Abstract
Prospective investigations of circulating vitamin D concentrations suggest inverse associations with colorectal cancer risk, although inconsistencies remain and few studies have examined the impact of season. The authors conducted a prospective case-control study of 239 colon cancer cases and 192 rectal cancer cases (diagnosed in 1993–2005) and 428 controls matched on age and blood collection date within the Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study, a cohort study of Finnish male smokers. Baseline serum 25-hydroxyvitamin D (25(OH)D) concentrations were categorized using a priori defined cutpoints of <25, 25–<37.5, 37.5–<50, 50–<75, and ≥75 nmol/L and by season-specific and season-standardized 25(OH)D quartiles. Conditional logistic regression models yielded multivariate-adjusted odds ratios for the predefined cutpoints of 0.63, 0.91, 0.73, 1.00 (referent), and 1.44 for colon cancer and 0.64, 0.58, 0.84, 1.00, and 0.76 for rectal cancer, respectively (all 95% confidence intervals included 1.00). Colon cancer risks were significantly elevated for the highest season-specific and season-standardized quartiles versus the lowest quartiles (OR = 2.11 (95% CI: 1.20, 3.69) and OR = 1.88 (95% CI: 1.07, 3.28), respectively), while rectal cancer risk estimates were null. These results provide no evidence to support an inverse association between vitamin D status and colon or rectal cancer risk; instead, they suggest a positive association for colon cancer.
Keywords: cohort studies, colorectal neoplasms, prospective studies, vitamin D
In 1980, ecologic findings that colorectal cancer mortality rates in the United States were inversely associated with sunlight exposure, with vitamin D as the suggested mechanism (1), generated interest in the relation between vitamin D and colorectal cancer. Recent reviews have concluded that low serum vitamin D concentrations are associated with increased risk of colorectal cancer (2–4); however, not all reviews interpret the data to be consistent and convincing (5).
Most prospective studies have supported the hypothesis of an inverse association (6–14); however, differences exist by anatomic subsite, and not all associations are statistically significant. In addition, all but 1 (12) of 9 (6–14) prospective studies matched subjects on the date of blood collection, but only 4 (9–12) used additional techniques to adjust for seasonal variation in vitamin D and its influence on the relation between vitamin D and colorectal cancer.
In addition to dietary and supplemental sources, vitamin D is synthesized from 7-dehydrocholesterol by the skin when exposed to solar radiation (15) and is then hydroxylated in the liver to form 25-hydroxyvitamin D (25(OH)D), the accepted biomarker of circulating vitamin D (16). 25(OH)D is further hydroxylated in the kidney and other organs to its active form, 1,25-dihydroxyvitamin D (1,25(OH)2D).
As part of a programmatic effort to comprehensively evaluate biomarkers of vitamin D and cancer, we conducted a nested case-control study within the Alpha-Tocopherol, Beta-Carotene Cancer Prevention (ATBC) Study, a cohort study of male smokers in Finland. Multiple approaches were used to address the issues related to seasonal variation in 25(OH)D concentrations.
MATERIALS AND METHODS
Study population
The ATBC Study was a randomized, double-blind, placebo-controlled, primary prevention trial involving daily supplementation with α-tocopherol (50 mg/day), β-carotene (20 mg/day), both, or placebo (17). From 1985 to 1988, the ATBC investigators recruited 29,133 men aged 50–69 years who smoked at least 5 cigarettes per day from southwestern Finland (latitude of study area, 60°N–64°N). Study supplementation continued for 5–8 years (median, 6.1 years) until death or trial closure (April 30, 1993). The study was approved by the institutional review boards of the US National Cancer Institute and the National Public Health Institute of Finland. Written informed consent was obtained from each participant prior to randomization.
Case identification and control selection
Cases (n = 428) were defined as incident colonic or rectal adenocarcinoma (International Classification of Diseases, Ninth Revision, codes 153 and 154, respectively), excluding cancers of the appendix and anus and cancers of unknown histology, identified through the Finnish Cancer Registry, with follow-up through April 30, 2005. Cases from a prior analysis (7) (diagnosed through November 1993) were excluded; therefore, all but 8 cases were diagnosed after November 1993. Three subjects who were diagnosed with both colon and rectal cancer on the same day were included in analyses of both sites, resulting in a total of 239 colon cancers and 192 rectal cancers. Cases of distal colon cancer (International Classification of Diseases, Ninth Revision, codes 153.2 and 153.3) and rectal cancer were also combined (n = 292). For cases diagnosed through April 1999 (116 colon and 89 rectum), 2 study physicians reviewed medical records for diagnostic confirmation and staging, and 1 pathologist reviewed histolopathologic or cytologic specimens where available. Information on colorectal cancer cases diagnosed since May 1999 (123 colon and 103 rectum) was derived solely from the Finnish Cancer Registry, which provides almost 100% case ascertainment (18). Controls were alive and cancer-free at the time of case diagnosis and were matched to cases (1:1) on age at randomization (±1 year) and date of baseline serum collection (±30 days).
Serum 25(OH)D determination
Fasting baseline serum samples were stored at –70°C. 25(OH)D was measured in early 2008 at Heartland Assays, Inc. (Ames, Iowa) using the DiaSorin Liaison 25(OH)D TOTAL assay platform by means of a direct, competitive chemiluminescence immunoassay (19, 20).
Each batch contained matched case/control sets and 4 or 6 blinded quality control samples, consisting of either standard reference material provided by the US National Institute of Standards and Technology (21) or an ATBC serum pool. Interbatch and intrabatch coefficients of variation, calculated using a nested components-of-variance analysis (22), were 12.7%–13.6% and 9.3%–11.0%, respectively, for the standard reference material and 12.3% and 10.5%, respectively, for the ATBC serum pool.
Statistical analysis
Wilcoxon rank-sum and chi-square tests (for continuous and categorical variables, respectively) were used to compare characteristics of cases and controls. Conditional logistic regression was used to determine odds ratios and 95% confidence intervals. The 25(OH)D concentrations were analyzed using several different approaches. First, cutpoints were defined on the basis of clinical definitions in the literature (15, 16, 23, 24) as <25, 25–<37.5, 37.5–<50, 50–<75, and ≥75 nmol/L. The referent category was defined as 50–<75 nmol/L because this includes the mean 25(OH)D concentration of the US population (24) and is the referent category used in several other analyses of 25(OH)D and cancer (12, 25–30). Although the top category had few subjects, it was retained because of interest in higher vitamin D concentrations. Results from both the overall models and those stratified on season of blood collection are presented in this manner. The second approach used season-specific quartiles of 25(OH)D, calculated on the basis of the distribution among the colorectal controls split by season and entered into the models as indicator variables. The third approach used season-standardized 25(OH)D quartiles (described below). Results from the latter 2 approaches are presented with the lowest quartile as the referent category. Tests for linear trend were conducted by assigning to each category an ordinal value (1–5 or 1–4 as necessary) and then treating this parameter as a continuous variable.
