Abstract
Purpose: The goal of the present work was to evaluate the accuracy of a plastic scintillation detector (PSD) system to perform in-phantom dosimetry during 192Ir high dose rate (HDR) brachytherapy treatments.
Methods: A PSD system capable of stem effect removal was built. A red–green–blue photodiode connected to a dual-channel electrometer was used to detect the scintillation light emitted from a green scintillation component and transmitted along a plastic optical fiber. A clinically relevant prostate treatment plan was built using the HDR brachytherapy treatment planning system. An in-house fabricated template was used for accurate positioning of the catheters, and treatment delivery was performed in a water phantom. Eleven catheters were inserted and used for dose delivery from 192Ir radioactive source, while two others were used to mimic dosimetry at the rectum wall and in the urethra using a PSD. The measured dose and dose rate data were compared to the expected values from the planning system. The importance of removing stem effects from in vivo dosimetry using a PSD during 192Ir HDR brachytherapy treatments was assessed. Applications for dwell position error detection and temporal verification of the treatment delivery were also investigated.
Results: In-phantom dosimetry measurements of the treatment plan led to a ratio to the expected dose of 1.003 ± 0.004 with the PSD at different positions in the urethra and 1.043 ± 0.003 with the PSD inserted in the rectum. Verification for the urethra of dose delivered within each catheter and at specific dwell positions led to average measured to expected ratios of 1.015 ± 0.019 and 1.014 ± 0.020, respectively. These values at the rectum wall were 1.059 ± 0.045 within each catheter and 1.025 ± 0.028 for specific dwell positions. The ability to detect positioning errors of the source depended of the tolerance on the difference to the expected value. A 5-mm displacement of the source was detected by the PSD system from 78% to 100% of the time depending on the acceptable range value. The implementation of a stem effect removal technique was shown to be necessary, particularly when calculating doses at specific dwell positions, and allowed decreasing the number of false-error detections—the detection of an error when it should not be the case—from 19 to 1 for a 5% threshold out of 43 measurements. The use of the PSD system to perform temporal verification of elapsed time by the source in each catheter—generally on the order of minutes—was shown to be in agreement within a couple of seconds with the treatment plan.
Conclusions: We showed that the PSD system used in this study, which was capable of stem effect removal, can perform accurate dosimetry during 192Ir HDR brachytherapy treatment in a water phantom. The system presented here shows some clear advantages over previously proposed dosimetry systems for HDR brachytherapy, and it has the potential for various online verifications of treatment delivery quality.
Keywords: in vivo dosimetry, plastic scintillation detectors, high dose rate, stem effect, brachytherapy
INTRODUCTION
The main challenge in radiation therapy is to deliver a prescribed radiation dose to the tumor while limiting the dose delivered to surrounding healthy organs and tissues. It is with this goal in mind that high-dose-rate (HDR) brachytherapy was developed. In HDR brachytherapy, a radioactive 192Ir source with an air kerma strength between 20 600 and 41 200 μGy m2 h−1 is placed for defined amounts of time along catheters inserted within the treatment site. Source positions and dwell times are determined to meet the requirements for the prescribed dose. The positioning and displacement of the 192Ir source are controlled with a remote afterloader. A multitude of anatomical sites have been treated with HDR brachytherapy, including the prostate, breast, lung, vagina, cervix, head and neck, and esophagus. HDR brachytherapy is also used for intraoperative radiation therapy, a procedure that initially involves surgically resecting the tumor and then irradiating the surgical bed.
An important characteristic of HDR brachytherapy is the fact that a radioactive source is used, which necessitates many considerations regarding security and safety. A variety of quality assurance procedures have been established, aiming to verify each step of the hypofractionated treatment. The ultimate quality assurance would be the use of an online in vivo dosimetry system, which would verify the dose delivered to the organs at risk and tumor in real-time while the treatment is dispensed. In vivo dosimetry would provide a more accurate measure of the dose being delivered during a fraction and could be compared to the treatment plan on the basis of a dwell position, catheter, or fraction analysis. The treatment could then be modified between fractions or even stopped, if necessary.
The search for an adequate dosimeter to perform accurate in vivo dosimetry during 192Ir HDR brachytherapy treatments is not new.1, 2, 3, 4, 5, 6, 7, 8, 9, 10 The various specificities and challenges to develop such a dosimeter make it a difficult task. Among the requirements, the dosimeter has to be small enough to fit into a catheter (inner diameter ∼1.4 mm) and must allow measurements to be taken near to and far from (up to 10 cm) the 192Ir radiation source. For example, the dose rate at 7 cm on the radial axis of the source is over 200 times smaller than the dose delivered at 0.5 cm.11 The detector response should also show dose linearity and energy independence in the range of interest. Furthermore, to avoid disturbing the dose planning and to measure the dose delivered to tissue and organs with minimal correction factors, the portion of the dosimeter irradiated by the source should be made of water-equivalent materials. Ionization chambers, diamond detectors, thermoluminescent dosimeters, diodes, metal oxide semiconductor field-effect transistors (MOSFETs), and scintillation detectors are among the detectors that have been considered for such an application. Comparison between these detectors was performed by Lambert et al.9 Although ionization chambers represent a gold standard for megavoltage Linac dosimetry, the high-dose gradients in 192Ir HDR brachytherapy make the bigger ion chambers useless for such an application, while the smaller chambers are not sensitive enough.9 Diamond detectors show adequate sensitivity but also show nonlinearity with increasing dose rate.3In vivo studies showed the usefulness of thermoluminescent dosimeters in brachytherapy to obtain measurements of the integrated dose delivered during a single treatment fraction.1, 2, 4 However, these detectors are not suited for giving a real-time measure of the dose because development time is required. Diodes show good accuracy, but their sensitivity changes with distance. Another drawback for in vivo dosimetry is the fact that diodes are silicon based, and silicon is not water equivalent.5 The non-water equivalence also holds true for MOSFET detectors, which showed energy dependence and variation in sensitivity with absorbed dose.6, 10, 12, 13 Nevertheless, implantable MOSFET dosimeters have been developed and are used for online dosimetry.6 The active size of the detector makes it a near point detector. Dosimetry that combined radioluminescence and optically stimulated luminescence was investigated by Andersen et al.14, 15 for online in vivo dosimetry in pulsed-dose-rate (PDR) brachytherapy. This technique uses an aluminum oxide (Al2O3:C) crystal as the luminescent material. Dose measurement was in agreement with the expected to a ratio of 1.04 ± 0.02. This technique was shown to be a very accurate method for in vivo detection of error during a cervix PDR brachytherapy study.15
The use of plastic scintillation detectors (PSDs) for in vivo192Ir HDR brachytherapy dosimetry was studied previously and showed good promise.7, 9, 16 The PSDs’ small size, near water equivalence, energy independence (for energies >100 keV), dose linearity, independence to dose rate, fast response, and temperature independence are among the characteristics that make this detector attractive for in vivo dosimetry.17, 18, 19 The main drawback of scintillation dosimetry is the emission of “stem effect light,” that is, light that is created in the optical fiber when it is struck by radiation. The purpose of the optical fiber is to guide the scintillation component to the photodetector. Different techniques have been suggested to eliminate this contamination component on the signal when performing dosimetry with PSDs for high-energy external-beam radiotherapy.20, 21 We previously studied the importance of implementing a stem effect removal technique over a wide range of clinically relevant 192Ir HDR brachytherapy conditions and the necessity for such a removal was obvious to ensure a good accuracy in a wide variety of PSD to source positions.22 The chromatic removal technique was shown to be the most accurate of the approaches investigated.
