Abstract
Endothelium-derived epoxyeicosatrienoic acids (EETs) relax vascular smooth muscle by activating potassium channels and causing membrane hyperpolarization. Recent evidence suggests that EETs act via a membrane binding site or receptor. To further characterize this binding site/receptor, we synthesized 20-iodo-14,15- epoxyeicosa-8Z-enoyl-3-azidophenylsulfonamide (20-I-14,15-EE8ZE-APSA), an EET analog with a photoactive azido group. 20-I-14,15- EE8ZE-APSA and 14,15-EET displaced 20-125I-14,15-epoxyeicosa-5Z-enoic acid binding to U937 cell membranes with Ki’s of 3.60 and 2.73 nM, respectively. The EET analog relaxed preconstricted bovine coronary arteries with an ED50 comparable to 14,15-EET. Using electrophoresis, 20-125I-14,15-EE8ZE-APSA labeled a single 47 KDa band in membranes from U937, smooth muscle and endothelial cells and bovine coronary arteries. In U937 cell membranes, the 47 KDa radiolabeling was inhibited in a concentration-dependent manner by 8,9-EET, 11,12-EET, and 14,15-EET (IC50’s = 444, 11.7 and 8.28 nM, respectively). The structurally unrelated EET ligands, miconazole, MS-PPOH and ketoconazole also inhibited the 47 KDa labeling. In contrast, radiolabeling was not inhibited by 8,9-dihydroxyeicosatrienoic acid, 5-oxo-eicosatetraenoic acid, a biologically inactive thiirane analog of 14,15-EET, the opioid antagonist naloxone, the thromboxane mimetic U46619 or the cannabinoid antagonist AM251. Radiolabeling was not detected in membranes from HEK293T cells expressing 79 orphan receptors. These studies indicate that vascular smooth muscle, endothelial and U937 cell membranes contain a high affinity EET binding protein that may represent an EET receptor. This EET photoaffinity labeling method with high signal to noise ratio may lead to new insights into the expression and regulation of the EET receptor.
Epoxyeicosatrienoic acids (EETs) are cytochrome P450 (CYP) metabolites of arachidonic acid (AA) (1–3). Four regioisomeric EETs (14,15-, 11,12-, 8,9- and 5,6-EET) are synthesized. Several CYP epoxygenases including CYP2C and CYP2J are capable of synthesizing the EETs. EETs have a variety of biological activities. They decrease inflammation, are antinocieceptive, promote angiogenesis, protect the heart and brain from ischemic-reperfusion injury and decrease platelet adhesion (4–9). Additionally, EETs function as endothelium-derived hyperpolarization factors (EDHF) in the coronary circulation (10–13). They are synthesized and released by the vascular endothelium in response to agonists such as bradykinin, acetylcholine, cyclic stretch and shear stress (13). EETs relax vascular smooth muscle by activating large conductance, calcium-activated potassium (BKCa) channels resulting in membrane hyperpolarization, a reduction in calcium influx through voltage activated calcium channels. 11,12-EET activation of smooth muscle cell BKCa channels requires intracellular GTP, but not ATP, and is blocked by the guanine nucleotide binding protein (G protein) inhibitor GTPβS and by an anti-Gαs antibody (14). Thus, a G protein, likely Gαs, mediates EET activation of BKCa channels.
Several lines of evidence suggest that EETs act through a specific binding site or receptor. Specific structural features are required for 14,15-EET to relax the bovine coronary artery (15). For full agonist activity, the structure must contain a 20 carbon chain, a S,R-cis-epoxide, a 8Z olefin and a carboxyl at carbon 1, i.e., 14(S),15(R)-cis-epoxyeicosa-8Z-enoic acid (14,15-EE8ZE). Partial reduction of the olefins to produce 14,15-EE5ZE results in an antagonist (16). The need for a specific enantiomer of the 14,15-epoxide suggests a specific binding site is involved in relaxation. When 14,15-EET was tethered to silica beads to maintain the EET extracellularly, 14,15-EET and the tethered 14,15-EET had similar activity in smooth muscle cells indicating an extracellular site of action (17). These studies suggest EETs act on a cell surface binding site or receptor.
The existence of both high and low affinity EET receptors or binding sites are supported by many physiological and biochemical experiments. The low affinity EET receptors and binding sites include fatty-acid binding protein (18), peroxisomal proliferator-activated receptor-α (PPAR) (19), PPAR-γ (20), peripheral benzodiazepine receptor (21), cannabinoid (CB) receptor (21), neurokinin receptor (21), dopamine receptor (21) and ATP-sensitive K channel (22), prostaglandin E (EP2) receptor (23) and thromboxane (TP) receptor (24). However, many physiological responses are elicited by lower concentrations of EETs suggesting the existence of high affinity EET receptors (13). To study the high affinity EET binding, Wong et al. pioneered the use of guinea pig mononuclear and U937 cells for direct radioligand binding studies (25–27). A high affinity specific binding site with a KD of 1–10 nM was characterized using 14,15-EET agonist radioligands such as 14,15-[3H]EET and 20-125I-14,15-epoxyeicosa-8Z-enoic acid (EE8ZE) and an EET antagonist, 20-125I-14,15-epoxyeicosa-5Z-enoic acid (EE5ZE) (26, 28–30). For example, 20-125I-14,15-EE8ZE showed high affinity, reversible specific binding that was inhibited by the G protein ligand GTPγS (29). Overall, these studies suggested 14,15-EET acts through an unknown G protein coupled receptor (GPCR).
Here, we developed and characterized a 14,15-EET agonist photoaffinity probe with a photoactive arylazido (AZ) group attached to the carboxyl end of 20-I-14,15-EE8ZE. This analog, 20-iodo-14,15-epoxyeicosa-8Z-enoyl-3-azidophenylsulfonamide (20-I-14,15-EE8ZE-APSA), may be radiolabeled as described for 20-125I-14,15-EE8ZE (29). The photoactive group is covalently bound to an EET-targeted receptor or binding site through covalent binding upon UV light-induced photolytic cross-linking. The labeled receptor/binding site is resolved by electrophoresis and detected by radioautography. The photoaffinity labeling method provides a high signal/noise ratio due to protein separation prior to detection, e.g., non-specifically labeled proteins are separated from specifically labeled proteins. This method also provides insights into the characteristics of the receptor/binding protein such as molecular weight, density and binding specificity. Using this method, we have characterized a 47 kDa protein that is photolabelled by 20-125I-14,15-EE8ZE-APSA. This photolabeled protein was detected in U937 cells, endothelial cells and vascular smooth muscle cells.
EXPERIMENTAL PROCEDURES
Materials
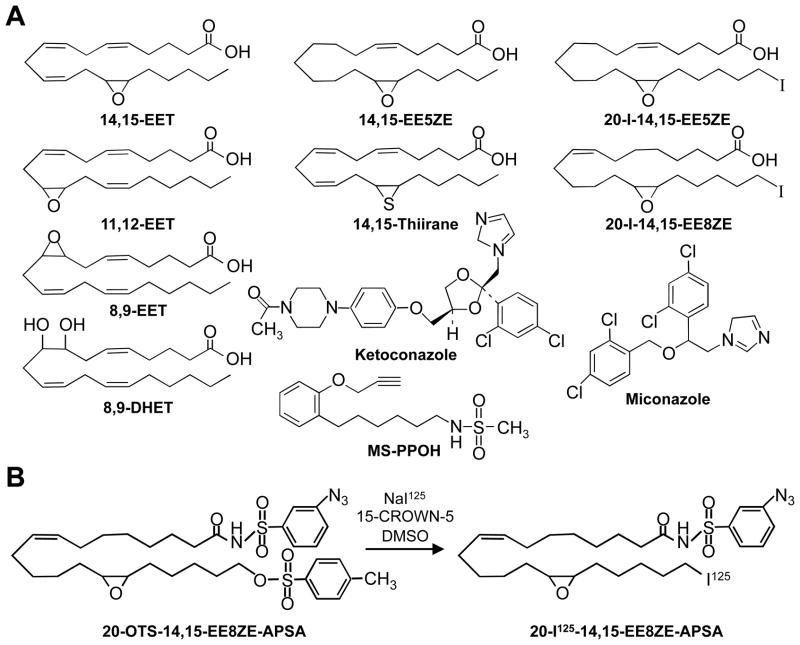
EET analogs were synthesized as previously described and their structures are indicated in Figure 1A (15, 28, 29). Chemicals and solvents were purchased from Sigma-Aldrich (St. Louis, MO).
Figure 1.
Chemical structures of EETs and EET analogs. A. Structures of chemicals used in the study. B. Chemical synthesis of 20-125I-14,15-EE8ZE-APSA (mAPSA isomer).
