Abstract
Objective
To examine the effects of human leukocyte antigen (HLA) alleles on HIV-1-related disease progression and central nervous system (CNS) impairment in children.
Design
572 HIV-1-infected children, identified as disease progressors or non-progressors, were selected from PACTG P152 and P300 through a case-cohort sampling scheme. Study endpoints were HIV-1-related disease progression-free survival and time to CNS impairment.
Methods
DNA was genotyped for HLA alleles using a Luminex 100 platform. Weighted Kaplan-Meier methods and Cox proportional hazards models were used to assess the effects of HLA alleles on study endpoints.
Results
Presence of the B-27 allele (n=20) was associated with complete protection against disease progression and CNS impairment over the median follow-up of 26 months (P<0.0001 for both). These findings held in multivariate analyses controlling for baseline covariates including race, gender, age, log HIV-1 RNA, CD4+ lymphocyte count and percent, weight for age z-score and treatment, and for other genotypes shown to affect HIV-1-related disease progression. Also, while the Cw-2 allele protected against disease progression (HR, 0.48; 95% CI: 0.28–0.81; P= 0.006), the A-24 allele was associated with more rapid CNS impairment (HR: 2.01; 95% CI: 1.04–3.88; P= 0.04). The HLA class II DQB1-2 allele was associated with a delayed disease progression (HR: 0.66; 95% CI: 0.47–0.92; P= 0.01) and CNS impairment (HR: 0.58; 95% CI: 0.36–0.93; P= 0.02).
Conclusions
Presence of B-27, Cw-2, or DQB1-2 alleles was associated with delayed HIV-1 disease progression, while B-27, A-24, and DQB1-2 alleles were associated with altered progression to CNS impairment in children.
Keywords: HLA alleles, Children, HIV-1-related disease, CNS impairment
INTRODUCTION
The human leukocyte antigen (HLA) system, which is also designated as the major histocompatibility complex (MHC), is located in a hypervariable region of human chromosome 6p21.1 There are approximately 128 expressed genes in the HLA system, most of which have not been found to be involved in the immune response.2,3 The ones which function in immunity are called classical HLA genes and are classified into two categories based on their structure and function3: class I (HLA-A, -B, -C, -E, -F, and -G) and class II (HLA-DR, -DQ, -DM, and -DP) genes.1 The class I and class II HLA molecules bind with peptides derived from endogenous and exogenous pathogens, respectively. The peptide-laden HLA molecules are then delivered to the surface of the infected cells.3 Class I HLA-peptide complexes can be recognized by CD8+ cytotoxic T lymphocytes (CTLs) and thus the infected cells can be lysed,3–6 whereas class II HLA-peptide complexes engage CD4+ T helper cells, which will produce important cytokines that help coordinate the immune response.4,6,8
HLA genes play a central role in susceptibility to many autoimmune and infectious diseases.1,7 In particular, several studies have shown a positive genetic association between HLA alleles and various aspects of HIV-1 infection and related opportunistic infections including hepatitis B and C, tuberculosis, and malaria.1,8 Among the HLA alleles, B-27 and B-57 have been shown to be associated with HIV-1-related disease progression and viral replication, respectively.9–15 However, all these studies have been performed in cohorts of adults. Since children have an immature and developing immune system that may respond to HLA differently from adults, the current research examined the associations between HLA class I or II alleles and HIV-1-related disease progression in children. Our findings demonstrate several significant associations of specific HLA alleles with HIV-1-related disease progression and central nervous system (CNS) impairment. Most notably, presence of the B-27 allele is strongly protective against HIV-1- related disease progression in children.
SUBJECTS AND METHODS
Subjects
The 572 children with evaluable HLA results screened for HLA class I and class II alleles in the present study were selected from Pediatric AIDS Clinical Trials Group (PACTG) protocols P15216 and P30017. P152 was a Phase III, randomized, double-blind trial in which the effectiveness of three antiretroviral treatment regimens (zidovudine (ZDV) monotherapy, didanosine (ddI) monotherapy, and ZDV plus ddI combination therapy) was compared in HIV-1-infected children aged 3 months to 18 years. P300 was a Phase II/III comparative, multicenter trial to compare the effects of ZDV plus lamivudine combination therapy versus ddI monotherapy versus ZDV plus ddI combination therapy with respect to disease progression in symptomatic HIV-1-infected infants and children aged 42 days to 15 years. The primary endpoints of the present study included progression free survival (PFS), which was defined as either time to progression to first clinical HIV-1-related disease endpoint or death, and time to CNS impairment (CNS free survival), which was defined as time to deterioration in brain growth, psychological function, and/or neurological status.
HLA typing
Total genomic DNA was extracted from peripheral blood mononuclear cells using mini blood DNA kits (QIAGEN, Carlsbad, CA). Whole Genome Amplification of DNA was done using QIAGEN WGA kits.18 HLA genotyping was done by multiplexing using Luminex 100 platform (Luminex Corp, Austin, TX) at Tepnel Lifecodes Corporation (Stamford, CT).
Design
A case-cohort design19 was used for the sampling of the study subjects. From a total of 1559 subjects enrolled, available study participants of P152/P300 (N=901) who had a defined disease progression status (i.e. progressors or non-progressors), a sample size of 600 subjects were selected for HLA genotyping. All cases defined as progressor (n=229) were included in the sample, while a sample of 371 subjects stratified by treatment, were randomly selected from the non-progressor group for genotyping. In the end, 572 patients (206 progressors and 366 non-progressors) with complete HLA genotyping results available were included in the analyses.
