Abstract
Hepatic ischemia/reperfusion (I/R) injury has a significant influence on the outcome of liver transplants. Inhibiting certain enzymatic reactions that occur during I/R injury may have a protective effect on the liver during transplantation. After reviewing the biochemical pathways involved in hepatic I/R injury, we describe a pharmacologic line of defense against this injury by means of the enzyme tissue inhibitor of metalloproteinase 3 (TIMP-3). Current results suggest that TIMP-3 will play a clinically relevant role in improving outcomes of liver transplants by reducing I/R injury to the donor liver.
Keywords: Hepatic ischemia/reperfusion, liver, TIMP-3
INTRODUCTION
Over the past 30 years, orthotopic liver transplantation (OLT) has become the treatment of choice for end-stage liver disease. The qualified success of OLT can be measured by its quantifiable need. The 16,000 potential recipients on the national waiting list far surpass the 6,000 organs available per year for transplantation. Approximately 17% of potential OLT recipients on the transplant list die while waiting for a transplant.1-3
Options for increasing the donor pool have included the use of living donors, split livers, and “marginal” livers. Marginal livers include those from donors who are older, have fatty livers, or have anticipated longer ischemic (deprived of blood flow) times. These marginal livers are at increased risk for the development of primary nonfunction after liver transplantation, a common cause for retransplantation and of early death after OLT. Optimization of marginal livers—by means of preserving healthy liver cells that would ordinarily die during preservation and transportation of the donor liver—would increase the utilization of these organs without increasing the associated perioperative morbidity and mortality. Although the precise cause of primary nonfunction remains multifactorial, preservation injuries from ischemia have been implicated.1,4-11
Tumor Necrosis Factor α
An ischemia/reperfusion (I/R) injury is a multifactorial event involving many agents. Tumor necrosis factor α (TNF-α) is a signaling molecule involved in the regulation of a wide spectrum of biologic processes, including cell proliferation, differentiation, apoptosis, lipid metabolism, and coagulation.12-18 TNF-α plays a critical paradoxical dual role in the prevention and promotion of I/R injury in liver tissue and other organs, including the brain, heart, and lungs.12-18 TNF-α has been implicated in a variety of disease states, such as rheumatoid arthritis, acquired immune deficiency syndrome, cancer, autoimmune diseases (immune complex diseases), lung fibrosis, multiple sclerosis, delayed-type hypersensitivity reactions in skin, and bacterial and parasitic infections.12-18
Injury Model
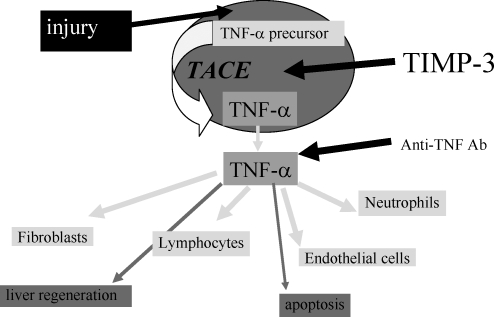
Several chronic disease states can be treated by regular infusions of anti–TNF-α antibodies.19 Transplantation, however, is unique. During reperfusion of an organ in the recipient, a surge of TNF-α occurs, resulting in significant inflammation mediated by a cytokine cascade (Figure 1). Infusions of TNF-α antibodies post reperfusion would remove the TNF-α but would not prevent the inflammatory cascade. This led to our research into the formation of active TNF-α by its converting enzyme and the hypothesis that blocking the converting enzyme in the donor may reduce TNF-α during reperfusion in the recipient (Figure 2).
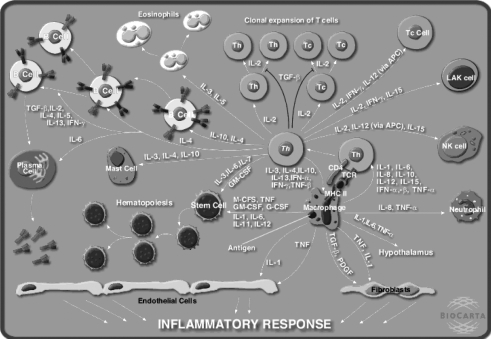
Figure 1.
