Deficiency for insulin-like 3 receptor RXFP2 causes an increase in uterine cyst formation in mice with deletion of Caveolin-1 gene.
Abstract
Gene mutations of insulin-like 3 (INSL3) peptide or its G protein-coupled receptor RXFP2 (relaxin family peptide receptor 2) lead to cryptorchidism. The role of INSL3 in adult females is less known, although INSL3 expression has been described in female reproductive organs. Caveolin-1 (CAV1), the main component of caveoli cell membrane invaginations, has been shown to play an important role in epithelial organization and stromal-epithelial interactions. We created a null allele of Cav1 mice by deleting its second exon through embryonic stem cell targeting. Immunohistochemical analysis demonstrated that CAV1 expression was primarily localized to endothelial blood vessel cells and the myometrium uterus, whereas the strongest expression of Rxfp2 was detected in the endometrial epithelium. By 12 months of age approximately 18% of Cav1−/− females developed single or multiple dilated endometrial cysts lined by a flattened, simple low epithelium. A deficiency for Rxfp2 on Cav1-deficient background led to more than a 2-fold increase in the incidence of uterine cysts (54–58%). Appearance of cysts led to a severe disorganization of uterine morphology. We have found that the cysts had an increased expression of β-catenin and estrogen receptor β in endometrial stromal and epithelial cells and increased epithelial proliferation. An analysis of simple dilated cysts in human patients for CAV1 expression did not show appreciable differences with control regardless of menstrual phase, suggesting an involvement of additional factors in human disease. The results of this study suggest a novel synergistic role of INSL3/RXFP2 and CAV1 in structural maintenance of the uterus.
Insulin-like 3 (INSL3) is a short peptide hormone that belongs to the insulin-relaxin superfamily. It is produced in embryonic and adult testicular Leydig cells and secreted into the blood. The INSL3 hormone plays a crucial role in transabdominal testicular descent inducing gubernacular differentiation (1). INSL3 signals through a G protein-coupled receptor RXFP2 (relaxin family peptide receptor 2) (2, 3). Deletion of INSL3 or its receptor RXFP2 in mutant mice causes high intraabdominal cryptorchidism (4–7). In some human populations, heterozygosity for the functionally deficient RXFP2 mutant allele is associated with cryptorchidism, suggesting gene dose dependence (8). In adult mouse testis, INSL3 has a protective antiapoptotic effect on male germ cells under GnRH antagonist-induced stress (9). The hormone was also implicated in osteoblast differentiation and bone metabolism in males (10). Additional functions in thyroid and prostate cancer progression have been recently described (11–14).
The role of INSL3 in females is much less defined. In female mice, the INSL3 mRNA first appears in 6-d-old ovary with INSL3 immunoreactivity detected in luteal cells (15). In human, cow, deer, and sheep, the INSL3 gene expression was detected at high levels in theca interna cells and in antral follicles (16–19). The RXFP2 expression has also been detected in various female reproductive tissues; however, the only phenotype described in INSL3-deficient mice was impaired fertility associated with an extension of the estrous cycle length (5), accelerated follicular atresia, luteolysis, and premature decay of corpora lutea, likely due to increased apoptosis (20). Correlation of INSL3 serum levels in women with polycystic ovarian syndrome related to LH and ovarian androgenic function has been described, specifically in normal-weight women with polycystic ovarian syndrome (21). We have shown that the human RXFP2 is well expressed in both normal myometrium and uterine leiomyoma (22).
Caveolin-1 (CAV1) is a principal structural component of caveolae, the small invaginations of the plasma membranes. The caveolae are present in majority of nonmitotic terminally differentiated cells, with numerous cell signaling molecules located within these structures in either a scaffolding binding-dependent or -independent manner. The role of caveolae and CAV1 has been extensively investigated over the last 20 yr in both healthy and neoplastic tissues. Four different groups produced targeted mutant alleles with deletion of the first two exons (23, 24), middle (25), or the last exon of Cav1 (26). Surprisingly, despite a wide expression of the gene and its important role in signal transduction and molecule trafficking, mice with the complete ablation of Cav1 were viable and fertile. Thorough analysis revealed numerous abnormalities in vascular, respiratory, muscle, and other systems of CAV1-deficient mice. Many of these defects were age dependent, manifesting only in older animals. A tumor suppressor role of CAV1 was suggested for some cancer and benign tumors (27). For example, Yang et al. (28) demonstrated that mice with Cav1 disruption gradually developed benign stromal lesions and compromised epithelial differentiation in several organs including uterus and ovary. Thus, we decided to investigate the effect of simultaneous deletion of CAV1 and RXFP2 on female reproductive organs.
Hyperplasia in the endometrium is a term applied when the ratio of glands to stroma has increased when compared with normal proliferative endometrium. The glands may show varying degrees of cystic dilation and mild to moderate crowding. This is in contrast to complex hyperplasia where the glands tend to be branched with prominent papillary infoldings. Cytological atypia is not a finding frequently associated with simple hyperplasia. The spectrum of hyperplasia in the endometrium is attributed most often to prolonged and persistent exposure to unopposed estrogen (29). Several mouse strains are prone to the development of uterine cysts in advanced age (30).
We decided to investigate the effect of simultaneous deletion of CAV1 and RXFP2 on female reproductive organs. We describe here a new knockout allele of Cav1 in mice. Analysis of the mutant females revealed dilated endometrial cysts in older animals. The frequency was significantly increased in animals with deletion of one or two alleles of Rxfp2 gene, suggesting synergetic effect of two genetic abnormalities. This study indicates a possible role of INSL3 in proper maintenance of female reproductive tract.
Materials and Methods
Production of Cav1 mutant mice
The animal studies were approved by the Baylor College of Medicine and Florida International University Institutional Committees on Animal Care and Use Committees in accordance with the principles and procedures outlined in the National Research Council publication Guide for Care and Use of Laboratory Research Involving Animals under Protocols 1835 and 4073.
