | 5.95 | 24.29 | −.06 | |
| Psychological functioning | ||||||
| Depression/anxiety | 0.37 | 0.39 | 0.53 | 0.64 | −.45 | −.34 |
| Anger/hostility | 14.18 | 3.66 | 15.94 | 4.80 | −.41 | −.31 |
| Maternal behavior | ||||||
| Maternal insensitivity | −0.21 | 0.51 | 0.14 | 1.23 | −.37 | −.29 |
| Maternal warmth | −0.08 | 0.99 | 0.06 | 1.01 | −.14 | −.07 |
| Maternal history of abuse | 9.05 | 4.90 | 11.42 | 6.16 | −.42 | −.27 |
Note. The measure of effect size is Cohen's d. Measures of prenatal and postnatal substance use reported in this table are based on self-report alone.
Effect size not controlling for maternal age and education.
Effect size after controlling for maternal age and education.
Results of MANOVAs with prenatal and postnatal use of other substances as the dependent variables also yielded a significant effect of group status, F(8, 120) = 4.35, p < .001. Univariate analyses were conducted with prenatal use of alcohol, cigarettes, and marijuana, in that order, followed by postnatal substance use, with alpha set at p < .007 (Bonferroni-type correction). Results indicated that women who used cocaine during pregnancy also smoked more cigarettes and consumed more alcoholic drinks during pregnancy (see Table 3).
Group Differences in Maternal Psychopathology, Child Abuse History, and Maternal Behavior
MANOVAs were conducted with maternal depression/anxiety, anger/hostility, and child abuse history as the dependent variables. Results indicated a significant multivariate effect of group status, F(3, 126) = 3.21, p < .05. Univariate analyses with alpha set at .015 indicated that cocaine-using mothers had higher levels of depression/anxiety compared with those in the comparison group and were more likely to report a history of child abuse (see Table 3). Next, MANOVAs with maternal behavior during feeding interaction as the dependent variables did not yield any significant group differences at the multivariate level, although mothers in the cocaine group tended to be more insensitive at the univariate level, with alpha set at .025 (see Table 3). When these analyses were repeated with maternal age and education as covariates, no significant multivariate effects were obtained and there were no significant effects at the univariate level. The effect sizes (Cohen's d) with these two variables as covariates are reported in Table 3.
Associations Among Study Variables
Associations among the variables in the conceptual model are presented in Table 4. We used the continuous measure of cocaine use (maternal self-reports) in these analyses as an indicator of the association between amount of use and other predictors and outcome variables. We chose to use binge drinking as an indicator of risky drinking during pregnancy. Several demographic variables were considered for inclusion in this model. These were maternal age, education, parity, and marital status. Only maternal education was associated with maternal insensitivity and with maternal substance use at p < .10. Mothers with lower years of education had higher substance use during pregnancy and the postnatal period, had higher depression and anger, and were more insensitive during play interactions. Thus, model testing was conducted with and without maternal education in the model. Among infant growth variables (birth weight, birth length, head circumference), only birth weight was associated with maternal insensitivity. Higher number of days of cocaine use, total number of cigarettes during pregnancy, and number of days of binge drinking during pregnancy were associated with higher postnatal depression and anger but not with maternal behavior during feeding. Higher number of days of cocaine use after delivery and higher number of days of binge drinking after delivery were both associated with higher maternal insensitivity. Higher number of cigarettes smoked after delivery was associated with higher depression and anger but not with maternal insensitivity during feeding.