Synthesis of Carrier Free 20-125I-14,15-EE8ZE-APSA
Carrier free 20-125I-14,15-EE8ZE-APSA (meta isomer) was synthesized from 20-tosyl (OTs)-14,15-EE8ZE-APSA using different conditions than were described for 20-I-14,15-EE8ZE-APSA synthesis (see Supplemental Material). (125I was used in designated areas and with appropriate precautions as designated by the radiation safety office of the Medical College of Wisconsin). Two mCi of carrier-free Na125I (0.8 nmol; 17.4 Ci/mg) in 20 μl was added to 20 μl of DMSO containing 640 nmol of 20-OTs-14,15-EE8ZE-APSA, and 20 μl of 15-crown-5 (Sigma Aldrich, St. Louis, MO) as a phase transfer reagent. The reaction was carried out at 25°C for 4–7 days with shaking 2 to 3 times daily and stopped by 10 μl of saturated Na2S2O3 aqueous solution. The reaction mixture was transferred to a 15 ml conical glass tube and extracted with 0.25 ml of diethyl ether four times. The extract was dried under N2 and purified by high performance liquid chromatography (HPLC) using a C18 reverse-phase column (Nucleosil; 5 μm; 4.6 × 250 mm; Phenomenex, Torrance, CA). A linear gradient of 50% to 100% solvent B in solvent A (solvent B: acetonitrile/glacial acetic acid = 999/1; solvent A: water) over 40 min was used to elute 20-125I-14,15-EE8ZE-APSA. 20-I-14,15-EE8ZE-APSA was used as a standard to mark the collection time. The specific activity of 20-125I-14,15-EE8ZE-APSA was calculated to be 2000 Ci/mmol.
Vascular Reactivity of Bovine Coronary Arteries
The protocols are the same as previously reported (10, 15). The left anterior descending branch of bovine coronary artery was used for tension measurements using a model FT-03C force transducer (Grass Instruments, Milford, MA), MacLab ETH-400 bridge amplifier, and MacLab 8e A/D converter (AD Instruments, Colorado Springs, CO) controlled by a Macintosh computer. Rings were pre-constricted with the thromboxane mimetic U46619 (20 nM), and relaxation responses to increasing concentrations of 14,15-EET or the ortho (o)APSA, meta (m)APSA or para (p)APSA isomers of 20-I-14,15-EE8ZE-APSA were measured. Results were expressed as % relaxation with the basal tension representing 100% relaxation.
Cell Culture and Membrane Preparation
Endothelial cells and smooth muscle cells were isolated and cultured as previously described (31, 32). Human endothelial cells were obtained from Invitrogen (Carlsbad, CA) and cultured according to the company’s recommendations. U937 cells were cultured in suspension in RPMI 1640 medium (Invitrogen) mixture containing 10% fetal bovine serum (HyClone Laboratories, Logan, UT), 25 mM HEPES, 2 mM L-glutamine, and 1 mM sodium pyruvate, 100 U/ml penicillin, 100 μg/ml streptomycin, and 0.25 μg/ml amphotericin B as previously reported (28–30) and harvested after reaching a density of 5 to 10 × 105 cell/ml.
To prepare cell or tissue mixed membranes, phosphate-buffered saline, pH 7.4, was used to wash the cells or tissue. After sonicating for 20 s in Hanks’ balanced salt solution with protease inhibitors (Roche), the lysates were centrifuged at 1,000g for 10 min and the supernatants were obtained. These supernatants were centrifuged again at 110,000g for 45 min, the pellets were resuspended in binding buffer (10 mM HEPES, 5 mM CaCl2, 5 mM MgCl2, and 5 mM EGTA, pH 7.4). This 100,000g pellet contains a mixture of membranes (29).
To prepare membranes enriched in plasma membranes, the U937 cell pellet was re-suspended in 10 ml of sucrose buffer (10 mM Tris Base, 0.25 M sucrose, pH 7.4) and sonicated for 20 sec five times. The homogenate was centrifuged at 700g for 15 min, and the supernatant was centrifuged at 66,000g for 45 min. The pellet was washed by re-suspended in 10 ml of sucrose buffer through sonication and centrifuged again at 66,000g for 45 min. The pellet was re-suspended in 5 ml of buffer, carefully layered onto top of sucrose gradient (5.5 ml of 40% sucrose and 5.5 ml of 30% sucrose) and centrifuged at 77,000g for 90 min. Middle interface was removed, mixed with three volumes of sucrose buffer, and centrifuged at 66,000g for 45 min. The pellet was re-suspended in binding buffer.
20-125I-14,15-EE5ZE Binding Assays
Using 20-125I-14,15-EE5ZE, U937 membrane binding assays were performed as previously reported (30). Briefly, 1–2 nM of 20-125I-14,15-EE5ZE, 50 μg of membrane protein, and different concentrations of the oAPSA, mAPSA or pAPSA isomers of 20-I-14,15-EE8ZE-APSA with or without 20 μM 14,15-EE5ZE were incubated for 15 min at 4°C. Following filtration with a Brandel 48-well harvester system (Brandel Inc., Gaithersburg, MD), the radioactivity was measured by gamma scintillation counting. The data were analyzed by Prism software (Prism Software Co., Orange, CA). Specific binding was calculated as the difference between the absence vs. presence of 14,15-EE5ZE.
Photoaffinity Labeling Using 20-125I-14,15-EE8ZE-APSA
The photoaffinity probe, 20-125I-14,15-EE8ZE-APSA (mAPSA isomer) (1 nM) was incubated with 400 μg of membranes for 15 min at 4°C with EET regioisomers or vehicle in a total volume of 100 μl. The reaction mixtures were exposed to UV light at a wavelength of 280 nm for 15 min. After photolysis, the light induced free radical reactions were stopped by addition of dithiothreitol containing sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) loading buffer and heated for 15 min at 100°C. The protein mixtures were further separated by denaturing SDS-PAGE. SDS-PAGE gels were stained by Biosafe Coomassie Blue for 30 min, destained with distilled water for 2 h, and dried under a vacuum gel dryer for 1 h. Radioactivity on the gel was determined by radioautography using Kodak X-ray film. The film was exposed to the gel at −80°C for 18–24 h. For concentration-dependent inhibition curves, the intensity of labeling was determined by ImageJ software (NIH, Bethesda, MD), and the data were normalized and plotted using GraphPad (GraphPad Software, LaJolla, CA) assuming a one-site competitive binding model. The labeling with the highest concentration of the inhibitor and no inhibitor were set as 0 and 100% labeling, respectively.
Overexpression and Photolabeling of GPCRs in HEK293T cells
HEK293 cells overexpressed GPCR cells were provided by Multispan Inc. (Hayward, California). Candidate GPCRs proteins were cloned in pMEX vectors and overexpressed in HEK293T cells in 96 well plates. An equal number of U937 cells was used as a positive control and vector transfected HEK293T cells were a negative control. Each well of cells was treated with 30 μl of lysis buffer (50 mM Tris pH 7.4, 150 mM NaCl, 0.5% Nonidet P-40, 1% deoxycholic acid, and protease inhibitor cocktail from Roche), incubated in ice for 30 min and homogenized by sonification. Cell lysates were incubated with 1 nM of 20-125I-14,15-EE8ZE-APSA at 4°C, photolabeled by exposure to UV light for 15 min and analyzed by electrophoresis and radioautography by the protocol described above.
Data analysis
Data are expressed as means ± S.E.M. Significance differences between individual groups were evaluated by Student t-test or ANOVA followed by Student-Newman-Keuls multiple comparison test. P values <0.05 were considered statistically significant.
RESULTS
Synthesis of Carrier free 20-125I-14,15-EE8ZE-APSA
The structural difference between the photoprobe, 20-125I-14,15-EE8ZE-APSA, and the radioligand, 20-125I-14,15-EE8ZE, is the addition of the phenylsulfonamide group containing a meta photoactive azide (29). The synthesis of the 20-125I-14,15-EE8ZE-APSA utilizes the 20-OTS-14,15-EE8ZE-APSA precursor (Figure 1B). While the synthesis of 20-125I-14,15-EE8ZE was carried out directly in acetone (29), these reaction conditions failed with 20-125I-14,15-EE8ZE-APSA synthesis. Other conditions were tested. Incubation of 20-OTs-14,15-EE8ZE-APSA with Na125I for 4–7 days in DMSO with the phase transfer reagent 15-crown-5 provided the best reaction yield for 20-125I-EE8ZE-APSA resulting in a specific activity of 2000 mCi/mmol (Figure 1B).
