Abstract
Liver transplantation radically changed the philosophy of hepatology practice, enriched multiple areas of basic science, and had pervasive ripple effects in law, public policy, ethics, and theology. Why organ engraftment was feasible remained enigmatic, however, until the discovery in 1992 of donor leukocyte microchimerism in long-surviving liver, and other kinds of organ recipients. Following this discovery, the leukocyte chimerism-associated mechanisms were elucidated that directly linked organ and bone marrow transplantation and eventually clarified the relationship of transplantation immunology to the immunology of infections, neoplasms, and autoimmune disorders. We describe here how the initially controversial paradigm shift mandated revisions of cherished dogmas. With the fresh insight, the reasons for numerous inexplicable phenomena of transplantation either became obvious or have become susceptible to discriminate experimental testing. The therapeutic implications of the “new immunology” in hepatology and in other medical disciplines, have only begun to be explored. Apart from immunology, physiologic investigations of liver transplantation have resulted in the discovery of growth factors (beginning with insulin) that are involved in the regulation of liver size, ultrastructure, function, and the capacity for regeneration. Such studies have partially explained functional and hormonal relationships of different abdominal organs, and ultimately they led to the cure or palliation by liver transplantation of more than 2 dozen hepatic-based inborn errors of metabolism. Liver transplantation should not be viewed as a purely technologic achievement, but rather as a searchlight whose beams have penetrated the murky mist of the past, and continue to potentially illuminate the future.
Legacy: Something immaterial, as a style or philosophy, that is passed from one generation to another. Anything handed down from, or as from, an ancestor.
During the quarter century that coincided with the birth and lifetime of the journal Hepatology (1981–2005), the philosophy and practice of hepatology were dramatically transformed by the wide acceptance of orthotopic liver transplantation. Numerous milestones in the development and use of this procedure had been reached between 1955 and 1980 (Table 1).1–36 The prodigious task that lay ahead in 1981 was diffusion of the complex new multidisciplinary enterprise into the national and international healthcare systems (Table 1). 37–73 The extent to which this was accomplished is evident in the 2004 Action Plan for Liver Disease Research designed to “…coordinate research efforts [to treat hepatic and biliary disease] across the NIH”.74
Table 1.
Milestones of Liver Transplantation
| Year | Description | Reference |
|---|---|---|
| 1955 | First article in the literature on auxiliary liver transplantation | 1 |
| 1956 | Concept of liver replacement first mentioned | 2 |
| 1958–1960 | Results reported of canine total hepatectomy and liver replacement without immunosuppression | 3,4 |
| 1960 | Abdominal multivisceral transplantation described in non-immunosuppressed dogs | 5 |
| 1963 | Azathioprine-prednisone cocktail introduced (kidneys first, then livers) and recognition of organ-induced tolerance | 6 |
| 1963 | Description of in situ preservation and procurement of cadaveric organs | 3,7 |
| 1963 | First attempts to transplant the human liver | 8 |
| 1964–1965 | Evidence reported of hepatotrophic (liver-supporting) factor(s) in portal venous blood | 9,10 |
| 1965 | Liver-induced tolerance under a short course of azathioprine reported in dogs | 11 |
| 1966 | First liver xenotransplantation on July 15, 1966 (chimpanzee donor) | 12 |
| 1966 | Clinical introduction of ALG for kidney, then liver recipients | 13 |
| 1966–1970 | Evidence that HLA matching would not be a major factor in cadaveric organ transplantation | 14,15 |
| 1967 | First 1-year survivals after human liver replacements | 16 |
| 1967–1968 | Acceptance of brain death concept | 17 |
| 1967–1969 | Liver-induced tolerance in pigs without immunosuppression | 18–20 |
| 1969 | First textbook of liver transplantation based on 25 Denver cases | 21 |
| 1969 | First palliation (or cure) of inborn error of metabolism with liver transplantation | 22 |
| 1973–1983 | Evidence accrued that the liver controls cholesterol homeostasis | 23–27 |
| 1973 | Description of the liver’s resistance to antibody-mediated rejection | 28 |
| 1973–1975 | Principal portal blood hepatotrophic factor identified as insulin | 29,30 |
| 1976 | Causes of failure analyzed in first 93 Colorado cases of liver transplantation | 31 |
| 1976 | Improved slush liver preservation permitted long-distance procurement | 32,33 |
| 1979 | Systematic use of arterial and venous grafts for cadaver liver revascularization | 34 |
| 1979 | Cyclosporine introduced for organ transplantation including 2 liver recipients | 35 |
| 1980 | Cyclosporine-steroid cocktail introduced clinically | 36 |
| 1981 | 80% 1-year liver recipient survival reported using cyclosporine-prednisone | 37 |
| 1982 | Review of progress in liver transplantation generates widespread interest of hepatologists | 38 |
| 1983 | Introduction of pump-driven venovenous bypass without anticoagulation | 39–41 |
| 1983–1995 | USA consensus development conference conclusion that liver transplantation is a service (1983) is followed by rapid proliferation of transplant centers worldwide | 42 |
| 1984 | Standardization of in situ preservation-procurement techniques for cadaver multiple organ donors | 43,44 |
| 1984 | Reversibility reported of B cell malignancies (PTLD) in liver and other organ recipients | 45 |
| 1984 | Reports of reduced-size liver grafts for pediatric recipients | 46,47 |
| 1984 | Liver transplantation of patient with hypercholesterolemia verified hypothesis that the liver is the site of cholesterol homeostasis | 48,49 |
| 1987–1989 | First successful transplantation of liver-containing multivisceral grafts | 50,51 |
| 1987 | University of Wisconsin (UW) solution improves preservation of liver and other organs | 52–54 |
| 1987 | Successful extensive use of livers from “marginal” donors reported | 55 |
| 1988 | National adoption of Pittsburgh point system for cadaver kidney and liver distribution complies with Organ Transplant Act of 1984 | 56,57 |
| 1989 | Popularization of the “piggyback” variation of liver transplantation | 58 |
| 1989 | Clinical introduction of FK 506-(tacrolimus)-based immunosuppression | 59,60 |
| 1989 | First report of split cadaver liver for transplantation into 2 recipients | 61 |
| 1990 | First use of live liver donors (left side fragments) | 62,63 |
| 1992–1993 | Discovery of donor leukocyte microchimerism in liver (and other organ) recipients, placing organ and bone marrow cell transplantation on common ground. | 64,65 |
| 1998 | Delineation of analogies between transplantation and infection immunology | 66 |
| 1994–1999 | Live donor transplantation of right side liver fragments | 67–69 |
| 2001 | Mechanism-based tolerogenic immunosuppression proposed | 70 |
| 2003 | Double knockout of porcine α1,3GT gene, revitalizing hopes of clinical xenotransplantation | 71 |
| 2003 | Clinical use of tolerogenic immunosuppression | 72 |
| 2005 | Mechanisms of recurrent hepatitis under transplant immunosuppression elucidated. | 73 |
Abbreviation: ALG, antilymphocyte globulin.
Research and Development Opportunities
The NIH plan was divided into 16 chapters, one of which was devoted exclusively to liver transplantation, with primary emphasis on clinical research. The liver transplant chapter began with the simple declarative sentence, “Liver transplantation is now the standard of care for patients with end stage liver disease or acute liver failure.”74 It was a proud statement from the government agency whose unfailing support had made this possible. But, had liver transplantation matured so completely that there is nothing left to do but fine tuning? This view is negated by links to liver transplantation in almost all of the 15 other chapters of the NIH prospectus. Most of these links were to targets of research opportunity that already had been enriched by, or even owed their provenance to, liver transplantation.
For example, techniques of liver procurement, preservation, and replacement are currently being adapted in non-transplant circumstances (e.g., for subtotal hepatic resections). The discovery that portal venous blood contains substances important for maintenance of liver size, function, and the capacity for regeneration was the beginning of the still-evolving special field of hepatotrophic physiology that is concerned with the functional and hormonal interrelationships of the different splanchnic organs.24,29,30,75–77 The hepatotrophic studies ultimately led to the cure or palliation with liver replacement of numerous hepatic-based inborn errors of metabolism,22,48,78 providing the first examples of what might be accomplished in the future with gene therapy and the application of stem cell biology. Finally, religious beliefs, concerns about medical ethics, and public policy or legal issues that surfaced 4 decades ago with the first attempts of liver transplantation42,79,80 remain as unresolved agenda items in the NIH master plan of 2004.
However, the most frequently identified potential research initiatives in the NIH strategic plan of 2004 concerned the immune response or the manifold consequences of modifying it, not just for transplantation but also in the context of hepatitis, HIV, and oncology (to which separate chapters of the plan were devoted). Using today’s sophisticated tools (particularly those of molecular biology), it now may be possible to expand the sphere of immunology in new directions, fill in knowledge gaps, explain long-standing enigmas, and contribute ultimately to better patient care. With this in mind, the following discussion considers specific issues of immunology that are central to the further development of liver transplantation and to improvement of treatment under multiple nontransplant circumstances.
The Relation of Alloengraftment to Acquired Immune Tolerance: The Historical View
Bone Marrow Transplantation
Transplantation immunology was brought to its current state by a series of events that began in 1943–44 with Medawar’s demonstration that rejection is an immune response.81 A year later, Owen discovered mixed blood cell chimerism in Free- martin cattle whose fused placentas had permitted fetal cross-circulation82; such animals were subsequently shown to be mutually tolerant. Then, in 1953–55, the strong association of donor leukocyte chimerism and acquired donor-specific tolerance was demonstrated in experiments in which allogeneic spleen and bone marrow cells were transplanted without immunosuppression into immunologically immature mice83 and into irradiated adult mouse recipients.84 After hematolymphopoietic cell engraftment, the recipients could accept all other donor tissues and organs. The mouse tolerance models escalated over the ensuing 15 years to clinical bone marrow transplantation in immunodeficient and irradiated patients. However, success depended on the use of HLA-matched donors. Otherwise, the penalty for engraftment was lethal graft-versus-host disease (GVHD): rejection of the host by the graft.
Organ Transplantation
In contrast to the “bench to bedside” chronology of bone marrow transplantation, organ transplantation (initially of the kidney) was accomplished in humans85,86 before proof of feasibility was demonstrated in an animal model and in the apparent absence of leukocyte chimerism. The first 6 kidney recipients with prolonged graft survival (1959–1962) were preconditioned with sublethal total body irradiation but were not infused with donor bone marrow cells.86–88 In 1960–61, daily post-transplantation azathioprine was shown to prolong kidney survival in dogs89 and taken to clinical trials. Used alone or with other cytotoxic agents, azathioprine was only marginally effective.90,91 However, its combination with prednisone made renal transplantation a practical service by exposing 2 features of the alloimmune response6 that later were demonstrated with liver and all other kinds of organ transplantation and under all other regimens of immunosuppression.
