Abstract
Mental health problems disproportionately affect women, particularly during childbearing years. We sought to estimate the prevalence of antepartum mental health problems and determine potential risk factors in a representative USA population. We examined data on 3,051 pregnant women from 11 panels of the 1996–2006 Medical Expenditure Panel Survey. Poor antepartum mental health was defined by self report of mental health conditions or symptoms or a mental health rating of “fair” or “poor.” Multivariate regression analyses modeled the odds of poor antepartum mental health; 7.8% of women reported poor antepartum mental health. A history of mental health problems increased the odds of poor antepartum mental health by a factor of 8.45 (95% CI, 6.01–11.88). Multivariate analyses were stratified by history of mental health problems. Significant factors among both groups included never being married and self-reported fair/poor health status. This study identifies key risk factors associated with antepartum mental health problems in a nationally representative sample of pregnant women. Women with low social support, in poor health, or with a history of poor mental health are at an increased risk of having antepartum mental health problems. Understanding these risk factors is critical to improve the long-term health of women and their children.
Keywords: Pregnancy, Mental health, Prevalence, Population-based, Antepartum mental health
Introduction
Serious mental health problems disproportionately affect women (Kessler 2003) and are particularly prevalent during childbearing years (Kornstein 2001; Weissman and Jensen 2002; Weissman and Olfson 1995). Among mothers, these disorders can have a profound negative impact on the long-term health (Davidson et al. 2000; Eaton 2002; Jonas and Lando 2000), quality of life (Gaynes et al. 2002; Hays et al. 1995; Wells et al. 1992), and well-being of their children (Billings and Moos 1983; Jaenicke et al. 1987; Lee and Gotlib 1989; Schwartz et al. 1990; Weissman et al. 1987; Zuckerman and Beardslee 1987). Moreover, pregnant women with poor mental health may be at increased risk for poor birth outcomes such as having a small for gestational age (Mutale et al. 1991), preterm, or low birth weight baby (Copper et al. 1996).
The frequency of mental health problems among pregnant and non-pregnant women is roughly the same (Kessler et al. 1994; Williams et al. 1995), although capturing the extent of the problem is challenging because experiences of pregnancy often mirror symptoms of depression and anxiety (Kelly et al. 2001; Klein and Essex 1994). Estimates of antepartum mental health problems range from 8.5% to 23%, reflecting substantial heterogeneity in study populations, research questions, and methods (Andersson et al. 2003; Evans et al. 2001; Holcomb et al. 1996; Johanson et al. 2000; Kelly et al. 2001; Vesga-Lopez et al. 2008). The prevalence of and risk factors for poor antepartum mental health have been investigated using a variety of: (1) mental health outcomes, including depression, anxiety, substance use disorders, or a combination thereof; and (2) potential risk factors and confounders including demographic, social, behavioral, or health factors. Furthermore, these studies’ methodologies varied in: (1) the measurement of mental health, utilization of diagnostic interviews, screeners, and/or self-reported symptoms; (2) measurement of risk factors and confounders; (3) the time during pregnancy when data were collected; and (4) the sample selection.
Most of the current literature on the correlates of poor mental health during pregnancy is from clinic-based studies of pregnant women. While these studies may have limited generalizability to the entire population (Bolton et al. 1998), they provide a valuable enumeration of evidence regarding which pregnant women are more likely to experience poor mental health. One of the most salient risk factors for poor antepartum mental health is having a prior history of such problems (Bilszta et al. 2008; Borjesson et al. 2005; Buesching et al. 1986; Marcus et al. 2003; Mora et al. 2009; Lancaster et al. 2010; Rich-Edwards et al. 2006). Women who are younger (Borjesson et al. 2005; Kearns et al. 1997; Paarlberg et al. 1996), African-American (Jesse and Swanson 2007; Orr et al. 2006), in poor physical health (Marcus et al. 2003), unemployed (Bolton et al. 1998; Borjesson et al. 2005; Marcus et al. 2003), or having their first baby (Kearns et al. 1997; Records and Rice 2007) may also be at greater a risk. In addition, women with limited or no social support (Demyttenaere et al. 1995; Jesse and Swanson 2007; Kearns et al. 1997; Lancaster et al. 2010; Lee et al. 2007; Leigh and Milgrom 2008; Rudnicki et al. 2001; Seguin et al. 1995; Westdahl et al. 2007), low socioeconomic status (Bolton et al. 1998; Holzman et al. 2006; Kearns et al. 1997; Marcus et al. 2003), public insurance (Canady et al. 2008; Jesse 2003; Jesse et al. 2005; Kermode et al. 2000; Lancaster et al. 2010), poor coping skills or sense of control (Bernazzani et al. 1997; Da Costa et al. 2000; Demyttenaere et al. 1995; Rudnicki et al. 2001), low self-esteem (Jesse and Swanson 2007; Jesse et al. 2005; Lee et al. 2007; Leigh and Milgrom 2008; Ritter et al. 2000), chronic stress (Jesse and Swanson 2007; Monk et al. 2008; Mora et al. 2009; Paarlberg et al. 1996; Seguin et al. 1995), alcohol or substance abuse problems (Marcus et al. 2003; Mora et al. 2009; Pajulo et al. 2001), or a history of childhood or adult abuse (Benedict et al. 1999; Holzman et al. 2006; Jesse and Swanson 2007; Lancaster et al. 2010; Leigh and Milgrom 2008; Nayak and Al-Yattama 1999) are more susceptible to having antepartum mental health problems. Pregnant women are also at an increased risk if they are unmarried (Bolton et al. 1998; Marcus et al. 2003; Orr et al. 2006; Westdahl et al. 2007), not living with a partner (Canady et al. 2008; Hobfoll et al. 1995; Lancaster et al. 2010; Marcus et al. 2003; Orr et al. 2006; Rich-Edwards et al. 2006; Seguin et al. 1995), or are unhappy in their marriage (Bilszta et al. 2008; Gotlib et al. 1989; Henry et al. 2004; Lee et al. 2007; Records and Rice 2007). Women who report stressful life events (Lancaster et al. 2010; Leigh and Milgrom 2008; Rubertsson et al. 2003; Seguin et al. 1995) or an unplanned or unwanted pregnancy (Lancaster et al. 2010; Lee et al. 2007; Mora et al. 2009; Rich-Edwards et al. 2006) are also at a greater risk according to previous research. A history of obstetrical problems, such as complications, miscarriage, abortion, or stillbirth also has been reported to elevate the risk of antepartum mental health problems (Larsson et al. 2004; Rubertsson et al. 2003). Job-related variables, such as the number of hours worked per week, may be associated with antepartum mental health status (Paarlberg et al. 1996); however, the importance of this variable is not well understood. As noted previously, these studies varied greatly in the specific outcomes, correlates, and confounders that were included in analyses, as well as the measurement and sampling methods that were used.
Importantly, many of the existing studies are based on convenience samples; in fact, we are aware of only one study on the prevalence and risk factors of antepartum psychological problems in a sample representative of the USA population (Vesga-Lopez et al. 2008). This study had several strengths, including the use of diagnostic interviews and a comparison group of non-pregnant women. However, the definition of psychopathology used in this study was a composite of many specific disorders, which reduces its ability to predict specific mental health problems. In addition to depression, anxiety, and other more severe mood disorders, the authors’ definition of psychopathology included any alcohol use disorder, any drug use disorder, and nicotine dependence. While nicotine dependence and mood disorders may have similar risk factors, these conditions are very distinct from one another. Moreover, the mechanisms by which these factors impact both poor birth outcomes and maternal and child postpartum outcomes (proximate and throughout the life course) are likely quite different. In order to understand the factors associated with poor mental health during pregnancy, a more narrow definition is important. The only other population-based studies to date are from Sweden (Borjesson et al. 2005; Larsson et al. 2004; Rubertsson et al. 2003). However, significant differences in the socio-environmental stresses and health care resources experienced by pregnant women in the USA may preclude the generalization of Swedish reports to USA populations.
In summary, additional research is needed to: (1) estimate the national prevalence in the USA of antepartum mental health problems across the pregnancy period; and (2) identify risk factors associated with antepartum mental health problems while controlling for confounders. The current study provides nationally representative prevalence estimates and investigates the independent associations of a wide variety of risk factors with poor antepartum mental health, while examining the effects of mental health before pregnancy.
