ABSTRACT
Cryptococcus neoformans is an important fungal pathogen of immunocompromised individuals, with a close relative, Cryptococcus gattii, emerging as a serious threat for the immunocompetent. During initial infection, C. neoformans colonizes the airspaces of the lungs, resulting in pneumonia, and subsequently migrates to the central nervous system (CNS). We sought to understand fungal carbon utilization during colonization of these fundamentally different niches within the host, in particular the roles of gluconeogenesis and glycolysis. We created mutants at key points in the gluconeogenesis/glycolysis metabolic pathways that are restricted for growth on lactate and glucose, respectively. A phosphoenolpyruvate carboxykinase mutant (the pck1∆ mutant), blocked for entry of 2- and 3-carbon substrates into gluconeogenesis and attenuated for virulence in a murine inhalation model, showed wild-type (WT) persistence in a rabbit cerebrospinal fluid (CSF) model of cryptococcosis. Conversely, both the pyruvate kinase (pyk1∆) and the hexose kinase I and II (hxk1∆/hxk2∆) mutants, which show impaired glucose utilization, exhibited severely attenuated virulence in the murine inhalation model of cryptococcosis and decreased persistence in the CNS in both the rabbit CSF and the murine inhalation models while displaying adequate persistence in the lungs of mice. These data suggest that glucose utilization is critical for virulence of C. neoformans and persistence of the yeast in the CNS.
IMPORTANCE
Cryptococcus neoformans is an emerging fungal pathogen of humans and is responsible for approximately 625,000 deaths annually among those suffering from HIV infection/AIDS. The ability of this fungus to persist in the host, coupled with its propensity to colonize the CNS, makes the understanding of nutrient acquisition in the host a primary concern. In this study, we report a requirement of glucose utilization for virulence of C. neoformans that is separate from its role in ATP production in the pathogen. Furthermore, we show that inhibition of glycolysis is a viable antifungal drug target, and impaired ATP production via the PYK1 deletion may serve as a model for dormant/chronic fungal infection in the host. Taken together, these results demonstrate the critical importance of understanding basic metabolic processes of the fungus in the context of host-pathogen interactions.
Introduction
Cryptococcus neoformans is a major human and animal pathogen responsible for cryptococcal meningoencephalitis in immunocompromised individuals. Found worldwide, C. neoformans is commonly isolated from environmental samples (1). Cryptococcosis is currently the third highest cause of mortality due to infectious diseases in sub-Saharan Africa (2), and an ongoing outbreak of Cryptococcus gattii infections is causing measurable human and animal morbidity and mortality in the Pacific Northwest (3–5). Unlike many other opportunistic fungal pathogens (e.g., Aspergillus fumigatus), C. neoformans appears to be specially adapted for survival in the human host and possesses a number of pathogenicity factors involved in this interaction (6). However, this pathogen does not appear to be part of the normal human flora, despite its intimate association with monocytes and the central nervous system (CNS) during disease (7–12).
Several virulence-associated traits have clearly been identified as required for cryptococcal pathogenesis, including growth at 37°C, production of capsule, melanin biosynthesis, secretion of phospholipase, and production of mannitol (13, 14). The polysaccharide capsule has received much attention due to its immunomodulatory properties (12) and is mainly composed of GXM (α1,3-linked mannose subunits with various xylosyl and β-glucuronyl substitutions), which varies by serotype. Interestingly, elimination of individual sugar components (e.g., xylose) results in virulence defects (15). Therefore, understanding the process of carbon assimilation by C. neoformans and therefore subsequent capsule biosynthesis is crucial for a proper understanding of virulence.
Recently, Barelle and colleagues demonstrated that the ascomycete yeast pathogen Candida albicans alters its carbon assimilation from nonfermentative (i.e., gluconeogenesis and glyoxylate cycle) to fermentative (i.e., glycolysis) metabolism during the onset of systemic infection in mice (16). In their study, they built upon previous studies showing the seemingly paradoxical importance of both fermentative and nonfermentative carbon metabolism in C. albicans virulence (17–19). Using mutants blocked in various core carbon metabolic pathways, Barelle and colleagues showed that the glyoxylate cycle and gluconeogenesis pathways were essential for optimal survival of C. albicans during its initial interactions with immune cells (16). Later, as the yeast cells colonize the bloodstream and internal organs of the mammalian host, glycolysis emerges as the main carbon assimilation pathway. This study emphasized the changes in carbon assimilation a pathogen must make to persist in spite of changing host defenses.
