Abstract
Few studies have examined multiple risk factors for mortality or formally compared their associations across specific causes of death. The authors used competing risks survival analysis to evaluate associations of lifestyle and dietary factors with all-cause and cause-specific mortality among 50,112 participants in the Nurses’ Health Study. There were 4,893 deaths between 1986 and 2004: 1,026 from cardiovascular disease, 931 from smoking-related cancers, 1,430 from cancers not related to smoking, and 1,506 from all other causes. Age, body mass index at age 18 years, weight change, height, current smoking and pack-years of smoking, glycemic load, cholesterol intake, systolic blood pressure and use of blood pressure medications, diabetes, parental myocardial infarction before age 60 years, and time since menopause were directly related to all-cause mortality, whereas there were inverse associations for physical activity and intakes of nuts, polyunsaturated fat, and cereal fiber. Moderate alcohol consumption was associated with decreased mortality. A model that incorporated differences in the associations of some risk factors with specific causes of death had a significantly better fit compared with a model in which all risk factors had common associations across all causes. In the future, this new model may be used to identify individuals at increased risk of mortality.
Keywords: diet, exercise, life style, mortality, obesity, smoking
Many epidemiologic studies have examined associations of individual lifestyle factors—including overweight and obesity, physical activity, smoking, dietary factors, and postmenopausal hormone use—with mortality (1–5). A limitation of most prior studies, however, has been their focus on one risk factor at a time; few studies have addressed the effects of multiple risk factors simultaneously. Furthermore, although some studies have examined the effects of risk factors on cause-specific mortality, few have done a formal comparison to determine whether there are significant differences in their associations across specific causes of death.
Use of a composite endpoint, such as all-cause mortality, can enhance statistical power if a risk factor has a fairly uniform effect on each component of the outcome but cannot identify differences between the associations of a risk factor with the different components. The purpose of this analysis, therefore, was to examine the associations of lifestyle and dietary factors with mortality among participants in the Nurses’ Health Study, applying a method for competing risks survival analysis to compare the effects of risk factors across specific causes of death. Then, based on this comparison, our goal was to develop a comprehensive model for all-cause mortality that incorporates the effects of important risk factors and allows them to have different or common effects across specific types of mortality.
MATERIALS AND METHODS
Study design and population
The Nurses’ Health Study is a prospective cohort study that began in 1976, when 121,700 US female registered nurses aged 30–55 years completed a mailed questionnaire about their lifestyle factors, health behaviors, and medical histories. Follow-up questionnaires have been sent to participants every 2 years since enrollment (6). This study was approved by the Partners Human Research Committee (Boston, Massachusetts); completion of the self-administered questionnaire was considered to imply informed consent.
Follow-up for this analysis began in 1986, the first year that a detailed assessment of physical activity was included on the questionnaire, because physical activity is an important risk factor for mortality in this population (2, 3). We excluded women who died or had a previous diagnosis of cardiovascular disease or cancer, other than nonmelanoma skin cancer, prior to return of the 1986 questionnaire. In addition, only participants with complete information on the risk factors of interest (including 3 dietary assessments in 1980, 1984, and 1986) were included.
Assessment of risk factors
We focused mainly on modifiable lifestyle and dietary factors previously shown to be associated with mortality in this cohort. We also included several conditions that are consequences of lifestyle (e.g., systolic blood pressure, diabetes), because of well-established associations with specific causes of death. All risk factors were assessed by self-report on the biennial questionnaires with a high degree of reliability and accuracy (7–13). Although there were repeated assessments of many of the risk factors during the follow-up period, we used only baseline values in this analysis to minimize the potential of clinical or subclinical disease affecting risk factor status.
Age was calculated from date of birth until the return date of the 1986 questionnaire. Height was assessed at the beginning of the study in 1976, and weight at age 18 years was reported in 1980; these were used to calculate body mass index at age 18. Weight change since age 18 years was calculated by subtracting weight at age 18 from current weight. Smoking status, quantity of cigarettes smoked, and duration of smoking were assessed on every questionnaire and used to calculate total pack-years of smoking. In 1986, women were asked to complete an 8-item questionnaire regarding the average amount of time spent per week in different physical activities. The reported time spent in each activity per week was multiplied by its typical expenditure requirements expressed in metabolic equivalents (METs) and added together to yield a MET-hours-per-week score (14, 15).
Dietary factors were assessed in 1980, 1984, and 1986 by using a semiquantitative food frequency questionnaire. Participants reported their average frequency of consumption of a commonly used portion size of specific foods during the previous year. Nutrient values were computed by multiplying the frequency of consumption of each food by the nutrient content of that portion, adding these products across all food items, and adjusting for total energy intake (16). We calculated total dietary glycemic load by multiplying the carbohydrate content of each food by its glycemic index, multiplying this value by the frequency of consumption, and summing these values for all foods (17). Averages of 1980, 1984, and 1986 values were computed for foods, nutrients, and glycemic load to reduce measurement error, an approach that has been used in many analyses of dietary factors (18). Questions about the consumption of beer, wine, and spirits were included as separate items on the food frequency questionnaire and were used to calculate total alcohol intake (1).
Menopausal status and age at menopause were assessed in 1986 and used to calculate time since menopause. Women were classified as postmenopausal at the first report of natural menopause or surgery with bilateral oophorectomy. Systolic blood pressure and use of medications to control blood pressure, personal history of diabetes, and parental myocardial infarction before age 60 years also were assessed via self-report.
Ascertainment of mortality
The main outcome was death from all causes, occurring after the return of the 1986 questionnaire but before June 1, 2004. Deaths were usually reported by families, and deaths among nonrespondents were identified by searching the National Death Index (19, 20). The cause of death was ascertained by review of the family reports, death certificates, and pertinent medical records. Deaths were grouped into 4 broad categories, according to the International Classification of Diseases, Eighth Revision (ICD-8): cardiovascular disease (ICD-8 codes 390.0–458.9 and 795.0–795.9); smoking-related cancers or cancers denoted by the 2004 Surgeon General's report to be caused by smoking (21, 22), including those of the lip, mouth, pharynx, esophagus (ICD-8 codes 140.0–150.0), larynx (ICD-8 codes 161.0–161.9), pancreas (ICD-8 codes 157.0–157.9), bladder and kidney (ICD-8 codes 188.0–189.9), cervix (ICD-8 code 180.0), stomach (ICD-8 codes 151.0–151.9), trachea and lung (ICD-8 codes 162.0–162.1), and acute myeloid leukemia (ICD-8 code 205.0); other cancers (ICD-8 codes 140.0–207.9, except those included in smoking-related cancers); and all other causes combined (all other codes not included in 1 of the previous 3 categories). These categories are consistent with those used in other recent Nurses’ Health Study analyses (2–4, 23, 24).