Our research draws upon a framework of perinatal health developed by Misra and colleagues (2003) that integrates the life course developmental perspective (Halfon and Hochstein 2002) with a model of health determinants (Evans and Stoddart 1990). This model posits that perinatal health is influenced by both cumulative effects of events across the lifespan and intergenerational effects. In addition, multiple determinants and their interactions likely influence women’s health during pregnancy. Central to this framework is the idea that key health determinants prior to and during pregnancy have an important impact on having poor antepartum mental health.
Methods
Data
Medical Expenditure Panel Survey
Data are from the household component of the 1996–2006 Medical Expenditure Panel Survey (MEPS). The household component of the MEPS collects information about medical conditions, health status, healthcare use, and expenditures. The survey has an overlapping panel design, gathering information through five rounds of data collection over a 2.5 year period. Each year, a new panel begins with a sample selected from the households who participated in the previous year’s National Health Interview Survey, which yields a nationally representative sample of the civilian, non-institutionalized population of the USA, with oversampling for blacks and Hispanics. Data are available from 11 panels of the 1996–2006 MEPS. Detailed methodology and a description of data available in MEPS are available at http://www.meps.ahrq.gov.meps.web.
Pregnancy detail files
At each round of data collection, if a woman in the household was pregnant, additional data specific to pregnancy were obtained. Because the pregnancy data are not publicly available, the Agency for Healthcare Research and Quality Data Center created a custom dataset incorporating pregnancy data from each of the 11 panels that was linked to the household component dataset.
Sample
Women included in the 11 panels of the pregnancy detail files who had pre-pregnancy and antepartum data were eligible for this analysis (n=3,552). Subjects were excluded from the dataset if they had missing information on age, race/ethnicity, education, marital/partner status, poverty threshold categories, or physical health status during pregnancy (n=76) or a zero person weight (n=313). Furthermore, women who reported other mental health conditions, besides depression or anxiety, and/or did not report depression, anxiety, or poor self-reported mental health (n=112), were excluded from the analysis. The final sample thus includes 3,051 pregnant women. Only one pregnancy per woman was included in the analysis. If a woman had more than one eligible pregnancy based on the previous exclusion criteria (n=309), a single pregnancy was randomly selected for inclusion in the analysis. A flag was subsequently created to document the number of pregnancies during the MEPS period.
Variable definitions
Dependent variable: antepartum mental health status
A woman was categorized as having poor antepartum mental health if she reported any of the following during an interview round when she was pregnant: (1) having a mental health condition; (2) “feeling sad, blue, or anxious about something”; or (3) being in “fair” or “poor” mental health when asked, “In general, would you say that your mental health is excellent, very good, good, fair, or poor?” (Cohen et al. 1996). A woman was categorized as having poor antepartum mental health if she met any of these conditions for poor mental health during a round that she also reported being pregnant. Self-reported conditions were assigned truncated 3-digit International Classification of Diseases, Ninth Revision (ICD-9) codes. ICD-9 codes 296 (episodic mood disorders), 300 (anxiety state, unspecified), and 311 (depression, unspecified), were considered indicators of poor mental health. While code 296 includes major depressive disorder and other episodic mood disorders, over 96% of women with depression in the sample were identified using code 311 (depression, unspecified).
Independent variables
Maternal and family sociodemographic variables included age, race/ethnicity (white non-Hispanic, Black non-Hispanic, other non-Hispanic, and Hispanic), education (no or some high school, high school graduate, some college, and college graduate or beyond), marital/partner status (married/living with partner, never married, and divorced, separated or widowed), region of the USA (West, Northeast, Midwest, and South), and metropolitan statistical area. Health insurance status was grouped into the following mutually exclusive categories: no health insurance, any publicly funded health insurance (Medicaid and/or Medicare), and private health insurance only. Socioeconomic status was measured using federal poverty thresholds, which adjusts income for family size. Family incomes were classified as below 100% of the federal poverty threshold, 100—199%, 200—399%, and 400% or more of the federal poverty threshold. Number of hours worked per week was dichotomized as less than 35 or 35 or more. Household composition was evaluated by the number of children under 5 years of age, number of children 5–17 years of age, and number of additional adults in the household or family unit (other than the pregnant woman and her partner).
Health-related risk factors
Smoking status while pregnant, substance abuse, the presence of a chronic medical condition, and the presence of sexually transmitted diseases or other urogenital infections, were each evaluated as dichotomous variables. Chronic conditions included: diabetes (ICD-9 code 250), chronic bronchitis (ICD-9 code 491), high cholesterol (ICD-9 code 272), primary hypertensive disease (ICD-9 codes 401–404), asthma (ICD-9 code 493), renal disease (ICD-9 codes 403, 404, 582, 583, 585–588), or hepatitis (ICD-9 code 070). Sexually transmitted diseases and other urogenital infections included: Herpes simplex (ICD-9 code 054), “other disorders of urethra and urinary tract” (ICD-9 code 599; includes urinary tract infection), pelvic inflammatory disease (ICD-9 codes 614–616), “other venereal diseases” (ICD-9 code 099), gonorrhea (ICD-9 code 098), HIV/AIDS (ICD-9 code 042), syphilis (ICD-9 codes 091–097), and trichomonas vaginalis (ICD-9 code 131).
Women who reported “fair” or “poor” health in response to the question, “In general, would you say that your health is excellent, very good, good, fair, or poor?” during any round of the MEPS while pregnant were considered to be in poor physical health during pregnancy. Pre-pregnancy BMI was divided into four categories: <20, 20–24.9, 25–25.9, and≥30. We used pre-pregnancy BMI data reported only in Round 3 of the MEPS, and as such BMI data were not available for all pregnant women during the study period.
Analytic approach
SAS 9.2 (SAS Institute Inc, Gary, NC) was used to construct the analytic files and STATA 10.1 (StataCorp LP, College Station, TX) was used to perform all analyses, accounting for the complex design of the MEPS. The standard errors were corrected due to clustering within strata and the primary sampling unit, and applied survey weights were used to produce estimates that account for the complex survey design, unequal probabilities of selection, and survey non-response.
Due to the high correlation between history of mental health problems and antepartum mental health, all analyses were stratified by history of poor mental health status. Women were categorized as having a history of poor mental health if they met either of the following criteria: (1) self report of anxiety or depression with a date of onset prior to their pregnancy; or (2) self report of “fair” or “poor” mental health during any round preceding the round in which they first reported being pregnant.
Descriptive analysis
Chi-square analyses were used to test for differences in sociodemographic and health characteristics by antepartum mental health status. Each subgroup was tested for statistical significance if differences were found in the overall chi-square tests.
Multivariate analysis
For the logistic regression analysis, models were fit to identify the factors that were associated with the poor antepartum mental health status stratified by history of poor mental health. Based on previous empirical literature, we chose to include the following variables in the models: age (five categories: 14–19, 20–24, 25–29, 30–34, 35+), race/ethnicity (two categories: non-Hispanic white, Hispanic or non-white), marital status (three categories: married/lives with partner, never married, divorced/separated/widowed), education (four categories: no or some high school, high school graduate, some college, and college graduate or beyond), ratio of family income to poverty threshold (four categories: <100%, 100–199%, 200–399%, and ≥400%), and physical health status during pregnancy (two categories: excellent/very good/good, fair/poor). Other variables, described in the Independent Variables section, were considered in the regression analyses; however, some were excluded due to concerns of collinearity or if they did not significantly contribute to the models. Of these other variables, only sexually transmitted diseases or other urogenital infections were significant and were thus included in the final analysis. In addition, we did not include smoking or BMI in the final analysis, since MEPS began collecting data for these variables starting in 2000, leading to inadequate sample size.
The University of Wisconsin—Madison Health Sciences Institutional Review Board considered this study exempt from review because the data were already collected and de-identified.