The vitamin D assays for this study were conducted in conjunction with ATBC studies of other cancer sites, which included a total of 1,221 controls. Based on monthly median 25(OH)D concentrations in this larger set of controls, season was defined as the “darker months” (November–April) versus the “sunnier months” (May–October). The season-standardized 25(OH)D values were calculated by regressing log-transformed 25(OH)D concentration against calendar week of blood collection in the 1,221 controls, using a locally weighted polynomial regression method, and creating quartiles of the residuals (20, 31). This method was also used to create a smoothed plot of predicted 25(OH)D values by calendar week of blood collection in the 1,221 controls.
Variables assessed for confounding included those shown in Table 1, as well as coastal residence, urban residence, marital status, education, vocational training, tooth loss, and intakes of butter, poultry, alcohol, iron, folate, and pyridoxine. Potential confounders were defined as covariables that were associated with either colon or rectal cancer or produced a >10% change in the 25(OH)D coefficients when added to the univariate models, and which were not likely to be direct determinants of 25(OH)D (e.g., vitamin D intake or supplement use). All potential confounders were then considered jointly and removed from the models if that produced a <10% change in the 25(OH)D coefficients (analyses were conducted separately for each outcome and each season). The resulting confounders for colon cancer were serum α-tocopherol, β-carotene, and retinol, and the confounders for rectal cancer were serum α-tocopherol, β-carotene, and years of smoking. Given the similarity between the colon and rectal cancer confounders and because the 25(OH)D risk estimates were not affected when all 4 covariables were included, these 4 identified confounders were retained in all models.
Table 1.
Selected Baseline Characteristics of Cases and Controls, Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study, Finland, 1985–2005
| Characteristic | Colon |
Rectum |
||||||||||||
| Cases (n = 239) |
Controls (n = 239) |
P Valuea | Cases (n = 192) |
Controls (n = 192) |
P Valuea | |||||||||
| Median | IQR | % | Median | IQR | % | Median | IQR | % | Median | IQR | % | |||
| Age, yearsb | 58 | 54–61 | 58 | 54–61 | 57 | 53–62 | 57 | 53–62 | ||||||
| Height, cm | 174 | 170–178 | 174 | 170–177 | 0.43 | 174 | 170–178 | 174 | 169–178 | 0.77 | ||||
| Weight, kg | 81.2 | 71.3–89.6 | 77.1 | 70.7–86.1 | 0.02 | 77.9 | 69.1–86.1 | 77.1 | 70.0–84.9 | 0.77 | ||||
| Body mass indexc | 26.6 | 24.1–29.8 | 26.0 | 23.7–28.4 | 0.04 | 25.7 | 23.3–28.5 | 25.6 | 23.7–27.7 | 0.81 | ||||
| Physical activity (heavy) | ||||||||||||||
| Leisure | 4.2 | 8.8 | 0.08 | 7.3 | 7.3 | 0.98 | ||||||||
| Occupational | 6.3 | 6.7 | 0.85 | 7.8 | 11.5 | 0.23 | ||||||||
| Family history of colorectal cancerd | 4.8 | 1.5 | 0.06 | 1.5 | 0.6 | 0.47 | ||||||||
| Smoking amount, cigarettes/day | 20 | 12–25 | 20 | 12–20 | 0.29 | 20 | 15–25 | 20 | 15–22 | 0.97 | ||||
| Smoking duration, years | 38 | 30–42 | 37 | 31–42 | 0.70 | 36 | 32–42 | 36 | 30–42 | 0.42 | ||||
| Blood collected in darker months (November–April)b | 69.5 | 66.5 | 61.5 | 62.0 | ||||||||||
| Supplement use | ||||||||||||||
| Calcium (yes) | 8.8 | 9.2 | 0.28 | 13.0 | 13.0 | 0.58 | ||||||||
| Vitamin D (yes) | 6.7 | 5.9 | 0.16 | 8.9 | 10.9 | 0.68 | ||||||||
| Daily dietary intakee | ||||||||||||||
| Energy, kcal | 2,531 | 2,086–3,102 | 2,648 | 2,214–3,160 | 0.31 | 2,622 | 2,149–3,138 | 2,549 | 2,136–3,061 | 0.57 | ||||
| Milk, g | 0 | 0–340 | 0 | 0–340 | 0.83 | 0 | 0–340 | 13.3 | 0–460 | 0.23 | ||||
| Fish, g | 31.0 | 21.5–46.8 | 34.9 | 20.6–56.3 | 0.36 | 34.0 | 20.7–54.0 | 32.7 | 20.3–47.9 | 0.22 | ||||
| Vitamin D, μg | 4.7 | 3.3–6.8 | 5.0 | 3.2–7.1 | 0.69 | 4.9 | 3.1–7.0 | 4.6 | 3.3–6.2 | 0.29 | ||||
| Calcium, mg | 1,316 | 966–1,728 | 1,354 | 1,007–1,775 | 0.37 | 1,303 | 966–1,578 | 1,378 | 1,016–1,708 | 0.17 | ||||
| Serum biomarkers | ||||||||||||||
| 25(OH)D, nmol/L | 32.4 | 22.5–49.3 | 29.6 | 20.5–45.7 | 0.14 | 34.6 | 21.3–50.9 | 31.2 | 22.1–45.6 | 0.63 | ||||
| α-tocopherol, mg/L | 11.7 | 9.9–13.4 | 11.7 | 10.4–14.0 | 0.14 | 11.5 | 9.4–13.7 | 11.2 | 9.8–13.5 | 0.98 | ||||
| β-carotene, μg/L | 159 | 104–256 | 193 | 127–299 | 0.001 | 167 | 105–263 | 184 | 131–280 | 0.08 | ||||
| Total cholesterol, mmol/L | 6.14 | 5.47–6.93 | 6.28 | 5.54–7.20 | 0.28 | 6.13 | 5.33–6.89 | 6.17 | 5.57–6.93 | 0.35 | ||||
| Retinol, μg/L | 602 | 520–683 | 572 | 505–662 | 0.06 | 580 | 500–655 | 572 | 494–670 | 0.90 | ||||
Abbreviations: 25(OH)D, 25-hydroxyvitamin D; IQR, interquartile range.
Based on the chi-squared test (for categorical variables) or the Wilcoxon rank-sum test (for continuous variables). All P values were 2-sided.
Matching factor.
Weight (kg)/height (m)2.
Family history of colon or rectal cancer, respectively; data were available for 82% of the colon cancer set and 78% of the rectal cancer set.
Dietary data were available on 95% of subjects.