Statistical analysis
Person-years of follow-up were accrued from the return date of the 1986 questionnaire until either death or the end of follow-up (May 31, 2004), whichever came first. We fit a Cox proportional hazards model to compute hazard ratios and 95% confidence intervals for associations between each risk factor and all-cause mortality, adjusting for all other risk factors. Follow-up time since 1986 (continuous, in months) was the time scale. Risk factors that had fairly linear associations with mortality were modeled as continuous variables after eliminating outliers (25). The difference between the 90th and 10th percentiles (shown in Table 1), which corresponds to the median of the top versus the median of the bottom quintile, was used as the unit for each continuous variable to provide standardized estimates across different risk factors. Smoking status, alcohol intake, nut consumption, systolic blood pressure, use of blood pressure medications, personal history of diabetes, and parental myocardial infarction before age 60 years were modeled as categorical variables. We tested the proportional hazards assumption by running separate models during the first 10 years (1986–1996) and during the second 8 years of follow-up (1996–2004); we also ran models that included interaction terms between each risk factor and time period. The associations of most risk factors with mortality were similar during the 2 time periods, as shown in Web Table 1 posted on the Journal’s Web site, http://aje.oupjournals.org.
Table 1.
Distributions of Risk Factors for Mortality Among 50,112 Participants in the Nurses’ Health Study in 1986
| Mean (SD) | Median | 10th Percentile | 90th Percentile | ||
| Continuous variables | |||||
| Age, years | 52.5 (7.1) | 52.3 | 42.9 | 62.3 | |
| Body mass index at age 18 years, kg/m2 | 21.4 (2.8) | 20.9 | 18.3 | 24.9 | |
| Weight change since age 18 years, kg | 7.8 (9.9) | 6.8 | −2.7 | 20.5 | |
| Height, inchesa | 64.5 (2.4) | 64.0 | 62.0 | 68.0 | |
| Smoking amount/duration, pack-yearsb | 22.2 (18.5) | 18.0 | 2.0 | 48.0 | |
| Physical activity, MET-hours/week | 12.9 (14.5) | 7.7 | 0.9 | 34.2 | |
| Polyunsaturated fat, % energy | 6.0 (1.2) | 5.9 | 4.5 | 7.6 | |
| Glycemic load, GL unitsc | 94.4 (16.6) | 94.0 | 74.0 | 115.0 | |
| Dietary cholesterol, mg/1,000 kcal per day | 179.1 (44.2) | 174.4 | 128.6 | 233.5 | |
| Cereal fiber, g/day, energy adjusted | 3.7 (1.6) | 3.4 | 2.0 | 5.8 | |
| Time since menopause, years | 5.0 (5.8) | 2.8 | 0.0 | 13.1 | |
| No. | % | ||||
| Categorical variables | |||||
| Smoking status | |||||
| Never | 22,698 | 45.3 | |||
| Past | 17,148 | 34.2 | |||
| Current | 10,266 | 20.5 | |||
We then used an extension of the Cox proportional hazards model, hereafter referred to as “competing risks survival analysis,” to fit a single model for mortality, assuming different associations of each risk factor with each specific cause of mortality (cardiovascular disease, smoking-related cancers, other cancers, and all other causes) (26). Specifically, the approach described by Lunn and McNeil (27) stratifies on event type and allows for estimation of the separate associations of each risk factor with the relative hazard of each outcome under a proportional hazards assumption. This can be readily implemented in a standard statistical software package using data augmentation, which requires that each subject have a separate observation for each outcome.
The initial full model assumed different associations of each risk factor with the 4 outcomes. We then fit a series of reduced models in which 1 risk factor at a time was constrained to have a single estimate across all 4 outcomes. In each of these reduced models, the effects of all other risk factors were allowed to be different, so that the reduced model differed from the initial full model for only 1 variable. Likelihood ratio tests for heterogeneity were used to compare the initial full model with each reduced model. We considered a P value for the likelihood ratio test (PLRT) of less than 0.10 to indicate that the associations of a particular risk factor were different across specific causes of death. All statistical tests were 2 sided.
A stepwise down approach was used to obtain the final competing risks model for mortality. We set the risk factor with the largest PLRT that was 0.10 or greater to have a single estimate across the 4 outcomes, and this became the new full model. The individual reduced models then were refit and compared with the new full model. These steps were repeated until we obtained a final model in which the only risk factors with different estimates across the 4 outcomes were those with PLRT < 0.10. We evaluated the performance of the final competing risks model compared with the standard Cox proportional hazards model for all-cause mortality, in which all risk factors were constrained to have equal effects across causes, using area under the receiver-operating characteristic curves (AUC or C statistics) (28) and likelihood ratio tests.
RESULTS
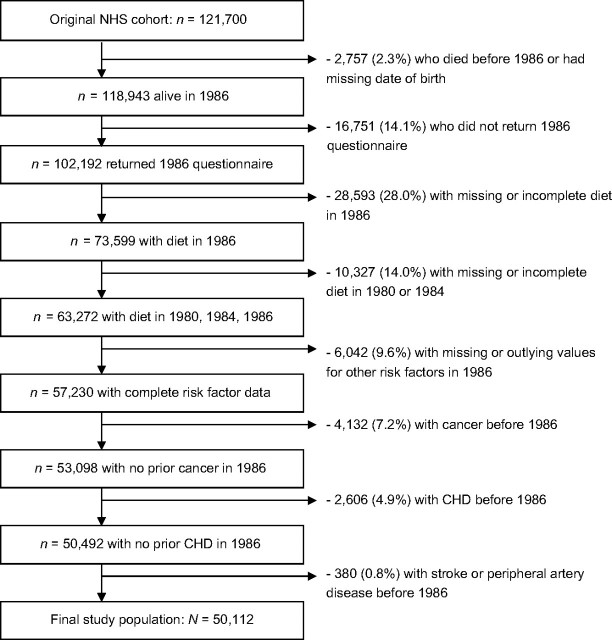
A total of 50,112 participants were included in the analysis. The numbers of participants excluded and the reasons for exclusion are shown in Figure 1. The mean age among eligible participants in 1986 was 52.5 years, and the distributions of the other risk factors are shown in Table 1. There were a total of 4,893 deaths between 1986 and 2004: 1,026 from cardiovascular disease (21%), 931 from smoking-related cancers (19%), 1,430 from cancers not related to smoking (29%), and 1,506 from all other causes (31%).