Results
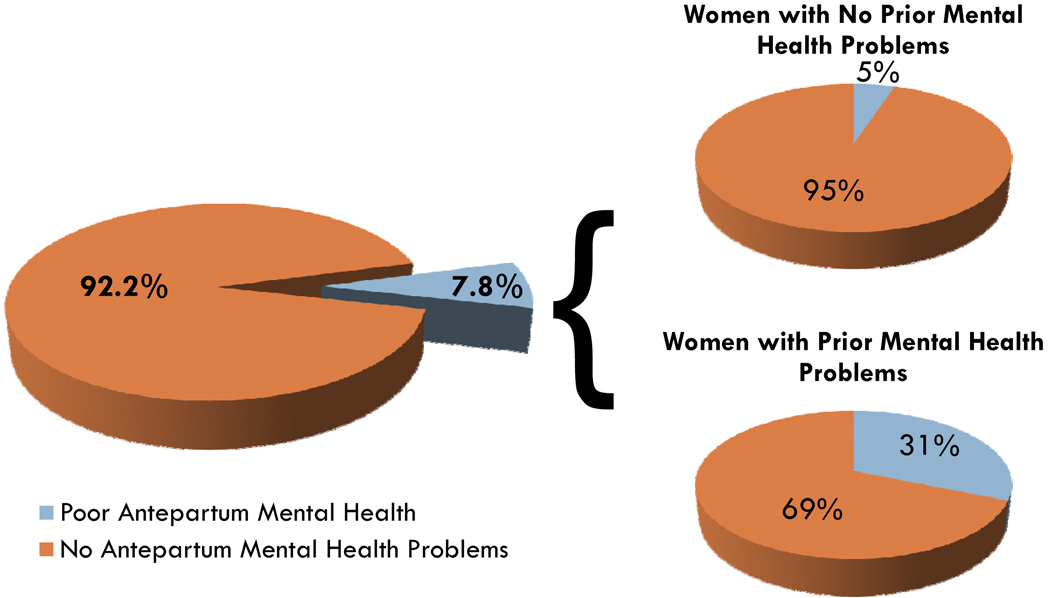
Overall, 7.8% of women in the USA reported poor mental health during their pregnancies. In our initial analysis, a history of poor mental health was the strongest correlate of poor antepartum mental health (unadjusted odds ratio (OR) [95% CI]: 8.45 [6.01–11.88]; adjusted OR [95% CI]: 6.23 [4.25–9.14]; data not shown). Hereafter, all reported results are stratified by history of pre-pregnancy mental health problems. Among women without pre-pregnancy mental health problems, 5% had poor antepartum mental health, while 31% of women with poor pre-pregnancy mental health had poor antepartum mental health (see Fig. 1).
Fig. 1.
Overall prevalence of antepartum mental health problems. The overall prevalence of mental health problems during pregnancy was 7.8%. The prevalence of mental health problems during pregnancy among women with no prior history of mental health problems was 5%. The prevalence of mental health problems during pregnancy among women with a prior history of mental health problems was 31%
Women without prior mental health problems
Among women without prior mental health problems, married women were less likely to have antepartum mental health problems and women who were never married were more likely to have antepartum mental health problems. High school graduates and women who rated their physical health status during pregnancy as “fair” or “poor” were more likely to have poor antepartum mental health. Women living below 100% of the poverty threshold and women with publicly funded insurance were also more likely to have poor antepartum mental health (see Table 1).
Table 1.
Sociodemographic characteristics of pregnant women in the USA, by history of and antepartum mental health status
| No history of mental health problems |
History of mental health problems |
|||||||
|---|---|---|---|---|---|---|---|---|
| Antepartum mental health Problems |
Chi- square Pvalue |
P value for category v. others |
Antepartum mental health Problems |
Chi- square P value |
P value for category v. others |
|||
| Noa | Yesb | Noa | Yesb | |||||
| Total, weightedc (%) | 4,275,176 (94.9%) | 231,849 (5.1%) | 353,276 (68.6%) | 161,816 (31.4%) | ||||
| Total, unweightedd (%) | 2,591 (94.8%) | 142 (5.2%) | 216 (67.9%) | 102 (32.1%) | ||||
| Maternal characteristics | ||||||||
| Age | 0.37 | 0.27 | ||||||
| 14–19 | 9.1% | 11.0% | 0.40 | 14.0% | 19.0% | 0.25 | ||
| 20–24 | 26.3% | 21.8% | 0.38 | 28.0% | 17.8% | 0.07 | ||
| 25–29 | 27.1% | 32.9% | 0.24 | 28.8% | 27.3% | 0.77 | ||
| 30–34 | 25.9% | 19.5% | 0.18 | 15.7% | 19.8% | 0.31 | ||
| 35+ | 11.7% | 14.8% | 0.41 | 13.5% | 16.2% | 0.53 | ||
| Race/Ethnicity | 0.38 | 0.31 | ||||||
| White (non-Hispanic) | 60.7% | 65.5% | 68.6% | 62.3% | ||||
| Non-white or Hispanic | 39.3% | 34.5% | 31.4% | 37.7% | ||||
| Race/Ethnicity | 0.64 | 0.34 | ||||||
| White (non-Hispanic) | 60.7% | 65.5% | 0.38 | 68.6% | 62.3% | 0.31 | ||
| Black (non-Hispanic) | 13.4% | 14.6% | 0.75 | 10.1% | 13.9% | 0.34 | ||
| Other (non-Hispanic) | 6.5% | 4.7% | 0.51 | 5.5% | 2.9% | 0.34 | ||
| Hispanic | 19.4% | 15.3% | 0.23 | 15.8% | 20.9% | 0.17 | ||
| Education status | 0.08 | 0.81 | ||||||
| No or some high school | 21.0% | 22.6% | 0.64 | 32.1% | 37.3% | 0.43 | ||
| High school graduate | 26.6% | 38.3% | 0.02 | 26.8% | 23.8% | 0.53 | ||
| Some college | 21.6% | 17.1% | 0.38 | 24.9% | 22.0% | 0.66 | ||
| College or beyond | 30.8% | 22.0% | 0.12 | 16.2% | 17.0% | 0.84 | ||
| Marital status | 0.03 | 0.01 | ||||||
| Married, lives with partner | 74.2% | 62.9% | 0.01 | 63.3% | 43.7% | 0.005 | ||
| Never married | 22.3% | 32.9% | 0.01 | 28.0% | 36.7% | 0.18 | ||
| Divorced, separated, widowed | 3.4% | 4.2% | 0.64 | 8.7% | 19.6% | 0.02 | ||
| Current smoker while pregnante | n=1,020 | n=65 | 0.35 | n=82 | n=26 | 0.0003 | ||
| Yes | 10.4% | 14.6% | 14.3% | 44.0% | ||||
| No | 89.6% | 85.4% | 85.7% | 56.0% | ||||
| Pre-pregnancy BMIf | n=676 | n=32 | 0.36 | n=81 | n=34 | 0.74 | ||
| <20 | 2.2% | 0.0% | na | 3.5% | 0.0% | na | ||
| 20–24.9 | 56.9% | 53.7% | 0.71 | 46.7% | 48.5% | 0.86 | ||
| 25–29.9 | 24.8% | 21.5% | 0.63 | 27.2% | 23.6% | 0.63 | ||
| 30 or more | 16.0% | 24.8% | 0.11 | 22.5% | 27.9% | 0.36 | ||
| Chronic medical conditions | 0.08 | 0.04 | ||||||
| Yes | 6.5% | 11.1% | 8.6% | 17.9% | ||||
| No | 93.5% | 88.9% | 91.4% | 82.1% | ||||
| Physical health status during pregnancy | <0.0001 | <0.0001 | ||||||
| Excellent, very good, or good | 95.0% | 71.3% | 87.8% | 64.8% | ||||
| Fair or poor | 5.0% | 28.7% | 12.2% | 35.2% | ||||
| Substance abuse | 0.50 | 0.04 | ||||||
| Yes | 0.5% | 1.0% | 0.7% | 5.4% | ||||
| No | 99.5% | 90.0% | 99.3% | 94.6% | ||||
| STD or urogenital infection | 0.67 | 0.003 | ||||||
| Yes | 5.1% | 4.3% | 3.7% | 14.8% | ||||
| No | 94.9% | 95.7% | 96.3% | 85.2% | ||||
| Antepartum rounds | 0.01 | 0.09 | ||||||
| 1 | 69.7% | 55.9% | 71.6% | 80.3% | ||||
| 2 | 30.3% | 44.4% | 28.4% | 19.7% | ||||
| Number of pregnancies during MEPS period | 0.001 | 0.20 | ||||||
| 1 | 89.1% | 96.7% | 90.5% | 85.6% | ||||
| 2 or more | 10.9% | 3.3% | 9.5% | 14.4% | ||||
| Number of hours worked per week | 0.40 | 0.16 | ||||||
| 0–34 | 51.8% | 56.6% | 62.4% | 72.6% | ||||
| 35+ | 48.2% | 43.4% | 37.6% | 27.4% | ||||
| Family characteristics | ||||||||
| Number of adults in household/family | 0.71 | 0.70 | ||||||
| 0 | 75.1% | 76.5% | 71.5% | 69.4% | ||||
| 1 or more | 24.9% | 23.5% | 28.5% | 30.6% | ||||
| Number of children (<5 years old) | 0.97 | 0.24 | ||||||
| 0 | 54.8% | 55.8% | 0.85 | 58.9% | 55.3% | 0.58 | ||
| 1 | 34.6% | 33.4% | 0.80 | 33.3% | 30.2% | 0.56 | ||
| 2 or more | 10.6% | 10.8% | 0.93 | 7.8% | 14.6% | 0.13 | ||
| Number of children (5–17 years old) | 0.24 | 0.68 | ||||||
| 0 | 67.3% | 60.2% | 0.19 | 55.9% | 52.6% | 0.61 | ||
| 1 | 19.8% | 27.8% | 0.06 | 24.9% | 28.0% | 0.58 | ||
| 2 | 8.9% | 8.9% | 0.99 | 14.4% | 16.8% | 0.59 | ||
| 3 or more | 4.0% | 3.2% | 0.61 | 4.7% | 2.5% | 0.30 | ||
