Abstract
The thalamus may be the critical brain area involved in sensory gating and the relay of respiratory mechanical information to the cerebral cortex for the conscious awareness of breathing. We hypothesized that respiratory mechanical stimuli in the form of tracheal occlusions would modulate the gene expression profile of the thalamus. Specifically, it was reasoned that conditioning to the respiratory loading would induce a state change in the medial thalamus consistent with a change in sensory gating and the activation of molecular pathways associated with learning and memory. In addition, respiratory loading is stressful and thus should elicit changes in gene expressions related to stress, anxiety, and depression. Rats were instrumented with inflatable tracheal cuffs. Following surgical recovery, they underwent 10 days (5 days/week) of transient tracheal occlusion conditioning. On day 10, the animals were sacrificed and the brains removed. The medial thalamus was dissected and microarray analysis of gene expression performed. Tracheal obstruction conditioning modulated a total of 661 genes (p < 0.05, log2 fold change ≥0.58), 250 genes were down-regulated and 411 up-regulated. There was a significant down-regulation of GAD1, GAD2 and HTR1A, HTR2A genes. CCK, PRKCG, mGluR4, and KCJN9 genes were significantly up-regulated. Some of these genes have been associated with anxiety and depression, while others have been shown to play a role in switching between tonic and burst firing modes in the thalamus and thus may be involved in gating of the respiratory stimuli. Furthermore, gene ontology and pathway analysis showed a significant modulation of learning and memory pathways. These results support the hypothesis that the medial thalamus is involved in the respiratory sensory neural pathway due to the state change of its gene expression profile following repeated tracheal occlusions.
Keywords: respiratory load, control of breathing, load compensation, stress
Introduction
The sensation of dyspnea, or breathlessness, is a prevalent symptom in numerous pulmonary and cardiovascular diseases. This conscious awareness of breathing requires the activation of higher brain centers. The neural control pathway to the higher centers is thought to be a gated process. Gating of incoming sensory information is a way to control what and how much information will be received by higher brain centers. Gating is thought to be a protective mechanism for humans and animals to prevent the conscious perception of unnecessary stimuli and instead attend only to the meaningful ones. One of the proposed brain areas functioning as a gate for respiratory stimuli activating the cerebral cortex is the thalamus (Chan and Davenport, 2008).
Eupneic breathing is usually not consciously perceived, meaning that respiratory afferents during normal breathing are gated out and do not reach higher brain centers. However, if ventilation changes sufficiently or breathing is attended to, the sensation is gated in and the animal becomes aware of its breathing (Chan and Davenport, 2008). This awareness is usually associated with distressing emotion (O’Donnell et al., 2007). The thalamus may be involved in respiratory gating based on evidence from several studies. Chen et al. (1992) showed that when respiratory drive was stimulated as measured by increased phrenic nerve activity, previously tonically active thalamic single units switched to rhythmic increases in firing that was associated with each respiration. Retrograde tracing experiments in cats indicated that phrenic afferents activate thalamocortical projections (Yates et al., 1994). Also, Zhang and Davenport (2003) showed that inspiratory occlusions activated thalamic neurons in cats and rats. Positron emission tomography (PET) studies in humans exposed to hypercapnia identified neuronal activation extending from the upper brainstem, up through the midbrain, hypothalamus, and thalamus (Corfield et al., 1995). Other PET and functional magnetic resonance imaging studies in humans have shown that voluntary hyperpnea, or the behavioral modulation of breathing, activates distinct cortical (primary sensorimotor cortices, supplementary motor, and premotor cortex) as well as subcortical (thalamus, globus pallidum, caudate, and cerebellum) structures (McKay et al., 2003). Previous studies using c-Fos, a marker for neuronal activity, have shown that intrinsic, transient tracheal obstruction (ITTO) in anesthetized animals activate neurons in the cerebral cortex as well as in the medial thalamus (Vovk et al., 2006). In addition, ITTO modulated the gene expression profile of the medial thalamus in anesthetized rats (Bernhardt et al., 2008), supporting the role of the medial thalamus as an integral component in the respiratory cortical neural pathway.
Respiratory information relayed through the thalamus reaches cortical areas for recognition and discrimination, as well as the limbic system for emotional processing (Davenport and Vovk, 2009). It is the interplay between these brain areas that are responsible for the generation of the perception of breathlessness. This feeling of breathlessness is an aversive sensation. Animals and humans alike modify their behavior to avoid feeling breathless. While it is known that repeated exposure to ITTO in a conscious state changes load compensation behavior (Pate et al., 2010), it is unknown if neuron plasticity in the medial thalamus is induced by conscious chronic exposure to ITTO. We hypothesized that repeated exposure to ITTO in a conscious animal would modulate the gene expression pattern of the medial thalamus. Repeated loading of the respiratory system via ITTO could change the state of thalamic gating, such that an increase in sensory threshold would result in less gating-in of aversive respiratory feedback.
Previously, we observed that, with ITTO in anesthetized rats, the serotonin receptor HTR2A was up-regulated (log2 fold change >1, p < 0.05; Bernhardt et al., 2008). Serotonin, as well as its receptors and transporter have been implicated in the stress response, anxiety, and depression (Graeff et al., 1996; Weisstaub et al., 2006; Heisler et al., 2007; Harada et al., 2008). Chronic exposure to tracheal occlusions in conscious rats is stressful and may show greater modulation of serotonin and/or its targets in the thalamus. In the present study, it was hypothesized that 10 days of exposure to 10 min trials of ITTO in chronically instrumented, conscious rats would induce state gene expression changes in the medial thalamus; specifically, up-regulation of genes that are associated with sensory gating and genes that have been implicated in the response to stress, anxiety, and/or depression.
Materials and Methods
Animals
Eight male Sprague-Dawley rats (299 ± 43.05 g, Harlan Labs) were housed two per cage in a temperature-controlled room (72°F) on a 12:12 light:dark cycle, and with free access to food and water. All animal experiments were approved by the Institutional Animal Care and Use Committee of the University of Florida. Rats were divided into an experimental group (n = 4) and a control group (n = 4). All rats underwent surgery for placement of the tracheal occluder.
Placement of tracheal occluder
Rats were anesthetized using inhaled isoflurane gas (2–5% in O2). Buprenorphine (0.01–0.05 mg/kg BW) and carprofen (5 mg/kg BW) were administered preoperatively via subcutaneous injection. The eyes were coated with petroleum ointment to prevent drying. Incision sites were shaved and sterilized with povidone–iodine topical antiseptic solution. The trachea was exposed ventrally in the neck via a skin incision and blunt dissection of surrounding connective tissues. An expandable cuff was sutured around the trachea, two cartilage rings caudal to the larynx. The actuating tube was routed subcutaneously and externalized, between the scapulae. The tube was anchored to the skin using the closing sutures. The neck incision was then closed using an interrupted suture pattern.