Figure 1.
Study population and exclusions in the Nurses’ Health Study between study initiation in 1976 and current follow-up analysis beginning in 1986. CHD, coronary heart disease; NHS, Nurses’ Health Study.
The associations of each risk factor with all-cause mortality are shown in Table 2. Risk of mortality increased sharply with age (hazard ratio (HR) per 19 years = 5.78). Both greater body mass index at age 18 years and weight change since age 18 were associated with increased risk, although the association was stronger for body mass index at age 18 (HR per 7 kg/m2 = 1.23). Height also was positively associated with all-cause mortality (HR per 6 inches = 1.16; 1 inch = 2.54 cm). Both current smoking (HR vs. never = 1.48) and greater total pack-years of smoking (HR per 46 pack-years = 2.08) were associated with increased mortality risk, whereas physical activity was associated with decreased risk (HR per 33 MET-hours/week = 0.87). For alcohol consumption, there was a small decrease in risk for 0.1 g/day–9.9 g/day and 10 g/day–29.9 g/day (HRs vs. none = 0.90 and 0.91, respectively), but not for ≥30 g/day. Glycemic load (HR per 41 units = 1.22) and cholesterol intake (HR per 105 mg/1,000 kcal = 1.17) were positively associated with mortality, whereas nut consumption (HR for ≥2 servings/week vs. none or almost none = 0.86), polyunsaturated fat intake (HR per 3% of total energy intake = 0.85), and cereal fiber intake (HR per 4 g = 0.84) were inversely associated with risk. Initial models also included other dietary factors (e.g., intakes of fruit and vegetables, trans fat), but these were eliminated from the final model because of nonsignificant associations with mortality after adjustment for other risk factors (data not shown). Systolic blood pressure (HR for ≥160 vs. <120 mm Hg = 1.49), use of blood pressure medications (HR = 1.19), personal history of diabetes (HR = 2.45), parental myocardial infarction before age 60 years (HR = 1.14), and time since menopause (HR per 13 years = 1.15) were positively associated with all-cause mortality. Based on multiplication of the individual hazard ratios, the hazard ratio for all-cause mortality for the “worst” versus the “best” risk profile for all of the modifiable risk factors (body mass index at age 18 years, weight change, smoking status and amount/duration, physical activity, alcohol intake, and all of the dietary factors) was 12.32.
Table 2.
Associations of Risk Factors With All-Cause Mortality From a Cox Proportional Hazards Model Among 50,112 Participants in the Nurses’ Health Study, 1986–2004a
| HRb | 95% CI | |
| Age (per 19 years)c | 5.78 | 5.06, 6.61 |
| Body mass index at age 18 years (per 7 kg/m2)d | 1.23 | 1.15, 1.31 |
| Weight change since age 18 years (per 23 kg)d | 1.07 | 1.00, 1.15 |
| Height (per 6 inches)e | 1.16 | 1.08, 1.24 |
| Smoking statusc | ||
| Never | 1.0 | Referent |
| Past | 0.96 | 0.89, 1.04 |
| Current | 1.48 | 1.33, 1.64 |
| Smoking amount/duration (per 46 pack-years)c | 2.08 | 1.92, 2.25 |
| Physical activity (per 33 MET-hours/week)c | 0.87 | 0.81, 0.93 |
| Alcohol intake, g/dayc | ||
| None | 1.0 | Referent |
| 0.1–9.9 | 0.90 | 0.84, 0.96 |
| 10–29.9 | 0.91 | 0.83, 1.00 |
| ≥30 | 1.03 | 0.90, 1.17 |
| Nut consumption, servings/weekc | ||
| None | 1.0 | Referent |
| ≤1 | 0.92 | 0.87, 0.98 |
| ≥2 | 0.86 | 0.77, 0.95 |
| Polyunsaturated fat (per 3% energy)f | 0.85 | 0.79, 0.91 |
| Glycemic load (per 41 units)f | 1.22 | 1.12, 1.34 |
| Dietary cholesterol (per 105 mg/1,000 kcal)f | 1.17 | 1.08, 1.26 |
| Cereal fiber (per 4 g, energy adjusted)f | 0.84 | 0.78, 0.91 |
| Systolic blood pressure, mm Hgc | ||
| <120 | 1.0 | Referent |
| 120–139 | 1.02 | 0.95, 1.09 |
| 140–149 | 1.22 | 1.12, 1.33 |
| 150–159 | 1.38 | 1.21, 1.58 |
| ≥160 | 1.49 | 1.25, 1.77 |
| Use of blood pressure medicationsg | 1.19 | 1.10, 1.28 |
| Personal history of diabetesh | 2.45 | 2.18, 2.76 |
| Parental MI before age 60 yearsh | 1.14 | 1.06, 1.22 |
| Time since menopause (per 13 years)c | 1.15 | 1.05, 1.25 |
Abbreviations: CI, confidence interval; HR, hazard ratio; MET, metabolic equivalent; MI, myocardial infarction.
Units for continuous variables correspond to the difference between the 90th and 10th percentile values.
Adjusted for all other risk factors in table.
From 1986 questionnaire.
From 1980 questionnaire.
From 1976 questionnaire; 1 inch = 2.54 cm.
Average of values from 1980, 1984, and 1986 questionnaires.
From 1988 questionnaire.
From 1986 or previous questionnaires.
The associations of each risk factor with cause-specific mortality are shown in Table 3. The relations of many factors with risk of death differed across causes. For example, age was more strongly associated with risk of death from cardiovascular disease (HR per 19 years = 6.98) and other causes (HR = 8.03) than from smoking-related cancers or other cancers, and the positive association for body mass index at age 18 years was stronger for risk of death from cardiovascular disease (HR per 7 kg/m2 = 1.61) than for any other causes. Weight change since age 18 years was positively associated with risk of death from cardiovascular disease (HR per 23 kg = 1.25) and other cancers (HR = 1.31), but inversely associated with risk of death from other causes (HR = 0.85). Current smoking was associated with increased risk of death from cardiovascular disease (HR vs. never = 2.02), smoking-related cancers (HR = 1.88), and other causes (HR = 1.57), but not from other cancers. Total number of pack-years of smoking was positively associated with risk of death from all of the causes, but most strongly for smoking-related cancers (HR per 46 pack-years = 3.54). Physical activity was most strongly inversely associated with risk of death from other causes (HR per 33 MET-hours/week = 0.76). The U-shaped association for alcohol consumption was observed only for risk of death from cardiovascular disease (HR for 0.1–9.9 g/day vs. none = 0.72) and other causes (HR = 0.83). Systolic blood pressure, use of blood pressure medications, and personal history of diabetes were positively associated with risk of death from cardiovascular disease and other causes, but not from smoking-related cancers or other cancers. Time since menopause was positively associated with risk of death from cardiovascular disease, smoking-related cancers, and other causes, but inversely associated with risk of death from other cancers. The hazard ratio for the “worst” versus the “best” risk profile for the modifiable risk factors was 24.14 for cardiovascular disease mortality, 16.02 for smoking-related cancer mortality, 5.73 for other cancer mortality, and 12.50 for other cause mortality.
