Abstract
This review, comprised of our own data and that of others, provides a summary overview of histone deacetylase (HDAC) inhibition on intestinal inflammation as well as inflammation-mediated carcinogenesis. Experimental colitis in mice represents an excellent in vivo model to define the specific cell populations and target tissues modulated by inhibitors of HDAC. Oral administration of either suberyolanilide hydroxamic acid (SAHA) or ITF2357 results in an amelioration in these models, as indicated by a significantly reduced colitis disease score and histological score. This effect was paralleled by suppression of proinflammatory cytokines at the site of inflammation as well as specific changes in the composition of cells within the lamina propria. In addition, tumor number and size was significantly reduced in two models of inflammation-driven tumorigenesis, namely interleukin (IL)-10–deficient mice and the azoxymethane–dextran sulfate sodium (DSS) model, respectively. The mechanisms affected by HDAC inhibition, contributing to this antiinflammatory and antiproliferative potency will be discussed in detail. Furthermore, with regard to the relevance in human inflammatory bowel disease, the doses of ITF2357 considered safe in humans and the corresponding serum concentrations are consistent with the efficacious dosing used in our in vivo as well as in vitro experiments. Thus, the data strongly suggest that HDAC inhibitors could serve as a therapeutic option in inflammatory bowel disease.
INTRODUCTION
Inflammatory bowel disease is a chronic disorder that accompanies patients throughout their lives. Two main forms can be distinguished, namely Crohn’s disease and ulcerative colitis. Whereas the treatment options have dramatically improved over the last decade, in particular because of the introduction of anti–tumor necrosis factor (TNF)-α strategies (infliximab and adalimumab), a relevant subset of patients still exists in whom satisfactory therapy is required.
To identify novel therapeutic targets, we must focus on the pathogenesis of the disease first. The genome-wide association studies of the last years have contributed significantly to the understanding of the pathogenesis of inflammatory bowel disease (IBD) (1). The results obtained from the genome-wide association studies not only confirmed the relevance of earlier characterized pathways, but equally opened novel avenues. If one must suggest a hypothesis on the etiology of IBD, one could simply say that the mucosal immune system is hyperresponsive to luminal antigens (for example, dietary factors, commensal bacteria) in genetically predisposed individuals (2).
A critical long-term complication of chronic inflammation (in particular, of the colon) is the development of colorectal cancer. The risk increases significantly with duration as well as with extension of disease; thus, a chronically active pan-colitis is associated with the highest risk (3). Historically, the idea that a chronic inflammatory condition represents a predisposing factor for developing cancer is well known, and other examples are chronic hepatitis, pancreatitis and esophagitis (4).
In the example of colorectal cancer, the mechanism by which inflammation induces cancer development has not been elucidated; however, it is clear that the sequence differs from the pathway activated in “non-inflammatory” colorectal cancer (5). Several lines of evidence, experimental as well as clinical data, suggest that antiinflammatory strategies and, as a consequence, mucosal healing contribute to preventing colorectal cancer. Thus, the identification of novel therapeutic strategies that exert antiinflammatory and possibly antiproliferative properties is desirable.
In addition to the identification of genetic polymorphisms associated with either Crohn’s disease or ulcerative colitis, this review will serve to shed light on a novel way of regulation that might result in a relevant impact in chronic intestinal inflammation, (namely epigenetic modifications and in particular changes driven by histone deacetylases [HDACs]).
Our own in vivo data from models of acute and chronic intestinal inflammation in mice as well as inflammation-driven tumorigenesis strongly indicate that inhibition of HDAC represents a highly effective therapeutic option. Recent human data (6) provide solid evidence that the novel HDAC inhibitor ITF2357 can be administered in doses sufficient to exert this antiinflammatory capacity; thus, it seems that these compounds might in fact provide a future pharmacological strategy for patients suffering from IBD.
REVIEW OF THE LITERATURE
Before analyzing the various pathways of how HDAC inhibition might affect the mucosal immune response, an overview of the cell populations known to contribute to intestinal inflammation will be provided. For defined cell populations as well as affected mechanisms, the current literature will be reviewed of how HDAC inhibition might influence their respective function and thus prevent or enhance the effector response.
Acetylation of Nonhistone Proteins
It is well known that histone acetyl transferases (HATs) do not acetylate histones exclusively, and equally HDACs do not only deacetylate histones. Thus, the term “histone” in this context is merely historical, and the use of lysine deacetylases (KDACs) best represents the biological activity of these molecules. While it has been described that distinct nonhistone proteins (for example, p65, STAT3 and HIF1α) are affected by HAT and HDAC, Choudhary et al. (7) recently demonstrated that various cellular networks, for example, RNA processing, DNA damage repair, cell cycle control and nuclear transport, are widely affected by protein acetylation. Analysis of the so-called “acetylome” (8) revealed a complex regulatory network of cellular protein acetylation with several thousand identified acetylation sites. Yet clearly defined signaling pathways, as known for other protein modifications, are still missing (9). These studies should be considered when discussing or investigating the effects of HDAC or HDAC inhibitors, respectively. Because inhibition of deacetylases produces open chromatin sites all over the genome, changes in the expression of distinct proteins are not necessarily attributed to these chromatin changes but are more likely attributed to the modulation of the cellular acetylome.
T Helper Cells
T lymphocytes represent the key cell population of the adaptive immune system. Once activated, T cells secrete cytokines and affect all other cell types within a local environment (macrophages, dendritic cells, neutrophils, epithelium, endothelial cells, stromal elements). Both human and murine studies revealed that different T-cell subpopulations are aberrantly activated in Crohn’s disease versus ulcerative colitis (10,11). A dominant role for the T helper cell type 1 (Th1)-mediated immune response could be described early on for models of intestinal inflammation (12). Recently, an additional Th subset was identified (so-called Th17 cells) (13). These cells produce interleukin (IL)-17 and IL-22, both proinflammatory cytokines capable of promoting local tissue destruction. Although Th17 cells are generated from Th0 cells by the combined effects of IL-6 and transforming growth factor (TGF)-β, there is an absolute requirement for IL-1 (14). In mice deficient in IL-1RI, there is no Th17 response (15). Once generated, Th17 cells are maintained by IL-23 (16). IL-23 belongs to the IL-12 family and shares the p40 subunit with IL-12. IL-17 as well as IL-22 can be found at increased levels in inflamed Crohn’s disease mucosa, suggesting that these cytokines contribute to the disease pathogenesis (17,18). Functional data from mice and men are in support of this hypothesis, as described in more detail below. An additional Th subset is the Th2 cell. These cells secrete IL-4, IL-5 and IL-13 (19). Th2 cells promote atopy with induction of IgE responses as well as eosinophil and mast cell activation. Ulcerative colitis was thought to represent a Th2-driven disease, but the absence of IL-4 in colonic tissue from ulcerative colitis patients and the observation that both IL-13 and interferon (IFN)-γ are found at elevated levels in ulcerative colitis mucosa changed this dogma (20). Recent data suggest that IL-13 originates from a natural killer (NK) T cell and targets the epithelial cell to become dysfunctional (10,21).
