Abstract
A healthy 25-year-old woman presents with worsening dysmenorrhea, pain of recent onset in the left lower quadrant, and dyspareunia. She has regular menstrual cycles, and her last menstrual period was 3 weeks before presentation. How should this patient be evaluated and treated?
THE CLINICAL PROBLEM
Endometriosis, a major contributor to pelvic pain and subfertility,1 is characterized by endometrial-like tissue outside the uterus (Fig. 1), primarily on the pelvic peritoneum, ovaries, and rectovaginal septum, and in rare cases on the diaphragm, pleura, and pericardium. Endometriosis affects 6 to 10% of women of reproductive age, 50 to 60% of women and teenage girls with pelvic pain, and up to 50% of women with infertility.2,3 Peritoneal disease, which is dependent on estrogen for growth, derives from retrograde menstruation of steroid hormone–sensitive endometrial cells and tissues (Fig. 2), which implant on peritoneal surfaces and elicit an inflammatory response. This response is accompanied by angiogenesis, adhesions, fibrosis, scarring, neuronal infiltration, and anatomical distortion (Fig. 1 and 2), resulting in pain and infertility.1,4–6 Although most women have retrograde menstruation, not all women with retrograde menstruation have endometriosis; affected women may have an immune dysfunction that interferes with clearing of the lesions.1 Since ovarian endometriomas are clonal and lesions can have genetic mutations, somatic mutations with resulting growth dysregulation also may be etiologic factors.1,4 Disease at distant sites is probably caused by lymphatic or hematogenous spread or meta-plastic transformation.
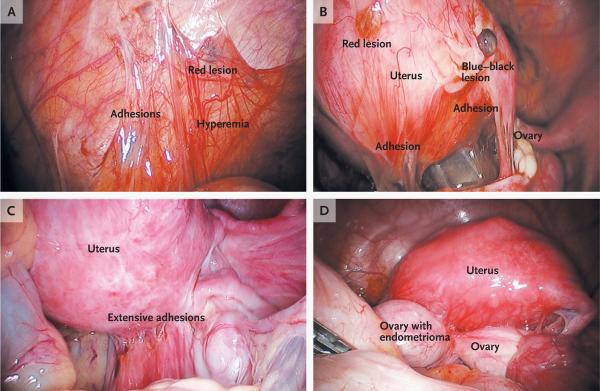
Figure 1. Peritoneal Lesions and an Ovarian Endometrioma Due to Endometriosis.
Panel A shows an endometriotic implant (red lesion), adhesions, and hyperemia in the peritoneum. Panel B shows peritoneal implants, including red and blue–black lesions and adhesions. Panel C shows extensive adhesions distorting the normal pelvic anatomy. Panel D shows an endometrioma adherent to the posterior uterus and distending the ovarian capsule. At surgery, it is difficult to distinguish visually an endometrioma from a cyst of the corpus luteum, a hemorrhagic cyst, or a simple cyst. Although the cyst fluid in endometriomas is thick and dark brown because it contains hemosiderin (hence, the name “chocolate cysts”), this color is not specific to endometriomas. (Images courtesy of Dr. Christopher Herndon, University of California, San Francisco.)
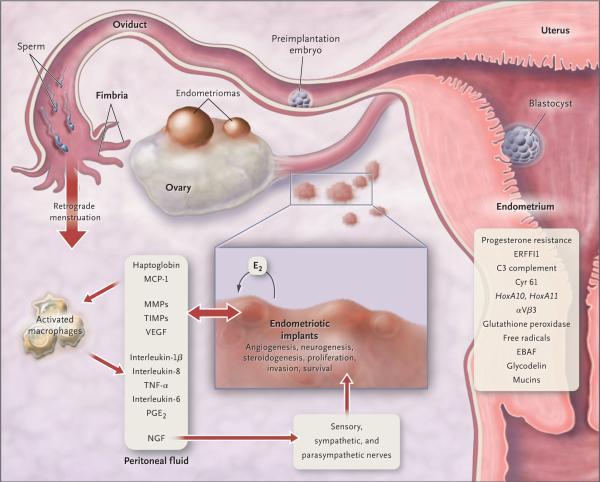
Figure 2. Pathophysiology of Pain and Infertility Associated with Endometriosis.
Retrograde transplanted endometrial tissue and cells attach to peritoneal surfaces, establish a blood supply, and invade nearby structures. They are infiltrated by sensory, sympathetic, and parasympathetic nerves and elicit an inflammatory response. Endometriotic implants secrete estradiol (E2) as well as prostaglandin E2 (PGE2), agents that attract macrophages (monocyte chemotactic protein 1 [MCP-1]), neurotrophic peptides (nerve growth factor [NGF]), enzymes for tissue remodeling (matrix metalloproteinases [MMPs]) and tissue inhibitors of MMPs (TIMPs), and proangiogenic substances such as vascular endothelial growth factor (VEGF) and interleukin-8. Lesions secrete haptoglobin, which decreases macrophage adhesion and phagocytic function. Lesions and activated macrophages, which are abundant in the peritoneal fluid in women with endometriosis, also secrete proinflammatory cytokines (interleukin-1β, interleukin-8, interleukin-6, and tumor necrosis factor α [TNF-α]). Local (and systemic) estradiol can stimulate lesion production of PGE2, which can activate pain fibers, enhance neuronal invasion of lesions by stimulating production of NGF and other neurotrophins, and promote sprouting of nociceptors that contribute to persistent inflammatory pain and inhibit neuronal apoptosis. Endometrial bleeding factor (EBAF) is misexpressed and may contribute to uterine bleeding. Infertility results from the toxic effects of the inflammatory process on gametes and embryos, compromised fimbrial function, and eutopic endometrium that is resistant to the action of progesterone and is inhospitable to embryonic implantation. HoxA10 and HoxA11 genes and αVβ3 integrin are not up-regulated by progesterone, and thus the endometrium is inhospitable to an implanting embryo. Endocrine-disrupting chemicals can contribute to progesterone resistance and perhaps immune dysfunction.1,4 ERFFI1 (ErbB receptor feedback inhibitor 1) is constitutively expressed and there is excess mitogenic signaling.