A hypothesis about the biochemical cascade that occurs during an inflammatory response. (Diagram provided by BioCarta.)
Figure 2.
A simplified interpretation of an injury, regulatory response of tumor necrosis factor α (TNF-α), and inhibitory role of tissue inhibitor of metalloproteinase 3 (TIMP-3). TACE, TNF-α converting enzyme.
TNF-α Converting Enzyme
In response to a stimulus (such as an I/R injury), the body's naturally occurring membrane-bound proTNF-α is enzymatically cleaved by TNF-α converting enzyme (TACE) into its biologically active form, TNF-α.20-22 TACE has been shown to be involved in a variety of physiologic or pathophysiologic processes.23-28 With the increasing use of expanded donor allografts, interest in I/R research in transplantation has been renewed.29
We hypothesize that the inhibition of TACE will reduce hepatic I/R injury during transplantation. Manipulation of this inhibition will enable the increased use of marginal livers that would otherwise be discarded, thereby increasing the donor pool of organs. The novel implementation of a pharmacologic line of defense against I/R injury is an innovation that renders nonviable livers into organs eligible for transplantation.
EXPERIMENTAL DATA
This review highlights the work done in the Transplantation Research Laboratory at the Ochsner Clinic Foundation. The focus of the laboratory has been to identify potential enzymatic targets in donor livers that could be pharmacologically treated to improve outcomes in liver transplantation.
TACE Upregulation
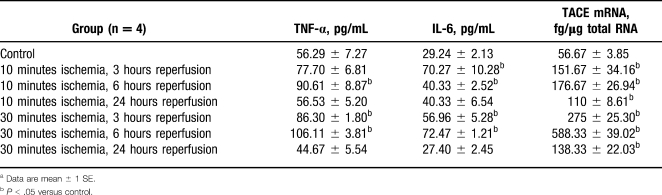
In a well-established rat model of partial warm hepatic I/R injury, we found that low levels of TACE were detected in normal liver tissue by both reverse transcriptase–polymerase chain reaction (RT-PCR) and Western blot.30-32 Ten minutes of warm ischemia resulted in a logarithmic rise in TACE messenger RNA (mRNA) levels, which peaked 6 hours after hepatic reperfusion. At 24 hours, TACE mRNA levels remained overexpressed, when compared to baseline, but had declined from the 6-hour peak. After 30 minutes of ischemia, hepatic TACE mRNA levels demonstrated a similar upregulated pattern of expression, although each time point had a 2-fold increase when compared to its 10-minute ischemia counterpart. Western blot analysis demonstrated a strong increase in TACE protein levels 6 hours after the ischemic injury. At 10 and 30 minutes of ischemia followed by reperfusion, TNF-α and TNF-α receptor mRNA levels were upregulated in a pattern similar to TACE mRNA levels. Serum TNF-α and interleukin 6 (IL-6) levels correlated with the observed increases in mRNA levels in the liver.30,31 This was the first reported documentation of TACE in liver tissue; this finding demonstrates that TACE is an inducible enzyme that is upregulated by ischemia.