Lastly, there are regulatory T-cell sub-populations. This is of particular importance, since the immunological defense of the intestine, in contrast to the systemic immune system, is one of suppression. Healthy individuals generally do not develop systemic immune responses against the commensal flora or dietary antigens. Regulatory cells are responsible for the immunologically suppressed milieu in the intestinal mucosa. One group of regulatory cells contributing to this milieu are regulatory T cells (Tregs) (22,23). Various subsets have been described as being involved in maintaining a condition of tolerance within the intestinal mucosa. With regard to HDACs and the available data, we restrict the view here on the FoxP3+ Treg population that requires the transcription factor FoxP3 (22,23). Remarkably, the absence of FoxP3 in humans (IPEX syndrome) (24,25) and mouse (Scurfy) is followed by an autoimmune endocrine disorder, immunodeficiency and an enteropathy mainly affecting the small bowel. In the following sections, we will describe in detail of how HDAC inhibition can modulate Treg number and function.
T helper cells type 1 and 2
In the nucleus of Th cells, class I HDAC proteins are present within SIN3 (yeast Switch INdependent) or NuRD (Nucleosome-Remodeling and histone Deacetylation complex) complexes (26) and are ubiquitously expressed (27). Here, the HDACs provide the deacetylase capability, whereas SIN3 provides the functionality to interact with various repressor molecules. NuRD complexes also promote histone deacetylation via HDAC1 and −2, but are, in addition, responsible for nucleosome remodeling via Mi-2 proteins, which build the central part of these complexes (28). Proliferating CD4+ T cells recruit Sin3-HDAC to the IFNγ locus. T-bet, the key transcription factor of Th1 cells, displaces Sin3-HDAC and allows HAT to acetylate the IFNγ locus, resulting in an IFNγ transcription and consequently a Th1 polarization (29). Thus, HDAC inhibitors are able to acetylate the IFNγ locus in naïve T cells, leading to a recruitment of transcription factors such as T-bet or STAT4. These mechanistic data are controversial, since the dominant effect of HDAC inhibitors is antiinflammatory and thus not driven by Th1 cells.
In addition, T-bet has been described to interact indirectly with HDAC3 and HDAC5, a member of class II HDAC (30). Here, T-bet demonstrates a binding capacity to GATA-3, the respective key transcription factor of Th2 cells, CBP/P300 and Sp1 to form a transcription factor complex. From within this complex, GATA-3 regulates FUT7 transcription by phosphorylation-dependent recruitment of HDAC3/HDAC5 and by competing with CBP/P300 in binding to the N terminus of T-bet (30). This transcriptional regulation by GATA-3/T-bet and HDAC3/HDAC5 interactions is a possible way to explain lineage-specific (Th1/Th2) protein expression, which is of particular interest in IBD, where the Th1/Th2 balance was taken for years as one feature of Crohn’s disease (Th1) or ulcerative colitis (Th2). Although this exact discrimination is not further supported according to the recent literature (31), the cytokine milieu at the site of inflammation represents a crucial characteristic, defining progress and severity of IBD, thus important both for diagnosis as well as for therapy.
A variety of HDAC inhibitors, such as butyrate, trichostatin A (TSA) or Script -aid, induce anergy in Th cells in vitro (32), as characterized by an inhibition of proliferation accompanied by a reduction of IL-2 production in Th1 cell cultures. For TSA and suberyolanilide hydroxamic acid (SAHA), this scenario was attributed in cell lines to chromatin remodeling at the IL-2 promoter as well as acetylation of transcription factors such as nuclear factor (NF)-κB (33,34).
Regulatory T cells and Th17 cells
Koenen et al. (35) reported that TSA as an HDAC inhibitor induces a profound negative effect on the emergence of IL-17-producing cells from Tregs. Whereas sorted regulatory T cells can be polarized from the Treg phenotype to Th17 cells in a defined cytokine milieu, this can be prevented by the presence of TSA (35).
A more specific concept was described for regulatory T cells (36). Administration of pan-HDAC inhibitors, but not class I–specific inhibitors in vivo in mice, increased Foxp3 gene expression, as well as the production and suppressive function of regulatory T cells in vitro. HDAC9 proved particularly important in regulating Foxp3-dependent suppression, as demonstrated by in vivo data in HDAC9-knockout mice (37). Optimal function of regulatory T cells required acetylation of several lysines in the forkhead domain of Foxp3 (38), connecting the results of Koenen et al. to the HDAC9/FoxP3 data. The same group recently suggested a link between the presence of HDAC9, Treg functionality and heat shock protein 70 (HSP70) expression. Although there was a clear link between the Treg function and HSP70 upregulation in HDAC9-knockout experiments, the direct interaction between HDAC9 and HSP70 has not been confirmed yet (39).
Dendritic Cells and Macrophages
Besides the described direct effects on Th cells, HDAC inhibitors are able to affect the influence of antigen-presenting cells on Th cells. Two different HDAC inhibitors, TSA and SAHA, have been described to modulate human dendritic cell (DC) function and particularly, the production of proinflammatory cytokines in vivo, namely IL-12/IL-23. Additionally, HDAC inhibitors were found to block the production of the Th1-attracting chemokines CXCL9, CXCL10 and CXCL11 in vitro (40). A similar effect was demonstrated for bone marrow–derived DCs and the HDAC inhibitors apicidin (41) as well as LAQ284 (42). The production of proinflammatory cytokines IL-12 and TNFα was suppressed, paralleled by an inhibited mitogen-activated protein (MAP) kinase activation and NF-κB translocation to the nucleus. The treated DCs were not able to induce a Th1 response or Th1 cell migration. Recently, pan-HDAC inhibitor (SAHA and ITF2357) treatment was shown to promote STAT3 acetylation in DCs (43,44), paralleled by an induction of in-doleamine 2,3-dioxygenase, which is known to be an inhibitor of T-cell activation, another link to the modulated DC effect on T cells. The particular effect of HDAC inhibition on DCs and macro -phages is further discussed within this issue by Pavan Reddy.