However, the use of a stem effect removal capable water-equivalent PSD system has never been investigated extensively toward the idea of performing in vivo dosimetry for 192Ir HDR brachytherapy as an online quality assurance method. The goal of the present work was to use the novel PSD system developed in our previous study22 and evaluate its accuracy under a clinically relevant irradiation procedure prepared with a treatment planning system (TPS). The PSD’s performance was examined in reproducible conditions, and a prostate HDR treatment plan was experimentally delivered using a water phantom and high-precision template. The system’s accuracy for dose measurement over each fraction, each catheter, and individual source positions was tested for clinically realistic source-to-detector geometry and dwell times. The use of the PSD system to perform various quality checks during treatment delivery was also investigated.
MATERIALS AND METHODS
PSD
The PSD system consisted of a 1-mm diameter × 3-mm long green scintillating fiber (BCF-60, Saint-Gobain Crystals and Detectors, Paris, France) coupled to a light-shielded 1-mm diameter × 7-m long polymethylmethacrylate optical fiber (Eska Premier GH-4001, Mitsubishi Rayon Co. Ltd., Tokyo, Japan). The fiber was stripped from its usual 2.2-mm outside diameter jacket for the length of the fiber inserted in the catheter and replaced by a thinner black jacket in order for the detector to fit snugly into a brachytherapy catheter with an inside diameter of ∼1.4 mm. The detector was completely light tight. The other end of the optical fiber was connected via a SubMiniature version A (SMA) adapter and connected to a red–green–blue photodiode (MCSiAT, Mazet, Germany). Green and blue channels of the photodiode were connected via triaxial cables to a dual-channel electrometer (SuperMax, Standard Imaging, Madison, WI). The electrometer was computer-controlled via an RS-232 cable, and the output currents of the photodiode were recorded at a rate of 10 Hz. The photodiode was enclosed in an aluminum box connected to a ground to protect it from electronic noise (setup shown in Fig. 1).
Figure 1.
Light detection portion of the plastic scintillation detector setup used in this study. The optical fiber was connected through a SMA connector and aligned to a red–green–blue photodiode (see inset). The blue and green outputs from the photodiode were connected via a triaxial cable to a double-input electrometer. The output of the electrometer was transmitted to a computer (not shown) via an RS-232 cable.
HDR brachytherapy planning and treatment delivery
A clinically realistic prostate treatment plan for 192Ir HDR brachytherapy was designed using Oncentra version 3.2 (Nucletron, Vennendaal, the Netherlands). The treatment was delivered in a water phantom (35 cm × 35 cm × 25 cm) using the template we described previously22 and is shown with the experimental setup in Fig. 2. The template was used for accurate positioning of the catheters along their radial axes. A total of 13 catheters were inserted and kept parallel to each other (see Fig. 2). The catheters were pulled taut and held tightly with clamp buttons to keep them as straight as possible. Because of the size of the buttons, the catheters were separated by a minimum distance of 1 cm. Two of the catheters were used for PSD placement at positions referred to as the “urethra” and “rectum wall” relative to the dose planning, while the remaining 11 catheters were used for treatment delivery inside a virtual prostate volume using a clinical loading pattern. The PSD in the phantom was surrounded by a minimum of 10 cm of water.
Figure 2.
Positioning template (developed in-house) with the catheters, connected to transfer tubes, inserted for treatment delivery. The inserted plastic scintillation detector is covered with a black coating.
In order to simulate a clinically realistic 192Ir HDR brachytherapy treatment plan, we loaded computed tomography images of an arbitrarily chosen patient with prostate cancer into the TPS. The target volume (prostate) and surrounding organs at risk (i.e., bladder, urethra, and rectum) were contoured, and catheters were reconstructed in the software to reproduce the experimental setup. Treatment was planned using the inverse planning system algorithm (IPSA) as well as manual loading. The goal of the optimization was to deliver 1500 cGy to the prostate volume and spare the organs at risk as much as possible. The dose distribution calculated by the TPS is the result of a calculation from data published by Daskalov et al.11 following the guidelines of the Task Group 43 on the dosimetry of interstitial brachytherapy sources.23 Treatment was delivered for different PSD placements. Source air kerma strength throughout the experiments varied between 23 000 and 29 600 μGy m2 h−1.