The first unexplained observation was that rejections that developed under azathioprine were easily reversed with the addition of large doses of prednisone rather than being inexorable as previously thought. Second, a successful rejection reversal frequently was succeeded by a greatly reduced requirement for maintenance immunosuppression (Fig. 1), suggesting that the graft was inherently tolerogenic.6 It also was learned that histocompatibility matching was not a prerequisite for success, that there was little threat of GVHD, and that perpetuation of organ graft survival almost always depended on lifetime drug treatment. In addition to these striking differences from bone marrow transplantation, none of the organ recipients were thought to have donor leukocyte chimerism (Table 2).
Fig. 1.
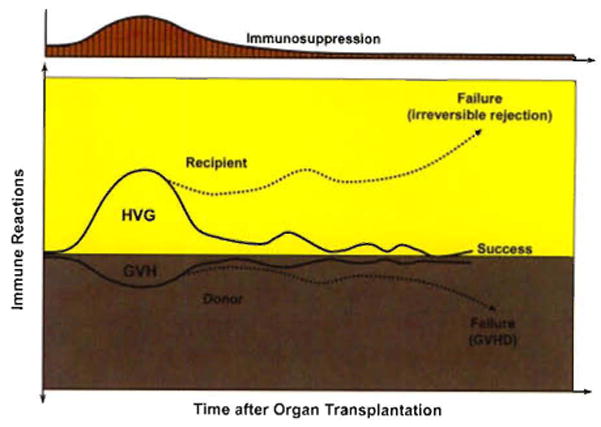
Contemporaneous host-versus-graft (HVG) and graft-versus-host (GVH) immune responses after transplantation. The presence of the weaker GVH response in organ recipients (the inverted curves on a gray background) was not recognized until the discovery of microchimerism in 1992.64,65 However, the demonstration in 1962–636 that the HVG response (rejection) is reversible and may be succeeded by a state of variable tolerance (yellow panel) allowed the practical development of immunosuppression-aided clinical organ transplantation.
Table 2.
Differences Between Clinical Organ Transplantation and Bone Marrow Transplantation
| Feature Organ | Transplantation Bone | Marrow Transplantation |
|---|---|---|
| Host cytoablation | No | Yes* |
| HLA matching | Not essential | Critical |
| Principal complication | Rejection | Graft-versus-host disease |
| Immunosuppression-free | Uncommon | Common |
| Term for success | Acceptance | Tolerance |
| Leukocyte chimerism | No | Yes |
Abreviation: HLA, human leukocyte antigen.
This therapeutic step allows a relatively unopposed graft-versus-host reaction and accounts for the other differences.
Because of these striking disparities, organ engraftment and successful bone marrow transplantation were considered for many years to involve fundamentally different mechanisms. Experimental therapeutic strategies were empirically developed with the objective of endowing organ recipients with the donor leukocyte chimerism-associated mechanisms of the bone marrow recipient while avoiding the penalty of GVHD.92,93 These strategies92,94–97 had in common the infusion of donor hematolymphopoietic cells into organ recipients that had been immunologically weakened by irradiation, antilymphoid antibody preparations, or other means (i.e., non-myelotoxic cytoreduction). Although encouraging experimental results have been reported,98,99 such protocols have not found a significant niche in clinical organ transplantation practice because of their complexity, risks, and unpredictable consequences.
Nevertheless, this body of experimental work demonstrated that the establishment of a hematolymphopoietic population composed of donor and recipient cells was possible in some models with a reasonably low risk of GVHD and could result in donor-specific tolerance, providing the donor cell contribution was at least 1% to 2% (“macrochimerism”). Levels below this (“microchimerism”) were interpreted as either negative findings or artifacts. By so doing, the historical paradigm that attributed bone marrow and organ engraftment to different mechanisms required no substantive revision.
A Unification of Bone Marrow and Organ Transplantation
The historical paradigm was not challenged until 1992. When small numbers of multilineage donor hematolymphopoietic cells (microchimerism) were found in animal and human recipients of long-surviving kidney and liver allografts,64,65,100–102 it was postulated that the mechanisms of organ engraftment differed only in degree from the leukocyte chimerism-dependent ones of successful bone marrow transplantation. Organ engraftment was now defined as a variable form of tolerance that resulted from “…responses of coexisting donor and recipient immune cells, each to the other, causing reciprocal clonal expansion followed by peripheral clonal deletion.”64,65 The graft-versus-host arm of the double immune reaction (the inverted curve in Fig. 1) usually was clinically inapparent.
The prerequisite for the donor-specific tolerance was migration of the graft’s passenger leukocytes to host lymphoid organs and induction there of the host-versus-graft response. Because the multilineage passenger leukocytes of an organ are of bone marrow origin, their hematogenous migration into the recipient was in essence the equivalent of a bone marrow cell infusion. Exhaustion and deletion of the antidonor response explained the characteristic rejection reversal and subsequent decline in need for immunosuppression in organ recipients (Fig. 1). Cytoablation of bone marrow, but not of organ recipients, was the apparent reason for essentially all of the differences between the two kinds of transplantation, including the high risk to the bone marrow recipient of GVHD, and the need to restrict marrow donors to those with a histocompatibility match103 (Table 2). Essentially all cytoablated bone marrow recipients have a small residual population of their own hematolymphopoietic cells (i.e., mirror image microchimerism) rather than complete bone marrow replacement.104
This unified view of transplantation was controversial,98,105–110 in part because it was incompatible with dogmas that made up much of the foundation of transplantation immunology. One point of contention was our view that historically rooted alternative engraftment mechanisms (listed in Table 3) were either epiphenomena or “variants or stages” of the clonal exhaustion-deletion that followed the key event of leukocyte migration.64 In addition, the role of small numbers of persistent cells (the microchimerism) in perpetuation of long-term graft survival was not yet clear. Finally, the crucial mechanism of clonal exhaustion-deletion was still generally considered to be only a theory. Although we did not know it at the time, precisely these issues were being addressed independently in Zurich but in the context of immune responsiveness and unresponsiveness to non-cytopathic microparasites (see below).
Table 3.
Mechanisms of Immune Nonreactivity*
Essential
|
Alternative
|
Modified from Starzl TE. J Am Coll Surg 2005;201:160–170. Reprinted with permission.
Tolerance From the Infection Perspective
The major histocompatibility complex–restricted mechanisms of T cell recognition of, and response to, noncytopathic microorganisms, and to allografts, had been elucidated by Zinkernagel and Dougherty in the 1970s,111–113 but the mechanisms of acquired immune nonreactivity remained a puzzle. During the early 1990s, Zinkernagel and his associates in Zurich formally proved in mouse infection models that the anti-pathogen T cell response could be exhausted and deleted114 and concluded that this was the explanation for the carrier state that may develop after infection with noncytopathic (intracellular) viruses.115 As with transplantation, the critical step was migration of the pathogen or its peptide to host lymphoid organs. The presence of virus that failed to reach these destinations was not recognized by the host. This was termed immune indifference (now called immune ignorance). In essence, Zinkernagel et al. had clarified the interrelationship of immunity and tolerance to pathogens, and had defined tolerance in terms of two essential mechanisms: immune ignorance and clonal exhaustion deletion. Moreover, they placed these mechanisms in the same dynamic context of antigen migration as that of transplantation.115
Once the viral antigen reached lymphoid destinations and induced a cytolytic T lymphocyte (CTL) response, the outcomes in the highly controlled models of lymphocytic choriomeningitis virus (LCMV) infection were determined by the balance between the amount of virus antigen and the number of induced antigen-specific CTL.115 Analogous to the maximal flood of passenger leukocytes migrating from a transplanted organ, the critical period was during the first few days or weeks of viral replication. If the CTL were induced in sufficient numbers, the result was disease control; if not, the result was clonal exhaustion-deletion and a carrier syndrome that ranged from asymptomatic to progressively more serious disease states. No matter what balance was established acutely, perpetuation of this balance (whether immunity, deletional tolerance, or some stage in between) depended on persistence of antigen with access to host lymphoid organs. The ability of small amounts of persistent virus to survive was attributed to its relocation in non-lymphoid sites that were inaccessible to host immune effector mechanisms.115 From these protected niches, the virus migrated secondarily to host lymphoid organs and could sustain immunity, or alternatively, maintain tolerance. Thus, persistent antigen was a two-edged sword.
The Boundary Between Immunity and Tolerance
The analogies between transplantation and infection were summarized in 1998 and generalized to other branches of immunology in the following statement: “…Migration and localization are the governing factors in immunologic responsiveness or unresponsiveness against infections, tumors and self, and against xenografts and allografts.”66 All of the clinical scenarios of transplantation, and those resulting from infection by noncytopathic pathogens, could be correlated with the routes of migration and the ultimate localization of the respective antigens. The “gray area” between unequivocal immunity and durable tolerance included a diversity of transplant outcomes short of outright acute irreversible rejection, as well as a panoply of analogous virus carrier states; all represented different degrees of partial tolerance. Such immunologic “compromises” included chronic allograft rejection and its analog, chronic hepatitis.66
The Rejection Option
Organ Transplantation
The antigen migration is essentially the same with transplantation of the liver and any other surgically revascularized whole organ (Fig. 2A). Movement of the passenger leukocytes is selective at first to host lymphoid organs, where a clonal antidonor T cell response is induced. The CTL then target the transplanted organ as well as the peripheralized cells of the source graft (Fig. 2A).
Fig. 2.
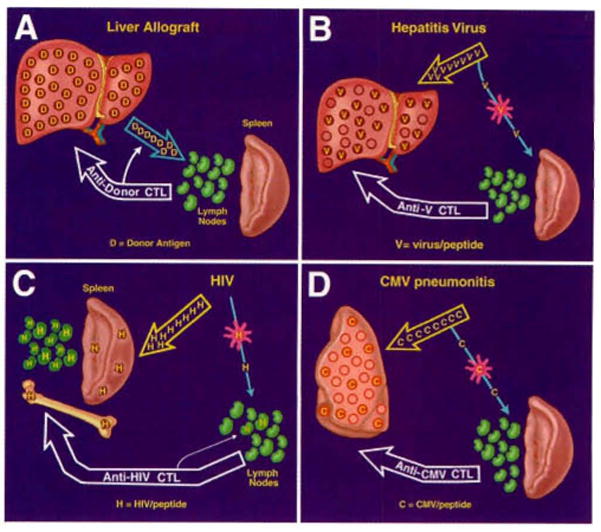
The consequences of antigen spread and localization (see text): (A) with liver or other kinds of organ transplantation; (B) with a viral hepatitis infection; (C) with a human immunodeficiency virus (HIV) infection; (D) with a pulmonary cytomegalovirus infection.