Alternatively, macrophage activation via lipopolysaccharide (LPS) alters the expression of HDAC in these cells. Class I as well as class II HDAC expression was induced after LPS stimulation of bone marrow–derived macro phages (45). While T-cell activation and polarization class II HDACs seem to play a dominant role, in macrophages, it was shown that class I HDACs abolish LPS-induced Cox-2 expression in RAW264.7 macrophages (45,46). In addition, HDAC2 was required for glucocorticoid-induced suppression of inflammatory mediator production from human alveolar macrophages (47). Thus, the inhibition of class I HDACs might provide a therapeutic target for the modulation of macrophage responses in inflammatory diseases.
As in DCs, LAQ284 was reported to inhibit monocyte chemotaxis (via MCP1-3 or CCL15/23) as well as Th1 molecules (CD40, IL12, IP10), whereas Th2 molecules remained unchanged (42). For LPS-mediated migration of macrophages themselves, butyrate and TSA reduced migration by suppression of Src enhancement and focal adhesion kinase activity in both RAW264.7 and rat peritoneal macrophages (48). For synovial macrophages, an inhibition of class I/II HDACs or class III nicotinamide adenine dinucleotide (NAD)-dependent sirtuin HDACs can suppress inflammatory activation of these cells from patients with rheumatoid arthritis. The HDAC inhibitors were reported to promote apoptosis as well as modulation of the expression of various proinflammatory cytokines (49). Cytokine inhibition equally shown for the HDAC inhibitor KBH-A42 in RAW 264.7 cells is paralleled by a suppressed MAP kinase phosphorylation (50).
Methylation
Because there are many ways for HDAC inhibitors to modulate cellular phenotypes, one should not underestimate the field of origin for these compounds, the epigenetic modification at the DNA or the histones. While it is known that various cross-talks between (histone) methylation and histone acetylation exist, their impact on T-cell polarization was shown recently by Lal et al. (51). They identified a highly conserved CpG island within a FoxP3 enhancer region that is methylated in peripheral CD4+ T cells but not in natural regulatory T cells. Here, this region was histone H3 acetylated and bound by Sp1 and TGF-inducible early gene-1 (TIEG1). Removing this methylation in non-Tregs resulted in an increased and stable FoxP3 expression. This effect is similar to other reported modulations of FoxP3 expression via methylation (52).
MicroRNA
Posttranscriptional regulation of gene expression by microRNA (miRNA) is another phenomenon among the epigenetic mechanisms functionally affected by HDAC inhibitors. LAQ824 is followed by rapid and significant changes in 40% of the different miRNA species expressed in SKBr3, a cancer cell line, with 22 miRNA species downregulated and 5 miRNAs upregulated (53). Although these experiments were not performed in immune cells, it is obvious that miRNA expression modulated by HDAC inhibitors represents a promising new field to investigate the mode of action of these compounds, since today, miRNAs are emerging as key regulators of the immune response (54). Thus, a recent study focused on the alteration of miR-146 by the HDAC inhibitors SAHA and ITF2357. This miRNA is widely known as a negative regulator of LPS-driven innate immune responses (55). At least in murine glia cells, this miRNA undergoes no differential expression in the presence of the HDAC inhibitors analyzed (56).
Regulation of Transcription Factors: NF-κB
Acetylation and deacetylation represent regulatory mechanisms for the activation or inactivation of selected transcription factors, such as NF-κB. In parallel to his-tones, this process is mediated by the balance of HAT/HDAC and can thus be modulated by HDAC inhibitors (57).
NF-κB represents a key transcription factor at the border between inflammation and cancer (4). Blocking the NF-κB pathway in myeloid cells in the azoxy-methane (AOM)–dextran sulfate sodium (DSS) model resulted in an inhibition of intestinal inflammation as well as in a decrease in tumor number and size. In contrast, blocking the NF-κB pathway in intestinal epithelial cells did not affect inflammation but did equally prevent tumorigenesis (58). This ambivalent character of NF-κB in intestinal epithelial versus myeloid cells within the inflamed lamina propria was supported by the data from other groups. Here, oral treatment of mice with the NF-κB essential modulator (NEMO)-binding peptide, an inhibitor of NF-κB, resulted in an amelioration of experimental colitis (59). In addition, selective inhibition of NF-κB in intestinal epithelial cells via cell-specific NEMO knockout was followed by severe chronic intestinal inflammation, thus further supporting the concept that NF-κB is essential in the control of epithelial integrity. In detail, NF-κB deficiency led to apoptosis of intestinal epithelial cells, impaired expression of antimicrobial peptides and facilitated translocation of bacteria into the mucosa (60).
How do HDACs participate in this critical signaling pathway? Chen et al. (61) suggested that the NF-κB subunit p65 acts as a substrate for either HAT or HDAC within the nucleus of an activated cell. While the acetylated form interacts weakly with IκBα, p65 can be deacetylated through a specific interaction with HDAC3 (61). This deacetylation promotes effective binding to IκBα and allows the nuclear export. Although this would implicate a translocation of p65 into the nucleus, and a subsequent hyperactivation of NF-κB by HDAC inhibition, there have been various studies describing contradictory results regarding the application of HDAC inhibitors in vitro (62–65). These conflicting data might in part be related to different cell lines, inhibitor concentrations as well as experimental design. Our own studies, where we could prove an antiinflammatory potency in addition to a tumor- preventing effect in multiple murine models, equally demonstrated a reduced translocation of p65 into the nucleus in vivo at the site of inflammation and tumorigenesis in the colon as well as in vitro using U937 cells (66). One study suggested a concept behind this effect by focusing on the transcription factor Stat1, which exists in an acetylated and deacetylated state. Inhibitors of HDAC induce Stat1 acetylation, and only acetylated Stat1 interacts with p65, resulting in a NF-κB inactivation by translocation of p65 to the cytoplasm (67).
Impact of HDAC in the Regulation of Experimental Colitis
As described in detail below, inhibition of HDAC results in an amelioration in various models of experimental colitis, as indicated by data from our group (66,68). As already introduced above, there are several classes and thus even more HDACs (69). The majority of currently available HDAC inhibitors are so-called pan-HDAC inhibitors, which inhibit the activity of a broad spectrum of HDACs (70). The ultimate goal will be to define the relevance of a single HDAC within the process of intestinal inflammation. At this point, there are only limited data on this issue.