| Number of infants born from pregnancy | 0.57 | 0.12 | ||||||
| 0 or missing | 35.0% | 32.1% | 51.7% | 42.1% | ||||
| 1 or more | 65.0% | 67.9% | 48.3% | 57.9% | ||||
| Ratio of family income to poverty threshold | 0.15 | 0.21 | ||||||
| <100% | 18.7% | 27.4% | 0.03 | 28.0% | 30.6% | 0.64 | ||
| 100–199% | 20.0% | 21.4% | 0.76 | 22.4% | 32.5% | 0.08 | ||
| 200–399% | 28.3% | 21.4% | 0.11 | 26.3% | 21.9% | 0.55 | ||
| ≥400% | 33.0% | 29.8% | 0.54 | 23.3% | 14.9% | 0.06 | ||
| Health insurance status | 0.01 | 0.64 | ||||||
| Private insurance only | 70.8% | 61.3% | 0.05 | 60.4% | 54.4% | 0.42 | ||
| Any publicly funded insurance | 16.4% | 27.5% | 0.01 | 25.6% | 30.9% | 0.32 | ||
| No insurance | 12.8% | 11.2% | 0.56 | 14.0% | 14.7% | 0.89 | ||
| Region of USA | 0.58 | 0.86 | ||||||
| Northeast | 16.7% | 20.7% | 0.37 | 10.1% | 9.4% | 0.75 | ||
| Midwest | 21.9% | 23.6% | 0.73 | 25.9% | 28.3% | 0.65 | ||
| South | 36.5% | 36.8% | 0.96 | 38.9% | 35.2% | 0.53 | ||
| West | 25.0% | 18.9% | 0.19 | 25.1% | 27.1% | 0.61 | ||
| MSA status | 0.18 | 0.70 | ||||||
| Urban | 84.8% | 79.2% | 83.5% | 85.4% | ||||
| Rural | 15.2% | 20.8% | 16.5% | 14.6% | ||||
| Survey respondent | 0.34 | 0.13 | ||||||
| Self | 73.3% | 77.3% | 68.9% | 77.8% | ||||
| Other | 26.7% | 22.7% | 31.1% | 22.2% | ||||
BMI body mass index, STD sexually transmitted disease, MEPS Medical Expenditure Panel Survey, MSA Metropolitan statistical area
Self-reported “excellent,” “very good,” or “good,” and no mental health conditions or symptoms reported
Self-reported “fair” or “poor,” or mental health conditions or symptoms reported
Weighted total for no history of mental health problems = 4,507,025; weighted total for history of mental health problems = 515,092
Unweighted total for no history of mental health problems = 2,733; unweighted total for history of mental health problems = 318
Data from 2000–2006 only
Data from 2000–2005 only
In the multivariate analysis, women who were never married were more likely than married women to have poor antepartum mental health (OR [95% CI]: 2.12 [1.25–3.60]). Women in “fair” or “poor” self-rated physical health during pregnancy were more likely to have antepartum mental health problems than women in “excellent,” “very good,” or “good” health (OR [95% CI]: 7.50 [4.52–12.43]), and women who were non-white or Hispanic were less likely to report poor antepartum mental health (OR [95% CI]: 0.56 [0.34–0.93]; see Table 2). There was no difference in the adjusted odds of poor antepartum mental health by family income or by health insurance status.
Table 2.
Multivariate analysesa of the odds of poor antepartum mental health, Stratified by history of mental health problems
| No history of mental health problems |
History of mental health problems |
|||
|---|---|---|---|---|
| Odds of poor/fair mental health during pregnancy |
Odds of poor/fair mental health during pregnancy |
|||
| OR | 95% CI | OR | 95% CI | |
| Maternal characteristics | ||||
| Age | ||||
| 14–19 | 0.73 | 0.32–1.65 | 0.51 | 0.16–1.63 |
| 20–24 | 0.70 | 0.34–1.43 | 0.24 | 0.08–0.70 |
| 25–29 | 1.41 | 0.74–2.68 | 0.49 | 0.19–1.24 |
| 30–34 | 1.00 | Reference | 1.00 | Reference |
| 35+ | 1.69 | 0.76–3.75 | 0.72 | 0.22–2.35 |
| Race/Ethnicity | ||||
| White (non-Hispanic) | 1.00 | Reference | 1.00 | Reference |
| Non-white or Hispanic | 0.56 | 0.34–0.93 | 0.87 | 0.45–1.71 |
| Education status | ||||
| No or some high school | 0.72 | 0.39–1.32 | 0.73 | 0.28–1.85 |
| High school graduate | 1.00 | Reference | 1.00 | Reference |
| Some college | 0.57 | 0.27–1.23 | 0.92 | 0.35–2.41 |
| College or beyond | 0.54 | 0.26–1.11 | 1.24 | 0.37–4.17 |
| Marital status | ||||
| Married, lives with partner | 1.00 | Reference | 1.00 | Reference |
| Never married | 2.12 | 1.25–3.60 | 2.69 | 1.25–5.80 |
| Divorced, separated, widowed | 1.19 | 0.42–3.32 | 3.96 | 1.34–11.74 |
| Sexually transmitted disease or urogenital infection | ||||
| Yes | 0.67 | 0.28–1.62 | 4.48 | 1.36–14.77 |
| No | 1.00 | Reference | 1.00 | Reference |
| Physical health status during pregnancy | ||||
| Excellent, very good, or good | 1.00 | Reference | 1.00 | Reference |
| Fair or poor | 7.50 | 4.52–12.43 | 3.90 | 1.88–8.11 |
| Family characteristics | ||||
| Ratio of family income to poverty threshold | ||||
| <100% | 1.00 | Reference | 1.00 | Reference |
| 100–199% | 0.80 | 0.43–1.49 | 1.59 | 0.72–3.51 |
| 200–399% | 0.67 | 0.34–1.32 | 0.72 | 0.24–2.17 |
| ≥400% | 0.97 | 0.47–1.99 | 0.81 | 0.29–2.26 |
OR odds ratio, CI confidence interval
Stratified logistic regression models are only adjusted for age, race/ethnicity, education status, marital status, sexually transmitted disease or urogenital infection, physical health status during pregnancy, and ratio of family income to poverty threshold
Women with prior mental health problems
Married women were less likely to have antepartum mental health problems while women who are divorced, separated or widowed were more likely to have poor antepartum mental health. “Fair” or “poor” self-rated physical health during pregnancy, current smoking, chronic medical conditions, substance abuse, and the presence of a sexually transmitted disease or urogenital infection were positively associated with antepartum mental health problems in this group (see Table 1).
In the multivariate analysis, those who were 20–24 years old were less likely than those who were 30–34 years old to have mental health problems during pregnancy (OR [95% CI]: 0.24 [0.08–0.70]). Women who were never married or who were divorced, separated, or widowed were more likely to have poor antepartum mental health (OR [95% CI]: 2.69 [1.25–5.80] and OR 3.96 [1.34–11.74], respectively). Those with a sexually transmitted disease (STD) or urogenital infection (OR [95% CI]: 4.48 [1.36–14.77]) or in “fair” or “poor” self-rated physical health during pregnancy (OR [95% CI]:3.90 [1.88–8.11]) were more likely to have mental health problems during pregnancy (see Table 2).
