Abstract
Understanding how individual identity is processed from faces remains a complex problem. Contrast reversal, showing faces in photographic negative, impairs face recognition in humans and demonstrates the importance of surface-based information (shading and pigmentation) in face recognition. We tested the importance of contrast information for face encoding in chimpanzees and rhesus monkeys using a computerized face-matching task. Results showed that contrast reversal (positive to negative) selectively impaired face processing in these two species, although the impairment was greater for chimpanzees. Unlike chimpanzees, however, monkeys performed just as well matching negative to positive faces, suggesting that they retained some ability to extract identity information from negative faces. A control task showed that chimpanzees, but not rhesus monkeys, performed significantly better matching face parts compared with whole faces after a contrast reversal, suggesting that contrast reversal acts selectively on face processing, rather than general visual-processing mechanisms. These results confirm the importance of surface-based cues for face processing in chimpanzees and humans, while the results were less salient for rhesus monkeys. These findings make a significant contribution to understanding the evolution of cognitive specializations for face processing among primates, and suggest potential differences between monkeys and apes.
Keywords: face recognition, contrast reversal, configuration, chimpanzee, rhesus monkey, evolution
1. Introduction
Faces are important social stimuli, yet many questions remain about how individual identity is extracted from faces and whether these processes are conserved among primates. Among humans, faces are processed using a combination of feature-based and configural strategies [1,2]. Several types of configural cue have been identified. The first-order configuration describes the presence and general arrangement of facial features on a face (e.g. eyes are above the nose), which is the same in every face. The second-order configuration, however, refers to the precise spacing of these features with regard to one another, which is unique in every face [3]. The importance of second-order configural cues for face recognition has been well documented using the face-inversion effect [4,5]. Turning a face upside down significantly impairs recognition by limiting one's ability to extract information about the spacing of features [3,6–8]. Other second-order cues include surface-based information such as the pigmentation of the face (e.g. non-uniformly distributed surface variations like texture and colour) and the shape information produced by shading differences reflected by the luminance profiles that affect the surface geometry of the face [9–11]. Although the majority of face-recognition studies focus on the spacing of features, surface-based cues appear to provide highly selective information regarding a face's identity. Therefore, an examination of surface-based cues may provide a better avenue for understanding the evolution of individual recognition.
One of the more widely used tests of the importance of surface-based cues is contrast reversal [12]. By reversing the contrast information in faces (e.g. showing faces in photographic negative), information about pigmentation and shading is lost and face discrimination performance can drop as much as 20 to 30 per cent [9–14]. Contrast reversal has little impact on faces that have been stripped of pigmentation, the recognition of facial expressions, a face's gender or whether the task involves recognition memory for faces [9,15,16]. Although the information encoded in surface-based cues has been shown to be important for object recognition, it appears to be strongest for encoding the identity of faces [14,16]. Nederhouser et al. [14] compared the effect of contrast reversal for faces with that of computer-generated objects (blobs). Subjects made approximately 20 per cent more errors matching faces of reversed contrast compared with faces of the same contrast, while there were no significant differences in performance matching blobs after a contrast reversal [14]. Contrast reversal produced greater deficits for pigmented faces compared with pigmented greebles (a three-dimensional set of man-made objects where, like faces, specific individuals can be discriminated at the subordinate level; e.g. greeble 1 versus greeble 2 [16]). However, those authors conclude that the main differences were quantitative. Performance dropped approximately 22 per cent when subjects matched contrast-reversed faces compared with same-polarity faces, but the drop was 11 per cent for greebles. In sum, contrast reversal can affect object recognition but it is quantitatively greater for faces, implicating its action on face-selective perceptual mechanisms. Because the effects of contrast reversal are minimal when the judgements are for facial expression, gender or in tasks involving recognition memory, it appears that contrast reversal selectively affects the perceptual analyses required to make judgements about facial identity.
The last 10 years has seen a rise in the number of studies on non-human primate face processing, with the majority focusing on the face-inversion effect (see [17] for a review of this literature); however, there remains considerable debate as to whether faces are processed in a manner similar to or different from humans. Only a handful of studies have specifically addressed the importance of second-order configural cues, using a variety of task manipulations, including individuation [18–24], manipulating the spacing of features [18,22,25,26] and the Thatcher illusion [27–29]. These studies have produced mixed results. Dahl et al. [18] and Sugita [26] reported that monkeys were able to detect subtle changes in the spacing of features, but Parr et al. [22] failed to show selective impairments in the face-recognition performance of monkeys after manipulations of second-order configural cues. Moreover, Parron & Fagot [29] showed that while baboons were able to detect changes in the first-order configuration (changing the position of the features), they failed to discriminate faces that had second-order configural changes (Thatcherized faces). These studies should be interpreted cautiously, as experimentally any manipulation of first-order configural features automatically alters their second-order configuration.
Two well-controlled studies provide favourable evidence for the individuation of faces in monkeys. Gothard et al. [19] used eye-tracking in combination with a visual paired comparison task to examine the ability of monkeys to discriminate conspecifics' faces. They first habituated the monkeys to two photographs of the same monkey shown with different head orientations. Then, they showed a new photograph of this same monkey plus a novel individual. Monkeys looked longer at the novel individual, suggesting that they could discriminate between the identity of the two monkeys, even though all the individual photographs and facial orientations were different. Pokorny & de Waal [24] trained capuchin monkeys to perform a four-choice oddity task. During the initial training, three identical photographs showed one monkey and the fourth showed a different monkey (odd stimulus). After learning to select the odd photograph, the test phase presented three different photographs of the same monkey plus a novel individual. Monkeys performed these trials above chance and also generalized their performance to novel photographs. Using a matching-to-sample (MTS) task, however, Parr and colleagues have consistently found that rhesus monkeys perform more poorly than chimpanzees in tasks that require them to individuate unfamiliar conspecifics' faces [21–23]. Not only do the monkeys require many trials before they learn to discriminate between two faces, but they often fail to generalize their performance to novel photographs [23]. Moreover, there are considerable individual differences in the performance across subjects and in response to specific face stimuli [21]. In contrast, chimpanzees have consistently performed well on individual discrimination tasks, suggesting that they are more tuned to individual identity than monkeys [23].