TACE Inhibition
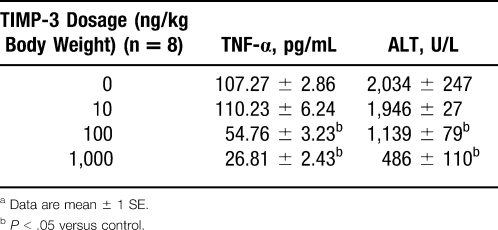
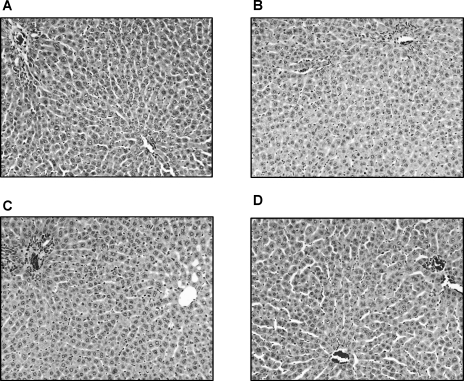
To further confirm the involvement of TACE and its role in hepatic I/R injury, we designed experiments to dampen TACE activity during hepatic I/R injury. Tissue inhibitor of metalloproteinase 3 (TIMP-3), a naturally occurring inhibitor of TACE, was given intraperitoneally 1 hour before hepatic I/R injury. Results are shown in Table 1. High doses of TIMP-3 markedly reduced both serum TNF-α and alanine aminotransferase (ALT) levels and preserved the architectural integrity during hepatic I/R injury (Table 2 and Figure 3).32 On the basis of these studies, we can conclude that TACE does play an important role in hepatic I/R injury. TIMP-3, through highly selective inhibition of TACE activity, does reduce hepatic I/R injury at both the biochemical and histologic levels.27,32-38
Table 1.
Serum Levels of Tumor Necrosis Factor α (TNF-α) and Interleukin 6 (IL-6) and Hepatic Levels of TNF-α Converting Enzyme (TACE) mRNA After Partial Hepatic Ischemia/Reperfusion Injurya
Table 2.
Serum Alanine Aminotransferase (ALT) and Tumor Necrosis Factor α (TNF-α) Levels After Partial Liver Ischemia (30 Minutes) and Reperfusion (6 Hours) in Animal Groups With or Without Tissue Inhibitor of Metalloproteinase 3 (TIMP-3) Treatmenta
Figure 3.
Histologic changes of hepatic ischemia/reperfusion injuries after treatment with tissue inhibitor of metalloproteinase 3 (TIMP-3) at different dosages. A. Control group (rats received saline injection). B–D. Treatment groups (rats received TIMP-3 at dose of 10, 100, and 1,000 ng/kg body weight, respectively) (hematoxylin-eosin, original magnifications ×100).
TNF-α Regulation
Further TACE inhibition studies progressed to a total warm ischemia model, which is more directly comparable to a potential transplant application; also, it eliminated other potential pitfalls of the partial warm ischemia model.
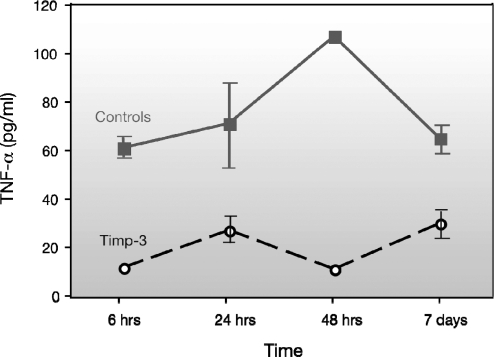
To illustrate TACE activity during total warm I/R injury, TIMP-3 was given 1 hour before surgery. At a dosage of 1,000 ng/kg body weight, TIMP-3 significantly decreased serum TNF-α levels at all 4 time points (6, 24, and 48 hours and 7 days) compared to controls. The control animals subjected to I/R injury had high levels of serum TNF-α up to 7 days after the injury (61.3–107.6 pg/mL). With TIMP-3 pretreatment, notable inhibition (62%–90%, P < .05) was present for the first 48 hours after injury (ranging from 11.6 pg/mL to 27.1 pg/mL). On day 7, although the pretreated animals recovered, with increased TNF-α levels, their levels were still 53% below the levels of the control animals, at only 30 pg/mL (Figure 4).
Figure 4.
Changes in tumor necrosis factor α (TNF-α) levels after tissue inhibitor of metalloproteinase 3 (TIMP-3) pretreatment.