The group of Wayne Hancock provided data characterizing the impact of HDAC9, a member of the class II HDACs (37). Background on their work showed that pan-HDAC inhibitors, but not class I–specific HDAC inhibitors, increased the function of Foxp3+ Tregs. As a consequence, these inhibitors prevented and reduced established colitis in mice. These pan-HDAC inhibitor– mediated effects were associated with increased numbers of Foxp3+ Tregs within the lamina propria. Because colitis was in addition associated with an increased local expression of HDAC9, HDAC9-knockout mice were subsequently investigated in models of experimental colitis, where they proved to be resistant. Tregs from HDAC9-knockout mice expressed increased levels of HSP70 compared with controls. Immunoprecipitation experiments indicated an interaction between HSP70 and Foxp3. Inhibition of HSP70 reduced the antiinflammatory efficacy of HDAC9-knockout Tregs, while Tregs that overex-pressed HSP70 exerted an enhanced suppressive function (39).
The therapeutic potency of manipulating FoxP3 acetylation via class II HDAC inhibition could be confirmed in other inflammatory models such as transplant rejection or arthritis (37,39,71,72).
Other Models of Inflammation
HDAC inhibition reduces cytokine production, particularly cytokines relevant to autoimmune/inflammatory diseases. Of particular importance is the effect of nanomolar concentrations of ITF2357 on TNFα and IFNγ gene expression and synthesis as well as the secretion of IL-1β in peripheral blood mononuclear cells in vitro (73). These results were confirmed in vivo investigating inflammatory models such as concanavalin A–induced hepatitis or LPS-induced shock in mice. This issue of Molecular Medicine discusses the effects of HDAC inhibitors on various inflammatory diseases and models such as transplant rejection or arthritis; however, we will focus on intestinal inflammation and carcinogenesis.
REVIEW OF OUR DATA
Antiinflammatory In Vitro
The described antiinflammatory properties could be confirmed by our group for various HDAC inhibitors independent of their structural classes in different human and murine cell types. Compounds included in these studies were SAHA, ITF2357, valproic acid, TSA and apicidin (68). They all proved to induce a suppression of proinflammatory cytokines. However, they differed significantly in their effective active concentration (for example, SAHA is 1,000 times more effective than valproic acid). Although the precise mechanism behind the antiinflammatory efficacy is still unknown, several studies offer possible explanations.
Antiinflammatory In Vivo
There are several models of chemically induced experimental colitis, each reflecting specific characteristics of the human disease. As an example, the models of trinitrobenzene- and oxazolone- induced colitis represent Th1- or Th2- mediated models of colitis, respectively (74,75). In contrast, the model of acute, short-duration DSS-induced colitis is not a model of immunological origin, but rather a model of mucosal inflammation resulting from the disruption of the epithelial barrier. Nevertheless, this model is well established and widely applied to evaluate novel pharmacological compounds for their antiinflammatory capacity.
The antiinflammatory potency of HDAC inhibitors was evaluated in these different models of colitis (68). HDAC inhibitors of different classes (the hydroxamic acids SAHA and ITF2357 as well as the short-chain fatty acid valproic acid) suppressed the inflammatory parameters in acute DSS-induced colitis, as evaluated by monitoring weight loss, bleeding and colon shortening as well as repeated examinations via lower endoscopy (66). Remarkably, five times lower doses of ITF2357 (10 mg/kg) when compared with SAHA (50 mg/kg) did suffice to exert the antiinflammatory effect in our in vivo models. In any of our in vivo studies, either substance was administered orally. This dosing resulted in a profound suppression of the production of proinflammatory cytokines at the site of inflammation for ITF2357 as well as for SAHA. Considering the human data available, here a dose of 50 mg orally resulted in a serum concentration of 104 nmol/L, sufficient to inhibit cytokine synthesis (6). From a study in children with juvenile idiopathic arthritis, we know that 1.5 mg/kg in two divided doses of ITF2357 is safe (76). Thus, the concentrations required for mediating the antiinflammatory potency in humans can be reached, thus indicating that these agents might become a therapeutic option in treating IBD.
These macroscopic data were paralleled by a reduction of proinflammatory cytokines at the site of inflammation in colon cultures and further underlined by a reduced histological inflammation score (68). The effect on trinitrobenzene-induced colitis was comparable. In both models, the tested hydroxamic acids were superior to valproic acid with regard to the antiinflammatory potency.
Although nearly all of the effects described above might contribute to the amelioration colitis, we observed an induction of apoptosis in the isolated lamina propria mononuclear cells from treated animals. This observation corresponds to that in healthy humans, where lamina propria mononuclear cells are highly susceptible to Fas-mediated apoptosis, whereas lamina propria mononuclear cells from patients with Crohn’s disease have been shown to be resistant to multiple apoptotic pathways (77). Here, an established treatment is represented by infliximab, an anti-TNFα antibody, resulting in monocyte- and caspase-3–dependent T-cell apoptosis (78).
Anti–NF-κB
As stated above, the effect on the activation of NF-κB has been discussed controversially (62–65). p65 is acetylated within the nucleus by the HAT p300 and deacetylated by HDAC3 (61), allowing for the relocation of p65 into the cytoplasm. Affecting this fundamental process with HDAC inhibitors should result in an accumulation of p65 in the nucleus and subsequently a hyperactivation of NF-κB. Although some groups could confirm this effect, others, including our own group, reported the opposite: in these studies, NF-κB activation was reduced and the translocation of p65 into the nucleus was inhibited. We could observe this effect on HDAC inhibitor–treated cell lines in vitro as well as in whole colon samples taken from in vivo–treated mice (66). Our data suggest that the compounds affect the pathway upstream of the separation of IκB and the p50/p65 complex.
Inflammation/Cancerogenesis
Given the profound antiinflammatory effect in the models of experimental colitis as well as the generally known anti -proliferative capacity of HDAC inhibitors, one would assume a strong tumor- preventing capacity of HDAC inhibition in cases where chronic inflammation is clearly associated with a subsequent tumorigenesis. To approach this hypothesis, two models of inflammation-associated cancerogenesis, mediated by chronic colitis, where tumors develop at the site of inflammation, were investigated.
The first model is the AOM/DSS model, which is based on a single injection of the carcinogen azoxymethane (AOM) followed by induction of a chronic DSS colitis (79). The development from initial dysplasia to full-grown adenomas was monitored by lower endoscopy in the living mice. Here, the continuous treatment with either SAHA or ITF2357 attenuated both tumor growth and development. Again, ITF2357, as seen in the colitis models, was more potent than SAHA (66,80).