Only one recent study has examined face processing in a chimpanzee using contrast-reversed images. Tomonaga & Imura [30] were interested in gaze detection and presented one chimpanzee with an oddity task where she was required to select the face that showed a different gaze direction than the distracters. Performance was impaired in trials in which the entire face, or just the eyes, were presented in their photographic negative. However, performance was good if the contrast reversal was for the face, but not the eyes. Because this study focused on gaze detection using human faces, it is unclear whether similar effects would be found for discriminations involving the identity of conspecifics' faces. Therefore, there is still much to be learned about the evolution of face processing, and whether human and non-human primates rely on similar visual features to code face identity.
Here, we present data on the importance of surface-based information for face discrimination in chimpanzees and rhesus monkeys using a contrast-reversal task. Subjects were first trained to match unfamiliar conspecifics' faces in their positive (P) or negative (N) greyscale contrast. After this training, the test sessions required them to match these original greyscale images either to the same face presented with reversed contrast (PN or NP), or to the same face presented in colour (PC or NC). This performance was contrasted with performance on the original training images, e.g. positive or negative. Based on previous studies showing inconclusive data on the salience of second-order configural cues in tasks of monkey face processing [22,31], it is expected that monkeys would be less sensitive to changes in the surface-based cues in a face than chimpanzees. Specifically, sensitivity to surface-based cues is expected to reveal significant deficits for trials in which there has been a contrast reversal (PP > PN, NN > NP) or a change in colour (PC > PN, NN > NC). Additionally, no differences are expected between performance on the NP compared with NC trials, as each of these contains a contrast reversal with the addition of colour reversal in the NC trials. Subjects should also show no differences in performance between PP and PC trials, in which the only manipulation is a change in colour. Finally, to examine whether contrast reversal acts selectively on face-processing mechanisms, these data are compared with a control condition in which subjects are required to match face parts after a contrast reversal. These data will make a significant contribution to the ongoing controversy surrounding the evolution of cognitive specializations for face processing among non-human primates.
2. Methods
(a). Subjects
Six chimpanzees (four males and two females) ranging in age from 16 to 22 years and six rhesus monkeys (two males and four females) 8 years aged participated voluntarily in these studies. The chimpanzees were raised by humans in peer groups at the Yerkes Primate Center until 4 years of age when they joined established social groups. The rhesus monkeys were raised in large social groups at the Yerkes Primate Center field station until 4 years of age, when they were relocated to the Yerkes main campus and housed in same-sex pairs. All subjects were tested daily in their home cage. Chimpanzees were tested using a computerized joystick testing system and the monkeys used a custom-designed touchscreen computer system [22]. Chimpanzees were reinforced for correct responses by hand while rhesus monkeys received treats delivered to a small cup located beneath the touchscreen. All subjects had extensive experience of performing computerized tasks of face recognition using MTS prior to this study [22,23,25].
(b). Stimuli
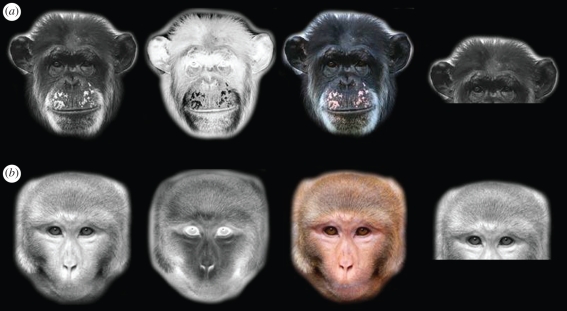
Stimuli consisted of conspecifics' faces, familiarized to subjects through an initial training procedure. Photographs of 60 novel individuals from each species were used. These photographs were taken with a digital camera (Canon SLR D50) from individuals unfamiliar to the subjects. Photographs were taken only in overcast weather to obtain high-quality facial images devoid of obvious shadows. These photographs were cropped to reveal only the face, placed on a black background, and sized to 300 pixels in height. Photographs were presented either in full RBG colour (colour, C), positive polarity greyscale (positive, P) or negative polarity greyscale (negative, N). Negative polarity was achieved by reversing the greyscale values so that 256 became 1, 255 became 2, etc. All photographic manipulations were done using Adobe Photoshop (CS2, www.adobe.com). Figure 1 illustrates an example of each image type for both chimpanzee and monkey faces.
Figure 1.
An illustration of each image type in (a) chimpanzees and (b) rhesus monkeys. From left to right: positive, negative, colour and the top face parts used in the control condition (shown positive contrast only).
Also included in the control task were the faces of 30 novel conspecifics cropped above the nostrils to show only the upper half of the face (figure 1). These face parts were edited to show both positive and negative contrast similar to that described above.
(c). Procedure
All subjects were tested using an MTS format. For the study of face identity encoding using contrast reversal, MTS is a preferred method in humans as it does not elicit biases inherent in other methodologies, such as same–different paradigms [14,15]. According to the MTS procedure, subjects are first shown a single image on the computer monitor, referred to as the sample. This is presented centrally against one of the four sides of the computer monitor. After orienting to this image, either by contacting it with the joystick-controlled cursor (chimpanzee), or touching it three times in rapid succession on the touchscreen (rhesus monkey), two additional images appeared simultaneously on the screen located equidistant from the sample on the opposite side of the monitor. One of these images (target) was identical to the sample while the other (foil) was a different exemplar from the same category (e.g. another conspecific's face with the same colour/polarity as the target). A correct response to the target image was followed by an inter-trial interval (ITI) of 2 s, while an incorrect response to the foil was followed by an ITI of 6 s and no food reinforcement. The next trial was then presented.