Presumably, C. neoformans also encounters several fundamentally different microenvironments in the host during the course of disease progression. For instance, we recently demonstrated that persistence of C. neoformans in the cerebrospinal fluid (CSF) microenvironment was influenced by a number of different cellular pathways, including maintenance of ionic balance and inositol metabolism (20). Previous studies touching on carbon metabolism and C. neoformans pathogenesis show mixed results regarding the role of nonpreferred carbon sources in disease progression (21–24). However, nonpreferred carbon sources such as lactate and acetate are likely important early in establishment of a pulmonary infection. For example, the acetyl-coenzyme A (CoA) synthase gene ACS1 has been demonstrated as necessary for full virulence in C. neoformans (21). In this same study, Hu and colleagues showed that Snf1, shown to be a major regulator of nonpreferred carbon utilization via the phosphorylation of the global carbon catabolite repressor Mig1 in Saccharomyces cerevisiae, is also required for full virulence in a murine inhalation model of cryptococcosis. However, there are differences between C. neoformans and other pathogenic fungi in the relationship of carbon utilization and virulence. For example, the glyoxylate shunt is not required for virulence in this basidiomycete pathogen as it is for the full virulence of the ascomycete C. albicans (18, 22, 24).
The ability of a pathogen to cause disease is inextricably linked to its ability to procure nutrients from its host, and carbon in particular. Therefore, a solid understanding of carbon acquisition by a microbial invader or tumor cell is essential for identifying potential targets for the development of drug therapies. This line of reasoning is currently being employed with regard to cancer treatment. Cancer cells are known to exhibit differential carbon metabolism compared to normal host tissue (25). For instance, malignant cells switch on proliferative carbon metabolism dependent on glycolysis for the production of their biomass, making them vulnerable to inhibitors of glycolysis (26). The apparent similarities observed between malignancies and eukaryotic microorganisms regarding carbon metabolism during host invasion (25) warrants further investigation of carbon metabolism during infection for unique weaknesses that can be targeted in treatment strategies for pathogenic fungi such as C. neoformans.
Therefore, we sought to understand the relationship of glucose anabolic and catabolic processes to pathogenicity in C. neoformans. To investigate the roles of glycolysis and gluconeogenesis in host colonization by Cryptococcus, we procured carbon metabolism mutants previously described as well as constructed additional mutants to block key steps in glycolysis/gluconeogenesis and carbon catabolite repression (CCR) (see Table S1 and Fig. S1 in the supplemental material) (21, 22). By examining the effects of these mutations on virulence at several body sites using both the murine inhalation (pulmonary infection and dissemination) and the rabbit CSF (direct inoculation of the subarachnoid space) models of cryptococcosis, we demonstrate that gluconeogenic carbon assimilation is sufficient for the initial establishment of infection but that glycolysis is critical for persistence of C. neoformans in the CNS and virulence in the host.
RESULTS
Role of gluconeogenesis in C. neoformans persistence in the CNS.
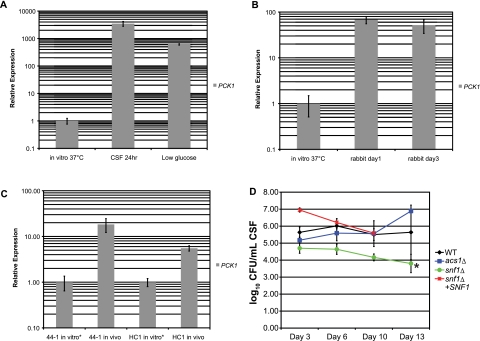
Since prior reports have demonstrated the importance of gluconeogenesis during lung infection by C. neoformans, we sought to establish a possible role for gluconeogenesis in survival of C. neoformans in the CNS. We used real-time PCR to measure expression of the phosphoenolpyruvate carboxykinase gene PCK1, which encodes the main gateway enzyme for entry of 2- and 3-carbon substrates into gluconeogenesis, under different growth conditions. We confirmed elevated PCK1 expression in low glucose concentrations, as expected from results obtained using S. cerevisiae (27), and also observed increased expression due to exposure to ex vivo pooled human CSF (Fig. 1A). This increased PCK1 expression in strain H99 was also observed in vivo in the rabbit CSF model of cryptococcosis and in cryptococcal strains taken directly from human patient CSF samples compared to the expression of PCK1 in these samples grown in vitro at 37°C (Fig. 1B and C). However, despite this elevated expression of PCK1, independent pck1∆ deletion mutants showed no defects in CNS persistence or viability in the rabbit CSF model (data not shown).
FIG 1 .