Analgesia and postoperative care
Preoperative analgesia consisted of buprenorphine (0.01–0.05 mg/kg BW) and carprofen (5 mg/kg BW) administered via subcutaneous injection. Following surgical instrumentation rats were administered warm normal saline (0.01–0.02 ml/g BW) to ensure proper hydration. Postoperative analgesia was provided for at least 3 days using buprenorphine (0.01–0.05 mg/kg BW given every 12–24 h) and carprofen (5 mg/kg BW given every 24 h). Rats were closely observed for any signs of distress or pain.
Experimental protocol
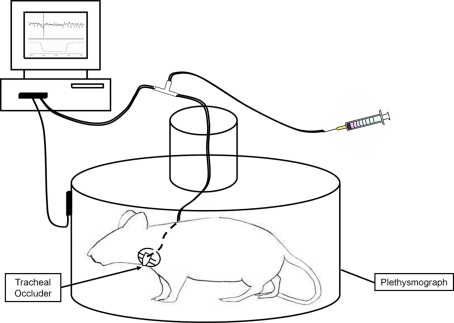
Rats were placed in a plethysmograph and the externalized occlude actuator was connected to a saline-filled syringe (Figure 1). Data collection for the control group consisted of 15 min of recording with no experimental manipulation. For the experimental group the syringe was used to inflate and deflate the cuff bladder. Inflation of the cuff compressed the trachea completely, occluding the airway during both inspiration and expiration. Deflation restored the trachea back to its original condition to allow unobstructed breathing. The experimental group received a 2.5-min background control recording period, followed by a 10-min experimental session of ITTO, and ended with a 2.5-min post-test control period. During the experimental session the rats underwent repeated trials of occlusions lasting for 3–10 breaths (duration of occlusion approximately 2.5–10 s) followed by approximately 30 s of recovery (deflation of cuff), so that the trachea was occluded at least 20 times within the 10-min trials (Figure 2). The control group animals were surgically prepared, handled and placed in the plethysmograph for the same duration as the experimental group except the control animals were not exposed to the ITTO. Experimental and control sessions were performed for 10 days (2 weeks of 5 days/week) at approximately the same time of day. On day 10, the rats were sacrificed via overdose of anesthetic. The medial thalamus was quickly excised, frozen in liquid nitrogen, and stored at −80°C until further use.
Figure 1.
Schematic of the experimental preparation for repeated ITTO conditioning in conscious rats. Rats were placed in a plethysmograph and the actuator tube of the tracheal cuff was connected to a saline-filled syringe.
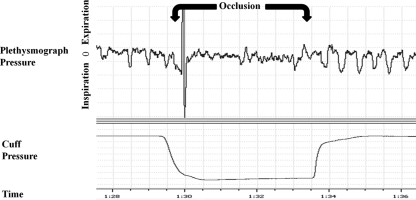
Figure 2.
Representative plethysmograph pressure traces for one occlusion trial on day 10. The period of occlusion is indicated on the plethysmograph pressure. The large deflection of the signal at the beginning of occlusion is ascribed to a movement artifact due to the rat’s withdrawal reflex at the onset of occlusion.
Total RNA isolation
Total RNA was isolated from medial thalamic tissue with RNA Stat-60 (Tel-test, Friendswood, TX, USA). About 10–20 mg of the frozen tissue was homogenized in Stat-60 and chloroform added. The mixture was vortexed for 15 s and centrifuged at 12,000 g for 15 min at 4°C. The upper aqueous phase containing the RNA was carefully extracted. The extraction step was repeated and the RNA precipitated with isopropanol. Following another centrifugation at 12,000 g for 40 min at 4°C, the pellet was washed twice with 80% ethanol and air dried. To inactivate RNases, the pellet was resuspended in 40 μl RNA secure (Ambion, Austin, TX, USA) following the manufacturer’s protocol. A total of 10 μg of RNA was treated with DNase to avoid contaminating DNA using DNA-free (Ambion, Austin, TX, USA) following the manufacturer’s protocol. A total of 10 μg of RNA was treated with DNase to avoid contaminating DNA using DNA-free (Ambion, Austin, TX, USA) following the manufacturer’s protocol. The quality of total RNA was assessed with the Agilent 2100 BioAnalyzer (Agilent Technologies, Palo Alto, CA, USA) and the quantity was determined on a NanoDrop spectrophotometer (NanoDrop Technologies, Wilmington, DE, USA).
Microarray hybridizations and analysis
Rat whole genome microarrays (Amadid: 014879) were purchased from Agilent Technologies (Palo Alto, CA, USA). The Agilent one-color microarray hybridization protocol (One-Color Microarray-Based Gene Expression Analysis, version 5.7, Agilent, Palo Alto, CA, USA) was used for microarray hybridizations following the manufacturer’s protocol and recommendations. One microgram of total RNA was used for all hybridizations. Complementary cDNA synthesis, cRNA labeling, and hybridizations were performed following the manufacturer’s kits and protocols (QuickAmp Labeling Kit; Agilent Technologies, Palo Alto, CA, USA). An Axon GenePix®4000B Microarray Scanner (Molecular Deviced Inc., Concord, ON, Canada) was used to scan the microarrays. Data were extracted from microarray images using Agilent Feature Extraction software (Agilent Technologies, Palo Alto, CA, USA). Consistent with the minimum information about a microarray experiment (MIAME) standards (Brazma et al., 2001), text versions of the raw data from this study have been deposited at the Gene Expression Omnibus website and are accessible through GEO series accession number GSE25152 (http://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE25152).
Linear models were firstly used to assess differential expression (Smyth et al., 2005), then an empirical Bayes method was used to moderate the SE. T-test for the samples was performed on normalized log2 transformed signal ratios of each probe individually, followed by multiple testing correction using a Benjamini–Hochberg approach (Benjamini and Hochberg, 1995). Genes were considered differentially expressed if the p-value was ≤0.05 and the log2 fold change was ≥ ± 0.58, corresponding to a 1.5-fold down- or up-regulation.
Gene ontology and pathway analysis
Gene ontology (GO) annotations were derived from similarity searches of the NCBI gene database. A blastn search for each of the 44,000 probes was performed to retrieve the GO annotation. Once the GO annotations were retrieved, a GO tree was built following the hierarchical structure for the whole array. Then, another GO tree for the significant regulated genes was built. The two trees were compared at each node by running a Fisher’s exact test (p ≤ 0.05) when traversing the tree branches. Significantly over-represented GO categories were identified by the Fisher’s p-value and the false discovery rate was determined. Some of the genes that showed significant modulation were scanned against the Pathway Studio ResNet database (Ariadne Genomics, Rockville, MD, USA). This database uses published information and catalogs the relationships between biological entities. Pathway Studio® (Ariadne Genomics) was used to identify and graphically display the functional interactions between the selected genes (Nikitin et al., 2003).