Agonist Activity of 20-I-14,15-EE8ZE-APSA
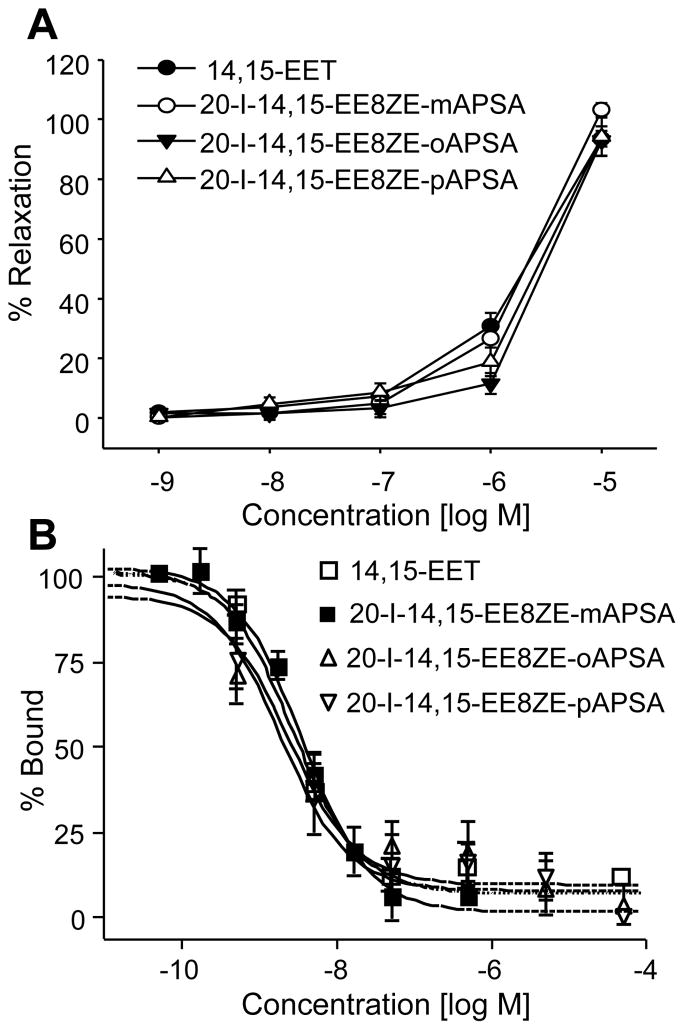
Previous structure-activity studies indicate that 20-I-14,15-EE8ZE is a EET receptor agonist (29). To determine whether the carbon-1 modification to 20-I-14,15-EE8ZE affects agonist activity, relaxation of U46619-pre-constricted bovine coronary artery rings to 14,15-EET and 20-I-14,15-EE8ZE-APSA isomers were compared. The oAPSA, mAPSA and pAPSA isomers of 20-I-14,15-EE8ZE-APSA relaxed the arterial rings to a similar extent as 14,15-EET with the agonists having similar EC50 values of approximately 2 μM (Figure 2A). Thus, the position of the AZ group did not affect activity. This experiment indicates that the photoprobe 20-I-14,15-EE8ZE-APSA is an EET agonist. These results are consistent with the agonist activity of other sulfonamide analogs of 14,15- and 11,12-EETs (15, 28, 33, 34).
Figure 2.
Agonist activity of 20-I-14,15-EE8ZE-APSA isomers. A. Effect of ortho (o)APSA, meta (m)APSA and para (p)APSA isomers of 20-I-14,15-EE8ZE-APSA and 14,15-EET on vascular tone in bovine coronary arteries. Bovine coronary artery rings was pre-constricted with U46619. Cumulative concentrations (10−9 to 10−5 M) of 14,15-EET or 20-I-14,15-EE8ZE-APSA isomers were added and isometric tension measured. Each value represents the mean ± SEM for n=6–7. B. Inhibition of 20-125I-14,15-EE5ZE specific binding to U937 membrane by 20-I-14,15-EE8ZE-APSA isomers and 14,15-EET. 20-125I-14,15-EE5ZE was incubated with increasing concentrations of 14,15-EET or 20-I-14,15-EE8ZE-APSA isomers for 15 min. Specific bindings were determined in presence or absence of 20 μM 14,15-EE5ZE. Specific binding obtained in the presence of vehicle represents 100% binding. Each value is the mean ± SEM for n=4.
Effect of 20-I-14,15-EE8ZE-APSA on 20-125I-14,15-EE5ZE Binding to Membranes of U937 Cells
A high affinity G protein coupled EET receptor exists on U937 cell membranes. The receptor has been characterized by several EET agonist and antagonist radioligands such as 3H-14,15-EET (agonist), 20-125I-14,15-EE8ZE (agonist), 14,15-EET-phenyl-125I-sulfonamide (agonist) and 20-125I-14,15-EE5ZE (antagonist) (25, 28–30). To test whether the photoprobe 20-I-14,15-EE8ZE-APSA binds to the EET receptor, 20-125I-14,15-EE5ZE was incubated with U937 membranes with and without various concentrations of 20-I-14,15-EE8ZE-APSA isomers or 14,15-EET (Figure 2B). The specific binding of 20-125I-14,15-EE5ZE was determined in presence and absence of 14,15-EE5ZE. The IC50’s were 3.60 nM, 2.4 nM, 1.9 nM and 2.73 nM for mAPSA, oAPSA and pAPSA isomers of 20-I-14,15-EE8ZE-APSA and 14,15-EET respectively. This indicates that 20-I-14,15-EE8ZE-APSA isomers have similar affinities for the U937 receptor site as 14,15-EET despite reduction of two double bonds and modification of carbon-1 and carbon-20 of the 14,15-EET structure. The mAPSA isomer was used for subsequent studies.
Photoaffinity Labeling by 20-125I-14,15-EE8ZE-APSA
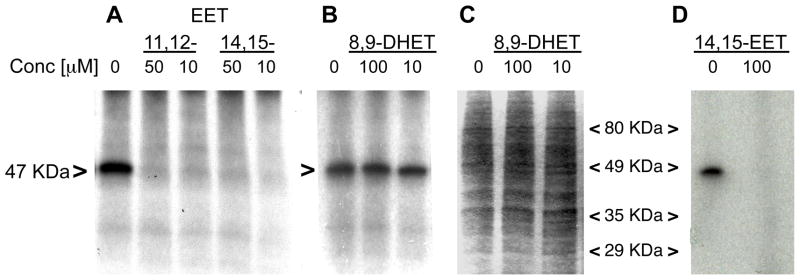
Mixed membranes (Figure 3A–C) and plasma membranes (Figure 3D) of U937 cells were incubated with 20-125I-14,15-EE8ZE-APSA (mAPSA isomer) with and without EETs, cross-linked by exposure to UV light and proteins resolved by electrophoresis. Autoradiograph (Figure 3A) detected the radiolabeling of a single 47 KDa band. The labeling of the 47 KDa protein was inhibited by co-incubation of the photoprobe with 11,12-EET or 14,15-EET. It was not inhibited by 8,9-DHET which was previously shown not to displace 20-125I-14,15-EE5ZE from its binding site (30) (Figure 3B). Coomassie blue staining of the gel shows equal loading of protein with the treatments (Figure 3C). 20-125I-14,15-EE8ZE-APSA similarly labeled a 47 KDa protein in plasma membranes (Figure 3D). The photolabeling was inhibited by co-incubation of the photoprobe with 14,15-EET. These findings suggest specific labeling of a 47 KDa EET binding site by the photoprobe.
Figure 3.
Photoaffinity labeling of U937 cell mixed membranes (A–C) and plasma membranes (D) with 20-125I-14,15-EE8ZE-APSA. 20-125I-14,15-EE8ZE-APSA (mAPSA isomer) (1 nM) was incubated with membranes, photo crosslinking was performed with UV light and proteins were separated by SDS-PAGE. Concentrations of EETs (nM) are indicated. Vehicle was used as a control. The photolabeled 47 KDa bands are indicated with “>”. A: Photolabeling of a 47 KDa protein in mixed membranes and inhibition by EET receptor agonists, 11,12-EET and 14,15-EET. B,C: Effect of 8,9-DHET on photolabeling of the 47 KDa protein in mixed membranes. Autoradiography (B) and Coomassie blue staining (C) of the same gel. (D) Photolabeling of a 47 KDa protein in plasma membranes and inhibition by 14,15-EET.
