Abstract
Aims/hypothesis
The aim of this study was to investigate the genetic aetiology of permanent diabetes mellitus with onset in the first 12 months of age.
Methods
We studied 46 probands with permanent, insulin-requiring diabetes with onset within the first 6 months of life (permanent neonatal diabetes mellitus [PNDM]/monogenic diabetes of infancy [MDI]) (group 1) and eight participants with diabetes diagnosed between 7 and 12 months of age (group 2). KCNJ11, INS and ABCC8 genes were sequentially sequenced in all patients. For those who were negative in the initial screening, we examined ERN1, CHGA, CHGB and NKX6-1 genes and, in selected probands, CACNA1C, GCK, FOXP3, NEUROG3 and CDK4. The incidence rate for PNDM/MDI was calculated using a database of Italian patients collected from 1995 to 2009.
Results
In group 1 we found mutations in KCNJ11, INS and ABCC8 genes in 23 (50%), 9 (19.5%) and 4 (8.6%) patients respectively, and a single homozygous mutation in GCK (2.1%). In group 2, we identified one incidence of a KCNJ11 mutation. No genetic defects were detected in other loci. The incidence rate of PNDM/MDI in Italy is estimated to be 1:210,287.
Conclusions/interpretation
Genetic mutations were identified in ~75% of non-consanguineous probands with PNDM/MDI, using sequential screening of KCNJ11, INS and ABCC8 genes in infants diagnosed within the first 6 months of age. This percentage decreased to 12% in those with diabetes diagnosed between 7 and 12 months. Patients belonging to the latter group may either carry mutations in genes different from those commonly found in PNDM/MDI or have developed an early-onset form of autoimmune diabetes.
Electronic supplementary material
The online version of this article (doi:10.1007/s00125-011-2094-8) contains supplementary material, which is available to authorised users.
Keywords: ABCC8 gene, Infancy-onset diabetes mellitus, INS gene, KCNJ11 gene, Monogenic diabetes of infancy, Neonatal diabetes mellitus, Non-syndromic diabetes
Introduction
Permanent diabetes with onset within the first 6 months following birth (permanent neonatal diabetes mellitus [PNDM]/monogenic diabetes of infancy [MDI]) is a rare condition associated with defects in genes that play major roles in pancreatic beta cell development and function; mutations in the genes encoding the ATP-sensitive potassium channel (KATP) subunits, KCNJ11 (also known as KIR6.2), ABCC8 (also known as SUR1) and insulin (INS), account for most cases of PNDM/MDI [1–6]. Mutations in both the KATP channel and INS genes can also cause diabetes with onset in childhood and adulthood [7–9]. In addition, KATP channel mutations are a prominent cause of transient neonatal diabetes mellitus (TNDM), a form of neonatal/infancy-onset diabetes that usually remits within 6 months of its onset [1, 2, 4]. Mutations in KATP channel genes that cause PNDM/MDI are usually activating, dominant and sporadic, but patients with recessive mutations in ABCC8 have also been described [10]. Patients with INS gene mutations, which can be either dominant [5, 6] or recessive [11], present with non-syndromic diabetes. In contrast, patients carrying mutations to KATP genes may also show variable degrees of motor/mental developmental delay with epilepsy (developmental delay, epilepsy, neonatal diabetes; DEND) or without epilepsy (intermediate DEND; iDEND). Moreover, individuals with some exceedingly rare forms of syndromic PNDM may bear recessive mutations in several genes, including PDX1, EIF2AK3, PTF1A, GLIS3, RFX6 or FOXP3 [12–18], while recessive mutations in glucokinase give rise to isolated neonatal diabetes [19]. In 2002 we reported the basis for the currently used definition of PNDM/MDI by providing strong evidence that permanent diabetes with onset within 6 months of life is not autoimmune, but rather genetic, in origin [20]. This conclusion was supported by the absence of type 1 diabetes mellitus autoantibodies in patients presenting with diabetes in the first 180 days of life. In the present investigation we have assessed the power of this temporal cut-off in defining neonatal/infancy-onset diabetes by performing sequential screening of KCNJ11, INS and ABCC8 genes in 54 patients with permanent diabetes: 46 patients with onset of the disease within 6 months from birth, and eight patients between 7 months and 1 year of age. In those who were negative for mutations in the initial screening, five other candidate genes (ERN1, CHGA, CHGB, NKX6-1 and CACNA1C) were sequenced. In specific patients, GCK, FOXP3, NEUROG3 and CDK4 genes and gene copy number variations were also evaluated.
Methods
Probands
A total of 46 patients with diabetes onset before 6 months of age (17, or 37%, with onset within the first 6 weeks of life) (group 1) and eight patients with diabetes diagnosed between 7 and 12 months (group 2) were included in this study. Of these 54 patients, 22 patients in group 1 and two in group 2 have not been reported previously. Most probands studied in the present investigation were of Italian descent, except for one of Moroccan (nd-VI/1), one of Albanian (nd-MI/3), one of Chinese (nd-MO/3) and one of Masai descent (nd-RM/6; Table 1). Family history disclosed consanguinity only in the Albanian family. In group 1, a male patient (deceased; nd-BR/1) presented with a phenotype resembling IPEX (immune dysregulation, polyendocrinopathy, enteropathy, X-linked) syndrome [18]. Two other infants had syndromic diabetes with a phenotype that was different from DEND/iDEND, or any other form caused by known genes [12–17]. One of these (group 1) suffered from episodes of liver insufficiency, with high ammonium and transaminase levels and low albumin, but with a mute liver biopsy; he also presented with anaemia that was successfully treated with erythropoietin (nd-NA/2). The other patient (group 2) showed muscle hypotrophy in the lower limbs, delayed puberty and retarded growth (mdi-RM-OBG/1).