Risk factors for endometriosis include obstruction of menstrual outflow (e.g., mullerian anomalies7), exposure to diethylstilbestrol in utero,8 prolonged exposure to endogenous estrogen (e.g., because of early menarche, late menopause, or obesity), short menstrual cycles, low birth weight,9 and exposure to endocrine-disrupting chemicals.10 Twin and family studies suggest a genetic component.11 Consumption of red meat and trans fats is associated with an increased risk of laparoscopically confirmed endometriosis, and eating fruits, green vegetables, and n−3 long-chain fatty acids is associated with a decreased risk.12 Prolonged lactation and multiple pregnancies are protective.9 Endometriosis is associated with increased risks of autoimmune diseases and ovarian endometrioid and clear-cell cancers, as well as other cancers, including non-Hodgkin's lymphoma and melanoma.1
Follow-up of women with pelvic pain and laparoscopically identified disease has shown that 17 to 29% of lesions resolve spontaneously, 24 to 64% progress, and 9 to 59% are stable over a 12-month period.13 Endometriosis is a major cause of disability and compromised quality of life in women and teenage girls.14 In the United States, the estimated costs of diagnosing endometriosis and treating associated pain and infertility totaled $22 billion in 2002.15
STRATEGIES AND EVIDENCE
EVALUATION
Chronic pelvic pain accounts for 10% of outpatient gynecologic visits.16 A complete medical, surgical, social, and family history should be obtained from patients who present with this symptom, and they should undergo a physical examination that includes a pelvic examination. Focal pain or tenderness on pelvic examination is associated with pelvic disease in 97% of patients and with endometriosis in 66% of patients.17 A pelvic mass, immobile pelvic organs, and rectovaginal nodules are suggestive of endometriosis but are not diagnostic because of their poor sensitivity and specificity. An evaluation of both the female patient and her male partner is indicated in cases of associated infertility.18
Pelvic pain due to endometriosis is usually chronic (lasting ≥6 months) and is associated with dysmenorrhea (in 50 to 90% of cases), dyspareunia, deep pelvic pain, and lower abdominal pain with or without back and loin pain. The pain can occur unpredictably and intermittently throughout the menstrual cycle or it can be continuous, and it can be dull, throbbing, or sharp, and exacerbated by physical acitivity.16,19 Bladder- and bowel-associated symptoms (nausea, distention, and early satiety) are typically cyclic.16,19 Pain often worsens over time and may change in character; infrequently, women report burning or hypersensitivity, symptoms that are suggestive of a neuropathic component.20 Symptoms overlap with those of several other gynecologic conditions (e.g., pelvic inflammatory disease, pelvic adhesions, ovarian cysts or masses, leiomyomata, and adenomyosis) and nongynecologic conditions and factors (e.g., irritable bowel syndrome, inflammatory bowel disease, interstitial cystitis, myofascial pain, depression, and a history of sexual abuse), making diagnosis challenging.16,19
DIAGNOSIS AND CLINICAL STAGING
Currently, the definitive method to diagnose and stage endometriosis and evaluate the recurrence of disease after treatment is visualization at surgery21 (Fig. 1). The revised scoring system of the American Society for Reproductive Medicine is used to determine the disease stage (ranging from I, indicating minimal disease, to IV, indicating severe disease) on the basis of the type, location, appearance, and depth of invasion of the lesions and the extent of disease and adhesions (see the table in the Supplementary Appendix, available with the full text of this article at NEJM.org). Although staging is useful in determining disease burden and management, the stage does not correlate with the severity of pain or predict the response to therapies for pain or infertility.21 Nonsurgical diagnostic approaches such as transvaginal ultrasonography and magnetic resonance imaging (MRI) perform poorly in the detection of peritoneal and ovarian implants and adhesions. However, both imaging methods perform well in detecting ovarian endometriomas, with ranges of 80 to 90% sensitivity and 60 to 98% specificity22 (Fig. 3). Because of its lower cost, transvaginal ultrasonography is preferred over MRI in the diagnosis of endometriomas. Doppler ultrasonography (Fig. 3A) may help in establishing the diagnosis; it shows characteristically scant blood flow to an endometrioma, normal flow to normal ovarian tissue, and enhanced flow to an ovarian tumor.22 Levels of CA-125 may be elevated in endometriosis, but this test is not recommended for diagnostic purposes because of poor sensitivity and specificity.23 The mean interval between the onset of pain and definitive (surgical) diagnosis is 10.4 years.24
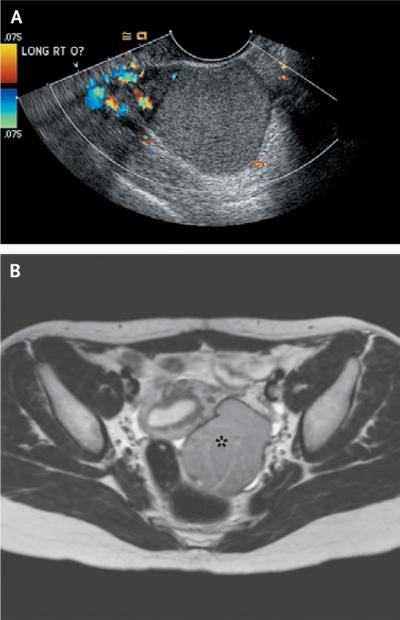
Figure 3. Radiographic Images of Endometriomas.