Real-time polymerase chain reaction
A total of six differentially expressed genes were confirmed by real-time polymerase chain reaction (RT-PCR). The genes tested were HTR1A (serotonin receptor 1A, NM_012585), CCK (cholecystokinin, NM_012829), CRHBP (corticotropin releasing hormone binding protein, NM_139183), HTR2A (serotonin receptor 2A, NM_017254), PRKCG (protein kinase C gamma, NM_012628), and GAD1 (glutamic acid decarboxylase 1, NM_017007), using 18S (NR_003286) as a control. TaqMan Probes were obtained from Applied Biosystems (Foster City, CA, USA). A TaqMan® RNA-to-CT™ 1-Step Kit was used following the manufacturer’s protocol (Applied Biosystems, Foster City, CA, USA). Briefly, 500 ng of total RNA was added to each well and nuclease-free water up to a total volume of 8.5 μl. The master mix was prepared as recommended by the manufacturer adding TaqMan® RT-PCR Mix (2×), TaqMan® Gene expression assay, and TaqMan® RT Enzyme mix (40×) up to a volume of 20 μl. All reactions were run in a 384-well plate. The plate was briefly centrifugated and run in the ABI 7900 (Applied Biosystems, Foster City, CA, USA) using the following conditions: RT step for 15 min at 48°C, enzyme activation step for 10 min at 95°C, annealing step for 1 min at 60°C, for 40 cycles. Data was analyzed using the ΔΔCt method (Applied Biosystems, Foster City, CA, USA).
Results
Modulation of gene expression profile following ITTO
Statistical analysis of the microarray data showed that a total of 661 genes were altered (p < 0.05, log2 fold change ≥0.58) following the 10-day occlusion protocol, with 250 down-regulated and 411 up-regulated (Supplementary Material). Table 1 shows some of the significantly differentially modulated genes. These genes were chosen based on their potential role thalamic neuron functioning, as well as their implication in stress, anxiety, and depression. The glutamic acid decarboxylase subtypes 1 and 2 (GAD1 and GAD2), corticotrophin releasing hormone binding protein (CRHBP), and the serotonin receptor subtypes 1A and 2A (HTR1A and HTR2A) were found to be significantly down-regulated. Up-regulated genes included short stature homeobox 2 (SHOX2), cholecystokinin (CCK), protein kinase C (PRKCG), metabotropic glutamate receptor subtype 4 (GRM4), and a potassium inwardly rectifying channel (KCNJ9).
Table 1.
Candidate genes significantly differentially regulated following chronic ITTO.
| Log fold change | p-value | Gene symbol | Description |
|---|---|---|---|
| −2.14 | 0.0043 | GAD1 | Glutamic acid decarboxylase 1 |
| −1.61 | 0.0256 | GAD2 | Glutamic acid decarboxylase 2 |
| −1.38 | 0.0039 | CRHBP | Corticotropin releasing hormone binding protein |
| −0.78 | 0.0396 | HTR1A | Serotonin receptor 1A |
| −0.59 | 0.0023 | HTR2A | Serotonin receptor 2A |
| +1.37 | 0.0009 | SHOX2 | Short stature homeobox 2 |
| +1.36 | 0.0402 | CCK | Cholecystokinin |
| +1.22 | 0.0040 | PRKCG | Protein kinase C, gamma |
| +1.09 | 0.0085 | GRM4 | Glutamate receptor, metabotropic 4 |
| +1.07 | 0.0060 | KCNJ9 | Potassium inwardly rectifying channel |
Gene expression confirmation by RT-PCR
Six genes were confirmed by RT-PCR. Both the statistical significance and fold change direction for the six genes were confirmed by RT-PCR. Table 2 shows the results from RT-PCR compared to the microarray results.
Table 2.
RT-PCR and Microarray results comparison (p < 0.01).
| Log fold change microarray | Log fold change RT-PCR | Gene symbol | TaqMan® Assay |
|---|---|---|---|
| −2.14 | −2.25 | GAD1 | Rn00690300_m1 |
| −1.38 | −3.54 | CRHBP | Rn00594854_m1 |
| −0.78 | −2.35 | HTR1A | Rn00561409_s1 |
| −0.59 | −2.07 | HTR2A | Rn00568473_m1 |
| +1.36 | +1.14 | CCK | Rn00563215_m1 |
Gene ontology and pathway analysis
Table 3 shows GO categories for biological processes that were significantly over-represented among the regulated genes (Supplementary Material). Neurotransmitter signaling and learning and memory were differentially modulated processes.
Table 3.
Significantly modulated gene ontology biological processes.
| Name | No. of entities | Overlap | p-value |
|---|---|---|---|
| Synaptic transmission | 247 | 17 | 2.19 e−12 |
| Learning and/or memory | 42 | 5 | 1.11 e−5 |
| Neurotransmitter transport | 62 | 5 | 7.55 e−5 |
| Neurotransmitter secretion | 48 | 4 | 3.60 e−4 |
| Regulation of neuronal synaptic plasticity | 26 | 3 | 7.85 e−4 |
| Regulation of neurotransmitter secretion | 27 | 3 | 8.79 e−4 |
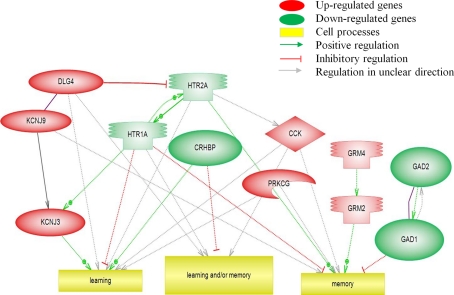
Pathway Studio was used to visualize changes of gene expression following tracheal occlusions. The balance between the activities of up- and down-regulated genes involved in these pathways determines the response. Figure 3 shows the significantly regulated genes and pathways involved in learning and/or memory. The up-regulated KCJN and GRM genes promote learning and/or memory, while the down-regulated HTR and GAD genes inhibited these pathways. Counteractive interactions between up- and down-regulated genes exist between the HTR and KCNJ pathways.
Figure 3.
Pathway analysis of transcripts (p < 0.05) involved in the biological processes of learning and/or memory.
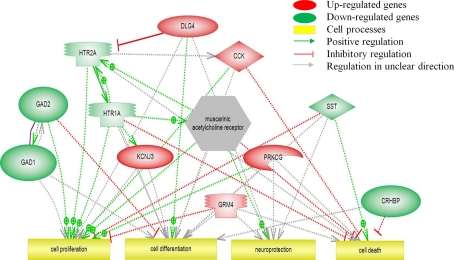
Modulated pathways for cellular processes are depicted in Figure 4. Cell proliferation is positively regulated by the down-regulated HTR and GAD genes and negatively regulated by the up-regulated GRM4, suggesting that this pathway could be less active following chronic exposure to ITTO. Cell differentiation appears to be promoted due to the positive regulation by DLH4 and the inhibition by GAD2. Several genes are involved in neuroprotection but the exact regulation is unknown. Cell death is inhibited by HTR1A and CRHBP but also by CCK, complicating a prediction on the direction of regulation on this pathway.
