Abstract
Lewy bodies (LBs), the pathological hallmark of Lewy body disease (LBD), contain α-synuclein, as well as other proteins. In this study, we examined the relationship of α-synuclein to two rate-limiting enzymes in neurotransmitter synthesis, tyrosine hydroxylase (TH), and choline acetyltransferase (ChAT). Double labeling immunohistochemistry for α-synuclein and TH revealed TH-immunoreactivity within LBs in catecholaminergic neurons in the substantia nigra and locus coeruleus, but not within LBs in cholinergic neurons in the pedunculopontine nucleus and nucleus basalis of Meynert. In contrast, ChAT-immunoreactivity within LBs was detected in cholinergic, but not within LBs in catecholaminergic neurons. The amygdala was devoid of TH and ChAT positive LBs, although a few Lewy neurites contained ChAT immunoreactivity. Further analysis revealed two distinct patterns of neurotransmitter immunoreactivity within LBs. One pattern had diffuse co-localization of TH or ChAT with α-synuclein as in cortical-type LBs, while the other had intense TH or ChAT immunoreactivity in the LB core surrounded by a peripheral rim of α-synuclein as in brainstem-type LBs. Levels of both TH and ChAT were higher in brainstem-type LBs than in the cytoplasm of the same neuron or in neurons from the same case devoid of LBs. Given the fact that LB-containing neurons have decreases in cytoplasmic TH and ChAT immunoreactivity, these results suggest LBs may disrupt cholinergic and catecholaminergic neurotransmitter production by sequestration of the rate limiting enzymes for acetylcholine and catecholamine synthesis.
Keywords: α-synuclein, choline acetyltransferase, Lewy bodies, locus coeruleus, nucleus basalis of Meynert, pedunculopontine nucleus, substantia nigra, tyrosine hydroxylase
Introduction
Lewy bodies (LBs), the pathological hallmark of Lewy body disease (LBD), are intracellular aggregates composed of over 300 proteins [6], the most well known being ubiquitin and α-synuclein [5, 20]. Based upon morphology, LBs can be classified as brainstem-type or cortical-type [4, 15, 19]. Brainstem-type LBs are intraneuronal spherical inclusions with a dense central core surrounded by a pale staining halo. In contrast, cortical-type LBs lack a dense central core and halo, and do not have a sharp boundary from the adjacent cytoplasm. Although these types correlate with the location (brainstem-type LBs being common in vulnerable brainstem nuclei and cortical-type LBs being common in the cortex), brainstem-type hyaline LBs can be detected in the cortex [8] and cortical-type LBs can be detected in subcortical nuclei [4]. This classification is solely based on morphology and not on biochemical composition or anatomy.
In addition to α-synuclein and ubiquitin, a number of other molecules, such as tau, neurofilament and various kinases have been noted in LBs [15, 18–20]. This indicates protein composition of LBs is heterogeneous and dependent upon the specific neuron in which the LB forms. For example, tyrosine hydroxylase (TH), the rate limiting enzyme in catecholamine synthesis has been described in LBs in catecholaminergic neurons of the brain, especially noradrenergic neurons of the locus coeruleus (LC) and dopaminergic neurons of the substantia nigra (SN) [11, 22]. Choline acetyltransferase (ChAT), the rate limiting enzyme in acetylcholine production, has not been studied in LBs. On the other hand, it is well established that there are significant cholinergic deficits in LBD [2, 12, 21]. ChAT is markedly reduced in LBD compared with age matched normals [2, 13, 14, 21], and there is extensive neuronal loss and LB pathology in cholinergic nuclei, including the nucleus basalis of Meynert (nbM) and the pedunculopontine nucleus (PPN) [7, 10, 17, 23].
The purpose of this study was to determine the relationship of TH and ChAT to LBs and the cytoplasmic levels of these neurotransmitter synthetic enzymes in neurons with and without LBs with single and double labeling immunohistochemistry in the nbM, SN, PPN and LC. Results were compared to the amygdala, a region of the brain highly vulnerable to LBs [16], but lacking in cholinergic and catecholaminergic neurons. We demonstrate cell type specificity for TH and ChAT immunoreactivity within LBs. Furthermore, the intensity of neurotransmitter enzyme immunoreactivity within LBs, especially brainstem-type LBs, was greater than the rest of the cytoplasm.