Table 3.
Associations of Risk Factors With Cause-specific Mortality From a Competing Risks Model Among 50,112 Participants in the Nurses’ Health Study, 1986–2004, Assuming Different Associations of All Risk Factors With Each Specific Causea
| Cardiovascular Disease (1,026 deaths) |
Smoking-related Cancers (931 deaths) |
Other Cancers (1,430 deaths) |
Other Causes (1,506 deaths) |
Pheterogeneity | |||||
| HRb | 95% CI | HRb | 95% CI | HRb | 95% CI | HRb | 95% CI | ||
| Age (per 19 years)c | 6.98 | 5.18, 9.39 | 5.17 | 3.80, 7.05 | 4.40 | 3.45, 5.61 | 8.03 | 6.27, 10.27 | 0.004 |
| Body mass index at age 18 years (per 7 kg/m2)d | 1.61 | 1.40, 1.84 | 1.22 | 1.04, 1.43 | 1.13 | 0.99, 1.29 | 1.06 | 0.93, 1.20 | <0.0001 |
| Weight change since age 18 years (per 23 kg)d | 1.25 | 1.09, 1.43 | 0.94 | 0.80, 1.11 | 1.31 | 1.16, 1.48 | 0.85 | 0.75, 0.96 | <0.0001 |
| Height (per 6 inches)e | 1.17 | 1.01, 1.37 | 1.14 | 0.97, 1.34 | 1.17 | 1.03, 1.34 | 1.14 | 1.00, 1.30 | 0.99 |
| Smoking statusc | |||||||||
| Never | 1.0 | Referent | 1.0 | Referent | 1.0 | Referent | 1.0 | Referent | <0.0001 |
| Past | 0.96 | 0.80, 1.15 | 1.03 | 0.83, 1.27 | 0.96 | 0.83, 1.10 | 1.05 | 0.90, 1.21 | |
| Current | 2.02 | 1.61, 2.54 | 1.88 | 1.48, 2.39 | 1.07 | 0.88, 1.31 | 1.57 | 1.30, 1.89 | |
| Smoking amount/duration (per 46 pack-years)c | 1.56 | 1.30, 1.87 | 3.54 | 3.02, 4.16 | 1.61 | 1.36, 1.91 | 2.01 | 1.74, 2.33 | <0.0001 |
| Physical activity (per 33 MET-hours/week)c | 0.88 | 0.75, 1.03 | 0.97 | 0.83, 1.14 | 0.91 | 0.81, 1.04 | 0.76 | 0.66, 0.86 | 0.07 |
| Alcohol intake, g/dayc | |||||||||
| None | 1.0 | Referent | 1.0 | Referent | 1.0 | Referent | 1.0 | Referent | 0.003 |
| 0.1–9.9 | 0.72 | 0.62, 0.84 | 1.03 | 0.88, 1.21 | 1.03 | 0.91, 1.17 | 0.83 | 0.74, 0.94 | |
| 10–29.9 | 0.73 | 0.59, 0.90 | 0.97 | 0.79, 1.19 | 1.12 | 0.95, 1.33 | 0.85 | 0.72, 1.00 | |
| ≥30 | 1.01 | 0.77, 1.33 | 1.08 | 0.81, 1.43 | 1.02 | 0.78, 1.32 | 1.01 | 0.80, 1.28 | |
| Nut consumption, servings/weekc | |||||||||
| None | 1.0 | Referent | 1.0 | Referent | 1.0 | Referent | 1.0 | Referent | 0.73 |
| ≤1 | 0.91 | 0.80, 1.03 | 0.93 | 0.81, 1.06 | 0.91 | 0.81, 1.02 | 0.95 | 0.85, 1.06 | |
| ≥2 | 0.82 | 0.65, 1.04 | 0.74 | 0.58, 0.95 | 0.95 | 0.79, 1.14 | 0.87 | 0.72, 1.05 | |
| Polyunsaturated fat (per 3% energy)f | 0.79 | 0.67, 0.93 | 0.98 | 0.83, 1.16 | 0.86 | 0.75, 0.99 | 0.79 | 0.69, 0.91 | 0.20 |
| Glycemic load (per 41 units)f | 1.19 | 0.98, 1.45 | 1.18 | 0.96, 1.45 | 1.14 | 0.97, 1.36 | 1.35 | 1.15, 1.58 | 0.55 |
| Dietary cholesterol (per 105 mg/1,000 kcal)f | 1.07 | 0.91, 1.26 | 1.12 | 0.94, 1.32 | 1.25 | 1.09, 1.43 | 1.20 | 1.05, 1.37 | 0.48 |
| Cereal fiber (per 4 g, energy adjusted)f | 0.82 | 0.69, 0.97 | 0.86 | 0.72, 1.03 | 0.82 | 0.71, 0.95 | 0.85 | 0.74, 0.98 | 0.96 |
| Systolic blood pressure, mm Hgc | 0.003 | ||||||||
| <120 | 1.0 | Referent | 1.0 | Referent | 1.0 | Referent | 1.0 | Referent | |
| 120–139 | 1.25 | 1.06, 1.48 | 0.87 | 0.75, 1.02 | 1.02 | 0.90, 1.15 | 1.03 | 0.91, 1.17 | |
| 140–149 | 1.72 | 1.42, 2.09 | 0.99 | 0.81, 1.22 | 1.10 | 0.93, 1.30 | 1.25 | 1.07, 1.47 | |
| 150–159 | 1.80 | 1.36, 2.38 | 1.34 | 0.98, 1.84 | 1.20 | 0.91, 1.58 | 1.42 | 1.11, 1.81 | |
| ≥160 | 2.58 | 1.91, 3.49 | 1.25 | 0.81, 1.92 | 1.04 | 0.70, 1.56 | 1.31 | 0.95, 1.81 | |
| Use of blood pressure medicationsg | 1.56 | 1.35, 1.81 | 0.96 | 0.80, 1.17 | 1.01 | 0.87, 1.17 | 1.23 | 1.07, 1.40 | <0.0001 |
| Personal history of diabetesh | 3.34 | 2.72, 4.10 | 1.18 | 0.80, 1.75 | 1.27 | 0.95, 1.70 | 3.58 | 2.97, 4.33 | <0.0001 |
| Parental MI before age 60 yearsh | 1.20 | 1.03, 1.39 | 1.10 | 0.93, 1.29 | 1.04 | 0.91, 1.20 | 1.21 | 1.07, 1.37 | 0.37 |
| Time since menopause (per 13 years)c | 1.32 | 1.11, 1.58 | 1.21 | 1.00, 1.47 | 0.90 | 0.76, 1.07 | 1.21 | 1.04, 1.41 | 0.01 |
Abbreviations: CI, confidence interval; HR, hazard ratio; MET, metabolic equivalent; MI, myocardial infarction.