Figure 4.
Pathway analysis of transcripts (p < 0.05) involved in cell processes.
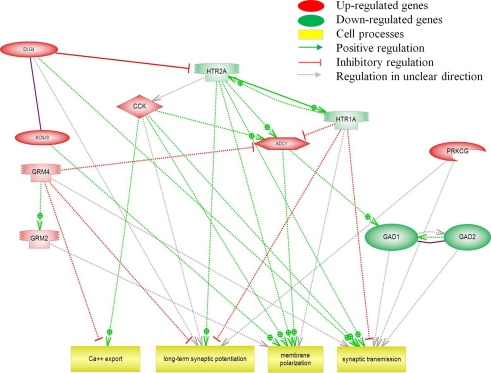
Figure 5 demonstrates the modulated pathways for cell signaling. Most up-regulated genes, except for GRM4, positively regulate calcium (Ca2+) export, membrane polarization, and synaptic transmission. The up-regulated DLG4 inhibits HTR1A, which was found to be down-regulated. HTR1A and HTR2A have opposing effects on long term synaptic potentiation, membrane polarization, and synaptic transmission.
Figure 5.
Pathway analysis of transcripts (p < 0.05) involved in cell signaling.
Discussion
The results of this study show ITTO modulation of gene expression in the medial thalamus. Thalamic gene difference between control rats and animals exposed to ITTO conditioning for 10 days had significant up-regulation of 250 and down-regulation of 411 genes in th medial thalamus. These results support out hypothesis that repeated exposure to ITTO in a conscious animal would modulate the gene expression pattern of the medial thalamus. In addition, using pathway and GO analyses, we observed a change in the stress, anxiety, and depression gene pathways and also modulation of the learning and/or memory, cellular processes, and cell signaling gene pathways. Hence we supported our hypothesis that 10 days of exposure to 10 min trials of ITTO in chronically instrumented, conscious rats would induce state gene expression changes in the medial thalamus, specifically in gene pathways mediating, stress, learned conditioned responses, and neural tissue remodeling.
Thalamic firing mode and sensory gating
Transmission in the thalamic relay neurons occurs via one of two neuronal firing modes, tonic and burst (Ramcharan et al., 2000). This response depends on the cells’ membrane potential and the activity of T- (transient) and L- (long-lasting) type Ca2+ channels (Sherman and Guillery, 2002; Cheong et al., 2008). Tonic firing occurs at relatively depolarized membrane potentials when the T-channels are inactivated, and the firing of single action potentials is dependent on suprathreshold activation. Burst firing occurs when the membrane is hyperpolarized. The T-channels are de-inactivated and the next suprathreshold depolarization activates the channels to produce an inward Ca2+ current. This results in a low-threshold, all-or-none voltage spike that is usually large enough to fire a high frequency cluster of action potentials. L-type Ca2+ channels are involved in the production of after-hyperpolarization, an important factor in determining the firing rate of neuronal cells. Specifically, Cheong et al. (2008) have shown that increased Ca2+ influx via these channels augments after-hyperpolarization, which leads to both decreased tonic-firing rates in thalamic relay neurons and increased gating of pain stimuli.
It has been proposed that switching between firing modes occurs in order to gate-out from the cerebral cortex extrinsic sensory noise (McCormick and Bal, 1994), or to assist the animal to attend to a specific stimulus (Sherman, 1996, 2001; Sherman and Guillery, 2002). During tonic firing each depolarization from a sensory stimulus produces one action potential. This linear relationship results in an accurate transmission of information through the thalamus to the cortex. Burst firing, on the other hand, is non-linear because spontaneous activity can occur in the absence of a stimulus. This high signal-to-noise ratio improves the initial detectability of a stimulus. Sherman and Guillery (2002) thus hypothesized that thalamic relay cells fire in burst mode when an important stimulus is detected, after which the cell switches to tonic mode for accurate relay of that input. A finding supporting this hypothesis is that tonic firing is increased the more alert the animal is (Ramcharan et al., 2000; Swadlow and Gusev, 2001). Furthermore, increased bursting and decreased tonic firing of thalamic neurons has been shown to reduce pain responses, suggesting that burst firing is associated with gating out of afferent sensory signals (Cheong et al., 2008).
Switching between firing modes requires a shift in membrane potential that is sufficiently sustained to inactivate or de-inactivate the T-channels. The slow metabotropic receptors are most likely responsible for the sustained voltage change. Specifically, studies have demonstrated that depolarization (inactivate T-channels) occurs through metabotropic glutamate receptors from the cortex and hyperpolarization (de-inactivate T-channels) through GABA receptors from reticular and/or interneuronal inputs (Sherman, 1996, 2001; Sherman and Guillery, 2002). Several neurotransmitters, such as serotonin, glutamate, acetylcholine, and norepinephrine, have been shown to facilitate the transition between thalamic firing modes (McCormick and Bal, 1994). In the present study, several genes were found to be significantly modulated following chronic ITTO and that could be important in the thalamic firing and thus gating activity.
KCNJ9 (or GIRK channel or Cir3 channels, potassium inwardly rectifying channel, subfamily J, member 9)
G-protein inwardly rectifying potassium (GIRK) channels mediate the synaptic actions of numerous neurotransmitters in the mammalian brain and play an important role in the regulation of neuronal excitability in most brain regions through activation of various G-protein-coupled receptors (Saenz del Burgo et al., 2008). Activation of GIRK channels causes membrane hyperpolarization, and thus the channels play an important role in the inhibitory regulation of neuronal excitability (Kobayashi et al., 2004). GIRK channels are widely expressed in brain nuclei and are co-expressed with serotonergic, GABAergic, glutamatergic, and cholinergic neurons throughout the brain (Saenz del Burgo et al., 2008). Thus, the interplay between the neurotransmitters and GIRK channels in addition to hyperpolarization could result in switching of the thalamic firing mode. GIRK2-deficient mice have been shown to exhibit reduced anxiety and elevated motor activity (Kobayashi et al., 2004); thus an up-regulation in GIRK as found in this study suggests increased anxiety which may be related to decreased sensory gating (Chan and Davenport, 2010).