Materials and Methods
Case material
Paraffin-embedded, formalin-fixed brain tissue samples were obtained from the brain bank for neurodegenerative disorders at Mayo Clinic in Jacksonville, Florida. The cases included LBD (N=16) and age-matched controls without significant neuropathologic abnormalities (N=16). The demographics of cases are summarized in Table 1. There were no significant differences between groups with respect to age or sex. Normals had a higher postmortem interval (PMI) when compared to LBD cases; however, PMI did not correlate with TH or ChAT staining intensities. All tissues had been collected with appropriate consent from brain donors and their legal next-of-kin, and the brain donation program is approved by the Institutional Review Board of Mayo Clinic.
Table 1.
Demographics of case material
| Pathologic group | LB Type | N (females) | Median Age at Death (range, in years) | Median PMI (range, in hrs) | Median Braak NFT Stage (range) |
|---|---|---|---|---|---|
| LBD | Diffuse (n=14); Transitional (n=2) | 16 (2) | 78 (61–85) | 4 (3–8) | III (II–IV) |
| Controls | NA | 16 (8) | 74 (51–94) | 21.5 (5–30) | I (0–III) |
Diffuse = diffuse cortical LBs; Transitional = limbic predominant LBs; NA = not applicable; NFT = neurofibrillary tangle; PMI= Post-mortem interval
Brain regions included in this study included the following: basal forebrain, which includes amygdala, hypothalamus and nbM; rostral midbrain at the level of the third nerve, which contains the SN; caudal midbrain at the level of the inferior colliculus, which contains the PPN; and rostral pons, which contains the LC. Specific nuclei were identified by immunohistochemistry for TH (SN and LC) and ChAT (nbM and PPN). The amygdala served as a negative control for neurotransmitter enzyme immunoreactivity.
Immunohistochemistry was performed using commercially available, well characterized, primary antibodies: LB509, a mouse monoclonal α-synuclein antibody (1:50; Zymed, South San Francisco, CA); TH, a rabbit polyclonal antibody (1:600; Affinity Bioreagents, Golden, CO); and AB144P, a goat polyclonal antibody to ChAT (1:200; Chemicon, Billerica, MA).
Immunofluorescent double labeling was performed in 5 μm thick paraffin sections. After removal of paraffin with xylenes and a graded series of alcohols, tissue sections were subjected to antigen retrieval by steaming in distilled water for 30 minutes and elimination of normal synaptic α-synuclein immunoreactivity by treatment with proteinase K [1] (DAKO, Carpinteria, CA) for five minutes. All sections were incubated in Protein Block Serum-Free (DAKO, Carpinteria, CA) for one hour, washed in 0.1M phosphate buffered saline (PBS), then incubated in a cocktail containing the α-synuclein antibody (LB509) with either rabbit anti-TH or goat anti-ChAT overnight at 4°C. The following day, sections were washed in 0.1M PBS and incubated with specific secondary reagent pairs – goat anti-rabbit (for TH) and goat anti-mouse (for α-synuclein) or donkey anti-goat (for ChAT) and donkey anti-mouse (for α-synuclein) with either Alexa Fluor 488 or 568 fluorochromes (1:500, Molecular Probes, Eugene, OR) for one hour at room temperature and washed in 0.1M PBS. To block autofluorescence, sections were treated with Sudan black (Sigma-Aldrich, St. Louis, MO) for two minutes, washed and coverslipped using VectaShield Mounting media (Vector Laboratories, Burlingame, CA). To ensure there was no preference to a particular fluorochrome, all slides were labeled with each antibody to both fluorochromes. There was no nonspecific cross-over artifact (data not shown).