Units for continuous variables correspond to the difference between the 90th and 10th percentile values.
Adjusted for all other risk factors in table.
From 1986 questionnaire.
From 1980 questionnaire.
From 1976 questionnaire; 1 inch = 2.54 cm.
Average of values from 1980, 1984, and 1986 questionnaires.
From 1988 questionnaire.
From 1986 or previous questionnaires.
In secondary analyses, we further explored the relation of height with individual causes of cardiovascular disease mortality, including coronary heart disease (407 deaths), stroke and cerebrovascular disease (266 deaths), and other cardiovascular disease (353 deaths), because of some previous studies suggesting that height may be inversely related to incidence of cardiovascular disease (29, 30). In these models, height was not associated with risk of mortality from coronary heart disease (HR per 6 inches = 1.04, 95% confidence interval (CI): 0.81, 1.33) or stroke (HR = 1.00, 95% CI: 0.74, 1.36), but was positively associated with other cardiovascular disease (HR per 6 inches = 1.52, 95% CI: 1.18, 1.98). This is consistent with an analysis from the Physicians’ Health Study showing that taller height was associated with increased risk of venous thromboembolism but not coronary events or stroke (31).
The results from the final competing risks model obtained from the stepwise down procedure, in which risk factors with PLRT ≥ 0.10 were set to have equal effects and all other risk factors had different effects across specific causes of death, are shown in Table 4. Consistent with the results from the original Cox proportional hazards model for all-cause mortality (Table 2), taller height, higher glycemic load, higher dietary cholesterol, and parental myocardial infarction before age 60 years were associated with increased risk of death from all causes, whereas greater nut consumption and cereal fiber intake were associated with decreased risk. Although polyunsaturated fat intake was inversely associated with death from all causes, we maintained its separate associations with different causes of death because the PLRT in the final competing risks model in Table 4, after equating the effects of other risk factors, was less than 0.10.
Table 4.
Associations of Risk Factors With Cause-specific Mortality From the Final Competing Risks Model Among 50,112 Participants in the Nurses’ Health Study, 1986–2004, Equating Risk Factors With Similar Effects and Allowing Other Risk Factors to Have Different Effectsa
| Cardiovascular Disease (1,026 deaths) |
Smoking-related Cancers (931 deaths) |
Other Cancers (1,430 deaths) |
Other Causes (1,506 deaths) |
|||||
| HRb | 95% CI | HRb | 95% CI | HRb | 95% CI | HRb | 95% CI | |
| Age (per 19 years)c | 6.92 | 5.15, 9.30 | 5.18 | 3.81, 7.04 | 4.43 | 3.48, 5.64 | 8.03 | 6.29, 10.26 |
| Body mass index at age 18 years (per 7 kg/m2)d | 1.60 | 1.40, 1.83 | 1.22 | 1.04, 1.43 | 1.14 | 1.00, 1.30 | 1.06 | 0.93, 1.19 |
| Weight change since age 18 years (per 23 kg)d | 1.24 | 1.09, 1.42 | 0.94 | 0.80, 1.10 | 1.32 | 1.17, 1.50 | 0.84 | 0.74, 0.96 |
| Height (per 6 inches)e | 1.16 (1.08, 1.24) | |||||||
| Smoking statusc | ||||||||
| Never | 1.0 | Referent | 1.0 | Referent | 1.0 | Referent | 1.0 | Referent |
| Past | 0.96 | 0.80, 1.15 | 1.03 | 0.83, 1.27 | 0.96 | 0.83, 1.10 | 1.04 | 0.90, 1.21 |
| Current | 2.04 | 1.62, 2.56 | 1.88 | 1.48, 2.39 | 1.08 | 0.88, 1.32 | 1.56 | 1.29, 1.88 |
| Smoking amount/duration (per 46 pack-years)c | 1.56 | 1.30, 1.87 | 3.54 | 3.02, 4.16 | 1.62 | 1.37, 1.92 | 2.00 | 1.73, 2.31 |
| Physical activity (per 33 MET-hours/week)c | 0.88 | 0.75, 1.03 | 0.97 | 0.83, 1.13 | 0.91 | 0.81, 1.04 | 0.76 | 0.66, 0.87 |
| Alcohol intake, g/dayc | ||||||||
| None | 1.0 | Referent | 1.0 | Referent | 1.0 | Referent | 1.0 | Referent |
| 0.1–9.9 | 0.72 | 0.63, 0.84 | 1.03 | 0.88, 1.21 | 1.04 | 0.92, 1.18 | 0.82 | 0.73, 0.93 |
| 10–29.9 | 0.73 | 0.60, 0.90 | 0.97 | 0.80, 1.18 | 1.15 | 0.98, 1.36 | 0.82 | 0.70, 0.96 |
| ≥30 | 1.04 | 0.81, 1.33 | 1.09 | 0.83, 1.40 | 1.07 | 0.81, 1.37 | 0.95 | 0.77, 1.18 |
| Nut consumption, servings/weekc | ||||||||
| None | 1.0 (Referent) | |||||||
| ≤1 | 0.92 (0.87, 0.98) | |||||||
| ≥2 | 0.86 (0.77, 0.95) | |||||||
| Polyunsaturated fat (per 3% energy)f | 0.78 | 0.67, 0.92 | 0.97 | 0.83, 1.14 | 0.89 | 0.78, 1.02 | 0.77 | 0.68, 0.88 |
| Glycemic load (per 41 units)f | 1.22 (1.12, 1.34) | |||||||
| Dietary cholesterol (per 105 mg/1,000 kcal)f | 1.17 (1.09, 1.26) | |||||||
| Cereal fiber (per 4 g, energy adjusted)f | 0.84 (0.78, 0.91) | |||||||
| Systolic blood pressure, mm Hgc | ||||||||
| <120 | 1.0 | Referent | 1.0 | Referent | 1.0 | Referent | 1.0 | Referent |
| 120–139 | 1.26 | 1.06, 1.48 | 0.87 | 0.75, 1.02 | 1.02 | 0.90, 1.15 | 1.03 | 0.91, 1.17 |
| 140–149 | 1.73 | 1.42, 2.10 | 0.99 | 0.81, 1.22 | 1.09 | 0.92, 1.29 | 1.26 | 1.07, 1.47 |
| 150–159 | 1.80 | 1.36, 2.39 | 1.34 | 0.97, 1.84 | 1.19 | 0.91, 1.57 | 1.42 | 1.11, 1.81 |
| ≥160 | 2.60 | 1.92, 3.51 | 1.25 | 0.82, 1.93 | 1.04 | 0.70, 1.56 | 1.31 | 0.95, 1.80 |
| Use of blood pressure medicationsg | 1.57 | 1.36, 1.82 | 0.96 | 0.79, 1.16 | 1.00 | 0.86, 1.16 | 1.23 | 1.08, 1.41 |
| Personal history of diabetesh | 3.31 | 2.70, 4.06 | 1.18 | 0.80, 1.74 | 1.28 | 0.95, 1.71 | 3.60 | 2.97, 4.34 |
| Parental MI before age 60 yearsh | 1.14 (1.06, 1.22) | |||||||
| Time since menopause (per 13 years)c | 1.33 | 1.11, 1.59 | 1.21 | 1.00, 1.48 | 0.90 | 0.76, 1.07 | 1.21 | 1.04, 1.41 |
Abbreviations: CI, confidence interval; HR, hazard ratio; MET, metabolic equivalent; MI, myocardial infarction.