GRM4 (or mGluR4, metabotropic glutamate receptor 4)
Glutamatergic neurotransmission is involved in most aspects of normal brain function and can be perturbed in many neuropathologic conditions (Niswender and Conn, 2010). l-glutamate is the major excitatory neurotransmitter in the central nervous system and activates both ionotropic and metabotropic glutamate receptors (Kew and Kemp, 2005). Grm4 belongs to group III metabotropic glutamate receptors. Agonists of group III mGluRs have been shown to exert antidepressant-like effects, possibly due to a decrease in excitatory glutamatergic neurotransmission (Klak et al., 2007). A recent study in protein lipase C-knockout mice demonstrated that switching between tonic and burst firing in thalamic neurons occurs through the simultaneous modulation of T- and L-type Ca2+ channels possibly through a transduction cascade that includes metabotropic glutamate receptors and protein kinase C (Cheong et al., 2008). These mice, which show decreased visceral pain responses, exhibited increased bursting and decreased tonic firing in thalamic neurons, suggesting that burst firing acts as an inhibitor of pain signal transmission to the cortex. Thus, the increase in GRM4 may be related to increased aversive affective responses to ITTO similar to human increased response to threat of load (Lang et al., 2010).
PRKCG (protein kinase C, gamma)
Protein kinase C (PRKC) is a family of serine- and threonine-specific protein kinases that can be activated by Ca2+ and the second messenger diacylglycerol (Huang, 1989). These molecules phosphorylate a wide variety of protein targets and are involved in diverse cellular signaling pathways. The gamma subunit of PRKC is expressed solely in neurons in the brain and spinal cord (Saito and Shirai, 2002). This specific kinase has been implicated in several neuronal functions, including long term potentiation and long term depression (Saito and Shirai, 2002). It has also been shown to associate directly with the GluR4 AMPA receptor subunit; GluR4 phosphorylation would allow for regulation of synaptic function and plasticity (Correia et al., 2003). As mentioned above, PRKC is involved in switching between burst and tonic firing. Specifically, down-regulation of PRKC activity in thalamic relay neurons has been attributed to reduced pain responses, or increased sensory gating (Cheong et al., 2008). In the present study, we found an up-regulation of PRKC, suggesting that chronic exposure to ITTO results in decreased gating in the thalamus.
CCK (cholecystokinin
Cholecystokinin is one of the most abundant neuropeptides in the brain and acts as a neurotransmitter and neuromodulator of dopamine, serotonin, endogenous opioids, GABA, and excitatory amino acids (Harro and Vasar, 1991). These characteristics support an important role in regulation of many behavioral phenomena, including anxiety (Moran and Schwartz, 1994) and learning and memory (Gulpinar and Yegen, 2004). Indeed, CCK agonists have been shown to be anxiogenic and CCK antagonists are anxiolytic in a variety of animal species (Harro et al., 1993; Rotzinger and Vaccarino, 2003). CCK has also been demonstrated to have close interaction with GABAergic inhibitory neurotransmission, mediated probably through CCK-B receptors, which could be the neurochemical substrate for anxious behavior (Harro and Vasar, 1991).Whole-cell patch clamp experiments have shown that CCK depolarizes somatosensory cortex neurons long-lastingly and thus may lead to prolonged discharge of these corticothalamic glutamatergic neurons and slow depolarization of thalamocortical neurons, shifting the firing mode from burst- to tonic-firing mode, thus being critical in sensory information processing (Chung et al., 2009). In the present study, ITTO conditioning up-regulated the CCK gene suggesting a shift in burst to tonic activity in the thalamus.
Chronic exposure to ITTO modulates genes involved in stress, anxiety, and depression
Chronic airway occlusion, as occurs in diseases such as chronic obstructive pulmonary disease, has been implicated in an increased incidence of anxiety and depression (Di Marco et al., 2006; Omachi et al., 2009). In the present study, several genes were found to be significantly altered following tracheal occlusions that play important roles in the development of depressive and other psychological disorders.
GAD1 (or GAD67, glutamate decarboxylase) and GAD2 (or GAD65)
GAD1 and GAD2 are two isoforms of glutamate decarboxylase. These enzymes catalyze the reaction to synthesize GABA from glutamate and are responsible for keeping cortical GABA levels at steady state (Soghomonian and Martin, 1998). Thus, a reduced expression of GAD1 and GAD2 may lead to decreased GABA levels and less inhibition of downstream targets. Decreased GAD67 expression has been implicated in schizophrenia and bipolar disorder with psychosis (Guidotti et al., 2000).
CRHBP (corticotrophin releasing hormone binding protein)
CRHBP is an important modulatory protein that negatively regulates corticotrophin releasing hormone (CRH) activity. CRHBP binds to CRH and thus reduces the ability of CRH to activate the CRHR1 and CRHR2 receptors (Jahn et al., 2002). CRHBP is a physiologically relevant reservoir of endogenous CRH, as 40–60% of human brain CRH is bound by CRHBP (Behan et al., 1995). CRH is released in response to stress (Holsboer and Ising, 2010). The reduction in CRHBP in our ITTO conditioned rats could lead to less binding of CRH and more free CRH which can then activate its receptors and elevate the stress response. In a CRHBP-deficient mouse model, Karolyi et al. (1999) have demonstrated increased anxiogenic behavior as tested on the elevated plus maze and open field. A decrease in CRHBP has also been suggested to play a role in the pathogenesis of major depressive disorder by inhibiting the function of CRH (Van Den Eede et al., 2005).
HTR (serotonin receptors)
The serotonin system has been shown to play a critical role in a variety of human psychopathological conditions, particularly mood and anxiety disorders (Hensler, 2006). Antidepressant treatment has thus focused on modulating serotonergic neurotransmission (Jones and Blackburn, 2002). One of the challenges of the serotonin system is the complexity of the pathway with 14 known receptor varieties categorized into seven receptor subtypes (Hoyer et al., 2002).
The HTR1A subtype exists on pre-synaptic neurons in raphe nuclei as well as on post-synaptic neurons in other brain regions, such as the hippocampus and the thalamus. Agonists have different effects depending on the location of the receptors, in that agonists to pre-synaptic receptors result in anxiolytic behaviors while agonists to post-synaptic receptors lead to anti-depressive behaviors (Schreiber and De Vry, 1993). Kennett et al. (1987) have demonstrated that the 5-HTR1A agonist 8-hydroxy-2-(di-n-propylamino) tetralin (8-OH-DPAT) may have rapid antidepressant properties. Partial HTR1A agonists may also be effective in the treatment of generalized anxiety disorder (Jones and Blackburn, 2002) and schizophrenia (Millan, 2000). HTR1A knockout mice show elevated anxiety levels in open field, elevated-zero maze, and novel-object assays (Heisler et al., 1998) and are less reactive, more anxious, and possibly less aggressive than the wild-types (Ramboz et al., 1998). Dysfunction of this receptor has been suggested to also play a role in the genesis of major depressive disorder in humans (Savitz et al., 2009). PET studies in patients with posttraumatic stress and panic disorders (Neumeister et al., 2004) and depression (Drevets et al., 1999) have shown reduced HTR1A receptor binding potential and reduced receptor availability (Nash et al., 2008).