Image acquisition and analysis
Stained sections from each area (LC, SN, PPN, nbM and amygdala) were evaluated with z-stack images on a Zeiss LSM 510 META laser scanning confocal microscope (Zeiss, Heidelberg, Germany) to study the qualitative aspects of double labeling for ChAT/α-synuclein and TH/α-synuclein in neurons with and without LBs. At least 20 x-y planar images (2 μm in thickness) were captured per group at 100x with oil immersion to create 5 μm z-stack reconstructions. This permitted representation of the full extent of signal intensities of TH/α-synuclein and ChAT/α-synuclein within neurons with and without LBs. Images were analyzed with the Zeiss LSM 510 META AIM software, version 4.0 (Zeiss, Heidelberg, Germany). ChAT or TH positive cells were grouped into one of three categories based on α-synuclein and neurotransmitter marker distribution: none (ChAT or TH positive cells without LBs), cortical-type (ChAT or TH positive cell with an even distribution of ChAT or TH within the α-synuclein-positive LB), or brainstem-type (ChAT or TH positive cell with ChAT or TH concentrated in the central domain of the LB, encircled by α-synuclein in its perimeter). Percentages of brainstem and cortical-type LBs were determined by dividing the number of cells with LBs (either cortical or brainstem) by the total number of cells (the sum of none, brainstem- and cortical-type LBs).
The neurons were examined to determine signal intensities for ChAT and TH within LBs and their subjacent cytoplasm. For quantitative analyses of intensity of immunoreactivity, a subset of neurons from LC, SN, PPN and nbM was analyzed in 5 pure LBD cases (1F:4M; average age: 73.4; range: 61–78 years) and 5 clinically normal cases (1F:4M; average age: 72.7; range: 63–81 years). A minimum of 20 cells per LB subtype and their subjacent cytoplasm were studied. The analysis included brainstem-type LBs, cortical-type LBs, neurons with no LBs from LBD and neurons with no LBs from normal controls. Images were acquired using z-stacks with images taken 2 μm apart at 100x magnification, with a 12-bit data depth, scan speed of 2 and 512 × 512 pixel frame size, to decrease saturation time. To avoid bias and the confounding variable of signal intensity bleaching over time, LBD and normal tissues were alternated during image acquisition. Average signal intensities in designated areas (cytoplasm and within LBs) were measured using ImageJ software, version 1.41o (National Institutes of Health, USA).
Statistical methods
Statistical analyses were preformed with Sigma Plot 11.0 (Systat Software, Inc., San Jose, CA) using Kruskal-Wallis One Way Analysis of Variance on Ranks and Mann-Whitney Rank Sum Tests where appropriate, with P< 0.05 considered significant.
Results
ChAT and TH co-localization with α-synuclein within LBs is cell type specific
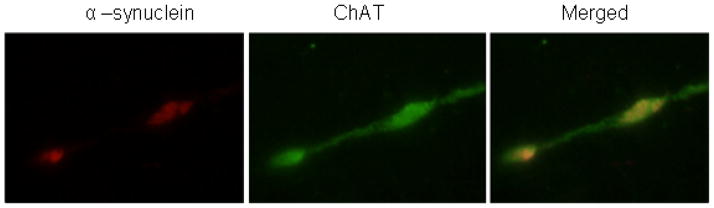
Using double labeling immunohistochemistry, we noted ChAT within LBs in PPN and nbM (Fig. 1), but not within LBs in catecholaminergic neurons of the LC and SN. Conversely, TH-immunoreactivity was present within LBs in the LC and SN, but not within LBs in cholinergic nuclei (Fig. 2). All LBs in TH and ChAT positive cells had at least some neurotransmitter immunoreactivity; however, no LBs in these cells contained only α-synuclein. Given the double dissociation of TH and ChAT immunoreactivity in cholinergic and catecholaminergic neurons, it is extremely unlikely that the results are due to antibody cross-reactivity. Moreover, in the amygdala, a brain region containing no perikaryal ChAT or TH immunopositive neurons, there was no immunoreactivity to either neurotransmitter synthetic enzyme within LBs, although a few Lewy neurites showed ChAT immunoreactivity (Fig. 3).