Subjects were first trained to match 15 trials showing either positive or negative contrast faces. The order of presentation was counterbalanced across subjects and all subjects completed each training set. A different set of photographs was used for the positive and negative training tasks so that no individual or photograph was repeated. Subjects received three repetitions of each trial during the daily training sessions (45 total trials), which continued until their performance exceeded 39 of 45 correct (86.67%) on a single testing session, or 38 of 45 (84.44%) over two sessions.
Once subjects reached either of these criteria, they were advanced to the test phase, which contained the original 15 training trials (either positive = PP or negative = NN), plus the same trials in which the two comparison images were presented in the reversed polarity (PN or NP), or in colour (PC or NC). Thus, the test trials contained 45 trials: 15 trials each of control, contrast-reversed or colour-reversed faces. Figure 2a illustrates an example of the three types of test trials presented to rhesus monkeys after positive training. Figure 2b illustrates an example of the three types of trials presented to chimpanzees after negative training. Subjects were given only four test sessions (4 × 45 trials = 180 trials), presented in a pseudo-random order such that each stimulus was seen exactly once per session. After completing the first testing session, subjects moved on to train with the opposite-polarity images using the same criteria and procedures as described above.
Figure 2.
An illustration of three types of test trials (a) following positive contrast training (illustrating rhesus monkeys): (left to right) positive–positive (PP), positive–negative (PN) and positive–colour (PC); and (b) following negative training (illustrating chimpanzees): negative–negative (NN), negative–positive (NP) and negative–colour (NC).
(d). Data analysis
An initial analysis compared the number of sessions required for subjects to reach the training criteria for the positive and negative contrast images. Next, in order to assess the magnitude of contrast-reversal effects, a 2 × 3 repeated-measure ANOVA was performed for each species, in which training image type (positive versus negative) and test image type (positive, negative and colour) were the within-subject factors. By conducting separate analyses for each species, the magnitude of p-values for each main effect and interaction could be compared, as opposed to simply looking for an overall main effect of species. Effect sizes are given using eta-squared (η2). Follow-up comparisons were performed where appropriate and adjusted for multiple comparisons using Bonferroni's correction procedure (α1 = α / no. of comparisons).
(e). Is contrast reversal face-specific?
To determine whether the effects of contrast reversal act specifically on face-processing and not general visual-processing mechanisms, a control condition was added. Subjects1 were presented with the faces of 30 new conspecifics that were edited to show only the top face part (figure 1), thus preventing the engagement of normal face-processing strategies. Using the same MTS format, subjects were presented with 15 trials in which the sample and correct comparison showed identical top face parts in their original positive contrast, while the non-matching comparison was a top face part from another individual. After reaching a criterion of greater than 85 per cent correct on these 15 trials, they were edited so that the comparison images were now presented in their photographic negative. The sample remained photographic positive. Subjects received two test sessions including these contrast-reversed trials, where each trial was seen twice in a session. Analyses then compared subjects' performances matching face parts versus whole faces across a change in contrast (P to N). If contrast reversal specifically affects the cognitive processes engaged by whole faces, then the magnitude of the contrast reversal is expected to be less for face parts than for whole faces.
3. Results
(a). Training sessions
One monkey subject failed to reach the training criterion with both positive and negative faces after 20 sessions and she was dropped from the study. One chimpanzee (Scott) failed to reach the formal training criterion on the negative images. His performance, however, exceeded 80 per cent on six of 20 sessions, so he was allowed to proceed to the testing phase. Two monkeys (Cw and Lm) failed to meet the formal training criterion on either the positive or negative sessions. The performance of Cw on the positive images exceeded 70 per cent for 15 sessions and 80 per cent for two sessions, and her performance exceeded 75 per cent on five of the negative training sessions. The performance of Lm on the positive images exceeded 70 per cent eight times, and her performance exceeded 80 per cent on three of the negative training sessions and 84.44 per cent on one session. Both of these females were allowed to advance to the test sessions. For the training analyses, the number of sessions to reach the criterion was set at 20 for these subjects.
Table 1 shows the number of training sessions that were required before subjects reached the performance criteria on positive and negative whole faces and face parts in the control task. A repeated-measures ANOVA using training stimuli (positive versus negative) as the within-subject factor revealed a significant main effect of training stimuli (F1,9 = 6.07, p < 0.04; effect size η2 = 0.40). Comparisons across species showed a near-significant difference between chimpanzees and rhesus monkeys on the positive whole faces (t(9) = 2.13, p = 0.06), where chimpanzees performed faster than monkeys, but no differences matching negative whole faces (t(9) = 0.95, p = 0.37) or face parts in the control task (t(8) = 0.19, p = 0.85). Overall, subjects reached the criterion fastest on the face parts.
Table 1.
Number of sessions required for subjects to reach the performance criteria for training on positive and negative faces. n.a., not applicable.
| subject | species | positive train | negative train | control train |
|---|---|---|---|---|
| Jarred | chimpanzee | 2 | 3 | n.a. |
| Lamar | chimpanzee | 1 | 6 | 3 |
| Scott | chimpanzee | 2 | 20a | 7 |
| Katrina | chimpanzee | 5 | 1 | 1 |
| Patrick | chimpanzee | 1 | 9 | 9 |
| Faye | chimpanzee | 7 | 17 | 5 |
| mean | chimpanzee | 3.0 | 9.3 | 5.0 |
| Rocket | rhesus | 3 | 10 | 1 |
| Samosa | rhesus | 8 | 11 | 5 |
| Onion | rhesus | 3 | 6 | 1 |
| Chewy | rhesus | 20a | 20a | 16 |
| Lemon | rhesus | 20a | 20a | 5 |
| mean | rhesus | 10.8 | 13.4 | 5.6 |
aPerformance for these subjects was stopped after 20 sessions. See text for details about criteria.