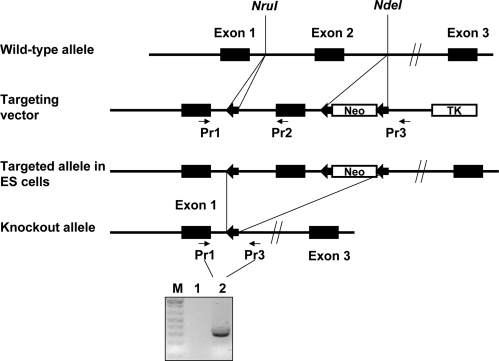
To produce a mouse with a mutant allele of the Cav1 gene, we used Cre/loxP approach (Fig. 1). A genomic clone containing promoter region and two exons and introns of the gene was isolated from 129 mouse genomic λ-phage library. The NruI and NdeI restriction sites within first and second introns were used for integration of loxP and floxed neocassette. The resulted construct was inserted into pBluescriptII SK vector 5′ to the herpes virus thymidine kinase (HSVtk) minigene driven by synthetic mutant polyoma-enhanced MC1 enhancer/promoter cassette. The vector was linearized with NotI and electroporated into AB2.1 embryonic stem (ES) cells. Recombinant clones were identified by PCR, with primers derived from the vector backbone (PGK promoter) and from the genomic sequence outside the targeting construct. To induce recombination, the ES clones were electroporated with a pCMV-Cre plasmid expressing CRE recombinase. The recombinant clones with deletion of second exon and floxed neocassette were identified using PCR with primers on both sides of the loxP sequences: Pr1, 5′-TAAATCACAGCCCAGGGAAA-3′; and Pr3, 5′-TGCACATGTGCCATGTTTCTAT-3′(Fig. 1). Three independent ES clones were injected into C57BL/6J blastocysts, which were implanted into pseudopregnant female mice using standard procedures in the Baylor College of Medicine Transgenic Core. Chimeric males were bred to C57BL/6J females to produce mice heterozygous for the targeted allele. The mutant animals were interbred to homozygosity for the mutant allele. Presence of the Cav1 mutant allele was detected by PCR from ear DNA with Pr1 and Pr3 primers; the wild-type allele was identified with primers Pr1 and Pr2, 5′-GACCACGTCGTCGTTGAGAT-3′ (Fig. 1).
Fig. 1.
Generation of mice with a mutant allele of the Cav1 gene. A targeting construct was prepared by insertion of loxP in NruI and floxed Neo cassette in NdeI restriction sites on both sides of exon 2. Neo, Neomycin resistance cassette; TK, herpes virus thymidine kinase minigene; the loxP sites are shown as the thick arrows; black boxes represent Cav1 exons. The wild-type chromosome is shown on top. After homologous integration the targeting construct is integrated into genomic DNA. After Cre-mediated recombination in ES cells, the knockout allele was produced (bottom). To verify the deletion of the second exon in ES clones, we used genomic PCR with primers located on both sides of the floxed region. The mutant allele (Cav1-) was detected through successful amplification of DNA fragment (lane 2), whereas there was no amplification in wild-type genomic DNA (lane 1). M, 1 Kb marker.
Mouse breeding
We used two other mutant strains in this study. Crsp mice had a 500+ kb deletion in the distal part of chromosome 5, which includes Rxfp2 gene (6). This mutant was derived on FVB inbred strain. A targeted mutation of Rxfp2 gene (Rxfp2tm1Aia, or Rxfp2− thereafter) was described previously (7) and was backcrossed to FVB mice for more than 15 generations. Homozygous crsp/crsp or Rxfp2−/− females were crossed to Cav1−/− males, and the resulted F1 diheterozygotes were backcrossed with Cav1−/−. The Cav1−/− animals were selected by PCR with Pr1 and Pr3 (see above). The presence of crsp or Rxfp2tm1Aia allele was analyzed by ear DNA PCR with primers TyEx1 and TyEx2 derived from tyrosinase minigene construct present in both mutations (6, 7). To obtain homozygous Cav1−/−, crsp/crsp mice, the crsp/+, Cav1−/− animals were bred inter se. The crsp/crsp females were identified by the presence of white spotting (6). The Cav1−/− males and females were fully viable and had normal fertility within the first 2–8 months when they were used for breeding. No apparent difference in fertility was detected between Cav1−/− females and crsp/+, Cav1−/− or Rxfp2−/+, Cav1−/− during the same breeding period (data not shown).
RNA isolation and RT-PCR
RNA was extracted with the TRIzol reagent (Life Technologies, Inc., Gaithersburg, MD). First-strand cDNA was synthesized using the oligo(dT) primer and RETROscript kit (Ambion, Inc., Austin, TX). The following primers were used for RT-PCR analysis of Cav1 expression: Pr1 and Cav1Ex3R, 5-TCCCTTCTGGTTCTGCAATC-3′.
β-Galactosidase staining
Rxfp2-icre mice with iCre transgene driven by Rxfp2 promoter (31) were crossed with R26R reporter strain to generate Rarb-icre, R26R mice. The uteri of 6-wk-old females were fixed in fresh 0.2% paraformaldehyde in PBS overnight at 4 C. The following day organs were rinsed two times in PBS and washed overnight at 4 C in PBS with 2 mm MgCl2 and 30% sucrose. Uteri were embedded in ornithine carboxytransferase compound and rapidly frozen in the −80 C freezer. Cryostat sections (15–20 μm) were prepared and postfixed for 5 min in 4% paraformaldehyde in PBS buffer. Sections were washed twice with PBS and subjected to X-Gal staining overnight at 37 C using Senescence β-Galactosidase Staining kit (Cell Signaling Technology, Inc., Danvers, MA).
Human patient samples
Archived human patient samples of normal proliferative and secretory endometrium (five of each) as well as those with simple hyperplasia without atypia or endometrial intraepithelial neoplasia, in curetting, were selected from the collection of Mount Sinai Medical Center Pathology Department. Dilated endometrial glands or cysts similar to what was observed in the mouse samples were present. Samples were chosen with this change demonstrated across the spectrum of normal and benign hyperplastic endometrium. The Institutional Review Board approved these studies.
Histology and immunohistochemistry
At the time of euthanasia the female reproductive organs were isolated and analyzed for the presence of neoplastic lesions or tumors. The tissues were fixed in 4% paraformaldehyde, paraffin embedded, sectioned, and stained with hematoxylin and eosin using standard protocols. The immunohistochemistry (IHC) was performed using the following antibodies: rabbit polyclonal to CAV1 (1:150; Santa Cruz Biotechnology, Inc., Santa Cruz, CA), rabbit polyclonal to β-catenin (1:1000; Abcam, Cambridge, MA), rabbit polyclonal to estrogen receptor α, estrogen receptor β, and to Ki67 (1:100, 1:300, and 1:1000 accordingly; Thermo Scientific, Rockford, IL). Detection was performed using a Vectastain ABC (avidin-biotin-peroxidase) kit (Vector Laboratories, Burlingame, CA) as recommended. The color was developed with diaminobenzidine as chromogen. Samples were counterstained with Harris Hematoxylin. Stained slides were examined with an Axio A1 Microscope (Carl Zeiss, Thornwood, NY), and images were captured by an AxioCam MRc5 charge-coupled device camera. The analysis of IHC expression was performed on three samples of each genotype in at least 10 different viewpoints and was examined by two investigators blindly. The intensity and the percent of positively stained cells were taken into consideration when the final staining was determined for the samples.