Stratum-specific subgroup models were fitted using the 25(OH)D season-specific quartile variable, with the lowest quartile serving as the referent category. Models stratified into separate subgroups based on age, body mass index, number of cigarettes smoked per day, calcium intake, serum α-tocopherol, β-carotene, and retinol concentrations (split at the median), physical activity, and α-tocopherol and β-carotene intervention groups were fitted using unconditional multivariate logistic regression, with additional adjustment for the matching factors. Analyses stratified on date of the case diagnosis (split at the median diagnosis date of November 15, 1999, for both colon and rectal cancer cases) were conducted conditionally. We evaluated effect modification statistically by comparing models with and without a cross-product term of 25(OH)D (categorical) and the effect modifier, using the log-likelihood test. Statistical analyses were performed using SAS software, version 9.1.3 (SAS Institute, Inc., Cary, North Carolina), and all P values were 2-sided.
RESULTS
Colon cancer cases had significantly higher average weight and body mass index and a lower serum β-carotene concentration than their respective controls, while rectal cancer cases did not differ from controls with respect to any of the baseline characteristics (Table 1). When colon and rectal cancer cases were combined, colorectal cancer cases had significantly higher weight (P = 0.04) and a lower serum β-carotene concentration (P = 0.0004) than controls. Both colon and rectal cancer cases had nonsignificantly higher 25(OH)D concentrations than controls (9.5% (P = 0.14) and 10.9% (P = 0.63), respectively).
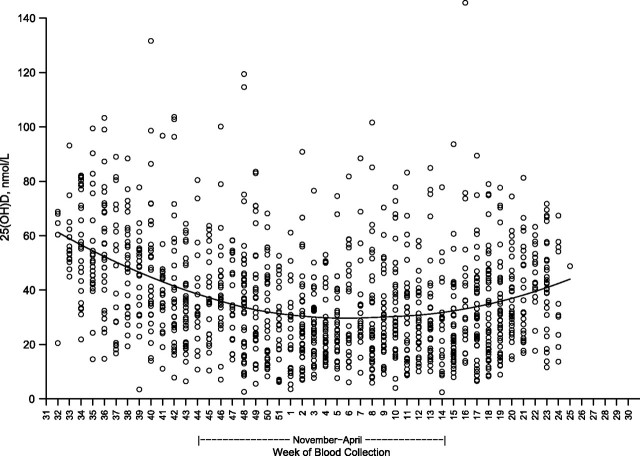
Figure 1 shows the expected seasonal variation in 25(OH)D concentrations, with values decreasing through the fall, being lowest in the winter months, and rising in late spring and early summer. There was substantial between-subject variation, however, such that high and low 25(OH)D concentrations were evident throughout the year. The median 25(OH)D concentration among controls with blood drawn in the darker months (November–April) was 27.8 nmol/L (interquartile range, 17.9–42.3), with a corresponding value for the sunnier months (May–October) of 41.7 nmol/L (interquartile range, 29.3–56.1). Serum was collected from few ATBC Study participants in June and August, and none was collected during July.
Figure 1.
Serum 25-hydroxyvitamin D (25(OH)D) concentrations according to calendar week of blood collection in the Alpha-Tocopherol, Beta-Carotene Cancer Prevention (ATBC) Study, Finland, 1985–2005. Each circle represents an individual 25(OH)D measurement, and the smoothed line shows the predicted 25(OH)D values calculated using locally weighted polynomial regression. Results were obtained among 1,221 controls (including colorectal controls) from several ATBC nested substudies with blood samples assayed in the same laboratory at the same time.
We observed no significant association between 25(OH)D concentration and colorectal, colon, or rectal cancer using the a priori defined cutpoints (Table 2), although the general pattern was for reduced risks within the lower 25(OH)D categories. Compared with the referent group, men with a 25(OH)D concentration less than 25 nmol/L had an approximately 30% lower colorectal cancer risk that was nearly statistically significant. The odds ratios comparing the highest 25(OH)D category (>75 nmol/L) with the lowest (<25 nmol/L) were 1.47 (95% CI: 0.72, 3.01), 2.28 (95% CI: 0.77, 6.78), and 1.19 (95% CI: 0.43, 3.28) for colorectal cancer, colon cancer, and rectal cancer, respectively. Combining distal colon cancer cases and rectal cancer cases produced patterns similar to those presented in Table 2 (data not shown), and there were few differences by season of blood collection in any of these models (Table 2).
Table 2.
Odds Ratios for the Association Between Serum 25-Hydroxyvitamin D and Risks of Colon and Rectal Cancer Obtained Using A Priori Defined Cutpoints, Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study, Finland, 1985–2005
| 25-Hydroxyvitamin D Concentration, nmol/L | Ptrenda | ||||||||||
| <25.0 |
25.0–<37.5 |
37.5–<50.0 |
50.0–<75.0 |
≥75.0 |
|||||||
| OR | 95% CI | OR | 95% CI | OR | 95% CI | OR | 95% CI | OR | 95% CI | ||
| Colorectal Cancer | |||||||||||
| Total | |||||||||||
| No. of cases/no. of controls | 143/155 | 105/105 | 73/77 | 85/72 | 22/19 | ||||||
| Crude OR | 0.77 | 0.51, 1.14 | 0.85 | 0.56, 1.27 | 0.79 | 0.50, 1.25 | 1.00 | Reference | 1.01 | 0.51, 2.02 | 0.22 |
| Multivariate-adjusted ORb | 0.68 | 0.45, 1.03 | 0.78 | 0.51, 1.20 | 0.78 | 0.49, 1.25 | 1.00 | Reference | 1.00 | 0.49, 2.03 | 0.07 |
| Darker monthsc | |||||||||||
| No. of cases/no. of controls | 115/126 | 71/66 | 48/48 | 40/37 | 7/7 | ||||||
| Crude OR | 0.78 | 0.47, 1.30 | 0.93 | 0.53, 1.61 | 0.86 | 0.47, 1.57 | 1.00 | Reference | 0.88 | 0.27, 2.87 | 0.40 |
| Multivariate-adjusted OR | 0.75 | 0.45, 1.27 | 0.94 | 0.53, 1.67 | 0.90 | 0.48, 1.68 | 1.00 | Reference | 0.91 | 0.27, 3.08 | 0.27 |
| Sunnier monthsd | |||||||||||
| No. of cases/no. of controls | 28/29 | 34/39 | 25/29 | 45/38 | 15/12 | ||||||
| Crude OR | 0.81 | 0.40, 1.65 | 0.75 | 0.40, 1.38 | 0.72 | 0.36, 1.46 | 1.00 | Reference | 1.07 | 0.45, 2.52 | 0.35 |