(b). Test sessions
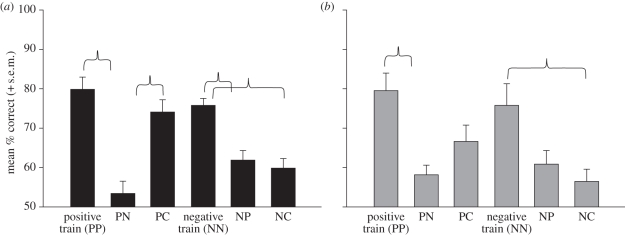
Analyses were performed separately for each species to provide an assessment of the impact of contrast reversal on their face discrimination performance. A 2 × 3 repeated-measures ANOVA for chimpanzees using training (positive versus negative) and test stimuli (positive, negative and colour) as the within-subject factors showed a significant main effect of the test conditions (F2,10 = 67.58, p < 0.001; effect size η2 = 0.93) and a significant interaction between the training and testing conditions (F2,10 = 11.36, p < 0.003; effect size η2 = 0.69). The same analysis for the rhesus monkeys showed a significant main effect of the test condition only (F2,8 = 15.16, p < 0.002; effect size η2 = 0.79). Figure 3 illustrates the mean performance (+ s.e.m.) for each species and each trial type. To understand these results, follow-up comparisons were performed using paired t-tests where the adjusted alpha was set at p < 0.017 (0.05/three comparisons). After positive training, chimpanzees showed significantly better performance matching PP-trained trials compared with PN trials (t(5) = 7.16, p < 0.001) and PC trials compared with PN trials (t(5) = 7.59, p < 0.001). However, no difference was observed in performance matching PP-trained trials compared with PC trials (t(5) = 2.53, p = 0.53). Rhesus monkeys also showed significantly better performance matching the PP-trained trials compared with PN trials (t(4) = 5.17, p < 0.007) and no difference in performance matching PP-trained trials to PC trials (t(4) = 3.26, p = 0.03). However, unlike chimpanzees, they showed no significant difference matching PC trials compared with PN trials (t(4) = 2.80, p = 0.049). Therefore, the performance of chimpanzees conformed to each of the predicted hypotheses whereas the rhesus monkeys did not show an advantage for a change in colour compared with a change in polarity, PN trials were matched as well as PC trials.
Figure 3.
Mean performance by (a) chimpanzees and (b) rhesus monkeys matching faces that differed in contrast or colour. The brackets note significant differences, p < 0.017 (corrected).
After the negative training, chimpanzees showed significantly better performance matching NN-trained trials compared with NP trials (t(5) = 5.12, p < 0.004) and NN-trained trials compared with NC trials (t(5) = 5.07, p < 0.004). However, no differences were observed in their performance matching NP trials compared with NC trials (t(5) = 0.68, p = 0.53). Rhesus monkeys were also significantly better matching NN-trained trials compared with NC trials (t(4) = 3.99, p < 0.017) and showed no significant difference matching NP trials to NC trials (t(4) = 0.79, p = 0.47). However, in contrast to chimpanzees, they also showed no significant difference for the contrast-reversed trials, matching NN-trained trials to NP trials (t(4) = 3.18, p = 0.034). Again, the performance of chimpanzees conformed to each of the predicted hypotheses, whereas after training on negative polarity images (NN), the rhesus monkeys failed to show any deficit generalizing to the contrast-reversed images (NP).
To provide further confirmation of the species differences described above, two additional ANOVAs were used to compare the performance between PC versus PN trials and NN versus NP trials, where species was included as a between-subject factor. A significant interaction between trial type and species would confirm the species differences described above. This interaction was significant for the PC versus PN comparison (F1,8 = 7.0, p < 0.03; effect size η2 = 0.47), and chimpanzees performed significantly worse after a reversal of contrast (PN) compared with a reversal of colour (PC), whereas the rhesus monkeys showed no difference. However, the interaction was not significant for the NN versus NP comparison (F1,8 = 0.02, p = 0.9; effect size η2 = 0.002). In the within-species ANOVAs reported above, chimpanzees showed the effect of contrast reversal by performing significantly better on NN compared with NP trials, but this failed to reach significance for the rhesus monkeys after the analysis controlled for multiple stimulus comparisons. Thus, when controlling for the variability within each species's performance owing to stimulus type, the effect of contrast reversal just failed to reach significance for monkeys (p < 0.03 versus p < 0.017); so it is not surprising that the post hoc ANOVA reported here showed no difference between species.
(c). Top-face-part control task
Monkeys required an average of 5.6 (s.e.m. = 2.75, range 1–16) repetitions of the positive training trials (positive top face parts) before their performance exceeded 85 per cent. Chimpanzees required an average of 5.0 (s.e.m. = 1.41, range 1–9) repetitions of the positive training trials (positive top face parts) before their performance exceeded 85 per cent. Trials to reach this training criterion did not differ significantly between the two species (t(8) = 0.19, p = 0.85; table 1). The effects of contrast reversal on face parts compared with whole faces was analysed using a repeated-measures ANOVA for each species where task (whole versus part) and contrast (positive versus negative) were the within-subject factors. For the rhesus monkeys, this revealed a significant main effect of task (F1,4 = 69.03, p < 0.001; effect size η2 = 0.95) and contrast (F1,4 = 56.41, p < 0.002; effect size η2 = 0.93), but no significant interaction between task and contrast (F1,4 = 1.94, p = 0.24; effect size η2 = 0.33). The effect of contrast reversal on the performance of rhesus monkeys matching whole faces was 21.33 per cent, indicating the drop in performance between PP and PN trials. This reduced to 10.67 per cent when matching face parts, although the difference was not significant.