Infection
The migratory principles after infection by noncytopathic microparasites are the same as those of passenger leukocytes, but the details differ because both the pathways of microorganism migration and the targets of the CTL response are dictated by the tropism of the various pathogens (Fig. 2B–D). For example, because of the liver tropism of the hepatitis viruses, the quantity of viral antigen that migrates to host lymphoid organs is small compared with that homing to the liver (Fig. 2B). At the lymphoid organs, hepatitis virus–specific CTL are induced by infected antigen-presenting cells (APC) displaying complexes of major histocompatibility complex molecules plus peptides derived from the pathogen. Because the induced CTL then destroy infected host cells no matter where these “non-self cells are located, the principal disease expression is hepatic (Fig. 2B). Infections with other intracellular viruses (Fig. 2C–D) are variations on the same theme (see later discussion).
The Tolerance Option Without Immunosuppression
Organ Transplantation
Exceptions to the outcome of immunity (i.e., rejection) in untreated transplant recipients are rare. However, lifetime tolerance to liver allografts occurs without therapeutic manipulation in approximately 20% of outbred pig recipients18–20,116,117 and with nearly 100% regularity using selected donor–recipient rat strain combinations,118–120 and most mouse strain combinations.121 Less well leukocyte–endowed mouse heart121,122 and kidney allografts123 also reliably self-induce tolerance, although in far fewer strain combinations. These models of spontaneous transplantation tolerance demonstrate that graft-induced immune non-reactivity is a normal potential option of the immune response (Fig. 3A). Importantly, the “spontaneous tolerance” may be abrogated in some of these models by administering immunosuppression.124–128
Fig. 3.
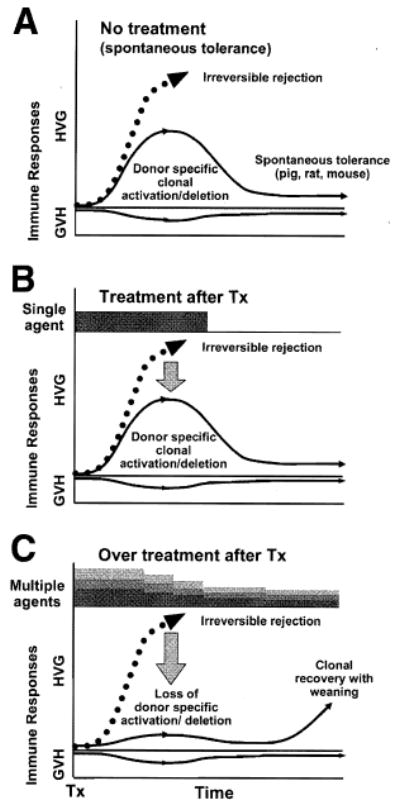
Organ-induced tolerance. (A) Spontaneous tolerance induced by the liver in experimental models18–21,118–121 in which the host-versus-graft immune response induced by the migratory donor leukocytes is too weak to eliminate the donor antigen and is exhausted and deleted (the rise and fall of the continuous thin line). Maintenance of engraftment depends on small numbers of persistent donor leukocytes (microchimerism). The less well–leukocyte-endowed heart and kidney also can “self- induce” engraftment but in a much smaller number of models. (B) Immunosuppression-aided tolerance in organ transplantation models in which the recipient response that normally would cause rejection (dashed line) is reduced into a deletable range (continuous thin line) with a short course of early post-transplantation immunosuppression. Lifetime tolerance after stopping immunosuppression was first convincingly demonstrated in canine liver recipients.11 (C) A self-defeating consequence of excessive prophylactic over-immunosuppression (depicted with multilayered bars) that subverts efficient clonal exhaustion- deletion. The initially over-treated recipient may be committed to unnecessarily high maintenance immunosuppression. This undesirable effect of too much immunosuppression was not recognized until 2001.70 Tx = Transplantation
Infection
After most infections by noncytopathic parasites, the balance between antigen and antigen-specific CTL tilts within a few weeks to immunity. The exceptions in which protective immunity frequently does not develop constitute a collection of some of the world’s most difficult-to-treat diseases [e.g., acquired immune deficiency syndrome (AIDS), hepatitis, and malaria].
Therapeutic Implications
Infections With Noncytopathic Microorganisms
In 1994, Zinkernagel and Hengartner discussed the comparative pathophysiology of HIV (a retrovirus) and hepatitis B virus (HBV; a DNA virus) in the context largely developed with their studies of the LCMV (an RNA virus). The authors argued that the consequences of HIV were caused principally by virus-specific cytotoxic T cell–mediated immunopathology rather than by direct cytolytic effects or any other mechanism.129 The essence of their argument is shown in Fig. 2C. After HIV antigen delivery to lymphoid organs by infected dendritic cells or other APC, a CTL response is induced that targets CD4 (T helper) and all other infected cells. With the selective destruction of infected CD4 cells and eventual decimation of the infected APC population, the inadequately renewed HIV-specific CTL clone is easily exhausted and deleted and, eventually, the entire CTL population dwindles. Thus, the immunodeficiency of AIDS may be prevented at the outset by a strong CTL response, or alternatively, the response may cause immunodeficiency. The outcome from the events shown in Fig. 2C depends on the balance established at an early time between the amount and localization of the virus versus the CTL response. A similar pathogenesis, but with variable targets and outcomes, pertains with all noncytopathic microparasites: for example, hepatitis (Fig. 2B) and cytomegalovirus (Fig. 2D).
From this point of view, Zinkernagel and Hengartner had suggested in 1994 that the foremost therapeutic objective under most circumstances is to limit the extent of the infected cell targets, or in clinical terms to reduce the viral load and restrict its spread.129 This has since been accomplished in HIV victims with zidovudine and protease inhibitors. Once a large target is established, the deliberate resurrection of a strong CTL response could have the disastrous consequence of widespread host cell killing. A better option under these circumstances might be the administration of T cell–directed immunosuppression in just the right amount to prevent the CTL-mediated destruction of massive numbers of host cells, but not so much that runaway viremia would merely expand the target for attack from a recovering CTL clone. Thus, the therapeutic aims in an intractable noncytopathic pathogen infection would be to predict, monitor, and equilibrate beneficial balances between pathogen distribution and the absence of an immunopathologic T cell response.
The Organ Engraftment Objective
Comparable antigen/CTL balances must be found and kept stable for successful transplantation. The best chance to establish a balance selectively favoring tolerance is during the first few post-transplantation weeks, during which the maximal donor leukocyte migration provides the optimal conditions for reciprocal exhaustion-deletion of the double immune response (Figs. 1, 3B). This one time only the window of opportunity could be narrowed or closed by so much prophylactic immunosuppression that clonal activation is subverted. To the extent this were to occur, later reduction of the primary over-treatment predictably would lead to recovery of the ineffectively deleted clone with the clinical consequence of delayed rejection (Fig. 3C).70 However, the penalty of too little immunosuppression during the critical early period may be irreversible rejection. In 2001, this dilemma, it was thought, could be addressed by application of one or both of the therapeutic principles depicted in combination in Fig. 4.70
Fig. 4.
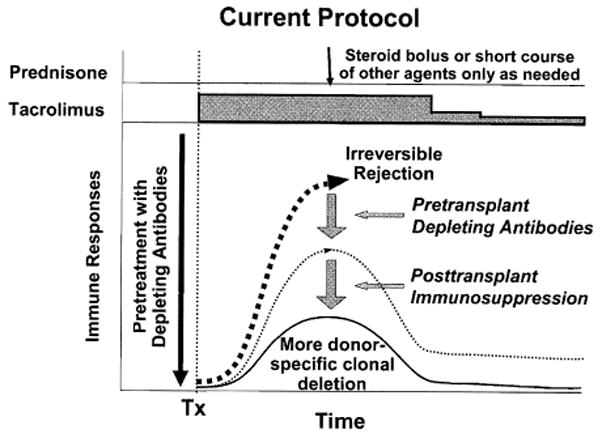
Protocol of “tolerance friendly” immunosuppression introduced at the University of Pittsburgh Medical Center.70 An antilymphoid antibody infusion before arrival of an allograft reduces the anticipated antidonor response into a more readily deletable range and allows maintenance treatment to begin with daily monotherapy to which other agents are added only for rejection. Weaning from the monotherapy may be possible later. The inverted curve at the bottom shows the usually silent graft-versus-host (GVH) reaction shown more clearly in Figure 1. Tx, transplantation
The first principle (shown alone in Fig. 3B) consists of administration of no more post-transplantation immunosuppression than the amount needed to prevent irreversible immune-mediated damage. Because of histocompatibility and other confounding parameters in the human population, such ideal immunosuppression in individual patients cannot be accurately predicted. With the second principle (recipient pretreatment), host global immune responsiveness can be reduced by nonmyeloablative conditioning before arrival of donor antigen, thereby bringing the anticipated donor-specific immune response into a more easily controllable and deletable range (Fig. 4). The 2 principles have been combined in a practical regimen using a single large dose of an antilymphoid antibody before transplantation followed by minimalistic post-transplantation tacrolimus monotherapy with the intent of eventual weaning (Fig. 5). Satisfactory results in liver (Fig. 6) and other kinds of organ recipients with reduced overall exposure to immunosuppression have been obtained,72,130–132 with the striking exception of liver recipients whose chronic end-stage hepatic failure was caused by hepatitis C virus (HCV)73,133 (see next section).
Fig. 5.
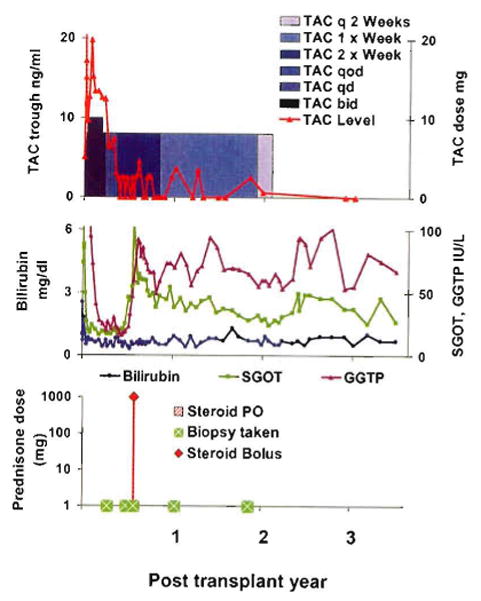
An example of the strategy shown in Fig. 4. A woman with a hepatic hemangioendothelioma who was infused with 5 mg/kg antithymocyte globulin (ATG, thymoglobulin) before liver allograft revascularization. Tacrolimus (TAC) monotherapy was reduced from daily to every other day at 100 days, and to once per week by 10 months. Treatment was stopped at 22 months. She has been immunosuppression-free for 11/2 years. Serum bilirubin and measures of hepatic parenchymal function have been normal throughout. Although enzyme levels have been stable, these have hovered at a high normal range or slightly above since a rejection at 6 months, which was treated with 1 g methylprednisolone. SGOT: serum glutamic-oxaloacetic transaminase, GGTP: gamma-glutamyl transpeptidase.