Sensitivity analysis
To generate more confidence in our results, we conducted a series of sensitivity analyses. First, since many of the women with unknown marital status were younger, a sensitivity analysis was conducted assuming all subjects with unknown marital status were “never married.” Results did not substantially change. Further sensitivity analyses were conducted using stricter criteria in the definition of poor mental health. In one analysis, women who self-reported “fair” mental health were not classified as having poor mental health. In another analysis, only those having a self-reported mental health of “fair” or “poor” were classified as having poor mental health. Both of these stricter definitions reduced the number of women categorized as having poor mental health and did not substantially change the estimates in the analysis of the subjects with no previous history of mental health problems. The sensitivity analysis of subjects with a previous history of mental health problems was inconclusive and showed unstable estimates with large confidence intervals due to limited sample size with decreased specificity. Results for these analyses are not shown.
Discussion
This study of a nationally representative sample of pregnant women contributes to our understanding of the prevalence of antepartum mental health problems and its correlates. We found the overall prevalence of poor mental health during pregnancy to be 7.8%, coinciding with previous clinic (Johanson et al. 2000) and population-based (Vesga-Lopez et al. 2008) reports. Specifically, our results are similar to Vesga-Lopez’s findings for any mood disorder and for any anxiety disorder (prevalence 8.5% and 12.2%, respectively). A history of mental health problems has been found to greatly increase the risk for poor antepartum mental health (Bilszta et al. 2008; Borjesson et al. 2005; Buesching et al. 1986; Marcus et al. 2003; Mora et al. 2009; Rich-Edwards et al. 2006; Seguin et al. 1995) and confound associations between other risk factors and poor mental health during pregnancy. Importantly, our study highlights the strong association between a history of mental health problems and antepartum mental health problems.
This finding suggests the need for greater focus on mental health screening for all women of reproductive age regardless of pregnancy status, but especially for women before they become pregnant. Primary care, often the first point of contact for health needs, is an ideal setting for screening. Research shows that patient care and services delivered with a primary care orientation is more likely to be efficient, equitable, and effective (Starfield 2008). The primary care setting may be the best means to ensure comprehensive health assessment and long-term, person-focused care (Starfield 2008); however, coordination of care is essential in order to educate all providers who encounter women at risk. Furthermore, mental health screening should not be limited to the primary care setting and could be conducted when women receive treatment for STDs or urogenital infections, upon hospital admittance, or during a routine OBGYN appointment during pregnancy. In an effort to move towards improved quality of care for women, comprehensive, preventive care, and coordination of care should be maintained longitudinally, before and during pregnancy, as opposed to only during pregnancy when women tend to interface with the healthcare system most frequently.
It is important to note that increased preventive mental health screening is necessary, but not sufficient to ameliorate widespread antepartum mental health problems. To help women manage their depression, coordination of care should facilitate referrals to mental health professionals. Accessibility of professional services and provider expertise should guide the recommendation of treatment options such as pharmacotherapy and psychotherapy. Accordingly, it is essential that health insurance policies include coverage for mental health screening and treatment to ensure that these barriers do not prevent women from receiving proper care.
Our study found that reporting an STD or urogenital infection was an important risk factor for antepartum mental health problems among those with a history of mental health problems. Given these findings, STD screenings may be a promising setting for health care providers to also screen for co-morbid mental health problems. It will be important for providers to become aware of the increased risk of mental health problems in their patients with STDs so as to facilitate the identification of such problems. This may be particularly important in order to ensure that mental health problems are identified in women before they become pregnant, allowing interventions to improve mental health before, during, and following pregnancy.
Our study found that not being married, a proxy for lack of social support, was an important correlate of poor antepartum mental health. Previous studies have shown that those who are married have better psychological well-being than those who are never married, divorced, or separated (Brown 2000), and that the emotional support of a spouse or partner elicits feelings of comfort and reinforces mutual feelings of love and respect, in addition to generating a sense of security (Jacobson 1986; Orr 2004). Other research has shown that the benefits of a supportive relationship, such as enhanced feelings of well-being, personal control, and positive effects, may help moderate the effects of antepartum mental health by making changes associated with pregnancy seem less stressful (Collins et al. 1993). Spousal support may be considered doubly important since it affects the health and well-being of the mothers and the birth outcomes of the unborn children (Feldman et al. 2000; Rubertsson et al. 2003). In the interest of improving maternal psychological well-being and birth outcomes, it is essential to identify women with prior mental health problems who exhibit depression-like symptoms during pregnancy, especially among un-partnered women who may lack social support (Rubertsson et al. 2003).
Our results show that non-white and Hispanic women without a history of mental health problems were less likely than white women to report poor antepartum mental health. This may be the result of underreporting due to patient beliefs and attitudes regarding mental illness, consistent with Cooper et al. (2003), who found that both African-Americans and Hispanics were more likely than whites to have negative beliefs about antidepressants and the effectiveness of treatment. It is also possible that the lack of association in our study stems from preferential diagnosis among white patients compared with African-American and Hispanic patients, as suggested by Borowsky et al. (2000), who reported that physician recognition of mental illness varies by race and ethnicity. Among pregnant women, missed diagnoses or negative views of treatment for depression may delay care or result in many women remaining untreated. This is of particular concern among African-American and Hispanic women, as studies indicate that underutilization of specialty mental health care is highest among these groups (Miranda and Cooper 2004).
Our results also suggest that age affects the likelihood of reporting poor antepartum mental health; specifically, those aged 20–24 years with a history of mental health problems were less likely to report poor mental health during pregnancy. Prior studies indicate that the median age of onset for major depressive disorder, any mood disorder and generalized anxiety disorder is just over 30 years, while the median age for any anxiety disorder is approximately 20 years earlier (Kessler et al. 2005). It is possible that the younger women with a history of poor mental health have a different etiologic profile compared to older women, based on median age of onset for the mental health conditions under study, which could partially explain the lower odds of reporting poor antepartum mental health. As such, younger women may be less likely to experience mood or depressive disorders associated with pregnancy due to the timing associated with psychological sequelae. However, because of the later onset of mood disorders, younger women have a significantly higher lifetime risk of developing mood disorders compared to older women (Kessler et al. 2005); thus, it will be especially important to monitor these women as they age and consider additional pregnancies.
It has been suggested that pregnancy may serve as a barometer for women’s overall health and mental and physical health problems during pregnancy may be predictive of long-term health outcomes (Lykke et al. 2009, 2010). With this in mind, it will be especially important for physicians to monitor women who have maternal depression and provide treatment that will be valuable for women’s health, both in the present and in the future.
Limitations
This study has some potential limitations. First, self-reported mental health items included in MEPS are not parallel to diagnostic criteria used in clinical settings. Given that self-reported global mental health status is not diagnostic, it may not adequately detect women who experience symptoms of depression. However, there is ample evidence that poor mental health is a robust phenomenon and self-reported measures are correlated with major depressive disorder (Hoff et al. 1997).
Due to the lack of data on lifetime mental health status in the MEPS, we had a limited ability to identify women with a history of poor mental health. It is possible that women who have experienced poor mental health to varying degrees throughout their lives may have been coded as not having any history of poor mental health.
Given current data availability, it is challenging to tease apart how various social factors interplay and impact antepartum mental health. Specifically, although it is commonly used in the existing literature as a proxy for social support (Orr 2004; Siegel and Kuykendall 1990), marital status and household composition may be limited in the ability to adequately capture the full impact of this important social factor and likely determinant of mental health. It is also difficult to distinguish genuine depressive episodes from the stresses common during pregnancy. Future research will need better measurement of the connections between antepartum mental health and across the life course risk factors such as life stress, social support and prior mental health problems.
Strengths
This study has some important strengths. Firstly, the results are based on national, population-based data, providing policy-makers and practitioners with a picture of the women at risk for antepartum mental health problems. Additionally, due to the large sample size and rich data set that 11 years of the MEPS provided, several key correlates of poor antepartum mental health could be investigated together in one model, allowing for adjusted estimates of the contributing effect of each characteristic.
Conclusions
This study extends previous clinical and population-based research on antepartum mental health problems among a nationally representative population-based sample of women. Regardless of pre-pregnancy mental health status, women who are not married and who are in poor physical health during pregnancy are more likely to be in poor mental health during pregnancy. These results highlight the importance of directing policy efforts toward providing pregnant women with ample social support as well as appropriate health care to deal with their physical and mental health problems. Identifying women who are at risk for mental health problems during pregnancy and providing adequate treatment options is important to ultimately improve the long-term health of women and their children.