For the chimpanzees, there was a significant main effect of task (F1,4 = 47.02, p < 0.002; effect size η2 = 0.92) and contrast (F1,4 = 37.71, p < 0.004; effect size η2 = 0.90), and a significant interaction between task and contrast (F1,4 = 9.60, p < 0.04; effect size η2 = 0.71). Chimpanzees showed a significant effect of contrast reversal in the face-part control task. Their performance after training to match positive face parts (88%, s.e.m. = 1.33) was significantly better than when these trials were presented in their photographic negative (76.33%, s.e.m. = 4.06; t(4) = 7.21, p < 0.002). However, the effect of contrast reversal was significantly greater for the whole-face task than the parts control task (t(4) = 3.10, p < 0.04). This was reflected by a 23.33 per cent drop in performance in the PP compared with PN trials, which reduced to an 11.67 per cent drop for the face parts.
4. Discussion
Results from this study showed strong effects of contrast reversal on face processing in chimpanzees, implying the importance of surface-based cues in processing face identity. However, the results from rhesus monkeys were weaker than chimpanzees and, at times, inconclusive. Three types of trial were presented after first training subjects to match whole faces in both their positive and negative polarity: the original training images (PP = positive or NN = negative), the training images reversed in contrast (PN or NP) and the training images reversed in colour (PC or NC). The experimental design enabled six possible predictions that would support the importance of surface-based cues. Compared with the original trained trials, performance was expected to decrease in both contrast-reversal conditions (PP > PN and NN > NP). After training with positive images, no differences were expected when presented with colour images (PP = PC), whereas subjects were expected to perform better on the negative-trained images compared with the colour-reversed condition (NN > NC). This is because the NC condition also contained a contrast reversal. After training on the positive images, subjects were expected to perform better on the colour reversal than the contrast reversal (PC > PN), whereas no differences were expected between these conditions after the negative training (NC = NP). Again, this was because both conditions also contained reversal of contrast.
The data from chimpanzees supported all six of these hypotheses, while the data from monkeys supported four. Monkeys' performance deviated from that of chimpanzees and the expected hypotheses in that they showed equivalent performance on the PC compared with PN trials. Therefore, after training with positive images, they showed as much of a deficit when the images changed in colour as when they changed in contrast. This effect was present when the data were analysed for each species, and when species was included in the analysis. The monkeys also failed to show the predicted advantage for the previously trained negative trials compared with the contrast-reversed trials (NN = NP), although this effect only failed to reach significance when the data were adjusted for multiple comparisons. This suggests that they were able to gain some information about individual identity from the contrast-reversed images, despite having previously received training on the negative faces.
Previous studies of contrast reversal in humans have met with criticism because the images presented during the test phase have been different from the images presented during the acquisition phase. Galper [12], for example, presented human subjects with positive images during a learning phase and then examined contrast-reversal effects by asking them to generalize to the negative images, but not vice versa. Thus, the images at test (negative polarity) differed from the way in which they were originally presented during the learning phase (positive polarity). This stimulus mismatch might explain the contrast-reversal effect in terms of a general stimulus change, without the need to invoke a general impairment in face-processing strategies. The present study avoided these problems in two ways. First, subjects were trained on both positive- and negative-contrast faces, and then tested on the same images in their reversed polarity. The effects of contrast reversal are expected in both directions (positive to negative and negative to positive). Second, we also included an important set of comparison trials that required subjects to match images across a change in colour, thus providing a stimulus mismatch between training and test conditions that was not based on image contrast. This helped to control for whether the observed differences in performance were the results of contrast changes specifically, or a stimulus mismatch between training and test conditions generally. There were two types of trial that could examine the impact of a change in contrast directly with a change in colour: PP versus PC and PC versus PN conditions. The performance of both monkeys and chimpanzees conformed to the predicted hypothesis, and there was no significant difference between their ability to match previously trained positive faces across a change of colour (PP = PC). Although the monkeys made twice as many errors (13%) on the PC trials compared with the chimpanzees (5.83%), this did not represent a significant drop in performance for either species. As predicted, the chimpanzees also performed significantly better on the PC compared with PN trials (a difference of 20.56 per cent), suggesting that they were impacted much more by a change in contrast than a change in colour. The monkeys, however, showed no significant difference in performance on this condition (PC = PN).
A control task was also performed to examine whether the impairments owing to contrast reversal operated selectively on face-processing mechanisms, or whether they affected visual processing more generally. As control stimuli, we presented the top part of conspecifics' faces (face parts) so as to match the visual complexity of face stimuli but not recruit the face-processing system. Like humans, the impact of contrast reversal on matching control stimuli (face parts) in chimpanzees and rhesus monkeys was about half that found for matching whole faces [16]. However, only chimpanzees showed a significant interaction between the task condition (whole faces versus face parts) and contrast, indicating that the effects of contrast reversal were significantly greater when chimpanzees matched whole faces compared with face parts.