Table 4. Correlations Among Study Variables.
| Variable | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. Maternal education | — | ||||||||||||||
| 2. Birth weight | .03 | — | |||||||||||||
| 3. No. days used cocaine—pregnancy | −.20* | −.16† | — | ||||||||||||
| 4. No. cigarettes—pregnancy | −.32 | −.28** | .47** | — | |||||||||||
| 5. No. days binged—pregnancy | −.26** | −.03 | .75** | .40** | — | ||||||||||
| 6. No. joints—pregnancy | −.19* | .01 | −.03 | −.02 | −.02 | — | |||||||||
| 7. No. days used cocaine—postnatal | −.18* | −.14 | .18* | .03 | .18* | .00 | — | ||||||||
| 8. No. cigarettes—postnatal | −.23** | −.24** | .17* | .60** | .10 | −.02 | .05 | — | |||||||
| 9. No. days binged—postnatal | −.12 | −.06 | .41** | .05 | .19* | −.03 | .85** | .12 | — | ||||||
| 10. No. joints used—postnatal | −.15† | −.04 | −.08 | −.05 | −.06 | .35** | −.02 | .00 | .00 | — | |||||
| 11. Maternal depression/anxiety | −.37** | .04 | .25** | .32** | .32** | .10 | −.03 | .33** | .05 | .14 | — | ||||
| 12. Maternal anger/hostility | −.32** | −.07 | .32** | .33** | .28** | .14 | .10 | .20* | .10 | .08 | .53** | — | |||
| 13. Maternal insensitivity | −.19* | .15† | .00 | .07 | .02 | −.04 | .21* | −.01 | .19* | .02 | .15 | −.12 | — | ||
| 14. Maternal warmth | .15 | .05 | .06 | .04 | .04 | −.09 | −.06 | .02 | .07 | −.12 | −.02 | .07 | .03 | — | |
| 15. Maternal history of abuse | −.25** | −.10 | .09 | .19* | .03 | .08 | .07 | .25** | .06 | −.04 | .40** | .36** | .09* | −.01 | — |
Note. Children in foster care were not included in these analyses.
p < .10.
p < .05.
p < .01.
Model Testing
The next step was to test the conceptual model depicted in Figure 1 predicting maternal insensitivity using path analysis. One primary purpose for this model testing was to examine whether the group differences and associations between maternal cocaine use and maternal behavior were due to cocaine per se but could be better explained by the context of maternal cocaine use such as other substance use during pregnancy and after delivery, maternal psychopathology, and higher infant risk. To maximize subject-to-parameter ratio and for parsimony, we dropped marijuana use from the model because it was not related to cocaine use or to maternal insensitivity. A baseline model was tested that included paths from maternal childhood history of abuse to maternal depression/anxiety, maternal anger/hostility, pregnancy, and postnatal substance use variables, as well as a direct path to maternal insensitivity. This baseline model also included paths from maternal psychopathology variables to maternal insensitivity, paths from pregnancy and postnatal substance use to maternal insensitivity, and paths from pregnancy substance use to birth weight and from birth weight to maternal insensitivity (as depicted in Figure 1). This model also included covariances among predictors (pregnancy and postnatal substance use and maternal psychopathology).1 After this model was estimated, the nonsignificant paths were trimmed, and the final model is depicted in Figure 2. This final model fit the data reasonably well: χ2(36, N = 130) = 58.35, p = .01, comparative fit index = .94, root-mean-square error of approximation = .069 (90% confidence limits = .03, .10). As indicated in Figure 2, mothers who reported higher child abuse histories were more depressed or anxious, more angry/hostile, and more likely to smoke during pregnancy and the postnatal period. When examined in the context of other substance use, maternal psychopathology, and infant birth weight, only maternal cocaine use in the postnatal period was a significant and unique predictor of maternal insensitivity during feeding. Maternal depression/anxiety was marginally associated with maternal insensitivity. Finally, although all of the pregnancy substance use variables were associated with lower infant birth weight, infant birth weight did not predict higher maternal insensitivity.
Figure 2.