PCK1 expression does not correlate with CNS persistence of C. neoformans. (A) PCK1 expression was measured by real-time PCR following exposure to either CSF or YNB-0.05% glucose (low glucose) for 24 h at 37°C. PCK1 expression following growth in YPD at 37°C (in vitro 37°C) was used as a positive control. (B) PCK1 expression in C. neoformans was measured by real-time PCR during rabbit infection. Expression levels in vivo were compared to those from yeast grown in YPD at 37°C (in vitro 37°C). (C) PCK1 expression levels were measured by real-time PCR for C. neoformans isolated from human CSF samples. C. neoformans strains 44-1 and HC1 were obtained from the Duke University Infectious Disease Specimen Repository, which houses specimens isolated from deidentified patients with cryptococcal meningitis. PCK1 transcripts for C. neoformans cultured from the CSF samples and grown in YPD at 37°C (“in vitro” samples) were compared to transcripts isolated directly from the yeast in CSF (“in vivo” samples). (D) Levels of persistence of the WT, acs1∆, snf1∆, or snf1∆ SNF1 mutant strains of C. neoformans were compared by measuring the number of CFU of each strain isolated from the CSF of male NZW rabbits over time as described previously (24, 42). Rabbits were infected with 108 CFU of either the WT, the acs1∆, the snf1∆, or the snf1∆ SNF1 strain, and fungal burden was assessed over the course of 2 weeks as described in Materials and Methods. Missing data for the snf1∆ SNF1 strain on day 13 reflects the mortality of all these rabbits before day 13. Statistical differences were assessed by an ANOVA comparison of the graphed lines using the fit model process in JMP version 8 (SAS Institute, Inc., Cary, NC) (*, P < 0.0001).
We also tested other previously published mutants related to nonpreferred carbon assimilation in the rabbit CSF model. A snf1∆ deletion mutant, which is thought to represent a major regulator of nonpreferred carbon utilization based on findings in S. cerevisiae, demonstrated moderately reduced persistence in the rabbit CSF model (Fig. 1D). The snf1∆ mutant exhibits increased sensitivity to sodium nitrite, decreased melanin production, and growth on acetate as a sole carbon source at 37°C and is avirulent in the murine inhalation model (21). Furthermore, a recent study using a serotype D strain of C. neoformans has also shown that SNF1 deletion impairs growth at 39°C, which is the normal body temperature for rabbits (28). Therefore, we also tested a major pathway for 2-carbon utilization via the tricarboxylic acid (TCA) cycle. ACS1 encodes acetyl-CoA synthase, and the acs1∆ mutant fails to grow on 2-carbon substrates and exhibits modestly attenuated virulence in the murine-inhalational model of cryptococcosis (21). However, the acs1∆ mutant showed no defects in CNS persistence in the rabbit CSF model (Fig. 1D). Taken together, these data regarding PCK1, SNF1, and ACS1 support the hypothesis that carbon assimilation by C. neoformans for disease production and yeast persistence in the CNS is likely via glycolysis.
Pyruvate kinase is required for C. neoformans pathogenesis but not persistence in the lung.
Since blocked entry of 2- and 3-carbon compounds into gluconeogenesis by deletion of ACS1 or PCK1 showed no adverse effect on persistence of C. neoformans in the CNS, we sought to define the specific role of glycolysis in persistence of the fungus in the CNS. Therefore, we blocked the exit of carbon from glycolysis via the deletion of the pyruvate kinase homolog PYK1. As expected, the pyk1∆ mutant strain was unable to grow on glucose, fructose, ribose, or glycerol but was viable on either acetate or lactate as a sole carbon source (see Fig. S2 in the supplemental material). Growth rate on lactate as measured by culture absorbance was comparable to that of the wild type (WT). The pyk1∆ mutant showed no defects in capsule or melanin production under inducing conditions and normal growth at 37°C, and urease activity was similar to that of the WT (data not shown). Thus, the deletion of PYK1 had no apparent effects on the classical virulence phenotypes.
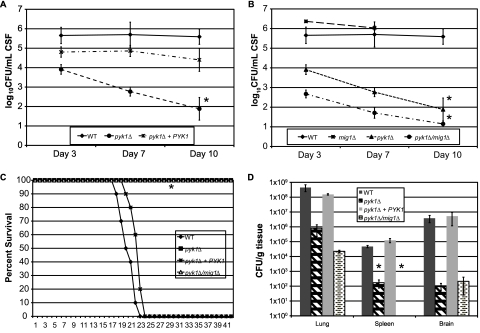
The pyk1∆ mutant did exhibit severely decreased persistence in the rabbit CSF model (Fig. 2A). Its inability to survive in this host microenvironment was profound. Since the pyk1∆ mutation restricts carbon utilization to nonpreferred carbon sources like lactate and/or acetate, and since utilization of nonpreferred carbon sources is restricted in the presence of glucose by CCR, we sought to alleviate the requirement of using lactate as a sole carbon substrate while allowing lactate use for energy production in the TCA cycle by impairing CCR. In fungi, two models exist for global regulation of carbon utilization. For molds such as Aspergillus nidulans, the Mig1p transcription factor homolog CreA is responsible for repressing the utilization of nonpreferred carbon sources in the presence of glucose (29). This is different from what was observed for S. cerevisiae, where CCR is regulated by three discrete mechanisms active at different concentrations of glucose (30). Since CreA is the main repressor in molds and Mig1p-mediated repression is active at glucose levels near those seen in human CSF (30, 31), we deleted a putative MIG1-related carbon catabolite repressor homolog in both WT and pyk1∆ backgrounds. The identity of the MIG1 gene was verified by real-time PCR (see Materials and Methods) because we observed repression of the key gluconeogenesis genes FBP1 and PCK1 in C. neoformans (3- and 169-fold repressed in the WT, respectively); these genes are known to be repressed by Mig1p in S. cerevisiae. The resulting pyk1∆/mig1∆ mutant strain should potentially utilize lactate for energy in the presence of glucose and allow utilization of numerous glycolytic metabolites as building blocks for DNA, proteins, etc.