In the second model, the IL-10 knockout mice were evaluated. In this model, no chemical carcinogen was required and the mice spontaneously developed colitis and, with age, dysplasia, which progressed to infiltrating carcinomas. This progression of tumor development was accelerated by a 5-day treatment with the cyclooxygenase 2 inhibitor celecoxib (80). Although HDAC inhibitors (ITF2357) were not administered until week 12, the treatment was associated with an inhibition of tumorigenesis within the following 4 weeks, demonstrating a high efficacy of ITF2357 in this therapeutic model of inflammation-mediated carcinogenesis (66) (Figure 1).
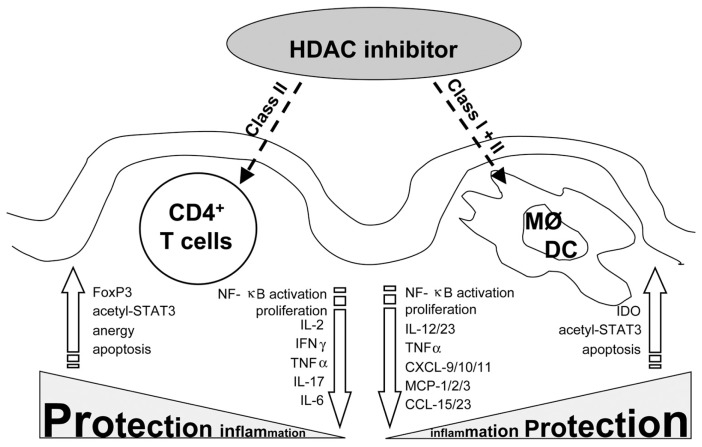
Figure 1.
HDAC inhibitors ameliorate intestinal inflammation. Orally administered HDAC inhibitors affect mucosal immune cells such as CD4+ T cells, DCs or macrophages. Whereas in T helper cells, class II HDACs act as the main targets for the antiinflammatory effect, in DCs or macrophages, both classes are relevant. In all three cell types, proinflammatory markers were downregulated (arrows at the center) and more protective markers were upregulated (arrows on the outside) leading to an overall protective effect within the inflamed mucosa.
CONCLUSION
There is solid evidence that in multiple models of inflammation and in various colitis models in particular, HDAC inhibitors exert an antiinflammatory capacity. This effect was proven from different groups in different species and a multitude of cell types. Additionally, as far as these simplified studies could reveal, most of the cell types involved in the inflammatory process are affected in an antiinflammatory manner. Beyond that, one promising feature of these compounds comprises the proven efficacy in models of experimental colitis when given orally. The HDAC inhibitors reach the site of inflammation and exert their therapeutic potency, whereas there are no side effects related to the gastrointestinal tract.
One characteristic that one has to bear in mind is the global effect of most of the HDAC inhibitors on HDACs in a living cell. They interfere with a plethora of cellular proteins that are involved in central processes, regarding survival, homeostasis or differentiation. Even though RNA expression assays demonstrated that only a small percentage of genes are actually affected by these compounds, the outcome of an HDAC inhibitor treatment is still unpredictable on the cellular level. The dose on which apoptosis or anergy or a diminished cytokine expression occurs, might differ between cell types, tissues and the given HDAC inhibitors. Additionally, various HDAC inhibitors, such as valproic acid, show effects beyond the inhibition of histone deacetylases (81).
While it is reasonable to continue probing the available HDAC inhibitors for their therapeutic capacity, this step should be paralleled by approaches aiming at inhibiting specific HDACs. This approach could be accomplished by applying either novel, second-generation HDAC inhibitors or alternative strategies such as in vivo RNA interference. A precedent request to the success of such trials would be a better understanding of the role of specific HDACs in the given cell types. Here, it proves to be of particular importance to investigate the polarization of effector cell types such as T helper cells or macrophages. While it was demonstrated by Hancock’s group that the inhibition of HDAC9 alone exerts a beneficial effect in experimental inflammatory models via enhancing the Tregs, other class II HDACs are supposed to be equally involved in the relevant cell types; thus, the specific inhibition might contribute to potential therapies. Future treatments should distinguish either between different cell types or between different effects on these cells by choosing a specific inhibition or a distinct concentration.
Footnotes
Online address: http://www.molmed.org
DISCLOSURE
The authors declare that they have no competing interests as defined by Molecular Medicine, or other interests that might be perceived to influence the results and discussion reported in this paper.
REFERENCES
- 1.Mathew CG. New links to the pathogenesis of Crohn disease provided by genome-wide association scans. Nat Rev Genet. 2008;9:9–14. doi: 10.1038/nrg2203. [DOI] [PubMed] [Google Scholar]
- 2.Baumgart DC, Carding SR. Inflammatory bowel disease: cause and immunobiology. Lancet. 2007;369:1627–40. doi: 10.1016/S0140-6736(07)60750-8. [DOI] [PubMed] [Google Scholar]
- 3.Soderlund S, et al. Decreasing time-trends of colorectal cancer in a large cohort of patients with inflammatory bowel disease. Gastroenterology. 2009;136:1561–67. doi: 10.1053/j.gastro.2009.01.064. [DOI] [PubMed] [Google Scholar]
- 4.Karin M, Greten FR. NF-kappaB: linking inflammation and immunity to cancer development and progression. Nat Rev Immunol. 2005;5:749–59. doi: 10.1038/nri1703. [DOI] [PubMed] [Google Scholar]
- 5.Tarmin L, et al. Adenomatous polyposis coli gene mutations in ulcerative colitis- associated dysplasias and cancers versus sporadic colon neoplasms. Cancer Res. 1995;55:2035–8. [PubMed] [Google Scholar]
- 6.Furlan A, et al. Pharmacokinetics, safety and inducible cytokine responses during a phase 1 trial of the oral histone deacetylase inhibitor ITF2357 (givinostat) Mol Med. 2011;17:353–362. doi: 10.2119/molmed.2011.00020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Choudhary C, et al. Lysine acetylation targets protein complexes and co-regulates major cellular functions. Science. 2009;325:834–40. doi: 10.1126/science.1175371. [DOI] [PubMed] [Google Scholar]