The transvaginal ultrasonogram in Panel A shows the ground-glass appearance of a 5-cm right ovarian endometrioma, with little flow to the mass but normal flow to the ovary. The red, yellow, and orange areas indicate blood flow toward the transducer, and the blue and green areas indicate blood flow away from the transducer. The T2-weighted magnetic resonance image in Panel B shows a left ovarian endometrioma (asterisk). (Images courtesy of Dr. Christopher Herndon, University of California, San Francisco.)
PAIN MANAGEMENT
Long-term treatment of patients with chronic pelvic pain associated with endometriosis involves repeated courses of medical therapy, surgical therapy, or both. In most cases, pain recurs within 6 to 12 months after completion of treatment.19,25
Medical Therapy
Empirical medical therapy is commonly initiated for pain control without surgical confirmation of disease. Such therapy is intended to reduce pain through a variety of mechanisms, including minimizing inflammation, interrupting or suppressing cyclic ovarian hormone production, inhibiting the action and synthesis of estradiol, and reducing or eliminating menses. Table 1 summarizes the indications for and side effects of various agents and approaches to the control of pain due to endometriosis.19,25
Table 1.
Medical and Surgical Therapies for Endometriosis-Related Pelvic Pain.*
| Treatment | Indication | Type of Therapy | Side Effects and Complications | Comments |
|---|---|---|---|---|
| Medical therapy | ||||
| NSAIDs | Dysmenorrhea | First-line | Nausea, vomiting, gastrointestinal irritation, drowsiness, headache | Initiate treatment at beginning of or just before menses; somewhat decreased menstrual flow |
| Combined oral contraceptives | ||||
| Cyclic | Dysmenorrhea | First-line | Nausea, weight gain, fluid retention, depression, breakthrough bleeding, breast tenderness, headache, decreased menstrual flow | |
| Continuous | Dysmenorrhea, noncyclic chronic pelvic pain | Second-line | Nausea, weight gain, fluid retention, depression, breakthrough bleeding, breast tenderness, headache, amenorrhea | |
| Progestins | ||||
| Medroxyprogesterone acetate | Dysmenorrhea, noncyclic chronic pelvic pain | Second-line | Nausea, weight gain, fluid retention, breakthrough bleeding, depression, amenorrhea, delayed return of ovulation | |
| Levonorgestrel intrauterine system | Dysmenorrhea, dyspareunia | Second- or third-line | Bloating, weight gain, headache, breast tenderness | Especially beneficial for symptomatic rectovaginal endometriosis; hypomenorrhea or amenorrhea for 6–12 mo; can be used for up to 5 yr; not FDA-approved for endometriosis |
| GnRH agonists | Dysmenorrhea, dyspareunia | Second- or third-line | Hypoestrogenism (vasomotor symptoms, vaginal dryness, decreased libido, irritability, loss of bone mineral density) | FDA-approved for endometriosis pain; estrogen-progestin add-back therapy used to mitigate loss of bone mineral density |
| Aromatase inhibitors | Dysmenorrhea, noncyclic chronic pelvic pain | Third-line | Hypoestrogenism, induction of ovulation | Combined with progestagens, combined oral contraceptives, and GnRH agonists because ovulation may be induced; not FDA-approved for endometriosis pain |
| Danazol | Dysmenorrhea, noncyclic chronic pelvic pain | Second- or third-line | Hyperandrogenic side effects (acne, edema, decreased breast size) | Side effects limit widespread use |
| Surgical therapy | ||||
| Laparoscopy | ||||
| Fulguration, ablation, and excision | Dysmenorrhea, noncyclic chronic pelvic pain, dyspareunia | First-or second-line | Risk associated with anesthesia and risk of infection, damage to internal organs, new adhesions, hemorrhage | First-line therapy for pelvic mass; commonly second-line therapy for pelvic pain resistant to medical therapy |
| Excision or drainage and ablation | Endometrioma >3 cm in diameter, chronic pelvic pain | First-line | Risk associated with anesthesia and risk of infection, damage to internal organs, new adhesions, hemorrhage | Potential for decreased ovarian reserve; excision is preferable to drainage and ablation |
| LPSN, nerve-pathway interruption (with conservative surgery) | Dysmenorrhea, dyspareunia, deep central pain | Third-line | Bleeding in the adjacent venous plexus, urinary urgency, constipation, painless first-stage labor | Technically challenging surgery; should be performed by surgeons with experience in LPSN |
| Hysterectomy, bilateral oo-phorectomy (abdominal, laparoscopic, total, or supracervical) | Noncyclic chronic pelvic pain | Fourth-line | Persistent or recurrent pain in 10% of patients, residual ovarian tissue | Reoperation may be necessary; measure FSH level to check ovarian remnant; add progestin to postoperative estrogen-replacement therapy for vasomotor symptoms |
| Adjunctive medical therapy after conservative surgery | ||||
| GnRH agonist | Dysmenorrhea, noncyclic chronic pelvic pain | Third-line | Hypoestrogenism | Used primarily in stage III or IV disease |
| Medroxyprogesterone acetate, danazol, combined oral contraceptives | Dysmenorrhea | Third-line | Medroxyprogesterone acetate: nausea, weight gain, fluid retention, breakthrough bleeding, depression; danazol: hyperandrogenic side effects (acne, edema, decreased breast size); combined oral contraceptives: nausea, weight gain, fluid retention, depression, breakthrough bleeding, breast tenderness, headache | Not commonly used |
FDA denotes Food and Drug Administration, FSH follicle-stimulating hormone, GnRH gonadotropin-releasing hormone, LPSN laparoscopic presacral neurectomy, and NSAID nonsteroidal antiinflammatory drug.