Fig. 6.
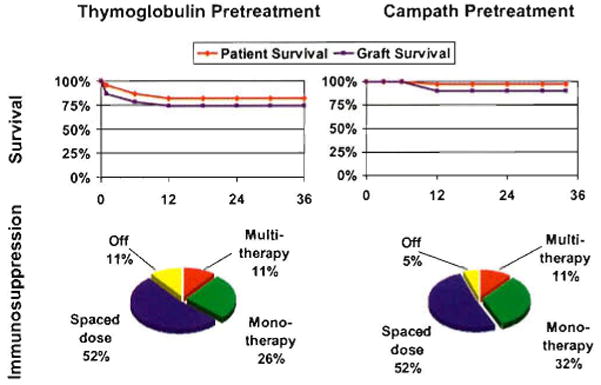
Patient and graft survival (above), and current immunosuppression (below) of hepatitis-free primary liver transplant recipients treated in Pittsburgh with the principles of immunosuppression shown in Fig. 4, and exemplified in Fig. 5. Left: pretreatment was with an infusion of 5 mg/kg rabbit ATG (thymoglobulin) (n = 23). Right: pretreatment was with 30 mg alemtuzumab (Campath®) (n = 38).
The frequency and extent to which drug weaning can be accomplished in hepatitis-free liver recipients with such tolerance-facilitating immunosuppression has yet to be determined. Complete stoppage of treatment has been attempted in only a few of these recent cases. However, the feasibility of liberation from immunosuppressive drugs is evident from observations of our first 210 liver recipients: 184 at the University of Colorado (between 1963 and 1980) and 26 at the University of Pittsburgh (in early 1981). Thirty-five (17%) of the 210 have now reached or passed their post-transplantation silver anniversary (Fig. 7). Thirty-two (15%) are still alive from 24.5 to 35.7 post-transplantation years (mean, 27.1 ± 3.1 SD), and 3 died after 25 years of lung disease (#42), widespread metastases from colon carcinoma (#82), and de novo HCV infection (#93).
Fig. 7.
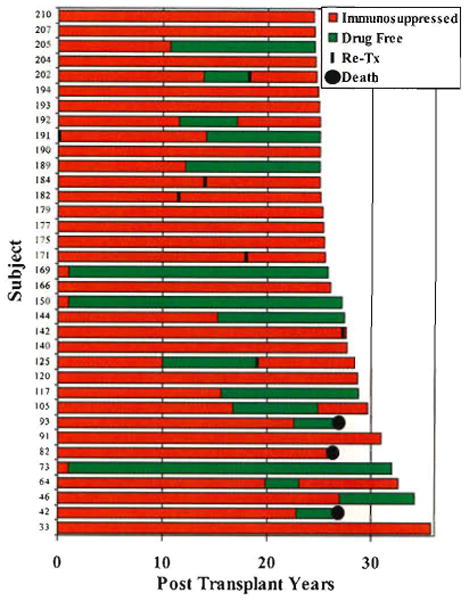
The 35 patients from the first 210 who survived 25 years or longer after liver replacement at the University of Colorado (n = 184) or University of Pittsburgh (n = 26). The times on (red) and off immunosuppression (green) in individual patients are displayed in the horizontal bars. Note that 6 of the 32 still-surviving recipients had retransplantation 11 to 28 years after the primary procedure.
Importantly, 16 of the 35 quarter-century survivors had periods of 3 to 31 years off immunosuppression, as indicated by the green portion of the horizontal bars in Fig. 7. In 5 of these 16 recipients, immunosuppression was resumed, but not because of breakthrough rejection in any case. The reason for treatment reinstitution in 2 patients was liver retransplantation necessitated by intractable biliary tract complications (#202 in Fig. 7) or because of HCV hepatitis (#125). A third recipient whose cyclosporine-based immunosuppression was stopped after 11 years was returned to treatment 6 years later because of cadaver kidney transplantation (#192). The other 2 recipients who were asymptomatic and had rejection-free biopsies were restarted on treatment because of patient and physician anxiety (#64 and #105, in Fig. 7).
The Price of Chronic Immunosuppression
The consequences of a decision to resume immunosuppression without a clear justification may not be evaluable for years or decades. When a detailed account of our 210 first recipients was published in the June 1993 issue of HEPATOLOGY,65 50 (23%) of the original patients had survived for at least a decade (maximum, 23 years). However, 7 had died 1 to 11 years after their 10th anniversary, leaving 43 (20.5%). Rejection had not caused any of the late deaths. Instead, the most common causes of death were hepatitis, other infections, malignant neoplasms, and drug-specific side effects. The same pattern has been apparent in the further shrinkage of survivors between 1993 and 2005 (from 43 to 32) for reasons other than rejection. Chronic immunosuppression clearly has been the principal direct or indirect cause of late mortality.
Organ Engraftment in HCV-Infected Patients
Although the importance of reducing or eliminating long-term immunosuppression is clear, when the tolerogenic strategy depicted in Figs. 4 and 5 was applied in Pittsburgh in HCV-infected liver recipients, the results fulfilled the 1994 prophecy of Zinkernagel and Hengartner129 (see earlier discussion). First, the lymphoid depletion caused viremia. Then, when later attempts were made to wean from tacrolimus monotherapy, the heavily infected liver graft was targeted by recovering CTL, resulting in early and severe recurrent HCV disease. With recognition by Eghtesad and Fung et al.73 of what had happened, the explanation also was apparent for the worldwide epidemic of HCV recurrence that was associated with the various regimens of viremia-inducing heavy multiple drug immunosuppression.134–138 Because strong prophylactic immunosuppression cannot be administered indefinitely without fatal consequences, such treatment eventually must be reduced. The consequent CTL recovery was leading to widespread destruction of infected allograft cells in the same way as with the weaning of our lymphoid-depleted patients.73
In contrast, light but continuous double-drug immunosuppression with tacrolimus and prednisone has allowed the systematic development of a relatively asymptomatic carrier state: a stable equilibrium between HCV and HCV-specific CTL.73 Thus, treatment protocols that minimize disease recurrence in HCV-infected liver allograft recipients must balance the desire to reduce immunosuppression or induce allotolerance with the need to prevent antiviral immunopathology. Curtailment of the epidemiologic implications of producing HCV-carrier recipients, and of the probability of insidious disease recurrence, will depend on containment of the viral load with yet to be developed HCV-specific drugs. One candidate is the protease inhibitor recently described in Hepatology by Reiser et al.139
Tumor Surveillance
By the late 1960s, convincing evidence existed that immunosuppression in organ recipients could result in accidental engraftment of donor malignancies, accelerated growth of tumor metastases, or the development of new malignancies (summarized in140,141 Because the highest risk from these consequences has been in liver recipients, liver transplant centers have become hotbeds of oncology research. Two tumors have been of particular interest because of their etiologic association with intracellular microorganisms: post-transplantation lymphoproliferative disorders (PTLD) with the Epstein-Barr virus, and hepatocellular carcinoma with HBV. The PTLD, most of which are B cell lymphomas, provided the first unequivocal proof of immune surveillance of a human malignancy when they disappeared after withdrawal of T cell–directed immunosuppression.45
The possibility of preventing virus-associated tumors with vaccines was demonstrated by Coggin, Larson, and Hilleman142 with proof of principle studies in primates of the SV40 virus, and extended to humans 20 years later with the recombinant HBV vaccine trials. The HBV- associated hepatoma scourge was virtually eliminated in immunized Asian populations.143 How the anti–HBV-induced antibodies produced by the vaccine interrelate with T cell immunity has been thoroughly studied with LCMV144 and explained by Klenerman.145 The research and therapeutic opportunities opened by these and other observations is too vast to dwell on here beyond emphasizing that the mechanisms of immune reactivity and non-reactivity to tumors are the same as those of transplantation.
Autoimmunity
The chimerism-dependent mechanisms of non-reactivity to allografts, all of which depend on mobile leukocytes, can be identified in a continuum of classical tolerance models that began with the observation by Owen of mixed blood cell chimerism in Freemartin cattle82 and ended in 1992 with the discovery of microchimerism in liver and other kinds of organ recipients (Fig. 8). Of historical interest, Paul Ehrlich recognized more than a century ago that a patient’s tissues could be destroyed by an immune system run amuck. Ehrlich’s term of “horror autotoxicus” is today’s autoimmune disease. He postulated that there must be mechanisms to prevent this by “…a regulatory contrivance as yet undescribed.”146 In the context of immunology described here, the contrivance that Ehrlich envisioned consisted of the mechanisms that have made transplantation feasible.
Fig. 8.
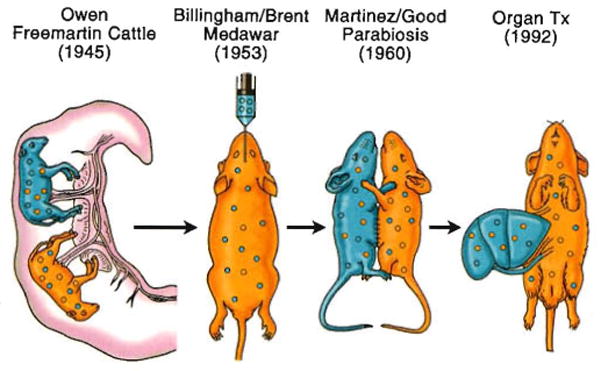
The relation of organ-induced engraftment (right) to classical models of spontaneous donor leukocyte chimerism-associated tolerance.
A Need for Closure
A coherent profile of immune function and governance has emerged from the studies of transplantation and infection summarized here. However, mechanisms of nonresponsiveness other than the essential ones of immune ignorance115,147–150 and clonal deletion (listed in Table 3) have generated a large body of historical151 and recent publications.152–155 In turn, these model-specific and poorly understood alternative mechanisms have become elements of recurring immunological dogmas and theories. Is assigning them essential roles in transplantation tolerance justifiable? Evidence of their existence derives from phenomena observed in experimental models in which the immune system is drastically perturbed (e.g., under conditions of lymphopenia) or the antigenic barriers to transplantation are significantly weakened (e.g., transplantation across minor histocompatibility mismatches). To complete the picture, it will be necessary to definitively determine the conditions for the development of these phenomena and to accurately assess their functional significance.
Conclusion
The article entitled “The Evolution of Liver Transplantation” published in 1982 in Hepatology concluded with the statement that “… what was inconceivable yesterday, and barely achievable today, often becomes routine tomorrow.”38 Liver transplantation proved to be a prime example. The accomplishment was more than the addition of the crucial centerpiece for the treatment of otherwise lethal end-stage hepatic disease, however. Liver transplantation was from the beginning, and will continue to be, an instrument of scientific discovery in multiple fields, and above all in immunology.