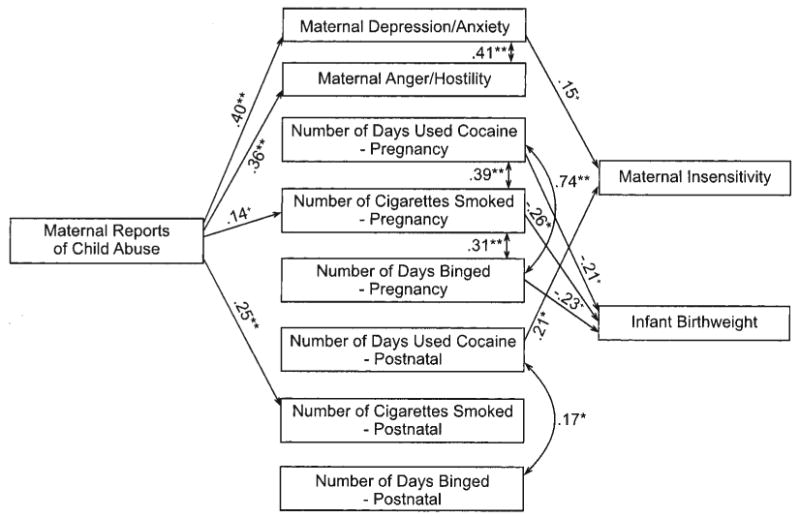
Final model. Not depicted in this model but included in model estimation were associations between the residuals of maternal depression/anxiety and pregnancy binge drinking (β = .16) and number of cigarettes smoked postnatally (β = .16). This model also included covariances between residuals of prenatal and postnatal substance use variables. These were not depicted in the figure because they were not significant in the context of model testing. +p < .10. *p < .05. **p < .01.
Discussion
Results from this study indicate that among substance abuse variables, only postnatal cocaine use was a unique predictor of maternal insensitivity when maternal behavior was examined in the context of polydrug exposure, maternal psychopathology, and higher infant risk. Although the analyses of group differences indicated that, as hypothesized, cocaine-using mothers displayed higher insensitivity during feeding interactions with their infants, this group difference was no longer significant after controlling for maternal education and age, and in model testing, it was not due to prenatal cocaine use per se but continued cocaine use after delivery. These results lend further support to a growing number of recent studies indicating the importance of examining continued postnatal substance abuse on maternal behavior during mother–infant interactions. For instance, Blackwell, Kirkhart, Schmitt, and Kaiser (1998) reported that the group of mothers who continued drug use in the postnatal period displayed a poorer quality of mother–infant interactions during feeding compared with those who abstained from drugs in the postnatal period at 6 months of infant age. Similarly, observation of maternal competence during mother–child play interactions at 18 months of child age was predicted only by ongoing maternal cocaine or opiate use and maternal rigidity in an intervention study (Schuler et al., 2002). Finally, Johnson et al. (2002) reported that the quality of mother–child interactions at child age of 3 years was most negative in the cocaine-using group if mothers continued to report cocaine use at 3 years of child age.
It is important to note that maternal cocaine use, cigarette smoking, and binge drinking during pregnancy were also associated with higher depression/anxiety. However, in the context of model testing when controlling for use of other substances, only binge drinking during pregnancy was uniquely associated with higher depression/anxiety in the postnatal period. A number of different studies have noted the co-occurrence of depression and heavy drinking among pregnant and postpartum women. For instance, Zuckerman, Amaro, Bauchner, and Cabral (1989) noted that alcohol consumption during pregnancy was significantly associated with depression. Others have noted that women who experience comorbid depression and alcohol problems during pregnancy are at greater risk for both depression and alcohol problems in the first postpartum year (Homish, Cornelius, Richardson, & Day, 2004). We did not measure depression during pregnancy in this study. Thus, it is unclear whether the association between pregnancy binge drinking and postnatal depression is due to stability of depression from pregnancy to the postnatal period. Among postnatal substances, maternal cigarette smoking after delivery was uniquely associated with maternal depression/anxiety. This is also similar to previous studies indicating a consistent association between maternal cigarette smoking and higher maternal depression and anxiety (D. W. Black, Zimmerman, & Coryell, 1999). Taken together, the results highlight the co-occurring nature of maternal substance use and maternal depression and anxiety during pregnancy and the postpartum period.