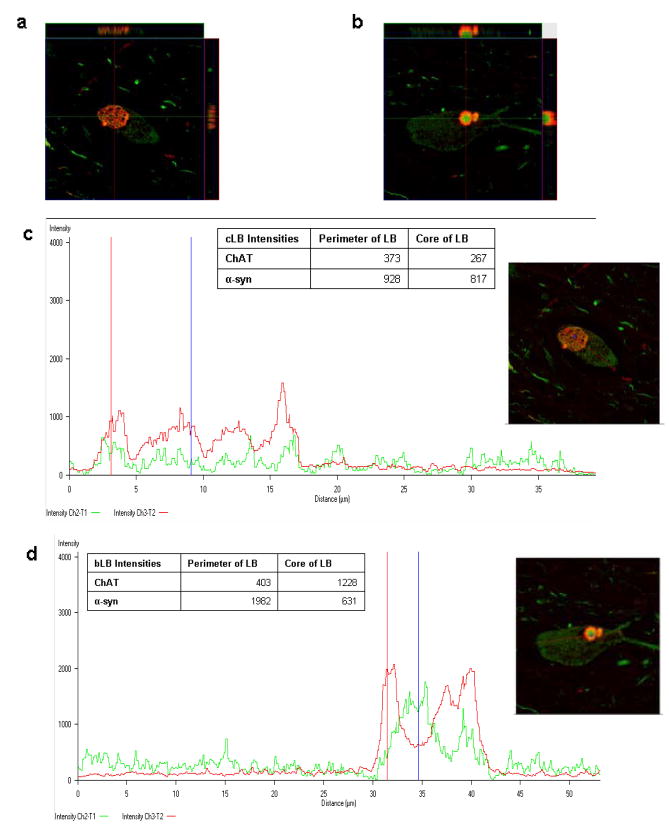
Fig. 1.
(a–b) 5 μm z-stack reconstructions of nbM cholinergic neurons labeled with ChAT (green) and α-synuclein (red; α-syn) to illustrate the x-y planar distributions. (c–d) Two dimensional intensity profiles of ChAT (intensity Ch2-T1, green) and α-syn (intensity Ch3-T2, red) throughout the entirely of the neurons; blue lines indicates the center of the LBs (blue marker in images on left) and the red lines indicates intensities at the perimeter of the LBs (red markers in images on left). Inset tables indicate relative quantitation of each marker. (a, c) a cortical-type LB distribution of ChAT in a nbM neuron, noted by equal distributions of ChAT and α-syn at the perimeter and core of the LB. (b, d) a brainstem-type LB distribution where ChAT intensity is greatest in the core of LB while α-syn is highest at the periphery. (All 100× magnification.)
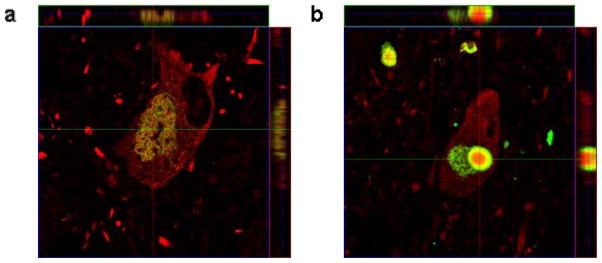
Fig. 2.
(a–b) 5 μm z-stack reconstructions of SN dopaminergic neurons labeled with TH (red) and α-synuclein (green) to illustrate the x-y planar distributions (100x magnification). (a) A cortical-type LB distribution of TH in a SN neuron and (b) a brainstem-type LB distribution of TH in a SN neuron.
Fig. 3.
A ChAT positive Lewy neurite in the amygdala. (100x magnification)
Analysis of z-stack images for ChAT (Fig. 1) and TH (Fig. 2) revealed two distinct patterns of transmitter synthetic enzyme within LBs. The first type resembled cortical-type LBs and showed equal distributions of α-synuclein and ChAT or TH throughout the inclusion (Figure 1a and c, Fig. 2a). The second type was similar to brainstem-type LBs and had intense staining of ChAT or TH in the center and α-synuclein at the periphery of the LB (Fig. 1b and d, Fig. 2b).
