Abstract
We present a 32-year-old, extremely obese, pregnant woman who developed severe hypotension and water intoxication after an accidental injection of large bolus of oxytocin during cesarean section under general anesthesia. The patient was initially thought to have an amniotic fluid embolism because of the abrupt hemodynamic changes developed immediately after fetal delivery and lack of recognition of medication error. It is highly recommended that careful attention should be paid not only to the possibility of hemodynamic deterioration and water intoxication if oxytocin is given rapidly in excessive doses, but to the confirmation of the proper use of the drug before it is injected.
Keywords: Oxytocin, Water intoxication
Oxytocin is best known for its contractile activity on uterine smooth muscle, and thus is still widely used to reduce uterine bleeding after delivery of the fetus. However, oxytocin when given in rapid bolus produces marked but short-lived hypotension and tachycardia [1-3]. Sometimes, this abrupt and severe hemodynamic depression may need to be distinguished from placental abruption, acute myocardial infarction, or a pulmonary embolism in patients undergoing delivery. Therefore, it is recommended to administer a dose of 5 international units of oxytocin by slow intravenous injection rather than a rapid bolus injection [1]. In addition, oxytocin may cause water intoxication via an anti-diuretic hormone-like activity when administered in excessive dose with electrolyte-free solution [4]. We report a case of 32-year-old, extremely obese, pregnant woman in which an inadvertent rapid injection of large bolus of oxytocin led to cardiovascular collapse initially and water intoxication later during the Cesarean delivery. In our case, early catastrophic event was regarded as an amniotic fluid embolism (AFE) because of the characteristic signs, including abrupt hemodynamic changes and respiratory distress [5].
Case Report
A 32-year-old multipara at 40 weeks' gestation was scheduled for elective cesarean section due to cephalopelvic disproportion under general anesthesia. She weighed 140 kg and was 161 cm in height, and thus the body mass index (BMI) was 54.0 kg/m2. Her medical history and laboratory data were unremarkable. On arrival in the operating room, her blood pressure and pulse rate were 152/98 mmHg and 87 beats/min, respectively and percutaneous oxygen saturation was 94% on room air. Anesthesia was induced with intravenous thiopental sodium 500 mg, succinylcholine 100 mg, and vecuronium 8 mg, and maintained with 1.2 vol% sevoflurane (inspired) until the fetal delivery and 1-3 vol% sevoflurane (inspired) thereafter in 50% nitrous oxide after tracheal intubation. The patient was mechanically ventilated at a tidal volume of 8 ml/kg, and the rate was adjusted to maintain the end-tidal carbon dioxide tension around 35 mmHg and oxygen saturation rose to 98%. The cesarean section was performed through a low midline incision and a 3.12 kg male was delivered 15 min after anesthetic induction. The Apgar scores were 8 at 1 min, and 9 at 5 min. Immediately after placental delivery, a bolus dose of 5 units oxytocin, carbetocin 100 µg, midazolam 4 mg followed by a continuous infusion of 25 units of oxytocin (Yuhan corp., Seoul, Korea) mixed in 1 L of Hartmann's solution was ordered to be given. Within a few min after their injections, her heart rate increased above 130 beats/min and arterial blood pressure decreased to 68/34 mmHg. Intravenous ephedrine 10 mg was given immediately followed by continuous infusion of dopamine 8-10 µg/kg/min with rapid administration of Hartman's and colloid solution (Voluven, Fresenius Kabi, Germanry). Thereafter systolic blood pressure increased up to 90 mmHg; however, her oxygen saturation gradually decreased to 82%, while the end-tidal carbon dioxide tension remained unchanged. We promptly discontinued the nitrous oxide, increased the FiO2 to 1.0, and changed her position from the Trendelenburg to the supine. The trachea was cleared several times because the breathing sounds were not clear with wheezing. The patient was given dexamethasone 10 mg and aminophylline 150 mg intravenously followed by a continuous infusion of aminophylline 350 mg mixed in 500 ml of 5% dextrose water. Her oxygen saturation increased to 89% at the time when the surgical procedure was completed.
We initially regarded the deterioration of hemodynamics as an amniotic fluid embolism (AFE), but could not confirm the diagnosis because the signs of a coagulopathy, including hemorrhage, disseminated intravascular coagulation, or inadequate uterine contractions, were lacking. It was also difficult to rule out an AFE because we could not obtain any blood sample for laboratory testing of AFE until the end of the surgery. All attempts to access the peripheral and central veins or even arteries were failed because of the patient's short, fatty neck and the peripheral vessels were not visible due to morbid obesity.