Nonsteroidal antiinflammatory drugs (NSAIDs) are commonly used to relieve dysmenorrhea, although one randomized, controlled trial showed no significant reduction in pain due to endometriosis with the use of NSAIDs as compared with placebo and no superiority of one NSAID over another.26 Combined oral contraceptives can be used cyclically or continuously for endometriosis-related pain and are commonly combined with NSAIDs, although they are associated with a 20 to 25% failure rate.19,25 This approach is first-line therapy in patients without contraindications to the use of combined oral contraceptives. A randomized, controlled trial27 showed the superiority of combined oral contraceptives over placebo in decreasing baseline pain scores for dysmenorrhea (by 45 to 52% vs. 14 to 17%, P<0.001) and the volume of ovarian endometriomas (by 48% vs. 32%, P = 0.04). In women with severe dysmenorrhea who have been treated with cyclic combined oral contraceptives, a switch to continuous combined oral contraceptives reduced pain scores by 58% within 6 months and by 75% at 2 years (P<0.001).28 Head-to-head randomized, nonblinded trials have shown that medroxyprogesterone acetate is as effective in controlling pain as combined oral contraceptives.29 In addition, in randomized, nonblinded studies, the levonorgestrel intrauterine system, which induces endometrial atrophy and associated amenorrhea, diminished endometriosis-associated pain and dysmenorrhea, as compared with regular follow-up with no treatment or treatment with a gonadotropin-releasing hormone (GnRH) agonist after conservative surgery.30
GnRH agonists effectively deplete the pituitary of endogenous gonadotropins and inhibit further synthesis, thus interrupting the menstrual cycle and resulting in a hypoestrogenic state, endometrial atrophy, and amenorrhea. In a systematic review of 15 randomized trials involving 1821 women, improvement in pain scores for dysmenorrhea with the use of GnRH agonists was 60 to 100%; these findings are similar to those with the use of danazol, antiprogestins, and combined oral contraceptives.31 Since GnRH agonist therapy has considerable side effects, including a hypoestrogenic state that may lead to bone loss of up to 13% over a period of 6 months (which is partly reversible on discontinuation of therapy), estrogen–progestagen add-back therapies are recommended.32 The “estrogen threshold hypothesis”33 suggests that maintaining estradiol levels between 30 and 45 pg per milliliter (109 and 164 pmol per liter) will maintain bone mineral density without stimulating disease. Indeed, scores for pelvic pain, tenderness, and dysmenorrhea improved with the use of regimens combining norethindrone acetate at a dose of 5 mg daily with a GnRH agonist, a conjugated equine estrogen at a dose of 0.625 mg, or both, but not when 5 mg of norethindrone acetate was combined with a higher dose (1.25 mg) of conjugated equine estrogen.34 At 1 year, bone mineral density was maintained at baseline levels in all groups that received add-back therapy. A meta-analysis of 15 randomized, controlled trials involving 910 women with symptomatic endometriosis revealed that estrogen–progestagen add-back therapy maintained bone density at the lumbar spine during and up to 12 months after GnRH agonist treatment.35 The effects of progestin-only add-back therapy on bone density have been inconsistent in studies involving adults35 and adolescents.36
Since endometriotic lesions express aromatase and synthesize their own estradiol (Fig. 2), suppression of ovarian estradiol production may not completely control pain. Limited studies involving small numbers of patients have shown that aromatase inhibitors (at doses lower than those used for breast-cancer treatment) are effective in reducing pelvic pain, with effects similar to those of other hormonal therapies.37 Aromatase inhibitors, however, are not approved by the Food and Drug Administration for endometriosis-related pain.
Danazol was an early treatment for endometriosis19; however, its androgenic side effects limit its clinical usefulness. Antiprogestagens such as mifepristone have been shown in small studies to reduce pain, but data from large randomized trials are lacking.20,29
Surgical Therapy
Surgical approaches to relieve endometriosis-related pain can be used as first-line therapy or initiated after failed medical therapies38 (Table 1). Surgical procedures include excision, fulguration, or laser ablation of endometriotic implants on the peritoneum, excision or drainage or ablation of endometriomas, resection of rectovaginal nodules, lysis of adhesions, and interruption of nerve pathways. Randomized, controlled trials have shown that at 6 months, laparoscopic ablation of endometriotic implants is 65% effective in reducing pain, as compared with a 22% rate of pain reduction associated with diagnostic laparoscopy alone.13 A small trial comparing laparoscopic ablation with GnRH agonist treatment showed similar pain reduction with the two approaches.29 Recurrence of pain requiring therapy is common (in 30 to 60% of patients) within 6 to 12 months after treatment.19,38 A combined analysis of data from two randomized trials involving 164 women that compared laparoscopic excision with drainage or ablation of endometriomas larger than 3 cm in diameter showed that excision resulted in less frequent recurrence of dysmenorrhea, dyspareunia, and pain, as well as reduced rates of further surgery.39
An alternative strategy for controlling endometriosis-related pain is interruption of nerve pathways. Whereas ablation of a segment of the uterosacral ligament has not proved effective, randomized, controlled trials have shown the superiority of laparoscopic ablation of endometriotic tissue combined with presacral neurectomy (removal of the nerve bundle within the boundaries of the interiliac triangle) over laparoscopic ablation alone in improving dysmenorrhea and reducing severe midline pain.40 Case series have shown that hysterectomy with bilateral salpingooophorectomy provided pain relief in 80 to 90% of women with debilitating symptoms that were refractory to medical or other surgical interventions; pain was reported to recur in 10% of the women within 1 to 2 years after surgery.19 Postoperative hormone replacement should include both estrogen and a progestagen, since estrogen alone may stimulate growth of microscopic disease.19