Table 1.
Clinical and genetic features of patients with diabetes onset within the first year of life studied in the present investigation
| Patient | T1D autoantibodies tested | Age at onset (days) | Gene variant | Other features | SU treatment |
|---|---|---|---|---|---|
| Group 1 | |||||
| nd-VI/1 | ICA, GADA, IA-2A | 1 | KCNJ11/V59A | DEND | Yes |
| nd-BR/1 | None | 2 | – | Diarrhoea | |
| nd-RM/4 | IAA, GADA, IA-2A | 2 | KCNJ11/R201S | Yes | |
| nd-MI/3 | IAA, GADA, IA-2A, ZnT8A | 2 | KCNJ11/R201C | Yes | |
| nd-PD/2 | None | 3 | ABCC8/L213P | DEND | Yes |
| nd-FI/1 | None | 15 | ABCC8/V324M; ABCC8/W688R | Yes | |
| nd-CT/2 | None | 27 | – | ||
| nd-MI/2 | ICA, GADA, IA-2A | 38 | KCNJ11/K170R | Yes | |
| nd-LE/2 | ICA, IAA, GADA, IA-2A | 39 | – | ||
| nd-PR/2 | None | 40 | ABCC8/L213P | iDEND | Yes |
| nd-NA/1 | None | 40 | KCNJ11/R201C | Yes + insulin | |
| nd-CT/1 | none | 60 | KCNJ11/V59M | iDEND | Yes |
| nd-NA/2 | ICA, GADA, IA-2A | 71 | ABCC8/A355T | Anaemia | Yes + insulin |
| nd-MO/3 | ICA, IAA, GADA | 73 | KCNJ11/H46Y | Yes | |
| nd-RM/4 | IAA, GADA, IA-2A | 80 | – | ||
| nd-TO/3 | GADA, IA-2A | 82 | – | ||
| nd-TS/2 | None | 120 | KCNJ11/V59M | iDEND | Yes |
| nd-RM/6 | None | 120 | KCNJ11/R195Ha | ||
| nd-RM/5 | IAA, GADA, IA-2A | 135 | KCNJ11/E322K | Yes | |
| nd-PI/1 | ICA | 141 | – | ||
| nd-BG/1 | GADA | 180 | ABCC8/S1054Na | ||
| nd-CES/3 | None | 190 | – | ||
| Group 2 | |||||
| mdi-RM/3 | None | 220 | KCNJ11/V59M | iDEND | Yes |
| mdi/NA-B/1 | ICA | 251 | – | ||
| mdi-PA/1 | ICA, IAA, GADA, IA-2A | 270 | – | ||
| mdi-RM-OBG/1 | IAA, GADA | 289 | – | Muscle hypotrophy | |
| mdi-CES/1 | ICA, IAA | 300 | – | ||
| mdi-RM-OBG/3 | IAA, GADA, IA-2A | 330 | – | ||
| mdi-RM-OBG/2 | IAA, GADA, IA-2A | 330 | – | ||
| mdi-NA/2 | GADA, IA-2A | 354 | – | ||
SU treatment denotes complete withdrawal of insulin therapy unless specified. Mutations KCNJ11/H46Y and KCNJ11/V59A were found in probands with Chinese and Moroccan ancestry, respectively. The Albanian patient carried mutation KCNJ11/R201C
T1D type 1 diabetes, SU sulfonylurea
aBenign gene variants
Type 1 diabetes autoantibodies
Twenty-seven patients in group 1 and all patients in group 2 tested negative for at least one type 1 diabetes autoantibody (i.e. ICA [islet cell autoantibodies], IAA [insulin autoantibodies], GADA [glutamic acid decarboxylase autoantibodies], IA-2A [tyrosine phosphatase-related proteins islet antigen 2 autoantibodies] or ZnT8A [zinc transporter 8 autoantibodies]) evaluated at the time of diagnosis.
Genetic testing
Informed consent was obtained at each local paediatric diabetes centre involved in the study. Genomic DNA was extracted from peripheral lymphocytes by DNeasy Tissue Isolation kit (Qiagen, Valencia, CA, USA), amplified by PCR and sequenced using a DNA automated sequencer (Applied Biosystems ABI 3730, Foster City, CA, USA). Based on their frequency, our current routine for new cases is to sequence KCNJ11 and INS genes at the same time and, if no mutation is found, the larger ABCC8 gene is examined [4, 6, 21]. In those patients where no mutation was identified, the entire coding region of the following candidate genes, ERN1, CHGA, CHGB, NKX6-1 (in patients in groups 1 and 2) and CACNA1C (in group 1 only), were sequenced. CACNA1C [22], which encodes voltage-dependent L-type calcium channel subunit alpha-1C (CAV1.2), CHGA, CHGB [23] and ERN1 [24] were chosen for their role in insulin secretion; NKX6-1 was selected because of its role in endocrine pancreas development [25]. In particular, ERN1 encodes inositol-requiring protein 1 (IRE1A), which is involved in the unfolded protein response and insulin biosynthesis [24]. FOXP3 and NEUROG3 were sequenced in the proband with IPEX-like features [18, 26], CDK4 in the proband with diabetes and muscle hypotrophy [27], and GCK in patients presenting with diabetes onset in the first week of life, together with low birthweight [19]. Primer sequences and PCR conditions for candidate genes are listed in Table 1 of the electronic supplementary material (ESM).