The proliferation in endometrial epithelial cells of the in WT, Cav1−/−, and Cav1−/−, Rxfp2−/+ females (at least three per group) was evaluated in five random fields with at least 500 cells counted at ×1000 magnification.
Statistical analysis
The frequency of the abnormal uteri in different groups was compared using two-sided Fisher's exact test. Differences were considered significant at P < 0.05.
Results
Production and characterization of Cav1 mutant mice
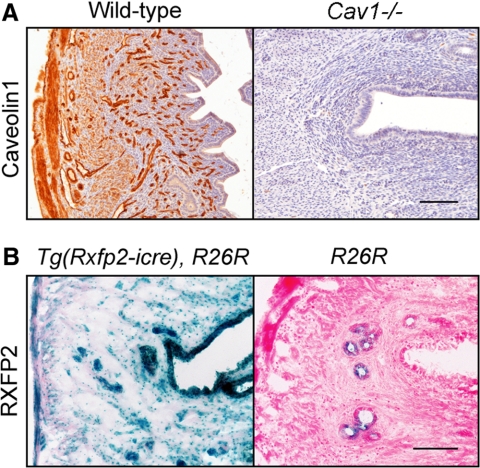
To produce mice with Cav1 deficiency, we used ES cells gene targeting. Our targeting construct was designed to introduce loxP sites on both sides of the second exon of Cav1 gene. Eight ES clones with homologous recombination were obtained; the correct integration of the targeting construct was verified by PCR using primers specific for both the construct and the Cav1 genomic sequence outside the vector. After Cre-induced recombination, numerous ES clones with deletion of the second exon were identified. Deletion of the second exon was confirmed by RT-PCR of the ES cell RNA with primers from first and third exons. Two bands were identified in recombinant clones. A small band corresponding to the Cav1 cDNA lacking second exon was detected in addition to the wild-type cDNA containing all three exons (data not shown). After blastocyst ES cell microinjection and production of chimeric animals, the germ-line transmission of the mutant allele was confirmed by PCR. The mutant mouse colony homozygous for Cav1− allele was established. The Cav1−/− males and females were fully viable and had normal fertility within the first 2–8 months when they were used for breeding. The deletion of the second exon of Cav1 led to a complete ablation of CAV1 expression in mutants as evident from the comparison of IHC staining with anti-CAV1 antibody in wild-type and mutant uterus (Fig. 2A). It is important to note that antibody used for these experiments recognizes an epitope encoded by the first exon of Cav1. Thus, these results further indicate that the truncated mRNA of Cav1− detected by RT-PCR is not translated.
Fig. 2.
The expression of Cav1 and Rxfp2 in mouse uterus. A, Immunohistochemical localization of CAV1 in wild-type uterus and an absence of staining in Cav1−/− uterus. Females (1 yr old) were used. Strong signal was detected in endothelial cells of the blood vessels and myometrium. Note an absence of staining in glandular endometrial epithelium. B, The β-galactosidase staining showing the Rxfp2-promoter-dependent expression of iCre recombinase in Rxfp2-iCre, R26R transgenic mice (left). No staining was detected in uterus of R26R controls (right). The most prominent blue staining is located in endometrial glandular epithelial cells and luminal epithelium. Two month old females were used. Magnification bar, 100 μm.
Expression of CAV1 and RXFP2 in uterus
To localize expression of CAV1 in mouse uterus, we used IHC. The CAV1 expression was readily detected in mouse uterus (Fig. 2A). Strongest expression was detected in endothelial cells of uterine vasculature. The CAV1 was also well expressed in circular and longitudinal layers of the myometrium. Weak or no expression was observed in endometrial stromal cells and epithelial cells of endometrial glands (Fig. 2A).
In the absence of reliable mouse anti-RXFP2 antibodies, we used Rxfp2-icre transgenic mice to localize Rxfp2 expression in mice with R26R reporter. The expression of iCre recombinase driven by Rxfp2 promoter caused a removal of floxed STOP codon and an activation of lacZ, thus marking the cells with Rxfp2 expression (Fig. 2B). The strongest expression of Rxfp2 was detected in endometrial gland epithelium; somewhat less staining was also present in endometrial stromal cells and in myometrium.
Endometrial cysts in Cav1- and Rxfp2-deficient females
Previously, a high incidence of benign stromal lesions and tumors was detected in mouse strains with other mutant allele of Cav1 (28). The high level of INSL3 receptor RXFP2 expression in uterus suggested some role of this hormone in female reproductive tract function. However, no female reproductive organ abnormalities have been reported for Rxfp2−/− females (6, 7). We generated mice with the Cav1−/− deletion heterozygous for crsp deletion. Analysis of Cav1−/−, crsp/+ females revealed a high incidence of uterine abnormalities. We have produced and examined a total of 55 females at 11–13 months of age with this genotype. Analysis of these animals revealed a high frequency of abnormal uterus (55%) (Table 1). The most common defect detected at macroscopic level was a presence of single or multiple uterine cysts (Table 1 and Fig. 3, A and B). Almost the same frequency of abnormalities (61%) was detected in females homozygous for crsp deletion. The incidence of detected abnormalities in Cav1−/− females derived from the same litters was almost 3 times lower (18.8%, P < 0.05). The crsp deletion encompasses more than 500 kb of genomic DNA (6). To prove that the Rxfp2 gene is responsible for the increase of cyst formation, we used mice with a targeted knockout allele of Rxfp2 (7). The Rxfp2− strain was maintained as a congenic line (B15 generation) on the same FVB background as coisogenic FVB-crsp line. The uterine abnormalities were present with the same frequency in females heterozygous for targeted mutation of Rxfp2 (58%) as in crsp/+ mice, confirming a causative role of Rxfp2 deficiency. We also detected an appearance of ovarian cysts with about the same frequency (3.8–7.7%) in all four groups (Table 1). Analysis of 5- to 7-month-old Cav1−/−, Rxfp2+/− females revealed 23.5% (four of 17) females with uterine cysts, suggesting an increase in cyst development in older animals.