Characterization of 47 KDa Labled Protein as a High Affinity EET Receptor
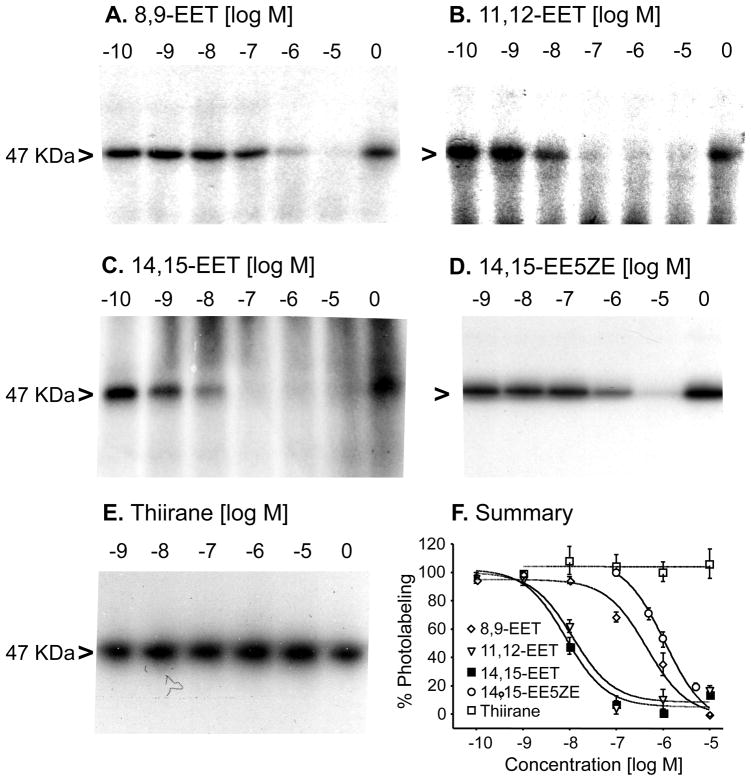
To determine whether the labeled 47 KDa protein is a high affinity binding site for EETs, EET agonists (8,9-EET, 11,12-EET, and 14,15-EET), an EET antagonist (14,15-EE5ZE), and structural similar but biologically inactive analog (14,15-Thiirane) were used to compete for the photoaffinity labeling (see Figure 1 for structures of the compounds) (15, 16). Figure 4A–4E are representative autoradiographs, and Figure 4F is a summary showing the concentration-dependent competition (10−10–10−5M) for photolabeling. 14,15-EET, 11,12-EET, 8,9-EET and 14,15-EE5ZE inhibited the photolabeling of U397 membranes with IC50 values 8.28, 11.2, 443.9, and 1097 nM, respectively (Table 1). The 14,15-thiirane did not alter the photolabeling. It appears that this 47 KDa protein is a high affinity receptor or binding site specifically for 11,12-EET and 14,15-EET, but lower affinity for 8,9-EET and 14,15-EE5ZE.
Figure 4.
Concentration-dependent inhibition of 20-125I-14,15-EE8ZE-APSA photolabeling of a 47 KDa protein in U937 membrane. U937 membrane were incubated with 20-125I-14,15-EE8ZE-APSA and indicated amount of the EET or EET analog following by the photolabeling protocol described in the methods. Labeling of 47 KDa receptor was indicated with “>”. Autoradiographs are shown in A–E. Inhibition of labeling by 8,9-EET (A), 11,12-EET (B), 14,15-EET (C), 14,15-EE5ZE (D) and 14,15-Thiirane analog (E). F is a summary of the inhibition of photolabeling EETs and EET analogs (14,15-EET, solid squares; 11,12-EET, triangles; 8,9-EET, diamond; 14,15-EE5ZE, circles; 14,15-Thiirane open squares)(n = 4).
Table 1.
Inhibition of 20-125I-14,15-EE8ZE-APSA Photolabeling by EETs and EET Analogs
| Competitor | Ki (95% Confidence Intervals) nM |
|---|---|
| 14,15-EET | 8.28 (4.74–14.5) |
| 11,12-EET | 11.2 (5.94–22.9) |
| 8,9-EET | 443.9 (246.6–799.1) |
| 14,15-EE5ZE | 1097 (690–1743) |
| 14,15-Thiirane | > 1×104 |
Structural Unrelated EET Receptor Antagonists Inhibit Labeling of the 47 KDa Protein
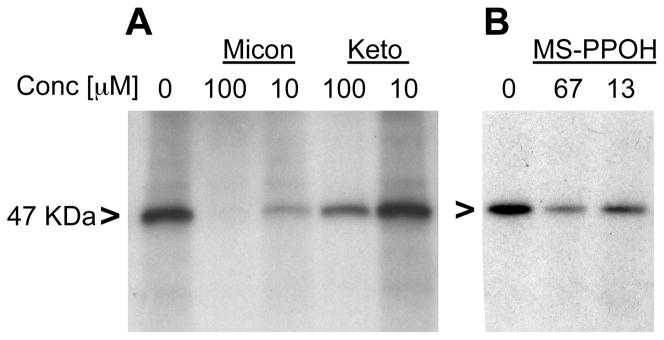
Three cytochrome P450 inhibitors, miconazole, N-(methylsulfonyl-2-(2-propynyloxy)-benzenehexanamide (MS-PPOH), and ketoconazole, also act as EET receptor ligands competing for 20-125I-EE5ZE binding to U937 membranes (30). Both miconazole and MS-PPOH block 14,15-EET-mediated relaxation of arterial rings. Thus, besides inhibiting cytochrome P450, these drugs, like 14,15-EE5ZE, act as EET antagonists. As a result, we tested the effect of these drugs on photolabeling. Miconazole completely blocked the photolabeling by 20-125I-14,15-EE8ZE-APSA while MS-PPOH and ketoconazole partially inhibited the photolabeling (Figure 5). The inhibition by three structurally unrelated EET antagonists further suggests that 47 KDa protein is an EET receptor/binding site.
Figure 5.
Inhibition of photolabeling by 20-125I-14,15-EE8ZE-APSA of the 47 KDa protein in U937 cell membranes by three structurally unrelated EET receptor ligands, miconazole, ketoconazole and MS-PPOH. Autoradiographs indicating: A, inhibition of labeling by various concentrations of miconazole and ketoconazole and B, inhibition of labeling by various concentrations of MS-PPOH. The experiment was repeated four times. 47 KDa protein is indicated with “>”.
Identification of 47 KDa EET Receptor/Binding Site in Vascular Cells and Arteries
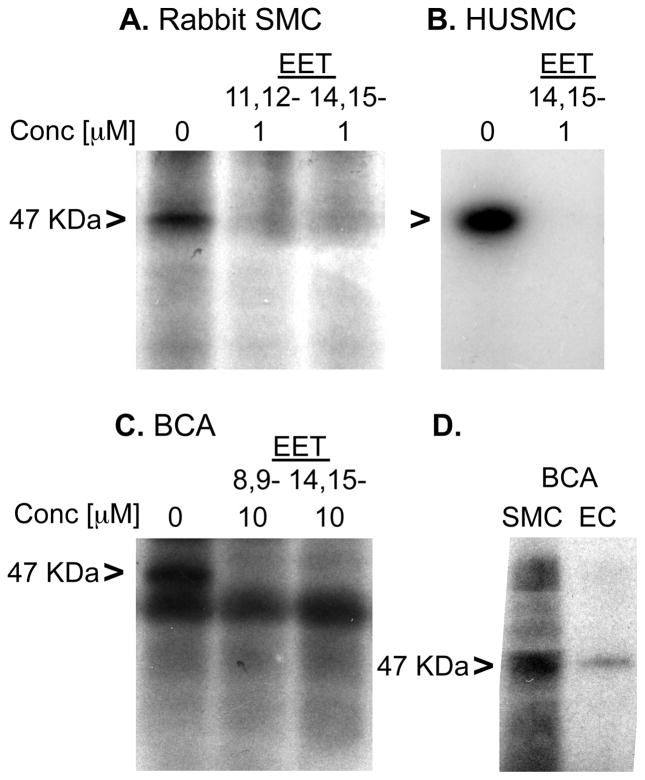
EETs relax vascular smooth muscle (10, 15). As with U937 cell membranes, 20-125I-14,15-EE8ZE-APSA labeled a 47 KDa protein in membranes from rabbit smooth muscle cells (Figure 6A), human smooth muscle cells (Figure 6B) and bovine coronary artery (Figure 6C). The photolabeling was also inhibited by co-incubation with 8,9-EET, 11,12-EET or 14,15-EET. A radiolabeled band was observed below the 47 KDa band in bovine coronary arterial membranes (Figure 6C). This labeling was not changed by co-incubation with 8,9-EET or 14,15-EET so represents a non-specifically labeled protein (Figure 6C). The photolabeling in the bovine coronary arteries appears to be due to the presence of the binding site in both smooth muscle and endothelial cells. Photolabeling of the 47 KDa protein was detected in cultured bovine coronary endothelial cells and smooth muscle cells; however, the expression was much greater in smooth muscle cells than endothelial cells (Figure 6D). The experiments revealed an EET receptor/binding site in smooth muscle cells and coronary arteries and to a lesser extend in endothelial cells. Photolabeling of the 47 KDa protein was also observed in membranes from the canine heart and rat kidney (data not shown).