Acknowledgments
We would like to acknowledge the generous funding that supported this research. TD was supported by a grant from the US Department of Health and Human Services, Office of the Assistant Secretary for Planning and Evaluation (3 U01 PE000003-06). EWH was supported by NIH (T32 HD049302; PI: Sarto). MAW was supported by a grant from the Institute for Research on Poverty (PI: Witt and DeLeire). LEW was supported by a grant from the Graduate School of the University of Wisconsin, Madison (PI: Witt) and a pre-doctoral NRSA Training Grant (T32 HS00083; PI: Smith). TM was supported by grants from the Center for Demography and Ecology (R24 HD047873) Center for Demography of Health and Aging (P30 AG17266), and a NIA Training Grant (T32 AG00129).
Contributor Information
Whitney P. Witt, Email: wwitt@wisc.edu, Department of Population Health Sciences, School of Medicine and Public Health, University of Wisconsin, 610 North Walnut Street, Office 503, Madison, WI 53726, USA.
Thomas DeLeire, Department of Population Health Sciences, School of Medicine and Public Health, University of Wisconsin, 610 North Walnut Street, Office 503, Madison, WI 53726, USA.
Erika W. Hagen, Department of Population Health Sciences, School of Medicine and Public Health, University of Wisconsin, 610 North Walnut Street, Office 503, Madison, WI 53726, USA
Margarete A. Wichmann, Department of Population Health Sciences, School of Medicine and Public Health, University of Wisconsin, 610 North Walnut Street, Office 503, Madison, WI 53726, USA
Lauren E. Wisk, Department of Population Health Sciences, School of Medicine and Public Health, University of Wisconsin, 610 North Walnut Street, Office 503, Madison, WI 53726, USA
Hilary A. Spear, Department of Population Health Sciences, School of Medicine and Public Health, University of Wisconsin, 610 North Walnut Street, Office 503, Madison, WI 53726, USA
Erika R. Cheng, Department of Population Health Sciences, School of Medicine and Public Health, University of Wisconsin, 610 North Walnut Street, Office 503, Madison, WI 53726, USA
Torsheika Maddox, Department of Population Health Sciences, School of Medicine and Public Health, University of Wisconsin, 610 North Walnut Street, Office 503, Madison, WI 53726, USA.
John Hampton, UW Comprehensive Cancer Center, Madison, USA.
References
- Andersson L, Sundstrom-Poromaa I, Bixo M, Wulff M, Bondestam K, Astrom M. Point prevalence of psychiatric disorders during the second trimester of pregnancy: a population-based study. Am J Obstet Gynecol. 2003;189(1):148–154. doi: 10.1067/mob.2003.336. [DOI] [PubMed] [Google Scholar]
- Benedict MI, Paine LL, Paine LA, Brandt D, Starlings R. The association of childhood sexual abuse with depressive symptoms during pregnancy, and selected pregnancy outcomes. Child Abuse Negl. 1999;23(7):659–670. doi: 10.1016/s0145-2134(99)00040-x. [DOI] [PubMed] [Google Scholar]
- Bernazzani O, Saucier JF, David H, Borgeat F. Psychosocial factors related to emotional disturbances during pregnancy. J Psychosom Res. 1997;42(4):391–402. doi: 10.1016/s0022-3999(96)00371-6. [DOI] [PubMed] [Google Scholar]
- Billings AG, Moos RH. Comparisons of children of depressed and nondepressed parents: a social-environmental perspective. J Abnorm Child Psychol. 1983;ll(4):463–485. doi: 10.1007/BF00917076. [DOI] [PubMed] [Google Scholar]
- Bilszta JLC, Tang M, Meyer D, Milgrom J, Ericksen J, Buist AE. Single motherhood versus poor partner relationship: outcomes for antenatal mental health. Aust NZ J Psychiatry. 2008;42(1):56–65. doi: 10.1080/00048670701732731. [DOI] [PubMed] [Google Scholar]
- Bolton HL, Hughes PM, Turton P, Sedgwick P. Incidence and demographic correlates of depressive symptoms during pregnancy in an inner London population. J Psychosom Obstet Gynaecol. 1998;19(4):202–209. doi: 10.3109/01674829809025698. [DOI] [PubMed] [Google Scholar]
- Borjesson K, Ruppert S, Bagedahl-Strindlund M. A longitudinal study of psychiatric symptoms in primiparous women: relation to personality disorders and sociodemographic factors. Arch Womens Ment Health. 2005;8(4):232–242. doi: 10.1007/s00737-005-0086-8. [DOI] [PubMed] [Google Scholar]
- Borowsky SJ, Rubenstein L, Meredith LS, Camp P, Jackson-Triche M, Wells KB. Who is at risk of nondetection of mental health problems in primary care? J Gen Intern Med. 2000;15(6):381–388. doi: 10.1046/j.1525-1497.2000.12088.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown SL. The effect of union type on psychological well-being: depression among cohabitors versus marrieds. J Health Soc Behav. 2000;41(3):241–255. [PubMed] [Google Scholar]
- Buesching DP, Glasser ML, Frate DA. Progression of depression in the prenatal and postpartum periods. Women Health. 1986;ll(2):61–78. [PubMed] [Google Scholar]
- Canady RB, Bullen BL, Holzman C, Broman C, Tian Y. Discrimination and symptoms of depression in pregnancy among African American and white women. Womens Health Issues. 2008;18(4):292–300. doi: 10.1016/j.whi.2008.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen JW, Monheit AC, Beauregard KM, Cohen SB, Lefkowitz DC, Potter DEB, Sommers JP, Taylor AK, Arnett RH. The medical expenditure panel survey: a national health information resource. Chicago: Blue Cross Blue Shield Assoc; 1996. pp. 373–389. [PubMed] [Google Scholar]
- Collins NL, Dunkel-Schetter C, Lobel M, Scrimshaw SC. Social support in pregnancy: psychosocial correlates of birth outcomes and postpartum depression. J Pers Soc Psychol. 1993;65(6):1243–1258. doi: 10.1037//0022-3514.65.6.1243. [DOI] [PubMed] [Google Scholar]
- Cooper LA, Gonzales JJ, Gallo JJ, Rost KM, Meredith LS, Rubenstein LV, Wang N, Ford DE. The acceptability of treatment for depression among African-American, Hispanic, and white primary care patients. Med Care. 2003;41(4):479–489. doi: 10.1097/01.MLR.0000053228.58042.E4. [DOI] [PubMed] [Google Scholar]
- Copper RL, Goldenberg RL, Das A, Elder N, Swain M, Norman G, Ramsey R, Cotroneo P, Collins BA, Johnson F, Jones P, Meier AM. The preterm prediction study: maternal stress is associated with spontaneous preterm birth at less than thirty-five weeks’ gestation. National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. Am J Obstet Gynecol. 1996;175(5):1286–1292. doi: 10.1016/s0002-9378(96)70042-x. [DOI] [PubMed] [Google Scholar]
- Da Costa D, Larouche J, Dritsa M, Brender W. Psychosocial correlates of prepartum and postpartum depressed mood. J Affect Disord. 2000;59(l):31–40. doi: 10.1016/s0165-0327(99)00128-7. [DOI] [PubMed] [Google Scholar]
- Davidson K, Jonas BS, Dixon KE, Markovitz JH. Do depression symptoms predict early hypertension incidence in young adults in the CARDIA study? Coronary artery risk development in young adults. Arch Intern Med. 2000;160(10):1495–1500. doi: 10.1001/archinte.160.10.1495. [DOI] [PubMed] [Google Scholar]
- Demyttenaere K, Lenaerts H, Nijs P, Van Assche FA. Individual coping style and psychological attitudes during pregnancy and predict depression levels during pregnancy and during postpartum. Acta Psychiatr Scand. 1995;91(2):95–102. doi: 10.1111/j.1600-0447.1995.tb09747.x. [DOI] [PubMed] [Google Scholar]
- Eaton WW. Epidemiologic evidence on the comorbidity of depression and diabetes. J Psychosom Res. 2002;53(4):903–906. doi: 10.1016/s0022-3999(02)00302-1. [DOI] [PubMed] [Google Scholar]