FIG 2 .
Uncoupling of glycolysis from oxidative phosphorylation results in complete attenuation of virulence, but not persistence, of C. neoformans. NZW rabbits were infected with either the WT, pyk1∆, or pyk1∆ PYK1 strain (A) or the WT, mig1∆, pyk1∆, or pyk1∆/mig1∆ strain (B) of C. neoformans as described in Materials and Methods. Differences between strains were assessed by an ANOVA using the fit model process in JMP version 8 (SAS Institute, Cary, NC) (*, P < 0.0001). Missing data for mig1∆ day 10 reflects the mortality of all these rabbits before day 10. (C) A/Jcr inbred mice were infected per nasally with the WT, pyk1∆, pyk1∆ PYK1, or pyk1∆/mig1∆ strain of C. neoformans. The mice were observed over the course of the experiment for clinical signs correlating with eventual mortality (*, P < 0.0001; log rank test). (D) Fungal burden was assessed for three mice per group from the virulence study described for panel C. Organs were removed at time of death (~21 days postinfection for the WT and pyk1∆ PYK1 strains; 42 days postinfection for the pyk1∆ and pyk1∆/mig1∆ strains) and processed for fungal burden as described elsewhere (70). Differences in numbers of CFU between each mutant strain and the WT for each organ were assessed using Student’s t test (*, P = 0.01). Missing data for pyk1∆ mig1∆ spleen counts reflect an absence of CFU detected in this sample.
Interestingly, the pyk1∆/mig1∆ mutant showed further decreased persistence in the rabbit CSF model compared to the pyk1∆ mutant strain (Fig. 2B), and this was not a CNS-site-specific finding, since both mutants exhibited severely attenuated virulence in the murine inhalation model (Fig. 2C). To further define the mechanism of the attenuated virulence in the murine inhalation model, fungal viability as measured by tissue colony counts was obtained from lungs, spleen, and brain of mice at time of death (or the end of the experiment for the mutant-infected mice). As shown in Fig. 2D, despite no apparent evidence of host disease, substantial numbers of viable cells of both the pyk1∆ and pyk1∆/mig1∆ mutants were recovered from mouse organs after 42 days postinfection. Additionally, colony counts were lower for the pyk1∆/mig1∆ mutant strain than for the pyk1∆ strain. Clearly, these mutants can survive for long periods of time in the murine host without producing disease yet are unable to do so in the rabbit CSF model where they do not produce disease and are eliminated from the site of infection.
Hexose kinase activity is important for C. neoformans pathogenesis.
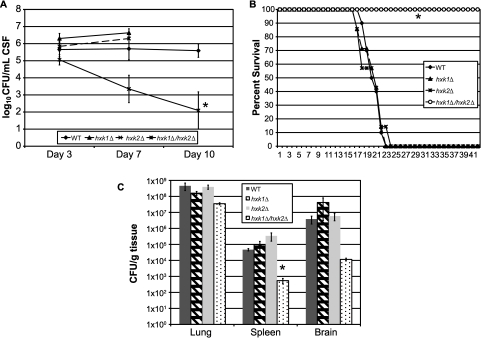
To more directly assess the mechanistic effect of glucose utilization on virulence of C. neoformans in the host, we blocked glycolysis at the entry point of glucose into the glycolysis pathway via the concurrent deletion of the putative hexose kinase homologs HXK1 and HXK2. Unlike the model yeast S. cerevisiae, which has three hexose kinase genes, C. neoformans possesses two putative hexose kinase genes, as observed in A. nidulans (22, 32). Whereas the single hexose kinase mutants and the WT strain were fully virulent, concurrent deletion of both putative hexose kinase genes by the creation of a double knockout mutation in C. neoformans significantly decreased fungal persistence in the rabbit CSF model (Fig. 3A). The hxk1∆/hxk2∆ mutant also showed profoundly attenuated virulence in the murine inhalation model of cryptococcosis, compared to the single mutants and the WT strain (Fig. 3B), without apparent defects in capsule production (data not shown). Surprisingly, subsequent analysis of the fungal census from various mouse organs obtained from the murine inhalation study revealed that both the single and the double mutant strains were recovered from mice at the time of death in similar numbers, yet the speeds of disease progression were dramatically different (i.e., at the end of the experiment [21 days postinfection for the hxk1∆ and hxk2∆ mutants and 42 days postinfection for the hxk1∆/hxk2∆ mutant]) (Fig. 3C).