Mothers who had higher depression/anxiety also tended to be more insensitive during feeding interactions with their infants. Thus, results indicate that in addition to postnatal cocaine use, another pathway to greater maternal insensitivity may be via maternal depression and anxiety. This finding lends further support to a relatively large literature emphasizing the influence of maternal depression on maternal behavior during mother–infant interactions (Cohn & Campbell, 1992). These results also emphasize the importance of considering other pathways of influence or associations with maternal behavior among substance abusing mothers. In particular, the results indicate that maternal depression and anxiety may play an important role in predicting maternal behavior apart from continued postnatal substance abuse.
Although there were no direct associations between maternal history of child abuse and maternal behavior, history of child abuse was associated with higher maternal psychopathology. Mothers with a childhood history of abuse reported higher depression/anxiety as well as higher anger/hostility. These results are similar to those reported in the psychiatric literature indicating a strong association between childhood history of abuse and adult depression among both men and women (Harkness & Wildes, 2002) and higher rates of anger/hostility among adults with childhood trauma (Hoglund & Nicholas, 1995). Although we hypothesized a direct association between childhood history of abuse and current maternal behavior, the association was indirect, via current maternal depression. Contrary to expectations, mothers with a history of abuse were not more likely to engage in illicit drug use during pregnancy, but they did report higher cigarette smoking both during and after pregnancy. Given the high rates of cigarette smoking during the prenatal and perinatal periods and the serious health consequences of cigarette smoking for both the mother and the infant, future studies might well examine the potential etiological role of childhood trauma in the continuation of cigarette smoking through pregnancy and postpartum periods.
Contrary to expectations, there were no group differences in maternal warmth or associations between maternal psychological functioning and maternal warmth. It is possible that this was due to the context in which maternal warmth was measured in this study, that of feeding. Although there was some variability in the measure of maternal warmth, variability on the measure of maternal insensitivity was much larger. Finally, contrary to the conceptual model, there was no unique association between infant birth weight and maternal behavior, although at the level of correlations, lower infant birth weight had a marginal association with higher maternal insensitivity. This indicates that once other maternal risk factors are considered, lower infant birth weight may not account for unique variance in maternal behavior. Previous studies reporting associations between infant birth weight and maternal behavior in prenatally exposed samples did not include other maternal risk factors in the same model (e.g., Singer et al., 2001). Future studies are needed to examine this issue more closely.
Taken together, the results add to a growing body of literature highlighting the importance of examining the postnatal caregiving environment as a predictor of child outcomes among children exposed to multiple substances, including cocaine, in the prenatal period. Indeed, a number of studies have noted that the caregiving environment of children exposed to cocaine and other substances in the prenatal period is often unstable and negative (Alessandri, Bendersky, & Lewis, 1998; Brown, Bakeman, Coles, Platzman, & Lynch, 2004; Singer et al., 2002). This is especially important given recent reports that cocaine-exposed children in nonmaternal, nonkin care in the first 2 years of life have more positive outcomes at 2 years of age compared with those raised by their biological parent (Brown et al., 2004). The current results indicate that two important aspects of this caregiving environment that may affect parenting directly are current maternal psychological functioning and postnatal substance use. Thus, it is possible that children raised in nonmaternal and nonkin care have more positive outcomes because they do not experience continued maternal substance use that affects maternal behavior concurrently. Future studies with a larger number of infants in nonmaternal care need to examine this issue more closely.
Little information is currently available regarding the degree of stability of prenatal and postnatal cocaine use or the pattern of cocaine use during pregnancy. In longitudinal cohorts, biomarkers such as urine and meconium are often used. Mothers or infants who test positive on the basis of such biomarkers at delivery represent recent or persistent users of cocaine, those who continued use throughout pregnancy. Those who do not test positive at delivery may represent light users or those with a declining pattern of cocaine use. Because of the complications and limitations of different methods of ascertainment, there are few empirical data to indicate the pattern of use and the association between prenatal and postnatal use. Ascertainment of postnatal use is often complicated by changes in caregivers in the postnatal period and the assessment of the primary caregiver, who may or may not be the biological mother. Current results suggested that when cocaine use is measured by maternal self-report, there is a linear decrease in the pattern of use from the month before conception to a month after delivery. However, these results need to be viewed with caution because they are retrospective and based on self-report alone. They also do not include children who were placed in nonmaternal care after delivery.