New variants in KATP genes were searched by DNA direct sequencing of genomic DNA from 50 normal controls.
The multiplex ligation-dependent probe amplification (MLPA) technique (MRC-Holland, Amsterdam, the Netherlands) was applied to search for ABCC8 gene deletions. In two patients with syndromic diabetes, copy number variations (CNVs) were also assessed. Patients’ genomic DNA was analysed with the GeneChip 6.0 microarray (Affymetrix, Santa Clara, CA, USA). Labelled DNA was hybridised for 16–18 h; the chip was washed, stained and scanned using a Scanner 3000 7G (Affymetrix). The generated file, containing a single intensity value calculated for each probe, was loaded into Genotyping Console 3.0.2 and the SNP 6.0 copy number calls from 270 International HapMap Project control samples (www.hapmap.org/) were used as a reference model file for comparison. CNVs were selected based on variables previously described [28].
PNDM/MDI incidence in Italy
Information was obtained from the Italian network database on early-onset diabetes (presentation within the first 24 months of life). The database works under the auspices of the Italian Society of Pediatric Endocrinology and Diabetology (ISPED) and collects clinical data from 45 paediatric diabetes clinics, which are the referral centres for the diagnosis and treatment of patients with neonatal and childhood diabetes in Italy.
Incidence rates per 100,000 live births were computed for cases that developed diabetes in the first 6 months of life and were born between 1995 and 2009. For comparison, we also calculated incidence rates utilising the old (1995) limit of ≤42 days of life for the diagnosis of PNDM [29]. Confidence intervals for incidence rates were estimated using the exact Poisson distribution.
Results
Disease-causing mutations in KATP genes
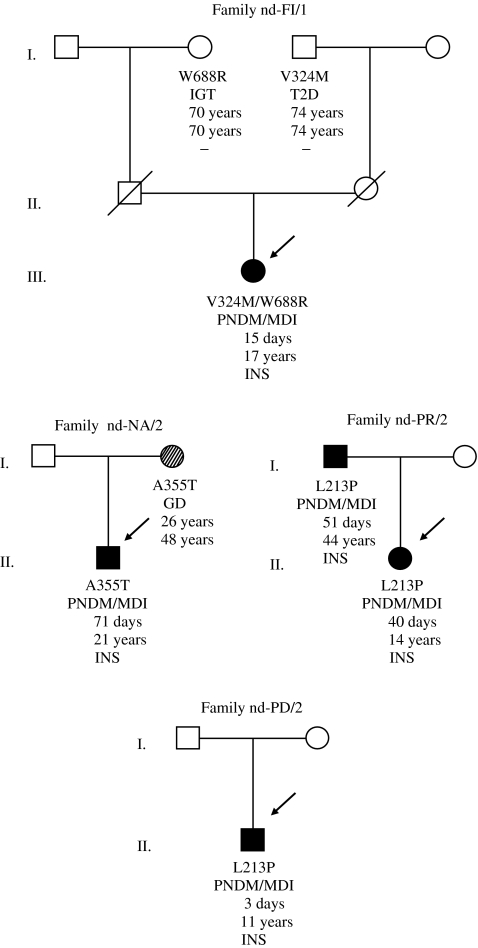
In group 1, heterozygous mutations in the KCNJ11 gene were identified in 23 patients (50%), including 14 probands previously reported by our group [21, 30–32]. All mutations were considered to be de novo, because parental DNA sequencing disclosed wild-type KCNJ11 genes. Table 1 shows mutations recently or previously identified in patients with PNDM/MDI, including H46Y (nd-MO/3) [33], V59A (nd-VI/1) [34], V59M, K170R, R201C and E322K (nd-RM/25; Table 1) [35]. We also identified a new substitution, arginine by serine, in codon 201 (R201S, c.601C>A), that we believe is novel (nd-RM/4). In Group 2, we found the previously described KCNJ11/V59M mutation [3, 21] in a patient with diabetes diagnosed at 220 days of life (mdi-RM/3). ABCC8 gene sequencing led to the identification of five heterozygous mutations in four probands from group 1. ABCC8/L213P (c.638T>C) was found to be a de novo mutation in a patient with complete DEND syndrome (nd-PD/2), and in a second proband with iDEND, who had inherited the mutation from her father (Fig. 1), who also presented with iDEND (nd-PR/2). A mutation in the same codon, ABCC8/L213R, has been previously reported in association with iDEND [36]. Of our patients, one proband was a compound heterozygote for the ABCC8/V324M mutation, previously described in patients with the transient form of the disease (TNDM) [36], who usually need insulin therapy for less than 1 year [4], and for the novel W688R (c.2062T>G) mutation (nd-FI/1). Residue W688 is conserved in dog, cow, mouse, chicken and zebra fish. Because both of the patient’s parents were deceased, we analysed the grandparents’ DNA and confirmed the ABCC8/V324M mutation in the maternal grandfather and the ABCC8/W688R mutation in the paternal grandmother (Fig. 1). After identifying the mutations, an OGTT was done in both patients; the carrier for V324M was diagnosed with diabetes (2 h plasma glucose 14 mmol/l) and the carrier for W688R presented with impaired glucose tolerance (2 h plasma glucose 8.4 mmol/l). Finally, a patient carrying the unreported ABCC8/A355T (c.1063G>A; nd-NA/2) variant inherited the mutation from his mother, who was diagnosed with gestational diabetes at the age of 26 years. Residue A355 is conserved in dog, cow, rat, mouse and chicken, but not in zebra fish, where it is substituted by glycine. None of the new KCNJ11 or ABCC8 mutations was detected in 100 chromosomes from normal controls.