Table 1.
Female reproductive tract abnormalities detected in 11- to 13-month-old females
| Genotype | Uterus |
Ovary |
|||||||
|---|---|---|---|---|---|---|---|---|---|
| Total | Normal | Cysts | % Cystic | P valuea | Total | Normal | Cysts | % Cystic | |
| Cav1−/−, +/+ | 13 | 3 | 18.8 | 16 | 15 | 1 | 6.3 | ||
| Cav1−/−, crsp/+ | 55 | 25 | 30 | 54.5 | 0.0209 | 55 | 53 | 2 | 3.6 |
| Cav1−/−, crsp/crsp | 13 | 5 | 8 | 61.5 | 0.0266 | 13 | 12 | 1 | 7.7 |
| Cav1−/−, Rxfp2−/+ | 36 | 15 | 21 | 58.3 | 0.0147 | 36 | 34 | 2 | 5.6 |
| Cav1−/−, Rxfp2−/+b | 17 | 13 | 4 | 23.5 | 17 | 17 | 1 | 0 | |
P values calculated in comparison with Cav1−/−, +/+ group.
Females (5–7 months of age).
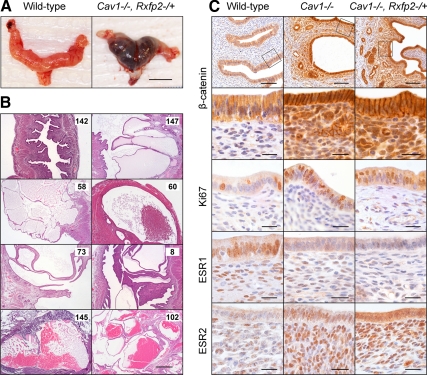
Fig. 3.
Endometrial cyst development in Cav1-deficient mice. A, Polycystic uterus from 12-month-old Cav1−/−, Rxfp2−/+ female (right image). Multiple cysts filled with blood are visible in both uterine horns. Normal uterus of wild-type female is shown on the left. Magnification bar, 10 mm. B, Cross-sections of representative uterine abnormalities in Cav1−/−. Top left image is a wild-type uterus (no. 142). Endometrial cysts in Cav1−/− (nos. 8 and 58), Cav1−/−, crsp/+ (nos. 73 and 102), Cav1−/−, Rxfp2+/− (nos. 60, 145, and 147). All animals are 11–13 months old. Note the presence of eiosinophilic secretion or blood cells in some cysts. Magnification bar, 500 μm. C, Expression of β-catenin (boxed areas are shown at higher magnification in a second row), Ki67, ESR1, and ESR2 in 11- to 13-month-old wild-type, Cav1−/−, and Cav1−/−, Rxfp2−/+ uteri. Note a significant increase of β-catenin, Ki67, and ESR2-positive cells. Magnification bars, 100 μm, row 1; 20 μm, rows 2–5.
Histological studies of abnormal uteri
The analysis of uterine lesions revealed the presence of grossly increased and expanded endometrial glands of various sizes (Fig. 3B). The dilated glands were lined by a flattened, simple low epithelium. Some of the cysts were filled with eosinophilic secretory or cellular material, and, in several cases, cysts were filled with blood. Severely dilated endometrial cysts often completely changed the normal histological organization of the uterus, displacing both the surrounding endometrial stromal and myometrium (Fig. 3B). We did not detect any significant difference in cyst appearance between females with or without deletion of Rxfp2. No cellular atypia, development of endometrial cancer, or metastases were detected in any of these mice.
The expression of estrogen receptor α (ESR1) was generally decreased in all 12-month-old females analyzed in this study compared with the younger animals. ESR1 was broadly expressed in endometrial stromal cells with intense nuclear immunoreactivity detected in glandular epithelium. Analysis of ESR1 expression in noncystic and cystic endometrium revealed a reduced level of ESR1 in the latter group (Fig. 3C). The decrease of ESR1 was evident in both stromal endometrial and flattened epithelial cells when compared with the age-matched wild-type uteri. The opposite staining pattern was detected for estrogen receptor β (ESR2). An increased nuclear staining was detected in both stromal and epithelial cells from the old Cav1−/− mice with or without Rxfp2 deficiency (Fig. 3C).
Recently, both INSL3 and related relaxin peptide were implicated in regulation of WNT/β-catenin signaling (32, 33). We therefore analyzed the expression of β-catenin in normal and cystic endometrium. β-Catenin was strongly expressed in glandular and, to a lesser degree, in stromal cells in wild-type endometrium (Fig. 3C). The expression was visibly increased in stromal cells in Cav1−/− mutants with or without Rxfp2-null allele compared with the wild-type tissues. Prominent nuclear localization of the β-catenin was noted in Cav1-deficient mice (Fig. 3C).
Cell proliferation was evaluated by examining Ki67 expression in Cav1−/− and Cav1−/−, Rxfp2−/+ endometrial glandular epithelium in animals without cysts and in animals with cysts (Fig. 3C). We detected an increase of proliferation (P < 0.01) in both cystic and normal endometrial epithelium in animals with cysts vs. those without cysts (Fig. 4). However, no difference between cystic or normal endometrium was found between mice of Cav1−/− and Cav1−/−, Rxfp2−/+ genotype.
Fig. 4.