| Multivariate-adjusted OR | 0.58 | 0.26, 1.27 | 0.56 | 0.28, 1.12 | 0.61 | 0.29, 1.27 | 1.00 | Reference | 0.98 | 0.39, 2.43 | 0.10 |
| Colon Cancer | |||||||||||
| Total | |||||||||||
| No. of cases/no. of controls | 77/87 | 63/60 | 40/44 | 47/40 | 12/8 | ||||||
| Crude OR | 0.73 | 0.42, 1.25 | 0.89 | 0.53, 1.50 | 0.76 | 0.42, 1.38 | 1.00 | Reference | 1.32 | 0.48, 3.60 | 0.22 |
| Multivariate-adjusted OR | 0.63 | 0.36, 1.12 | 0.91 | 0.52, 1.59 | 0.73 | 0.39, 1.36 | 1.00 | Reference | 1.44 | 0.49, 4.26 | 0.11 |
| Darker months | |||||||||||
| No. of cases/no. of controls | 62/70 | 46/43 | 28/28 | 25/21 | 5/4 | ||||||
| Crude OR | 0.73 | 0.38, 1.44 | 0.90 | 0.46, 1.75 | 0.83 | 0.38, 1.79 | 1.00 | Reference | 1.10 | 0.23, 5.27 | 0.36 |
| Multivariate-adjusted OR | 0.65 | 0.32, 1.31 | 0.90 | 0.44, 1.84 | 0.78 | 0.35, 1.75 | 1.00 | Reference | 1.00 | 0.19, 5.18 | 0.24 |
| Sunnier months | |||||||||||
| No. of cases/no. of controls | 15/17 | 17/17 | 12/16 | 22/19 | 7/4 | ||||||
| Crude OR | 0.74 | 0.28, 1.96 | 0.88 | 0.37, 2.10 | 0.67 | 0.26, 1.73 | 1.00 | Reference | 1.44 | 0.38, 5.47 | 0.40 |
| Multivariate-adjusted OR | 0.48 | 0.15, 1.54 | 0.66 | 0.23, 1.90 | 0.49 | 0.16, 1.48 | 1.00 | Reference | 2.14 | 0.44, 10.35 | 0.12 |
| Rectal Cancer | |||||||||||
| Total | |||||||||||
| No. of cases/no. of controls | 68/70 | 42/46 | 33/33 | 39/32 | 10/11 | ||||||
| Crude OR | 0.79 | 0.44, 1.44 | 0.74 | 0.39, 1.41 | 0.82 | 0.41, 1.68 | 1.00 | Reference | 0.77 | 0.29, 2.02 | 0.58 |
| Multivariate-adjusted OR | 0.64 | 0.34, 1.22 | 0.58 | 0.29, 1.17 | 0.84 | 0.40, 1.75 | 1.00 | Reference | 0.76 | 0.28, 2.07 | 0.21 |
| Darker months | |||||||||||
| No. of cases/no. of controls | 55/58 | 25/24 | 20/20 | 16/13 | 2/3 | ||||||
| Crude OR | 0.80 | 0.36, 1.75 | 0.85 | 0.32, 2.23 | 0.83 | 0.31, 2.21 | 1.00 | Reference | 0.58 | 0.09, 3.87 | 0.74 |
| Multivariate-adjusted OR | 0.68 | 0.29, 1.59 | 0.70 | 0.24, 2.02 | 0.99 | 0.35, 2.84 | 1.00 | Reference | 0.74 | 0.10, 5.47 | 0.34 |
| Sunnier months | |||||||||||
| No. of cases/no. of controls | 13/12 | 17/22 | 13/13 | 23/19 | 8/8 | ||||||
| Crude OR | 0.91 | 0.31, 2.61 | 0.64 | 0.27, 1.55 | 0.85 | 0.29, 2.45 | 1.00 | Reference | 0.87 | 0.28, 2.72 | 0.64 |
| Multivariate-adjusted OR | 0.81 | 0.24, 2.66 | 0.61 | 0.23, 1.65 | 0.94 | 0.30, 2.90 | 1.00 | Reference | 0.85 | 0.25, 2.93 | 0.58 |
Abbreviations: CI, confidence interval; OR, odds ratio.
All P values were 2-sided.
Conditional logistic regression models with adjustment for years of smoking and serum α-tocopherol, β-carotene, and retinol concentrations.
November–April.
May–October.
Based on season-specific quartiles, the risk of colon cancer was significantly elevated for higher 25(OH)D concentrations (Ptrend = 0.01; Table 3), with a similar pattern being observed for colorectal cancer (Ptrend = 0.02; data not shown). Elevated colon cancer risk was also noted for the higher quartiles in the season-standardized models (Table 3), although the colorectal cancer association was weaker (Ptrend = 0.12; data not shown). These risk patterns for colon cancer were similar for subjects with blood drawn during the darker months and subjects with blood drawn during the sunnier months, but only in the darker months were the associations statistically significant (in the season-specific and season-standardized models, odds ratios for the highest quartile versus the lowest were 2.26 (95% confidence interval (CI): 1.12, 4.57; Ptrend = 0.03) and 2.10 (95% CI: 1.08, 4.10; Ptrend = 0.08), respectively). Threshold relations were suggested whereby subjects in the lowest quartiles had the lowest colon cancer risk. The rectal cancer association was generally null in the season-specific, season-standardized, and season-stratified (not shown) models, although the odds ratios for the second quartile versus the first quartile were significantly reduced (Table 3).
Table 3.
Odds Ratios for the Association Between Serum 25-Hydroxyvitamin D and Risks of Colon and Rectal Cancer Obtained Using Season-Specific and Season-Standardized (Residuals) Quartiles, Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study, Finland, 1985–2005
| Quartile of 25-Hydroxyvitamin D Concentration |
Ptrenda | ||||||||
| 1 |
2 |
3 |
4 |
||||||
| OR | 95% CI | OR | 95% CI | OR | 95% CI | OR | 95% CI | ||
| Colon cancer | |||||||||
| Season-specific modelb | |||||||||
| No. of cases/no. of controls | 49/72 | 57/50 | 65/58 | 68/59 | |||||
| Multivariate-adjusted ORc | 1.00 | Reference | 1.97 | 1.13, 3.45 | 1.96 | 1.14, 3.36 | 2.11 | 1.20, 3.69 | 0.01 |
| Season-standardized modeld | |||||||||
| No. of cases/no. of controls | 54/71 | 61/51 | 54/55 | 70/62 | |||||
| Multivariate-adjusted OR | 1.00 | Reference | 1.77 | 1.01, 3.08 | 1.39 | 0.81, 2.40 | 1.88 | 1.07, 3.28 | 0.06 |
| Rectal cancer | |||||||||
| Season-specific model | |||||||||
| No. of cases/no. of controls | 48/37 | 39/58 | 48/50 | 57/47 | |||||
| Multivariate-adjusted OR | 1.00 | Reference | 0.46 | 0.24, 0.88 | 0.78 | 0.41, 1.51 | 1.18 | 0.62, 2.26 | 0.31 |
| Season-standardized model | |||||||||
| No. of cases/no. of controls | 51/37 | 35/57 | 49/53 | 57/45 | |||||
| Multivariate-adjusted OR | 1.00 | Reference | 0.41 | 0.21, 0.77 | 0.69 | 0.37, 1.28 | 1.13 | 0.61, 2.07 | 0.57 |
Abbreviations: CI, confidence interval; OR, odds ratio; Q, quartile.
All P values were 2-sided.