Fig. 1.
Pedigrees of four families with mutations in ABCC8; from top to bottom: mutation, phenotype, age at presentation/diagnosis of diabetes, current age and initial therapy for diabetes. For the two grandparents of proband nd-FI/1, OGTT tests were performed at 70 (W688R) and 74 (V324M) years of age. GD, gestational diabetes; IGT, impaired glucose tolerance; INS, insulin
Polymorphisms—KATP genes
In the patient of Masai descent (nd-RM/6) we detected the variant KCNJ11/R195H (c.584G>A; single nucleotide polymorphism: rs5217; Table 1), while in the proband nd-BG/1 we found the variant ABCC8/S1054>N (c.3161G>A, numbered according to [36]; Table 1), inherited from the patient’s father, who had normal glucose tolerance. MLPA of the ABCC8 gene did not disclose any deletion in this patient. Thus, we currently classify ABCC8/S1054N as a rare, benign variant, taking into account the fact that residue S1054 is conserved from dog through to mouse, but not in chicken and zebra fish, where is substituted by cysteine and valine, respectively.
No additional mutations were found in INS (6), GCK (19) or any of the other genes (FOXP3, NEUROG3, ERN1, CHGA, CHGB, NKX6-1, CACNA1C and CDK4) screened at this time.
Clinical features in patients with diabetes and negative for KCNJ11, INS and ABCC8
Patients from group 1, negative in the genetic screening for genes commonly causing PNDM/MDI (hereafter called PNDM/MDI ‘X’) had a higher birthweight (median 3,164 g) than patients with KCNJ11 mutations (median 2,460 g; p < 0.0003), and similar to individuals carrying INS mutations (Table 2). Moreover, in patients with PNDM/MDI ‘X’, diabetes was diagnosed 1 month later (median) than carriers of mutations of KATP genes.
Table 2.
Birthweight and age at diabetes onset of probands with KCNJ11, INS and ABCC8 mutations and of unknown genetic origin
| Gene | KCNJ11 | INS | ABCC8 | Unknown |
|---|---|---|---|---|
| n | 22 | 9 | 4 | 8 |
| Birthweight (g) | 2,460 ± 394.34 | 3,050 ± 203.88† | 2,825 ± 663.79 | 3,150 ± 204.57‡ |
| Age at diagnosis (days) | 53 ± 59.7 | 86 ± 45.6 | 275 ± 30.0 | 82 ± 68.9 |
Birthweight and age at diabetes onset are median values±SD
Birthweight was not available for one patient carrying a KCNJ11 mutation, and for a second patient born in Kenya (nd-RM/6). The patient with IPEX-like features (birthweight 990 g, born at 22 weeks of gestation) was not included in the group of unknown genetic aetiology (PNDM/MDI ‘X’)
p value vs KCNJ11: †0.009, ‡0.003
Incidence of PNDM/MDI in Italy
The incidence of PNDM/MDI was calculated at 1:210,287 (95% CI 1:300,300–1:151,976) live births for years 1995–2009. Of note, 22 out of 23 patients (95%) included in the database for years 2000–2009 (incidence rate: 1:213,198, 95% CI 1:336,700–1:142,045) have been subjected to genetic screening for KCNJ11, INS and ABCC8 and 15 (68%) carried a causative mutation in either KCNJ11 or INS. The incidence rate (years 1995–2009) was found to be 1:473,146 when calculated using the original definition of neonatal diabetes as insulin-requiring hyperglycaemia with onset within the first 6 weeks of life [29].
Discussion
In 2002 we suggested that the cause of diabetes in patients with disease onset within 6 months of life might be genetic [20]. At that time only one gene, GCK [19], was known to be linked with rare cases of insulin-deficient diabetes, while IPF1 (also known as PDX1), EIF2AK3 and FOXP3 had been associated with early onset, syndromic diabetes [12, 13, 18]. More recently, mutations in the KCNJ11, ABCC8 and INS genes have been identified, which cause diabetes with onset in the first 6 months of life [3–6] and beyond [7–9, 37]. In the present study, we report the identification, by sequential sequencing of the three latter genes, of a disease-causing mutation in 36 probands with diabetes diagnosed before 6 months of age and in a single patient with diabetes onset after 6 months but before 1 year of age. In Group 1, novel mutations in KCNJ11/R201S, ABCC8/L213P and ABCC8/W688R were identified. The latter was found in a patient who also carried the TNDM-causing ABCC8/V324M mutation [36]. PNDM/MDI can result from recessive ABCC8 mutations, usually one with a mild activating effect and the other with loss-of-function, which are observed in recessive hyperinsulinism [10]. In our case, however, we favour the hypothesis that both ABCC8/V324M and ABCC8/W688R are mildly activating, based on the fact that W688R is associated with impaired glucose tolerance in the paternal grandmother. In contrast, we cannot explain at this time the extremely different phenotypes we observed in the carriers of the mutation ABCC8/A355T, who show either PNDM/MDI or gestational diabetes. Previously, phenotypic variability in ABCC8 mutation carriers has been described in families in which the proband presents with TNDM and first-degree relatives carry the same mutation showing early-onset type 2 diabetes or even normal glucose tolerance [7, 38]. Because the patient with ABCC8/A355T also has a liver-related and haematologic phenotype, it is conceivable that he may carry a mutation in another locus that impacts on glucose metabolism. Functional studies are definitely needed to firmly establish the impact of ABCC8/A355T on insulin secretion.