Historically, studies of individual recognition have focused on the ability of subjects to detect subtle changes in the spacing of facial features, which are unique in every face [32]. However, surface-based cues describe an additional, highly salient source of information about individual identity, including the texture and pigmentation of a face, and the shading information produced from its local features [9,11]. Both pigmentation and shape from shading information relate specifically to the luminance profiles that affect the surface geometry of faces and objects. These luminance profiles provide the contrast information from shadows that is necessary to delineate the borders of an object or face, in addition to the depth information that helps to define its shape [33]. By reversing the contrast of a face, information about the spacing of local features is preserved, while information about surface-based cues is disrupted. In their early study, Bruce & Langton [9] reported that contrast reversal only affected the ability to extract information from pigmented faces. A subject's performance identifying non- pigmented faces was unaffected by negation. Later, Kemp et al. [11] suggested that contrast reversal had a greater impact on the ability to extract shape from shading information compared with pigmentation, although altering both sources of surface-based cues produced the strongest impairments in face identification.
It is still somewhat unclear from human studies how information from the first-order configuration of faces, the spacing of features and surface-based cues become integrated to aid the recognition and memory of specific individuals. Rossion [34] has suggested that holistic processing is responsible for integrating the first-order configuration of facial features and the precise spacing of these features into a single perceptual whole. When holistic processing is impaired, as is the case for inverted faces, subjects show greater impairment in the ability to detect subtle changes in the spacing of features compared with the presence of specific features. Several human studies have suggested that the mechanisms involved in processing surface-based cues may be independent of those involved in processing the spacing of features, and may even be unaffected by the standard manipulations of holistic processing. The effects of inversion, for example, persist for negative faces, and contrast reversal had little or no impact on the performance of subjects on the face-composite task [9,35]. Moreover, subjects were able to detect subtle changes in the spacing of facial features in negative faces [15,36]. One confounding study, however, showed that the Thatcher illusion was less prominent in negative faces compared with positive faces [37]. The Thatcher illusion refers to the grotesque appearance of an upright face in which the eyes and mouth have been inverted. This affects both second-order configural cues in that the spacing of features is altered when the inner features are inverted, and holistic processing in that the Thatcher illusion disappears when faces are inverted. Thus, holistic processing appears to be relatively unaffected by surface-based cues, although both are presumed to be necessary for the accurate representation of face identity.
Finally, surface-based cues reflect both the pigmentation of faces and the shape from shading information produced by shadows unique to the surface geometry of each face. However, it remains unclear how comparable these features are across different species of non-human primates, or the extent to which these cues can be compared with human faces. This raises concerns about whether similar selection pressures for detecting surface-based cues would be operating in species whose faces showed differences in feature shape and pigmentation. The presence of hair, its colour and the pattern of skin pigmentation among primates are highly variable. Chimpanzee faces, for example, are relatively free of hair compared with rhesus monkeys, and rhesus monkeys also have a characteristic darker whorl around their cheek region that shows considerable variability across individuals. Additionally, although all faces contain the same first-order features (eyes, nose and mouth etc.), the size and shape of these features varies considerably across species. Chimpanzees have a large prognathic mouth and heavy brow ridge compared with monkeys or humans. Monkeys and chimpanzees lack everted reddish lips and the fatty cheeks that characterize human faces. Although it is difficult to quantify the differences in the pigmentation and feature morphology of primate faces, it would not be unreasonable to conclude that some of this variation could have played a role in the species differences and underlying evolutionary patterns reported here.
In conclusion, the present study demonstrated that chimpanzees are more sensitive than rhesus monkeys to changes in surface-based cues in a face-matching task. Explanations for performance based on the presentation of different stimulus types during the training and test sessions, general difficulties encoding negative faces or problems with differential memory demands for the contrast-reversal trials cannot adequately explain these species differences. Rather, the data support a weaker role of surface-based cues in the face-matching skills of monkeys compared with chimpanzees and humans. These results are consistent with previous studies reporting quantitatively weaker performance by rhesus monkeys compared with chimpanzees in tasks involving the individuation of faces and sensitivity to second-order configural cues [22,23]. While numerous studies have reported the ability of monkeys to rapidly orient to faces, attend to eye gaze and direction, and show selective neuronal responses to face-like stimuli [26,38–44], there is, at present, no compelling evidence that monkeys share the same sensitivity to second-order configural cues as chimpanzees or humans (see [17] for a review). We should emphasize that this lack of evidence is not evidence that the skill is lacking: there are simply not enough data to make a firm conclusion, and on at least four of the six comparisons reported here, the monkey's performance was comparable to the chimpanzees. However, on two key comparisons (PC versus PN and NN versus NP), the performance of the rhesus monkeys failed to provide strong evidence for the importance of surface-based cues. There is still much to be learned about the evolution of face processing in primates, and whether humans and monkeys encode face identity using similar visual features. Together, these data suggest that surface-based cues may reflect a unique and relatively unexplored dimension of face processing that could shed important light on the evolution of human face-processing abilities.
Acknowledgements
The Yerkes National Primate Research Center is fully accredited by the American Association for Accreditation of Laboratory Animal Care.
This investigation was supported by RR-00165 from the NIH/NCRR to the Yerkes National Primate Research Center, and R01-MH068791 to LA Parr. Thanks to Daniel Brubaker, Erin Siebert and Kim Baldesare for assistance with animal testing and stimulus preparation, the animal care staff at the Yerkes National Primate Research Center and three anonymous reviewers.
Endnote
One chimpanzee passed away after the completion of the whole-face task and therefore could not participate in the control condition. The control condition includes five chimpanzee subjects and five monkey subjects.