ChAT and TH with respect to LB subtypes
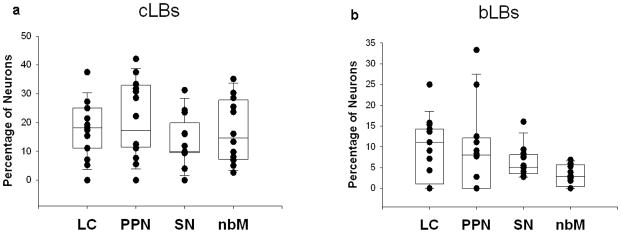
In order to understand if the two patterns indeed correlated with the profiles of brainstem- and cortical-type LBs, we examined whether LB types from double labeling immunohistochemistry (Fig. 4) correlated with distribution of brainstem- and cortical-type LBs as previously described in histologic preparations [3, 19]. The nbM contained fewer brainstem-type LBs compared to the SN, LC and PPN, but the difference did not reach statistical significance (Fig. 4). There were no differences in the percentages of cortical-type LBs (Fig. 4). All areas contained a greater proportion of cortical-type LBs than brainstem-type LBs (Ps < 0.009).
Fig. 4.
Box plots of percentage of neurons with cortical- (a, cLBs) or brainstem- type LBs (b, bLBs) in LC, PPN, SN and nbM. All counts were done on 5 μm paraffin sections double labeled for α-synuclein and either ChAT or TH. (Boxes show median, 25th and 75th percentile; whisker plots show 5th and 95th percentile.)
Neurotransmitter immunoreactivity in cytoplasm of neurons with and without LBs
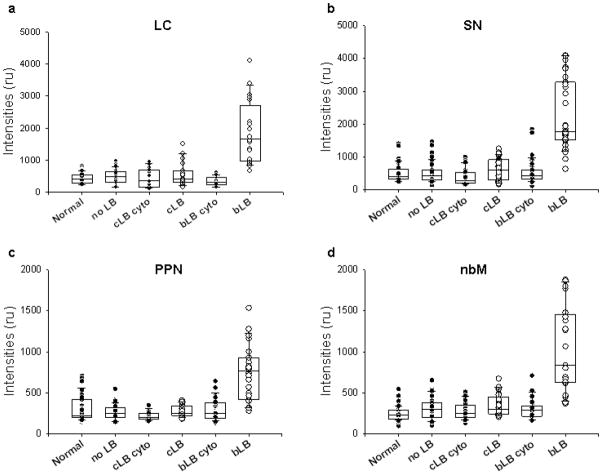
The average intensities of TH and ChAT cytoplasmic immunoreactivity in LBD neurons without LBs was not different from TH and ChAT cytoplasmic immunoreactivity in neurons of normal controls in the SN, LC, PPN and nbM (Fig. 5). On the other hand, average intensities of TH and ChAT immunoreactivity were significantly lower in all areas analyzed (cytoplasm of neurons in normal brains, cytoplasm of neurons in LBD cases with no LBs, cytoplasm of neurons with cortical-type LBs and cytoplasm of neurons with brainstem-type LBs) compared to TH and ChAT immunoreactivity within brainstem-type LBs (Fig. 5 and Table 2). When comparing average intensities of cytoplasmic TH and ChAT in neurons with cortical-type LBs to average intensities of TH and ChAT within cortical-type LBs, there were also differences in the SN, PPN and nbM, but not in the LC (Table 2b). These differences, however, were not as great as those seen for brainstem-type LBs. The only other significant difference was lower intensity of cytoplasmic TH immunoreactivity in SN neurons with cortical-type LBs compared to TH immunoreactivity in the cytoplasm of SN neurons from normal controls. A similar trend was also noted in the LC (Fig 5a and b, Table 2a).
Fig. 5.
Box plots of median signal intensities in relative units (ru) of TH (a, b) and ChAT (c, d) in the LC (a), SN (b), PPN (c) and nbM (d) in cytoplasm (cyto) (black dots) and in LBs (white dots) in cells with cortical-type LBs (cLB), brainstem-type LBs (bLB) and no LBs, as well as the cytoplasm of neurons in normal controls (normal). See Table 2 for statistical comparisons. (Boxes show median, 25th and 75th percentile; whisker plots show 5th and 95th percentile.)
Table 2.