In the present work, the mutation detection rate of KCNJ11, INS and ABCC8 genes was 78.2% (36/46) for patients in group 1 and 12.5% (1/8) for patients in group 2. After sequencing the same three genes, Støy et al. [39] obtained comparable results, with a 63% and 6.6% detection rate in patients for permanent diabetes before or after 6 months of life, respectively. Our results from group 1 were not influenced by type 1 diabetes autoantibody status, which was unknown in 15 (40%) patients who harboured a disease-causing mutation [6, 21, 30–32] (Table 1).
Sequencing of KCNJ11, INS and ABCC8 in patients from group 2 was justified by the fact that mutations in these genes have previously been detected in patients with diabetes onset in infancy, childhood and even adulthood [7–9, 37, 38 and F. Barbetti, unpublished observations], eliciting a negative result in >85% of cases examined. Screening of candidate genes in these and other patients was also negative [present study; 40]. Consequently, the autoimmune or genetic aetiology for seven of the remaining patients in group 2 remains open. Four patients tested negative for IAA, which are found at an increased rate in individuals with diabetes onset before 5 years of age, and one of these four was negative for all autoantibodies commonly used as tools for the diagnosis of type 1 diabetes. However, the latter patient may still harbour autoantibodies against ZnT8A, which can be detected as a single autoantibody in patients with type 1 diabetes previously classified as autoantibody-negative on the basis of existing markers (i.e. IAA, GADA, IA-2A and ICA) [9, 41]. Of note, one patient who was negative to GADA at diagnosis (and carrying HLA-DR3/DR4) and was initially included in group 2 became weakly GADA positive during the study; this individual was therefore re-classified as having type 1 diabetes and was not investigated further. A limitation/weakness of our study is that the HLA status of most patients in group 2 was unknown, and type 1 diabetes autoantibodies were not thoroughly tested.
Conversely, we think that the nine patients in group 1 who had no disease-causing mutation identified, and possibly the patient carrying the ABCC8/A355T mutation, who showed clinical features not associated with mutations to KATP channel genes, are likely to carry a mutation in a locus that has not, as yet, been found. Seven of these patients presented with non-syndromic diabetes and birthweight close to normal (Table 2) and were not good candidates for screening of any of the known genes giving rise to syndromic diabetes [12–17] or hyperglycaemia in the first week of life [42, 43]. We therefore sequenced genes that we considered to be candidates, based on the phenotypic consequences of their manipulation/ablation in mice [22–25], with no success. Because hundreds of genes can have an impact on pancreatic beta cell function [44] we believe that new approaches, such as whole exome/genome sequencing, should be used in the future for PNDM/MDI gene discovery [45, 46].
The incidence rate of PNDM/MDI in Italy, utilising the 6 month limit as a cut-off for diagnosis of neonatal/infancy-onset diabetes, is about 1:210,000 live births. This result is comparable to recent reports from Slovakia, the UK, the Netherlands and Poland [47, 48], showing an incidence of PNDM/MDI of 1:215,417 [47] and 1:260,000 [48], respectively. In summary, a molecular genetic diagnosis can be reached in ~75% of patients with diabetes onset ≤6 months of age when KCNJ11, INS and ABCC8 genes are sequenced in populations with a low consanguinity rate. Moreover, because the status of type 1 diabetes autoimmunity was unknown in about 40% of patients in this group, we also conclude that knowledge of type 1 diabetes autoantibodies is not a prerequisite to proceeding to genetic screening, which we recommend for all patients with diabetes onset before 6 months of age. The incidence of PNDM/MDI varies, as expected, if different age cut-offs for disease onset are used as criteria for aetiological (genetic vs autoimmune) diagnosis.
Electronic supplementary material
Below is the link to the electronic supplementary material.
PCR and sequencing conditions for candidate gene screening (PDF 320 kb)
Acknowledgements
Part of this work has been supported by the Fondazione Roma grant ‘Molecular mechanisms in the pathogenesis of type 2 diabetes mellitus and its cardiovascular complications’ to F. Barbetti and by the Transnational European research grant on Rare Diseases (ERANET 09 RARE 005). Funding for L. Aguilar-Bryan is from the Thrasher Research Fund.