Statistical Analyses
In order to reweight contributions in the case-cohort sample to represent the contributions from the full cohort, a sampling weight for each subject in the case-cohort sample was defined by the number of subjects in the overall P152/P300 cohorts for the respective disease groups (progressor and non-progressor) divided by the corresponding number in the case-cohort sample. Thus, the weight for the progressor group was 1 since all cases from the cohorts were included, while the non-progressor group was assigned a larger weight of 1.81 ([901-229]/[600-229]) since the sampled subjects in that group were under-represented relative to the full cohorts. In the weighted analyses, contributions to estimators and other quantities, such as partial likelihoods, were multiplied by these weights. If the patients included in the case-cohort sample were a random subset of the corresponding group from P152 and P300, then the weighted estimators would provide consistent estimates of the corresponding quantities from the full P152 and P300 cohort. The weighted partial likelihood computed was used for estimating hazard ratios and testing effects. This essentially provided the weighted pseudo-likelihood estimators.20 The variance of the partial likelihood estimators was assessed with the robust variance.21, 22
Analyses of the association between HLA allele markers and the time to progression free survival (PFS) and CNS impairment were performed using weighted Kaplan Meier estimates (Wald statistic p-values with robust variance estimate were reported.). Cox proportional hazards models with weighted estimates were used to investigate the effects of the allele specific markers on PFS and CNS endpoints in univariate and multivariate analyses controlling for baseline covariates including age, gender, race, CD4+ lymphocyte count and percent, change of CD4+ lymphocyte count from baseline to week 24 and 48, log HIV-1 RNA, change of log HIV-1 RNA from baseline to week 24 and 48 on study, and weight for age z-score (WAZ score), as well as other genetic markers identified to significantly affect PFS or CNS impairment in this cohort of children (CR5-wt/Δ32, CX3CR1-249-V/I, -280-T/M, SDF-1–180-G/A, and MBL2-A/O for PFS and -280-T/M, SDF-1–180-G/A, and MBL2-P/Q for CNS impairment). Hazard ratios (HR) of time to PFS or CNS events between the two comparison groups defined by HLA allele markers and their 95% confidence intervals (CIs) are also presented.
RESULTS
Baseline characteristics of studied children
The children evaluated were 58% non-Hispanic Black, 28% Hispanic, 12% non-Hispanic White, and 2% other or unknown race/ethnicity (Table 1). Ages ranged from 72 days to 17 years; 54% were females. Of the 572 subjects for whom complete data were available, 206 were progressors and 366 were non-progressors. Twenty-one percent of study participants were followed for <12 months, 39% for ≥12–23 months, 27% for >23–35 months and 13% for ≥35 months. Weight growth failure and single CNS event were the leading causes of disease progression. At study entry, progressors had significantly higher log10 RNA copies/ml and lower CD4+ lymphocyte counts and percentages, WAZ scores, and cognitive scores (Table 1). The median follow-up time was 26.0 months for the 572 subjects included in the analysis.
Table 1.
Baseline characteristics by disease progression status.
| Characteristic | Disease Progression Status | ||||
|---|---|---|---|---|---|
| Total (N=572) | Non-Progressor (N=366) | Progressor (N=206) | P-Value | ||
| Gender | Male | 265 (46%) | 171 (47%) | 94 (46%) | 0.861* |
| Female | 307 (54%) | 195 (53%) | 112 (54%) | ||
| Race/Ethnicity | Black | 333 (58%) | 219 (60%) | 114 (55%) | 0.105* |
| Hispanic | 160 (28%) | 90 (25%) | 70 (34%) | ||
| White | 68 (12%) | 49 (13%) | 19 (9%) | ||
| Asian/Pacific Islander | 1 (0%) | 1 (0%) | 0 (0%) | ||
| unknown | 10 (2%) | 7 (2%) | 3 (1%) | ||
| Follow-up time | <12 months | 119 (21%) | 18 (5%) | 101 (49%) | <.001* |
| >=12–23 months | 222 (39%) | 150 (41%) | 72 (35%) | ||
| >=23–35 months | 154 (27%) | 129 (35%) | 25 (12%) | ||
| >=35 months | 77 (13%) | 69 (19%) | 8 (4%) | ||
| PFS events | No | 366 (64%) | 366 (100%) | 0 (0%) | <.001* |
| Yes | 206 (36%) | 0 (0%) | 206 (100%) | ||