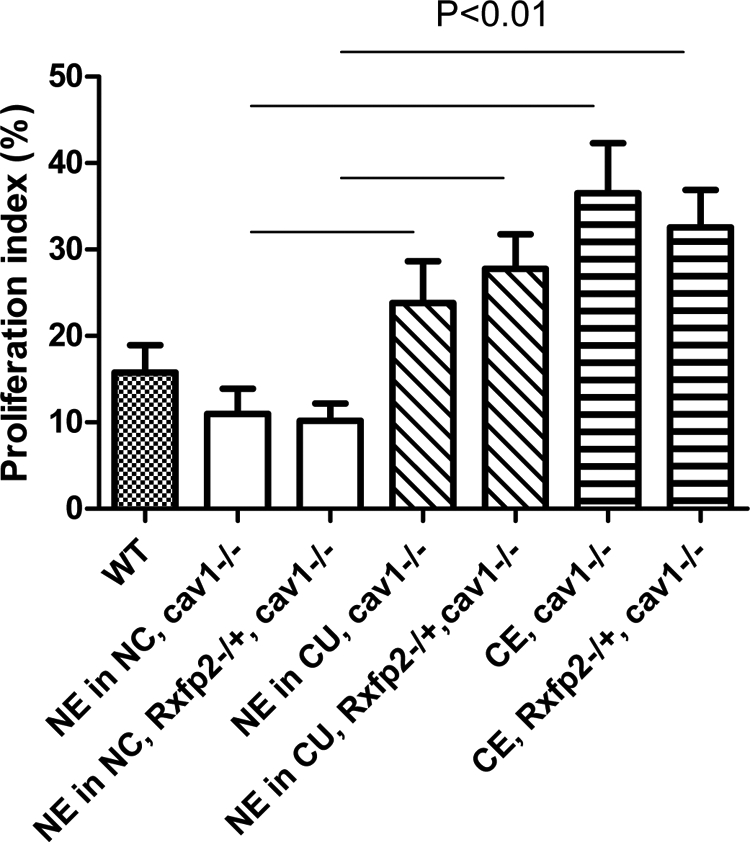
An increase of epithelial endometrial cell proliferation in cystic uteri. The proliferation index was calculated as a percentage of Ki67-positive cells in normal (NE) and cystic (CE) epithelium in uteri without cysts (NC) or with cysts (CU). Genotype of the animals is indicated. WT, Age-matched wild-type females. Each group represented by three to six 11- to 13-month-old animals. Differences between NE in animals without cysts and NE or CE in animals with cysts are significant (P < 0.01).
Analysis of CAV1 expression in human hyperplastic endometrium
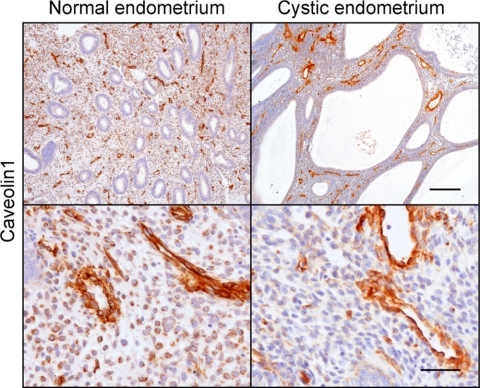
Our observations led us to hypothesize that altered CAV1 expression could play a role in the abnormal endometrial proliferation that leads to and/or characterizes human hyperplastic endometrium. To explore this question, we analyzed the expression of CAV1 in endometrial specimens collected from women found to have dilated cystic endometrium at the time they underwent curetting. We specifically chose to examine these specimens because, histologically, these lesions resemble our murine phenotype. Ten affected patient and 10 age-matched control samples were analyzed using IHC. Similar to the mouse endometrium, human endometrium had weak CAV1 staining in stromal cells and no expression in the endometrial glandular epithelium (Fig. 5). Although some variation in CAV1 expression was detected among specimens, there were no appreciable differences between proliferative or secretory endometrium and dilated glands in normal and hyperplastic samples (Fig. 5).
Fig. 5.
CAV1 expression in normal human endometrium and in endometrium with dilated cysts. There is a strong CAV1 staining in endothelial cells of blood vessels, weak CAV1 staining in stromal endometrial cells, and no expression in the endometrial glandular epithelium. Normal endometrium is on the left, endometrium with dilated cysts is on the right. Magnification bars, 200 μm, top; 50 μm, bottom.
Discussion
We analyzed the effect of INSL3 receptor deficiency on female reproductive tract. We have shown that RXFP2 is well expressed in different tissue compartments of the uterus. We report here a production of a new knockout allele of Cav1 gene in mice. Similar to the previously reported Cav1-deficient mice, the Cav1−/− animals were viable and fertile. We then produced Cav1−/− mice with 500 kb crsp deletion encompassing the Rxfp2 gene in the proximal part of chromosome 5. The analysis of 1-yr-old Cav1-deficient females with wild-type chromosome 5, heterozygous for crsp, or crsp/crsp homozygous revealed an increase in the incidence of endometrial cysts in three groups (18%, 54%, 61%). Single or multiple dilated endometrial cysts, often filled with secretion material, cell debris, eosinophilic substance, or blood cells, were detected. Mice with targeted mutation of Rxfp2 demonstrated the same increase in cyst formation on Cav1−/− background as in crsp/+, Cav1−/− females, indicating that the deficiency of INSL3/RXFP2 signaling was responsible for the increase in uterine abnormalities.
The significance of CAV1 expression in different human malignancies is still being debated. A number of reports have demonstrated the loss of CAV1 expression in various human cancers. On the other hand, a poor survival and prognosis in various adenocarcinomas associated with CAV1 overexpression were reported (34). Although the incidence of malignant tumors was not reported in mice with deletion of the gene, the Cav1−/− mice have been found to be more sensitive to carcinogen-induced hyperplasia and tumorigenesis (35). Female mice with deletion of Cav1 showed an increase and early onset of breast cancer in transgenic mice expressing a dominant transforming oncogene, polyoma middle T (PyMT), or Erbb2 (HER2) v-erb-b2 erythroblastic leukemia viral oncogene homolog 2 (34). A recent study demonstrated an increase of benign lesions in mice deficient for Cav1, including abnormalities of male and female reproductive organs (28, 36).
The data presented here demonstrated that the deficiency for Rxfp2 caused an increase in uterine disorganization and the development of endometrial cysts on CAV1-deficient background. No such defects have been previously detected in Insl3- or Rxfp2-deficient animals, indicating a synergetic effect of Cav1− deficiency on the development of this abnormality. Notably, the deletion of even one functional Rxfp2 allele was sufficient to cause the abnormality, demonstrating a dose dependency previously reported for this gene in the development of cryptorchidism (8).