Duality of interest
The authors declare that there is no duality of interest associated with this manuscript.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Abbreviations
- CNV
Copy number variation
- DEND
Developmental delay, epilepsy, neonatal diabetes
- GADA
Glutamic acid decarboxylase autoantibodies
- IA-2A
Tyrosine phosphatase-related proteins islet antigen 2 autoantibodies
- IAA
Insulin autoantibodies
- ICA
Islet-cell antibodies
- iDEND
Intermediate DEND
- IPEX
Immune dysregulation, polyendocrinopathy, enteropathy, X-linked
- ISPED
Italian Society of Pediatric Endocrinology and Diabetology
- KATP
ATP-sensitive potassium channel
- MDI
Monogenic diabetes of infancy
- MLPA
Multiplex ligation-dependent probe amplification
- PNDM
Permanent neonatal diabetes mellitus
- TNDM
Transient neonatal diabetes mellitus
- ZnT8A
Zinc transporter 8 autoantibodies
Appendix
Members of the ISPED Diabetes Study Group are:
Alessandria: R. Lera; Ancona: V. Cherubini; Arezzo: A. R. Fifi; Bari, Giovanni XXIII Hospital: C. Torelli, E. Frezza; Bari, University: L. Cavallo; Bologna: S. Zucchini; Bolzano: B. Pasquino, P. Kaufmann; Cagliari, Brotzu Hospital: P. Frongia; Cagliari, University: N. Zedda, C. Ripoli; Caltanissetta: A. La Loggia; Campobasso: M. Cicchetti; Catania: D. Lo Presti; Catanzaro: F. Citriniti; Cesena: M. Pocecco; Chieti: F Chiarelli, S. Tumini; Como: M. Del Donno; Cosenza: M. R. De Marco; Crotone: N. Lazzaro; Feltre (Belluno): A.Vergerio; Ferrara: P. Banin; Firenze:S. Toni, L. Lenzi; Genova: R. Lorini, G. D’Annunzio; Livorno: S. Lucchesi; Locri: F. Mammì; Lucca: C. Meossi; Mantova: F. Bontempi; Massa Carrara: D. Pardi; Matera: R. Taccardi; Messina: F. Lombardo; Milano: F. Meschi, R. Bonfanti; Modena: L. Iughetti; Napoli, Federico II: A. Franzese, P. Buono; Napoli, SUN: F. Prisco, D. Iafusco; Novara: F. Cadario; Nuoro: A. F. Milia; Olbia: L. Bellu, G. Piredda; Padova: C. M. Monciotti; Palermo: F. Cardella; Parma: M. Vanelli, G. Chiari; Pavia: V. Calcaterra; Perugia: G. De Giorgi; Pisa: G. Federico; Potenza: F. Citro; Rimini: A. Marsciani; Roma, Bambino Gesù Hospital: M. Cappa, C. Bizzarri; Roma, Sapienza University: N. Sulli; Roma, Tor Vergata University: F. Barbetti, M.L. Manca Bitti; San Giovanni Rotondo (Foggia): M. Delvecchio; Sanremo-Imperia: M. Cotellessa; Sassari: A. M. Marinaro; Thiene (Vicenza): R. Pesavento; Torino: F. Cerutti, I. Rabbone; Tortona (Alessandria): F. Fontana; Trento: V. Cauvin, M. Bellizzi; Treviglio (Bergamo): L. Gargantini; Trieste: G. Tonini, E. Faleschini; Varese: A. Salvatoni; Vercelli: M. Trada; Verona: L. Pinelli; Viterbo: C. Araldi.
References
- 1.Aguilar-Bryan L, Bryan J. Neonatal diabetes mellitus. Endocr Rev. 2008;29:265–291. doi: 10.1210/er.2007-0029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Murphy R, Ellard S, Hattersley AT. Clinical implications of a molecular genetic classification of monogenic β-cell diabetes. Nat Clin Pract Endocrinol Metab. 2008;4:200–213. doi: 10.1038/ncpendmet0778. [DOI] [PubMed] [Google Scholar]
- 3.Gloyn AL, Pearson ER, Antcliff JF, et al. Activating mutations in the gene encoding the ATP-sensitive potassium channel subunit Kir6.2 gene and permanent neonatal diabetes. N Engl J Med. 2004;350:1838–1849. doi: 10.1056/NEJMoa032922. [DOI] [PubMed] [Google Scholar]
- 4.Babenko AP, Polak M, Cave H, et al. Activating mutations in the ABCC8 gene in neonatal diabetes mellitus. N Engl J Med. 2006;355:456–466. doi: 10.1056/NEJMoa055068. [DOI] [PubMed] [Google Scholar]
- 5.Støy J, Edghill EL, Flanagan SE, et al. Neonatal Diabetes International Collaborative Group. Insulin gene mutations as a cause of permanent neonatal diabetes. Proc Natl Acad Sci USA. 2007;104:15040–15044. doi: 10.1073/pnas.0707291104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Colombo C, Porzio O, Liu M, et al. Seven mutations in the human insulin gene linked to permanent neonatal/infancy-onset diabetes mellitus. J Clin Invest. 2008;118:2148–2156. doi: 10.1172/JCI33777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yorifuji T, Nagashima K, Kurokawa K, et al. The C42R mutation in the Kir6.2 (KCNJ11) gene as a cause of transient neonatal diabetes, childhood diabetes, or later-onset, apparently type 2 diabetes mellitus. J Clin Endocrinol Metab. 2005;90:3174–3178. doi: 10.1210/jc.2005-0096. [DOI] [PubMed] [Google Scholar]
- 8.Molven A, Ringdal M, Nordbø AM, et al. Mutations in the insulin gene can cause MODY and autoantibody-negative type 1 diabetes. Diabetes. 2008;57:1131–1135. doi: 10.2337/db07-1467. [DOI] [PubMed] [Google Scholar]