| Disease progressions | No event | 366 (64%) | 366 (100%) | 0 (0%) | |
| Death | 24 (4%) | 0 (0%) | 24 (12%) | ||
| >=2 opportunistic infections | 7 (1%) | 0 (0%) | 7 (3%) | ||
| Weight growth failure | 65 (11%) | 0 (0%) | 65 (32%) | ||
| Malignancy | 1 (0%) | 0 (0%) | 1 (0%) | ||
| CDC clinical Dx Category C | 9 (2%) | 0 (0%) | 9 (4%) | ||
| Single CNS Event | 66 (12%) | 0 (0%) | 66 (32%) | ||
| Single CNS Event + other event | 13 (2%) | 0 (0%) | 13 (6%) | ||
| CNS deterioration | 21 (4%) | 0 (0%) | 21 (10%) | ||
| Age (years) at randomization | N | 572 | 366 | 206 | <.001** |
| Median | 1.84 | 2.53 | 0.98 | ||
| Baseline CD4 percent | N | 564 | 360 | 204 | <.001** |
| Median | 24.49 | 26.89 | 21 | ||
| Baseline CD4 count/mm^3 | N | 564 | 360 | 204 | 0.004** |
| Median | 824.50 | 900.50 | 575.68 | ||
| Baseline log10 RNA copies/ml | N | 447 | 289 | 158 | <.001** |
| Median | 5.25 | 4.98 | 5.89 | ||
| Baseline cognitive score | N | 532 | 345 | 187 | 0.001** |
| Median | 83 | 84 | 78 | ||
| Baseline WAZ score | N | 572 | 366 | 206 | <.001** |
| Median | −0.83 | −0.47 | −1.42 | ||
Fisher’s Exact Test
T-Test with Unequal Variance
Associations of homozygosity or heterozygosity of HLA alleles with HIV-1-related progression-free survival and CNS impairment
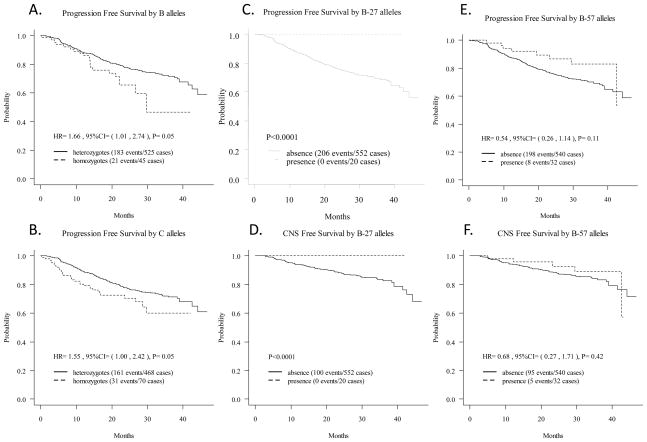
Among the HLA class I alleles, presence of homozygous class I B alleles (HR [homozygotes vs. heterozygotes]: 1.66; p= 0.05) (Table 2, Figure 1A) was associated with a more rapid disease progression. This trend, however, lost significance after adjusting for baseline covariates and for other genotypes. Presence of homozygous C alleles (HR [homozygotes vs. heterozygotes]: 1.55; p= 0.05) (Table 2, Figure 1B) was associated with a more rapid disease progression. This effect remained significant after adjusting for other genotypes (HR, [homozygotes vs. heterozygotes]: 1.63; p= 0.04) (Table 2) but lost significance after adjusting for baseline covariates. Among the HLA class II alleles, presence of the homozygous DRB1 allele showed a trend toward a more rapid disease progression (HR [homozygotes vs. heterozygotes]: 1.48; p= 0.08) (Table 2). However, the effect of the homozygous DRB1 allele lost significance after adjusting for baseline covariates and other genotypes (Table 2). When comparing the effects of homoygosity vs. heteroygosity of HLA alleles on CNS impairment, only presence of class II DRB1 homozygous allele was found to be marginally associated with higher CNS impairment (HR [homozygotes vs. heterozygotes]: 1.67; p= 0.09) (Table 3). This association became stronger after adjusting for other genotypes (HR [homozygotes vs. heterozygotes]: 1.82; p= 0.05) and was lost after adjusting for baseline covariates (Table 3).
Table 2.
Unadjusted and adjusted hazards ratios for progression-free survival (PFS) by HLA alleles.
| Alleles | Comparisons | Unadjusted HR (95%CI) | p-value | Adjusteda HR (95%CI) | p-value | Adjustedb HR (95%CI) | p-value |
|---|---|---|---|---|---|---|---|
| A | homozygotes vs. heterozygotes | 0.82 (0.44,1.51) | 0.52 | 2.23 (0.77,6.48) | 0.14 | 0.86 (0.46,1.62) | 0.64 |
| B | homozygotes vs. heterozygotes | 1.66 (1.01,2.74) | 0.05 | 1.63 (0.62,4.34) | 0.32 | 1.55 (0.90,2.66) | 0.11 |
| C | homozygotes vs. heterozygotes | 1.55 (1.00,2.42) | 0.05 | 0.80 (0.35,1.84) | 0.6 | 1.63 (1.02,2.60) | 0.04 |
| DRB1 | homozygotes vs. heterozygotes | 1.48 (0.95,2.29) | 0.08 | 1.60 (0.77,3.30) | 0.2 | 1.46 (0.92,2.31) | 0.11 |
| B-27 | presence vs. absence | - | *<0.0001 | - | - | - | - |
| B-57 | presence vs. absence | 0.54 (0.26,1.14) | 0.11 | 0.74 (0.15,3.73) | 0.72 | 0.51 (0.23,1.14) | 0.1 |
| Cw-2 | presence vs. absence | 0.48 (0.28,0.81) | 0.006 | 1.16 (0.48,2.78) | 0.75 | 0.46 (0.27,0.79) | <0.0001 |
| Cw-6 | presence vs. absence | 1.43 (0.99,2.07) | 0.06 | 1.43 (0.59,3.49) | 0.43 | 1.41 (0.96,2.06) | 0.08 |
| DQB1-2 | presence vs. absence | 0.66 (0.47,0.92) | 0.01 | 0.57 (0.32,1.03) | 0.06 | 0.67 (0.48,0.94) | 0.02 |
Adjusted for baseline covariates: race, gender, age, baseline log HIV-1 RNA, change of log HIV-1 RNA from baseline to week 24 and 48, baseline CD4+ lymphocyte count and percent, change of CD4+ lymphocyte count from baseline to week 24 and 48, Baseline WAZ Score and treatment.