Calibration of the PSD
The stem effect removal technique used for this experiment was described previously20, 24, 25 and consisted of a chromatic method for which measurements were made in two separate wavelength regions. The blue and green channels were used throughout this experiment. The dose and dose rate measurements were linked to measurements in these channels following equations:
| (1) |
or
| (2) |
where
| (3) |
For a green scintillating component, M1 corresponds to the current or charge readout of the green channel and M2 corresponds to the current or charge readout of the blue channel. To determine the two constants in either equation, one performs the basic technique of taking measurements in two known dose conditions.24 Determination of the “a” and “b” factors is then subject to the uncertainty in dose determination for both conditions. Equation (2) constitutes a rearrangement of Eq. (1) proposed by Guillot et al.25 that allows for calculation of the b factor under a null-dose condition [Eq. (3)], a procedure useful for application to HDR brachytherapy, since it means only one non-null dose need to be accurately determined. We first determined the b factor by performing a null dose-to-scintillator measurement by placing the scintillator more than 10 cm away from the source, shielding it in a thick lead block (>4 cm = 8 half-value layers), and then irradiating the optical fiber only. The b factor value was 0.600 (dimensionless). Factor a was calculated by taking the average of different factor a calculations from non-null dose situations. The a factor was calculated to be 7.159 cGy∕pC-s.
Positioning
While the necessity for accurate radial positioning was fulfilled using the template (Fig. 2), positioning accuracy of the source relative to the PSD along the longitudinal axis of the catheter was done by adjusting the catheters index length and planning positions in such a way that dose delivery was relatively symmetric along that axis.
In-phantom dosimetry
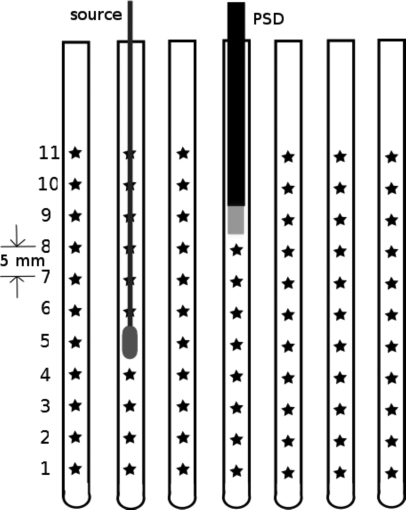
In-phantom dosimetry was performed with the PSD at different positions (see system of coordinates in Fig. 3). Each treatment delivery was repeated three times for each of the detector positions. The source delivery catheter was positioned to mimic the urethra at positions aligned sequentially to positions #1, #4, #8, #9, and #10. Dosimetry was also performed with the PSD aligned at position #7 in a catheter inserted for dose assessment of the rectal wall. Signal was acquired at a rate of 10 Hz; integration of data led to dose deposited per catheter and per treatment. Dosimetry was also performed for individual dwell positions, but only the dose rates for dwell times over 5 s were analyzed, as this was the rise time necessary for the current to increase to its true value. This limitation is a physical property of the electrometer and could be resolved in the future with a hardware modification. However, it should be mentioned that no charges are lost in the overall process. As there is enough time in between dose delivery from different catheters, the integral measurement of dose from source displacement in each catheter and over the entire treatment are not affected by this particular issue. For the different dosimetry values, doses and dose rates obtained through calculation with the TPS were used as reference data.
Figure 3.
System of coordinates for the source and PSD positioning used in this study. The consecutive dwell positions were separated by 5 mm. The source (along with its cable) is represented in the second catheter from the left, while the PSD is shown in the middle catheter. The end of the PSD represents the scintillation component while the rest of it is the optical fiber. The stars represent the possible dwell positions along the catheters.
Definition of uncertainties
Two different uncertainties were defined: the source-to-detector position uncertainty and the measurement uncertainty. The uncertainty in positioning was estimated to be of 0.2 mm in the r direction and 0.4 mm in the z direction. It takes into account the holes drilling accuracy of the template (from a computer-controlled milling machine), space between catheter wall and detector∕source, the ability to vertically align the source with the PSD’s scintillating element at a certain position as well as the nonperfect rigidity of the catheters. The effect of the positioning uncertainty on dose was assessed with the TPS and its value was applied to the treatment planning values in the results section in order to distinguish it from the measurement uncertainty. The uncertainty on the measurements was calculated from the standard deviation of multiple repeated measurements. Any other sources of uncertainty other than the one related to the repeated measurements were disregarded.
Error detection
In a clinical application of the dosimeter for dose verification, an error detection system could be implemented based on detection of discrepancies with the planned dose, and ruling it an error when it falls out of a predefined acceptable range. We evaluated such an application for the PSD system used in this study by forcing of a 5-mm error in positioning. The numbers of true-errors and false-accurate readings were calculated and compared for various amounts of acceptable error, ranging from 3% to 20%. Measurements with the PSD at positions #8 and #10 were used to simulate the effect of −5 mm and +5 mm errors, respectively, in the positioning of the catheters along the longitudinal axis in comparison with measurements with the PSD at position #9 (the expected value).
Importance of stem effect removal
The importance of using a stem effect removal technique was also revisited by comparing the dosimetric data obtained in this study to the dose distribution that would be obtained from the same data points but without having a stem effect removal technique implemented.22 The same set of current measurements was used as when implementing the chromatic removal technique, except that the measurements from the blue and green channels were added and then multiplied by a single calibration factor. A factor of 5.597 cGy∕pC-s was determined using the same procedure as used to determine the a factor in the chromatic technique. Position #1 in the urethra for the PSD placement was used as a clinically relevant condition in which production of stem effect could be important. Dosimetry over the whole treatment, for each catheter, and for each dwell position was performed. For different threshold values, we evaluated the number of errors that would be flagged by the system depending on whether or not a stem effect removal technique was implemented. As we assumed that all source positions are considered to be accurate, any error revealed by the measuring system could then be considered as being a false-error.