Adjunctive Medical Therapy
In women with advanced disease (stage III or IV), moderate-to-severe dysmenorrhea, and noncyclic pelvic pain, postoperative medical therapy may improve pain management by providing control of recurrent microscopic or residual disease. A meta-analysis of six randomized trials that compared 3 to 6 months of postoperative treatment with a GnRH agonist, danazol, or combined oral contraceptives with no postoperative treatment or placebo revealed a significant reduction in pain scores at the conclusion of therapy in the active-treatment groups, although the benefits were inconsistent with longer follow-up (to 18 months) after discontinuation of therapy.41 The mean interval between surgery and symptom recurrence requiring alternative therapy was significantly longer for patients who received postoperative treatment with GnRH agonists (>24 months) than for patients who received placebo (12 months).41
MANAGEMENT OF INFERTILITY
A large meta-analysis of randomized trials evaluating ovarian suppression with combined oral contraceptives, GnRH agonists, medroxyprogesterone acetate, or danazol as compared with placebo or no treatment in women with various stages of endometriosis showed no significant differences in spontaneous pregnancy or live birth rates.42 Thus, these agents are not recommended for the treatment of infertility and should not delay the pursuit of effective fertility therapies.18,25
Gonadotropin therapy and intrauterine insemination, as well as in vitro fertilization (IVF), are efficacious treatments in women with infertility and endometriosis.18,25 In a large randomized trial comparing four treatment strategies in 932 couples with stage I or II endometriosis or unexplained infertility, cumulative pregnancy rates during four treatment cycles were as follows: intracervical insemination (10%), intrauterine insemination (18%), gonadotropin therapy and intracervical insemination (19%), and gonadotropin therapy and intrauterine insemination (33%).43 A meta-analysis of 14 randomized, controlled trials showed that women with endometriosis were less likely than women with tubal-factor infertility to conceive by means of IVF (odds ratio, 0.81; 95% confidence interval [CI], 0.72 to 0.91).44 In a systematic review of three randomized trials including 165 women with advanced endometriosis and infertility, GnRH agonist therapy for 3 to 6 months before IVF, as compared with no treatment before this procedure, significantly increased the live birth rate (odds ratio, 9.19; 95% CI, 1.08 to 78.22).45
Ablation of endometriotic lesions with lysis of adhesions is recommended for the treatment of infertility related to stage I or II endometriosis.18,25 In a controlled trial involving 341 women with infertility who underwent diagnostic laparoscopy, those randomly assigned to ablation of stage I or II endometriotic lesions had a significantly higher cumulative pregnancy rate at 3 years than untreated patients (31% vs. 17%).46 A smaller trial did not show a significantly higher pregnancy rate with laparoscopic ablation, but a meta-analysis combining these trials showed a significant difference in rates of pregnancy and live births between groups.46 In an observational study involving 216 women with infertility and severe endometriosis, the cumulative pregnancy rate at 2 years was 63% among those who had undergone laparotomy with treatment of lesions and adhesions, as compared with 45% among those who had undergone laparoscopy alone.47 Two randomized trials showed that excision of endometriomas larger than 3 cm in diameter, as compared with drainage and ablation, resulted in significantly higher pregnancy rates,39 although ovarian surgery may diminish ovarian reserve in women with advanced disease.18,25
AREAS OF UNCERTAINTY
There is a lack of data from randomized trials to inform the optimal management of endometriosis (medical vs. surgical) with respect to pain relief, pain recurrence, and future fertility. Studies of interventions for pelvic pain often have high rates of placebo effects (approximately 40 to 45% improvement in symptoms). A recently proposed scoring system specifically for endometriosis-related chronic pelvic pain48 awaits validation. A noninvasive diagnostic test with high sensitivity and specificity for endometriosis is lacking, although transcriptomic and proteomic approaches are under study. There is a lack of data from randomized trials evaluating the effects of different surgical therapies and robotic-assisted laparoscopy on pain and fertility, as compared with each other and with medical therapies, and assessing the effects of different add-back therapies on pain and on bone density. It is uncertain whether leaving endometriosis untreated accelerates the age-related decline in fertility. Although the pathogenesis of endometriosis and associated pain and infertility remains incompletely understood, therapies aimed at correcting progesterone resistance (e.g., selective progesterone-receptor modulators) and systemic immune dysfunction, as well as those targeting angiogenesis, inflammation, neurotropism, and pain transmission, including a neuropathic component, warrant further study. Oral GnRH antagonists and other small molecules that suppress circulating estradiol levels to the range suggested by the estrogen threshold hypothesis (30 to 45 pg per milliliter)49 also warrant investigation. Studies of complementary or alternative therapies are needed. A randomized, sham-controlled trial involving 18 adolescents and young women showed the efficacy of Japanese-style acupuncture for endometriosis-related pain,50 and small observational studies suggest reductions in endometriosis-associated pain after transcutaneous electrical nerve stimulation, hypogastric nerve block, physical therapy, or complementary medicine approaches14; however, data from large, randomized, controlled trials to confirm these findings are lacking.
GUIDELINES
Several professional organizations have published guidelines for the evaluation and treatment of endometriosis-related pain and infertility. Table 2 lists key recommendations of these societies,16,18,19,25,51 which involve a multidisciplinary approach, including psychological support, for women with chronic pelvic pain, infertility, or both. The recommendations provided here are generally concordant with these guidelines.
Table 2.