Units for continuous variables correspond to the difference between the 90th and 10th percentile values.
Adjusted for all other risk factors in table.
From 1986 questionnaire.
From 1980 questionnaire.
From 1976 questionnaire; 1 inch = 2.54 cm.
Average of values from 1980, 1984, and 1986 questionnaires.
From 1988 questionnaire.
From 1986 or previous questionnaires.
Table 1.
Continued
| No. | % | Median | 10th Percentile | 90th Percentile | |
| Alcohol intake, g/day | |||||
| None | 17,216 | 34.4 | |||
| 0.1–9.9 | 22,115 | 44.1 | |||
| 10–29.9 | 8,240 | 16.4 | |||
| ≥30 | 2,541 | 5.1 | |||
| Nut consumption, servings/week | |||||
| None | 18,892 | 37.7 | |||
| ≤1 | 25,857 | 51.6 | |||
| ≥2 | 5,363 | 10.7 | |||
| Systolic blood pressure, mm Hg | |||||
| <120 | 23,309 | 46.5 | |||
| 120–139 | 18,836 | 37.6 | |||
| 140–149 | 5,966 | 11.9 | |||
| 150–159 | 1,375 | 2.7 | |||
| ≥160 | 626 | 1.3 | |||
| Use of blood pressure medications | 6,419 | 12.8 | |||
| Personal history of diabetes | 1,085 | 2.2 | |||
| Parental MI before age 60 years | 9,025 | 18.0 |
Abbreviations: GL, glycemic load; MET, metabolic equivalent; MI, myocardial infarction; SD, standard deviation.
One inch = 2.54 cm.
Among past and current smokers only.
Glycemic load = glycemic index (%) × grams of carbohydrate per serving. One unit of glycemic load approximates the glycemic effect of 1 gram of glucose.
To evaluate the performance of the final competing risks model in Table 4, we first examined the area under the curve (AUC or C statistic) with an approach described by Chambless and Diao (28), which uses recursive calculation over the ordered time of events, analogous to the Kaplan-Meier approach to survival function estimation. The AUC for the standard Cox proportional hazards model for all-cause mortality (Table 2) was 0.74 at 5 years, 0.75 at 10 years, and 0.76 at 15 years into the follow-up period; these values were almost identical for the final competing risks model in Table 4, indicating similar discriminatory accuracy.
Because the C statistic may not be very sensitive to change for time-to-event data (32), we also used likelihood ratio tests to compare the models. The difference in the log likelihoods of the Cox proportional hazards model for all-cause mortality (Table 2) and the final competing risks model (Table 4) was 652.96 (df = 54; P < 0.0001), indicating a significant improvement with the competing risks model. The fit of the final competing risks model (Table 4) was not significantly different from that of the initial full model (Table 3) (difference in log likelihoods = 13.14, df = 21, P = 0.90).
DISCUSSION
Our findings confirm those from other studies, showing strong and significant associations between lifestyle and dietary factors and risk of mortality in middle-aged women (1–5, 23, 24, 33–40). Having a low body mass index at age 18 years, minimizing weight gain during adulthood, not smoking or decreasing the amount of smoking, and engaging in physical activity are associated with lower overall mortality risk. Small-to-moderate amounts of alcohol and larger amounts of polyunsaturated fats, nuts, and cereal fiber also are related to lower mortality, whereas higher cholesterol intake and higher glycemic load are associated with greater mortality. Additional factors such as taller height, high systolic blood pressure and use of blood pressure medications, personal history of diabetes, and earlier menopause also may contribute to increased mortality risk. Women with the “worst” risk profile for modifiable lifestyle and dietary factors have more than 12 times greater risk of all-cause mortality compared with those having the “best” risk profile.
From a methodological standpoint, there are several advantages to using competing risks survival analysis to analyze the composite endpoint of all-cause mortality. Unlike Cox proportional hazards regression, in which risk factors are constrained to have common associations with all components of the outcome, this method allows for some risk factors to have different associations with different causes of death. In contrast to polytomous logistic regression (26), the survival analysis approach offers precise incorporation of censoring times, direct estimation of relative hazard ratios, and the use of stratification to accommodate heterogeneity in the effects of age and/or time (31). As we have shown, likelihood ratio tests can still be used in the setting of competing risks survival analysis to evaluate heterogeneity, and various measures can be used to examine discriminatory accuracy. Previous analyses in this cohort have focused primarily on examining the effect of a single risk factor on mortality, rather than developing a more comprehensive model that incorporates the effects of multiple risk factors. The major advantage of the competing risks approach is that the effects of each risk factor can be estimated and formally compared across different causes of death; then, based on these comparisons, a single model for all-cause mortality can be developed in which risk factors can have different or common effects across specific causes of mortality.