There are a number of limitations of this study. First, the accurate assessment of substance use both prenatally and postnatally is difficult. Pregnant and postpartum women are often hesitant to divulge substance use information, particularly illicit substances such as cocaine. The use of multiple methods such as self-report, hair, and urine during the prenatal period partially mitigated this limitation, although it is worth noting that the analyses using continuous measures of substance abuse were based on self-reported use alone. A second limitation is that the measure of history of child abuse was based on retrospective reports, with obvious biases that need to be considered in interpretation of the data. Finally, the measures of maternal behavior were limited to a single context, that of feeding, and the session was limited to a brief, 10-min period. We chose to focus on feeding interactions because they provide a naturalistic context in which a majority of mother–infant interactions in the first month of life occur. However, future studies that measure maternal behavior in multiple contexts will be better able to address the issue of generalizability of results to contexts other than feeding. Finally, we did not measure maternal lack of empathy or remorse in this study. It is possible that the addition of such variables would be related to both substance use and the quality of maternal behavior.
Despite these limitations, the findings are important because they lend further support to the importance of the postnatal caregiving environment in predicting maternal behavior and potential child outcomes. The results highlight the role of continued maternal substance use in the postpartum period and the importance of postnatal maternal depression/anxiety in predicting maternal behavior.
Acknowledgments
This study was made possible by Grant 1R01DA013190-01A2 from the National Institute on Drug Abuse. We thank the parents and infants who participated in this study and the research staff who were responsible for conducting numerous assessments with these families. Special thanks go to Claire Coles for consultation with regard to the conceptual model and for collaboration on this study, to Amol Lele and Luther Robinson for collaboration on data collection at Women's and Children's Hospital of Buffalo, and to Michael Ray for his collaboration on data collection at Sisters of Charity Hospital of Buffalo.
Footnotes
Given the associations between maternal education and substance use as well as maternal behavior, the model was estimated with maternal education as a covariate. The inclusion of maternal education in the model did not change the parameter estimates, and the parameter estimate reflecting the unique association between maternal education and maternal insensitivity was nonsignificant. Thus, maternal education was not included in the final model.
Contributor Information
Rina D. Eiden, University at Buffalo, State University of New York
Arianne Stevens, University at Buffalo, State University of New York.
Pamela Schuetze, Buffalo State College, State University of New York, and University at Buffalo, State University of New York.
Laura E. Dombkowski, Buffalo State College, State University of New York
References
- Alessandri SM, Bendersky M, Lewis M. Cognitive functioning in 8- to 18-month-old drug-exposed infants. Developmental Psychology. 1998;34:565–573. doi: 10.1037//0012-1649.34.3.565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amaro H, Zuckerman B, Cabral H. Drug use among adolescent mothers: Profile of risk. Pediatrics. 1989;84:144–151. [PubMed] [Google Scholar]