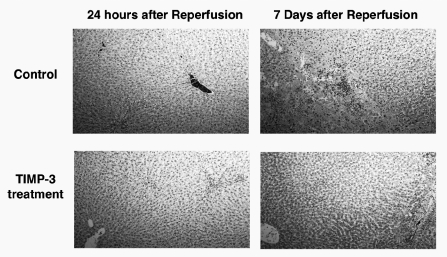
At 24 hours after reperfusion, both control and TIMP-3–treated liver samples showed mild edema. At 7 days after reperfusion, TIMP-3–treated liver samples continued to show only mild edema. Control liver samples showed more pronounced ischemic changes, with hepatocyte collapse, necrosis, and hemorrhage (Figure 5).
Figure 5.
Histology slides of liver samples at 24 hours and 7 days after ischemia (hematoxylin-eosin, original magnifications ×100). TIMP-3, tissue inhibitor of metalloproteinase 3.
HUMAN STUDIES
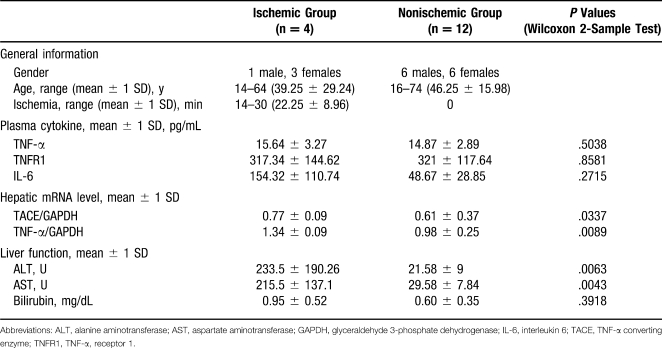
In a pilot study, the presence of TACE in human donor liver tissues was assessed. During organ procurement, liver biopsies and plasma samples were collected from 16 deceased donors. Four of the donors had documented hepatic ischemia within 48 hours of procurement. TNF-α, TNF-α receptor 1, and IL-6 were measured by enzyme-linked immunosorbent assay in donor plasma sampled at the time of procurement. Hepatic TACE, TNF-α, and glyceraldehyde 3-phosphate dehydrogenase mRNAs were detected by RT-PCR. Serum ALT, aspartate aminotransferase, and bilirubin levels were obtained and analyzed (Table 3).
Table 3.
Analysis of Human Pilot Study of the Role of Tumor Necrosis Factor α (TNF-α ) in Donors
This study is the first to document the existence of TACE in human liver tissue: TACE appears to be upregulated in donor livers in response to ischemia. Further study is necessary to evaluate the value of TACE as a potential target of inhibition within donor livers. Extrapolating from our animal studies, TACE inhibition may ameliorate human hepatic injury from ischemia. If true, inhibiting TACE in organ donors might protect donor livers from ischemic injury and perhaps even allow the salvage of organs from donors who have sustained severe ischemia. This could potentially expand the donor liver pool for transplantation.
CURRENT RESEARCH
Because of its role and its potential inhibition during the transplant event, TACE appears to be a clinically viable target for improving “marginal” livers. Currently, the laboratory is examining the effects of TACE inhibition on liver apoptosis (programmed cell death) and liver regeneration. The degree of TACE inhibition may alter the balance of apoptosis and regeneration. Understanding this balance is the next step to developing a clinically useful pharmacologic modifier to increase the potential pool of donor organs and thus reduce the deaths of patients on the waitlist.