References
- 1.Tanaka J. W., Farah M. J. 1993. Parts and wholes in face recognition. Q. J. Exp. Psychol. A 46, 225–245 [DOI] [PubMed] [Google Scholar]
- 2.Rhodes G. 1988. Looking at faces: first-order and second-order features as determinants of facial appearance. Perception 17, 43–63 10.1068/p170043 (doi:10.1068/p170043) [DOI] [PubMed] [Google Scholar]
- 3.Maurer D., Le Grand R., Mondloch C. J. 2002. The many faces of configural processing. Trends Cogn. Sci. 6, 255–260 10.1016/S1364-6613(02)01903-4 (doi:10.1016/S1364-6613(02)01903-4) [DOI] [PubMed] [Google Scholar]
- 4.Yin R. K. 1969. Looking at upside-down faces. J. Exp. Psychol. 81, 141–145 10.1037/h0027474 (doi:10.1037/h0027474) [DOI] [Google Scholar]
- 5.Valentine T. 1988. Upside-down faces: a review of the effects of inversion upon face recognition. Brit. J. Psychol. 79, 471–491 [DOI] [PubMed] [Google Scholar]
- 6.Barton J. J. S., Keenan J. P., Bass T. 2001. Discrimination of spatial relations and features in faces: effects of inversion and viewing duration. Br. J. Psychol. 93, 527–549 10.1348/000712601162329 (doi:10.1348/000712601162329) [DOI] [PubMed] [Google Scholar]
- 7.Freire A., Lee K., Symons L. A. 2000. The face-inversion effect as a deficit in the encoding of configural information: direct evidence. Perception 29, 159–170 10.1068/p3012 (doi:10.1068/p3012) [DOI] [PubMed] [Google Scholar]
- 8.Leder H., Candrian G., Huber O., Bruce V. 2001. Configural features in the context of upright and inverted faces. Perception 30, 73–83 10.1068/p2911 (doi:10.1068/p2911) [DOI] [PubMed] [Google Scholar]
- 9.Bruce V., Langton S. 1994. The use of pigmentation and shading information in recognizing the sex and identities of faces. Perception 23, 803–822 10.1068/p230803 (doi:10.1068/p230803) [DOI] [PubMed] [Google Scholar]
- 10.Johnston A., Hill H., Carman N. 1992. Recognising faces: effects of lighting direction, inversion, and brightness reversal. Perception 21, 365–375 10.1068/p210365 (doi:10.1068/p210365) [DOI] [PubMed] [Google Scholar]
- 11.Kemp R., Pike G., White P., Musselmen A. 1996. Perception and recognition of normal and negative faces: the role of shape from shading and pigmentation cues. Perception 25, 37–52 10.1068/p250037 (doi:10.1068/p250037) [DOI] [PubMed] [Google Scholar]
- 12.Galper R. E. 1970. Recognition of faces in photographic negative. Psychon. Sci. 19, 207–208 [Google Scholar]
- 13.Liu C. H., Chaudhuri A. 1997. Face recognition with multi-tone and two-tone photographic negatives. Perception 26, 1289–1296 10.1068/p261289 (doi:10.1068/p261289) [DOI] [PubMed] [Google Scholar]
- 14.Nederhouser M., Yue X., Mangini M. C., Biederman I. 2007. The deleterious effect of contrast reversal on recognition is unique to faces, not objects. Vis. Res. 47, 2134–2142 10.1016/j.visres.2007.04.007 (doi:10.1016/j.visres.2007.04.007) [DOI] [PubMed] [Google Scholar]
- 15.White M. 2001. Effect of photographic negation on matching the expressions and identities of faces. Perception 30, 969–981 10.1068/p3225 (doi:10.1068/p3225) [DOI] [PubMed] [Google Scholar]
- 16.Voung X., Peissig J., Harrison M., Tarr M. 2005. The role of surface pigmentation for recognition revealed by contrast reversal in faces and greebles. Vis. Res. 45, 1213–1223 10.1016/j.visres.2004.11.015 (doi:10.1016/j.visres.2004.11.015) [DOI] [PubMed] [Google Scholar]
- 17.Parr L. A. 2010. The evolution of face processing in nonhuman primates. Phil. Trans. R. Soc. B. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dahl C. D., Logothetis N. K., Hoffman K. L. 2007. Individuation and holistic processing of faces in rhesus monkeys. Proc. R. Soc. Lond. B 274, 2069–2076 10.1098/rspb.2007.0477 (doi:10.1098/rspb.2007.0477) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gothard K. M., Brooks K. B., Peterson M. A. 2009. Multiple perceptual strategies used by macaque monkeys for face recognition. Anim. Cogn. 12, 155–167 10.1007/s10071-008-0179-7 (doi:10.1007/s10071-008-0179-7) [DOI] [PubMed] [Google Scholar]
- 20.Parr L. A., de Waal F. B. M. 1999. Visual kin recognition in chimpanzees. Nature 399, 647–648 10.1038/21345 (doi:10.1038/21345) [DOI] [PubMed] [Google Scholar]
- 21.Parr L. A., Heintz M., Lonsdorf E., Wroblewski E. In press Visual kin recognition in nonhuman primates: inbreeding avoidance or male distinctiveness? J. Comp. Psychol. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Parr L. A., Heintz M., Pradhan G. 2008. Rhesus monkeys (Macaca mulatta) lack face expertise. J. Comp. Psychol. 122, 390–402 10.1037/0735-7036.122.4.390 (doi:10.1037/0735-7036.122.4.390) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Parr L. A., Winslow J. T., Hopkins W. D., de Waal F. B. M. 2000. Recognizing facial cues: individual recognition in chimpanzees (Pan troglodytes) and rhesus monkeys (Macaca mulatta). J. Comp. Psychol. 114, 47–60 10.1037/0735-7036.114.1.47 (doi:10.1037/0735-7036.114.1.47) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pokorny J. J., de Waal F. B. M. 2009. Face recognition in capuchin monkeys (Cebus apella). J. Comp. Psychol. 123, 151–160 10.1037/a0014073 (doi:10.1037/a0014073) [DOI] [PubMed] [Google Scholar]