It was then revealed that 25 units of oxytocin were inadvertently administered as a single bolus simultaneously with 5 units of oxytocin and carbetocin 100 µg instead of mixed with Hartman's solution. Until that time, 1.0 L of Hartman's solution and 500 ml of colloid solution had already been infused. To prevent the development of water intoxication due to oxytocin, Hartman's solution was exchanged with 0.9% normal saline and lasix 20 mg and 25% mannitol 12.5 g were intravenously administered. Upon completion of surgery, a central venous catheter was inserted through the right femoral vein to withdraw blood for measurements of plasma electrolytes and coagulation profiles and to administer drugs and fluids. The plasma Na+ and K+ concentrations measured just before the end of anesthesia were 118 and 3.9 mmol/L, respectively, and thus lasix 20 mg was additionally administered intravenously in an attempt to treat the water intoxication. There was no evidence of coagulopathy with the normal coagulation profiles, including the activated prothrombin time (20.9 sec), fibrinogen (324.1 mg/dl), platelet count (329 × 109/L), and fibrinogen degradation products (FDP; 10.5 µg/ml). The estimated blood loss, amount of fluid infused, and volume of urine during the operation were 1,000 ml, 1,800 ml, and 150 ml, respectively.
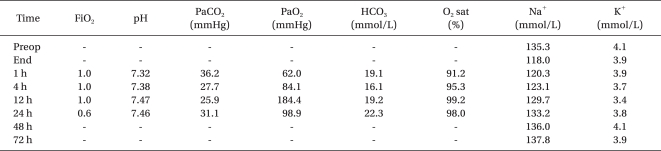
The patient was transferred to the intensive care unit (ICU) while intubated because her respiration was not sufficient to remove the endotracheal tube, despite the reversal of neuromuscular block with pyridostigmine 20 mg. On arrival at the ICU, her blood pressure and heart rate were 83/35 mmHg and 124 beats/min, respectively. Then, the patient received a continuous infusion of norepinephrine at a rate of 0.05-0.2 µg/kg/min in addition to dopamine and thereby the blood pressure increased to 105/52 mmHg. An arterial line was placed into the right dorsalis pedis artery to continuously monitor blood pressure and to collect blood samples. The blood gas analysis was as follows: pH, 7.32; PaCO2, 36.2 mmHg; PaO2, 62.0 mmHg; and O2 saturation, 91.2%. At that time, the pulse oximetry reading was 90% on mechanical ventilation, which was set in the synchronized intermittent mandatory ventilation mode using the following parameters: tidal volume, 500 ml; and respiratory rate, 8 breaths/min with a FiO2 of 1.0 without positive end-expiratory pressure support. Chest x-ray and computed tomography (CT) obtained at the completion of surgery showed no evidence of pulmonary edema or embolism. The infusion of dopamine and norepinephrine was discontinued the night of surgery because the hemodynamics had been stabilized. She regained consciousness 4 hours after cessation of anesthetics, but was still drowsy, agitated, and nauseated despite of the absence of lesions in the brain CT. It was not until the next morning that she was fully awake and well-oriented. The Na+ concentration measured 1 and 4 hours after surgery increased to 120.3 and 123.1 mmol/L, respectively. The last supplemental dose of lasix (10 mg) was given intravenously. The arterial blood gas data were gradually improved over time (Table 1), and thus extubated the day following surgery. The Na+ concentration gradually returned to the normal range 24 hours after the surgery (Table 1). She was transferred to the general ward without any problems after a 3 day ICU stay.
Table 1.
Perioperative Changes of Arterial Blood Gas Data and Electrolytes
Preop: preoperative, End: end of surgery, FiO2: fraction of inspired oxygen, PaCO2: arterial carbon-dioxide pressure, PaO2: arterial oxygen pressure, HCO3: bicarbonate, O2 sat: arterial oxygen saturation.
Discussion
The patient presented herein had severe hypotension and water intoxication caused by an inadvertent large bolus injection of oxytocin instead of a continuous infusion. This mistake was made by an inexperienced assistant nurse in the process of taking order from the anesthesiologist. To avoid iatrogenic medication errors, it is suggested that anesthesiologists supervise their assistants with vigilance and assistants confirm the order before they execute it.