Acknowledgments
Supported by National Institutes of Health Grant RO1DK64207, and an unrestricted gift from Mr. Robert Eberly.
Abbreviations
- GVHD
graft-versus-host disease
- LCMV
lymphocytic choriomeningitis virus
- CTL
cytolytic T lymphocyte
- APC
antigen-presenting cells
- HBV
hepatitis B virus
- HCV
hepatitis C virus
- PTLD
post-transplantation lymphoproliferative disorders
Footnotes
Potential conflict of interest: Nothing to report.
References
- 1.Welch CS. A note on transplantation of the whole liver in dogs. Transplant Bull. 1955;2:54–55. [Google Scholar]
- 2.Cannon JA. Brief report. Transplant Bull. 1956;3:7. [Google Scholar]
- 3.Starzl TE, Kaupp HA, Jr, Brock DR, Lazarus RE, Johnson RV. Reconstructive problems in canine liver homotransplantation with special reference to the postoperative role of hepatic venous flow. Surg Gynecol Obstet. 1960;111:733–743. [PMC free article] [PubMed] [Google Scholar]
- 4.Moore FD, Wheeler HB, Demissianos HV, Smith LL, Balankura O, Abel K, et al. Experimental whole organ transplantation of the liver and of the spleen. Ann Surg. 1960;152:374–387. [PMC free article] [PubMed] [Google Scholar]
- 5.Starzl TE, Kaupp HA., Jr Mass homotransplantation of abdominal organs in dogs. Surg Forum. 1960;11:28–30. [PMC free article] [PubMed] [Google Scholar]
- 6.Starzl TE, Marchioro TL, Waddell WR. The reversal of rejection in human renal homografts with subsequent development of homograft tolerance. Surg Gynecol Obstet. 1963;117:385–395. [PMC free article] [PubMed] [Google Scholar]
- 7.Marchioro TL, Huntley RT, Waddell WR, Starzl TE. Extracorporeal perfusion for obtaining postmortem homografts. Surgery. 1963;54:900–911. [PMC free article] [PubMed] [Google Scholar]
- 8.Starzl TE, Marchioro TL, Von Kaulla KN, Hermann G, Brittain RS, Waddell WR. Homotransplantation of the liver in humans. Surg Gynecol Obstet. 1963;117:659–676. [PMC free article] [PubMed] [Google Scholar]
- 9.Starzl TE, Marchioro TL, Rowlands DT, Jr, Kirkpatrick CH, Wilson WEC, Rifkind D, et al. Immunosuppression after experimental and clinical homotransplantation of the liver. Ann Surg. 1964;160:411–439. doi: 10.1097/00000658-196409000-00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Marchioro TL, Porter KA, Dickinson TC, Faris TD, Starzl TE. Physiologic requirements for auxiliary liver homotransplantation. Surg Gynecol Obstet. 1965;121:17–31. [PMC free article] [PubMed] [Google Scholar]
- 11.Starzl TE, Marchioro TL, Porter KA, Taylor PD, Faris TD, Herrmann TJ, et al. Factors determining short- and long-term survival after orthotopic liver homotransplantation in the dog. Surgery. 1965;58:131–155. [PMC free article] [PubMed] [Google Scholar]
- 12.Starzl TE. Orthotopic heterotransplantation. In: Starzl TE, editor. Experience in Hepatic Transplantation. Philadelphia: WB Saunders; 1969. pp. 408–421. [Google Scholar]
- 13.Starzl TE, Marchioro TL, Porter KA, Iwasaki Y, Cerilli GJ. The use of heterologous antilymphoid agents in canine renal and liver homotransplantation and in human renal homotransplantation. Surg Gynecol Obstet. 1967;124:301–318. [PMC free article] [PubMed] [Google Scholar]
- 14.Terasaki PI, Vredevoe DL, Mickey MR, Porter KA, Marchioro TL, Faris TD, et al. Serotyping for homotransplantation. VI. Selection of kidney donors for thirty-two recipients. Ann NY Acad Sci. 1966;129:500–520. doi: 10.1111/j.1749-6632.1966.tb12873.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Starzl TE, Porter KA, Andres G, Halgrimson CG, Hurwitz R, Giles G, et al. Long-term survival after renal transplantation in humans: With special reference to histocompatibility matching, thymectomy, homograft glomerulonephritis, heterologous ALG, and recipient malignancy. Ann Surg. 1970;172:437–472. doi: 10.1097/00000658-197009000-00011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Starzl TE, Groth CG, Brettschneider L, Penn I, Fulginiti VA, Moon JB, et al. Orthotopic homotransplantation of the human liver. Ann Surg. 1968;168:392–415. doi: 10.1097/00000658-196809000-00009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Definition of irreversible coma: report of the ad hoc committee of the Harvard Medical School to examine the definition of brain death. JAMA. 1968;205:337. [PubMed] [Google Scholar]
- 18.Cordier G, Garnier H, Clot JP, Camplez P, Gorin JP, Clot Ph, et al. La greffe de foie orthotopique chez le porc. Mem Acad Chir (Paris) 1966;92:799–807. [PubMed] [Google Scholar]
- 19.Peacock JH, Terblanche J. Orthotopic homotransplantation of the liver in the pig. In: Read AE, editor. The Liver. London: Butterworth; 1967. p. 333. [Google Scholar]
- 20.Calne RY, White HJO, Yoffa DE, Binns RM, Maginn RR, Herbertson RM, et al. Prolonged survival of liver transplants in the pig. Br Med J. 1967;4:645–648. doi: 10.1136/bmj.4.5580.645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Starzl TE. Experience in Hepatic Transplantation. Philadelphia: WB Saunders; 1969. pp. 1–533. (with the assistance of Putnam CW) [Google Scholar]
- 22.DuBois RS, Giles G, Rodgerson DO, Lilly J, Martineau G, Halgrimson CG, et al. Orthotopic liver transplantation for Wilson’s disease. Lancet. 1071;1:505–508. doi: 10.1016/s0140-6736(71)91121-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Starzl TE, Chase HP, Putnam CW, Porter KA. Portacaval shunt in hyperlipoproteinemia. Lancet. 1973;2:940–944. doi: 10.1016/s0140-6736(73)92599-3. [DOI] [PubMed] [Google Scholar]
- 24.Starzl TE, Lee IY, Porter KA, Putnam CW. The influence of portal blood upon lipid metabolism in normal and diabetic dogs and baboons. Surg Gynecol Obstet. 1975;140:381–396. [PMC free article] [PubMed] [Google Scholar]
- 25.Starzl TE, Chase HP, Ahrens EH, Jr, McNamara DJ, Bilheimer DW, Schaefer EJ, et al. Portacaval shunt in patients with familial hypercholesterolemia. Ann Surg. 1983;198:273–283. doi: 10.1097/00000658-198309000-00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Starzl TE. The Puzzle People. Pittsburgh: University of Pittsburgh Press; 1992. The little drummer girls; pp. 318–333. [Google Scholar]
- 27.Brown MS, Golstein JL. Lipoprotein receptors in the liver: control signals for plasma cholesterol traffic. J Clin Invest. 1983;72:743–747. doi: 10.1172/JCI111044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Starzl TE, Ishikawa M, Putnam CW, Porter KA, Picache R, Husberg BS, et al. Progress in and deterrents to orthotopic liver transplantation, with special reference to survival, resistance to hyperacute rejection, and biliary duct reconstruction. Transplant Proc. 1974;6:129–139. [PMC free article] [PubMed] [Google Scholar]
- 29.Starzl TE, Francavilla A, Halgrimson CG, Francavilla FR, Porter KA, Brown TH, et al. The origin, hormonal nature, and action of hepatotrophic substances in portal venous blood. Surg Gynecol Obstet. 1973;137:179–199. [PMC free article] [PubMed] [Google Scholar]
- 30.Starzl TE, Porter KA, Putnam CW. Intraportal insulin protects from the liver injury of portacaval shunt in dogs. Lancet. 1975;2:1241–1246. doi: 10.1016/s0140-6736(75)92076-0. [DOI] [PubMed] [Google Scholar]
- 31.Starzl TE, Porter KA, Putnam CW, Schroter GPJ, Halgrimson CG, Weil R, III, et al. Orthotopic liver transplantation in ninety-three patients. Surg Gynecol Obstet. 1976;142:487–505. [PMC free article] [PubMed] [Google Scholar]
- 32.Wall WJ, Calne RY, Herbertson BM, Baker PG, Smith DP, Underwood J, et al. Simple hyptothermic preservation for transporting human livers long distance for homotransplantation. Transplantation. 1977;23:210–216. doi: 10.1097/00007890-197703000-00002. [DOI] [PubMed] [Google Scholar]
- 33.Benichou J, Halgrimson CG, Weil R, III, Koep LJ, Starzl TE. Canine and human liver preservation for 6 to 18 hour by cold infusion. Transplantation. 1977;24:407–411. doi: 10.1097/00007890-197712000-00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Starzl TE, Halgrimson CG, Koep LJ, Weil R, III, Taylor PD. Vascular homografts from cadaveric organ donors. Surg Gynecol Obstet. 1979;149:737. [PMC free article] [PubMed] [Google Scholar]
- 35.Calne RY, Rolles K, White DJ, Thiru S, Evans DB, McMaster P, et al. Cyclosporin A initially as the only immunosuppressant in 34 recipients of cadaveric organs: 32 kidneys, 2 pancreases, and 2 livers. Lancet. 1979;2:1033–1036. doi: 10.1016/s0140-6736(79)92440-1. [DOI] [PubMed] [Google Scholar]
- 36.Starzl TE, Weil R, III, Iwatsuki S, Klintmalm G, Schroter GPJ, Koep LJ, et al. The use of cyclosporin A and prednisone in cadaver kidney transplantation. Surg Gynecol Obstet. 1980;151:17–26. [PMC free article] [PubMed] [Google Scholar]
- 37.Starzl TE, Klintmalm GBG, Porter KA, Iwatsuki S, Schroter GPJ. Liver transplantation with use of cyclosporin A and prednisone. N Engl J Med. 1981;305:266–269. doi: 10.1056/NEJM198107303050507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Starzl TE, Iwatsuki S, Van Thiel DH, Gartner JC, Zitelli BJ, Malatack JJ, et al. Evolution of liver transplantation. Hepatology. 1982;2:614–636. doi: 10.1002/hep.1840020516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Denmark SW, Shaw BW, Jr, Starzl TE, Griffith BP. Veno-venous bypass without systemic anticoagulation in canine and human liver transplantation. Surg Forum. 1983;34:380–382. [PMC free article] [PubMed] [Google Scholar]