Temporal verification
Because PSDs have the potential to provide temporal information in addition to dosimetric data, we tested our system’s ability to measure the duration of dose delivery inside each catheter. The time elapsed by the source in each catheter was defined as the interval for which the dose rate measured was more than three times over the standard deviation of the background signal. The time calculated from the measurements was then compared to the expected time from the treatment plan report.
RESULTS
In-phantom dosimetry
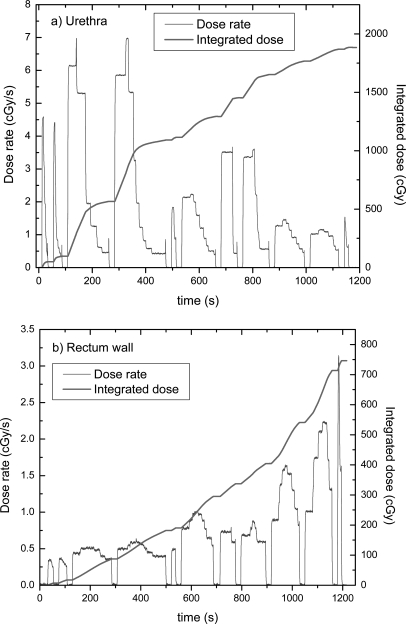
The treatment plan was delivered while the PSD was inserted in a specific position. Figure 4(a) shows the acquired dose rate and integrated dose when the PSD was inserted in the urethra at position #9. Figure 4(b) shows the delivery of the same treatment, but with the PSD inserted in the rectum at position #7. The total dose delivered to the PSD per treatment, dose delivered to the PSD per catheter, and dose rate to the PSD for specific dwell positions are given in Table Table I.. The ratio of measured dose to planned dose ranged from (0.996 ± 0.032) to (1.005 ± 0.015) with the PSD in the urethra and was (1.043 ± 0.003) when the PSD was positioned at the rectal wall.
Figure 4.
Acquired dose rate and integrated dose were measured as a function of time when the PSD was inserted (a) in the urethra and (b) at the rectum wall. The PSD was aligned to dwell position #9 for the urethra and position #7 for the rectum wall.
Table 1.
Doses measured with the PSD and planned doses for different PSD positions in the urethra and the rectum. Measured to planned ratio was also calculated. Measured values are reported as the average and standard deviation from three repeated measurements.
| PSD position | Measured dose (cGy) | Planned dose (cGy) | Measured∕planned |
|---|---|---|---|
| Urethra—pos. 1 | 1363 ± 2 | 1368 ± 42 | 0.996 ± 0.032 |
| Urethra—pos. 4 | 1851 ± 2 | 1845 ± 4 | 1.003 ± 0.003 |
| Urethra—pos. 8 | 1904 ± 2 | 1895 ± 2 | 1.005 ± 0.002 |
| Urethra—pos. 9 | 1883 ± 2 | 1875 ± 6 | 1.004 ± 0.004 |
| Urethra—pos. 10 | 1750 ± 2 | 1742 ± 23 | 1.005 ± 0.015 |
| Rectum—pos. 7 | 745 ± 2 | 714 ± 1 | 1.043 ± 0.003 |
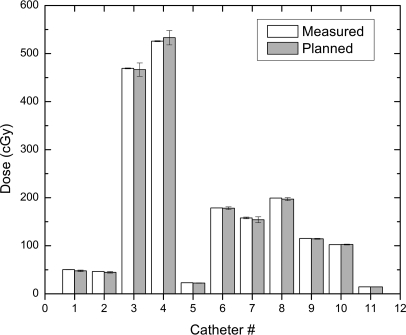
Figure 5 shows the dose to the PSD for the 11 individual catheters with the PSD at position #9 in the urethra. Good agreement was observed between the catheters and the treatment plan, regardless of whether a high or low dose was delivered to the PSD; the doses ranged from about 20 to 530 cGy. Ratios between the measured and expected doses are summarized in Table Table II..
Figure 5.
Dose measured in each catheter with the plastic scintillation detector in the urethra aligned to dwell position #9 and dose obtained from the planning system. Error bars for measured data were obtained from the standard deviation of three repeated measurements.
Table 2.
Ratio of the measured to the expected dose per catheter and dose rate per dwell position for different PSD positions in the urethra and rectum. Data shown here represent the average accuracies over the different catheters and different dwell positions along with their associated standard deviations.
| PSD position | Measured∕planned per catheter | Measured∕planned per dwell position |
|---|---|---|
| Urethra—pos. 1 | 0.993 ± 0.021 | 0.994 ± 0.030 |
| Urethra—pos. 4 | 1.000 ± 0.015 | 0.995 ± 0.023 |
| Urethra—pos. 8 | 1.013 ± 0.014 | 1.011 ± 0.025 |
| Urethra—pos. 9 | 1.015 ± 0.019 | 1.014 ± 0.020 |
| Urethra—pos. 10 | 1.013 ± 0.018 | 1.007 ± 0.016 |
| Rectum—pos. 7 | 1.059 ± 0.045 | 1.025 ± 0.028 |
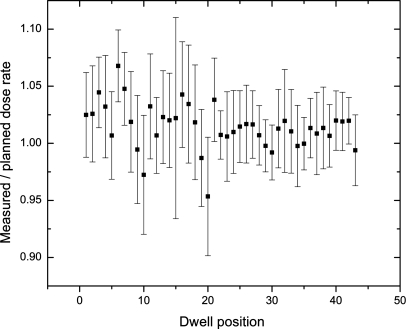
The dose rate for the 43 dwell positions with dwell times over 5 s were measured and compared to the expected dose rate obtained from the TPS. Figure 6 shows the ratios calculated when the PSD was inserted in the urethra and placed at position #9. These ratios ranged from about 0.953 to 1.068, with an average value of 1.014 ± 0.020. The averages were calculated for all PSD positions and are shown in Table Table II.. The ratios obtained with the PSD at the rectal wall ranged from 0.981 to 1.109, with an average value of 1.025 ± 0.028.