Oxytocin is generally used in accordance with a protocol involving continuous infusion of diluted oxytocin (typically 10 mU/ml) at a rate of 1 mU/min initially, with dose increment of no greater than 1 mU/min every 30 min [6]. However, oxytocin causes cardiovascular effects [2] mainly due to a decrease in systemic vascular resistance, varying from sinus tachycardia, mild hypotension, ST changes to even cardiac arrest in compromised patient [3]. In addition, an antidiuretic hormone-like effect of oxytocin may lead to water intoxication which could be life-threatening. Woodhouse reported that infusion of 240 units of syntocinon mixed in electrolyte free water over 32 hours in a gravida led to comatose state for 24 hours [4]. In our case we experienced abrupt hemodynamic changes after an accidental injection of large bolus of oxytocin. The reduced blood pressure did not recover soon and lasted for about 6 hours, which is not consistent with previous reports [1,2] and made us misdiagnosed the signs as AFE until we were aware of the medication error. Delayed recovery of blood pressure could not thoroughly explained by oxytocin overdose alone because of its short biologic half-life. Thus prolonged hypotension may have been contributed to combinations of oxytocin overdose and other factors such as unrecognized hypovolemia or anaphylactoid reaction to oxytocin, although the latter is less likely to be the cause of hypotension than former because she already had an experience of delivery without any problem, in which oxytocin was used.
A decrease of arterial oxygen saturation was also one of the factors that led us to confuse the signs with AFE. In hindsight the unexplained hypoxemia represented in this case is considered to be attributed to secretion, positional change and mainly the mechanical factors of obesity rather than an AFE because it was relatively well-managed with merely a change in the original position, tracheal suction, and bronchodilators. Obesity impairs respiratory function, mainly expiratory reserve volume and functional residual capacity which further decrease below 50% of the pre-operative values respectively under general anesthesia [7].
Another reason we did not exclude an AFE early during surgery was that we could not sample the blood for laboratory confirmation of a coagulopathy, which is considered to be essential for the clinical criteria of AFE [8], because she was morbidly obese. It would have been helpful for the early decision to rule out AFE if we had secured the intravenous or intra-arterial line before anesthetic induction. Thus, the central venous line in the beginning is recommended to manage a catastrophe if an obese patient is scheduled for surgery.
We could not confirm the diagnosis of AFE even until the revelation because this patient did not show typical clinical manifestations of AFE including coagulopathy, uterine atony, seizures, cardiac arrest and even pulmonary edema except for only sudden changes of hemodynamics and reduced oxygen saturation. In addition, the hemodynamic changes were not intractable and responded relatively well to treatment compared with the typical appearance of AFE. As a whole, it was not considered as AFE because not all of the signs were consistent with an AFE.
Water intoxication, a kind of acute hyponatremia, is characterized by confusion, lethargy, vomiting, convulsion, periods of violent behavior, coma, and death due to cerebral edema. It rarely occurs even when a large bolus of oxytocin is administered, unless a large quantity of electrolyte free water is administered [9]. Nevertheless, in our case water intoxication was developed with the use of isotonic and colloid solutions. It has been reported that the continuous infusion of oxytocin decreases free water clearance regardless of water load [10]. Thus 1 L of 5% dextrose water infused at delivery room before transfer to operating room might have contributed to water intoxication in this case.
The signs and symptoms of water intoxication depend not only on the serum sodium concentration, but on its rate of decrease [11]. A serum sodium concentration of 120 mmol/L may cause mild symptoms, including confusion or restlessness, while a serum sodium concentration below 110 mmol/L is associated with seizures or coma, which are associated with a high mortality rate and thus require immediate treatment [12]. Although, the water intoxication is confirmed by measuring the serum electrolyte concentrations, its occurrence might be neglected if signs and symptoms are mild or patients are under general anesthesia as in our case. Even after the end of anesthesia, the patient showed no abnormalities on a brain CT or neurologic signs, but only mild symptoms, including drowsiness, nausea, and delayed recovery of consciousness, although it has not been proven to be related to water intoxication. It would be very helpful for early detection of water intoxication if the patient had spinal or epidural block.
Treatment of water intoxication includes an immediate discontinuance of the infusion of oxytocin and free water. Electrolyte-free water should be replaced with isotonic saline, while improving excretion of free water with furosemide or mannitol may be helpful. Administration of hypertonic saline depends on the presence of the neurologic symptoms and should be used with great caution since rapid correction might be associated with neurologic sequela, such as central pontine myelinolysis [13]. Thus, it is generally recommended that correction should be kept at a rate < 12 mmol/L per 24 hours and < 18 mmol/L per 48 hours [14]. In our case, there were no suspicious symptoms or signs suggesting neurologic complications, as we corrected the serum sodium concentration as recommended [14].