FIG 3 .
Glucose utilization via hexose kinase is required for virulence but not persistence of C. neoformans. (A) NZW rabbits were infected with either the WT, the hxk1∆, the hxk2∆, or the hxk1∆/hxk2∆ strain of C. neoformans as described in Materials and Methods. Differences between strains were assessed by an ANOVA using the fit model process in JMP version 8 (SAS Institute, Inc., Cary, NC) (*, P = 0.001). Missing data for the hxk1∆ and hxk2∆ strains for day 10 reflect the mortality of all these rabbits before day 10. (B) A/Jcr inbred mice were infected per nasally with the WT, hxk1∆, hxk2∆, or hxk1∆/hxk2∆ strain of C. neoformans. The mice were observed over the course of the experiment for clinical signs correlating with eventual mortality (*, P < 0.0002; log rank test). (C) Fungal burden was assessed for three mice per group from the virulence study described for panel B. Organs were removed at the time of death (~21 days postinfection for the WT, hxk1∆, and hxk2∆ strains; 42 days postinfection for the hxk1∆/hxk2∆ strain) and processed for fungal burden as described elsewhere (70). Differences in numbers of CFU between each mutant strain and the WT for each organ were assessed using Student’s t test (*, P = 0.01).
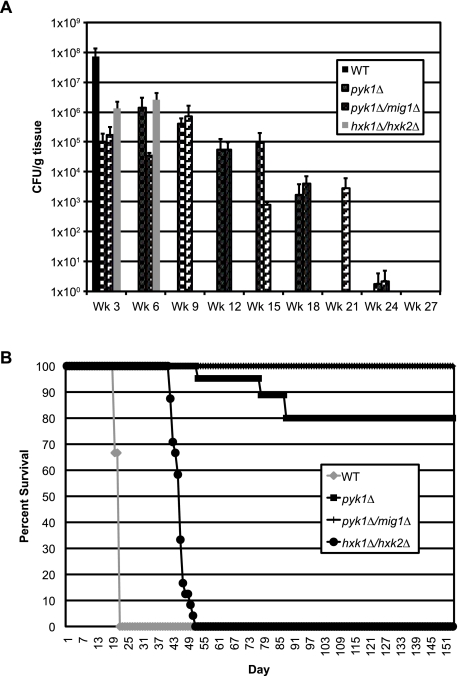
Given the results of the pyk1∆, pyk1∆/mig1∆, and hxk1∆/hxk2∆ mutants in both animal models, we elected to study the long-term effects of these mutations on yeast survival in the murine inhalation model. Mice were infected with either the WT, the pyk1∆, the pyk1∆/mig1∆, or the hxk1∆/hxk2∆ strain and evaluated every 3 weeks for fungal burden in both brain and lung tissues. These mutants did not successfully establish a persistent infection in the brains of these mice during the duration of the study (data not shown). However, all 3 mutants successfully colonized the lungs (Fig. 4A), and evaluation of these mice for signs of morbidity revealed differences in virulence in this model not previously observed (Fig. 4B). Surprisingly, the hxk1∆/hxk2∆ mutant was shown to be virulent and caused mortality in these mice after 6 weeks postinfection. The pyk1 mutant also displayed some virulence capacity, although by 21 weeks postinfection, this strain was cleared by the mice. Interestingly, impairment of CCR in the pyk1∆ background via the concurrent deletion of MIG1 completely abolished mortality compared to the level for the pyk1∆ single mutant and conferred prolonged persistence of the yeast in the lung (Fig. 4). Despite their common effects on glucose assimilation via glycolysis, either at the point of entry of glucose into glycolysis (hxk1∆/hxk2∆) or directly before entry of pyruvate into the TCA cycle (pyk1∆), virulence is severely affected, yet the final dispositions of these glycolysis mutants within the host are clearly different.
FIG 4 .
Pyruvate kinase mutants exhibit enhanced persistence in the lung without disease production. A/Jcr inbred mice were infected per nasally with the WT, pyk1∆, pyk1∆/mig1∆, or hxk1∆/hxk2∆ strain of C. neoformans in order to assess the persistence of these strains in vivo. (A) Fungal burden in lung tissues was assessed for three mice per strain every 3 weeks following infection as described elsewhere (70). Brain tissues from these mice showed no fungal burden over the course of the experiment. (B) The mice in the fungal burden assay were also observed over the course of the experiment for clinical signs correlating with eventual mortality (≥15% loss of body weight, lack of grooming, etc.). The survival of mice not sampled at the predetermined endpoints was assessed for similarity to the WT level. All mutant strains showed statistically significant differences from the WT (P < 0.0001; log rank test).