Cutpoints for season-specific quartiles: darker months (November–April)—Q1, ≤18.3; Q2, >18.3 and ≤26.9; Q3, >26.9 and ≤42.0; Q4, >42.0 nmol/L; sunnier months (May–October)—Q1, ≤27.0; Q2, >27.0 and ≤38.7; Q3, >38.7 and ≤53.4; Q4, >53.4 nmol/L.
Conditional logistic regression models with adjustment for years of smoking and serum α-tocopherol, β-carotene, and retinol concentrations.
Cutpoints for season-standardized quartiles: Q1, ≤3.10; Q2, >3.10 and ≤3.47; Q3, >3.47 and ≤3.85; Q4, >3.85. Season-standardized quartiles were created by calculating residuals from the regression line of log-transformed 25-hydroxyvitamin D concentration against calendar week of blood collection.
Exploratory stratum-specific subgroup analyses for colon cancer indicated greater elevated risk for the highest season-specific quartile versus the lowest among older men (i.e., age >57 years; odds ratio (OR) = 2.13, 95% CI: 1.03, 4.39) and subjects in the α-tocopherol supplementation arm (OR = 2.67, 95% CI: 1.21, 5.88). For rectal cancer, risk appeared greater for the highest quartile among men with higher serum β-carotene concentrations (≥185 μg/L; OR = 1.74, 95% CI: 0.69, 4.37) and those in the β-carotene supplementation arm (OR = 1.66, 95% CI: 0.68, 4.04). None of the tests for interaction by stratum were statistically significant, however. Risk of rectal cancer (but not colon cancer) appeared to differ on the basis of follow-up year of case diagnosis (highest quartile vs. lowest: for cases diagnosed through November 1999, OR = 2.24, 95% CI: 0.75, 6.67; for cases diagnosed after November 1999, OR = 0.59, 95% CI: 0.23, 1.52). However, this interaction was not statistically significant (P = 0.13).
DISCUSSION
Neither colon cancer risk nor rectal cancer risk was inversely associated with circulating 25(OH)D concentrations. In fact, most odds ratios for categories below the referent level (50–<75 nmol/L) suggested lower risk. Results from analyses using season-specific and seasonally adjusted 25(OH)D cutpoints yielded similar patterns, with significantly elevated risks of colon cancer in the higher quartiles, although rectal cancer risk was significantly reduced in the second quartile versus the first quartile. Across all models, risk patterns did not differ consistently by season of blood collection.
Vitamin D plays a role in calcium homeostasis and regulation of bone mineralization by enhancing the absorption of calcium in the kidney and intestine (15), and higher calcium intake has been associated with reduced colorectal cancer risk (5). Besides its impact on calcium metabolism, higher vitamin D status may protect against cancer by reducing cellular proliferation and angiogenesis or inducing differentiation and apoptosis (15). Vitamin D could also act locally in the colon to inhibit carcinogenesis, since both 1-α-vitamin D hydroxylase, the enzyme that metabolizes 25(OH)D to 1,25(OH)2D, and the vitamin D receptor, which binds the active 1,25(OH)2D molecule, are expressed in the colon and elsewhere (15), and direct binding of harmful secondary bile acids by vitamin D receptor and activation of detoxifying enzymes has been shown (32). However, our findings are not consistent with these hypotheses of a protective association for vitamin D.
Clinical trials of vitamin D supplementation and colorectal cancer have been limited, and results do not support a beneficial effect for higher exposure (14, 33, 34). For example, in the Women's Health Initiative, the hazard ratio for colorectal cancer incidence after 7 years of supplementation with vitamin D (400 IU) and calcium (1,000 mg) was 1.08 (95% CI: 0.86, 1.34); however, all participants were permitted to self-supplement, complicating the trial's interpretation (14). Research on polymorphisms in vitamin D pathway genes, especially the vitamin D receptor, is inconsistent for colorectal cancer (35), as is evidence for an association with vitamin D intake (36). In only 4 of 12 prospective studies did investigators report lower colorectal cancer risk with higher vitamin D intakes (2), and risk was decreased by only 6% in a meta-analysis of dietary vitamin D (37).
Because the substantial contribution of sun exposure to vitamin D status is not captured in dietary studies, circulating 25(OH)D is a more accurate measure of overall vitamin D exposure. Prospective studies generally indicate an association between higher 25(OH)D concentrations and reduced colorectal cancer risk (6–14, 38) or mortality (39) or improved survival (40)—findings that are also supported by a recent meta-analysis (41). However, many reported risk estimates are not statistically significant, and some differ within study by anatomic subsite. While the majority of studies have observed inverse associations for 25(OH)D, risks or trends were significantly inverse in only 4 (11–14) of 7 prospective colorectal cancer studies (7, 9–14), 2 (10, 12) of 8 colon cancer studies (6–13), and 3 (9) (men only) (11, 13) of 6 rectal cancer studies (7, 9–13). Investigators in 2 studies reported nonsignificant positive associations for colon cancer (OR = 2.1, P-trend = 0.12) (9) and rectal cancer (OR = 3.32, P-trend = 0.08) (10). By far the largest study was the European Prospective Investigation into Cancer and Nutrition (EPIC), with 785 colon cancer cases and 463 rectal cancer cases; however, even with such large numbers, the inverse association for rectal cancer failed to reach statistical significance (12). With regard to subsite differences, inverse associations were significant for colon cancer only in EPIC (12) and the Health Professionals Follow-up Study (10) and, for rectal cancer, only in the Multiethnic Cohort Study (11) and a Japanese cohort study (9). Our findings indicate a significant positive association for colon cancer only. Interestingly, the 2 studies with significant inverse findings for rectal cancer either included a large percentage of Japanese Americans (11) or consisted entirely of Japanese participants (9). Other than the Multiethnic Cohort Study, which also included participants from Latino, African-American, and white populations but showed no evidence of heterogeneity among the racial/ethnic groups (11), data on minority groups are lacking.
Blood concentrations of 25(OH)D vary substantially by season (42), and we noted such a pattern, with the highest 25(OH)D concentrations being observed in the summer and fall. We used a multifaceted approach to adjust for this variation in our analyses, including tightly matching controls to cases on the date of blood collection (within 30 days), conducting season-stratified analyses, and using season-specific cutpoints, to decrease the influence of date of blood collection on quantile placement. In a simulation study, the association between 25(OH)D and disease risk was biased toward the null when seasonal variation was ignored but biased away from the null when results were adjusted for season of blood collection, but quantiles were not based on season-specific cutpoints (43). We also calculated season-standardized 25(OH)D values as residuals from a regression on date of blood collection. Our findings were generally similar across all approaches, although the positive association for colon cancer was statistically significant only in the season-specific and season-standardized models, which may have more precisely adjusted for season-related sun exposure. All but 1 (12) of the previous prospective serologic studies matched subjects on the date of blood collection, but only 4 included additional adjustment for seasonal variation in 25(OH)D (9–12): EPIC adjusted for season of blood collection and used season-standardized values (12), 1 study tested season-specific cutpoints (11), and 2 studies conducted season-stratified analyses (9, 10). In the Health Professionals Follow-up Study, an inverse relation between 25(OH)D and colorectal cancer was observed based only on blood collected during the winter months (10).