Figure 6.
Photoaffinity labeling of 47 KDa receptor in vascular cells and tissues. Autoradiography indicating photolabeling in A. Rabbit smooth muscle cell membranes (RSMC), B. Bovine coronary artery membranes (BCA), C. Human smooth muscle cell (HUSMC) and D. Bovine coronary artery endothelial cells and smooth muscle cells. 11,12-EET and 14,15-EET were used to inhibit photolabeling. Autoradiographs are shown. 47 KDa positions are indicated with “>”.
Use of 20-125I-14,15-EE8ZE-APSA to Screen Candidate Receptors
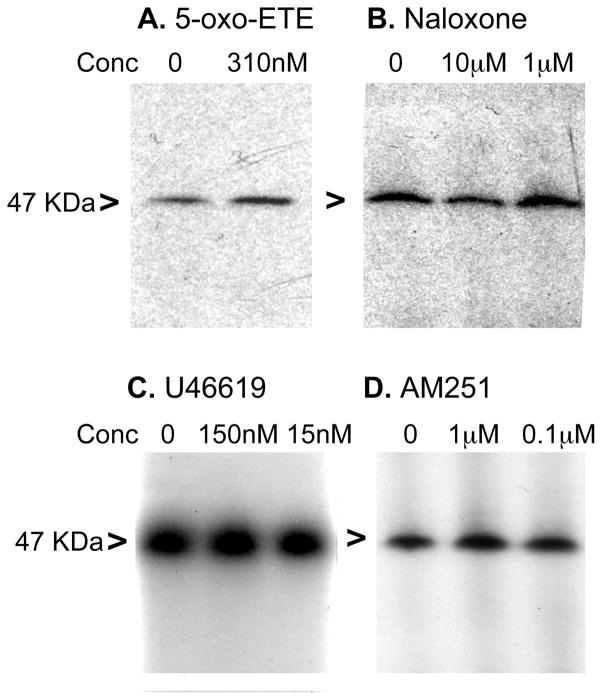
Ligand competition with 20-125I-14,15-EE8ZE-APSA was used to screen receptors known to be present on U937 cell membranes (35–38). Some of these receptors have been suggested to mediate the action of EETs (24, 39). Since the EET receptor has the characteristics of a GPCR (14, 29), only ligands for GPCRs were tested. The photolabeling of a 47 kDa protein in U937 cell membranes by 20-125I-14,15-EE8ZE-APSA was not inhibited by 5-oxo-eicosatetraenoic acid (a 5-oxo-ETE receptor agonist), naloxone (an opioid antagonist), U46619 (a TP receptor agonist) or AM251 (a CB1 receptor antagonist) suggesting these candidate receptors are not EET receptors (Figure 7).
Figure 7.
Effect of candidate receptor agonists and antagonists on photolabeling of U937 membranes with 20-125I-14,15-EE8ZE-APSA. Candidate receptor ligands were used to compete with the photoprobe for labeling of the 47 KDa protein. U937 membrane were labeled with 1 nM of 20-125I-14,15-EE8ZE-APSA with or without (vehicle control) indicated ligands. Autoradiographs are show: A, 5-oxo-ETE, a 5-oxo-ETE receptor agonist. B, Naloxone, a opioid receptor antagonist. C, U46619, a thromboxane (TP) receptor agonist. D, AM251, a cannabinoid (CB)-1 receptor antagonist. 47 KDa protein is indicated with “>”.
GPCRs without known ligands or related to receptors with lipid ligands are likely candidates for the EET receptor (40, 41). The photoaffinity labeling method was used to screen 79 such receptors (Table 2). HEK293T cells were transiently transfected with one of 79 N-terminal FLAG-tagged GPCRs and the expression of receptor protein confirmed by flow cytometry using a FLAG specific antibody. Vector transfected HEK293T cell lyastes were used as a blank control. Each of the 79 GPCRs cell lysates and blank control were incubated with 20-125I-14,15-EE8ZE-APSA, photolabeled and analyzed by radioautography. Lysate from an equal number of U937 cells was used a positive control. No radiolabeled bands were observed in any of the lysates of the 79 GPCR expressing cells or the blank control. However, under identical conditions, U937 cells showed a single 47 KDa radiolabeled band. Thus, photolabeling detected the endogenous EET receptor in U937 cells but did not detect a receptor in HEK293T cells over-expressing GPCRs. Thus, the 79 GPCRs in Table 2 are not the EET receptor labeled by 20-125I-14,15-EE8ZE-APSA.
TABLE 2. GPCRs overexpressed in HEK293T cells and tested for photolabeling with 20-125I-14,15-EE8ZE-APSA.
Orphan Receptors Tested
| ADMR | GPR35 | GPR88 | GPR172A |
| CD97 | GPR37L1 | GPR89 | GPR173 |
| EB12 | GPR40 | GPR97 | GPR174 |
| EMR1 | GPR41 | GPR101 | GPR175 |
| ETLD1 | GPR43 | GPR107 | GPR176 |
| G2A | GPR45 | GPR114 | GPR182 |
| GPR1 | GPR52 | GPR116 | GRCA |
| GPR3 | GPR56 | GPR120 | MAS1 |
| GPR6 | GPR61 | GPR133 | MAS1L |
| GPR12 | GPR62 | GPR135 | MRGD |
| GPR15 | GPR63 | GPR139 | MRGE |
| GPR18 | GPR64 | GPR143 | MRGF |
| GPR19 | GPR75 | GPR146 | MRGX3 |
| GPR21 | GPR78 | GPR148 | MRGX4 |
| GPR22 | GPR80 | GPR151 | OPN3 |
| GPR25 | GPR82 | GPR153 | P2RY8 |
| GPR26 | GPR83 | GPR155 | P2Y10 |
| GPR31 | GPR84 | GPR160 | PSGR |
| GPR32 | GPR85 | GPR161 | RAIG3 |
| GPR34 | GPR87 | GPR171 | TM7SF1 |
ADMR=GPR182; EMR1=EGF-like module containing, mucin-like hormone receptor-like 1; GPR=G-protein coupled receptor; MAS=Mas receptor; MRG=MAS-related GPR; OPN=Opsin; P2RY=Purinergic receptor P2Y; PSGR=Olfactor receptor 51E2; RAIG3=Retinoic acid-induced gene 3, GPR; TM7SF1=Transmembrane 7 superfamily member 1.
DISCUSSION
While many physiological and biochemical activities have been described for the EETs (4, 7, 42–44), the initiation step in the actions of EETs has not been defined. Several lines of evidence implicate a cell surface, high affinity, G-protein coupled receptor as the initial step (17, 26, 29, 30); however, further characterization and identification of this receptor is needed. Photoaffinity labeling has been a useful approach to receptor identification. These studies describe the characterization of the first EET photoaffinity probe, 20-I-14,15-EE8ZE-APSA, and its use to characterize the EET receptor. 20-I-14,15-EE8ZE-APSA relaxed the bovine coronary artery with a similar ED50 as 14,15-EET and displaced the EET antagonist radioligand 20-125I-14,15-EE5ZE from its binding site on U937 cell membranes with an IC50 similar to 14,15-EET. Thus, 20-I-14,15-EE8ZE-APSA is high affinity agonist ligand for the EET binding site/receptor. Importantly, this photoactive ligand can be radiolabeled with high specific activity.
When incubated with U937 membranes and cross-linked with UV light, 20-125I-14,15-EE8ZE-APSA radiolabeled a single protein band of 47 KDa. The radiolabeling of the 47 KDa protein was specific for the EET structure since EET agonists (8,9-EET, 11,12-EET, and 14,15-EET) and the structurally-related EET antagonist (14,15-EE5ZE). In contrast, the inactive EET analogs 14,15-thiirane and 8,9-DHET did not inhibit the radiolabeling. The IC50’s of the EET analogs for inhibition of labeling by 20-125I-14,15-EE8ZE-APSA suggest the 47 KDa protein is a high affinity receptor for 11,12-EET and 14,15-EET but has lower affinity for 8,9-EET and 14,15-EE5ZE. This affinity ranking order, 11,12-EET = 14,15-EET > 8,9-EET=14,15-EE5ZE and no binding for 8,9-DHET and 14,15-thiirane is same as was determined in radioligand binding assays and correlates with agonist activity (15, 29, 30).