C. neoformans glycolysis mutants exhibit different host stress susceptibilities during infection.
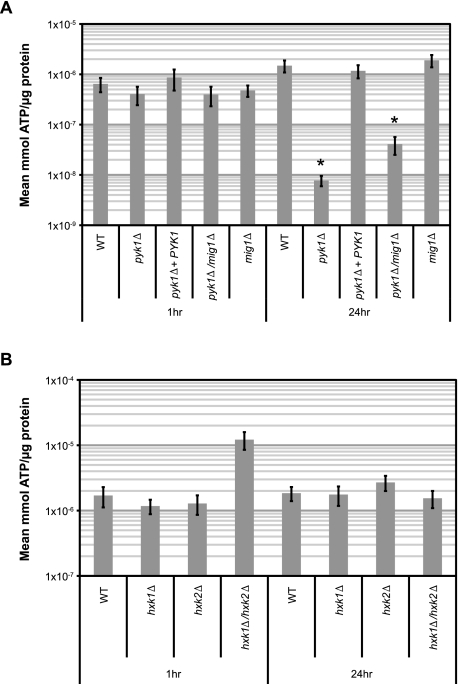
To understand the mechanism(s) surrounding the impact of glycolytic interruptions on host outcomes, and since glycolysis provides metabolites for energy production via oxidative phosphorylation, we quantified ATP production in the pyk1∆ mutants following exposure to human CSF in vitro to determine a possible link between energy production and virulence in the subarachnoid space. Following 1 h of CSF exposure, the pyk1∆ mutant possessed equal amounts of ATP per µg protein compared to the WT (Fig. 5A). However, after 24 h of CSF exposure, the pyk1∆ mutant exhibited significantly less ATP than the WT. Removing carbon catabolite repression in the pyk1∆ background (i.e., pyk1∆/mig1∆) did not completely alleviate this energy production deficiency in ex vivo human CSF (Fig. 5A). To further differentiate whether this deficiency was due specifically to the loss of glucose utilization or more generally to the blocked entry of numerous carbon metabolites into oxidative phosphorylation, we also examined ATP production in the hexose kinase mutants (Fig. 5B). The hxk1∆/hxk2∆ mutant is completely unable to grow on glucose as a sole carbon source but can utilize other carbon sources, such as glycerol or lactate (see Fig. S2 in the supplemental material). Interestingly, no statistically significant differences were observed between the hexose kinase single or double mutants and the WT, supporting the conclusion that the ATP production defect in CSF is due to the impaired ability of the pyk1∆ mutants to move carbon metabolites into the TCA cycle as substrates for oxidative phosphorylation.
FIG 5 .
Mutations blocking glucose utilization produce varied effects on ATP production in ex vivo human CSF. WT and mutant strains of C. neoformans were grown in YPL shake culture at 37°C to saturation, then centrifuged, washed with sterile PBS, and resuspended in filter-sterilized pooled human CSF. These CSF shake cultures were then incubated at 37°C, with 15-ml aliquots removed at 1 h and 24 h, and processed as described elsewhere (67). (A) Measurement of ATP levels in the WT, pyk1∆, pyk1∆ PYK1, pyk1∆/mig1∆, and mig1∆ strains following exposure to ex vivo CSF for 1 h or 24 h. Differences between mutant strains and the WT at each time point were compared using Student’s t test (*, P < 0.005). (B) Measurement of ATP levels in the WT, hxk1∆, hxk2∆, and hxk1∆/hxk2∆ strains following exposure to ex vivo CSF for 1 h or 24 h. Differences between mutant strains and the WT at each time point were compared using Student’s t test (α = 0.05).
To determine the cause of the attenuation in virulence in the hxk1∆/hxk2∆ mutant if energy production is not an issue, we performed a macrophage-killing assay. This assay allowed us to assess the ability of strains to survive and proliferate in an intracellular environment. As shown in Fig. S3 in the supplemental material, defects at the beginning and end of glycolysis result in reduced survival within host cells. In fact, the hxk1∆/hxk2∆ mutant is particularly susceptible to killing by macrophages, as this mutant was significantly reduced in survival compared to all other mutant strains tested in this macrophage assay. The hxk1∆/hxk2∆ mutant result is at least somewhat explained due to this mutant’s increased susceptibility to hydrogen peroxide and nitric oxide killing compared to that of the other mutant strains that show no added sensitivity compared to the WT (H2O2 MIC = 250 µM for the hxk1∆/hxk2∆ mutant versus 2,000 µM for the WT; NO− MIC = 62.5 µM for the hxk1∆/hxk2∆ mutant versus 250 µM for the WT).