Statistical comparisons of TH and ChAT immunoreactivity
| 2a. Four-way comparison of cytoplasmic neurotransmitter enzyme intensity in normal controls to neurons with no LBs to neurons with cLB and to neurons with bLB |
| TH |
| LC (p=0.057) |
| SN (p =0.019) |
| cLB cytoplasm vs. normal |
|
|
| ChAT |
| PPN (p=0.158) |
| nbM (p=0.301) |
| ANOVA on Ranks with Dunn’s Method for pairwise comparisons. |
| 2b. Comparison of neurotransmitter enzyme intensity in cytoplasm to LB, by type | |
|---|---|
| TH | |
| LC | |
| bLB vs. bLB cytoplasm (p<0.001) | |
| cLB vs. cLB cytoplasm (p=0.365) | |
| SN | |
| bLB vs. bLB cytoplasm (p<0.001) | |
| cLB vs. cLB cytoplasm (p<0.001) | |
|
| |
| ChAT | |
| PPN | |
| bLB vs. bLB cytoplasm (p<0.001) | |
| cLB vs. cLB cytoplasm (p=0.003) | |
| nbM | |
| bLB vs. bLB cytoplasm (p<0.001) | |
| cLB vs. cLB cytoplasm (p=0.056) | |
Mann-Whitney Rank Sum Test.
Discussion
The results of this study demonstrate both TH and ChAT are present in a subset of LBs. TH was detected in catecholaminergic neurons of the LC and SN, and CHAT was detected in cholinergic neurons of the PPN and nbM. In a region lacking cholinergic and catecholaminergic neurons, the amygdala, none of the LBs had ChAT or TH immunoreactivity. The results are not due to TH or ChAT antibody cross reactivity with α-synuclein since TH was only detected within LBs in catecholaminergic neurons, and ChAT was only detected within LBs in cholinergic neurons.
On closer examination, two distinct patterns of neurotransmitter enzyme immunoreactivity were detected within LBs, which we have operationally defined as brainstem-type and cortical-type LBs based upon morphologic appearance (Figs. 1 and 2). In brainstem-type LBs, the neurotransmitter enzyme was concentrated in the center of the LB, with less co-localization with α-synuclein in the periphery of the LB, while in cortical-type LBs, there was uniform co-localization of neurotransmitter enzyme and α-synuclein throughout the LB. Further investigation showed that neurotransmitter enzyme levels were increased within brainstem-type LBs compared to the cytoplasm of the same neurons, as well as neurons in the same nucleus without LBs (Fig. 5). This fact was also noted when comparing neurotransmitter synthetic enzyme levels in cortical-type LBs to the cytoplasm of the same neurons, although the difference was not as great (Table 2). These data support the concept that the composition of LBs is heterogeneous depending on cell type and that neurotransmitter synthetic enzymes may be sequestered within LBs in defined neuronal types.
The decrease in neurotransmitter enzyme level in the cytoplasm of neurons with LBs was most clearly evident in neurons with brainstem-type LBs in all regions evaluated, and a similar trend was also detected in neurons with cortical-type LBs. It is worth noting that in the SN, the cytoplasm of neurons with cortical-type LBs had significantly lower TH than the cytoplasm of SN neurons in normals (Fig. 5b), with a similar trend in the LC (Fig 5a). These findings may indicate that with an increased sequestration within LBs, the neuron is unable to maintain even basal levels of neurotransmitter synthetic enzymes.
Another issue to consider is the enzymatic efficiency of a neurotransmitter enzyme within LBs compared to the normal location in the neuronal cytoplasm. It may be speculated that access to precursor molecules and proper disposition of the neurotransmitter would be impeded by the abnormal granulofilamentous structural properties of the LB, and thus would contribute to the decrease in neurotransmitter production in LB-containing neurons. This decrease in neurotransmitter production would contribute to the abnormal neurotransmitter phenotype characterized in LBD cholinergic and catecholaminergic neurons. A limitation of this study is the lack of information on neurotransmitter levels, which we hypothesize would be reduced in proportion to the degree of neurotransmitter enzyme sequestration within LBs. Even though TH and ChAT are rate-limiting enzymes, the net effect of their sequestration within LBs on tissue transmitter levels would be difficult to interpret. A means of assessing transmitter production at the cellular level would be needed to address this question, but at present such a method is not available for human postmortem tissues.