In an acute ITTO study in anesthetized rats we have shown that the serotonin receptor HTR2A was up-regulated following one 10 min occlusion trial (Bernhardt et al., 2008). This is in general agreement with the suggestion that anxiety is the result of a hypersensitive serotonin system (Heisler et al., 1998). However, contrary to the acute ITTO response, in the present study we found 10 days of chronic exposure to ITTO down-regulated HTR2A and HTR1A receptor genes. Thus, chronic ITTO conditioning down-regulation of HTR1A and HTR2A suggests serotonergic receptor expression may be reduced either as a result of excessive release if serotonin or an adaptive hyposensitivity response of thalamic neurons with 10 days of ITTO conditioning. The down-regulation of serotonin receptors may therefore be related to anxiety (Heisler et al., 1998) or impulsivity and depression (Schreiber and De Vry, 1993).
Chronic exposure to ITTO modulates pathways involved in learning and memory, cell processes, and cell signaling
Ten days of ITTO resulted in a behavioral adaption in order to cope with the stress of the occlusion trials. These adaptations were characterized by decreased exploratory behavior, increased submissive state, and even breath holding (Pate et al., 2010). This finding is consistent with learned helplessness. Memory of previous occlusion trials may, thus, alter molecular pathways of learning and/or memory. Indeed, in the present study, most of the genes that were found to be up-regulated have been shown to increase learning and/or memory, while the ones that were down-regulated decrease these pathways (Figure 3). The modulation of the learning/memory gene pathway suggests a behavioral conditioning effect of ITTO in response to 10 days of occlusion exposure.
Modulated cellular processes included cell proliferation, differentiation, neuroprotection, and cell death. A common trend in the analysis of these pathways was the inhibitory regulation of cell death by up-regulated genes, while neuroprotection was positively regulated. Cell proliferation seemed to be down-regulated because the genes that are activating this process showed decreased expression. However, cell differentiation appeared to be increased, both by positive regulation of up-regulated genes as well as by inhibitory regulation of down-regulated ones suggesting thalamic neural remodeling in response to chronic exposure to ITTO.
Differentially regulated genes involved in cell signaling were Ca2+ transport, membrane polarization, synaptic potentiation, and transmission. Involvement of these genes is less clear and often in opposite directions. The balance of up- and down-regulation of genes in these pathways may be important in determining the final outcome behaviors. The GO category synaptic transmission was one of the most significantly modulated biological processes (Table 3) suggesting thalamic neuronal functional reconfiguration in response to 10 days of ITTO conditioning.
Conclusion
This study identified 661 genes in the medial thalamus significantly modulated by repeated exposure to ITTO in conscious rats. Thalamic genes related to anxiety, depression, and sensory information processing were significantly changed. These results are consistent with the thalamus gating respiratory mechanosensory information to the somatosensory and affective cortices. We also found the pattern of gene expression with chronic ITTO different from acute (10 min) exposure to ITTO. Chronic ITTO has a conditioning effect on behavioral responses of rats (Pate et al., 2010). The ITTO elicited changes in the state gene expression profile of the medial thalamus suggests modulation of the learning and/or memory, cellular processes and cell signaling gene pathways. Modulation of these pathways may provide a neuromolecular foundation for respiratory afferent information processing. A change in processing of information to higher brain centers may be the functional basis for the somatosensory and affective behavioral responses to ITTO respiratory conditioning. The respiratory load compensation response has been shown to be altered in conscious rats following 10 days of repeated ITTO (Pate et al., 2010). This alteration may be due to a change in thalamic neuronal function that, in part, regulates the behavioral control of breathing.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary Material
Supplementary Material for this article can be found online at http://www.frontiersin.org/respiratory_physiology/10.3389/ fphys.2011.00024/abstract
Acknowledgments
Support for B. Lynn Escalon was provided by the US Army Environmental Quality Research Program.
References
- Behan D. P., Heinrichs S. C., Troncoso J. C., Liu X. J., Kawas C. H., Ling N., De Souza E. B. (1995). Displacement of corticotropin releasing factor from its binding protein as a possible treatment for Alzheimer’s disease. Nature 378, 284–287 10.1038/378284a0 [DOI] [PubMed] [Google Scholar]
- Bernhardt V., Denslow N., Liu L., Pate K., Vovk A., Davenport P. W. (2008). Tracheal occlusion modulation of gene expression in the medial thalamus. Am. J. Respir. Crit. Care Med. 177, A745 [Google Scholar]
- Brazma A., Hingamp P., Quackenbush J., Sherlock G., Spellman P., Stoeckert C., Aach J., Ansorge W., Ball C. A., Causton H. C., Gaasterland T., Glenisson P., Holstege F. C., Kim I. F., Markowitz V., Matese J. C., Parkinson H., Robinson A., Sarkans U., Schulze-Kremer S., Stewart J., Taylor R., Vilo J., Vingron M. (2001). Minimum information about a microarray experiment (MIAME)-toward standards for microarray data. Nat. Genet. 29, 365–371 [DOI] [PubMed] [Google Scholar]
- Chan P. Y., Davenport P. W. (2008). Respiratory-related evoked potential measures of respiratory sensory gating. J. Appl. Physiol. 105, 1106–1113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan P. Y., Davenport P. W. (2010). The role of nicotine on respiratory sensory gating measured by respiratory-related evoked potentials. J. Appl. Physiol. 108, 662–669 [DOI] [PubMed] [Google Scholar]
- Chen Z., Eldridge F. L., Wagner P. G. (1992). Respiratory-associated thalamic activity is related to level of respiratory drive. Respir. Physiol. 90, 99–113 [DOI] [PubMed] [Google Scholar]