DISCUSSION
Previous studies by others investigating various carbon metabolic pathways in fungal pathogens have shown the importance that carbon metabolism plays in fungal infection. For example, isocitrate lyase is a gateway enzyme for the glyoxylate shunt that allows for the utilization of 2-carbon compounds such as acetate and ethanol (18, 33, 34). The glyoxylate shunt has been shown to play a significant role in the virulence of numerous plant and animal pathogenic fungal species and interacts with the β-oxidation pathway for fatty acid utilization, as isocitrate lyase mutants in other fungal systems are also unable to utilize fatty acids as sole carbon sources (18, 35–38). However, these data from other fungi stand in direct contrast to observations in C. neoformans and A. fumigatus, where carbon acquisition via the glyoxylate shunt is not required for virulence (22, 24, 39, 40).
In the current study, we have further defined the carbon assimilation pathways in C. neoformans necessary for growth in discrete host microenvironments. We employed two different yet complementary models of C. neoformans virulence, the murine inhalation model and the rabbit CSF model (41, 42). The murine inhalation model corresponds closely to the mechanism of human infection, whereas the rabbit CSF model allows us to specifically probe the influence of the various carbon utilization pathways on CNS persistence. The linkage of gluconeogenesis via Pck1 (see Fig. S1 in the supplemental material) to C. neoformans virulence was first made using a random-insertion mutagenesis screen that identified the virulence-associated DEAD box RNA helicase gene VAD1 by the Williamson laboratory (23). PCK1 is regulated in part by Vad1, encodes a key enzyme for entry of 2- and 3-carbon compounds into gluconeogenesis, and is upregulated in response to low glucose levels. The pck1∆ deletion strain of C. neoformans exhibits severely attenuated virulence in a murine inhalation model of cryptococcosis (23). Using a separately constructed pck1∆ strain, we have shown that although the gene is highly upregulated in the low-glucose environment of the CSF, loss of PCK1 yields no impact on persistence of Cryptococcus in the rabbit CSF model of cryptococcosis (Fig. 1).
Likewise, a prior study demonstrated roles for the nonpreferred carbon utilization regulatory kinase gene SNF1 and the acetyl-CoA synthase gene ACS1 in C. neoformans virulence in a murine inhalation model (21). Interestingly, these mutations either moderately affected or had no effect on persistence of C. neoformans in the rabbit CSF model of disease, which places different host site demands on the yeast regarding establishment of infection compared to those in the murine inhalation model (Fig. 1). These gluconeogenesis-related gene data, along with previously published data on ICL1 (24) and MLS1 (22), which demonstrate no defects in virulence due to lost glyoxylate-shunt function, clearly show that gluconeogenesis is not primarily utilized by C. neoformans to procure nonpreferred carbon (e.g., lactate, acetate, etc.) during growth in the CNS.
The obvious alternative to using gluconeogenesis for carbon acquisition in the CNS is glycolysis. Previous studies of cryptococcosis have touched on pathways linked to glycolysis but have not explicitly addressed the glycolysis pathway and its linkage to the virulence composite of C. neoformans. For example, the trehalose biosynthesis pathway has been studied for both C. neoformans and C. gattii (43, 44). In the most recent study, the relationship between trehalose biosynthesis and hexose kinase activity was demonstrated via the ability of individual hexose kinase deletion mutants to suppress the temperature-sensitive (ts) phenotype of the trehalose pathway mutants (43). However, the specific effect of hexose kinase deletions on the virulence composite was not addressed. Therefore, in this study we showed that hexose kinase activity for glucose utilization is critical both for virulence in the murine inhalation model of cryptococcosis and for acute persistence in the rabbit CSF model (Fig. 3 and 4).
The defined roles of hexose kinase in glucose catabolite repression and glucose metabolism (reviewed in reference 45) allowed us to begin to tease apart glucose utilization versus energy production in regard to their effect on virulence and growth of C. neoformans in the host. Three different mechanisms for glucose catabolite repression that are dependent on the level of extracellular glucose have been described for S. cerevisiae: the Mig1p-independent pathway (very low glucose), the Ras/cyclic AMP (cAMP) pathway (low glucose), and the Mig1p-dependent pathway (high glucose) (30). In the Mig1p-mediated glucose catabolite repression pathway, Hxk2p interacts with Mig1p to enter the nucleus and bind as a complex to glucose-repressible genes (reviewed in reference 45). Our data show a profound attenuation of virulence due to blocked metabolism of glucose in the hxk1∆/hxk2∆ double mutant, in spite of adequate persistence of this mutant strain in the host (Fig. 3B and C and 4). However, ATP production in ex vivo CSF was unaffected by deletion of both hexose kinases (Fig. 5B). Therefore, these data suggest that in the absence of normal Hxk2/Mig1-mediated glucose repression (due to a lack of Hxk2), nonpreferred carbon sources are able to adequately sustain survival of the yeast in the host, with delayed expression of disease symptoms.