Figure 6.
Ratio of the dose rate measured at different dwell positions over the treatment to the calculated dose rate from the planning system. The plastic scintillation detector was inserted in the urethra aligned to dwell position #9 and only positions with dwell times over 5 s were selected (total of 43 positions). Uncertainty on measurements was obtained using the standard deviation from ten consecutive data points (1 s span) for the same dwell position.
Error detection
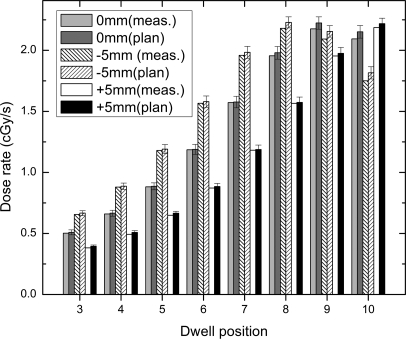
Our data revealed that it was easier to detect errors at the individual dwell position level as there is no chance for compensating effects from a lower dose read by a higher one, as could happen by integrating over individual catheter or over the entire treatment. The results presented in the following will refer to error detection by using the measurement from individual dwell positions. Figure 7 shows the measured and planned dose rates for the three conditions used in the error detection evaluation (+5 mm, −5 mm, 0 mm) for different dwell positions inside an arbitrarily chosen catheter. The error detection application described earlier was tested for various thresholds, and Table Table III. shows the number of true-errors and false-accurate doses determined for a −5 mm displacement of the catheters. The number of detected errors increased as the threshold decreased. Differences between the TPS and the measurements were rarely observed.
Figure 7.
Dose rates measured (meas.) and planned (plan) for different plastic scintillation detector positions in the urethra mimicking positioning errors of 5 mm in either direction. Shown is the dose delivered in catheter #6.
Table 3.
Number of true-errors and false-accurate detected using the PSD and expected from planning in function of the acceptable range. A −5 mm displacement of the catheters was introduced in the plan and the dose then delivered. When the PSD-measured dose rate exceeded the acceptable range value, an error was reported. The case represented here is from treatment delivery with the PSD inserted in the urethra and expected to be aligned with position #9.
| Acceptable range | |||||
|---|---|---|---|---|---|
| 3% | 5% | 10% | 20% | ||
| PSD | True-errors | 43 | 43 | 40 | 35 |
| False-accurate | 0 | 0 | 3 | 8 | |
| Plan | True-errors | 43 | 41 | 40 | 35 |
| False-accurate | 0 | 2 | 3 | 8 | |
Importance of stem effect removal
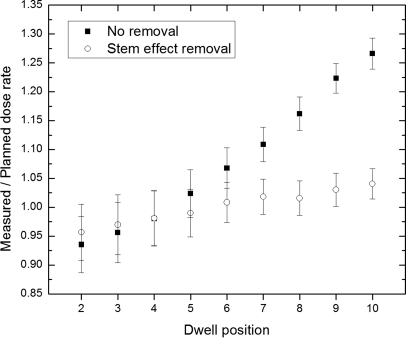
A demonstration of the importance of including a stem effect removal approach in a PSD system was performed by comparing results that were obtained without any stem effect removal to the results obtained with the approach implemented throughout this study. The ratio to the expected dose for the treatment was (1.006 ± 0.031) without stem effect removal and (0.996 ± 0.030) with it. The average ratio to expected dose delivery within each catheter was (1.016 ± 0.029) without the removal of the stem effect and (0.993 ± 0.021) with stem effect removal. Figure 8 shows the ratio of measured to expected dose—for all the dwell positions inside one of the catheters—on the basis of whether or not a stem effect removal technique was implemented. When no stem effect removal was performed, discrepancies between the expected and measured values up to 25% were observed. The detection of false-errors—situations ruled as an error, but that are not one in reality—with and without the use of a stem effect removal technique is reported in Table Table IV.. It shows that the use of a stem effect removal technique should considerably decrease the number of false-errors reported.
Figure 8.
Ratio of dose rate between the measured and planned values with and without chromatic removal of the stem effect. The plastic scintillation detector was inserted in the urethra aligned to dwell position #1, and the data shown here are for dose delivery in catheter #4. Uncertainty on measurements was calculated from the standard deviation from ten consecutive data points (1 s span) for the same dwell position.
Table 4.
Number of false-errors that would be flagged as a function of the acceptable range for a situation with and without chromatic removal of the stem effect.
| False-errors | ||||
|---|---|---|---|---|
| Chromatic removal | No removal | |||
| Error detection threshold | 3% | 9 | 27 | |
| 5% | 1 | 19 | ||
| 10% | 0 | 10 | ||
| 20% | 0 | 3 | ||
Temporal verification
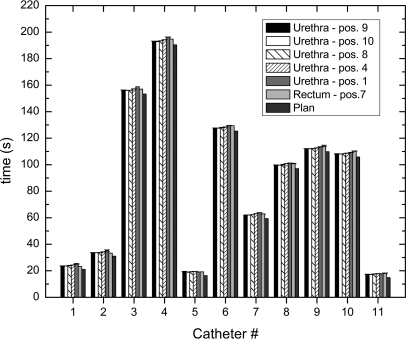
The time elapsed by the source in each of the catheters was measured and compared with the planned time. Figure 9 shows the calculated time in each catheter for each of the different PSD positions along with the treatment plan time. The calculated treatment times were smaller than the measured times by 2–4 s.
Figure 9.