- 40.Shaw BW, Jr, Martin DJ, Marquez JM, Kang YG, Bugbee AC, Iwatsuki S, et al. Venous bypass in clinical liver transplantation. Ann Surg. 1984;200:524–534. doi: 10.1097/00000658-198410000-00013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Griffith BP, Shaw BW, Jr, Hardesty RL, Iwatsuki S, Bahnson HT, Starzl TE. Veno-venous bypass without systemic anticoagulation for transplantation of the human liver. Surg Gynecol Obstet. 1985;160:270–272. [PMC free article] [PubMed] [Google Scholar]
- 42.National Institutes of Health Consensus Development Conference on Liver Transplantation. Sponsored by the National Institute of Arthritis, Diabetes, and Digestive and Kidney Diseases and the National Institutes of Health Office of Medical Applications of Research. Hepatology. 1984;4(1 Suppl):1S–110S. [PubMed] [Google Scholar]
- 43.Starzl TE, Hakala TR, Shaw BW, Jr, Hardesty RL, Rosenthal TJ, Griffith BP, et al. A flexible procedure for multiple cadaveric organ procurement. Surg Gynecol Obstet. 1984;158:223–230. [PMC free article] [PubMed] [Google Scholar]
- 44.Starzl TE, Miller C, Broznick B, Makowka L. An improved technique for multiple organ harvesting. Surg Gynecol Obstet. 1987;165:343–348. [PMC free article] [PubMed] [Google Scholar]
- 45.Starzl TE, Nalesnik MA, Porter KA, Ho M, Iwatsuki S, Griffith BP, et al. Reversibility of lymphomas and lymphoproliferative lesions developing under cyclosporin-steroid therapy. Lancet. 1984;1:583–587. doi: 10.1016/s0140-6736(84)90994-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bismuth H, Houssin D. Reduced–sized orthotopic liver graft in hepatic transplantation in children. Surgery. 1984;95:367–370. [PubMed] [Google Scholar]
- 47.Broelsch CE, Neuhaus P, Burdelski M, Berngau U, Pichlmayr R. Orthotope transplantation von Lebegmenten bei mit Gallengangsatresien. (Orthotopic transplantation of hepatic segments in infants with biliary atresia) In: Kolsowski L, editor. Chirurgisches Forum 1984, F Experim U Klimische Forschung Hrsga. Berlin: Springer–Verlag; 1984. pp. 105–109. [Google Scholar]
- 48.Starzl TE, Bilheimer DW, Bahnson HT, Shaw BW, Jr, Hardesty RL, Griffith BP, et al. Heart-liver transplantation in a patient with familial hypercholesterolemia. Lancet. 1984;1:1382–1383. doi: 10.1016/s0140-6736(84)91876-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Bilheimer DW, Goldstein JL, Grundy SC, Starzl TE, Brown MS. Liver transplantation provides low-density-lipoprotein receptors and lowers plasma cholesterol in a child with homozygous familial hypercholesterolemis. N Engl J Med. 1984;311:1658–1664. doi: 10.1056/NEJM198412273112603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Starzl TE, Rowe MI, Todo S, Jaffe R, Tzakis A, Hoffman AL, et al. Transplantation of multiple abdominal viscera. JAMA. 1989;261:1449– 1457. [PMC free article] [PubMed] [Google Scholar]
- 51.Grant D, Wall W, Mimeault R, Zhong R, Ghent C, Garcia B, et al. Successful small-bowel/liver transplantation. Lancet. 1990;335:181–184. doi: 10.1016/0140-6736(90)90275-a. [DOI] [PubMed] [Google Scholar]
- 52.Jamieson NV, Sundberg R, Lindell S, Laravuso M, Kalayoglu M, Southard JH, et al. Successful 24- to 30-hour preservation of the canine liver: a preliminary report. Transplant Proc. 1988;20(Suppl l):945–947. [Google Scholar]
- 53.Kalayoglu M, Sollinger HW, Stratta RJ, D’Alessandro AM, Hoffmann RM, Pirsch JD, et al. Extended preservation of the liver for clinical transplantation. Lancet. 1988;1:617–619. doi: 10.1016/s0140-6736(88)91416-x. [DOI] [PubMed] [Google Scholar]
- 54.Todo S, Nery J, Yanaga K, Podesta L, Gordon RD, Starzl TE. Extended preservation of human liver grafts with UW solution. JAMA. 1989;26l:711–714. [PMC free article] [PubMed] [Google Scholar]
- 55.Makowka L, Gordon RD, Todo S, Ohkohchi N, Marsh JW, Tzakis AG, et al. Analysis of donor criteria for the prediction of outcome in clinical liver transplantation. Transplant Proc. 1987;19:2378–2382. [PMC free article] [PubMed] [Google Scholar]
- 56.Starzl TE, Hakala T, Tzakis A, Gordon R, Stieber A, Makowka L, et al. A multifactorial system for equitable selection of cadaveric kidney recipients. JAMA. 1987;257:3073–3075. [PMC free article] [PubMed] [Google Scholar]
- 57.Starzl TE, Gordon RD, Tzakis A, Staschak S, Fioravanti V, Broznick B, et al. Equitable allocation of extrarenal organs: With special reference to the liver. Transplant Proc. 1988;20:131–138. [PMC free article] [PubMed] [Google Scholar]
- 58.Tzakis A, Todo S, Starzl TE. Orthotopic liver transplantation with preservation of the inferior vena cava. Ann Surg. 1989;210:649–652. doi: 10.1097/00000658-198911000-00013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Starzl TE, Todo S, Fung J, Demetris AJ, Venkataramanan R, Jain A. FK 506 for human liver, kidney and pancreas transplantation. Lancet. 1989;2:1000–1004. doi: 10.1016/s0140-6736(89)91014-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Todo S, Fung JJ, Starzl TE, Tzakis A, Demetris AJ, Kormos R, et al. Liver, kidney, and thoracic organ transplantation under FK 506. Ann Surg. 1990;212:295–305. doi: 10.1097/00000658-199009000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Pichlmayr R, Ringe B, Gubernatis G, Hauss J, Bunzendahl H. Transplantation einer spenderleber auf Zwis Empfanger (Split liver transplantation) Eine neue methode in der weitzentwicklung der lebesegment transplantation. Langenbecks Arch Chir. 1988;373:127–130. [PubMed] [Google Scholar]
- 62.Strong RW, Lynch SV, Ong TH, Matsunami H, Koido Y, Balderson GA. Successful liver transplantation from a living donor to her son. N Engl J Med. 1990;322:1505–1507. doi: 10.1056/NEJM199005243222106. [DOI] [PubMed] [Google Scholar]
- 63.Broelsch CE, Emond JC, Whitington PF, Thistlethwaite JR, Baker AL, Lichtor JL. Application of reduced-size liver transplants as split grafts, auxiliary orthotopic grafts, and living related segmental transplants. Ann Surg. 1990;212:368–375. doi: 10.1097/00000658-199009000-00015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Starzl TE, Demetris AJ, Murase N, Ildstad S, Ricordi C, Trucco M. Cell migration, chimerism, and graft acceptance. Lancet. 1992;339:1579–1582. doi: 10.1016/0140-6736(92)91840-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Starzl TE, Demetris AJ, Trucco M, Murase N, Ricordi C, Ildstad S, et al. Cell migration and chimerism after whole-organ transplantation: the basis of graft acceptance. Hepatology. 1993;17:1127–1152. [PMC free article] [PubMed] [Google Scholar]
- 66.Starzl TE, Zinkernagel R. Antigen localization and migration in immunity and tolerance. N Engl J Med. 1998;339:1905–1913. doi: 10.1056/NEJM199812243392607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Yamaoka Y, Washida M, Honda K, Tanaka K, Mori K, Shimahara Y, et al. Liver transplantation using a right lobe graft from a living related donor. Transplantation. 1994;57:1127–1130. [PubMed] [Google Scholar]
- 68.Wachs M, Bak T, Karrer F, Everson GT, Shrestha R, Trouillot TE, et al. Adult living donor liver transplantation using a right hepatic lobe. Transplantation. 1998;66:1313–1316. doi: 10.1097/00007890-199811270-00008. [DOI] [PubMed] [Google Scholar]
- 69.Marcos A, Fisher RA, Ham JM, Shiffman ML, Sanyal AJ, Luketic VA, et al. Right lobe living donor liver transplantation. Transplantation. 1999;68:798–803. doi: 10.1097/00007890-199909270-00012. [DOI] [PubMed] [Google Scholar]
- 70.Starzl TE, Zinkernagel R. Transplantation tolerance from a historical perspective. Nature Reviews: Immunology. 2001;1:233–239. doi: 10.1038/35105088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Phelps CJ, Koike C, Vaught TD, Boone J, Wells KD, Chen SH, et al. Production of α1,3-galactosyltransferase-deficient pigs. Science. 2003;299:411–414. doi: 10.1126/science.1078942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Starzl TE, Murase N, Abu–Elmagd K, Gray EA, Shapiro R, Eghtesad B, et al. Tolerogenic immunosuppression for organ transplantation. Lancet. 2003;361:1502–1510. doi: 10.1016/s0140-6736(03)13175-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Eghtesad B, Fung JJ, Demetris AJ, Murase N, Ness R, Bass DC, et al. Mechanism-based principles of immunosuppression for liver transplantation in HCV-infected patients. Liver Transpl. 2005;11:1343–1352. doi: 10.1002/lt.20536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Anonymous. National Institutes of Health, NIH Pub. No. 04-5491. U.S. Department of Health and Human Services; 2004. Dec, Action Plan for Liver Disease Research; pp. 1–93. [Google Scholar]
- 75.Francavilla A, Hagiya M, Porter KA, Polimeno L, Ihara I, Starzl TE. Augmenter of liver regeneration (ALR): its place in the universe of hepatic growth factors. Hepatology. 1994;20:747–757. [PubMed] [Google Scholar]
- 76.Starzl TE, Porter KA, Kashiwagi N, Lee IY, Russell WJI, Putnam CW. The effect of diabetes mellitus on portal blood hepatotrophic factors in dogs. Surg Gynecol Obstet. 1975;140:549–562. [PMC free article] [PubMed] [Google Scholar]
- 77.Starzl TE, Porter KA, Kashiwagi N, Putnam CW. Portal hepatotrophic factors, diabetes mellitus and acute liver atrophy, hypertrophy and regeneration. Surg Gynecol Obstet. 1975;141:843–858. [PMC free article] [PubMed] [Google Scholar]
- 78.Starzl TE, Demetris AJ, Van Thiel DH. Medical progress: liver transplantation. N Engl J Med [Part I] 1989;321:1014–1022. doi: 10.1056/NEJM198910123211505. [DOI] [PMC free article] [PubMed] [Google Scholar]; [Part II] 1989;321:1092–1099. [Google Scholar]