In contrast to the present findings, an early analysis within the ATBC Study suggested an inverse association, particularly for 25(OH)D and rectal cancer, although risk estimates were based on only 146 cases and up to 8 years of follow-up and were not statistically significant (7). The earlier study used a radioimmunoassay to measure 25(OH)D, but the coefficients of variation and the average 25(OH)D concentrations were similar to those presented here, and applying similarly derived cutpoints from the earlier analysis to the present data yielded risk patterns consistent with the current findings. For example, odds ratios for quartiles 1–4 were 1.0 (reference), 1.41, 1.86, and 1.71 (95% CI for quartile 4: 0.95, 3.07) for colon cancer and 1.0, 0.58, 1.02, and 1.41 (95% CI for quartile 4: 0.72, 2.76) for rectal cancer. Given the shorter observation period, reverse causality could have influenced the previous results to some degree; however, exclusion of cases diagnosed within 2 years of blood collection did not attenuate those risks, and we found no evidence of reduced risks with higher 25(OH)D for subjects in the present analysis with shorter follow-up.
Our findings could have been due to the low vitamin D status of the study population, which was a consequence of the high latitude's limiting synthesis of vitamin D in skin, especially during the winter (44), very few blood collections in the summer months, and low use of supplemental vitamin D. However, 2 other studies that observed a nonsignificantly increased risk with higher 25(OH)D for colon (9) and rectal (10) cancer had higher 25(OH)D concentrations than were found in ATBC. Other studies have reported increased risk of pancreatic (30, 45, 46) and upper gastrointestinal (47, 48) cancer with higher 25(OH)D concentrations in ATBC and other populations. The risk patterns in the current ATBC colorectal analysis differed from those in the EPIC analysis of Europeans (12), which used similar a priori defined cutpoints and the same referent category as our analysis. More than half of the EPIC participants were current or former smokers, and while adjustment for smoking status did not appear to alter the risk estimates, results stratified on smoking status were not presented in the published article (12).
The 25(OH)D biomarker integrates vitamin D exposure from diet, supplements, and sunlight, with an approximately 2- to 3-week half-life (2). The prospective evaluation of 25(OH)D 8–20 years after baseline minimized any effect of cancer on vitamin D status, but the long follow-up could be a limitation if exposures changed over time. Differences in the length of follow-up among studies do not appear to explain inconsistencies in the results found across studies. As with most prospective studies, we relied on a single measurement of 25(OH)D to represent long-term vitamin D exposure; however, correlations of 0.52–0.70 have been reported for 25(OH)D measured in samples collected 3–14 years apart (49–51). Another limitation, due to the design of the parent study, was that the analysis included only male smokers, leaving open the possibility that the vitamin D-colorectal cancer relation is somehow altered among smokers and that our findings may not be generalizable to nonsmoking populations or to women. Some investigators have reported gender differences in the vitamin D-colorectal cancer association (9, 10), while others have not (11, 12). Whether an interaction with smoking exists has not been adequately addressed, and future researchers should examine this issue, especially given that approximately 20% of the US population currently smokes (52). Few cohort studies will have the statistical power to prospectively examine vitamin D-cancer associations among smokers; however, planned pooled analyses should be able to address this issue.
In summary, high 25(OH)D concentrations were not associated with lower risk of colorectal cancer in this prospective study. In fact, our data suggested greater colon cancer risk in men with higher circulating 25(OH)D concentrations. This could have resulted from the very long period of observation, the fact that the cohort consisted solely of smokers, or the low overall vitamin D status of this population, although insufficiently high levels might be expected to yield a null finding. In future studies, researchers should specifically examine the association by smoking status, sex, and race/ethnicity and should employ rigorous methods to adjust and control for seasonal variation in circulating vitamin D concentrations.
Acknowledgments
Author affiliations: Division of Cancer Epidemiology and Genetics, National Cancer Institute, Bethesda, Maryland (Stephanie J. Weinstein, Kai Yu, Demetrius Albanes); Heartland Assays, Inc., Ames, Iowa (Ronald L. Horst); Information Management Services, Inc., Silver Spring, Maryland (Jason Ashby); and Department of Chronic Disease Prevention, National Institute for Health and Welfare, Helsinki, Finland (Jarmo Virtamo).
This work was supported in part by the Intramural Research Program of the US National Institutes of Health and the National Cancer Institute. Additionally, this research was supported by US Public Health Service contracts N01-CN-45165, N01-RC-45035, N01-RC-37004, and HHSN261201000006C from the National Cancer Institute.
The authors thank Dr. Karen Phinney of the US National Institute of Standards and Technology for providing the Vitamin D in Human Serum (SRM 972) used as quality control material.
Dr. Ronald Horst is the President and Chief Executive Officer of Heartland Assays, Inc.
Conflict of interest: none declared.