In summary, we present herewith a morbidly obese patient who developed severe hypotension and water intoxication following an accidental intravenous bolus injection of large amount of oxytocin during cesarean section. An extreme vigilance for confirming drug before its injection is required to avoid unexpected medication errors.
References
- 1.Pinder AJ, Dresner M, Calow C, Shorten GD, O'Riordan J, Johnson R. Haemodynamic changes caused by oxytocin during caesarean section under spinal anaesthesia. Int J Obstet Anesth. 2002;11:156–159. doi: 10.1054/ijoa.2002.0970. [DOI] [PubMed] [Google Scholar]
- 2.Thomas JS, Koh SH, Cooper GM. Haemodynamic effects of oxytocin given as i.v. bolus or infusion on women undergoing Caesarean section. Br J Anaesth. 2007;98:116–119. doi: 10.1093/bja/ael302. [DOI] [PubMed] [Google Scholar]
- 3.Thomas TA, Cooper GM. Maternal deaths from anaesthesia. An extract from Why mothers die 1997-1999, the Confidential enquiries into maternal deaths in the United Kingdom. Br J Anaesth. 2002;89:499–508. [PubMed] [Google Scholar]
- 4.Woodhouse DR. Water intoxication associated with high dose syntocinon infusion. Med J Aust. 1980;1:34. doi: 10.5694/j.1326-5377.1980.tb134581.x. [DOI] [PubMed] [Google Scholar]
- 5.Gist RS, Stafford IP, Leibowitz AB, Beilin Y. Amniotic fluid embolism. Anesth Analg. 2009;108:1599–1602. doi: 10.1213/ane.0b013e31819e43a4. [DOI] [PubMed] [Google Scholar]
- 6.Parker KL, Schimmer BP. Pituitary hormones and their hypothalamic releasing factors. In: Hardman JG, Limbird LE, editors. Goodman & Gillman's the Pharmacological Basis of Therapeutics. 10th ed. USA: McGrow-Hill; 2001. p. 1559. [Google Scholar]
- 7.Roizen MF, Fleisher LA. Anesthetic implications of concurrent diseases. In: Miller RD, editor. Miller's Anesthesia. 6th ed. San Francisco: Elsevier Churchill Livingstone; 2005. p. 1032. [Google Scholar]
- 8.Clark SL, Hankins GD, Dudley DA, Dildy GA, Porter TF. Amniotic fluid embolism: analysis of the national registry. Am J Obstet Gynecol. 1995;172:1158–1167. doi: 10.1016/0002-9378(95)91474-9. [DOI] [PubMed] [Google Scholar]
- 9.Moen V, Irestedt L. Water intoxication following labour and surgery: blaming oxytocin - the easy way out? Acta Anaesthesiol Scand. 2009;53:1226. doi: 10.1111/j.1399-6576.2009.02078.x. [DOI] [PubMed] [Google Scholar]
- 10.Saunders WG, Munsick RA. Antidiuretic potency of oxytocin in women post partum. Am J Obstet Gynecol. 1966;95:5–11. doi: 10.1016/0002-9378(66)90622-3. [DOI] [PubMed] [Google Scholar]
- 11.Prough DS, Wolf SW, Funston JS, Svensen CH. Acid-base, fluid and electrolytes. In: Barash PG, Cullen BF, Stoelting PK, editors. Clinical Anesthesia. 5th ed. Philadelphia: Lippincott-Raven Publishers; 2006. p. 189. [Google Scholar]
- 12.O'hara JF, Jr, Cywinski JB, Monk TG. The renal system and anesthesia for urologic surgery. In: Barash PG, Cullen BF, Stoelting PK, editors. Clinical Anesthesia. 5th ed. Philadelphia: Lippincott-Raven Publishers; 2006. p. 1028. [Google Scholar]
- 13.Laureno R, Karp BI. Myelinolysis after correction of hyponatremia. Ann Intern Med. 1997;126:57–62. doi: 10.7326/0003-4819-126-1-199701010-00008. [DOI] [PubMed] [Google Scholar]
- 14.Sterns RH, Cappuccio JD, Silver SM, Cohen EP. Neurologic sequelae after treatment of severe hyponatremia: a multicenter perspective. J Am Soc Nephrol. 1994;4:1522–1530. doi: 10.1681/ASN.V481522. [DOI] [PubMed] [Google Scholar]