Major Guidelines from Professional Societies for the Diagnosis and Management of Endometriosis-Related Pain and Infertility.*
| Condition | Recommendation |
|---|---|
| Pain† | |
| Diagnosis | Surgery is the preferred method for the diagnosis of pelvic pain and a pelvic mass (e.g., endometrioma), but it is not required before initiating empirical therapy, after consideration of other conditions in a differential diagnosis. There should be a low threshold for the evaluation of endometriosis in adolescents because the diagnosis is often missed in this age group. |
| Treatment | Initial treatment is a trial of nonsteroidal antiinflammatory drugs and hormonal therapy (combined oral contraceptives). All hormonal drugs that have been studied (combined oral contraceptives, progestins, GnRH agonists, and danazol) are similarly effective, but their side effects and costs differ. If a GnRH agonist is used, estrogen-progestin add-back therapy is recommended; GnRH agonists are not recommended for adolescents because of their effects on bone. The levonorgestrel intrauterine system is effective in selected patients. Laparoscopic uterosacral nerve ablation is not effective. |
| Infertility | |
| Diagnosis | Both the male and female partner should undergo a full evaluation. |
| Treatment | Superovulation with intrauterine insemination provides a benefit. Ovarian suppression is not effective in promoting spontaneous pregnancy. The use of a GnRH agonist for 3–6 mo before in vitro fertilization and surgical ablation of endometriosis for stage I or II disease are beneficial. Excision of endometriomas >;3 cm in diameter is of benefit, although there is potential for diminished ovarian reserve. |
CONCLUSIONS AND RECOMMENDATIONS
The patient described in the vignette has symptoms of pain that are highly suggestive of endometriosis. After a thorough medical, surgical, gynecologic, and family history has been obtained, a pelvic examination should be performed. If the pelvic examination reveals adnexal pain or tenderness with or without fullness, the patient should undergo transvaginal ultrasonography to look for an ovarian endometrioma or other pelvic disease, although peritoneal disease will not be detected by this imaging method. In patients such as the woman described in the vignette, NSAIDs and cyclical combined oral contraceptives are recommended as first-line therapy in the absence of contraindications. If the pain persists, a switch to continuous combined oral contraceptives for 3 to 6 months or a levonorgestrel intrauterine system is warranted. If these approaches are not effective, GnRH agonist therapy with estrogen–progestin add-back therapy is appropriate. Laparoscopy would be indicated to evaluate and treat persistent pain, a pelvic mass, or both. The patient should be counseled about the association of endometriosis with infertility, but she should also be reassured that she may not have a problem conceiving and that treatment for endometriosis-associated infertility is often effective.
Footnotes
This Journal feature begins with a case vignette highlighting a common clinical problem. Evidence supporting various strategies is then presented, followed by a review of formal guidelines, when they exist. The article ends with the author's clinical recommendations.
Dr. Giudice reports receiving consulting fees from Bayer Schering Pharma, Neurocrine Biosciences, Endo Pharmaceuticals, and Schering-Plough, holding stock in Merck and Pfizer, and serving on advisory committees for the Endometriosis Association and Baylor College of Medicine. No other potential conflict of interest relevant to this article was reported.
Disclosure forms provided by the author are available with the full text of this article at NEJM.org.
REFERENCES
- 1.Giudice LC, Swiersz RO. LM, Burney Endometriosis. In: Jameson JL, De Groot LJ, editors. Endocrinology. 6th ed. Elsevier; New York: 2010. pp. 2356–70. [Google Scholar]
- 2.Goldstein DP, deCholnoky C, Emans SJ, Leventhal JM. Laparoscopy in the diagnosis and management of pelvic pain in adolescents. J Reprod Med. 1980;24:251–6. [PubMed] [Google Scholar]
- 3.Eskenazi B, Warner ML. Epidemiology of endometriosis. Obstet Gynecol Clin North Am. 1997;24:235–58. doi: 10.1016/s0889-8545(05)70302-8. [DOI] [PubMed] [Google Scholar]
- 4.Bulun SE. Endometriosis. N Engl J Med. 2009;360:268–79. doi: 10.1056/NEJMra0804690. [DOI] [PubMed] [Google Scholar]
- 5.Berkley KJ, Rapkin AJ, Papka RE. The pains of endometriosis. Science. 2005;308:1587–9. doi: 10.1126/science.1111445. [DOI] [PubMed] [Google Scholar]
- 6.Tokushige N, Markham R, Russell P, Fraser IS. Nerve fibres in peritoneal endometriosis. Hum Reprod. 2006;21:3001–7. doi: 10.1093/humrep/del260. [DOI] [PubMed] [Google Scholar]
- 7.Sanfilippo JS, Wakim NG, Schikler KN, Yussman MA. Endometriosis in association with uterine anomaly. Am J Obstet Gynecol. 1986;154:39–43. doi: 10.1016/0002-9378(86)90389-3. [DOI] [PubMed] [Google Scholar]