Epoxide hydrolase and cytochrome P450 inhibitors were screened for binding to the EET receptor using the 20-125I-14,15-EE5ZE as the radioligand (30). Miconazole and MSPPOH inhibited binding with Ki’s of 350 nM and 1558 nM, respectively. Ketoconazole (50 μM) inhibited binding by 50%. The concentrations of the drugs that inhibited binding of the EET radioligand differed from the concentrations that inhibited cytochrome P450 (30). Thus, inhibition of EET binding was not associated with cytochrome P450 inhibition. Other cytochrome P450 inhibitors such as sulfaphenazole and proadifen and epoxide hydrolase inhibitors did not alter binding of the radioligand. In addition, miconazole and MSPPOH blocked 14,15-EET-induced relaxations of bovine coronary arteries. Proadifen did not alter 14,15-EET relaxations. These studies indicate that MSPPOH and miconazole, besides inhibiting cytochrome P450, act as EET antagonists like 14,15-EE5ZE. Miconazole, MSPPOH, ketoconazole and 14,15-EE5ZE inhibited 20-125I-14,15-EE8ZE-APSA photolabeling of the 47 KDa protein. These data are consistent with our previous conclusion that these drugs are EET antagonists and suggests that the 47 KDa protein is the binding site for 20-125I-14,15-EE5ZE and an EET receptor.
The initial characterization of the EET photoprobe used U937 cell membranes since this cell line has been a model system to study the EET receptor (26, 29, 30). However, the photolabeling of the 47 KDa protein by 20-125I-14,15-EE8ZE-APSA was also observed in membranes from rabbit, bovine and human vascular smooth muscle cells, bovine coronary arterial endothelial cells, bovine coronary artery, canine heart and rat kidney. As with U937 cells, this labeling was inhibited by EET agonists. Since EETs relax vascular smooth muscle (10), inhibit adhesion molecule expression on endothelial cells (4), alter renal tubular and vascular function (45) and cause cardioprotection (8), a protein involved in binding EETs and initiating EET actions would be expected in these cells and tissues.
Several studies indicate the utility of the EET photoprobe in characterizing the EET receptor. These photolabeling experiments reveal for the first time that the EET binding site/receptor has a molecular weight of 47 KDa. This information may be used to eliminate a number of proteins that may bind EETs. For example: CYP2J and CYP2C synthesize EETs, fatty acid binding proteins and PPARs bind EETs and soluble epoxide hydrolase converts EETs to DHETs (4, 18–20, 46). However, these proteins cannot represent the EET binding site labeled by the photoprobe since the molecular weights of CYPs, PPARs and epoxide hydrolase exceed 47 KDa and fatty acid binding proteins are less than 47 KDa. By the same logic, BKCa channel proteins, TRPV4 channels, EP2 receptor and the TP receptor can be eliminated (47–49) despite suggestions that these proteins may function as EET receptors (24, 50).
The photoprobe made EET receptor screening simple and applicable on a relatively large scale. With small amounts of protein, candidate EET receptor proteins can be tested directly for photolabeling. Additionally, competition by specific receptor agonists or antagonists for photolabeling by 20-125I-14,15-EE8ZE-APSA can be used to eliminate other known receptors. For example, AM251 (a CB1 receptor antagonist), 5-oxo-ETE (a 5-oxo-ETE receptor agonist), naloxone (an opioid antagonist) and U46619 (a thromboxane receptor agonist) failed to inhibit photolabeling thereby eliminating these receptors as possible EET receptors. Thus, while high concentrations of EETs may inhibit TP receptors or activate EP2 receptors (23, 24), the TP and EP receptors are not the high affinity EET receptor.
A large number of GPCRs have no known ligand so are termed orphan receptors (40). Many of these GPCRs may bind lipid mediators (41). For this reason, a group of 79 GPCRs that bind lipids, are related phylogenetically to lipid binding receptors or are express in EET-responding tissues were tested for 14,15-EET binding by photolabeling. None of the 79 GPCRs were photolabeled by 20-125I-14,15-EE8ZE-APSA. When U937 membranes were treated in an identical manner at the same time, the photoprobe labeled a 47 KDa protein. Since the method is capable of labeling and detecting the endogenous EET receptor in U937 cells, it should certainly detect an overexpressed GPCR that binds 14,15-EET. Thus, it appears that none of the 79 GPCRs represent the high affinity EET receptor labeled by 20-I-14,15-EE8ZE-APSA. Of particular note, free fatty acid receptors and receptors that bind modified or oxidized fatty acids such as GPR2A, GPR18, GPR40, GPR41, GPR43, GPR84 and GPR120 were not labeled by the photoprobe (51–53).
The photoprobe that is characterized herein is an analog of 14,15-EET and may only label an EET receptor or receptors with high affinity for 14,15-EET and possibly 11,12-EET such as the 47 KDa protein. The failure to detect receptor labeling with 20-125I-14,15-EE8ZE-APSA in HEK293T cells overexpressing orphan GPCRs does not exclude the possibility that these GPCRs are receptors for other EET regioisomers or low affinity EET receptors. Photoprobes for the other EET regioisomers may be needed to identify other EET receptors.
In summary, 20-I-14,15-EE8ZE-APSA is the first EET photoaffinity probe and has EET agonist activity. This photoprobe labeled a 47 KDa high affinity EET binding protein in U937 cells and vascular cells. Since photolabeling was inhibited by several EET agonist (8,9-EET, 11,12-EET, and 14,15-EET) and EET antagonists (14,15-EE5ZE, miconazole, ketoconazole and MS-PPOH), we propose this 47 KDa protein as a high affinity EET receptor. Used in a same way as an antibody, the photoprobe could be a molecular marker for the EET receptor in normal cells and tissues and in disease and improve our understanding of the expression and regulation of the EET receptor.
Supplementary Material
Acknowledgments
This work was supported by a grant from the National Heart, Lung and Blood Institute (HL-51055), the National Institute of General Medical Sciences (GM-31278) and the Robert A. Welch Foundation (GL-625910)
We thank Mr. Daniel Goldman, Ms. Sarah Christian and Mr. Michael Szadkowski for their technical assistance and Mrs. Gretchen Barg for her secretarial assistance. The authors thank Dr. Kathryn M. Gauthier for the helpful discussions and assistance with the figures. We also thank Helena Mancebo, Ph.D. and Multispan Inc. (Hayward, California) for GPCR overexpressing HEK293T cells.
ABBREVIATIONS
- EET
epoxyeicosatrienoic acid
- CYP
cytochrome P450
- 14,15-EE8ZE
14,15-cis-epoxy-eicosa-8(Z)-enoic acid
- 14,15-EE5ZE
14,15-cis-epoxy-eicosa-5(Z)-enoic acid
- 20-I-14,15-EE8ZE-APSA
20-iodo-14,15-epoxyeicosa-8Z-enoyl-3-azidophenylsulfonamide
- 8,9-DHET
8,9-dihydroxyeicosatrienoic acid
- MSPPOH
N-(methylsulfonyl-2-(2-propynyloxy)-benzenehexanamide
- U46619
9-11-dideoxy-11α,9a-epoxymethano-prostaglandin F2α
- EDRF
endothelium-derived relaxation factor
- EDHF
endothelium-derived hyperpolarizing factor
- 20-I-14,15-EE8ZE
20-iodo-14,15-epoxyeicosa-8(Z)-enoic acid
- OTs
20-tosyl
- HPLC
high-performance liquid chromatography
- G protein
guanine nucleotide binding protein
- GPCR
G protein-coupled receptor
- BKCa
large-conductance Ca2+ -activated K+ channel
- TP
thromboxane prostanoid receptor
- EP
prostaglandin E prostanaoid receptor
- CB
cannabinoid
Footnotes
Methods for the synthesis of 20-OTs-14,15-EE8ZE-APSA and 20-I-14,15-EE8ZE-APSA are provided as online supplemental material. It is accessible free of charge at the following address (http://pubs.acs.org).