This study also has several limitations. We did not update risk factors in the competing risks model, using only participants’ risk factor status at the beginning of the follow-up period, which would attenuate associations. We made this decision to avoid the possibility of clinical or subclinical disease affecting risk factor status. However, using repeated exposure assessments and focusing on a single outcome would be more powerful for addressing possible causal associations, which has been done in this cohort previously (39). Restriction of the population to nurses could reduce the generalizability of the results, but it also controls for socioeconomic status. Moreover, important strengths of this population are the excellent follow-up rate and high-quality data on risk factors. Although a large number of participants in the original cohort were excluded because of missing or incomplete data on diet or other risk factors, the mortality risk over the 18-year follow-up period among all participants who returned the 1986 questionnaire (12.7%) was very similar to the risk among those with complete data on diet and other risk factors (11.7%), suggesting that selection bias is not a major issue. Finally, although we examined the internal validity of our model, we did not validate it in an external population. This would be an important next step before the model can be used for risk prediction.
In summary, this study showed that many lifestyle and dietary factors are related to risk of mortality in middle-aged women, and the associations for some risk factors differ according to type of mortality. We developed a competing risks model for all-cause mortality and found that it had a significantly better fit compared with a standard model in which all of the risk factors had common associations across all causes of death. In the future, this new model may be used to identify individuals at increased risk of mortality.
Supplementary Material
Acknowledgments
Author affiliations: Division of General Medicine and Primary Care, Department of Medicine, Brigham and Women's Hospital and Harvard Medical School, Boston, Massachusetts (Heather J. Baer); Department of Epidemiology, Harvard School of Public Health, Boston, Massachusetts (Heather J. Baer, Frank B. Hu, Susan E. Hankinson, Walter C. Willett, Meir Stampfer, Bernard Rosner); Division of Preventive Medicine, Department of Medicine, Brigham and Women's Hospital and Harvard Medical School, Boston, Massachusetts (Robert J. Glynn); Department of Biostatistics, Harvard School of Public Health, Boston, Massachusetts (Robert J. Glynn, Bernard Rosner); Department of Nutrition, Harvard School of Public Health, Boston, Massachusetts (Frank B. Hu, Walter C. Willett); Channing Laboratory, Department of Medicine, Brigham and Women's Hospital and Harvard Medical School, Boston, Massachusetts (Frank B. Hu, Susan E. Hankinson, Walter C. Willett, Meir Stampfer, Bernard Rosner); and Alvin J. Siteman Cancer Center and Department of Surgery, Washington University School of Medicine, St. Louis, Missouri (Graham A. Colditz).
This work was supported by the National Cancer Institute, National Institutes of Health (P01 CA87969). G. A. C. also is supported by an American Cancer Society Clinical Research Professorship.
Conflict of interest: none declared.
Glossary
Abbreviations
- AUC
area under the curve
- CI
confidence interval
- HR
hazard ratio
- ICD-8
International Classification of Diseases, Eighth Revision
- MET
metabolic equivalent
- PLRT
P value for the likelihood ratio test
References
- 1.Fuchs CS, Stampfer MJ, Colditz GA, et al. Alcohol consumption and mortality among women. N Engl J Med. 1995;332(19):1245–1250. doi: 10.1056/NEJM199505113321901. [DOI] [PubMed] [Google Scholar]
- 2.Hu FB, Willett WC, Li T, et al. Adiposity as compared with physical activity in predicting mortality among women. N Engl J Med. 2004;351(26):2694–2703. doi: 10.1056/NEJMoa042135. [DOI] [PubMed] [Google Scholar]
- 3.Rockhill B, Willett WC, Manson JE, et al. Physical activity and mortality: a prospective study among women. Am J Public Health. 2001;91(4):578–583. doi: 10.2105/ajph.91.4.578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kenfield SA, Stampfer MJ, Rosner BA, et al. Smoking and smoking cessation in relation to mortality in women. JAMA. 2008;299(17):2037–2047. doi: 10.1001/jama.299.17.2037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Grodstein F, Stampfer MJ, Colditz GA, et al. Postmenopausal hormone therapy and mortality. N Engl J Med. 1997;336(25):1769–1775. doi: 10.1056/NEJM199706193362501. [DOI] [PubMed] [Google Scholar]
- 6.Colditz GA, Hankinson SE. The Nurses’ Health Study: lifestyle and health among women. Nat Rev Cancer. 2005;5(5):388–396. doi: 10.1038/nrc1608. [DOI] [PubMed] [Google Scholar]
- 7.Troy LM, Hunter DJ, Manson JE, et al. The validity of recalled weight among younger women. Int J Obes Relat Metab Disord. 1995;19(8):570–572. [PubMed] [Google Scholar]
- 8.Willett W, Stampfer MJ, Bain C, et al. Cigarette smoking, relative weight, and menopause. Am J Epidemiol. 1983;117(6):651–658. doi: 10.1093/oxfordjournals.aje.a113598. [DOI] [PubMed] [Google Scholar]
- 9.Wolf AM, Hunter DJ, Colditz GA, et al. Reproducibility and validity of a self-administered physical activity questionnaire. Int J Epidemiol. 1994;23(5):991–999. doi: 10.1093/ije/23.5.991. [DOI] [PubMed] [Google Scholar]
- 10.Willett WC, Sampson L, Stampfer MJ, et al. Reproducibility and validity of a semiquantitative food frequency questionnaire. Am J Epidemiol. 1985;122(1):51–65. doi: 10.1093/oxfordjournals.aje.a114086. [DOI] [PubMed] [Google Scholar]
- 11.Giovannucci E, Colditz G, Stampfer MJ, et al. The assessment of alcohol consumption by a simple self-administered questionnaire. Am J Epidemiol. 1991;133(8):810–817. doi: 10.1093/oxfordjournals.aje.a115960. [DOI] [PubMed] [Google Scholar]
- 12.Colditz GA, Martin P, Stampfer MJ, et al. Validation of questionnaire information on risk factors and disease outcomes in a prospective cohort study of women. Am J Epidemiol. 1986;123(5):894–900. doi: 10.1093/oxfordjournals.aje.a114319. [DOI] [PubMed] [Google Scholar]