- Cheong E., Lee S., Choi B. J., Sun M., Lee C. J., Shin H. S. (2008). Tuning thalamic firing modes via simultaneous modulation of T- and L-type Ca2 channels controls pain sensory gating in the thalamus. J. Neurosci. 28, 13331–13340 10.1523/JNEUROSCI.3013-08.2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung L., Moore S. D., Cox C. L. (2009). Cholecystokinin action on layer 6b neurons in somatosensory cortex. Brain Res. 1282, 10–19 10.1016/j.brainres.2009.05.061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corfield D. R., Fink G. R., Ramsay S. C., Murphy K., Harty H. R., Watson J. D., Adams L., Frackowiak R. S., Guz A. (1995). Evidence for limbic system activation during CO2-stimulated breathing in man. J. Physiol. 488(Pt 1), 77–84 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Correia S. S., Duarte C. B., Faro C. J., Pires E. V., Carvalho A. L. (2003). Protein kinase C gamma associates directly with the GluR4 alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionate receptor subunit. Effect on receptor phosphorylation. J. Biol. Chem. 278, 6307–6313 [DOI] [PubMed] [Google Scholar]
- Davenport P. W., Vovk A. (2009). Cortical and subcortical central neural pathways in respiratory sensations. Respir. Physiol. Neurobiol. 167, 72–86 [DOI] [PubMed] [Google Scholar]
- Di Marco F., Verga M., Reggente M., Maria Casanova F., Santus P., Blasi F., Allegra L., Centanni S. (2006). Anxiety and depression in COPD patients: the roles of gender and disease severity. Respir. Med. 100, 1767–1774 [DOI] [PubMed] [Google Scholar]
- Drevets W. C., Frank E., Price J. C., Kupfer D. J., Holt D., Greer P. J., Huang Y. Y., Gautier C., Mathis C. (1999). PET imaging of serotonin 1A receptor binding in depression. Biol. Psychiatry 46, 1375–1387 [DOI] [PubMed] [Google Scholar]
- Graeff F. G., Guimaraes F. S., De Andrade T. G., Deakin J. F. (1996). Role of 5-HT in stress, anxiety, and depression. Pharmacol. Biochem. Behav. 54, 129–141 [DOI] [PubMed] [Google Scholar]
- Guidotti A., Auta J., Davis J. M., Di-Giorgi-Gerevini V., Dwivedi Y., Grayson D. R., Impagnatiello F., Pandey G., Pesold C., Sharma R., Uzunov D., Costa E. (2000). Decrease in reelin and glutamic acid decarboxylase67 (GAD67) expression in schizophrenia and bipolar disorder: a postmortem brain study. Arch. Gen. Psychiatry 57, 1061–1069 [DOI] [PubMed] [Google Scholar]
- Gulpinar M. A., Yegen B. C. (2004). The physiology of learning and memory: role of peptides and stress. Curr. Protein Pept. Sci. 5, 457–473 [DOI] [PubMed] [Google Scholar]
- Harada K., Yamaji T., Matsuoka N. (2008). Activation of the serotonin 5-HT(2C) receptor is involved in the enhanced anxiety in rats after single-prolonged stress. Pharmacol. Biochem. Behav. 89, 11–16 [DOI] [PubMed] [Google Scholar]
- Harro J., Vasar E. (1991). Cholecystokinin-induced anxiety: how is it reflected in studies on exploratory behaviour? Neurosci. Biobehav. Rev. 15, 473–477 [DOI] [PubMed] [Google Scholar]
- Harro J., Vasar E., Bradwejn J. (1993). CCK in animal and human research on anxiety. Trends Pharmacol. Sci. 14, 244–249 [DOI] [PubMed] [Google Scholar]
- Heisler L. K., Chu H. M., Brennan T. J., Danao J. A., Bajwa P., Parsons L. H., Tecott L. H. (1998). Elevated anxiety and antidepressant-like responses in serotonin 5-HT1A receptor mutant mice. Proc. Natl. Acad. Sci. U.S.A. 95, 15049–15054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heisler L. K., Zhou L., Bajwa P., Hsu J., Tecott L. H. (2007). Serotonin 5-HT(2C) receptors regulate anxiety-like behavior. Genes Brain Behav. 6, 491–496 10.1111/j.1601-183X.2007.00316.x [DOI] [PubMed] [Google Scholar]
- Hensler J. G. (2006). Serotonergic modulation of the limbic system. Neurosci. Biobehav. Rev. 30, 203–214 [DOI] [PubMed] [Google Scholar]
- Holsboer F., Ising M. (2010). Stress hormone regulation: biological role and translation into therapy. Annu. Rev. Psychol. 61, 81–109, C101–C111. [DOI] [PubMed] [Google Scholar]
- Hoyer D., Hannon J. P., Martin G. R. (2002). Molecular, pharmacological and functional diversity of 5-HT receptors. Pharmacol. Biochem. Behav. 71, 533–554 [DOI] [PubMed] [Google Scholar]
- Huang K. P. (1989). The mechanism of protein kinase C activation. Trends Neurosci. 12, 425–432 10.1016/0166-2236(89)90091-X [DOI] [PubMed] [Google Scholar]
- Jahn O., Eckart K., Brauns O., Tezval H., Spiess J. (2002). The binding protein of corticotropin-releasing factor: ligand-binding site and subunit structure. Proc. Natl. Acad. Sci. U.S.A. 99, 12055–12060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones B. J., Blackburn T. P. (2002). The medical benefit of 5-HT research. Pharmacol. Biochem. Behav. 71, 555–568 [DOI] [PubMed] [Google Scholar]
- Karolyi I. J., Burrows H. L., Ramesh T. M., Nakajima M., Lesh J. S., Seong E., Camper S. A., Seasholtz A. F. (1999). Altered anxiety and weight gain in corticotropin-releasing hormone-binding protein-deficient mice. Proc. Natl. Acad. Sci. U.S.A. 96, 11595–11600 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennett G. A., Dourish C. T., Curzon G. (1987). Antidepressant-like action of 5-HT1A agonists and conventional antidepressants in an animal model of depression. Eur. J. Pharmacol. 134, 265–274 [DOI] [PubMed] [Google Scholar]
- Kew J. N., Kemp J. A. (2005). Ionotropic and metabotropic glutamate receptor structure and pharmacology. Psychopharmacology (Berl.) 179, 4–29 [DOI] [PubMed] [Google Scholar]
- Klak K., Palucha A., Branski P., Sowa M., Pilc A. (2007). Combined administration of PHCCC, a positive allosteric modulator of mGlu4 receptors and ACPT-I, mGlu III receptor agonist evokes antidepressant-like effects in rats. Amino Acids 32, 169–172 10.1007/s00726-006-0316-z [DOI] [PubMed] [Google Scholar]
- Kobayashi T., Washiyama K., Ikeda K. (2004). Inhibition of G protein-activated inwardly rectifying K+ channels by various antidepressant drugs. Neuropsychopharmacology 29, 1841–1851 10.1038/sj.npp.1300484 [DOI] [PubMed] [Google Scholar]
- Lang P. J., Wangelin B. C., Bradley M. M., Versace F., Davenport P. W., Costa V. D. (2010). Threat of suffocation and defensive reflex activation. Psychophysiology 48, 393–396 10.1111/j.1469-8986.2010.01076.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCormick D. A., Bal T. (1994). Sensory gating mechanisms of the thalamus. Curr. Opin. Neurobiol. 4, 550–556 [DOI] [PubMed] [Google Scholar]