- 8.Norris KL, Lee JY, Yao TP. Acetylation goes global: the emergence of acetylation biology. Sci. Signal. 2009;2:pe76. doi: 10.1126/scisignal.297pe76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Spange S, Wagner T, Heinzel T, Kramer OH. Acetylation of non-histone proteins modulates cellular signalling at multiple levels. Int J Biochem Cell Biol. 2009;41:185–98. doi: 10.1016/j.biocel.2008.08.027. [DOI] [PubMed] [Google Scholar]
- 10.Fuss IJ, et al. Nonclassical CD1d-restricted NK T cells that produce IL-13 characterize an atypical Th2 response in ulcerative colitis. J Clin Invest. 2004;113:1490–7. doi: 10.1172/JCI19836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fuss IJ, et al. Disparate CD4+ lamina pro-pria (LP) lymphokine secretion profiles in inflammatory bowel disease: Crohn’s disease LP cells manifest increased secretion of IFN-gamma, whereas ulcerative colitis LP cells manifest increased secretion of IL-5. J Immunol. 1996;157:1261–70. [PubMed] [Google Scholar]
- 12.Stuber E, Strober W, Neurath M. Blocking the CD40L-CD40 interaction in vivo specifically prevents the priming of T helper 1 cells through the inhibition of interleukin 12 secretion. J Exp Med. 1996;183:693–8. doi: 10.1084/jem.183.2.693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Weaver CT, Hatton RD, Mangan PR, Harrington LE. IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu Rev Immunol. 2007;25:821–52. doi: 10.1146/annurev.immunol.25.022106.141557. [DOI] [PubMed] [Google Scholar]
- 14.Acosta-Rodriguez EV, Napolitani G, Lanzavecchia A, Sallusto F. Interleukins 1beta and 6 but not transforming growth factor-beta are essential for the differentiation of interleukin 17-producing human T helper cells. Nat Immunol. 2007;8:942–9. doi: 10.1038/ni1496. [DOI] [PubMed] [Google Scholar]
- 15.Zhang X, et al. Interleukin-1 receptor signaling is required to overcome the effects of pertussis toxin and for efficient infection- or vaccination-induced immunity against Bordetella pertussis. Infect Immun. 2011;79:527–41. doi: 10.1128/IAI.00590-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhou L, et al. IL-6 programs T(H)-17 cell differentiation by promoting sequential engagement of the IL-21 and IL-23 pathways. Nat Immunol. 2007;8:967–74. doi: 10.1038/ni1488. [DOI] [PubMed] [Google Scholar]
- 17.Fujino S, et al. Increased expression of interleukin 17 in inflammatory bowel disease. Gut. 2003;52:65–70. doi: 10.1136/gut.52.1.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fuss IJ, et al. Both IL-12p70 and IL-23 are synthesized during active Crohn’s disease and are down-regulated by treatment with anti-IL-12 p40 monoclonal antibody. Inflamm Bowel Dis. 2006;12:9–15. doi: 10.1097/01.mib.0000194183.92671.b6. [DOI] [PubMed] [Google Scholar]
- 19.Umetsu DT, DeKruyff RH. Th1 and Th2 CD4+ cells in the pathogenesis of allergic diseases. Proc Soc Exp Biol Med. 1997;215:11–20. doi: 10.3181/00379727-215-44109. [DOI] [PubMed] [Google Scholar]
- 20.Bouma G, Strober W. The immunological and genetic basis of inflammatory bowel disease. Nat Rev Immunol. 2003;3:521–33. doi: 10.1038/nri1132. [DOI] [PubMed] [Google Scholar]
- 21.Heller F, et al. Interleukin-13 is the key effector Th2 cytokine in ulcerative colitis that affects epithelial tight junctions, apoptosis, and cell restitution. Gastroenterology. 2005;129:550–64. doi: 10.1016/j.gastro.2005.05.002. [DOI] [PubMed] [Google Scholar]
- 22.Stephens GL, Shevach EM. Foxp3+ regulatory T cells: selfishness under scrutiny. Immunity. 2007;27:417–9. doi: 10.1016/j.immuni.2007.08.008. [DOI] [PubMed] [Google Scholar]
- 23.Zheng Y, Rudensky AY. Foxp3 in control of the regulatory T cell lineage. Nat Immunol. 2007;8:457–62. doi: 10.1038/ni1455. [DOI] [PubMed] [Google Scholar]
- 24.Ruemmele FM, Brousse N, Goulet O. Autoimmune enteropathy: molecular concepts. Curr Opin Gastroenterol. 2004;20:587–91. doi: 10.1097/00001574-200411000-00014. [DOI] [PubMed] [Google Scholar]
- 25.Ochs HD, Gambineri E, Torgerson TR. IPEX, FOXP3 and regulatory T-cells: a model for autoimmunity. Immunol Res. 2007;38:112–21. doi: 10.1007/s12026-007-0022-2. [DOI] [PubMed] [Google Scholar]
- 26.McDonel P, Costello I, Hendrich B. Keeping things quiet: roles of NuRD and Sin3 co-repressor complexes during mammalian development. Int J Biochem Cell Biol. 2009;41:108–16. doi: 10.1016/j.biocel.2008.07.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Grozinger CM, Hassig CA, Schreiber SL. Three proteins define a class of human histone deacetylases related to yeast Hda1p. Proc Natl Acad Sci U S A. 1999;96:4868–73. doi: 10.1073/pnas.96.9.4868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Feng Q, Zhang Y. The MeCP1 complex represses transcription through preferential binding, remodeling, and deacetylating methylated nucleosomes. Genes Dev. 2001;15:827–832. doi: 10.1101/gad.876201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chang S, Collins PL, Aune TM. T-bet dependent removal of Sin3A-histone deacetylase complexes at the Ifng locus drives Th1 differentiation. J Immunol. 2008;181:8372–81. doi: 10.4049/jimmunol.181.12.8372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chen GY, Osada H, Santamaria-Babi LF, Kannagi R. Interaction of GATA-3/T-bet transcription factors regulates expression of sialyl Lewis X homing receptors on Th1/Th2 lymphocytes. Proc Natl Acad Sci U S A. 2006;103:16894–9. doi: 10.1073/pnas.0607926103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sarra M, Pallone F, Macdonald TT, Monteleone G. IL-23/IL-17 axis in IBD. Inflamm Bowel Dis. 2010;16:1808–13. doi: 10.1002/ibd.21248. [DOI] [PubMed] [Google Scholar]
- 32.Edens RE, Dagtas S, Gilbert KM. Histone deacetylase inhibitors induce antigen specific anergy in lymphocytes: a comparative study. Int Immunopharmacol. 2006;6:1673–81. doi: 10.1016/j.intimp.2006.07.001. [DOI] [PubMed] [Google Scholar]