- Bauman PS, Dougherty FE. Drug-addicted mothers' parenting and their children's development. International Journal of the Addictions. 1983;18:291–302. doi: 10.3109/10826088309039348. [DOI] [PubMed] [Google Scholar]
- Baumgartner WA, Hill VA, Blahd WH. Hair analysis for drugs of abuse. Journal of Forensic Science. 1989;34:1433–1453. [Google Scholar]
- Belsky J. The determinants of parenting: A process model. Child Development. 1984;55:83–96. doi: 10.1111/j.1467-8624.1984.tb00275.x. [DOI] [PubMed] [Google Scholar]
- Black DW, Zimmerman M, Coryell W. Cigarette smoking and psychiatric disorder in a community sample. Annals of Clinical Psychiatry. 1999;11:129–136. doi: 10.1023/a:1022355826450. [DOI] [PubMed] [Google Scholar]
- Black M, Schuler M, Nair P. Prenatal drug exposure: Neurodevelopmental outcome and parenting environment. Journal of Pediatric Psychology. 1993;18:605–620. doi: 10.1093/jpepsy/18.5.605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blackwell P, Kirkhart K, Schmitt D, Kaiser M. Cocaine/polydrug affected dyads: Implications for infant cognitive development and mother–infant interaction during the first six postnatal months. Journal of Applied Developmental Psychology. 1998;19:235–248. [Google Scholar]
- Brandon TH, Copeland AL, Saper ZL. Programmed therapeutic messages as a smoking treatment adjunct: Reducing the impact of negative affect. Health Psychology. 1995;14:41–47. doi: 10.1037//0278-6133.14.1.41. [DOI] [PubMed] [Google Scholar]
- Brown JV, Bakeman R, Coles CD, Platzman KA, Lynch ME. Prenatal cocaine exposure: A comparison of 2-year-old children in parental and nonparental care. Child Development. 2004;75:1282–1295. doi: 10.1111/j.1467-8624.2004.00739.x. [DOI] [PubMed] [Google Scholar]
- Brown JV, Bakeman R, Coles CD, Sexson WR, Demi AS. Maternal drug use during pregnancy: Are preterm and full-term infants affected differently? Developmental Psychology. 1998;34:540–554. doi: 10.1037//0012-1649.34.3.540. [DOI] [PubMed] [Google Scholar]
- Bulik CM, Prescott CA, Kendler KS. Features of childhood sexual abuse and the development of psychiatric and substance use disorders. British Journal of Psychiatry. 2001;179:444–449. doi: 10.1192/bjp.179.5.444. [DOI] [PubMed] [Google Scholar]
- Burns K, Chethik L, Burns W, Clark R. Dyadic disturbances on cocaine-abusing mothers and their infants. Journal of Clinical Psychology. 1991;47:316–319. doi: 10.1002/1097-4679(199103)47:2<316::aid-jclp2270470220>3.0.co;2-1. [DOI] [PubMed] [Google Scholar]
- Burns K, Chethik L, Burns W, Clark R. The early relationship of drug abusing mothers and their infants: An assessment at eight to twelve months of age. Journal of Clinical Psychology. 1997;53:279–287. doi: 10.1002/(sici)1097-4679(199704)53:3<279::aid-jclp11>3.0.co;2-f. [DOI] [PubMed] [Google Scholar]
- Buss AH, Perry M. The Aggression Questionnaire. Journal of Personality and Social Psychology. 1992;63:452–459. doi: 10.1037//0022-3514.63.3.452. [DOI] [PubMed] [Google Scholar]
- Chatoor I. Mother–Infant/Toddler Feeding Scale. 1986. Unpublished measure. [Google Scholar]
- Chatoor I, Egan J. Diagnosis and treatment of feeding disorders in infants and young children. Abstract of paper presented at the 33rd Annual Meeting of the American Academy of Child and Adolescent Psychiatry; Los Angeles. 1986. Sep, [Google Scholar]
- Chatoor I, Schaefer S, Dickson I, Egan I. Non-organic failure to thrive: A developmental perspective. Pediatric Annals. 1984;13:829–835. [PubMed] [Google Scholar]
- Cohn JF, Campbell SB. Influence of maternal depression on infant affect regulation. In: Cicchetti D, Toth S, editors. Developmental perspectives on depression. 4th. Rochester, NY: University of Rochester Press; 1992. pp. 103–130. [Google Scholar]