REFERENCES
- 1.Ploeg R. J., D'Alessandro A. M., Knechtle S. J., et al. Risk factors for primary dysfunction after liver transplantation: a multivariate analysis. Transplantation. 1993;55((4)):807–813. doi: 10.1097/00007890-199304000-00024. [DOI] [PubMed] [Google Scholar]
- 2.Starzl T. E., Demetris A. J., Van Thiel D. Liver transplantation (2) N Eng J Med. 1989;321((16)):1092–1099. doi: 10.1056/NEJM198910193211606. [DOI] [PubMed] [Google Scholar]
- 3.2001 Annual Report of the U.S. Organ Procurement and Transplantation Network and the Scientific Registry for Transplant Recipients: Transplant Data 1991-2000. Rockville, MD: US Department of Health and Human Services, Health Resources and Services Administration, Office of Special Programs, Division of Transplantation; Richmond, VA: United Network for Organ Sharing; Ann Arbor, MI: University Renal Research and Education Association; [Google Scholar]
- 4.Busuttil R. W., Tanaka K. The utility of marginal donors in liver transplantation [review] Liver Transpl. 2003;9((7)):651–663. doi: 10.1053/jlts.2003.50105. [DOI] [PubMed] [Google Scholar]
- 5.Kupiec-Weglinski J. W., Busuttil R. W. Ischemia and reperfusion injury in liver transplantation. Transplant Proc. 2005;37((4)):1653–1656. doi: 10.1016/j.transproceed.2005.03.134. [DOI] [PubMed] [Google Scholar]
- 6.Belzer F. O., Southard J. H. Principles of solid-organ preservation by cold storage. Transplantation. 1988;45((4)):673–676. doi: 10.1097/00007890-198804000-00001. [DOI] [PubMed] [Google Scholar]
- 7.Todo S., Nery J., Yanaga K., et al. Extended preservation of human liver grafts with SALINE solution. JAMA. 1989;261:711–714. [PMC free article] [PubMed] [Google Scholar]
- 8.2004 Annual Report of the U.S. Organ Procurement and Transplantation Network and the Scientific Registry of Transplant Recipients: Transplant Data 1994-2003. Rockville, MD: Department of Health and Human Services, Health Resources and Services Administration, Healthcare Systems Bureau, Division of Transplantation; Richmond, VA: United Network for Organ Sharing; Ann Arbor, MI: University Renal Research and Education Association; [Google Scholar]
- 9.Harvey P. R., Iu S., McKeown C. M., Petrunka C. N., Ilson R. G., Strasberg S. M. Adenine nucleotide tissue concentrations and liver allograft viability after cold preservation and warm ischemia. Transplantation. 1988;45((6)):1016–1020. doi: 10.1097/00007890-198806000-00004. [DOI] [PubMed] [Google Scholar]
- 10.Cisneros C., Guillén F., Gomez R., et al. Analysis of warm ischemia time for prediction of primary nonfunction of the hepatic graft. Transplant Proc. 1991;23((3)):1976. [PubMed] [Google Scholar]
- 11.Clavien P. A., Harvey P. R., Strasberg S. M. Preservation and reperfusion injuries in liver allografts: an overview and synthesis of current studies. Transplantation. 1992;53((5)):957–978. doi: 10.1097/00007890-199205000-00001. [DOI] [PubMed] [Google Scholar]
- 12.Arumugam T. V., Shiels I. A., Woodruff T. M., Granger D. N., Taylor S. M. The role of the complement system in ischemia-reperfusion injury. Shock. 2004;21((5)):401–409. doi: 10.1097/00024382-200405000-00002. [DOI] [PubMed] [Google Scholar]
- 13.Carini R., Albano E. Recent insights on the mechanisms of liver preconditioning. Gastroenterology. 2003;125((5)):1480–1491. doi: 10.1016/j.gastro.2003.05.005. [DOI] [PubMed] [Google Scholar]