- 79.Wolstenholme GEW, O’Connor M, editors. Ethics in Medical Progress: With Special Reference to Transplantation. Boston: Little, Brown; 1966. pp. 1–249. The Symposium held in London at the CIBA House was attended by 25 invited physicians and surgeons (including TES, Keith Reemtsma, and Joseph E. Murray), ethicists, religious leaders, medical philosophers, judges, and lawyers. [Google Scholar]
- 80.Burger WF. The law and medical advances. Ann Intern Med. 1967;67(Suppl 7):15–18. doi: 10.7326/0003-4819-67-3-15. Symposium on Medical Ethics of the American Association of Physicians with presentations by: Peter Medawar, Joshua Lederburg, Thomas Starzl, and Warren Burger (subsequently Chief Justice of the United States Supreme Court) [DOI] [PubMed] [Google Scholar]
- 81.Medawar PB. The behavior and fate of skin autografts and skin homografts in rabbits. J Anat. 1944;78:176–199. [PMC free article] [PubMed] [Google Scholar]
- 82.Owen RD. Immunogenetic consequences of vascular anastomoses beween bovine twins. Science. 1945;102:400–401. doi: 10.1126/science.102.2651.400. [DOI] [PubMed] [Google Scholar]
- 83.Billingham RE, Brent L, Medawar PB. “Actively acquired tolerance” of foreign cells. Nature. 1953;172:603–606. doi: 10.1038/172603a0. [DOI] [PubMed] [Google Scholar]
- 84.Main JM, Prehn RT. Successful skin homografts after the administration of high dosage × radiation and homologous bone marrow. J Natl Cancer Inst. 1955;15:1023–1029. [PubMed] [Google Scholar]
- 85.Murray JE, Merrill JP, Dammin GJ, Dealy JB, Jr, Walter CW, Brooke MS, et al. Study of transplantation immunity after total body irradiation: clinical and experimental investigation. Surgery. 1960;48:272–284. [PubMed] [Google Scholar]
- 86.Hamburger J, Vaysse J, Crosnier J, Tubiana M, Lalanne CM, Antoine B, et al. Transplantation of a kidney between nonmonozygotic twins after irradiation of the receiver. Good function at the fourth month. Presse Med. 1959;67:1771–1775. [Google Scholar]
- 87.Hamburger J, Vaysse J, Crosnier J, Auvert J, Lalanne CL, Hopper J., Jr Renal homotransplantation in man after radiation of the recipient. Am J Med. 1962;32:854–871. doi: 10.1016/0002-9343(62)90032-3. [DOI] [PubMed] [Google Scholar]
- 88.Kuss R, Legrain M, Mathe G, Nedey R, Camey M. Homologous human kidney transplantation: experience with six patients. Postgrad Med J. 1962;38:528–531. doi: 10.1136/pgmj.38.443.528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Calne RY. Inhibition of the rejection of renal homografts in dogs by purine analogues. Transplant Bull. 1961;28:445–461. [PubMed] [Google Scholar]
- 90.Murray JE, Merrill JP, Harrison JH, Wilson RE, Dammin GJ. Prolonged survival of human–kidney homografts by immunosuppressive drug therapy. N Engl J Med. 1963;268:1315–1323. doi: 10.1056/NEJM196306132682401. [DOI] [PubMed] [Google Scholar]
- 91.Hopewell J, Calne RY, Beswick I. Three clinical cases of renal transplantation. Br Med J. 1964;I:411–413. doi: 10.1136/bmj.1.5380.396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Slavin S, Strober S, Fuks Z, Kaplan HS. Induction of specific tissue transplantation tolerance using fractionated total lymphoid irradiation in adult mice: long–term survival of allogeneic bone marrow and skin grafts. J Exp Med. 1977;146:34–48. doi: 10.1084/jem.146.1.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Ildstad ST, Sachs DH. Reconstitution with syngeneic plus allogeneic or xenogeneic bone marrow leads to specific acceptance of allografts or xenografts. Nature. 1984;307:168–170. doi: 10.1038/307168a0. [DOI] [PubMed] [Google Scholar]
- 94.Sharabi Y, Sachs DH. Mixed chimerism and permanent specific transplantation tolerance induced by a non-lethal preparative regimen. J Exp Med. 1989;169:493–502. doi: 10.1084/jem.169.2.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Colson YL, Li H, Boggs SS, Patrene KD, Johnson PC, Ildstad ST. Durable mixed allogeneic chimerism and tolerance by a non-lethal radiation-based cytoreductive approach. J Immunol. 1996;157:2820–2829. [PubMed] [Google Scholar]
- 96.Storb R, Yu C, Wagner JL, Deeg HJ, Nash RA, Kiem HP, et al. Stable mixed hematopoietic chimerism in DLA-identical littermate dogs given sublethal total body irradiation before and pharmacological immunosuppression after marrow transplantation. Blood. 1997;89:3048–3054. [PubMed] [Google Scholar]
- 97.Thomas JM, Eckhoff DE, Contreras JL, Lobashevsky AL, Hubbard WJ, Moore JK, et al. Durable donor-specific T and B cell tolerance in rhesus macaques induced with peritransplantation anti–CD3 immunotoxin and deoxyspergualin. Transplantation. 2000;69:2497–2503. doi: 10.1097/00007890-200006270-00007. [DOI] [PubMed] [Google Scholar]
- 98.Wekerle T, Sykes M. Mixed chimerism as an approach for the induction of transplantation tolerance. Transplantation. 1999;68:459–467. doi: 10.1097/00007890-199908270-00001. [DOI] [PubMed] [Google Scholar]
- 99.Cosimi AB, Sachs DH. Mixed chimerism and transplantation tolerance. Transplantation. 2004;77:943–946. doi: 10.1097/01.tp.0000117779.23431.3f. [DOI] [PubMed] [Google Scholar]
- 100.Starzl TE, Demetris AJ, Trucco M, Ramos H, Zeevi A, Rudert WA, et al. Systemic chimerism in human female recipients of male livers. Lancet. 1992;340:876–877. doi: 10.1016/0140-6736(92)93286-v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Starzl TE, Demetris AJ, Trucco M, Ricordi C, Ildstad S, Terasaki PI, et al. Chimerism after liver transplantation for type IV glycogen storage disease and Type I Gaucher’s disease. N Engl J Med. 1993;328:745–749. doi: 10.1056/NEJM199303183281101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Starzl TE, Demetris AJ, Trucco M, Zeevi A, Ramos H, Terasaki P, et al. Chimerism and donor-specific nonreactivity 27 to 29 years after kidney allotransplantation. Transplantation. 1993;55:1272–1277. doi: 10.1097/00007890-199306000-00012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Starzl TE, Demetris AJ. Transplantation milestones: viewed with one- and two-way paradigms of tolerance. JAMA. 1995;273:876–879. doi: 10.1001/jama.273.11.876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Przepiorka D, Thomas ED, Durham DM, Fisher L. Use of a probe to repeat sequence of the Y chromosome for detection of host cells in peripheral blood of bone marrow transplant recipients. Am J Clin Pathol. 1991;95:201–206. doi: 10.1093/ajcp/95.2.201. [DOI] [PubMed] [Google Scholar]
- 105.Schlitt HJ, Hundrieser J, Ringe B, Pichlmayr R. Donor-type microchimerism associated with graft rejection eight years after liver transplantation. N Engl J Med. 1994;330:646–647. doi: 10.1056/NEJM199403033300919. [DOI] [PubMed] [Google Scholar]
- 106.Burlingham WJ, Grailer AP, Fechner JH, Kusaka S, Trucco M, Kocova M, et al. Microchimerism linked to cytotoxic T lymphocyte functional unresponsiveness (clonal anergy) in a tolerant renal transplant recipient. Transplantation. 1995;59:1147–1155. [PubMed] [Google Scholar]
- 107.Wood K, Sachs DH. Chimerism and transplantation tolerance. cause and effect. Immunol Today 17. 1996;12:584–588. doi: 10.1016/s0167-5699(96)10069-4. [DOI] [PubMed] [Google Scholar]
- 108.Bushell A, Pearson TC, Morris PJ, Wood KJ. Donor-recipient microchimerism and tolerance induction. Transplantation. 1996;61:170–172. [Google Scholar]
- 109.Miller J, Mathew JM, Esquenazi V. Toward tolerance to human organ transplants. a few additional corollaries and questions. Transplantation. 2004;77:940–942. doi: 10.1097/01.tp.0000117781.50131.55. [DOI] [PubMed] [Google Scholar]
- 110.Ko S, Deiwick A, Jager MD, Dinkel A, Rohde F, Fischer R, et al. The functional relevance of passenger leukocytes and microchimerism for heart allograft acceptance in the rat. Nature Med. 1999;5:1292–1297. doi: 10.1038/15248. [DOI] [PubMed] [Google Scholar]
- 111.Zinkernagel RM. Restriction by H-2 gene complex of transfer of cell-mediated immunity to Listeria monocytogenes. Nature. 1974;251:230–233. doi: 10.1038/251230a0. [DOI] [PubMed] [Google Scholar]
- 112.Doherty PC, Zinkernagel RM. A biological role for the major histocompatibility antigens. Lancet. 1975:1406–1409. doi: 10.1016/s0140-6736(75)92610-0. [DOI] [PubMed] [Google Scholar]
- 113.Zinkernagel RM, Doherty PC. The discovery of MHC restriction. Immunol Today. 1997;18:14–17. doi: 10.1016/s0167-5699(97)80008-4. [DOI] [PubMed] [Google Scholar]
- 114.Moskophidis D, Lechner F, Pircher H, Zinkernagel RM. Virus persistence in acutely infected immunocompetent mice by exhaustion of antiviral cytotoxic effector T cells. Nature. 1993;362:758–761. doi: 10.1038/362758a0. [DOI] [PubMed] [Google Scholar]
- 115.Zinkernagel RM, Moskophidis D, Kundig T, Oehen S, Pircher H, Hengartner H. Effector T-cell induction and T-cell memory versus peripheral deletion of T cells. Immunol Rev. 1993;131:199–223. doi: 10.1111/j.1600-065x.1993.tb01517.x. [DOI] [PubMed] [Google Scholar]
- 116.Starzl TE. Experience in Hepatic-Transplantation. Philadelphia: W.B. Saunders; 1969. Rejection in unmodified animals; p. 184. [Google Scholar]
- 117.Calne RY, Sells RA, Pena JR, Davis DR, Millard PR, Herbertson BM. Induction of immunological tolerance by porcine liver allografts. Nature. 1969;223:472–474. doi: 10.1038/223472a0. [DOI] [PubMed] [Google Scholar]
- 118.Kamada N, Brons G, Davies HffS. Fully allogeneic liver grafting in rats induces a state of systemic nonreactivity to donor transplantation antigens. Transplantation. 1980;29:429–431. doi: 10.1097/00007890-198005000-00021. [DOI] [PubMed] [Google Scholar]