- 25.Parr L. A., Heintz M., Akamagwuna U. 2006. Three studies on configural face processing by chimpanzees. Brain Cogn. 62, 30–42 10.1016/j.bandc.2006.03.006 (doi:10.1016/j.bandc.2006.03.006) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sugita Y. 2008. Face perception in monkeys reared with no exposure to faces. Proc. Natl Acad. Sci. USA 105, 394–398 10.1073/pnas.0706079105 (doi:10.1073/pnas.0706079105) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Adachi I., Chou D. P., Hampton R. R. 2009. Thatcher effect in monkeys demonstrates conservation of face perception across primates. Curr. Biol. 19, 1270–1273 10.1016/j.cub.2009.05.067 (doi:10.1016/j.cub.2009.05.067) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Dahl C. D., Logothetis N. K., Bülthoff H. H., Wallraven C. 2010. The Thatcher illusion in humans and monkeys. Proc. R. Soc. B 277, 2973–2981 10.1098/rspb.2010.0438 (doi:10.1098/rspb.2010.0438) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Parron C., Fagot J. 2008. Baboons (Papio papio) spontaneously process the first-order but not the second-order configural properties of faces. Am. J. Primatol. 70, 415–422 10.1002/ajp.20503 (doi:10.1002/ajp.20503) [DOI] [PubMed] [Google Scholar]
- 30.Tomonaga M., Imura T. 2010. Visual search for human gaze direction by a chimpanzee (Pan troglodytes). PLoS ONE 5, e9131. 10.1371/journal.pone.0009131 (doi:10.1371/journal.pone.0009131) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Parron C., Fagot J. 2008. Baboons (Papio papio) spontaneously process the first-order but not second-order configural properties of faces. Am. J. Primatol. 70, 415–422 10.1002/ajp.20503 (doi:10.1002/ajp.20503) [DOI] [PubMed] [Google Scholar]
- 32.Mondloch C. J., Le Grand R., Maurer D. 2002. Configural face processing develops more slowly than featural face processing. Perception 31, 553–566 10.1068/p3339 (doi:10.1068/p3339) [DOI] [PubMed] [Google Scholar]
- 33.Cavanagh P., Leclerc Y. G. 1989. Shape from shadows. J. Exp. Psychol. Human 15, 3–27 10.1037/0096-1523.15.1.3 (doi:10.1037/0096-1523.15.1.3) [DOI] [PubMed] [Google Scholar]
- 34.Rossion B. 2008. Picture-plane inversion leads to qualitative changes of face perception. Acta Psychol. 128, 274–289 10.1016/j.actpsy.2008.02.003 (doi:10.1016/j.actpsy.2008.02.003) [DOI] [PubMed] [Google Scholar]
- 35.Hole G. J., George P. A., Dunsmore V. 1999. Evidence for holistic processing of faces viewed as photographic negatives. Perception 28, 341–359 10.1068/p2622 (doi:10.1068/p2622) [DOI] [PubMed] [Google Scholar]
- 36.Kemp R., McManus C., Pigott T. 1990. Sensitivity to the displacement of facial features in negative and inverted images. Perception 19, 531–543 10.1068/p190531 (doi:10.1068/p190531) [DOI] [PubMed] [Google Scholar]
- 37.Lewis M. B., Johnston R. A. 1997. The Thatcher illusion as a test of configural disruption. Perception 26, 225–227 10.1068/p260225 (doi:10.1068/p260225) [DOI] [PubMed] [Google Scholar]
- 38.Gothard K. M., Battaglia F. P., Erickson C. A., Spitler K. M., Amaral D. G. 2007. Neural responses to face expression and face identity in the monkey amygdala. J. Neurophys. 97, 1671–1683 10.1152/jn.00714.2006 (doi:10.1152/jn.00714.2006) [DOI] [PubMed] [Google Scholar]
- 39.Hasselmo M. E., Rolls E. T., Baylis G. C. 1989. The role of expression and identity in the face-selective responses of neurons in the temporal visual cortex of the monkey. Biobehav. Brain Res. 32, 203–218 10.1016/S0166-4328(89)80054-3 (doi:10.1016/S0166-4328(89)80054-3) [DOI] [PubMed] [Google Scholar]
- 40.Hattori Y., Kano F., Tomonaga M. 2010. Differential sensitivity to conspecific and allospecific cues in chimpanzees and humans: a comparative eye-tracking study. Biol. Lett. 6, 610–613 10.1098/rsbl.2010.0120 (doi:10.1098/rsbl.2010.0120) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kano F., Tomonaga M. 2009. How chimpanzees look at pictures: a comparative eye-tracking study. Proc. R. Soc. B 276, 1949–1955 10.1098/rspb.2008.1811 (doi:10.1098/rspb.2008.1811) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Parr L. A., Hecht E., Barks S. K., Preuss T. M., Votaw J. R. 2009. Face processing in the chimpanzee brain. Curr. Biol. 19, 50–53 10.1016/j.cub.2008.11.048 (doi:10.1016/j.cub.2008.11.048) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Perrett D. I., Hietanen J. K., Oram M. W., Benson P. J. 1992. Organization and functions of cells responsive to faces in the temporal cortex. Phil. Trans. R. Soc. Lond. B 335, 23–30 10.1098/rstb.1992.0003 (doi:10.1098/rstb.1992.0003) [DOI] [PubMed] [Google Scholar]
- 44.Pinsk M. A., DeSimone K., Moore T., Gross C. G., Kastner S. 2005. Representations of faces and body parts in macaque temporal cortex: a functional MRI study. Proc. Natl Acad. Sci. USA 102, 6996–7001 10.1073/pnas.0502605102 (doi:10.1073/pnas.0502605102) [DOI] [PMC free article] [PubMed] [Google Scholar]