- 8.Missmer SA, Hankinson SE, Spiegel-man D, Barbieri RL, Michels KB, Hunter DJ. In utero exposures and the incidence of endometriosis. Fertil Steril. 2004;82:1501–8. doi: 10.1016/j.fertnstert.2004.04.065. [DOI] [PubMed] [Google Scholar]
- 9.Missmer SA, Hankinson SE, Spiegel-man D, Barbieri RL, Marshall LM, Hunter DJ. Incidence of laparoscopically confirmed endometriosis by demographic, anthropometric, and lifestyle factors. Am J Epidemiol. 2004;160:784–96. doi: 10.1093/aje/kwh275. [DOI] [PubMed] [Google Scholar]
- 10.Diamanti-Kandarakis E, Bourguignon JP, Giudice LC, et al. Endocrine-disrupting chemicals: an Endocrine Society scientific statement. Endocr Rev. 2009;30:293–342. doi: 10.1210/er.2009-0002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Montgomery GW, Nyholt DR, Zhao ZZ, et al. The search for genes contributing to endometriosis risk. Hum Reprod Update. 2008;14:447–57. doi: 10.1093/humupd/dmn016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Missmer SA, Chavarro JE, Malspeis S, et al. A prospective study of dietary fat consumption and endometriosis risk. Hum Reprod. 2010 March 23; doi: 10.1093/humrep/deq044. Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sutton CJ, Pooley AS, Ewen SP, Haines P. Follow-up report on a randomized controlled trial of laser laparoscopy in the treatment of pelvic pain associated with minimal to moderate endometriosis. Fertil Steril. 1997;68:1070–4. doi: 10.1016/s0015-0282(97)00403-2. [DOI] [PubMed] [Google Scholar]
- 14.D'Hooghe T, Hummelshoj L. Multi-disciplinary centres/networks of excellence for endometriosis management and research: a proposal. Hum Reprod. 2006;21:2743–8. doi: 10.1093/humrep/del123. [DOI] [PubMed] [Google Scholar]
- 15.Simoens S, Hummelshoj L, D'Hooghe T. Endometriosis: cost estimates and methodological perspective. Hum Reprod Update. 2007;13:395–404. doi: 10.1093/humupd/dmm010. [DOI] [PubMed] [Google Scholar]
- 16.ACOG Practice Bulletin no. 51: chronic pelvic pain. Obstet Gynecol. 2004;103:589–605. [PubMed] [Google Scholar]
- 17.Ripps BA, Martin DC. Focal pelvic tenderness, pelvic pain and dysmenorrhea in endometriosis. J Reprod Med. 1991;36:470–2. [PubMed] [Google Scholar]
- 18.Practice Committee of the American Society for Reproductive Medicine Endometriosis and infertility. Fertil Steril. 2006;86(Suppl):S156–S160. doi: 10.1016/j.fertnstert.2006.08.014. [DOI] [PubMed] [Google Scholar]
- 19.Idem Treatment of pelvic pain associated with endometriosis. Fertil Steril. 2008;90(Suppl):S260–S269. doi: 10.1016/j.fertnstert.2008.08.057. [DOI] [PubMed] [Google Scholar]
- 20.Evans S, Moalem-Taylor G, Tracey DJ. Pain and endometriosis. Pain. 2007;132(Suppl 1):S22–S25. doi: 10.1016/j.pain.2007.07.006. [DOI] [PubMed] [Google Scholar]
- 21.Revised American Society for Reproductive Medicine classification of endometriosis: 1996. Fertil Steril. 1997;67:817–21. doi: 10.1016/s0015-0282(97)81391-x. [DOI] [PubMed] [Google Scholar]
- 22.Brosens I, Puttemans P, Campo R, Gordts S, Kinkel K. Diagnosis of endometriosis: pelvic endoscopy and imaging techniques. Best Pract Res Clin Obstet Gynaecol. 2004;18:285–303. doi: 10.1016/j.bpobgyn.2004.03.002. [DOI] [PubMed] [Google Scholar]
- 23.Mol BW, Bayram N, Lijmer JG, et al. The performance of CA-125 measurement in the detection of endometriosis: a meta-analysis. Fertil Steril. 1998;70:1101–8. doi: 10.1016/s0015-0282(98)00355-0. [DOI] [PubMed] [Google Scholar]
- 24.Hadfield R, Mardon H, Barlow D, Kennedy S. Delay in the diagnosis of endometriosis: a survey of women from the USA and the UK. Hum Reprod. 1996;11:878–80. doi: 10.1093/oxfordjournals.humrep.a019270. [DOI] [PubMed] [Google Scholar]
- 25.Kennedy S, Bergqvist A, Chapron C, et al. ESHRE guideline for the diagnosis and treatment of endometriosis. Hum Reprod. 2005;20:2698–704. doi: 10.1093/humrep/dei135. [DOI] [PubMed] [Google Scholar]
- 26.Allen C, Hopewell S, Prentice A. Nonsteroidal antiinflammatory drugs for pain in women with endometriosis. Cochrane Database Syst Rev. 2005;4:CD004753. doi: 10.1002/14651858.CD004753.pub2. [DOI] [PubMed] [Google Scholar]
- 27.Harada T, Momoeda M, Taketani Y, Hoshiai H, Terakawa N. Low-dose oral contraceptive pill for dysmenorrhea associated with endometriosis: a placebo-controlled, double-blind, randomized trial. Fertil Steril. 2008;90:1583–8. doi: 10.1016/j.fertnstert.2007.08.051. [DOI] [PubMed] [Google Scholar]
- 28.Vercellini P, Frontino G, De Giorgi O, Pietropaolo G, Pasin R, Crosignani PG. Continuous use of an oral contraceptive for endometriosis-associated recurrent dysmenorrhea that does not respond to a cyclic pill regimen. Fertil Steril. 2003;80:560–3. doi: 10.1016/s0015-0282(03)00794-5. [DOI] [PubMed] [Google Scholar]
- 29.Vercellini P, Cortesi I, Crosignani PG. Progestins for symptomatic endometriosis: a critical analysis of the evidence. Fertil Steril. 1997;68:393–401. doi: 10.1016/s0015-0282(97)00193-3. [DOI] [PubMed] [Google Scholar]
- 30.Abou-Setta AM, Al-Inany HG, Farquhar CM. Levonorgestrel-releasing intrauterine device (LNG-IUD) for symptomatic endometriosis following surgery. Cochrane Database Syst Rev. 2006;4:CD005072. doi: 10.1002/14651858.CD005072.pub2. [DOI] [PubMed] [Google Scholar]