Adjusted for other genotypes: CCR5-wt/Δ32, CX3CR1-249-V/I, -280-T/M, SDF-1-180-G/A, and MBL2-A/O.
The p-value is from the robust score test of the Cox model in order to account for the sampling weight in the variance estimation. Since none of the subjects possessing the B-27 allele was observed to have a HIV-1-related disease progression endpoint during follow-up, no HR and 95% CI estimates can be calculated.
Figure 1.
Association of HLA Class I B or C allele heterozygosity; B-27, and B-57 alleles with PFS and CNS Free Survival.
Table 3.
Unadjusted and Adjusted Hazards Ratios for CNS endpoint by HLA Alleles.
| Alleles | Comparisons | Unadjusted HR (95%CI) | p-value | Adjusteda HR (95%CI) | p-value | Adjusted HR (95%CI) | p-value |
|---|---|---|---|---|---|---|---|
| A | homozygotes vs. heterozygotes | 0.90 (0.40,2.00) | 0.79 | 2.88 (0.53,15.50) | 0.22 | 0.99 (0.44,2.22) | 0.98 |
| B | homozygotes vs. heterozygotes | 1.33 (0.63,2.81) | 0.45 | 1.64 (0.49,5.44) | 0.42 | 1.35 (0.63,2.91) | 0.44 |
| C | homozygotes vs. heterozygotes | 1.25 (0.66,2.39) | 0.5 | 0.83 (0.28,2.45) | 0.73 | 1.34 (0.70,2.58) | 0.38 |
| DRB1 | homozygotes vs. heterozygotes | 1.67 (0.93,3.01) | 0.09 | 1.77 (0.68,4.58) | 0.24 | 1.82 (1.00,3.29) | 0.05 |
| A-1 | presence vs. absence | 1.59 (0.93,2.72) | 0.09 | 1.03 (0.32,3.31) | 0.97 | 1.66 (0.96,2.86) | 0.07 |
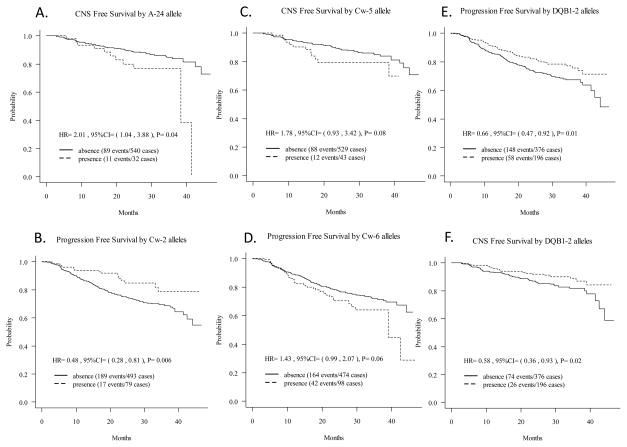
| A-24 | presence vs. absence | 2.01 (1.04,3.88) | 0.04 | 2.84 (0.76,10.64) | 0.12 | 2.04 (1.07,3.89) | 0.03 |
| B-27 | presence vs. absence | - | *<0.0001 | - | - | - | - |
| B-57 | presence vs. absence | 0.68 (0.27,1.71) | 0.42 | 0.35 (0.04,2.79) | 0.32 | 0.62 (0.23,1.66) | 0.34 |
| Cw-5 | presence vs. absence | 1.78 (0.93,3.42) | 0.08 | 4.76 (1.52,14.90) | 0.01 | 1.96 (1.01,3.79) | 0.05 |
| DQB1-2 | presence vs. absence | 0.58 (0.36,0.93) | 0.02 | 0.53 (0.21,1.34) | 0.18 | 0.61 (0.37,0.99) | 0.04 |
Adjusted for baseline covariates: race, gender, age, baseline log HIV-1 RNA, change of log HIV-1 RNA from baseline to week 24 and 48, baseline CD4+ lymphocyte count and percent, change of CD4+ lymphocyte count from baseline to week 24 and 48, Baseline WAZ Score and treatment.
Adjusted for other genotypes: CX3CR1-280-T/M, SDF-1-180-G/A, and MBL2-P/Q
The p-value is from the robust score test of the Cox model in order to account for the sampling weight in the variance estimation. Since none of the subjects possessing the B-27 allele was observed to have a CNS endpoint during follow-up, no HR and 95% CI estimates can be calculated.