Time elapsed for dose delivery in each of the 11 catheters for different plastic scintillation detector positions (pos.) in comparison with the expected time from the planning system. The error bars represent the standard deviation for three repeated deliveries.
DISCUSSION
Our results demonstrate the feasibility for real-time dosimetry by showing the accuracy of PSDs in 192Ir HDR brachytherapy treatments for a clinically relevant prostate treatment plan delivered in a water phantom. Figures 4a, 4b show the type of dosimetric information that could be available in real-time to the staff performing an HDR treatment. These figures show that most of the dwell positions could be temporally resolved. Unfortunately, the 5-s rise time of the current system represents a limitation for real-time measurements. Nevertheless, this issue could be resolved with a slight modification in the hardware of the electrometer. Even with this limitation, we showed that the PSD system was able to perform accurate dosimetry over the entire fraction delivery for each catheter and even down to specific dwell positions with dwell times over 5 s, and this was for clinically relevant air kerma strengths between 23 000 and 29 600 μGy m2 h−1. The source activity was in the lower part of the clinical range. Using a source with a higher activity would have enhanced the acuity of the PSD for dose measurements as the signal-to-noise ratio of each measurement would have been higher.
Doses and dose rates obtained with the PSD system were in good agreement with expected values obtained with the TPS for the entire fraction, for each catheter and for individual source positions. Values obtained from dosimetry at the rectal wall were less accurate than those performed in the urethra, as the PSD located at the rectal wall was on average farther from the source. This can be explained by a lower signal-to-noise ratio under these conditions of measurements. In the case we would have considered only the five closest catheters to the PSD (higher average dose rates), besides the 11 considered catheters, the ratio to the expected dose would have fallen from (1.059 ± 0.045) to (1.022 ± 0.025). Improvements in the signal-to-noise ratio of the electro-optical system would help to increase the accuracy of the detector under such conditions.
The use of the PSD dosimetry system to detect errors was shown to be accurate and relevant. Although a different number of errors were detected depending on the chosen acceptable range value, the only condition for which the experimental detection was slightly different from the treatment plan was in the 5% acceptable range. The plan led to a ratio just under the 5% threshold (ruled accurate), while the measurement led to a ratio just over the threshold (ruled an error). In a practical application, it will be important to consider the positioning and detector uncertainties when choosing the error threshold, particularly, if flagging an error will lead to altering the HDR treatment plan or halting treatment delivery. In practice, presence of measurements that are over the threshold could lead to the verification of PSD and catheters placement via imaging approaches in order to point at and correct the problem.
We also demonstrated that implementing a stem effect removal technique is important, especially when interested in performing dosimetry down to each dwell position. The observed discrepancies without a stem effect removal technique implemented should be considered unacceptable for performing in vivo dosimetry—with values differing from the expected ones by up to more than 25%. The study on false-error detection emphasized even more the necessity for a stem effect removal technique. The use of the chromatic stem effect removal technique considerably decreased the number of false-errors detected, decreasing the number from 19 to 1 for a 5% threshold out of 43 measurements. It decreased the number from 10 and 3 to 0 for the 10% and 20% thresholds, respectively. The many errors detected when the threshold was 3% indicate this threshold is too low for the actual system. The impact of stem effect was described here for a particular treatment plan. As we saw, there could be situations in which the PSD can perform well without the implementation of a stem effect removal technique, but there could be other clinically relevant situations in which the discrepancies would be even greater than those shown here. This could lead to an erroneous conclusion regarding the accuracy of dose delivery.
Although not the main purpose of the present article, the use of the PSD to perform temporal verification showed good promise. The fact that measured times were always a little longer than the expected times arose from two factors. First, the times defined on the treatment delivery report were calculated by adding the individual dwell times in the catheter, without taking into account the source transit time. The dose delivered to the point of interest, even if very small, was not taken into account in the TPS.26 Second, the rise or decay time must be considered. The time between the moment the 192Ir source left the catheter and the moment the PSD system output went below the threshold value was probably on the order of a couple of seconds. The latter probably represents the main explanation for the time difference between the recorded and expected values. However, even with this discrepancy, the system could be used for temporal verification, as most of the hypothetical errors (e.g., transfer tube inversions) would be detected. Temporal verification would be a good addition to a dosimetry verification system and would help in tracking the causes and effects of possible errors, including timer errors.
Previous work on developing real-time in vivo scintillation detectors for 192Ir brachytherapy was performed by other groups. Cartwright et al.16 developed a multidetector system to map dose on the rectal wall during 192Ir HDR brachytherapy. They used an electron-multiplying charge-coupled camera as a photodetector, as previously described by Archambault et al.,17 for in vivo dosimetry during intensity-modulated radiation therapy treatments. However, Cartwright et al. did not implement any stem effect removal technique in their system. They showed a 2%–3% accuracy for their detectors when using them under what could be considered as negligible stem effect conditions. However, their system failed to perform measurements within this accuracy range when irradiating under conditions susceptible to stem effect. This issue was discussed in length in one of our previous works.22 Even though the inclusion of a relevant and efficient stem effect removal technique represents an additional challenge in the development of the detector, we have shown the increase in versatility and accuracy of the PSD system to be very important and necessary in its future application in the clinic. Nevertheless, it should be noted from the study of Cartwright et al.16 that multiple PSDs were used, and this had the advantage that a detected error by a specific PSD could then be confirmed by at least one other detector. It might reduce the possibility for false-error or false-accurate statements from the system.