Relatively little is known about neurotransmitter enzyme accumulation within LBs. As far as we could discern, this has been previously reported only for TH [9, 11, 22]. The current study is the first to show ChAT immunoreactivity within LBs. The present results also suggest sequestration of neurotransmitter synthetic enzymes may have adverse effects on the neuronal phenotype in that the relative levels of neurotransmitter enzyme were decreased in the cytoplasm of the same neuron, possibly reflecting failure of the cell to compensate for the enzyme within LBs. It is unclear if the observed findings are relevant to other neurons vulnerable to LBs. Sequestration of neurotransmitter synthetic enzymes within LBs remains to be investigated for serotonergic neurons (tryptophan hydroxylase), noradrenergic neurons (dopamine beta-hydroxylase), adrenergic neurons (phenyl-ethanolamine-N-methyl transferase), histaminergic neurons (histidine decarboxylase) and for GABAergic neurons (glutamic acid decarboxylase). It remains a possibility that sequestration of neurotransmitter synthetic enzymes within LBs may not be restricted to TH and ChAT.
Elucidation of the molecular nature of LBs is crucial to understanding the functional consequences of neurodegeneration in LBD. The results of the current study bring to light a possible untoward effect of LB pathology, namely, sequestration of rate-limiting enzymes responsible for neurotransmitter production, which may contribute to neurotransmitter deficiencies and abnormal symptoms of LBD.
Acknowledgments
The authors thank Monica Castanedes Casey, David Personett, Linda Rousseau and Virginia Phillips for their histopathological expertise and technical support. The authors would also like to thank Dr. Tanis Ferman for constructive advice. This research would not be possible without brain donations from patients and their families. This study was supported by grants from the National Institutes of Health (R01-AG15866, P50-AG16574, P50-NS40256) and Mayo Foundation for Research & Education.
References
- 1.Beach TG, White CL, Hamilton RL, et al. Evaluation of alpha-synuclein immunohistochemical methods used by invited experts. Acta Neuropathol. 2008;116:277–288. doi: 10.1007/s00401-008-0409-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dickson DW, Davies P, Mayeux R, et al. Diffuse Lewy body disease. Neuropathological and biochemical studies of six patients. Acta Neuropathol. 1987;75:8–15. doi: 10.1007/BF00686786. [DOI] [PubMed] [Google Scholar]
- 3.Gomez-Tortosa E, Newell K, Irizarry MC, Sanders JL, Hyman BT. alpha-Synuclein immunoreactivity in dementia with Lewy bodies: morphological staging and comparison with ubiquitin immunostaining. Acta Neuropathol. 2000;99:352–357. doi: 10.1007/s004010051135. [DOI] [PubMed] [Google Scholar]
- 4.Kosaka K. Lewy bodies in cerebral cortex, report of three cases. Acta Neuropathol. 1978;42:127–134. doi: 10.1007/BF00690978. [DOI] [PubMed] [Google Scholar]
- 5.Kuzuhara S, Mori H, Izumiyama N, Yoshimura M, Ihara Y. Lewy bodies are ubiquitinated. A light and electron microscopic immunocytochemical study. Acta Neuropathol. 1988;75:345–353. doi: 10.1007/BF00687787. [DOI] [PubMed] [Google Scholar]
- 6.Leverenz JB, Umar I, Wang Q, et al. Proteomic identification of novel proteins in cortical lewy bodies. Brain Pathol. 2007;17:139–145. doi: 10.1111/j.1750-3639.2007.00048.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lippa CF, Smith TW, Perry E. Dementia with Lewy bodies: choline acetyltransferase parallels nucleus basalis pathology. J Neural Transm. 1999;106:525–535. doi: 10.1007/s007020050176. [DOI] [PubMed] [Google Scholar]