- 9.Bonfanti R, Colombo C, Nocerino V, et al. Insulin gene mutations as cause of diabetes in children negative for five type 1 diabetes autoantibodies. Diab Care. 2009;32:123–125. doi: 10.2337/dc08-0783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ellard S, Flanagan SE, Girard CA, et al. Permanent neonatal diabetes caused by dominant, recessive, or compound heterozygous SUR1 mutations with opposite functional effects. Am J Hum Genet. 2007;81:375–382. doi: 10.1086/519174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Garin I, Edghill EL, Akerman I, et al. Recessive mutations in the INS gene result in neonatal diabetes through reduced insulin biosynthesis. Proc Natl Acad Sci USA. 2010;107:3105–3110. doi: 10.1073/pnas.0910533107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Stoffers DA, Zinkin NT, Stanojevic V, Clarke WL, Habener JF. Pancreatic agenesis attributable to a single nucleotide deletion in the human IPF1 gene coding sequence. Nat Genet. 1997;15:106–110. doi: 10.1038/ng0197-106. [DOI] [PubMed] [Google Scholar]
- 13.Delepine M, Nicolino M, Barrett T, Golamaully M, Lathrop GM, Julier C. EIF2AK3, encoding translation initiation factor 2-alpha kinase 3, is mutated in patients with Wolcott–Rallison syndrome. Nat Genet. 2000;25:406–409. doi: 10.1038/78085. [DOI] [PubMed] [Google Scholar]
- 14.Sellick GS, Barker KT, Stolte-Dijkstra I, et al. Mutations in PTF1A cause pancreatic and cerebellar agenesis. Nat Genet. 2004;36:1301–1305. doi: 10.1038/ng1475. [DOI] [PubMed] [Google Scholar]
- 15.Senee V, Chelala C, Duchatelet S, et al. Mutations in GLIS3 are responsible for a rare syndrome with neonatal diabetes mellitus and congenital hypothyroidism. Nat Genet. 2006;38:682–687. doi: 10.1038/ng1802. [DOI] [PubMed] [Google Scholar]
- 16.Smith SB, Qu HQ, Taleb N, et al. Rfx6 directs islet formation and insulin production in mice and humans. Nature. 2010;463:775–780. doi: 10.1038/nature08748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Senee V, Vattem KM, Delepine M, et al. Wolcott–Rallison syndrome: clinical, genetic, and functional study of EIF2AK3 mutations and suggestion of genetic heterogeneity. Diabetes. 2004;53:1876–1883. doi: 10.2337/diabetes.53.7.1876. [DOI] [PubMed] [Google Scholar]
- 18.Bennett CL, Christie J, Ramsdell F, et al. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat Genet. 2001;27:20–21. doi: 10.1038/83713. [DOI] [PubMed] [Google Scholar]
- 19.Njolstad PR, Sovik O, Cuesta-Munoz A, et al. Neonatal diabetes mellitus due to complete glucokinase deficiency. N Engl J Med. 2001;344:1588–1592. doi: 10.1056/NEJM200105243442104. [DOI] [PubMed] [Google Scholar]
- 20.Iafusco D, Stazi MA, Cotichini R, et al. Permanent diabetes mellitus in the first year of life. Diabetologia. 2002;45:798–804. doi: 10.1007/s00125-002-0837-2. [DOI] [PubMed] [Google Scholar]
- 21.Massa O, Iafusco D, D’Amato E. KCNJ11 activating mutations in Italian patients with permanent neonatal diabetes. Hum Mutat. 2005;25:22–27. doi: 10.1002/humu.20124. [DOI] [PubMed] [Google Scholar]
- 22.Sinnegger-Brauns MJ, Hetzenauer A, Huber IG, et al. Isoform-specific regulation of mood behavior and pancreatic beta cell and cardiovascular function by L-type Ca2+ channels. J Clin Invest. 2004;113:1430–1439. doi: 10.1172/JCI20208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Obermüller S, Calegari F, King A, et al. Defective secretion of islet hormones in chromogranin-B deficient mice. PLoS ONE. 2010;5:e8936. doi: 10.1371/journal.pone.0008936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lipson KL, Fonseca SG, Ishigaki S, et al. Regulation of insulin biosynthesis in pancreatic beta cells by an endoplasmic reticulum-resident protein kinase IRE1. Cell Metab. 2006;4:245–254. doi: 10.1016/j.cmet.2006.07.007. [DOI] [PubMed] [Google Scholar]
- 25.Sander M, Sussel L, Conners J, et al. Homeobox gene Nkx6.1 lies downstream of Nkx2.2 in the major pathway of beta-cell formation in the pancreas. Development. 2000;127:5533–5540. doi: 10.1242/dev.127.24.5533. [DOI] [PubMed] [Google Scholar]
- 26.Wang J, Cortina G, Wu SV, et al. Mutant neurogenin-3 in congenital malabsorptive diarrhea. N Engl J Med. 2006;335:270–280. doi: 10.1056/NEJMoa054288. [DOI] [PubMed] [Google Scholar]
- 27.Mettus RV, Rane SG. Characterization of the abnormal pancreatic development, reduced growth and infertility in CDK4 mutant mice. Oncogene. 2003;22:8413–8421. doi: 10.1038/sj.onc.1206888. [DOI] [PubMed] [Google Scholar]
- 28.Bernardini L, Alesi V, Loddo S, et al. High-resolution SNP arrays in mental retardation diagnostics: how much do we gain? Eur J Hum Genet. 2010;18:178–185. doi: 10.1038/ejhg.2009.154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.von Mühlendahl KE, Herkenhoff H. Long-term course of neonatal diabetes. N Engl J Med. 1995;333:704–708. doi: 10.1056/NEJM199509143331105. [DOI] [PubMed] [Google Scholar]
- 30.Tonini G, Bizzarri C, Bonfanti R, et al. Sulphonylurea treatment outweighs insulin therapy in short-term metabolic control of patients with permanent neonatal diabetes mellitus due to activating mutations of the KCNJ11 gene. Diabetologia. 2006;49:2210–2213. doi: 10.1007/s00125-006-0329-x. [DOI] [PubMed] [Google Scholar]