The INSL3/RXFP2 ligand/receptor pair strongly resembles closely related relaxin/RXFP1 system (37). Both hormones and corresponding receptors have similar structure and at least in receptor-transfected cells, both hormones activate cAMP. Relaxin receptor RXFP1 is expressed mainly in myometrium and in the lamina propria under the luminal epithelium (22, 38, 39). Estrogen receptor-dependent proliferative relaxin effects on epithelial cells are mediated by stromal cells (40). Moreover, it was demonstrated that relaxin caused an increase in uterine weight and stimulation of endometrial angiogenesis and inhibited endometrial levels of estrogen receptor α and progesterone receptor, while maintaining endometrial connective tissue integrity (41). It is possible that the defect in different tissue components of uterus or an absence of proper cell-cell interactions may also play a role in the phenomenon described here. Indeed, we showed that Rxfp2 is strongly expressed in endometrial epithelium, whereas CAV1 was mainly detected in myometrial cells in addition to the endothelial cells lining blood vessels. Thus, an absence of proper cell-cell signaling in double mutants might be critical for the proper maintenance of uterine structure and for the loss of integrity of endometrial glands.
An increase in the frequency of disorganization in the CAV1/RXFP2-deficient females vs. CAV1-deficient females was likely independent of estrogen receptor α signaling. Previously the effect of CAV1 on both expression of ESR1 and its function has been described (36, 42, 43). Although we detect some decrease in ESR1 expression in all older Cav1−/− females analyzed in this study, no significant difference was detected between Cav1-deficient groups with or without Rxfp2 mutation. Analysis of estrogen receptor β expression detected the notable increase in the endometrium of Cav1−/− females with or without Rxfp2 deficiency when compared with the wild type. These results correspond to the reported findings in human samples from women with endometrial hyperplasia (44). An increase in ESR2 might be also responsible for the increased proliferation of endometrial cells but at the same time for the absence of cancerous transformation in cystic epithelium in these mice. The other critical cell signaling system involved in endometrial function is WNT/ β-catenin. It was shown that an overexpression of β-catenin induced hyperproliferation and tumor formation in different components of the uterus (45, 46). The conditional deletion of Dicer using Amhr2-cre causes a formation of cysts in oviducts with corresponding misregulation of several WNT genes (47, 48). We have detected strong nuclear staining for β-catenin in endometrial stromal cells of Cav1-deficient mice, both in cystic and normal parts of the endometrium in affected animals; however, no differences were detected between Cav1−/−, Rxfp2+/+ and Cav1−/−, Rxfp2−/+ groups. It is possible, therefore, that the overexpression of β-catenin was a direct effect of gene deletions. The epithelial endometrial cells demonstrated a higher proliferation index than the normal epithelium in animals without cysts, perhaps reflecting the loss of normal stromal-epithelial interaction in Cav1−/− mice. Thus, it is possible that a combination of different signaling pathways affected by CAV1 and RXFP2 deficiency in different cellular components leads to the uterine disorganization. The impact of such changes on the uterine function and their contribution to the Cav1−/− phenotype remain to be elucidated.
The CAV1 expression present in normal and benign tumors has been shown previously to recede in high-grade sarcomas, consistent with the tumor-suppressing role of this protein (27). We have analyzed the expression of CAV1 in a small group of human patients with dilated glands in normal and hyperplastic endometrium. No significant differences have been recognized in these samples. Further analysis of the RXFP2 expression in various human abnormalities leading to endometrial malfunction will be required; a development of reliable anti-RXFP2 antibodies will be essential for such studies.
In summary, we have shown that reduced INSL3/RXFP2 signaling on CAV1-deficient background caused disorganization of the uterine morphology accompanied with development of simple dilated endometrial cysts. The results of the study highlight previously unrecognized functional significance of INSL3/RXFP2 in female reproductive tract.
Acknowledgments
We thank Anne Truong (Baylor College of Medicine) for excellent technical support with histochemical analysis and mouse genotyping and the Baylor College of Medicine Transgenic Core (Director, Dr. Franco DeMayo) for the help with Cav1-mutant mice production.
This work was supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development/National Institute of Health grant R01HD37067 (to A.I.A.), and a Chinese Student Council scholarship (to Z.L.).
Disclosure Summary: The authors have nothing to disclose.
Footnotes
- CAV1
- Caveolin-1
- ES
- embryonic stem
- ESR1
- estrogen receptor α
- ESR2
- estrogen receptor β
- IHC
- immunohistochemistry
- INSL3
- insulin-like 3
- RXFP2
- relaxin family peptide receptor 2.
References
- 1. Bogatcheva NV, Agoulnik AI. 2005. INSL3/LGR8 role in testicular descent and cryptorchidism. Reprod Biomed Online 10:49–54 [DOI] [PubMed] [Google Scholar]
- 2. Bogatcheva NV, Truong A, Feng S, Engel W, Adham IM, Agoulnik AI. 2003. GREAT/LGR8 is the only receptor for insulin-like 3 peptide. Mol Endocrinol 17:2639–2646 [DOI] [PubMed] [Google Scholar]
- 3. Kumagai J, Hsu SY, Matsumi H, Roh JS, Fu P, Wade JD, Bathgate RA, Hsueh AJ. 2002. INSL3/Leydig insulin-like peptide activates the LGR8 receptor important in testis descent. J Biol Chem 277:31283–31286 [DOI] [PubMed] [Google Scholar]