- 14.Chen T., Zamora R., Zuckerbraun B., Billiar T. R. Role of nitric oxide in liver injury. Curr Mol Med. 2003;3((6)):519–526. doi: 10.2174/1566524033479582. [DOI] [PubMed] [Google Scholar]
- 15.Ke B., Shen X. D., Gao F., Busuttil R. W., Kupiec-Weglinski J. W. Interleukin 13 gene transfer in liver ischemia and reperfusion injury: role of Stat6 and TLR4 pathways in cytoprotection. Hum Gene Ther. 2004;15((7)):691–698. doi: 10.1089/1043034041361244. [DOI] [PubMed] [Google Scholar]
- 16.Oreopoulos G. D., Wu H., Szaszi K., et al. Hypertonic preconditioning prevents hepatocellular injury following ischemia/reperfusion in mice: a role for interleukin 10. Hepatology. 2004;40((1)):211–220. doi: 10.1002/hep.20281. [DOI] [PubMed] [Google Scholar]
- 17.Teoh N., Leclercq I., Pena A. D., Farrell G. Low-dose TNF-alpha protects against hepatic ischemia-reperfusion injury in mice: implications for preconditioning. Hepatology. 2003;37((1)):118–128. doi: 10.1053/jhep.2003.50009. [DOI] [PubMed] [Google Scholar]
- 18.Teoh N. C., Farrell G. C. Hepatic ischemia reperfusion injury: pathogenic mechanisms and basis for hepatoprotection. J Gastroenterol Hepatol. 2003;18((8)):891–902. doi: 10.1046/j.1440-1746.2003.03056.x. [DOI] [PubMed] [Google Scholar]
- 19.Hyrich K. L., Deighton C., Watson K. D., et al. Benefit of anti-TNF therapy in rheumatoid arthritis patients with moderate disease activity. Rheumatology (Oxford) 2009;48((10)):1323–1327. doi: 10.1093/rheumatology/kep242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Black R. A., Rauch C. T., Kozlosky C. J., et al. A metalloproteinase disintegrin that releases tumour-necrosis factor-alpha from cells. Nature. 1997;385((6618)):729–733. doi: 10.1038/385729a0. [DOI] [PubMed] [Google Scholar]
- 21.Moss M. L., Jin S. L., Milla M. E., et al. Cloning of a disintegrin metalloproteinase that processes precursor tumour-necrosis factor-α [erratum in Nature. 1997;386(6626):738] Nature. 1997;385((6618)):733–736. doi: 10.1038/385733a0. [DOI] [PubMed] [Google Scholar]
- 22.Mezyk R., Bzowska M., Bereta J. Structure and functions of tumor necrosis factor-alpha converting enzyme. Acta Biochim Pol. 2003;50((3)):625–645. [PubMed] [Google Scholar]
- 23.Black R. A. TIMP3 checks inflammation [comment on Nat Genet. 2004;36(9):969-977] Nat Genet. 2004;36((9)):934–935. doi: 10.1038/ng0904-934. [DOI] [PubMed] [Google Scholar]
- 24.Akatsu T., Nakamura M., Satoh M., Hiramori K. Increased mRNA expression of tumour necrosis factor-alpha and its converting enzyme in circulating leucocytes of patients with acute myocardial infarction. Clin Sci (Lond) 2003;105((1)):39–44. doi: 10.1042/CS20020367. [DOI] [PubMed] [Google Scholar]
- 25.Hurtado O., Cárdenas A., Lizasoain I., et al. Up-regulation of TNF-alpha convertase (TACE/ADAM17) after oxygen-glucose deprivation in rat forebrain slices. Neuropharmacology. 2001;40((8)):1094–1102. doi: 10.1016/s0028-3908(01)00035-1. [DOI] [PubMed] [Google Scholar]