Glossary
Abbreviations
- ATBC
Alpha-Tocopherol, Beta-Carotene Cancer Prevention
- CI
confidence interval
- EPIC
European Prospective Investigation into Cancer and Nutrition
- 1,25(OH)2D
1,25-dihydroxyvitamin D
- 25(OH)D
25-hydroxyvitamin D
- OR
odds ratio
References
- 1.Garland CF, Garland FC. Do sunlight and vitamin D reduce the likelihood of colon cancer? Int J Epidemiol. 1980;9(3):227–231. doi: 10.1093/ije/9.3.227. [DOI] [PubMed] [Google Scholar]
- 2.International Agency for Research on Cancer. Vitamin D and Cancer. Lyon, France: International Agency for Research on Cancer; 2009. [Google Scholar]
- 3.van der Rhee H, Coebergh JW, Vries ED. Sunlight, vitamin D and the prevention of cancer: a systematic review of epidemiological studies. Eur J Cancer Prev. 2009;18(6):458–475. doi: 10.1097/CEJ.0b013e32832f9bb1. [DOI] [PubMed] [Google Scholar]
- 4.Giovannucci E. Epidemiological evidence for vitamin D and colorectal cancer. J Bone Miner Res. 2007;22(suppl 2):V81–V85. doi: 10.1359/jbmr.07s206. [DOI] [PubMed] [Google Scholar]
- 5.Chung M, Balk EM, Brendel M, et al. Vitamin D and Calcium: A Systematic Review of Health Outcomes. (Evidence report no. 183) Rockville, MD: Agency for Healthcare Research and Quality; 2009. (AHRQ publication no. 09-E015) [Google Scholar]
- 6.Braun MM, Helzlsouer KJ, Hollis BW, et al. Colon cancer and serum vitamin D metabolite levels 10–17 years prior to diagnosis. Am J Epidemiol. 1995;142(6):608–611. doi: 10.1093/oxfordjournals.aje.a117682. [DOI] [PubMed] [Google Scholar]
- 7.Tangrea J, Helzlsouer K, Pietinen P, et al. Serum levels of vitamin D metabolites and the subsequent risk of colon and rectal cancer in Finnish men. Cancer Causes Control. 1997;8(4):615–625. doi: 10.1023/a:1018450531136. [DOI] [PubMed] [Google Scholar]
- 8.Garland CF, Comstock GW, Garland FC, et al. Serum 25-hydroxyvitamin D and colon cancer: eight-year prospective study. Lancet. 1989;2(8673):1176–1178. doi: 10.1016/s0140-6736(89)91789-3. [DOI] [PubMed] [Google Scholar]
- 9.Otani T, Iwasaki M, Sasazuki S, et al. Plasma vitamin D and risk of colorectal cancer: the Japan Public Health Center-Based Prospective Study. Br J Cancer. 2007;97(3):446–451. doi: 10.1038/sj.bjc.6603892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wu K, Feskanich D, Fuchs CS, et al. A nested case-control study of plasma 25-hydroxyvitamin D concentrations and risk of colorectal cancer. J Natl Cancer Inst. 2007;99(14):1120–1129. doi: 10.1093/jnci/djm038. [DOI] [PubMed] [Google Scholar]
- 11.Woolcott CG, Wilkens LR, Nomura AM, et al. Plasma 25-hydroxyvitamin D levels and the risk of colorectal cancer: the Multiethnic Cohort Study. Cancer Epidemiol Biomarkers Prev. 2010;19(1):130–134. doi: 10.1158/1055-9965.EPI-09-0475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Jenab M, Bueno-de-Mesquita HB, Ferrari P, et al. Association between pre-diagnostic circulating vitamin D concentration and risk of colorectal cancer in European populations: a nested case-control study. BMJ. 2010;340 doi: 10.1136/bmj.b5500. b5500. (doi: 10.1136/bmj.b5500) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Feskanich D, Ma J, Fuchs CS, et al. Plasma vitamin D metabolites and risk of colorectal cancer in women. Cancer Epidemiol Biomarkers Prev. 2004;13(9):1502–1508. [PubMed] [Google Scholar]
- 14.Wactawski-Wende J, Kotchen JM, Anderson GL, et al. Calcium plus vitamin D supplementation and the risk of colorectal cancer. N Engl J Med. 2006;354(7):684–696. doi: 10.1056/NEJMoa055222. [DOI] [PubMed] [Google Scholar]
- 15.Holick MF. Vitamin D deficiency. N Engl J Med. 2007;357(3):266–281. doi: 10.1056/NEJMra070553. [DOI] [PubMed] [Google Scholar]
- 16.Holick MF. Vitamin D status: measurement, interpretation, and clinical application. Ann Epidemiol. 2009;19(2):73–78. doi: 10.1016/j.annepidem.2007.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.The ATBC Cancer Prevention Study Group. The Alpha-Tocopherol, Beta-Carotene Lung Cancer Prevention Study: design, methods, participant characteristics, and compliance. Ann Epidemiol. 1994;4(1):1–10. doi: 10.1016/1047-2797(94)90036-1. [DOI] [PubMed] [Google Scholar]
- 18.Korhonen P, Malila N, Pukkala E, et al. The Finnish Cancer Registry as follow-up source of a large trial cohort—accuracy and delay. Acta Oncol. 2002;41(4):381–388. doi: 10.1080/028418602760169442. [DOI] [PubMed] [Google Scholar]
- 19.Wagner D, Hanwell HE, Vieth R. An evaluation of automated methods for measurement of serum 25-hydroxyvitamin D. Clin Biochem. 2009;42(15):1549–1556. doi: 10.1016/j.clinbiochem.2009.07.013. [DOI] [PubMed] [Google Scholar]
- 20.Gallicchio L, Helzlsouer KJ, Chow WH, et al. Circulating 25-hydroxyvitamin D and the risk of rarer cancers: design and methods of the Cohort Consortium Vitamin D Pooling Project of Rarer Cancers. Am J Epidemiol. 2010;172(1):10–20. doi: 10.1093/aje/kwq116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Material Measurement Laboratory, National Institute of Standards and Technology. Certificate of Analysis. Standard Reference Material 972. Vitamin D in Human Serum. Gaithersburg, MD: National Institute of Standards and Technology; 2009. ( https://www-s.nist.gov/srmors/view_detail.cfm?srm=972). (Accessed October 14, 2010) [Google Scholar]
- 22.Fears TR, Ziegler RG, Donaldson JL, et al. Reproducibility studies and interlaboratory concordance for androgen assays in female plasma. Cancer Epidemiol Biomarkers Prev. 2000;9(4):403–412. [PubMed] [Google Scholar]
- 23.Looker AC, Dawson-Hughes B, Calvo MS, et al. Serum 25-hydroxyvitamin D status of adolescents and adults in two seasonal subpopulations from NHANES III. Bone. 2002;30(5):771–777. doi: 10.1016/s8756-3282(02)00692-0. [DOI] [PubMed] [Google Scholar]