- Derogatis LR. The Brief Symptom Inventory (BSI) administration, scoring, and procedures manual. Minneapolis, MN: National Computer Systems; 1993. [Google Scholar]
- Eiden RD. Maternal substance use and mother–infant feeding interactions. Infant Mental Health Journal. 2001;22:497–511. [Google Scholar]
- Eiden RD, Chavez F, Leonard KE. Parent–infant interactions in alcoholic and control families. Development and Psychopathology. 1999;11:745–762. doi: 10.1017/s0954579499002308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Espinosa M, Beckwith L, Howard J, Tyler R, Swanson K. Maternal psychopathology and attachment in toddlers of heavy cocaine-using mothers. Infant Mental Health Journal. 2001;23:316–333. [Google Scholar]
- Fink LA, Bernstein D, Handelsman L, Foote J, Lovejoy M. Initial reliability and validity of the Childhood Trauma Interview: A new multidimensional measure of childhood interpersonal trauma. American Journal of Psychiatry. 1995;152:1329–1335. doi: 10.1176/ajp.152.9.1329. [DOI] [PubMed] [Google Scholar]
- Gottwald SR, Thurman SK. The effects of prenatal cocaine exposure on mother infant interaction and infant arousal in the newborn period. Topics in Early Childhood Special Education. 1994;14:217–231. [Google Scholar]
- Handler A, Kistin N, Davis F, Ferre C. Cocaine use during pregnancy: Perinatal outcomes. American Journal of Epidemiology. 1991;133:818–825. doi: 10.1093/oxfordjournals.aje.a115961. [DOI] [PubMed] [Google Scholar]
- Hans SL, Bernstein VJ, Henson LG. The role of psychopathology in the parenting of drug-dependent women. Development and Psychopathology. 1999;11:957–977. doi: 10.1017/s0954579499002400. [DOI] [PubMed] [Google Scholar]
- Harkness KL, Wildes JE. Childhood adversity and anxiety versus dysthymia co-morbidity in major depression. Psychological Medicine. 2002;32:1239–1249. doi: 10.1017/s0033291702006177. [DOI] [PubMed] [Google Scholar]
- Hawley TL, Halle TG, Drasin RE, Thomas NG. Children of addicted mothers: Effects of the ‘crack epidemic’ in the caregiving environment and the development of preschoolers. American Journal of Orthopsychiatry. 1995;65:364–379. doi: 10.1037/h0079693. [DOI] [PubMed] [Google Scholar]
- Hoglund CL, Nicholas KB. Shame, guilt, and anger in college students exposed to abusive family environments. Journal of Family Violence. 1995;10:141–157. [Google Scholar]
- Hollingshead AB. Four-factor index of social status. Yale University, Department of Sociology; New Haven, CT: 1975. Unpublished manuscript. [Google Scholar]
- Homish GG, Cornelius JR, Richardson GA, Day NL. Antenatal risk factors associated with postpartum comorbid alcohol use and depressive symptomatology. Alcoholism: Clinical and Experimental Research. 2004;28:1242–1248. doi: 10.1097/01.alc.0000134217.43967.97. [DOI] [PubMed] [Google Scholar]
- Institute of Medicine. Pathways of addiction: Opportunities in drug abuse research. Washington, DC: National Academies Press; 1996. [PubMed] [Google Scholar]
- Jameson PB, Gelfand D, Kulcsar E, Teti DM. Mother–toddler interaction patterns associated with maternal depression. Developmental Psychopathology. 1997;9:557–590. [PubMed] [Google Scholar]
- Johnson AL, Morrow C, Accornero VH, Xue L, Anthony JC, Bandstra ES. Maternal cocaine use: Estimated effects on mother–child play interactions in the preschool period. Journal of Developmental and Behavioral Pediatrics. 2002;23:191–202. doi: 10.1097/00004703-200208000-00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson HL, Rosen TS. Mother–infant interaction in a multirisk population. American Journal of Orthopsychiatry. 1990;60:281–288. doi: 10.1037/h0079181. [DOI] [PubMed] [Google Scholar]