Associations of HLA class I alleles with HIV-1-related progression-free survival and CNS impairment
Strikingly, among the HLA class I alleles, presence of the B-27 allele provided complete protection against HIV-related disease progression as none of the 20 children possessing the B-27 allele experienced progression of HIV-1-related disease (robust score test, p <0.0001) (Figure 1C) or CNS impairment (robust score test, p <0.0001) (Figure 1D) during a median follow-up time of 26 months. Presence of HLA class I B-57 allele, which has been found to be associated with protection against progression to AIDS in adults, did not show significant protection against disease progression (p=0.11) (Table 2, Figure 1E) or CNS impairment (p=0.42) (Table 3, Figure 1F) in children. Presence of the A-24 allele was associated with a more rapid HIV-1-related CNS disease progression (HR [presence vs. absence]: 2.01; p= 0.04) (Table 3, Figure 2A); this remained significant after adjusting for other genotypes (HR [presence vs. absence]: 2.04; p= 0.03) (Table 3). However, presence of the A-24 allele did not show any significant effect on the rate of the overall disease progression.
Figure 2.
Association of HLA Class I A-24, Cw-2, Cw-5, Cw-6, and HLA Class II DQB1-2 alleles with PFS and CNS Free Survival.
Among the HLA class I C alleles, presence of Cw-2 was significantly associated with protection against HIV-1-related disease progression (HR [presence vs. absence]: 0.48; p= 0.006) (Table 2, Figure 2B) which remained significant after controlling for other genotypes (HR [presence vs. absence]: 0.46; p <0.0001); however, no significant association was observed with CNS impairment. Additionally, presence of the Cw-5 allele showed a marginal association with progression of CNS disease (HR [presence vs. absence]: 1.78; p= 0.08) (Table 3, Figure 2C), and the results remained consistent after adjusting for baseline characteristics (HR [presence vs. absence]: 4.76; p= 0.01) and for other genotypes (HR [presence vs. absence]: 1.96; p= 0.05) (Table 3). Presence of the Cw-6 allele also showed a marginal association with HIV-1-related disease progression (HR [presence vs. absence]: 1.43; p= 0.06) (Table 3, Figure 2D), and the results remained consistent after adjusting for other genotypes (HR [presence vs. absence]: 1.41; p= 0.08). This effect of the Cw-6 allele was lost, however, when adjusted for baseline covariates.
Associations of HLA class II alleles with HIV-1-related progression-free survival and CNS impairment
Presence of the HLA class II DQB1-2 allele was protective against HIV-1-related disease progression (HR [presence vs. absence]: 0.66; p= 0.01) (Table 2, Figure 2E). This effect remained significant after adjusting for other genotypes (HR [presence vs. absence]:0.67; p= 0.02), but became marginal after adjusting for baseline covariates (HR [presence vs. absence]: 0.57; p= 0.06). Presence of the HLA class II DQB1-2 allele was also protective against HIV-1-related CNS impairment (HR [presence vs. absence]:0.58; p= 0.02) (Table 3, Figure 2F), which remained significant after adjusting for other genotypes (HR [presence vs. absence]: 0.61; p= 0.04), but lost significance after adjusting for baseline covariates.
Associations of B-27 allele with changes of CD4+ lymphocyte count and log HIV-1 RNA viral load overtime
No association was observed between the HLA B-27 allele and changes in viral load and absolute CD4 count from baseline to 24 and 48 weeks. Additionally, protective effects of the B-27 allele against disease progression or CNS impairment remained when adjusted for changes in CD4+ lymphocyte count and log RNA over the follow-up period in a multivariate Cox proportional hazards model (data not shown).
DISCUSSION
Although specific HLA alleles have been found in previous studies to be related to HIV-1-related disease progression in adults, there is little information on the association between HLA variants and HIV-1-related disease progression in children. The data presented here identified several novel associations in a large cohort of children with HLA genotyped from the pre-HAART era. The most striking finding was the complete protection from disease progression by presence of the B-27 allele. Another surprising finding, however, was the lack of significant protective association with presence of the B-57 allele which has been found in previous adult studies to be strongly associated with protection against HIV-1-related disease progression. In addition to overall disease progression, we were able to examine the association of HLA alleles with CNS impairment and identified that presence of the B27, A-24 and DQB1-2 alleles was associated with more rapid progression to CNS impairment. These effects remained significant after adjusting for CD4 lymphocyte counts, viral load and other genotypes known to affect HIV-1-related disease.
It was also found that heterozygosity for HLA class I B and C alleles was associated with delayed progression of HIV-1-related disease in children. These findings are in agreement with those of previous studies performed in HIV-1-infected adults.23 Additionally, the results provide additional support for the hypothesis of over dominant selection (heterozygote advantage) which proposes that the heterozygous HLA loci encode a wider range of HLA molecules which can bind to a greater array of peptides derived from intracellular and extracellular pathogens and trigger the downstream immune response. In contrast, the homozygous loci encode a much narrower range of HLA molecules and thus immune escape often occurs.24, 25 HIV-1 is characterized by its remarkable antigenic variation and rapid mutations, which require a more diverse immune response.