- 8.Masliah E, Galasko D, Wiley CA, Hansen LA. Lobar atrophy with dense-core (brain stem type) Lewy bodies in a patient with dementia. Acta Neuropathol. 1990;80:453–458. doi: 10.1007/BF00307702. [DOI] [PubMed] [Google Scholar]
- 9.Mori F, Nishie M, Kakita A, Yoshimoto M, Takahashi H, Wakabayashi K. Relationship among alpha-synuclein accumulation, dopamine synthesis, and neurodegeneration in Parkinson disease substantia nigra. J Neuropathol Exp Neurol. 2006;65:808–815. doi: 10.1097/01.jnen.0000230520.47768.1a. [DOI] [PubMed] [Google Scholar]
- 10.Nakano I, Hirano A. Parkinson’s disease: neuron loss in the nucleus basalis without concomitant Alzheimer’s disease. Ann Neurol. 1984;15:415–418. doi: 10.1002/ana.410150503. [DOI] [PubMed] [Google Scholar]
- 11.Nakashima S, Ikuta F. Tyrosine hydroxylase protein in Lewy bodies of parkinsonian and senile brains. J Neurol Sci. 1984;66:91–96. doi: 10.1016/0022-510x(84)90144-8. [DOI] [PubMed] [Google Scholar]
- 12.Perry E, Court J, Goodchild R, et al. Clinical neurochemistry: developments in dementia research based on brain bank material. J Neural Transm. 1998;105:915–933. doi: 10.1007/s007020050102. [DOI] [PubMed] [Google Scholar]
- 13.Perry E, Court J, Goodchild R, et al. Clinical neurochemistry: developments in dementia research based on brain bank material. Journal of Neural Transmission. 1998;105:915–933. doi: 10.1007/s007020050102. [DOI] [PubMed] [Google Scholar]
- 14.Perry EK, McKeith I, Thompson P, et al. Topography, extent, and clinical relevance of neurochemical deficits in dementia of Lewy body type, Parkinson’s disease, and Alzheimer’s disease. Ann N Y Acad Sci. 1991;640:197–202. doi: 10.1111/j.1749-6632.1991.tb00217.x. [DOI] [PubMed] [Google Scholar]
- 15.Pollanen MS, Dickson DW, Bergeron C. Pathology and biology of the Lewy body. J Neuropathol Exp Neurol. 1993;52:183–191. doi: 10.1097/00005072-199305000-00001. [DOI] [PubMed] [Google Scholar]
- 16.Popescu A, Lippa CF, Lee VM, Trojanowski JQ. Lewy bodies in the amygdala: increase of alpha-synuclein aggregates in neurodegenerative diseases with tau-based inclusions. Arch Neurol. 2004;61:1915–1919. doi: 10.1001/archneur.61.12.1915. [DOI] [PubMed] [Google Scholar]
- 17.Rinne JO, Ma SY, Lee MS, Collan Y, Roytta M. Loss of cholinergic neurons in the pedunculopontine nucleus in Parkinson’s disease is related to disability of the patients. Parkinsonism Relat Disord. 2008;14:553–557. doi: 10.1016/j.parkreldis.2008.01.006. [DOI] [PubMed] [Google Scholar]
- 18.Schmidt ML, Murray J, Lee VM, Hill WD, Wertkin A, Trojanowski JQ. Epitope map of neurofilament protein domains in cortical and peripheral nervous system Lewy bodies. Am J Pathol. 1991;139:53–65. [PMC free article] [PubMed] [Google Scholar]
- 19.Shults CW. Lewy bodies. Proc Natl Acad Sci U S A. 2006;103:1661–1668. doi: 10.1073/pnas.0509567103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Spillantini MG, Schmidt ML, Lee VM, Trojanowski JQ, Jakes R, Goedert M. Alpha-synuclein in Lewy bodies. Nature. 1997;388:839–840. doi: 10.1038/42166. [DOI] [PubMed] [Google Scholar]
- 21.Tiraboschi P, Hansen LA, Alford M, et al. Cholinergic dysfunction in diseases with Lewy bodies. Neurology. 2000;54:407–411. doi: 10.1212/wnl.54.2.407. [DOI] [PubMed] [Google Scholar]
- 22.Wakabayashi K, Takahashi H, Ohama E, Ikuta F. Parkinson’s disease: an immunohistochemical study of Lewy body-containing neurons in the enteric nervous system. Acta Neuropathol. 1990;79:581–583. doi: 10.1007/BF00294234. [DOI] [PubMed] [Google Scholar]
- 23.Whitehouse PJ, Hedreen JC, White CL, 3rd, Price DL. Basal forebrain neurons in the dementia of Parkinson disease. Ann Neurol. 1983;13:243–248. doi: 10.1002/ana.410130304. [DOI] [PubMed] [Google Scholar]