- 26.Romera C., Hurtado O., Botella S. H., et al. In vitro ischemic tolerance involves upregulation of glutamate transport partly mediated by the TACE/ADAM17-tumor necrosis factor-alpha pathway. J Neurosci. 2004;24((6)):1350–1357. doi: 10.1523/JNEUROSCI.1596-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Goto T., Ishizaka A., Kobayashi F., et al. Importance of tumor necrosis factor-alpha cleavage process in post-transplantation lung injury in rats. Am J Respir Crit Care Med. 2004;170((11)):1239–1246. doi: 10.1164/rccm.200402-146OC. [DOI] [PubMed] [Google Scholar]
- 28.Gilles S., Zahler S., Welsch U., Sommerhoff C. P., Becker B. F. Release of TNF-alpha during myocardial reperfusion depends on oxidative stress and is prevented by mast cell stabilizers. Cardiovasc Res. 2003;60((3)):608–616. doi: 10.1016/j.cardiores.2003.08.016. [DOI] [PubMed] [Google Scholar]
- 29.Vassalli P. The pathophysiology of tumor necrosis factors. Annu Rev Immunol. 1992;10:411–452. doi: 10.1146/annurev.iy.10.040192.002211. [DOI] [PubMed] [Google Scholar]
- 30.Tang Z-Y., Loss G., Nair S., Eason J., Cohen A. Increased hepatic expression of tumor necrosis factor-alpha-converting enzyme (TACE) and TNF-α after ischemia/reperfusion injury in rat model. J Gastrointest Surg. 2005;9:530. [Google Scholar]
- 31.Tang Z-Y., Loss G., Eason J., Cohen A. Role of tumor necrosis factor-alpha-converting enzyme (TACE) in hepatic ischemia/reperfusion injury rat model. Hepatology. 2004;40((4)):699a. [Google Scholar]
- 32.Tang Z-Y., Loss G., Carmody I., Cohen A. J. TIMP-3 ameliorates hepatic ischemia/reperfusion injury through inhibition of tumor necrosis factor-alpha-converting enzyme activity in rats. Transplantation. 2006;82((11)):1518–1523. doi: 10.1097/01.tp.0000243381.41777.c7. [DOI] [PubMed] [Google Scholar]
- 33.Uhlmann D., Gaebel G., Armann B., et al. Attenuation of proinflammatory gene expression and microcirculatory disturbances by endothelin A receptor blockade after orthotopic liver transplantation in pigs. Surgery. 2006;139((1)):61–72. doi: 10.1016/j.surg.2005.07.006. [DOI] [PubMed] [Google Scholar]
- 34.Jahnke C., Mehrabi A., Golling M., et al. Evaluation of microperfusion disturbances in the transplanted liver after Kupffer cell destruction using GdCl3: an experimental porcine study. Transplant Proc. 2006;38((5)):1588–1595. doi: 10.1016/j.transproceed.2006.02.067. [DOI] [PubMed] [Google Scholar]
- 35.Wang X., Feuerstein G. Z., Xu L., et al. Inhibition of tumor necrosis factor-alpha-converting enzyme by a selective antagonist protects brain from focal ischemic injury in rats. Mol Pharmacol. 2004;65((4)):890–896. doi: 10.1124/mol.65.4.890. [DOI] [PubMed] [Google Scholar]
- 36.Zhang Y., Xu J., Levin J., et al. Identification and characterization of 4-[[4-(2-butynyloxy)phenyl]sulfonyl]-N-hydroxy-2,2-dimethyl-(3S) thiomorpholinecarboxamide (TMI-1), a novel dual tumor necrosis factor-alpha-converting enzyme/matrix metalloprotease inhibitor for the treatment of rheumatoid arthritis. J Pharmacol Exp Ther. 2004;309((1)):348–355. doi: 10.1124/jpet.103.059675. [DOI] [PubMed] [Google Scholar]
- 37.Doggrell S. A. TACE inhibition: a new approach to treating inflammation. Expert Opin Investig Drugs. 2002;1((7)):1003–1006. doi: 10.1517/13543784.11.7.1003. [DOI] [PubMed] [Google Scholar]
- 38.Newton R. C., Solomon K. A., Covington M. B., et al. Biology of TACE inhibition. Ann Rheum Dis. 2001;60((suppl 3)):iii25–iii32. doi: 10.1136/ard.60.90003.iii25. [DOI] [PMC free article] [PubMed] [Google Scholar]