- Evans RG, Stoddart GL. Producing health, consuming health care. Soc Sci Med. 1990;31(12):1347–1363. doi: 10.1016/0277-9536(90)90074-3. [DOI] [PubMed] [Google Scholar]
- Evans J, Heron J, Francomb H, Oke S, Golding J. Cohort study of depressed mood during pregnancy and after childbirth. BMJ. 2001;323(7307):257–260. doi: 10.1136/bmj.323.7307.257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feldman PJ, Dunkel-Schetter C, Sandman CA, Wadhwa PD. Maternal social support predicts birth weight and fetal growth in human pregnancy. Psychosom Med. 2000;62(5):715–725. doi: 10.1097/00006842-200009000-00016. [DOI] [PubMed] [Google Scholar]
- Gaynes BN, Burns BJ, Tweed DL, Erickson P. Depression and health-related quality of life. J Nerv Ment Dis. 2002;190(12):799–806. doi: 10.1097/00005053-200212000-00001. [DOI] [PubMed] [Google Scholar]
- Gotlib IH, Whiffen VE, Mount JH, Milne K, Cordy NI. Prevalence rates and demographic characteristics associated with depression in pregnancy and the postpartum. J Consult Clin Psychol. 1989;57(2):269–274. doi: 10.1037//0022-006x.57.2.269. [DOI] [PubMed] [Google Scholar]
- Halfon N, Hochstein M. Life course health development: an integrated framework for developing health, policy, and research. Milbank Q. 2002;80(3):433–479. doi: 10.1111/1468-0009.00019. iii. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hays RD, Wells KB, Sherbourne CD, Rogers W, Spritzer K. Functioning and well-being outcomes of patients with depression compared with chronic general medical illnesses. Arch Gen Psychiatry. 1995;52(1):11–19. doi: 10.1001/archpsyc.1995.03950130011002. [DOI] [PubMed] [Google Scholar]
- Henry AL, Beach AJ, Stowe ZN, Newport DJ. The fetus and maternal depression: implications for antenatal treatment guidelines. Clin Obstet Gynecol. 2004;47(3):535–546. doi: 10.1097/01.grf.0000135341.48747.f9. [DOI] [PubMed] [Google Scholar]
- Hobfoll SE, Ritter C, Lavin J, Hulsizer MR, Cameron RP. Depression prevalence and incidence among inner-city pregnant and postpartum women. J Consult Clin Psychol. 1995;63(3):445–453. doi: 10.1037//0022-006x.63.3.445. [DOI] [PubMed] [Google Scholar]
- Hoff RA, Bruce ML, Kasl SV, Jacobs SC. Subjective ratings of emotional health as a risk factor for major depression in a community sample. Br J Psychiatry. 1997;170:167–172. doi: 10.1192/bjp.170.2.167. [DOI] [PubMed] [Google Scholar]
- Holcomb WL, Jr, Stone LS, Lustman PJ, Gavard JA, Mostello DJ. Screening for depression in pregnancy: characteristics of the Beck Depression Inventory. Obstet Gynecol. 1996;88(6):1021–1025. doi: 10.1016/s0029-7844(96)00329-8. [DOI] [PubMed] [Google Scholar]
- Holzman C, Eyster J, Tiedje LB, Roman LA, Seagull E, Rahbar MH. A life course perspective on depressive symptoms in mid-pregnancy. Matern Child Health J. 2006;10(2):127–138. doi: 10.1007/s10995-005-0044-0. [DOI] [PubMed] [Google Scholar]
- Jacobson DE. Types and timing of social support. J Health Soc Behav. 1986;27(3):250–264. [PubMed] [Google Scholar]
- Jaenicke C, Hammen C, Zupan B, Hiroto D, Gordon D, Adrian C, Burge D. Cognitive vulnerability in children at risk for depression. J Abnorm Child Psychol. 1987;15(4):559–572. doi: 10.1007/BF00917241. [DOI] [PubMed] [Google Scholar]
- Jesse DE. Prenatal psychosocial needs: differences between a TennCare group and a privately insured group in Appalachia. J Health Care Poor Underserved. 2003;14(4):535–549. doi: 10.1353/hpu.2010.0670. [DOI] [PubMed] [Google Scholar]
- Jesse DE, Swanson MS. Risks and resources associated with antepartum risk for depression among rural southern women. Nurs Res. 2007;56(6):378–386. doi: 10.1097/01.NNR.0000299856.98170.19. [DOI] [PubMed] [Google Scholar]
- Jesse DE, Walcott-McQuigg J, Mariella A, Swanson MS. Risks and protective factors associated with symptoms of depression in low-income African American and Caucasian women during pregnancy. J Midwifery Womens Health. 2005;50(5):405–410. doi: 10.1016/j.jmwh.2005.05.001. [DOI] [PubMed] [Google Scholar]
- Johanson R, Chapman G, Murray D, Johnson I, Cox J. The North Staffordshire Maternity Hospital prospective study of pregnancy-associated depression. J Psychosom Obstet Gynaecol. 2000;21(2):93–97. doi: 10.3109/01674820009075614. [DOI] [PubMed] [Google Scholar]
- Jonas BS, Lando JF. Negative affect as a prospective risk factor for hypertension. Psychosom Med. 2000;62(2):188–196. doi: 10.1097/00006842-200003000-00006. [DOI] [PubMed] [Google Scholar]
- Kearns RA, Neuwelt PM, Hitchman B, Lennan M. Social support and psychological distress before and after childbirth. Oxford: Blackwell Science Ltd.; 1997. pp. 296–308. [Google Scholar]
- Kelly RH, Russo J, Katon W. Somatic complaints among pregnant women cared for in obstetrics: normal pregnancy or depressive and anxiety symptom amplification revisited? Gen Hosp Psychiatry. 2001;23(3):107–113. doi: 10.1016/s0163-8343(01)00129-3. [DOI] [PubMed] [Google Scholar]
- Kermode M, Fisher J, Jolley D. Health insurance status and mood during pregnancy and following birth: a longitudinal study of multiparous women. Aust NZ J Psychiatry. 2000;34(4):664–670. doi: 10.1080/j.1440-1614.2000.00741.x. [DOI] [PubMed] [Google Scholar]
- Kessler RC. Epidemiology of women and depression. J Affect Disord. 2003;74(1):5–13. doi: 10.1016/s0165-0327(02)00426-3. [DOI] [PubMed] [Google Scholar]
- Kessler RC, McGonagle KA, Zhao S, Nelson CB, Hughes M, Eshleman S, Wittchen HU, Kendler KS. Lifetime and 12-month prevalence of DSM-III-R psychiatric disorders in the United States. Results from the National Comorbidity Survey. Arch Gen Psychiatry. 1994;51(1):8–19. doi: 10.1001/archpsyc.1994.03950010008002. [DOI] [PubMed] [Google Scholar]
- Kessler RC, Berglund P, Demler O, Jin R, Merikangas KR, Walters EE. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):593–602. doi: 10.1001/archpsyc.62.6.593. [DOI] [PubMed] [Google Scholar]
- Klein M, Essex M. Pregnant or depressed? The effect of overlap between symptoms of depression and somatic complaints of pregnancy on rates of major depression in the second trimester. Depression. 1994;2:308–314. [Google Scholar]
- Kornstein SG. The evaluation and management of depression in women across the life span. J Clin Psychiatry. 2001;62 Suppl 24:11–17. [PubMed] [Google Scholar]
- Lancaster CA, Gold KJ, Flynn HA, Yoo H, Marcus SM, Davis MM. Risk factors for depressive symptoms during pregnancy: a systematic review. Am J Obstet Gynecol. 2010;202(l):5–14. doi: 10.1016/j.ajog.2009.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larsson C, Sydsjo G, Josefsson A. Health, sociodemographic data, and pregnancy outcome in women with antepartum depressive symptoms. Obstet Gynecol. 2004;104(3):459–466. doi: 10.1097/01.AOG.0000136087.46864.e4. [DOI] [PubMed] [Google Scholar]
- Lee CM, Gotlib IH. Maternal depression and child adjustment: a longitudinal analysis. J Abnorm Psychol. 1989;98(1):78–85. doi: 10.1037//0021-843x.98.1.78. [DOI] [PubMed] [Google Scholar]
- Lee AM, Lam SK, Mun Lau SMS, Chong CSY, Chui HW, Fong DYT. Prevalence, course, and risk factors for antenatal anxiety and depression. Obstet Gynecol. 2007;110(5):1102–1112. doi: 10.1097/01.AOG.0000287065.59491.70. [DOI] [PubMed] [Google Scholar]