- 13.Colditz GA, Stampfer MJ, Willett WC, et al. Reproducibility and validity of self-reported menopausal status in a prospective cohort study. Am J Epidemiol. 1987;126(2):319–325. doi: 10.1093/aje/126.2.319. [DOI] [PubMed] [Google Scholar]
- 14.Ainsworth BE, Haskell WL, Leon AS, et al. Compendium of physical activities: classification of energy costs of human physical activities. Med Sci Sports Exerc. 1993;25(1):71–80. doi: 10.1249/00005768-199301000-00011. [DOI] [PubMed] [Google Scholar]
- 15.Ainsworth BE, Haskell WL, Whitt MC, et al. Compendium of physical activities: an update of activity codes and MET intensities. Med Sci Sports Exerc. 2000;32(suppl 9):S498–S504. doi: 10.1097/00005768-200009001-00009. [DOI] [PubMed] [Google Scholar]
- 16.Willett WC, Stampfer MJ. Implications of total energy intake for epidemiologic analyses. In: Willett WC, editor. Nutritional Epidemiology. New York, NY: Oxford University Press; 1998. [Google Scholar]
- 17.Halton TL, Willett WC, Liu S, et al. Low-carbohydrate-diet score and the risk of coronary heart disease in women. N Engl J Med. 2006;355(19):1991–2002. doi: 10.1056/NEJMoa055317. [DOI] [PubMed] [Google Scholar]
- 18.Willett WC, editor. Nutritional Epidemiology. New York, NY: Oxford University Press; 1998. [Google Scholar]
- 19.Rich-Edwards JW, Corsano KA, Stampfer MJ. Test of the National Death Index and Equifax Nationwide Death Search. Am J Epidemiol. 1994;140(11):1016–1019. doi: 10.1093/oxfordjournals.aje.a117191. [DOI] [PubMed] [Google Scholar]
- 20.Stampfer MJ, Willett WC, Speizer FE, et al. Test of the National Death Index. Am J Epidemiol. 1984;119(5):837–839. doi: 10.1093/oxfordjournals.aje.a113804. [DOI] [PubMed] [Google Scholar]
- 21.New Surgeon General's report expands list of smoking-related diseases. FDA Consum. 2004;38(4):9. [PubMed] [Google Scholar]
- 22.The 2004 United States Surgeon General's Report: The Health Consequences of Smoking. N S W Public Health Bull. 2004;15(5–6):107. [PubMed] [Google Scholar]
- 23.Kenfield SA, Wei EK, Rosner BA, et al. Burden of smoking on cause-specific mortality: application to the Nurses’ Health Study. Tob Control. 2010;19(3):248–254. doi: 10.1136/tc.2009.032839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhang C, Rexrode KM, van Dam RM, et al. Abdominal obesity and the risk of all-cause, cardiovascular, and cancer mortality: sixteen years of follow-up in US women. Circulation. 2008;117(13):1658–1667. doi: 10.1161/CIRCULATIONAHA.107.739714. [DOI] [PubMed] [Google Scholar]
- 25.Rosner B. Fundamentals of Biostatistics. Belmont, CA: Wadsworth Publishing Company; 1995. [Google Scholar]
- 26.Glynn RJ, Rosner B. Methods to evaluate risks for composite end points and their individual components. J Clin Epidemiol. 2004;57(2):113–122. doi: 10.1016/j.jclinepi.2003.02.001. [DOI] [PubMed] [Google Scholar]
- 27.Lunn M, McNeil D. Applying Cox regression to competing risks. Biometrics. 1995;51(2):524–532. [PubMed] [Google Scholar]
- 28.Chambless LE, Diao G. Estimation of time-dependent area under the ROC curve for long-term risk prediction. Stat Med. 2006;25(20):3474–3486. doi: 10.1002/sim.2299. [DOI] [PubMed] [Google Scholar]
- 29.Hebert PR, Rich-Edwards JW, Manson JE, et al. Height and incidence of cardiovascular disease in male physicians. Circulation. 1993;88(4 pt 1):1437–1443. doi: 10.1161/01.cir.88.4.1437. [DOI] [PubMed] [Google Scholar]
- 30.Rich-Edwards JW, Manson JE, Stampfer MJ, et al. Height and the risk of cardiovascular disease in women. Am J Epidemiol. 1995;142(9):909–917. doi: 10.1093/oxfordjournals.aje.a117738. [DOI] [PubMed] [Google Scholar]
- 31.Glynn RJ, Rosner B. Comparison of risk factors for the competing risks of coronary heart disease, stroke, and venous thromboembolism. Am J Epidemiol. 2005;162(10):975–982. doi: 10.1093/aje/kwi309. [DOI] [PubMed] [Google Scholar]
- 32.Pencina MJ, D'Agostino RB, Sr, D'Agostino RB, Jr, et al. Evaluating the added predictive ability of a new marker: from area under the ROC curve to reclassification and beyond. Stat Med. 2008;27(2):157–172. doi: 10.1002/sim.2929. discussion 207–212. [DOI] [PubMed] [Google Scholar]
- 33.Chan AT, Manson JE, Feskanich D, et al. Long-term aspirin use and mortality in women. Arch Intern Med. 2007;167(6):562–572. doi: 10.1001/archinte.167.6.562. [DOI] [PubMed] [Google Scholar]
- 34.Colditz GA. Oral contraceptive use and mortality during 12 years of follow-up: the Nurses’ Health Study. Ann Intern Med. 1994;120(10):821–826. doi: 10.7326/0003-4819-120-10-199405150-00002. [DOI] [PubMed] [Google Scholar]
- 35.Hu FB, Stampfer MJ, Solomon CG, et al. The impact of diabetes mellitus on mortality from all causes and coronary heart disease in women: 20 years of follow-up. Arch Intern Med. 2001;161(14):1717–1723. doi: 10.1001/archinte.161.14.1717. [DOI] [PubMed] [Google Scholar]
- 36.Kawachi I, Colditz GA, Stampfer MJ, et al. Smoking cessation in relation to total mortality rates in women. A prospective cohort study. Ann Intern Med. 1993;119(10):992–1000. doi: 10.7326/0003-4819-119-10-199311150-00005. [DOI] [PubMed] [Google Scholar]
- 37.Manson JE, Willett WC, Stampfer MJ, et al. Body weight and mortality among women. N Engl J Med. 1995;333(11):677–685. doi: 10.1056/NEJM199509143331101. [DOI] [PubMed] [Google Scholar]
- 38.Must A, Jacques PF, Dallal GE, et al. Long-term morbidity and mortality of overweight adolescents. A follow-up of the Harvard Growth Study of 1922 to 1935. N Engl J Med. 1992;327(19):1350–1355. doi: 10.1056/NEJM199211053271904. [DOI] [PubMed] [Google Scholar]
- 39.van Dam RM, Li T, Spiegelman D, et al. Combined impact of lifestyle factors on mortality: prospective cohort study in US women [electronic article] BMJ. 2008;337 doi: 10.1136/bmj.a1440. a1440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.van Dam RM, Willett WC, Manson JE, et al. The relationship between overweight in adolescence and premature death in women. Ann Intern Med. 2006;145(2):91–97. doi: 10.7326/0003-4819-145-2-200607180-00006. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.