- 4. Zimmermann S, Steding G, Emmen JM, Brinkmann AO, Nayernia K, Holstein AF, Engel W, Adham IM. 1999. Targeted disruption of the Insl3 gene causes bilateral cryptorchidism. Mol Endocrinol 13:681–691 [DOI] [PubMed] [Google Scholar]
- 5. Nef S, Parada LF. 1999. Cryptorchidism in mice mutant for Insl3. Nat Genet 22:295–299 [DOI] [PubMed] [Google Scholar]
- 6. Overbeek PA, Gorlov IP, Sutherland RW, Houston JB, Harrison WR, Boettger-Tong HL, Bishop CE, Agoulnik AI. 2001. A transgenic insertion causing cryptorchidism in mice. Genesis 30:26–35 [DOI] [PubMed] [Google Scholar]
- 7. Gorlov IP, Kamat A, Bogatcheva NV, Jones E, Lamb DJ, Truong A, Bishop CE, McElreavey K, Agoulnik AI. 2002. Mutations of the GREAT gene cause cryptorchidism. Hum Mol Genet 11:2309–2318 [DOI] [PubMed] [Google Scholar]
- 8. Bogatcheva NV, Ferlin A, Feng S, Truong A, Gianesello L, Foresta C, Agoulnik AI. 2007. T222P mutation of the insulin-like 3 hormone receptor LGR8 is associated with testicular maldescent and hinders receptor expression on the cell surface membrane. Am J Physiol Endocrinol Metab 292:E138–E144 [DOI] [PubMed] [Google Scholar]
- 9. Kawamura K, Kumagai J, Sudo S, Chun SY, Pisarska M, Morita H, Toppari J, Fu P, Wade JD, Bathgate RA, Hsueh AJ. 2004. Paracrine regulation of mammalian oocyte maturation and male germ cell survival. Proc Natl Acad Sci USA 101:7323–7328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Ferlin A, Pepe A, Gianesello L, Garolla A, Feng S, Giannini S, Zaccolo M, Facciolli A, Morello R, Agoulnik AI, Foresta C. 2008. Mutations in the insulin-like factor 3 receptor are associated with osteoporosis. J Bone Miner Res 23:683–693 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Hombach-Klonisch S, Bialek J, Radestock Y, Truong A, Agoulnik AI, Fiebig B, Willing C, Weber E, Hoang-Vu C, Klonisch T. 2010. INSL3 has tumor-promoting activity in thyroid cancer. Int J Cancer 127:521–531 [DOI] [PubMed] [Google Scholar]
- 12. Klonisch T, Bialek J, Radestock Y, Hoang-Vu C, Hombach-Klonisch S. 2007. Relaxin-like ligand-receptor systems are autocrine/paracrine effectors in tumor cells and modulate cancer progression and tissue invasiveness. Adv Exp Med Biol 612:104–118 [DOI] [PubMed] [Google Scholar]
- 13. Klonisch T, Müller-Huesmann H, Riedel M, Kehlen A, Bialek J, Radestock Y, Holzhausen HJ, Steger K, Ludwig M, Weidner W, Hoang-Vu C, Hombach-Klonisch S. 2005. INSL3 in the benign hyperplastic and neoplastic human prostate gland. Int J Oncol 27:307–315 [PubMed] [Google Scholar]
- 14. Klonisch T, Mustafa T, Bialek J, Radestock Y, Holzhausen HJ, Dralle H, Hoang-Vu C, Hombach-Klonisch S. 2005. Human medullary thyroid carcinoma: a source and potential target for relaxin-like hormones. Ann NY Acad Sci 1041:449–461 [DOI] [PubMed] [Google Scholar]
- 15. Zimmermann S, Schöttler P, Engel W, Adham IM. 1997. Mouse Leydig insulin-like (Ley I-L) gene: structure and expression during testis and ovary development. Mol Reprod Dev 47:30–38 [DOI] [PubMed] [Google Scholar]
- 16. Hombach-Klonisch S, Kauffold J, Rautenberg T, Steger K, Tetens F, Fischer B, Klonisch T. 2000. Relaxin-like factor (RLF) mRNA expression in the fallow deer. Mol Cell Endocrinol 159:147–158 [DOI] [PubMed] [Google Scholar]
- 17. Bathgate R, Balvers M, Hunt N, Ivell R. 1996. Relaxin-like factor gene is highly expressed in the bovine ovary of the cycle and pregnancy: sequence and messenger ribonucleic acid analysis. Biol Reprod 55:1452–1457 [DOI] [PubMed] [Google Scholar]
- 18. Roche PJ, Butkus A, Wintour EM, Tregear G. 1996. Structure and expression of Leydig insulin-like peptide mRNA in the sheep. Mol Cell Endocrinol 121:171–177 [DOI] [PubMed] [Google Scholar]
- 19. Bamberger AM, Ivell R, Balvers M, Kelp B, Bamberger CM, Riethdorf L, Löning T. 1999. Relaxin-like factor (RLF): a new specific marker for Leydig cells in the ovary. Int J Gynecol Pathol 18:163–168 [PubMed] [Google Scholar]
- 20. Spanel-Borowski K, Schäfer I, Zimmermann S, Engel W, Adham IM. 2001. Increase in final stages of follicular atresia and premature decay of corpora lutea in Insl3-deficient mice. Mol Reprod Dev 58:281–286 [DOI] [PubMed] [Google Scholar]
- 21. Gambineri A, Patton L, De Iasio R, Palladoro F, Pagotto U, Pasquali R. 2007. Insulin-like factor 3: a new circulating hormone related to luteinizing hormone-dependent ovarian hyperandrogenism in the polycystic ovary syndrome. J Clin Endocrinol Metab 92:2066–2073 [DOI] [PubMed] [Google Scholar]
- 22. Li Z, Burzawa JK, Troung A, Feng S, Agoulnik IU, Tong X, Anderson ML, Kovanci E, Rajkovic A, Agoulnik AI. 2009. Relaxin signaling in uterine fibroids. Ann NY Acad Sci 1160:374–378 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Razani B, Engelman JA, Wang XB, Schubert W, Zhang XL, Marks CB, Macaluso F, Russell RG, Li M, Pestell RG, Di Vizio D, Hou H, Jr, Kneitz B, Lagaud G, Christ GJ, Edelmann W, Lisanti MP. 2001. Caveolin-1 null mice are viable but show evidence of hyperproliferative and vascular abnormalities. J Biol Chem 276:38121–38138 [DOI] [PubMed] [Google Scholar]
- 24. Zhao YY, Liu Y, Stan RV, Fan L, Gu Y, Dalton N, Chu PH, Peterson K, Ross J, Jr, Chien KR. 2002. Defects in caveolin-1 cause dilated cardiomyopathy and pulmonary hypertension in knockout mice. Proc Natl Acad Sci USA 99:11375–11380 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Cao G, Yang G, Timme TL, Saika T, Truong LD, Satoh T, Goltsov A, Park SH, Men T, Kusaka N, Tian W, Ren C, Wang H, Kadmon D, Cai WW, Chinault AC, Boone TB, Bradley A, Thompson TC. 2003. Disruption of the caveolin-1 gene impairs renal calcium reabsorption and leads to hypercalciuria and urolithiasis. Am J Pathol 162:1241–1248 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Drab M, Verkade P, Elger M, Kasper M, Lohn M, Lauterbach B, Menne J, Lindschau C, Mende F, Luft FC, Schedl A, Haller H, Kurzchalia TV. 2001. Loss of caveolae, vascular dysfunction, and pulmonary defects in caveolin-1 gene-disrupted mice. Science 293:2449–2452 [DOI] [PubMed] [Google Scholar]