Presence of the B-27 allele was found to provide a strong protective benefit against HIV-1-related disease progression in children. Our findings are consistent with those of a number of adult studies which showed a significant association between presence of the B-27 allele and delayed HIV-1-related disease progression.10–13,26,27 However, we did not observe any significant association of B-27 allele with change in CD4 lymphocyte count and viral load when followed up from baseline to week 24 and 48 period. Presence of the B-27 molecule protects HIV-1-infected individuals against disease progression by binding to a conserved HIV-1 epitope in p24 gag, which resides on the capsid of the HIV-1 virus, and then activating the HLA-B27-restricted cytotoxic T lymphocyte (CTL) responses.8–9
In our study, we didn’t see any significant difference in HIV-1 disease progression conferred by the presence/absence of the B-57 allele, although a trend toward protection against disease progression and CNS impairment was observed. However, two previous studies15, 27 have shown that the frequency of the B-57 allele was dramatically higher in non-progressors than in progressors. Another recent study26 also showed that the B-57 allele was associated with disease progression delay. Other research has demonstrated that B-57 could bind to multiple HIV peptides and trigger a strong B-57-restricted CTL immune response.28–30 A recent study hypothesized that HLA-B-57 provides protection to haploidentical infants by driving and maintaining a fitness-attenuating mutation in p24-Gag.31 It is of interest, therefore, that in our cohort of children, presence of the B-57 allele failed to be associated with altered HIV-1-related disease progression. Reasons for our findings are unclear. However, because HLA types for the mothers were unavailable in this study, we were unable to assess any potential beneficial effects that might be present when mother-infant pairs are haploidentical at HLA-B-57. Additionally, NK cells from individuals with KIR3DL1 (killer Ig-like receptor 3DL1) and HLA-B-57 genotypes have been found to have increased functional potential, particularly in the KIR3DL1 (+) NK cell subset.32 Therefore, knowledge of the KIR genotype in our patients might provide additional insights into the associations of HLA-B-57 with HIV-1-related disease progression and CNS impairment.
In our current study, presence of the Cw-2 allele was associated with slower HIV-1-related disease progression, while presence of the Cw-5 allele was associated with more rapid CNS impairment. The mechanisms underlying these associations are not clear; however, similar to the B-27 allele,8–9 it is likely that these alleles place structural or functional constraints on the virus that alters its ability to generate CTL escape mutants.
In previous studies, it has been demonstrated that A-11, B-27, B-51, B-57, B-58, Cw-2, and Cw-14 alleles were associated with delayed HIV-1-related disease progression, while B-35, B-53, and Cw-4 alleles were associated with more rapid disease progression.33 However, many of these alleles such as A-1, A-23, B-35, B-53, and Cw-4 were not found to significantly alter the HIV-1-related disease progression in our cohort of children. The reasons for these differences are unknown. However, we have also observed differences in association studies conducted in children and adults for other host genetic allelic variants for HIV-1-related disease progression.34–36 It is likely that the developing immune system of children is at least partly responsible for these observed differences. Additionally, genetic backgrounds in the populations studied, as well as virus clade differences among the U.S. and African cohorts, may account for some of the differences observed.
Among the class II HLA alleles, presence of the DQB1-2 allele was associated with protection against HIV-1-related disease progression and CNS impairment in our cohort of children. Although several DQB1 alleles and DQ haplotypes have been shown to be associated with resistance to HIV-1 infection,34 it is the first time to our knowledge that the DQB1-2 allele has been reported to be associated with delayed disease progression. The associations of DQ alleles and haplotypes with resistance and susceptibility to HIV-137 emphasize the importance of the DQ alleles in anti-HIV-1 immunity.
Another novel finding from our study is the association of B-27 and DQB1-2 alleles with the progression of HIV-1-related CNS impairment in addition to the disease progression. Although there have been no previous reports on the association between HLA alleles and HIV-1-related CNS impairment, our finding that such associations were similar to the associations between HLA alleles and HIV-1-related disease progression implies that HLA may affect HIV-1-related disease progression and CNS impairment by a common mechanism. We identified two new HLA class I alleles (A-24, Cw-5) which were associated with CNS impairment, but not with the overall HIV-1-related disease progression. We also found that presence of the DRB1–3-DQB1–6 allele was marginally associated with more rapid disease progression and significantly associated with CNS impairment. These findings require further validation in additional cohorts.
There are a number of limitations to our study. First, the children did receive one to two antiretroviral drugs. However, these treatments were prior to effective antiretroviral therapy and disease progression was common. Additionally, when we controlled for treatment, no changes in statistical significance of the tests for any of the associations were observed. In this study, we specifically examined associations between HLA genotypes of HIV-1-infected children and time to disease progression without regard to maternal HLA genotypes. Although maternal HLA genotypes may provide useful information, the data presented here can be applied to infected children without regard to their mother’s HLA type or mother-child HLA concordance.
This study has a number of major advantages over previous studies on roles of HLA alleles in HIV-1-infected individuals. First, this is the largest cohort of children from the U.S. in the pre-HAART era evaluated for HLA associations with time to HIV-1-related disease progression. Second, children participating in this study had comprehensive evaluations for clinical endpoints including clinical disease progression and CNS impairment. Third, because we have previously evaluated a large number of host genetic variants in this cohort of children, we were able to assess the effects of HLA genotypes controlling for other specific allelic variants which were previously identified as being associated with disease progression and/or CNS impairment.
In summary, it was found that presence of B-27, Cw-2, or DQB1-2 alleles was associated with delayed HIV-1 disease progression in HIV-1 infected children, while B-27, A-24 and DQB1-2 alleles were found to significantly alter the progression to CNS impairment. These associations remained for the majority of the alleles in multivariate analyses controlling for baseline covariates including age, gender, race, CD4+ lymphocyte count and percent, change of CD4+ lymphocyte count and change of log HIV-1 RNA from baseline to week 24 and 48, treatments, and WAZ score, as well as other genetic markers identified to significantly affect PFS or CNS impairment in this cohort of children. These findings suggest that specific HLA alleles are critical to elicit an effective immune response to HIV-1 infection in children.