- McKay L. C., Evans K. C., Frackowiak R. S., Corfield D. R. (2003). Neural correlates of voluntary breathing in humans. J. Appl. Physiol. 95, 1170–1178 [DOI] [PubMed] [Google Scholar]
- Millan M. J. (2000). Improving the treatment of schizophrenia: focus on serotonin (5-HT)(1A) receptors. J. Pharmacol. Exp. Ther. 295, 853–861 [PubMed] [Google Scholar]
- Moran T. H., Schwartz G. J. (1994). Neurobiology of cholecystokinin. Crit. Rev. Neurobiol. 9, 1–28 [PubMed] [Google Scholar]
- Nash J. R., Sargent P. A., Rabiner E. A., Hood S. D., Argyropoulos S. V., Potokar J. P., Grasby P. M., Nutt D. J. (2008). Serotonin 5-HT1A receptor binding in people with panic disorder: positron emission tomography study. Br. J. Psychiatry 193, 229–234 10.1192/bjp.bp.107.041186 [DOI] [PubMed] [Google Scholar]
- Neumeister A., Bain E., Nugent A. C., Carson R. E., Bonne O., Luckenbaugh D. A., Eckelman W., Herscovitch P., Charney D. S., Drevets W. C. (2004). Reduced serotonin type 1A receptor binding in panic disorder. J. Neurosci. 24, 589–591 10.1523/JNEUROSCI.4921-03.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nikitin A., Egorov S., Daraselia N., Mazo I. (2003). Pathway studio – the analysis and navigation of molecular networks. Bioinformatics 19, 2155–2157 10.1093/bioinformatics/btg290 [DOI] [PubMed] [Google Scholar]
- Niswender C. M., Conn P. J. (2010). Metabotropic glutamate receptors: physiology, pharmacology, and disease. Annu. Rev. Pharmacol. Toxicol. 50, 295–322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Donnell D. E., Banzett R. B., Carrieri-Kohlman V., Casaburi R., Davenport P. W., Gandevia S. C., Gelb A. F., Mahler D. A., Webb K. A. (2007). Pathophysiology of dyspnea in chronic obstructive pulmonary disease: a roundtable. Proc. Am. Thorac. Soc. 4, 145–168 [DOI] [PubMed] [Google Scholar]
- Omachi T. A., Katz P. P., Yelin E. H., Gregorich S. E., Iribarren C., Blanc P. D., Eisner M. D. (2009). Depression and health-related quality of life in chronic obstructive pulmonary disease. Am. J. Med. 122, 778 e9–778.e15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pate K. M., Scheuer D. A., Davenport P. W. (2010). Behavioral and physiological changes associated with chronic tracheal obstructions in conscious rats. FASEB J. 24, 79923.19897662 [Google Scholar]
- Ramboz S., Oosting R., Amara D. A., Kung H. F., Blier P., Mendelsohn M., Mann J. J., Brunner D., Hen R. (1998). Serotonin receptor 1A knockout: an animal model of anxiety-related disorder. Proc. Natl. Acad. Sci. U.S.A. 95, 14476–14481 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramcharan E. J., Gnadt J. W., Sherman S. M. (2000). Burst and tonic firing in thalamic cells of unanesthetized, behaving monkeys. Vis. Neurosci. 17, 55–62 [DOI] [PubMed] [Google Scholar]
- Rotzinger S., Vaccarino F. J. (2003). Cholecystokinin receptor subtypes: role in the modulation of anxiety-related and reward-related behaviours in animal models. J. Psychiatry Neurosci. 28, 171–181 [PMC free article] [PubMed] [Google Scholar]
- Saenz del Burgo L., Cortes R., Mengod G., Zarate J., Echevarria E., Salles J. (2008). Distribution and neurochemical characterization of neurons expressing GIRK channels in the rat brain. J. Comp. Neurol. 510, 581–606 [DOI] [PubMed] [Google Scholar]
- Saito N., Shirai Y. (2002). Protein kinase C gamma (PKC gamma): function of neuron specific isotype. J. Biochem. 132, 683–687 [DOI] [PubMed] [Google Scholar]
- Savitz J., Lucki I., Drevets W. C. (2009). 5-HT(1A) receptor function in major depressive disorder. Prog. Neurobiol. 88, 17–31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreiber R., De Vry J. (1993). 5-HT1A receptor ligands in animal models of anxiety, impulsivity and depression: multiple mechanisms of action? Prog. Neuropsychopharmacol. Biol. Psychiatry 17, 87–104 [DOI] [PubMed] [Google Scholar]
- Sherman S. M. (1996). Dual response modes in lateral geniculate neurons: mechanisms and functions. Vis. Neurosci. 13, 205–213 [DOI] [PubMed] [Google Scholar]
- Sherman S. M. (2001). Thalamic relay functions. Prog. Brain Res. 134, 51–69 10.1016/S0079-6123(01)34005-0 [DOI] [PubMed] [Google Scholar]
- Sherman S. M., Guillery R. W. (2002). The role of the thalamus in the flow of information to the cortex. Philos. Trans. R Soc. Lond. B. Biol. Sci. 357, 1695–1708 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smyth G. K., Michaud J., Scott H. S. (2005). Use of within-array replicate spots for assessing differential expression in microarray experiments. Bioinformatics 21, 2067–2075 10.1093/bioinformatics/bti270 [DOI] [PubMed] [Google Scholar]
- Soghomonian J. J., Martin D. L. (1998). Two isoforms of glutamate decarboxylase: why? Trends Pharmacol. Sci. 19, 500–505 [DOI] [PubMed] [Google Scholar]
- Swadlow H. A., Gusev A. G. (2001). The impact of ‘bursting’ thalamic impulses at a neocortical synapse. Nat. Neurosci. 4, 402–408 [DOI] [PubMed] [Google Scholar]
- Van Den Eede F., Van Broeckhoven C., Claes S. J. (2005). Corticotropin-releasing factor-binding protein, stress and major depression. Ageing Res. Rev. 4, 213–239 [DOI] [PubMed] [Google Scholar]
- Vovk A., Pate K. M., Davenport P. (2006). “Respiratory response to transient, reversible tracheal obstruction in rats,” in 2006 Neuroscience Meeting Planner (Atlanta, GA: Society for Neuroscience; ). [Google Scholar]
- Weisstaub N. V., Zhou M., Lira A., Lambe E., Gonzalez-Maeso J., Hornung J. P., Sibille E., Underwood M., Itohara S., Dauer W. T., Ansorge M. S., Morelli E., Mann J. J., Toth M., Aghajanian G., Sealfon S. C., Hen R., Gingrich J. A. (2006). Cortical 5-HT2A receptor signaling modulates anxiety-like behaviors in mice. Science 313, 536–540 10.1126/science.1123432 [DOI] [PubMed] [Google Scholar]
- Yates J. S., Davenport P. W., Reep R. L. (1994). Thalamocortical projections activated by phrenic nerve afferents in the cat. Neurosci. Lett. 180, 114–118 [DOI] [PubMed] [Google Scholar]
- Zhang W., Davenport P. W. (2003). Activation of thalamic ventroposteriolateral neurons by phrenic nerve afferents in cats and rats. J. Appl. Physiol. 94, 220–226 [DOI] [PubMed] [Google Scholar]