- LaGasse LL, Messinger D, Lester BM, Seifer R, Tronick EZ, Bauer CR, et al. Prenatal drug exposure and maternal and infant feeding behaviour. Archives of Disease in Childhood: Fetal and Neonatal Edition. 2003;88:F391–F399. doi: 10.1136/fn.88.5.F391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Littman A, Parmelee B. Medical correlation of infant development. Pediatrics. 1978;61:474. doi: 10.1542/peds.61.3.470. [DOI] [PubMed] [Google Scholar]
- Magura S, Freeman RC, Siddiqi Q, Lipton DS. The validity of hair analysis for detecting cocaine and heroin use among addicts. International Journal of the Addictions. 1992;27:51–69. doi: 10.3109/10826089109063462. [DOI] [PubMed] [Google Scholar]
- Molitor A, Mayes LC, Ward A. Emotion regulation behavior during a separation procedure in 18-month-old children of mothers using cocaine and other drugs. Development and Psychopathology. 2003;15:39–54. doi: 10.1017/s0954579403000038. [DOI] [PubMed] [Google Scholar]
- Neuspiel DR, Hamel SC, Hochberg E, Greene J, Campbell J. Maternal cocaine use and infant behavior. Neurotoxicology and Teratology. 1991;13:229–233. doi: 10.1016/0892-0362(91)90015-o. [DOI] [PubMed] [Google Scholar]
- O'Connor MJ, Sigman M, Kasari C. Interactional model for the association among maternal alcohol use, mother–infant interaction, and infant cognitive development. Infant Behavior and Development. 1993;16:177–192. [Google Scholar]
- Oro AS, Dixon SD. Perinatal cocaine and methamphetamine exposure: Maternal and neonatal correlates. Journal of Pediatrics. 1987;111:571–578. doi: 10.1016/s0022-3476(87)80125-7. [DOI] [PubMed] [Google Scholar]
- Schuck AM, Widom CS. Childhood victimization and alcohol symptoms in females: Causal inferences and hypothesized mediators. Child Abuse and Neglect. 2001;25:1069–1092. doi: 10.1016/s0145-2134(01)00257-5. [DOI] [PubMed] [Google Scholar]
- Schuler ME, Nair P, Black MM. Ongoing maternal drug use, parenting attitudes, and a home intervention: Effects on mother–child interaction at 18 months. Journal of Developmental and Behavioral Pediatrics. 2002;23:87–94. doi: 10.1097/00004703-200204000-00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singer L, Arendt R, Minnes S, Farkas K, Yamashita T, Kliegman R. Increased psychological distress in post-partum, cocaineusing mothers. Journal of Substance Abuse. 1995;7:165–174. doi: 10.1016/0899-3289(95)90002-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singer L, Hawkins S, Huang J, Davillier M, Baley J. Developmental outcomes and environmental correlates of very low birthweight, cocaine-exposed infants. Early Human Development. 2001;2267:1–13. doi: 10.1016/s0378-3782(01)00182-7. [DOI] [PubMed] [Google Scholar]
- Singer L, Salvator A, Arendt R, Minnes S, Farkas K, Kliegman R. Effects of cocaine/polydrug exposure and maternal psychological distress on infant birth outcomes. Neurotoxicology and Teratology. 2002;24:127–135. doi: 10.1016/s0892-0362(01)00208-2. [DOI] [PubMed] [Google Scholar]
- Snyder JJ, Huntley D. Troubled families and troubled youth: The development of antisocial behavior and depression in children. In: Leone PE, editor. Understanding troubled and troubling youths. Newbury Park, CA: Sage; 1990. pp. 194–225. [Google Scholar]
- Sobell MB, Sobell LC, Klajner F, Pavan D, Basian E. The reliability of a timeline method for assessing normal drinker college students' recent drinking history. Addictive Behaviors. 1986;11:149–162. doi: 10.1016/0306-4603(86)90040-7. [DOI] [PubMed] [Google Scholar]
- Zuckerman B, Amaro H, Bauchner H, Cabral H. Depressive symptoms during pregnancy: Relationship to poor health behaviors. American Journal of Obstetrics and Gynecology. 1989;160:1107–1111. doi: 10.1016/0002-9378(89)90170-1. [DOI] [PubMed] [Google Scholar]