Acknowledgments
Sources of Grant Support: Supported by grants from the National Institute of Allergy and Infectious Diseases, the International Maternal Pediatric Adolescent AIDS Clinical Trials (IMPAACT) Network (AI-069536) and 5R01MH085608.
Footnotes
Presented in part: XVI Conference on Retroviruses and Opportunistic Infections, Montreal, Canada, February 8-11, 2009 (poster presentation no. 893).
Informed consent was obtained from study participants. This study followed the human experimentation guidelines of the US Department of Health and Human Services and the UCSD review board.
References
- 1.Blackwell JM, Jamieson SE, Burgner D. HLA and infectious diseases. Clin Microbiol Rev. 2009;22:370–385. doi: 10.1128/CMR.00048-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Aguado B, Bahram S, Beck S, et al. Complete sequence and gene map of a human major histocompatibility complex. The MHC sequencing consortium. Nature. 1999;401:921–923. doi: 10.1038/44853. [DOI] [PubMed] [Google Scholar]
- 3.Klein J, Sato A. The HLA system. First of two parts. N Engl J Med. 2000;343:702–709. doi: 10.1056/NEJM200009073431006. [DOI] [PubMed] [Google Scholar]
- 4.Gandhi RT, Walker BD. Immunologic control of HIV-1. Annu Rev Med. 2002;53:149–172. doi: 10.1146/annurev.med.53.082901.104011. [DOI] [PubMed] [Google Scholar]
- 5.McMichael AJ, Rowland-Jones SL. Cellular immune responses to HIV. Nature. 2001;410:980–987. doi: 10.1038/35073658. [DOI] [PubMed] [Google Scholar]
- 6.Norris PJ, Rosenberg ES. CD4(+) T helper cells and the role they play in viral control. J Mol Med. 2002;80:397–405. doi: 10.1007/s00109-002-0337-3. [DOI] [PubMed] [Google Scholar]
- 7.Klein J, Sato A. The HLA system. Second of two parts. N Engl J Med. 2000;343:782–786. doi: 10.1056/NEJM200009143431106. [DOI] [PubMed] [Google Scholar]
- 8.Carrington M, O’Brien SJ. The influence of HLA genotype on AIDS. Annu Rev Med. 2003;54:535–551. doi: 10.1146/annurev.med.54.101601.152346. [DOI] [PubMed] [Google Scholar]
- 9.Schneidewind A, Brockman MA, Sidney J, et al. Structural and functional constraints limit options for cytotoxic T-lymphocyte escape in the immunodominant HLA-B27-restricted epitope in human immunodeficiency virus type 1 capsid. J Virol. 2008;82:5594–5605. doi: 10.1128/JVI.02356-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kaslow R, Carrington M, Apple R, et al. Influence of combinations of human major histocompatibility complex genes on the course of HIV-1 infection. Nat Med. 1996;2:405–411. doi: 10.1038/nm0496-405. [DOI] [PubMed] [Google Scholar]
- 11.McNeil A, Yap P, Gore S, et al. Association of HLA types A1-B8-DR3 and B27 with rapid and slow progression of HIV disease. Q J Med. 1996;89:177–185. doi: 10.1093/qjmed/89.3.177. [DOI] [PubMed] [Google Scholar]
- 12.Magierowska M, Theodorou I, Debré P, et al. Combined genotypes of CCR5, CCR2, SDF1 and HLA genes can predict the long-term non-progressor status in HIV-1-Infected individuals. Blood. 1999;93:936–941. [PubMed] [Google Scholar]
- 13.Easterbrook P. Long-term non-progression in HIV infection: definitions and epidemiological issues. J Infect. 1999;38:71–73. doi: 10.1016/s0163-4453(99)90071-3. [DOI] [PubMed] [Google Scholar]
- 14.Mathieu A, Paladini F, Vacca A, et al. The interplay between the geographic distribution of HLA-B27 alleles and their role in infectious and autoimmune diseases: a unifying hypothesis. Autoimmun Rev. 2009;8:420–425. doi: 10.1016/j.autrev.2009.01.003. [DOI] [PubMed] [Google Scholar]
- 15.Migueles SA, Sabbaghian MS, Shupert WL, et al. HLA B*5701 is highly associated with restriction of virus replication in a subgroup of HIV-infected long term nonprogressors. Proc Natl Acad Sci USA. 2000;97:2709–2714. doi: 10.1073/pnas.050567397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Englund JA, Baker CJ, Raskino C, et al. Zidovudine, didanosine, or both as the initial treatment for symptomatic HIV-infected children. AIDS Clinical Trials Group (ACTG) Study 152 Team. N Engl J Med. 1997;336:1704–1712. doi: 10.1056/NEJM199706123362403. [DOI] [PubMed] [Google Scholar]
- 17.McKinney RE, Jr, Johnson GM, Stanley K, et al. A randomized study of combined zidovudine-lamivudine versus didanosine monotherapy in children with symptomatic therapy-naive HIV-1 infection. The Pediatric AIDS Clinical Trials Group Protocol 300 Study Team. J Pediatr. 1998;133:500–508. doi: 10.1016/s0022-3476(98)70057-5. [DOI] [PubMed] [Google Scholar]