References
- 1.Capdevila J, Chacos N, Werringloer J, Prough RA, Estabrook RW. Liver microsomal cytochrome P450 and the oxidative metabolism of archidonic acid. Proc Natl Acad Sci USA. 1981;78:5362–5366. doi: 10.1073/pnas.78.9.5362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fitzpatrick FA, Murphy RC. Cytochrome P-450 metabolism of arachidonic acid: Formation and biological actions of “epoxygenase”-derived eicosanoids. Pharmacol Reviews. 1988;40:229–241. [PubMed] [Google Scholar]
- 3.Zeldin DC. Epoxygenase pathways of arachidonic acid metabolism. J Biol Chem. 2001;276:36059–36062. doi: 10.1074/jbc.R100030200. [DOI] [PubMed] [Google Scholar]
- 4.Node K, Huo Y, Ruan X, Yang B, Spiecker M, Ley K, Zeldin D, Liao J. Anti-inflammatory properties of cytochrome P450 epoxygenase-derived eicosanoids. Science. 1999;285:1276–1279. doi: 10.1126/science.285.5431.1276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Munzenmaier DH, Harder DR. Cerebral microvascular endothelial cell tube formation: Role of astrocytic epoxyeicosatrienoic acid release. Am J Physiol. 2000;278:H1163–1167. doi: 10.1152/ajpheart.2000.278.4.H1163. [DOI] [PubMed] [Google Scholar]
- 6.Krotz F, Riexinger T, Buerkle MA, Nithipatikom K, Gloe T, Sohn HY, Campbell WB, Pohl U. Membrane potential-dependent inhibition of platelet adhesion to endothelial cells by epoxyeicosatrienoic acids. Arterioscler Thromb Vasc Biol. 2003;24:595–600. doi: 10.1161/01.ATV.0000116219.09040.8c. [DOI] [PubMed] [Google Scholar]
- 7.Larsen BT, Campbell WB, Gutterman DD. Beyond vasodilation: Non-vasomotor roles of epoxyeicosatrienoic acids in the cardiovascular system. TIPS. 2007;28:32–38. doi: 10.1016/j.tips.2006.11.002. [DOI] [PubMed] [Google Scholar]
- 8.Gross GJ, Gauthier KM, Moore J, Falck JR, Hammock BD, Campbell WB, Nithipatikom K. Effects of the selective EET antagonist, 14,15-EEZE, on cardioprotection produced by exogenous and endogenous EETs in the canine heart. Am J Physiol. 2008;294:H2838–H2844. doi: 10.1152/ajpheart.00186.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Terashvili M, Tseng LF, Wu HE, Narayanan J, Hart LM, Falck JR, Pratt PF, Harder DR. Antinociception produced by 14,15-epoxyeicosatrienoic acid is mediated by the activation of beta-endorphin and met-enkephalin in the rat ventrolateral periaqueductal gray. J Pharmacol Exp Ther. 2008;326:614–622. doi: 10.1124/jpet.108.136739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Campbell WB, Gebremedhin D, Pratt PF, Harder DR. Identification of epoxyeicosatrienoic acids as endothelium-derived hyperpolarizing factors. Circ Res. 1996;78:415–423. doi: 10.1161/01.res.78.3.415. [DOI] [PubMed] [Google Scholar]
- 11.Fisslthaler B, Popp R, Kiss L, Potente M, Harder DR, Fleming I, Busse R. Cytochrome P450 2C is an EDHF synthase in coronary arteries. Nature. 1999;401:493–497. doi: 10.1038/46816. [DOI] [PubMed] [Google Scholar]
- 12.Campbell WB, Falck JR. Arachidonic acid metabolites as endothelium-derived hyperpolarizing factors. Hypertension. 2007;49:590–596. doi: 10.1161/01.HYP.0000255173.50317.fc. [DOI] [PubMed] [Google Scholar]
- 13.Campbell WB, Fleming I. Epoxyeicosatrienoic acids and endothelium-dependent responses. Pflugers Arch. 2010;459:881–895. doi: 10.1007/s00424-010-0804-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Li PL, Campbell WB. Epoxyeicosatrienoic acids activate potassium channels in coronary smooth muscle through guanine nucleotide binding protein. Circ Res. 1997;80:877–884. doi: 10.1161/01.res.80.6.877. [DOI] [PubMed] [Google Scholar]
- 15.Falck JR, Krishna UM, Reddy YK, Kumar PS, Reddy KM, Hittner SB, Deeter C, Sharma KK, Gauthier KM, Campbell WB. Comparison of the vasodilatory properties of 14,15-EET analogs: Structural requirements for dilation. Am J Physiol. 2003;284:H337–H349. doi: 10.1152/ajpheart.00831.2001. [DOI] [PubMed] [Google Scholar]
- 16.Gauthier KM, Deeter C, Krishna UM, Reddy YK, Bondlela M, Falck JR, Campbell WB. 14,15-Epoxyeicosa-5(Z)-enoic acid: A selective epoxyeicosatrienoic acid antagonist that inhibits endothelium-dependent hyperpolarization and relaxation in coronary arteries. Circ Res. 2002;90:1028–1036. doi: 10.1161/01.res.0000018162.87285.f8. [DOI] [PubMed] [Google Scholar]
- 17.Snyder GD, Krishna UM, Falck JR, Spector AA. Evidence for a membrane site of action for 14,15-EET on expression of aromatase in vascular smooth muscle. Am J Physiol. 2002;283:H1936–H1942. doi: 10.1152/ajpheart.00321.2002. [DOI] [PubMed] [Google Scholar]
- 18.Widstrom RL, Norris AW, Spector AA. Binding of cytochrome P450 monooxygenase and lipoxygenase pathway products by heart fatty acid-binding protein. Biochemistry. 2001;40:1070–1076. doi: 10.1021/bi001602y. [DOI] [PubMed] [Google Scholar]
- 19.Cowart LA, Wei S, Hsu MH, Johnson EF, Krishna UM, Falck JR, Capdevila J. The CYP4A isoforms hydroxylate epoxyeicosatrienoic acids to form high affinity peroxisome proliferator-activated receptor ligands. J Biol Chem. 2002;20:35105–35112. doi: 10.1074/jbc.M201575200. [DOI] [PubMed] [Google Scholar]
- 20.Liu Y, Zhang Y, Schmelzer K, Lee TS, Fang X, Zhu Y, Spector AA, Gill S, Morisseau C, Hammock BD, Shyy JYJ. The antiinflammatory effect of laminar flow: the role of PPARγ. epoxyeicosatrienoic acids and soluble epoxide hydrolase. Proc Nat Acad Sci USA. 2005;102:16747–16752. doi: 10.1073/pnas.0508081102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Inceoglu B, Schmelzer KR, Morisseau C, Jinks SL, Hammock BD. Soluble epoxide hydrolase inhibition reveals novel biological functions of epoxyeicosatrienoic acids (EETs) Prostag and Other Lipid Mediat. 2007;82:42–49. doi: 10.1016/j.prostaglandins.2006.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lu T, Ye D, Wang X, Seubert JM, Graves JP, Bradbury A, Zeldin DC, Lee HC. Cardiac and vascular Katp channels in rats are activated by endogenous epoxyeicosatrienoic acids through different mechanisms. J Physiol. 2006;575:627–644. doi: 10.1113/jphysiol.2006.113985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yang C, Kwan YW, Au AL, Poon CC, Zhang Q, Chan SW, Lee SM, Leung GP. 14,15-Epoxyeicosatrienoic acid induces vasorelaxation through prostaglandin EP(2) receptors in rat mesenteric artery. Prostag and Other Lipid Mediat. 2010;93:44–51. doi: 10.1016/j.prostaglandins.2010.06.004. [DOI] [PubMed] [Google Scholar]
- 24.Behm DJ, Ogbonna A, Wu C, Burns-Kurtis CL, Douglas SA. Epoxyeicosatrienoic acids function as selective, endogenous antagonists of native thromboxane receptors: Identification of a novel mechanism of vasodilation. J Pharmacol Exp Ther. 2009;328:231–239. doi: 10.1124/jpet.108.145102. [DOI] [PubMed] [Google Scholar]
- 25.Wong PY, Lin KT, Yan YT, Ahern D, Iles J, SYS, Bhatt RK, Falck JR. 14(R), 15(S)-Epoxyeicosatrienoic acid receptor in guinea pig mononuclear cell membranes. J Lipid Mediat Cell Signal. 1993;6:199–208. [PubMed] [Google Scholar]
- 26.Wong PYK, Lai PS, Shen SY, Belosludtsev YY, Falck JR. Post-receptor signal transduction and regulation of 14(R), 15(S)-epoxyeicosatrienoic acid (14,15-EET) binding in U-937 cells. J Lipid Med Cell Signal. 1997;16:155–169. doi: 10.1016/s0929-7855(97)00005-9. [DOI] [PubMed] [Google Scholar]