- Leigh B, Milgrom J. Risk factors for antenatal depression, postnatal depression and parenting stress. BMC Psychiatry. 2008;8:11. doi: 10.1186/1471-244X-8-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lykke JA, Langhoff-Roos J, Sibai BM, Funai EF, Triche EW, Paidas MJ. Hypertensive pregnancy disorders and subsequent cardiovascular morbidity and type 2 diabetes mellitus in the mother. Hypertension. 2009;53(6):944–951. doi: 10.1161/HYPERTENSIONAHA.109.130765. [DOI] [PubMed] [Google Scholar]
- Lykke JA, Paidas MJ, Damm P, Triche EW, Kuczynski E, Langhoff-Roos J. Preterm delivery and risk of subsequent cardiovascular morbidity and type-II diabetes in the mother. BJOG. 2010;117(3):274–281. doi: 10.1111/j.1471-0528.2009.02448.x. [DOI] [PubMed] [Google Scholar]
- Marcus SM, Flynn HA, Blow FC, Barry KL. Depressive symptoms among pregnant women screened in obstetrics settings. J Women’s Health Gend-Based Med. 2003;12(4):373–380. doi: 10.1089/154099903765448880. [DOI] [PubMed] [Google Scholar]
- Miranda J, Cooper LA. Disparities in care for depression among primary care patients. J Gen Intern Med. 2004;19(2):120–126. doi: 10.1111/j.1525-1497.2004.30272.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Misra DP, Guyer B, Allston A. Integrated perinatal health framework. A multiple determinants model with a life span approach. Am J Prev Med. 2003;25(l):65–75. doi: 10.1016/s0749-3797(03)00090-4. [DOI] [PubMed] [Google Scholar]
- Monk C, Leight KL, Fang YX. The relationship between women’s attachment style and perinatal mood disturbance: implications for screening and treatment. Arch Womens Ment Health. 2008;11(2):117–129. doi: 10.1007/s00737-008-0005-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mora PA, Bennett IM, Elo IT, Mathew L, Coyne JC, Culhane JF. Distinct trajectories of perinatal depressive symptomatology: evidence from growth mixture modeling. Am J Epidemiol. 2009;169(1):24–32. doi: 10.1093/aje/kwn283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mutale T, Creed F, Maresh M, Hunt L. Life events and low birthweight—analysis by infants preterm and small for gestational age. Br J Obstet Gynaecol. 1991;98(2):166–172. doi: 10.1111/j.1471-0528.1991.tb13363.x. [DOI] [PubMed] [Google Scholar]
- Nayak MB, Al-Yattama M. Assault victim history as a factor in depression during pregnancy. Obstet Gynecol. 1999;94(2):204–208. doi: 10.1016/s0029-7844(99)00267-7. [DOI] [PubMed] [Google Scholar]
- Orr ST. Social support and pregnancy outcome: a review of the literature. Clin Obstet Gynecol. 2004;47(4):842–855. doi: 10.1097/01.grf.0000141451.68933.9f. [DOI] [PubMed] [Google Scholar]
- Orr ST, Blazer DG, James SA. Racial disparities in elevated prenatal depressive symptoms among black and white women in eastern North Carolina. Ann Epidemiol. 2006;16(6):463–468. doi: 10.1016/j.annepidem.2005.08.004. [DOI] [PubMed] [Google Scholar]
- Paarlberg KM, Vingerhoets A, Passchier J, Heiner A, Dekker GA, vanGeijn HR. Psychosocial factors as predictors of maternal well being and pregnancy-related complaints. J Psychosom Obstet Gynaecol. 1996;17(2):93–102. doi: 10.3109/01674829609025669. [DOI] [PubMed] [Google Scholar]
- Pajulo M, Savonlahti E, Sourander A, Helenius H, Piha J. Antenatal depression, substance dependency and social support. J Affect Disord. 2001;65(1):9–17. doi: 10.1016/s0165-0327(00)00265-2. [DOI] [PubMed] [Google Scholar]
- Records K, Rice M. Psychosocial correlates of depression symptoms during the third trimester of pregnancy. JOGN Nurs. 2007;36(3):231–242. doi: 10.1111/j.1552-6909.2007.00140.x. [DOI] [PubMed] [Google Scholar]
- Rich-Edwards JW, Kleinman K, Abrams A, Harlow BL, McLaughlin TJ, Joffe H, Gillman MW. Sociodemographic predictors of antenatal and postpartum depressive symptoms among women in a medical group practice. J Epidemiol Community Health. 2006;60(3):221–227. doi: 10.1136/jech.2005.039370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ritter C, Hobfoll SE, Lavin J, Cameron RP, Hulsizer MR. Stress, psychosocial resources, and depressive symptomatology during pregnancy in low-income, inner-city women. Health Psychol. 2000;19(6):576–585. doi: 10.1037//0278-6133.19.6.576. [DOI] [PubMed] [Google Scholar]
- Rubertsson C, Waldenstrom U, Wickberg B. Depressive mood in early pregnancy: prevalence and women at risk in a national Swedish sample. J Reprod Infant Psychol. 2003;21(2):113–123. [Google Scholar]
- Rudnicki SR, Graham JL, Habboushe DF, Ross RD. Social support and avoidant coping: correlates of depressed mood during pregnancy in minority women. Women Health. 2001;34(3):19–34. doi: 10.1300/J013v34n03_02. [DOI] [PubMed] [Google Scholar]
- Schwartz CE, Dorer DJ, Beardslee WR, Lavori PW, Keller MB. Maternal expressed emotion and parental affective disorder: risk for childhood depressive disorder, substance abuse, or conduct disorder. J Psychiatr Res. 1990;24(3):231–250. doi: 10.1016/0022-3956(90)90013-g. [DOI] [PubMed] [Google Scholar]
- Seguin L, Potvin L, St-Denis M, Loiselle J. Chronic stressors, social support, and depression during pregnancy. Obstet Gynecol. 1995;85(4):583–589. doi: 10.1016/0029-7844(94)00449-N. [DOI] [PubMed] [Google Scholar]
- Siegel JM, Kuykendall DH. Loss, widowhood, and psychological distress among the elderly. J Consult Clin Psychol. 1990;58(5):519–524. doi: 10.1037//0022-006x.58.5.519. [DOI] [PubMed] [Google Scholar]
- Starfield B. The future of primary care: refocusing the system. N Engl J Med. 2008;359(20):2087–2091. doi: 10.1056/NEJMp0805763. [DOI] [PubMed] [Google Scholar]
- Vesga-Lopez O, Blanco C, Keyes K, Olfson M, Grant BF, Hasin DS. Psychiatric disorders in pregnant and postpartum women in the United States. Arch Gen Psychiatry. 2008;65(7):805–815. doi: 10.1001/archpsyc.65.7.805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weissman MM, Jensen P. What research suggests for depressed women with children. J Clin Psychiatry. 2002;63(7):641–647. doi: 10.4088/jcp.v63n0717. [DOI] [PubMed] [Google Scholar]
- Weissman MM, Olfson M. Depression in women: implications for health care research. Science. 1995;269(5225):799–801. doi: 10.1126/science.7638596. [DOI] [PubMed] [Google Scholar]
- Weissman MM, Gammon GD, John K, Merikangas KR, Warner V, Prusoff BA, Sholomskas D. Children of depressed parents. Increased psychopathology and early onset of major depression. Arch Gen Psychiatry. 1987;44(10):847–853. doi: 10.1001/archpsyc.1987.01800220009002. [DOI] [PubMed] [Google Scholar]
- Wells KB, Burnam MA, Rogers W, Hays R, Camp P. The course of depression in adult outpatients. Results from the Medical Outcomes Study. Arch Gen Psychiatry. 1992;49(10):788–794. doi: 10.1001/archpsyc.1992.01820100032007. [DOI] [PubMed] [Google Scholar]
- Westdahl C, Milan S, Magriples U, Kershaw TS, Rising SS, Ickovics JR. Social support and social conflict as predictors of prenatal depression. Obstet Gynecol. 2007;110(1):134–140. doi: 10.1097/01.AOG.0000265352.61822.1b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams JB, Spitzer RL, Linzer M, Kroenke K, Hahn SR, deGruy FV, Lazev A. Gender differences in depression in primary care. Am J Obstet Gynecol. 1995;173(2):654–659. doi: 10.1016/0002-9378(95)90298-8. [DOI] [PubMed] [Google Scholar]
- Zuckerman BS, Beardslee WR. Maternal depression: a concern for pediatricians. Pediatrics. 1987;79(1):110–117. [PubMed] [Google Scholar]