- 18.Singh KK, Spector SA. Fidelity of whole-genome amplification of blood spot DNA for HLA typing and SNP analyses. Clin Genet. 2007;72:156–159. doi: 10.1111/j.1399-0004.2007.00808.x. [DOI] [PubMed] [Google Scholar]
- 19.Prentice L. A case-cohort design for epidemiologic cohort studies and disease prevention trials. Biometrika. 1986;73:1–11. [Google Scholar]
- 20.Chen K, Lo S-H. Case-cohort and case-control analysis with Cox’s model. Biometrika. 1999;86:755–764. [Google Scholar]
- 21.Barlow WE. Robust variance estimation for the case-cohort design. Biometrics. 1994;50:1064–1072. [PubMed] [Google Scholar]
- 22.Therneau TM, Grambsch PM. Modeling Survival Data. New York: Springer; 2000. [Google Scholar]
- 23.Carrington M, Nelson GW, Martin MP, et al. HLA and HIV-1: Heterozygote advantage and B*35-Cw*04 disadvantage. Science. 1999;283:1748–1752. doi: 10.1126/science.283.5408.1748. [DOI] [PubMed] [Google Scholar]
- 24.Doherty PC, Zinkernagel RM. Enhanced immunological surveillance in mice heterozygous at the H-2 gene complex. Nature. 1975;256:50–52. doi: 10.1038/256050a0. [DOI] [PubMed] [Google Scholar]
- 25.Zinkernagel RM. Immunology taught by viruses. Science. 1996;271:173–178. doi: 10.1126/science.271.5246.173. [DOI] [PubMed] [Google Scholar]
- 26.Gao X, O’Brien TR, Welzel TM, et al. HLA-B alleles associate consistently with HIV heterosexual transmission, viral load, and progression to AIDS, but not susceptibility to infection. AIDS. 2010;24:1835–1840. doi: 10.1097/QAD.0b013e32833c3219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Salgado M, Simón A, Sanz-Minguela B, et al. J Acquir Immune Defic Syndr. 2010. Nov 13, An additive effect of protective host genetic factors correlates with HIV-non progression status. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 28.Klein MR, van der Burg SH, Hovenkamp E, et al. Characterization of HLAB57-restricted human immunodeficiency virus type 1 Gag- and RT-specific cytotoxic T lymphocyte responses. J Gen Virol. 1998;79:2191–2201. doi: 10.1099/0022-1317-79-9-2191. [DOI] [PubMed] [Google Scholar]
- 29.Goulder PJ, Bunce M, Krausa P, et al. Novel, cross-restricted, conserved, and immunodominant cytotoxic T lymphocyte epitopes in slow progressors in HIV type 1 infection. AIDS Res Hum Retroviruses. 1996;12:1691–1698. doi: 10.1089/aid.1996.12.1691. [DOI] [PubMed] [Google Scholar]
- 30.Gillespie GM, Kaul R, Dong T, et al. Cross-reactive cytotoxic T lymphocytes against a HIV-1 p24 epitope in slow progressors with B-57. AIDS. 2002;16:961–972. doi: 10.1097/00002030-200205030-00002. [DOI] [PubMed] [Google Scholar]
- 31.Schneidewind A, Tang Y, Brockman MA, et al. Maternal transmission of human immunodeficiency virus escape mutations subverts HLA-B57 immunodominance but facilitates viral control in the haploidentical infant. J Virol. 2009;83:8616–8627. doi: 10.1128/JVI.00730-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Boulet S, Song R, Kamya P, et al. HIV Protective KIR3DL1 and HLA-B Genotypes Influence NK Cell Function Following Stimulation with HLA-Devoid Cells. J Immunol. 2010;184:2057–2064. doi: 10.4049/jimmunol.0902621. [DOI] [PubMed] [Google Scholar]
- 33.Dean M, Carrington M, O’Brien SJ. Balanced polymorphism selected by genetic versus infectious human disease. Ann Rev Genomics Hum Genet. 2002;3:263–292. doi: 10.1146/annurev.genom.3.022502.103149. [DOI] [PubMed] [Google Scholar]
- 34.Singh KK, Barroga CF, Hughes MD, et al. Genetic influence of CCR5, CCR2, and SDF1 variants on human immunodeficiency virus 1 (HIV-1)-related disease progression and neurological impairment, in children with symptomatic HIV-1 infection. J Infect Dis. 2003;188:1461–1472. doi: 10.1086/379038. [DOI] [PubMed] [Google Scholar]
- 35.Singh KK, Hughes MD, Chen J, et al. Lack of protective effects of interleukin-4 -589-C/T polymorphism against HIV-1-related disease progression and central nervous system impairment, in children. J Infect Dis. 2004;189:587–592. doi: 10.1086/381460. [DOI] [PubMed] [Google Scholar]
- 36.Singh KK, Hughes MD, Chen J, et al. Impact of MCP-1-2518-G allele on the HIV-1 disease of children in the United States. AIDS. 2006;20:475–478. doi: 10.1097/01.aids.0000200540.09856.58. [DOI] [PubMed] [Google Scholar]
- 37.Hardie RA, Luo M, Bruneau B, et al. Human leukocyte antigen-DQ alleles and haplotypes and their associations with resistance and susceptibility to HIV-1 infection. AIDS. 2008;22:807–816. doi: 10.1097/QAD.0b013e3282f51b71. [DOI] [PubMed] [Google Scholar]