- 31.Masia R, Koster JC, Tumini S, et al. An ATP-binding mutation (G334D) in KCNJ11 is associated with a sulfonylurea-insensitive form of DEND (Developmental Delay, Epilepsy, and Neonatal Diabetes) Diabetes. 2007;56:328–336. doi: 10.2337/db06-1275. [DOI] [PubMed] [Google Scholar]
- 32.Koster JC, Cadario F, Kurata HT, et al. The G53D mutation in Kir6.2 (KCNJ11) is associated with neonatal diabetes and motor dysfunction in adulthood that is improved with sulfonylurea therapy. J Clin Endocrinol Metab. 2008;93:1054–1061. doi: 10.1210/jc.2007-1826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Flanagan SE, Edghill EL, Gloyn AL, Ellard S, Hattersley AT. Mutations in KCNJ11, which encodes Kir6.2, are a common cause of diabetes diagnosed in the first months of life, with the phenotype determined by genotype. Diabetologia. 2006;49:1190–1197. doi: 10.1007/s00125-006-0246-z. [DOI] [PubMed] [Google Scholar]
- 34.Winkler M, Lutz R, Russ U, Quast U, Bryan J. Analysis of two KCNJ11 neonatal diabetes mutations, V59G and V59A, and the analogous KCNJ8 I60G substitution: differences between the channel subtypes formed with SUR1. J Biol Chem. 2009;284:6752–6762. doi: 10.1074/jbc.M805435200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Vaxillaire M, Populaire C, Busiah K, et al. Kir6.2 mutations are a common cause of permanent neonatal diabetes in a large cohort of French patients. Diabetes. 2004;53:2719–2722. doi: 10.2337/diabetes.53.10.2719. [DOI] [PubMed] [Google Scholar]
- 36.Flanagan SE, Clauin S, Bellané-Chantelot C, et al. Update of mutations in the genes encoding the pancreatic beta-cell KATP channel subunits Kir6.2 (KCNJ11) and sulfonylurea receptor 1 (ABCC8) in diabetes mellitus and hyperinsulinism. Hum Mutat. 2009;30:170–180. doi: 10.1002/humu.20838. [DOI] [PubMed] [Google Scholar]
- 37.Boesgaard TW, Pruhova S, Andersson EA, et al. Further evidence that mutations in INS can be a rare cause of Maturity Onset Diabetes of the Young (MODY) BMC Med Genet. 2010;11:42. doi: 10.1186/1471-2350-11-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Flanagan SE, Patch A-M, Mackay DJG, et al. Mutations in ATP-sensitive K+ channel genes cause transient neonatal diabetes and permanent diabetes in childhood or adulthood. Diabetes. 2007;56:1930–1937. doi: 10.2337/db07-0043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Støy J, Greeley SA, Paz VP, et al. Diagnosis and treatment of neonatal diabetes: a United States experience. Pediatr Diabetes. 2008;9:450–459. doi: 10.1111/j.1399-5448.2008.00433.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nocerino V, Colombo C, Bonfanti R, Iafusco D, Barbetti F. Mutations in hIAPP and NEUROG3 genes are not a common cause of permanent neonatal/infancy/childhood onset diabetes. Diabet Med. 2009;26:660–661. doi: 10.1111/j.1464-5491.2009.02726.x. [DOI] [PubMed] [Google Scholar]
- 41.Wenzlau JM, Moua O, Sarkar SA, et al. SlC30A8 is a major target of humoral autoimmunity in type 1 diabetes and a predictive marker in prediabetes. Ann NY Acad Sci. 2008;1150:256–259. doi: 10.1196/annals.1447.029. [DOI] [PubMed] [Google Scholar]
- 42.Njolstad PR, Sagen JV, Bjorkhaug L, et al. Permanent neonatal diabetes caused by glucokinase deficiency: inborn error of the glucose–insulin signaling pathway. Diabetes. 2003;52:2854–2860. doi: 10.2337/diabetes.52.11.2854. [DOI] [PubMed] [Google Scholar]
- 43.Nicolino M, Claiborn KC, Senée V, et al. A novel hypomorphc PDX1 mutation responsible for permanent neonatal diabetes with subclinical exocrine deficiency. Diabetes. 2010;59:733–740. doi: 10.2337/db09-1284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kutlu B, Burdick D, Baxter D, et al. Detailed transcriptome atlas of the pancreatic beta cell. BMC Med Genomics. 2009;2:3. doi: 10.1186/1755-8794-2-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Choi M, Scholl UI, Ji W, et al. Genetic diagnosis by whole exome capture and massively parallel DNA sequencing. Proc Nat Acad Sci USA. 2009;106:19096–19101. doi: 10.1073/pnas.0910672106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bonnefond A, Durand E, Sand O, et al. Molecular diagnosis of neonatal diabetes mellitus using next-generation sequencing of whole exome. PLoS ONE. 2010;5:e13630. doi: 10.1371/journal.pone.0013630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Stanik J, Gasperikova D, Paskova M, et al. Prevalence of permanent neonatal diabetes in Slovakia and successful replacement of insulin with sulfonylurea therapy in KCNJ11 and ABCC8 mutation carriers. J Clin Endocrinol Metab. 2007;92:1276–1282. doi: 10.1210/jc.2006-2490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Slingerland AS, Shields BM, Flanagan SE, et al. Referral rates for diagnostic testing support an incidence of permanent neonatal diabetes in three European countries of at least 1 in 260,000 live births. Diabetologia. 2009;52:1683–1685. doi: 10.1007/s00125-009-1416-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
PCR and sequencing conditions for candidate gene screening (PDF 320 kb)