- 27. Wiechen K, Sers C, Agoulnik A, Arlt K, Dietel M, Schlag PM, Schneider U. 2001. Down-regulation of caveolin-1, a candidate tumor suppressor gene, in sarcomas. Am J Pathol 158:833–839 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Yang G, Timme TL, Naruishi K, Fujita T, Fattah el MA, Cao G, Rajagopalan K, Troung LD, Thompson TC. 2008. Mice with cav-1 gene disruption have benign stromal lesions and compromised epithelial differentiation. Exp Mol Pathol 84:131–140 [DOI] [PubMed] [Google Scholar]
- 29. Pickar JH. 2009. The endometrium–from estrogens alone to TSECs. Climacteric 12:463–477 [DOI] [PubMed] [Google Scholar]
- 30. Malinin GI, Malinin IM. 1972. Age-related spontaneous uterine lesions in mice. J Gerontol 27:193–196 [DOI] [PubMed] [Google Scholar]
- 31. Feng S, Bogatcheva NV, Truong A, Korchin B, Bishop CE, Klonisch T, Agoulnik IU, Agoulnik AI. 2007. Developmental expression and gene regulation of insulin-like 3 receptor RXFP2 in mouse male reproductive organs. Biol Reprod 77:671–680 [DOI] [PubMed] [Google Scholar]
- 32. Kaftanovskaya EM, Feng S, Huang Z, Tan Y, Barbara AM, Kaur S, Truong A, Gorlov IP, Agoulnik AI. 2011. Suppression of insulin-like3 receptor reveals the role of β-catenin and Notch signaling in gubernaculum development. Mol Endocrinol 25:170–183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Liu S, Vinall RL, Tepper C, Shi XB, Xue LR, Ma AH, Wang LY, Fitzgerald LD, Wu Z, Gandour-Edwards R, deVere White RW, Kung HJ. 2008. Inappropriate activation of androgen receptor by relaxin via β-catenin pathway. Oncogene 27:499–505 [DOI] [PubMed] [Google Scholar]
- 34. Sloan EK, Ciocca DR, Pouliot N, Natoli A, Restall C, Henderson MA, Fanelli MA, Cuello-Carrión FD, Gago FE, Anderson RL. 2009. Stromal cell expression of caveolin-1 predicts outcome in breast cancer. Am J Pathol 174:2035–2043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Capozza F, Williams TM, Schubert W, McClain S, Bouzahzah B, Sotgia F, Lisanti MP. 2003. Absence of caveolin-1 sensitizes mouse skin to carcinogen-induced epidermal hyperplasia and tumor formation. Am J Pathol 162:2029–2039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Mercier I, Casimiro MC, Zhou J, Wang C, Plymire C, Bryant KG, Daumer KM, Sotgia F, Bonuccelli G, Witkiewicz AK, Lin J, Tran TH, Milliman J, Frank PG, Jasmin JF, Rui H, Pestell RG, Lisanti MP. 2009. Genetic ablation of caveolin-1 drives estrogen-hypersensitivity and the development of DCIS-like mammary lesions. Am J Pathol 174:1172–1190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Bathgate RA, Ivell R, Sanborn BM, Sherwood OD, Summers RJ. 2006. International Union of Pharmacology LVII: recommendations for the nomenclature of receptors for relaxin family peptides. Pharmacol Rev 58:7–31 [DOI] [PubMed] [Google Scholar]
- 38. Kamat AA, Feng S, Bogatcheva NV, Truong A, Bishop CE, Agoulnik AI. 2004. Genetic targeting of relaxin and insulin-like factor 3 receptors in mice. Endocrinology 145:4712–4720 [DOI] [PubMed] [Google Scholar]
- 39. Yao L, Agoulnik AI, Cooke PS, Meling DD, Sherwood OD. 2008. Relaxin acts on stromal cells to promote epithelial and stromal proliferation and inhibit apoptosis in the mouse cervix and vagina. Endocrinology 149:2072–2079 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Yao L, Cooke PS, Meling DD, Shanks RD, Jameson JL, Sherwood OD. 2010. The effect of relaxin on cell proliferation in mouse cervix requires estrogen receptor α binding to estrogen response elements in stromal cells. Endocrinology 151:2811–2818 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Goldsmith LT, Weiss G, Palejwala S, Plant TM, Wojtczuk A, Lambert WC, Ammur N, Heller D, Skurnick JH, Edwards D, Cole DM. 2004. Relaxin regulation of endometrial structure and function in the rhesus monkey. Proc Natl Acad Sci USA 101:4685–4689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Schlegel A, Wang C, Pestell RG, Lisanti MP. 2001. Ligand-independent activation of oestrogen receptor α by caveolin-1. Biochem J 359:203–210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Wang X, Feng S, Zhang H, Wang Y, Cui Y, Wang Z, Liu J, Zou W. 2010. RNA inference-mediated caveolin-1 down-regulation decrease estrogen receptor α (ERα) signaling in human mammary epithelial cells. Mol Biol Rep 38:761–768 [DOI] [PubMed] [Google Scholar]
- 44. Villavicencio A, Bacallao K, Avellaira C, Gabler F, Fuentes A, Vega M. 2006. Androgen and estrogen receptors and co-regulators levels in endometria from patients with polycystic ovarian syndrome with and without endometrial hyperplasia. Gynecol Oncol 103:307–314 [DOI] [PubMed] [Google Scholar]
- 45. Jeong JW, Lee HS, Franco HL, Broaddus RR, Taketo MM, Tsai SY, Lydon JP, DeMayo FJ. 2009. β-Catenin mediates glandular formation and dysregulation of β-catenin induces hyperplasia formation in the murine uterus. Oncogene 28:31–40 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Tanwar PS, Lee HJ, Zhang L, Zukerberg LR, Taketo MM, Rueda BR, Teixeira JM. 2009. Constitutive activation of β-catenin in uterine stroma and smooth muscle leads to the development of mesenchymal tumors in mice. Biol Reprod 81:545–552 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Nagaraja AK, Andreu-Vieyra C, Franco HL, Ma L, Chen R, Han DY, Zhu H, Agno JE, Gunaratne PH, DeMayo FJ, Matzuk MM. 2008. Deletion of Dicer in somatic cells of the female reproductive tract causes sterility. Mol Endocrinol 22:2336–2352 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Gonzalez G, Behringer RR. 2009. Dicer is required for female reproductive tract development and fertility in the mouse. Mol Reprod Dev 76:678–688 [DOI] [PMC free article] [PubMed] [Google Scholar]