- 119.Zimmerman FA, Davies HS, Knoll PP, Gocke JM, Schmidt T. Orthotopic liver allografts in the rat. Transplantation. 1984;37:406–410. doi: 10.1097/00007890-198404000-00019. [DOI] [PubMed] [Google Scholar]
- 120.Murase N, Starzl TE, Tanabe M, Fujisaki S, Miyazawa H, Ye Q, et al. Variable chimerism, graft versus host disease, and tolerance after different kinds of cell and whole organ transplantation from Lewis to Brown-Norway rats. Transplantation. 1995;60:158–171. doi: 10.1097/00007890-199507000-00009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Qian S, Demetris AJ, Murase N, Rao AS, Fung JJ, Starzl TE. Murine liver allograft transplantation: tolerance and donor cell chimerism. Hepatology. 1994;19:916–924. doi: 10.1002/hep.1840190418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Corry RJ, Winn HJ, Russell PS. Primary vascularized allografts of hearts in mice: the role of H-2D, H-2K, and non-H-2 antigens in rejection. Transplantation. 1973;16:343–350. doi: 10.1097/00007890-197310000-00010. [DOI] [PubMed] [Google Scholar]
- 123.Russell PS, Chase CM, Colvin RB, Plate JMD. Kidney transplants in mice: an alalysis of the immune status of mice bearing long-term H-2 incompatible transplants. J Exp Med. 1978;147:1449–1468. doi: 10.1084/jem.147.5.1449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Wang C, Sun J, Sheil AG, McCaughan GW, Bishop GA. A short course of methylprednisolone immunosuppression inhibits both rejection and spontaneous acceptance of rat liver allografts. Transplantation. 2001;72:44–51. doi: 10.1097/00007890-200107150-00011. [DOI] [PubMed] [Google Scholar]
- 125.Smiley ST, Csizmadia V, Gao W, Turka LA, Hancock WW. Differential effects of cyclosporine A, methylprednisolone, mycophenolate, and rapamycin and CD 154 induction and requirement for NFKB: implications for tolerance induction. Transplantation. 2000;70:415–419. doi: 10.1097/00007890-200008150-00005. [DOI] [PubMed] [Google Scholar]
- 126.Larsen CP, Elwood ET, Alexander DZ, Ritchie Sc, Hendrix R, Tucker-Burden C, et al. Long-term acceptance of skin and cardiac allografts after blocking CD40 and CD28 pathways. Nature. 1996;381:434–438. doi: 10.1038/381434a0. [DOI] [PubMed] [Google Scholar]
- 127.Myburgh JA. Total lymphoid irradiation in transplantation: experimental background and results in 70 patients. In: Messmer K, Stein M, editors. Pathways in Applied Immunology. Berlin: Springer-Verlag; 1991. pp. 87–93. [Google Scholar]
- 128.Kirk AD, Burkly LC, Batty DS, Baumgartner RE, Berning JD, Buchanan K, et al. Treatment with humanized monoclonal antibody against CD154 prevents acute renal allograft rejection in nonhuman primates. Nat Med. 1999;5:686–693. doi: 10.1038/9536. [DOI] [PubMed] [Google Scholar]
- 129.Zinkernagel RM, Hengartner H. T-cell mediated immunopathology versus direct cytolysis by virus: implications for HIV and AIDS. Immunol Today. 1994;15:262–268. doi: 10.1016/0167-5699(94)90005-1. [DOI] [PubMed] [Google Scholar]
- 130.Shapiro R, Jordan M, Basu A, Scantlebury V, Potdar S, Tan H, et al. Kidney transplantation under a tolerogenic regimen of recipient pretreatment and low-dose postoperative immunosuppression, with subsequent weaning. Ann Surg. 2003;238:520–527. doi: 10.1097/01.sla.0000089853.11184.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Shapiro R, Basu A, Tan H, Gray E, Kahn A, Randhawa P, et al. Kidney transplantation under minimal immunosuppression after pretransplant lymphoid depletion with thymoglobulin or campath. J Am Coll Surg. 2005;200:505–515. doi: 10.1016/j.jamcollsurg.2004.12.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.McCurry K, Iacano A, Zeevi A, Yousem S, Zaldonis D, Girnita A, et al. Early outcomes in human lung transplantation utilizing thymoglobulin or campath 1H for recipient pretreatment followed by posttransplant tacrolimus near-monotherapy. J Thorac Cardiovasc Surg. 2005;130:528–537. doi: 10.1016/j.jtcvs.2004.09.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Marcos A, Eghtesad B, Fung JJ, Fontes P, Patel K, deVera M, et al. Use of alemtuzumab and tacrolimus monotherapy for cadaveric liver transplantation: with particular reference to hepatitis C virus. Transplantation. 2004;78:966–971. doi: 10.1097/01.tp.0000142674.78268.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Curry MP. Hepatitis B and Hepatitis C viruses in liver transplantation. Transplantation. 2004;78:955–963. doi: 10.1097/01.tp.0000140927.63952.58. [DOI] [PubMed] [Google Scholar]
- 135.Berenguer M, Crippin J, Gish R, Bass N, Bostrom A, Netto G, et al. A model to predict severe HCV-related disease following liver transplantation. Hepatology. 2003;38:34–41. doi: 10.1053/jhep.2003.50278. [DOI] [PubMed] [Google Scholar]
- 136.Neumann UP, Berg T, Bahra M, Puhl G, Guckelberger O, Langrehr JM, et al. Long-term outcome of liver transplants for chronic hepatitis C: a 10-year follow-up. Transplantation. 2004;77:226–231. doi: 10.1097/01.TP.0000101738.27552.9D. [DOI] [PubMed] [Google Scholar]
- 137.International Liver Transplantation Society Experts Panel. Report of the first International Liver Transplantation Society expert panel consensus conference on liver transplantation and hepatitis C. In: Wiesner RH, Sorrell MF, Villamil F, editors. Liver Transpl; Liver Transplantation and Hepatitis C. A Single Topic and Consensus Development Symposium; 2003. pp. S1–S119. [DOI] [PubMed] [Google Scholar]
- 138.Berenguer M, Prieto M, San Juan F, Rayon JM, Martinez F, Carrasco D, et al. Contribution of donor age to the recent decrease in patient survival among HCV-infected liver transplant recipients. Hepatology. 2002;36:202–210. doi: 10.1053/jhep.2002.33993. [DOI] [PubMed] [Google Scholar]
- 139.Reiser M, Hinrichsen H, Benhamou Y, Reesink HW, Wedemeyer H, Avendano C, et al. Antiviral efficacy of NS3-serine protease inhibitor BILN-2061 in patients with chronic genotype 2 and 3 hepatitis C. Hepatology. 2005;41:832–835. doi: 10.1002/hep.20612. [DOI] [PubMed] [Google Scholar]
- 140.Starzl TE. Experience in Hepatic Transplantation. Philadelphia: WB Saunders; 1969. Late results and complications; pp. 348–393. [Google Scholar]
- 141.Starzl TE, Penn I, Putnam CW, Groth CG, Halgrimson CG. Iatrogenic alterations of immunologic surveillance in man and their influence on malignancy. Transplant Rev. 1971;7:112–145. doi: 10.1111/j.1600-065x.1971.tb00465.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Coggin JH, Larson VM, Hilleman MR. Prevention of SV40 virus tumor-igenesis by irradiated, disrupted and iododeoxyuridine treated tumor cell antigens. Proceedings of the Society for Experimental Biology and Medicine. 1967;124:774–784. doi: 10.3181/00379727-124-31851. [DOI] [PubMed] [Google Scholar]
- 143.Hilleman MR. Personal historical chronicle of six decades of basic and applied research in virology, immunology, and vaccinology. Immunol Rev. 1999;170:7–27. doi: 10.1111/j.1600-065x.1999.tb01325.x. [DOI] [PubMed] [Google Scholar]
- 144.Bachmann MF, Hunziker L, Zinkernagel RM, Storni T, Kopf M. Maintenance of memory CTL responses by T helper cells and CD40-CD40 ligand: antibodies provide the key. Eur J Immunol. 2004;34:317–326. doi: 10.1002/eji.200324717. [DOI] [PubMed] [Google Scholar]
- 145.Klenerman P. Commentary: T cells get by with a little help from their friends. Eur J Immunol. 2004;34:313–316. doi: 10.1002/eji.200324844. [DOI] [PubMed] [Google Scholar]
- 146.Ehrlich P, Morgenroth J. Über Hämolysine. Fünfte Mittheilung Berl. Klin. Wschr. Vol. 38. 1901. pp. 598–604. Reproduced in (1957) The Collected Papers of Paul Ehrlich, ed. Himmelweit, F. London: Pergamon, Vol 2. pp. 234–245 (with English translation pp. 246–255) [Google Scholar]
- 147.Frey JR, Wenk P. Experimental studies on the pathogenesis of contact eczema in the guinea pig. Int Arch Allergy Appl Immunol. 1957;11:81– 100. doi: 10.1159/000228405. [DOI] [PubMed] [Google Scholar]
- 148.Barker CF, Billingham RE. The role of afferent lymphatics in the rejection of skin homografts. J Exp Med. 1968;128:197–221. doi: 10.1084/jem.128.1.197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Lakkis FG, Arakelov A, Konieczny BT, Inoue Y. Immunologic ignorance of vascularized organ transplants in the absence of secondary lymphoid tissue. Nat Med. 2000;6:686–688. doi: 10.1038/76267. [DOI] [PubMed] [Google Scholar]
- 150.Lafferty KJ, Prowse SJ, Simeonovic CJ. Immunobiology of tissue transplantation: a return to the passenger leukocyte concept. Annu Rev Immunol. 1983;1:143–173. doi: 10.1146/annurev.iy.01.040183.001043. [DOI] [PubMed] [Google Scholar]
- 151.Brent L. A History of Transplantation Immunology. London: Academic Press; 1997. pp. 182–338. [Google Scholar]
- 152.Waldmann H. Transplantation tolerance-where do we stand? Nat Med. 1999;5:1245–1248. doi: 10.1038/15197. [DOI] [PubMed] [Google Scholar]
- 153.Walker LSK, Abbas AK. The enemy within: keeping self-reactive T cells at bay in the periphery. Nat Rev Immunol. 2002;2:11–19. doi: 10.1038/nri701. [DOI] [PubMed] [Google Scholar]
- 154.Lechler R, Ng WF, Steinman RM. Dendritic cells in transplantation: friend or foe? Immunity. 2001;14:357–368. doi: 10.1016/s1074-7613(01)00116-9. [DOI] [PubMed] [Google Scholar]
- 155.Jonuleit H, Schmitt E, Schuler G, Knop J, Enk AH. Induction of inter-leukin 10-producing, nonproliferating CD4(+) T cells with regulatory properties by repetitive stimulation with allogeneic immature human dendritic cells. J Exp Med. 2000;192:1213–1212. doi: 10.1084/jem.192.9.1213. [DOI] [PMC free article] [PubMed] [Google Scholar]