- 27.Wong PYK, Lai PS, Falck JR. Mechanism and signal transduction of 14(R), 15 (S)-epoxyeicosatrienoic acid (14,15-EET binding in guinea pig monocytes. Prostag Other Lipid Med. 2000;62:321–333. doi: 10.1016/s0090-6980(00)00079-4. [DOI] [PubMed] [Google Scholar]
- 28.Yang W, Holmes BB, Gopal VR, Kishore RVK, Sangras B, Yi XY, Falck JR, Campbell WB. Characterization of 14,15-epoxyeicosatrienoyl-sulfonamides as 14,15-epoxyeicosatrienoic acid agonists: Use for studies of metabolism and ligand binding. J Pharmacol Exp Ther. 2007;321:1023–1031. doi: 10.1124/jpet.107.119651. [DOI] [PubMed] [Google Scholar]
- 29.Yang W, Tuniki VR, Anjaiah S, Falck JR, Hillard CJ, Campbell WB. Characterization of epoxyeicosatrienoic acid binding site in U937 membranes using a novel radiolabeled agonist, 20–125I-14,15-epoxyeicosa-8(Z)-enoic acid. J Pharmacol Exp Ther. 2008;324:1019–1027. doi: 10.1124/jpet.107.129577. [DOI] [PubMed] [Google Scholar]
- 30.Chen Y, Falck JR, Tuniki VR, Campbell WB. 20–125Iodo-14,15-epoxyeicosa-5Z-enoic acid: a high affinity radioligand used to characterize the epoxyeicosatrienoic acid antagonist binding site. J Pharmacol Exp Ther. 2009;331:1137–1145. doi: 10.1124/jpet.109.157818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Revtyak GE, Johnson AR, Campbell WB. Cultured bovine coronary arterial endothelial cells synthesize HETEs and prostacyclin. Am J Physiol. 1988;254:C8–C19. doi: 10.1152/ajpcell.1988.254.1.C8. [DOI] [PubMed] [Google Scholar]
- 32.Campbell WB, Deeter C, Gauthier KM, Ingraham RH, Falck JR, Li PL. 14,15-Dihydroxyeicosatrienoic acid relaxes bovine coronary arteries by activation of KCa channels. Am J Physiol. 2002;282:H1656–H1664. doi: 10.1152/ajpheart.00597.2001. [DOI] [PubMed] [Google Scholar]
- 33.Imig JD, Inscho EW, Deichmann PC, Reddy KM, Falck JR. Afferent arteriolar vasodilation to the sulfonimide analog of 11,12-epoxyeicosatrienoic acid involves protein kinase A. Hypertension. 1999;33:408–413. doi: 10.1161/01.hyp.33.1.408. [DOI] [PubMed] [Google Scholar]
- 34.Gauthier KM, Falck JR, Reddy LM, Campbell WB. 14,15-EET analogs: Characterization of structural requirements for agonist and antagonist activity in bovine coronary arteries. Pharmacol Res. 2004;49:515–524. doi: 10.1016/j.phrs.2003.09.014. [DOI] [PubMed] [Google Scholar]
- 35.Bouaboula M, Rinaldi M, Carayon P, Carillon C, Delpech B, Shire D, LeFur G, Caselias P. Cannabinoid-receptor expression in human leukocytes. Europ J Biochem. 1993;214:173–180. doi: 10.1111/j.1432-1033.1993.tb17910.x. [DOI] [PubMed] [Google Scholar]
- 36.Erlemann KR, Rokash J, Powell WS. Oxidative stress stimulates the synthesis of the eosinophil chemoattractant 5-oxa-6,8,11,14-eicosatetraenoic acid by inflammatory cells. J Biol Chem. 2004;279:40376–40384. doi: 10.1074/jbc.M401294200. [DOI] [PubMed] [Google Scholar]
- 37.Royal W, Leander MV, Bissonnette R. Retinoid-induced mu opioid receptor expression by phytohemagglutinin-stimulated U937 cells. J Neurovirol. 2005;11:157–165. doi: 10.1080/13550280590922766. [DOI] [PubMed] [Google Scholar]
- 38.Chen JK, Chen J, Imig JD, Wei S, Hachey DL, Guthi JS, Falck JR, Capdevila JH, Harris RC. Identification of novel endogenous cytochrome P450 arachidonate metabolites with high affinity for cannabinoid receptors. J Biol Chem. 2008;283:24514–24524. doi: 10.1074/jbc.M709873200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Gross GJ, Baker JE, Hsu AK, Wu HE, Falck JR, Nithipatikom K. Evidence for a role of opioids in epoxyeicosatrienoic acid-induced cardioprotection in rat hearts. Am J Physiol. 2010;298:H2201–2207. doi: 10.1152/ajpheart.00815.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wise A, Jupe SC, Rees S. The identification of ligands at orphan G-protein coupled receptors. Annu Rev Pharmacol Toxicol. 2004;44:43–66. doi: 10.1146/annurev.pharmtox.44.101802.121419. [DOI] [PubMed] [Google Scholar]
- 41.Im DS. Discovery of new G protein-coupled receptors for lipid mediators. J Lipid Res. 2004;45:410–418. doi: 10.1194/jlr.R300006-JLR200. [DOI] [PubMed] [Google Scholar]
- 42.Chen JK, Capdevila J, Harris RC. Cytochrome P450 epoxygenase metabolism of arachidonic acid inhibits apoptosis. Mol Cell Biol. 2001;21:6322–6331. doi: 10.1128/MCB.21.18.6322-6331.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chen JK, Falck JR, Reddy KM, Capdevila J, Harris RC. Epoxyeicosatrienoic acids and their sulfonimide derivatives stimulate tyrosine phosphorylation and induce mitogenesis in renal epithelial cells. J Biol Chem. 1998;273:29254–29261. doi: 10.1074/jbc.273.44.29254. [DOI] [PubMed] [Google Scholar]
- 44.Fleming I, Fissllthaler B, Michaelis UR, Kiss L, Popp R, Busse R. The coronary endothelium-derived hyperpolarizing factor (EDHF) stimulates multiple signalling pathways and proliferation of vascular cells. Pfluegers Arch. 2001;442:511–518. doi: 10.1007/s004240100565. [DOI] [PubMed] [Google Scholar]
- 45.Imig JD. Epoxide hydrolase and epoxygenase metabolites as therapeutic targets for renal diseases. Am J Physiol. 2005;289:F496–F503. doi: 10.1152/ajprenal.00350.2004. [DOI] [PubMed] [Google Scholar]
- 46.Spector AA, Fang X, Snyder GD, Weintraub NL. Epoxyeicosatrienoic acids (EETs): Metabolism and biochemical function. Prog Lipid Res. 2004;43:55–90. doi: 10.1016/s0163-7827(03)00049-3. [DOI] [PubMed] [Google Scholar]
- 47.Knaus HG, Eberhart A, Koch ROA, Munujos P, Schmalhofer WA, Warmke JW, Kaczorowski GJ, Garcia ML. Characterization of tissue-expressed a subunits of the high conductance Ca2+-activated K+ channel. J Biol Chem. 1995;270:22434–22439. doi: 10.1074/jbc.270.38.22434. [DOI] [PubMed] [Google Scholar]
- 48.Nilius B, Vriens J, Prenen J, Droogmans G, Voets T. TRPV4 calcium entry channel: a paradigm for gating diversity. Am J Physiol. 2004;286:C195–C205. doi: 10.1152/ajpcell.00365.2003. [DOI] [PubMed] [Google Scholar]
- 49.Pfister SL, Kotulock DA, Campbell WB. Vascular smooth muscle thromboxane A2 receptors mediate arachidonic acid-induced sudden death in rabbits. Hypertension. 1997;29:303–309. doi: 10.1161/01.hyp.29.1.303. [DOI] [PubMed] [Google Scholar]
- 50.Earley S, Heppner TJ, Nelson MT, Brayden JE. TRPV4 forms a novel Ca2+ signaling complex with ryanodine receptors and BK Ca channels. Circ Res. 2005;97:1270–1279. doi: 10.1161/01.RES.0000194321.60300.d6. [DOI] [PubMed] [Google Scholar]
- 51.Obinata H, Hattori T, Nakane S, Tatei K, Izumi T. Identification of 9-hydroxyoctadecadienoic acid and other oxidized free fatty acids as ligands of teh G protein-coupled receptor G2A. J Biol Chem. 2005;280:40676–40683. doi: 10.1074/jbc.M507787200. [DOI] [PubMed] [Google Scholar]
- 52.Wang J, Wu X, Simonavicius N, Tian H, Ling L. Medium-chain fatty acids as ligands for orphan G protein-coupled receptor GPR84. J Biol Chem. 2006;281:34457–34464. doi: 10.1074/jbc.M608019200. [DOI] [PubMed] [Google Scholar]
- 53.Morgan NG, Dhayal S. G-protein coupled receptors mediating long chain fatty acid signalling in pancreatic beta-cell. Biochem Pharmacol. 2009;78:1419–1427. doi: 10.1016/j.bcp.2009.07.020. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.