The development and application of another type of scintillation detector was performed by Andersen et al.14, 15 Their system, for which detection was based on radioluminescence and optically stimulated luminescence of an Al2O3 component attached to a polymethylmethacrylate optical fiber, was applied to 192Ir PDR brachytherapy treatments. They reported situations where the stem effect was up to 30% of the signal of interest in their detector.15 This supports the results of the present study as well as our previous findings.22 PDR brachytherapy makes use of a lower activity 192Ir source compared to HDR brachytherapy, but for longer dwell times. The sensitivity of the detector is then more crucial for PDR applications while there is less stress on temporal resolution of the detector. Andersen et al. showed a good agreement between measurements and calculated values from their TPS other than for the situations where the stem effect was predominant.14, 15 Their dosimetry system was used to perform time-resolved measurement during a clinical cervix PDR brachytherapy in vivo study.15 They assessed the ability to detect interchange of transfer tubes and applicator displacements. The authors showed their error detection to be particularly accurate when investigating down to dwell positions, being able to detect most of the errors for displacements of over 15 mm. A quick glance at our recorded data allowed us to determine that using measurement at the level of individual dwell position leads to a better efficiency in detecting source-to-detector positioning errors in comparison to the dose measurement per catheter or over the treatment. This is in agreement with the findings from Andersen et al.,15 even if the brachytherapy application was different (PDR vs HDR). We found our PSD system to be able to detect 5 mm offsets. However, this cannot be directly compared quantitatively to the results of Andersen et al. as the measurement conditions and error definitions were different. Still, our study was a step forward as they were based on mechanically imposed errors in positioning instead of being based on hypothetical errors from an uncertainty modeling.15 It should be mentioned that such an error analysis was easier to implement since our study was conducted in a water phantom and not in patients as it was the case for Andersen et al.15 Nevertheless, the important conclusion is that it confirms the positioning-error detection ability of scintillation detectors for 192Ir brachytherapy.
In order to move forward with the development of an in vivo clinical application of the PSDs for dose and position error detection, it will be important to ensure a proper positioning of the detector relatively to the dose delivery catheters. The PSDs will have to be grossly inserted in the organs of interest and their exact positioning relatively to organs and dose delivery catheters calculated during the pretreatment imaging step. In the case the detector is not evident to see on images (likely, due to its water equivalence), the use of small radio-opaque markers might be necessary. This issue will be important to address.
CONCLUSION
The PSD system used in this study was shown to be a good fit for dosimetry during 192Ir HDR brachytherapy treatments. A clinically relevant treatment was planned and delivered to a phantom, and system feasibility for dose verification over the whole treatment, for each specific catheter, and for specific dwell positions was demonstrated. Scintillation detectors show advantages that make them more suited to the particular characteristics and requirements of HDR brachytherapy in vivo dosimetry than other detectors. Our PSD system outperformed previously developed plastic scintillation detectors. Its accuracy was shown to be dependent on the dosimeter positioning and could be enhanced by an increase of the signal-to-noise ratio. The implementation of a stem effect removal approach increased its accuracy. The system was shown to be capable of detecting source misplacements and even allowed for temporal verification of treatment delivery. The latter represents potential treatment verification applications for 192Ir HDR brachytherapy using our PSD system.
ACKNOWLEDGMENTS
This work was supported in part by a National Cancer Institute of Canada grant (017133), a Natural Sciences and Engineering Research Council Discovery grant (262105), and a National Cancer Institute grant (1R01CA120198-01A2). F.T.-P. was supported by the Natural Sciences and Engineering Research Council and Fonds Québécois de la Recherche sur la Nature et les Technologies. We would also like to thank Kathryn Carnes from UT MD Anderson Cancer Center, Department of Scientific Publications for her help in editing this article.
References
- Meigooni A., Meli J., and Nath R., “Influence of the variation of energy spectra with depth in the dosimetry of 192Ir using LiF TLD,” Phys. Med. Biol. 33, 1159–1170 (1988). 10.1088/0031-9155/33/10/005 [DOI] [PubMed] [Google Scholar]
- Kirov A. S., Williamson J. F., Meigooni A. S., and Zhu Y., “TLD, diode and Monte Carlo dosimetry of an 192-Ir source for high dose-rate brachytherapy,” Phys. Med. Biol. 40, 2015–2036 (1995). 10.1088/0031-9155/40/12/002 [DOI] [PubMed] [Google Scholar]
- Rustgi S. N., “Application of a diamond detector to brachytherapy dosimetry,” Phys. Med. Biol. 43, 2085–2094 (1998). 10.1088/0031-9155/43/8/007 [DOI] [PubMed] [Google Scholar]
- Brezovich I. A., Duan J., Pareek P. N., Fiveash J., and Ezekiel M., “In vivo urethral dose measurements: A method to verify high dose rate prostate treatments,” Med. Phys. 27, 2297–2301 (2000). 10.1118/1.1312811 [DOI] [PubMed] [Google Scholar]
- Waldhausl C., Wamberside A., Potter R., and Georg D., “In-vivo dosimetry for gynaecological brachytherapy: Physical and clinical considerations,” Radiother. Oncol. 77, 310–317 (2005). 10.1016/j.radonc.2005.09.004 [DOI] [PubMed] [Google Scholar]