- 33.Moreira JM, Scheipers P, Sorensen P. The histone deacetylase inhibitor trichostatin A modulates CD4+ T cell responses. BMC Cancer. 2003;3:30. doi: 10.1186/1471-2407-3-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Matsuoka H, Fujimura T, Mori H, Aramori I, Mutoh S. Mechanism of HDAC inhibitor FR235222-mediated IL-2 transcriptional repression in Jurkat cells. Int Immunopharmacol. 2007;7:1422–32. doi: 10.1016/j.intimp.2007.05.022. [DOI] [PubMed] [Google Scholar]
- 35.Koenen HJ, et al. Human CD25highFoxp3pos regulatory T cells differentiate into IL-17-producing cells. Blood. 2008;112:2340–52. doi: 10.1182/blood-2008-01-133967. [DOI] [PubMed] [Google Scholar]
- 36.Wang L, de Zoeten EF, Greene MI, Hancock WW. Immunomodulatory effects of deacetylase inhibitors: therapeutic targeting of FOXP3+ regulatory T cells. Nat Rev Drug Discov. 2009;8:969–81. doi: 10.1038/nrd3031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tao R, et al. Deacetylase inhibition promotes the generation and function of regulatory T cells. Nat Med. 2007;13:1299–307. doi: 10.1038/nm1652. [DOI] [PubMed] [Google Scholar]
- 38.Wang L, Tao R, Hancock WW. Using his-tone deacetylase inhibitors to enhance Foxp3(+) regulatory T-cell function and induce allograft tolerance. Immunol Cell Biol. 2009;87:195–202. doi: 10.1038/icb.2008.106. [DOI] [PubMed] [Google Scholar]
- 39.de Zoeten EF, Wang L, Sai H, Dillmann WH, Hancock WW. Inhibition of HDAC9 increases T regulatory cell function and prevents colitis in mice. Gastroenterology. 2010;138:583–94. doi: 10.1053/j.gastro.2009.10.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bosisio D, et al. Blocking TH17-polarizing cytokines by histone deacetylase inhibitors in vitro and in vivo. J Leukoc Biol. 2008;84:1540–8. doi: 10.1189/jlb.0708401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jung ID, et al. Apicidin, the histone deacetylase inhibitor, suppresses Th1 polarization of murine bone marrow-derived dendritic cells. Int J Immunopathol Pharmacol. 2009;22:501–15. doi: 10.1177/039463200902200227. [DOI] [PubMed] [Google Scholar]
- 42.Brogdon JL, et al. Histone deacetylase activities are required for innate immune cell control of Th1 but not Th2 effector cell function. Blood. 2007;109:1123–30. doi: 10.1182/blood-2006-04-019711. [DOI] [PubMed] [Google Scholar]
- 43.Sun Y, et al. Cutting edge: negative regulation of dendritic cells through acetylation of the nonhistone protein STAT-3. J Immunol. 2009;182:5899–903. doi: 10.4049/jimmunol.0804388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Reddy P, et al. Histone deacetylase inhibition modulates indoleamine 2,3-dioxygenase-dependent DC functions and regulates experimental graft-versus-host disease in mice. J Clin Invest. 2008;118:2562–73. doi: 10.1172/JCI34712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Aung HT, et al. LPS regulates proinflammatory gene expression in macrophages by altering histone deacetylase expression. FASEB J. 2006;20:1315–27. doi: 10.1096/fj.05-5360com. [DOI] [PubMed] [Google Scholar]
- 46.Deng WG, Zhu Y, Wu KK. Role of p300 and PCAF in regulating cyclooxygenase-2 promoter activation by inflammatory mediators. Blood. 2004;103:2135–42. doi: 10.1182/blood-2003-09-3131. [DOI] [PubMed] [Google Scholar]
- 47.Ito K, et al. Histone deacetylase 2-mediated deacetylation of the glucocorticoid receptor enables NF-kappaB suppression. J Exp Med. 2006;203:7–13. doi: 10.1084/jem.20050466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Maa MC, et al. Butyrate reduced lipopolysaccharide-mediated macrophage migration by suppression of Src enhancement and focal adhesion kinase activity. J Nutr Biochem. 2010;21:1186–92. doi: 10.1016/j.jnutbio.2009.10.004. [DOI] [PubMed] [Google Scholar]
- 49.Grabiec AM, et al. Histone deacetylase inhibitors suppress inflammatory activation of rheumatoid arthritis patient synovial macrophages and tissue. J Immunol. 2010;184:2718–28. doi: 10.4049/jimmunol.0901467. [DOI] [PubMed] [Google Scholar]
- 50.Choi Y, et al. Histone deacetylase inhibitor KBH-A42 inhibits cytokine production in RAW 264.7 macrophage cells and in vivo endotoxemia model. Exp Mol Med. 2008;40:574–81. doi: 10.3858/emm.2008.40.5.574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lal G, et al. Epigenetic regulation of Foxp3 expression in regulatory T cells by DNA methylation. J Immunol. 2009;182:259–273. doi: 10.4049/jimmunol.182.1.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Polansky JK, et al. DNA methylation controls Foxp3 gene expression. Eur J Immunol. 2008;38:1654–63. doi: 10.1002/eji.200838105. [DOI] [PubMed] [Google Scholar]
- 53.Scott GK, Mattie MD, Berger CE, Benz SC, Benz CC. Rapid alteration of microRNA levels by histone deacetylase inhibition. Cancer Res. 2006;66:1277–81. doi: 10.1158/0008-5472.CAN-05-3632. [DOI] [PubMed] [Google Scholar]
- 54.Schickel R, Boyerinas B, Park SM, Peter ME. MicroRNAs: key players in the immune system, differentiation, tumorigenesis and cell death. Oncogene. 2008;27:5959–74. doi: 10.1038/onc.2008.274. [DOI] [PubMed] [Google Scholar]
- 55.Baltimore D, Boldin MP, O’Connell RM, Rao DS, Taganov KD. MicroRNAs: new regulators of immune cell development and function. Nat Immunol. 2008;9:839–45. doi: 10.1038/ni.f.209. [DOI] [PubMed] [Google Scholar]
- 56.Faraco G, et al. Histone deacetylase (HDAC) inhibitors reduce the glial inflammatory response in vitro and in vivo. Neurobiol Dis. 2009;36:269–79. doi: 10.1016/j.nbd.2009.07.019. [DOI] [PubMed] [Google Scholar]