- 24.Looker AC, Pfeiffer CM, Lacher DA, et al. Serum 25-hydroxyvitamin D status of the US population: 1988–1994 compared with 2000–2004. Am J Clin Nutr. 2008;88(6):1519–1527. doi: 10.3945/ajcn.2008.26182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zeleniuch-Jacquotte A, Gallicchio L, Hartmuller V, et al. Circulating 25-hydroxyvitamin D and risk of endometrial cancer: Cohort Consortium Vitamin D Pooling Project of Rarer Cancers. Am J Epidemiol. 2010;172(1):36–46. doi: 10.1093/aje/kwq114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zheng W, Danforth KN, Tworoger SS, et al. Circulating 25-hydroxyvitamin D and risk of epithelial ovarian cancer: Cohort Consortium Vitamin D Pooling Project of Rarer Cancers. Am J Epidemiol. 2010;172(1):70–80. doi: 10.1093/aje/kwq118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Abnet CC, Chen Y, Chow WH, et al. Circulating 25-hydroxyvitamin D and risk of esophageal and gastric cancer: Cohort Consortium Vitamin D Pooling Project of Rarer Cancers. Am J Epidemiol. 2010;172(1):94–106. doi: 10.1093/aje/kwq121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gallicchio L, Moore LE, Stevens VL, et al. Circulating 25-hydroxyvitamin D and risk of kidney cancer: Cohort Consortium Vitamin D Pooling Project of Rarer Cancers. Am J Epidemiol. 2010;172(1):47–57. doi: 10.1093/aje/kwq115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Purdue MP, Freedman DM, Gapstur SM, et al. Circulating 25-hydroxyvitamin D and risk of non-Hodgkin lymphoma: Cohort Consortium Vitamin D Pooling Project of Rarer Cancers. Am J Epidemiol. 2010;172(1):58–69. doi: 10.1093/aje/kwq117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Stolzenberg-Solomon RZ, Jacobs EJ, Arslan AA, et al. Circulating 25-hydroxyvitamin D and risk of pancreatic cancer: Cohort Consortium Vitamin D Pooling Project of Rarer Cancers. Am J Epidemiol. 2010;172(1):81–93. doi: 10.1093/aje/kwq120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Borkowf CB, Albert PS, Abnet CC. Using lowess to remove systematic trends over time in predictor variables prior to logistic regression with quantile categories. Stat Med. 2003;22(9):1477–1493. doi: 10.1002/sim.1507. [DOI] [PubMed] [Google Scholar]
- 32.Makishima M, Lu TT, Xie W, et al. Vitamin D receptor as an intestinal bile acid sensor. Science. 2002;296(5571):1313–1316. doi: 10.1126/science.1070477. [DOI] [PubMed] [Google Scholar]
- 33.Trivedi DP, Doll R, Khaw KT. Effect of four monthly oral vitamin D3 (cholecalciferol) supplementation on fractures and mortality in men and women living in the community: randomised double blind controlled trial. BMJ. 2003;326(7387):469. doi: 10.1136/bmj.326.7387.469. (doi: 10.1136/bmj.326.7387.469) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lappe JM, Travers-Gustafson D, Davies KM, et al. Vitamin D and calcium supplementation reduces cancer risk: results of a randomized trial. Am J Clin Nutr. 2007;85(6):1586–1591. doi: 10.1093/ajcn/85.6.1586. [DOI] [PubMed] [Google Scholar]
- 35.McCullough ML, Bostick RM, Mayo TL. Vitamin D gene pathway polymorphisms and risk of colorectal, breast, and prostate cancer. Annu Rev Nutr. 2009;29:111–132. doi: 10.1146/annurev-nutr-080508-141248. [DOI] [PubMed] [Google Scholar]
- 36.World Cancer Research Fund, American Institute for Cancer Research. Food, Nutrition, Physical Activity, and the Prevention of Cancer: A Global Perspective. Washington, DC: American Institute for Cancer Research; 2007. [Google Scholar]
- 37.Huncharek M, Muscat J, Kupelnick B. Colorectal cancer risk and dietary intake of calcium, vitamin D, and dairy products: a meta-analysis of 26,335 cases from 60 observational studies. Nutr Cancer. 2009;61(1):47–69. doi: 10.1080/01635580802395733. [DOI] [PubMed] [Google Scholar]
- 38.Gorham ED, Garland CF, Garland FC, et al. Optimal vitamin D status for colorectal cancer prevention: a quantitative meta analysis. Am J Prev Med. 2007;32(3):210–216. doi: 10.1016/j.amepre.2006.11.004. [DOI] [PubMed] [Google Scholar]
- 39.Freedman DM, Looker AC, Chang SC, et al. Prospective study of serum vitamin D and cancer mortality in the United States. J Natl Cancer Inst. 2007;99(21):1594–1602. doi: 10.1093/jnci/djm204. [DOI] [PubMed] [Google Scholar]
- 40.Ng K, Meyerhardt JA, Wu K, et al. Circulating 25-hydroxyvitamin D levels and survival in patients with colorectal cancer. J Clin Oncol. 2008;26(18):2984–2991. doi: 10.1200/JCO.2007.15.1027. [DOI] [PubMed] [Google Scholar]
- 41.Gandini S, Boniol M, Haukka J, et al. Meta-analysis of observational studies of serum 25-hydroxyvitamin D levels and colorectal, breast and prostate cancer and colorectal adenoma [published online ahead of print May 6, 2010] Int J Cancer. doi: 10.1002/ijc.25439. (doi: 10.1002/ijc.25439) [DOI] [PubMed] [Google Scholar]
- 42.Nakamura K, Nashimoto M, Yamamoto M. Summer/winter differences in the serum 25-hydroxyvitamin D3 and parathyroid hormone levels of Japanese women. Int J Biometeorol. 2000;44(4):186–189. doi: 10.1007/s004840000067. [DOI] [PubMed] [Google Scholar]
- 43.Wang Y, Jacobs EJ, McCullough ML, et al. Comparing methods for accounting for seasonal variability in a biomarker when only a single sample is available: insights from simulations based on serum 25-hydroxyvitamin D. Am J Epidemiol. 2009;170(1):88–94. doi: 10.1093/aje/kwp086. [DOI] [PubMed] [Google Scholar]
- 44.Holick MF. Vitamin D: importance in the prevention of cancers, type 1 diabetes, heart disease, and osteoporosis. Am J Clin Nutr. 2004;79(3):362–371. doi: 10.1093/ajcn/79.3.362. [DOI] [PubMed] [Google Scholar]
- 45.Stolzenberg-Solomon RZ, Hayes RB, Horst RL, et al. Serum vitamin D and risk of pancreatic cancer in the Prostate, Lung, Colorectal, and Ovarian Screening Trial. Cancer Res. 2009;69(4):1439–1447. doi: 10.1158/0008-5472.CAN-08-2694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Stolzenberg-Solomon RZ, Vieth R, Azad A, et al. A prospective nested case-control study of vitamin D status and pancreatic cancer risk in male smokers. Cancer Res. 2006;66(20):10213–10219. doi: 10.1158/0008-5472.CAN-06-1876. [DOI] [PubMed] [Google Scholar]
- 47.Chen W, Dawsey SM, Qiao YL, et al. Prospective study of serum 25(OH)-vitamin D concentration and risk of oesophageal and gastric cancers. Br J Cancer. 2007;97(1):123–128. doi: 10.1038/sj.bjc.6603834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Abnet CC, Chen W, Dawsey SM, et al. Serum 25(OH)-vitamin D concentration and risk of esophageal squamous dysplasia. Cancer Epidemiol Biomarkers Prev. 2007;16(9):1889–1893. doi: 10.1158/1055-9965.EPI-07-0461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Platz EA, Leitzmann MF, Hollis BW, et al. Plasma 1,25-dihydroxy- and 25-hydroxyvitamin D and subsequent risk of prostate cancer. Cancer Causes Control. 2004;15(3):255–265. doi: 10.1023/B:CACO.0000024245.24880.8a. [DOI] [PubMed] [Google Scholar]
- 50.Hofmann JN, Yu K, Horst RL, et al. Long-term variation in serum 25-hydroxyvitamin D concentration among participants in the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial. Cancer Epidemiol Biomarkers Prev. 2010;19(4):927–931. doi: 10.1158/1055-9965.EPI-09-1121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Jorde R, Sneve M, Hutchinson M, et al. Tracking of serum 25-hydroxyvitamin D levels during 14 years in a population-based study and during 12 months in an intervention study. Am J Epidemiol. 2010;171(8):903–908. doi: 10.1093/aje/kwq005. [DOI] [PubMed] [Google Scholar]
- 52.Cigarette smoking among adults—United States, 2007. MMWR Morb Mortal Wkly Rep. 2008;57(45):1221–1226. [PubMed] [Google Scholar]