- Kinhikar R. A., Sharma P. K., Tambe C. M., and Deshpande D. D., “Dosimetric evaluation of a new OneDose MOSFET for Ir-192 energy,” Phys. Med. Biol. 51, 1261–1268 (2006). 10.1088/0031-9155/51/5/015 [DOI] [PubMed] [Google Scholar]
- Lambert J., McKenzie D. R., Law S., Elsey J., and Suchowerska N., “A plastic scintillation dosimeter for high dose rate brachytherapy,” Phys. Med. Biol. 51, 5505–5516 (2006). 10.1088/0031-9155/51/21/008 [DOI] [PubMed] [Google Scholar]
- Das R., Toye W., Kron T., Williams S., and Duchesne G., “Thermoluminescence dosimetry for in-vivo verification of high dose rate brachytherapy for prostate cancer,” Australas. Phys. Eng. Sci. Med. 30, 178–184 (2007). 10.1007/BF03178424 [DOI] [PubMed] [Google Scholar]
- Lambert J., Nakano T., Law S., Elsey J., McKenzie D. R., and Suchowerska N., “In vivo dosimeters for HDR brachytherapy: A comparison of a diamond detector, MOSFET, TLD, and scintillation detector,” Med. Phys. 34, 1759–1765 (2007). 10.1118/1.2727248 [DOI] [PubMed] [Google Scholar]
- Fagerstrom J. M., Micka J. A., and DeWerd L. A., “Response of an implantable MOSFET dosimeter to 192-Ir HDR radiation,” Med. Phys. 35, 5729–5737 (2008). 10.1118/1.3013574 [DOI] [PubMed] [Google Scholar]
- Daskalov G. M., Loffler E., and Williamson J. F., “Monte-Carlo-aided dosimetry of a new high dose-rate brachytherapy source,” Med. Phys. 25, 2200–2208 (1998). 10.1118/1.598418 [DOI] [PubMed] [Google Scholar]
- Qi Z.-Y., Deng X.-W., Huang S.-M., Lu J., Lerch M., Cutajar D., and Rosenfeld A., “Verification of the plan dosimetry for high dose rate brachytherapy using metal-oxide-semiconductor field effect transistor detectors,” Med. Phys. 34, 2007–2013 (2007). 10.1118/1.2736288 [DOI] [PubMed] [Google Scholar]
- Zilio V., Joneja O., Popowski Y., Rosenfeld A., and Chawla R., “Absolute depth-dose-rate measurements for an 192Ir HDR brachytherapy source in water using MOSFET detectors,” Med. Phys. 33, 1532–1539 (2006). 10.1118/1.2198168 [DOI] [PubMed] [Google Scholar]
- Andersen C. E., Nielsen S. K., Greilich S., Helt-Hansen J., Lindegaard J. C., and Tanderup K., “Characterization of fiber-optic coupled Al2O3:C luminescence dosimetry system for online in vivo dose verification during 192Ir brachytherapy,” Med. Phys. 36, 708–718 (2009). 10.1118/1.3063006 [DOI] [PubMed] [Google Scholar]
- Andersen C. E., Nielsen S. K., Lindegaard J. C., and Tanderup K., “Time-resolved in vivo luminescence dosimetry for online error detection in pulsed dose-rate brachytherapy,” Med. Phys. 36, 5033–5043 (2009). 10.1118/1.3238102 [DOI] [PubMed] [Google Scholar]
- Cartwright L. E., Suchowerska N., Yin Y., Lambert J., Haque M., and McKenzie D. R., “Dose mapping of the rectal wall during brachytherapy with an array of scintillation dosimeters,” Med. Phys. 37, 2247–2255 (2010). 10.1118/1.3397446 [DOI] [PubMed] [Google Scholar]
- Archambault L., Briere T. M., Ponisch F., Beaulieu L., Kuban D. A., Lee A., and Beddar S., “Toward a real-time in vivo dosimetry system using plastic scintillation detectors,” Int. J. Radiat. Oncol. Biol. Phys. 78, 280–287 (2010). 10.1016/j.ijrobp.2009.11.025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beddar A. S., Mackie T. R., and Attix F. H., “Water-equivalent plastic scintillation detectors for high-energy beam dosimetry: II. Properties and measurements,” Phys. Med. Biol. 37, 1901–1913 (1992). 10.1088/0031-9155/37/10/007 [DOI] [PubMed] [Google Scholar]
- Beddar A. S., Mackie T. R., and Attix F. H., “Water-equivalent plastic scintillation detectors for high-energy beam dosimetry: I. Physical characteristics and theoretical considerations,” Phys. Med. Biol. 37, 1883–1900 (1992). 10.1088/0031-9155/37/10/006 [DOI] [PubMed] [Google Scholar]
- Archambault L., Beddar A. S., Gingras L., Roy R., and Beaulieu L., “Measurement accuracy and Cerenkov removal for high performance, high spatial resolution scintillation dosimetry,” Med. Phys. 33, 128–135 (2006). 10.1118/1.2138010 [DOI] [PubMed] [Google Scholar]
- Beddar A. S., Mackie T. R., and Attix F. H., “Cerenkov light generated in optical fibres and other light pipes irradiated by electron beams,” Phys. Med. Biol. 37, 925–935 (1992). 10.1088/0031-9155/37/4/007 [DOI] [Google Scholar]
- Therriault-Proulx F., Beddar S., Briere T. M., Archambault L., and Beaulieu L., “Technical note: Removing the stem effect when performing Ir-192 HDR brachytherapy in vivo dosimetry using plastic scintillation detectors—A relevant and necessary step,” Med. Phys. 38, 2176–2179 (2011). 10.1118/1.3562902 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nath R., Anderson L. L., Luxton G., Weaver K. A., Williamson J. F., and Meigooni A. S., “Dosimetry of interstitial brachytherapy sources: Recommendations of the AAPM Radiation Therapy Committee Task Group No. 43,” Med. Phys. 22, 209–234 (1995). 10.1118/1.597458 [DOI] [PubMed] [Google Scholar]
- Fontbonne J. M., Iltis G., Ban G., Battala A., Vernhes J. C., Tillier J., Bellaize N., LeBrun C., Tamain B., Mercier K., and Motin J. C., “Scintillating fiber dosimeter for radiation therapy accelerator,” IEEE Trans. Nucl. Sci. 49, 2223–2227 (2002). 10.1109/TNS.2002.803680 [DOI] [Google Scholar]
- Guillot M., Gingras L., Archambault L., Beddar S., and Beaulieu L., “Spectral method for the correction of the Cerenkov light effect in plastic scintillation detectors: A comparison study of calibration procedures and validation in Cerenkov light-dominated situations,” Med. Phys. 38, 2140–2150 (2011). 10.1118/1.3562896 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahoo N., “Measurement of transit time of a remote after-loading high dose rate brachytherapy source,” Med. Phys. 28, 1786–1790 (2001). 10.1118/1.1386779 [DOI] [PubMed] [Google Scholar]