- 57.Kiernan R, et al. Post-activation turn-off of NF-kappa B-dependent transcription is regulated by acetylation of p65. J Biol Chem. 2003;278:2758–66. doi: 10.1074/jbc.M209572200. [DOI] [PubMed] [Google Scholar]
- 58.Greten FR, et al. IKKbeta links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell. 2004;118:285–96. doi: 10.1016/j.cell.2004.07.013. [DOI] [PubMed] [Google Scholar]
- 59.Shibata W, et al. Cutting edge: the IkappaB kinase (IKK) inhibitor, NEMO-binding domain peptide, blocks inflammatory injury in murine colitis. J Immunol. 2007;179:2681–5. doi: 10.4049/jimmunol.179.5.2681. [DOI] [PubMed] [Google Scholar]
- 60.Nenci A, et al. Epithelial NEMO links innate immunity to chronic intestinal inflammation. Nature. 2007;446:557–61. doi: 10.1038/nature05698. [DOI] [PubMed] [Google Scholar]
- 61.Chen L, Fischle W, Verdin E, Greene WC. Duration of nuclear NF-kappaB action regulated by reversible acetylation. Science. 2001;293:1653–7. doi: 10.1126/science.1062374. [DOI] [PubMed] [Google Scholar]
- 62.Ashburner BP, Westerheide SD, Baldwin AS., Jr The p65 (RelA) subunit of NF-kappaB interacts with the histone deacetylase (HDAC) corepressors HDAC1 and HDAC2 to negatively regulate gene expression. Mol Cell Biol. 2001;21:7065–77. doi: 10.1128/MCB.21.20.7065-7077.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Imre G, Gekeler V, Leja A, Beckers T, Boehm M. Histone deacetylase inhibitors suppress the inducibility of nuclear factor-kappaB by tumor necrosis factor-alpha receptor-1 down-regulation. Cancer Res. 2006;66:5409–18. doi: 10.1158/0008-5472.CAN-05-4225. [DOI] [PubMed] [Google Scholar]
- 64.Liu H, et al. Trichostatin A regulates hGCN5 expression and cell cycle on daudi cells in vitro. J Huazhong Univ Sci Technolog Med Sci. 2006;26:534–7. doi: 10.1007/s11596-006-0512-9. [DOI] [PubMed] [Google Scholar]
- 65.Takada Y, Gillenwater A, Ichikawa H, Aggarwal BB. Suberoylanilide hydroxamic acid potentiates apoptosis, inhibits invasion, and abolishes osteoclastogenesis by suppressing nuclear factor-kappaB activation. J Biol Chem. 2006;281:5612–22. doi: 10.1074/jbc.M507213200. [DOI] [PubMed] [Google Scholar]
- 66.Glauben R, et al. Histone deacetylases: novel targets for prevention of colitis-associated cancer in mice. Gut. 2008;57:613–622. doi: 10.1136/gut.2007.134650. [DOI] [PubMed] [Google Scholar]
- 67.Kramer OH, et al. Acetylation of Stat1 modulates NF-kappaB activity. Genes Dev. 2006;20:473–485. doi: 10.1101/gad.364306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Glauben R, et al. Histone hyperacetylation is associated with amelioration of experimental colitis in mice. J Immunol. 2006;176:5015–22. doi: 10.4049/jimmunol.176.8.5015. [DOI] [PubMed] [Google Scholar]
- 69.Marks PA, Dokmanovic M. Histone deacetylase inhibitors: discovery and development as anticancer agents. Expert Opin Investig Drugs. 2005;14:1497–511. doi: 10.1517/13543784.14.12.1497. [DOI] [PubMed] [Google Scholar]
- 70.Bertrand P. Inside HDAC with HDAC inhibitors. Eur J Med Chem. 2010;45:2095–116. doi: 10.1016/j.ejmech.2010.02.030. [DOI] [PubMed] [Google Scholar]
- 71.Reilly CM, et al. The histone deacetylase inhibitor trichostatin A upregulates regulatory T cells and modulates autoimmunity in NZB/W F1 mice. J Autoimmun. 2008;31:123–30. doi: 10.1016/j.jaut.2008.04.020. [DOI] [PubMed] [Google Scholar]
- 72.Saouaf SJ, et al. Deacetylase inhibition increases regulatory T cell function and decreases incidence and severity of collagen-induced arthritis. Exp Mol Pathol. 2009;87:99–104. doi: 10.1016/j.yexmp.2009.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Leoni F, et al. The histone deacetylase inhibitor ITF2357 reduces production of pro-inflammatory cytokines in vitro and systemic inflammation in vivo. Mol Med. 2005;11:1–15. doi: 10.2119/2006-00005.Dinarello. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Neurath MF, Fuss I, Kelsall BL, Stuber E, Strober W. Antibodies to interleukin 12 abrogate established experimental colitis in mice. J Exp Med. 1995;182:1281–90. doi: 10.1084/jem.182.5.1281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Boirivant M, Fuss IJ, Chu A, Strober W. Oxazolone colitis: a murine model of T helper cell type 2 colitis treatable with antibodies to interleukin 4. J Exp Med. 1998;188:1929–39. doi: 10.1084/jem.188.10.1929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Vojinovic J, et al. Safety and efficacy of an oral histone deacetylase inhibitor in systemic-onset juvenile idiopathic arthritis. Arthritis Rheum. 2011;63:1452–8. doi: 10.1002/art.30238. [DOI] [PubMed] [Google Scholar]
- 77.Neurath MF, et al. Regulation of T-cell apoptosis in inflammatory bowel disease: to die or not to die, that is the mucosal question. Trends Immunol. 2001;22:21–6. doi: 10.1016/s1471-4906(00)01798-1. [DOI] [PubMed] [Google Scholar]
- 78.Lugering A, et al. Infliximab induces apoptosis in monocytes from patients with chronic active Crohn’s disease by using a caspase-dependent pathway. Gastroenterology. 2001;121:1145–57. doi: 10.1053/gast.2001.28702. [DOI] [PubMed] [Google Scholar]
- 79.Tanaka T, et al. A novel inflammation- related mouse colon carcinogenesis model induced by azoxymethane and dextran sodium sulfate. Cancer Sci. 2003;94:965–73. doi: 10.1111/j.1349-7006.2003.tb01386.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Hegazi RA, et al. Celecoxib and rofecoxib potentiate chronic colitis and premalignant changes in interleukin 10 knockout mice. Inflamm Bowel Dis. 2003;9:230–36. doi: 10.1097/00054725-200307000-00003. [DOI] [PubMed] [Google Scholar]
- 81.Chateauvieux S, Morceau F, Dicato M, Diederich M. Molecular and therapeutic potential and toxicity of valproic acid. J Biomed Biotechnol. 2010;2010;479364 doi: 10.1155/2010/479364. [DOI] [PMC free article] [PubMed] [Google Scholar]