- 31.Prentice A, Deary AJ, Goldbeck-Wood S, Farquhar C, Smith SK. Gonadotrophin-releasing hormone analogues for pain associated with endometriosis. Cochrane Database Syst Rev. 2000;2:CD000346. doi: 10.1002/14651858.CD000346. [DOI] [PubMed] [Google Scholar]
- 32.Al Kadri H, Hassan S, Al-Fozan HM, Hajeer A. Hormone therapy for endometriosis and surgical menopause. Cochrane Database Syst Rev. 2009;1:CD005997. doi: 10.1002/14651858.CD005997.pub2. [DOI] [PubMed] [Google Scholar]
- 33.Barbieri RL. Hormone treatment endometriosis: the estrogen threshold hypothesis. Am J Obstet Gynecol. 1992;166:740–5. doi: 10.1016/0002-9378(92)91706-g. [DOI] [PubMed] [Google Scholar]
- 34.Hornstein MD, Surrey ES, Weisberg GW, Casino LA. Leuprolide acetate depot and hormonal add-back in endometriosis: a 12-month study. Obstet Gynecol. 1998;91:16–24. doi: 10.1016/s0029-7844(97)00620-0. [DOI] [PubMed] [Google Scholar]
- 35.Sagsveen M, Farmer JE, Prentice A, Breeze A. Gonadotrophin-releasing hormone analogues for endometriosis: bone mineral density. Cochrane Database Syst Rev. 2003;4:CD001297. doi: 10.1002/14651858.CD001297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Divasta AD, Laufer MR, Gordon CM. Bone density in adolescents treated with a GnRH agonist and add-back therapy for endometriosis. J Pediatr Adolesc Gynecol. 2007;20:293–7. doi: 10.1016/j.jpag.2007.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Nawathe A, Patwardhan S, Yates D, Harrison GR, Khan KS. Systematic review of the effects of aromatase inhibitors on pain associated with endometriosis. BJOG. 2008;115:818–22. doi: 10.1111/j.1471-0528.2008.01740.x. [DOI] [PubMed] [Google Scholar]
- 38.Jacobson TZ, Duffy JM, Barlow D, Koninckx PR, Garry R. Laparoscopic surgery for pelvic pain associated with endometriosis. Cochrane Database Syst Rev. 2009;4:CD001300. doi: 10.1002/14651858.CD001300.pub2. [DOI] [PubMed] [Google Scholar]
- 39.Hart RJ, Hickey M, Maouris P, Buckett W. Excisional surgery versus ablative surgery for ovarian endometriomata. Cochrane Database Syst Rev. 2008;2:CD004992. doi: 10.1002/14651858.CD004992.pub3. [DOI] [PubMed] [Google Scholar]
- 40.Proctor ML, Latthe PM, Farquhar CM, Khan KS, Johnson NP. Surgical interruption of pelvic nerve pathways for primary and secondary dysmenorrhoea. Cochrane Database Syst Rev. 2005;4:CD001896. doi: 10.1002/14651858.CD001896.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yap C, Furness S, Farquhar C. Pre and post operative medical therapy for endometriosis surgery. Cochrane Database Syst Rev. 2004;3:CD003678. doi: 10.1002/14651858.CD003678.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hughes E, Brown J, Collins JJ, Farquhar C, Fedorkow DM, Vandekerckhove P. Ovulation suppression for endometriosis. Cochrane Database Syst Rev. 2007;3:CD000155. doi: 10.1002/14651858.CD000155.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Guzick DS, Carson SA, Coutifaris C, et al. Efficacy of superovulation and intrauterine insemination in the treatment of infertility. N Engl J Med. 1999;340:177–83. doi: 10.1056/NEJM199901213400302. [DOI] [PubMed] [Google Scholar]
- 44.Barnhart K, Dunsmoor-Su R, Coutifaris C. Effect of endometriosis on in vitro fertilization. Fertil Steril. 2002;77:1148–55. doi: 10.1016/s0015-0282(02)03112-6. [DOI] [PubMed] [Google Scholar]
- 45.Sallam HN, Garcia-Velasco JA, Dias S, Arici A. Long-term pituitary down-regulation before in vitro fertilization (IVF) for women with endometriosis. Cochrane Database Syst Rev. 2006;1:CD004635. doi: 10.1002/14651858.CD004635.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Jacobson TZ, Barlow DH, Koninckx PR, Olive D, Farquhar C. Laparoscopic surgery for subfertility associated with endometriosis. Cochrane Database Syst Rev. 2002;4:CD001398. doi: 10.1002/14651858.CD001398. [DOI] [PubMed] [Google Scholar]
- 47.Al-Inany HG. Evidence may change with more trials: concepts to be kept in mind. Hum Reprod. 2000;15:2447–8. [PubMed] [Google Scholar]
- 48.Vincent K, Kennedy S, Stratton P. Pain scoring in endometriosis: entry criteria and outcome measures for clinical trials: report from the Art and Science of Endometriosis meeting. Fertil Steril. 2010;93:62–7. doi: 10.1016/j.fertnstert.2008.09.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Struthers RS, Nicholls AJ, Grundy J, et al. Suppression of gonadotropins and estradiol in premenopausal women by oral administration of the nonpeptide gonadotropin-releasing hormone antagonist elagolix. J Clin Endocrinol Metab. 2009;94:545–51. doi: 10.1210/jc.2008-1695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wayne PM, Kerr CE, Schnyer RN, et al. Japanese-style acupuncture for endometriosis-related pelvic pain in adolescents and young women: results of a randomized sham-controlled trial. J Pediatr Adolesc Gynecol. 2008;21:247–57. doi: 10.1016/j.jpag.2007.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.ACOG committee opinion: number 310, April 2005: endometriosis in adolescents. Obstet Gynecol. 2005;105:921–7. doi: 10.1097/00006250-200504000-00058. [DOI] [PubMed] [Google Scholar]