We have also demonstrated the importance of glucose metabolism to virulence by examining the role of pyruvate kinase, located at the opposite end of the glycolytic pathway from hexose kinase (see Fig. S1 in the supplemental material). In these studies, blocking glycolysis via deletion of PYK1 again results in attenuated virulence, albeit apparently for a different reason. ATP production in the pyk1∆ mutant was severely impaired after only 24 h of exposure to ex vivo CSF (Fig. 5A); this demonstrates a mechanism for the adverse effect on persistence in the host CSF through reduction in energy formation (Fig. 2A and D). However, glucose catabolite repression is still active in the pyk1∆ mutant, and even though ample lactate is present in CSF, it may not be available for use. Therefore, we attempted to alleviate Mig1-mediated glucose catabolite repression through the deletion of the candidate MIG1 gene in the pyk1∆ background and were able to partially restore ATP production (Fig. 5A). Interestingly, persistence in the host CSF was made even worse than that obtained with the single pyk1∆ mutant (Fig. 2B and D). In other fungal systems, deletion of MIG1 (or its homolog creA) results in reduced/altered growth (46–48). It is possible that loss of Mig1-mediated repression in the pyk1∆/mig1∆ mutant further impairs growth by allowing many carbon metabolic pathways to be activated due to the availability of all present carbon sources in the host. The putative activation of these metabolic pathways may simply outstrip the ability of available lactate to provide adequate ATP via oxidative phosphorylation, due to the block at the step of Pyk1. Alternatively, loss of Mig1-mediated repression may lead to increased activity of alternate glucose repression pathways, as has been described for deletion of HXK2 in budding yeast (49), or other pleiotropic effects, as has been demonstrated for SNF1. Additional experiments will need to be performed to address this issue.
Taken together, the hxk1∆/hxk2∆ and pyk1∆ data support the hypothesis that blocks at different steps in glycolysis can result in completely different mechanisms for virulence attenuation. The hexose kinase double mutant still possesses the ability to produce ATP (Fig. 5B), but it shows increased sensitivity to reactive oxygen species (ROS) and reduced intracellular growth likely due to blocked movement of glucose-6-phosphate into the pentose phosphate pathway (50, 51). The hexose kinase and pyk1∆ single mutants also exhibited decreased survival in the macrophage assay but did not demonstrate particular sensitivity to ROS or nitric oxide, suggesting that the decreased survival in macrophages may have been more closely related to the use of glucose in the media necessitated by this assay. Still, the hxk1∆/hxk2∆ mutant did show significantly decreased survival in macrophages compared to the corresponding single mutants and displays increased sensitivity to ROS compared to all other strains in this study, suggesting that the hxk1∆/hxk2∆ mutant phenotype in vivo may be at least partially due to decreased survival in macrophages. Interestingly, this increased ROS sensitivity should slow the proliferation of the organism in the host, yet our data show that it may eventually adapt and kill the host (Fig. 3C and 4B). Alternatively, blocking efficient ATP (energy) production by deletion of PYK1 (Fig. 5A) produces a strain that does not completely die in the host but persists for long periods of time (Fig. 4). In the rabbit CSF model of acute infection, both blocks in glycolysis provide substantial challenges for the survival of the yeast in this biological fluid and the CNS site of infection (Fig. 2A and 3A).
Mechanistically, it has previously been shown that activated macrophages inhibit growth of C. neoformans in vitro (52). The specific mechanism of growth inhibition by macrophages was first described in regard to interactions with neoplastic tumor cells and involved the inhibition of mitochondrial oxidative phosphorylation in the neoplastic cells (53, 54), which is phenotypically similar to the effect observed in C. neoformans, another eukaryote. It is possible that this yeast interaction with activated macrophages in immunocompetent hosts results in dormancy of the fungus through reduction or elimination of its energy production, which is then able to reestablish infection later when host immune conditions are reduced and thus more conducive for yeast growth by eliminating the block in energy production (55). The pyk1∆ mutation actually mimics impaired oxidative phosphorylation, as evidenced by the decrease in ATP production observed in CSF (Fig. 5A), and further shows greatly impaired proliferation in association with an activated macrophage-like cell line (see Fig. S3 in the supplemental material). These data, along with the long-term persistence data (Fig. 4), allude to the establishment of a dormant-like state in the pyk1∆ mutants due to disrupted oxidative phosphorylation since these cells are viable without causing disease. Therefore, we believe that the pyk1∆ mutants may serve as good model strains for studying the proposed yeast energy impact on dormancy of C. neoformans.
The impact of ATP production on persistence has implications for future antifungal development. For example, the importance of glucose metabolism on cancer growth is the subject of considerable research and is fairly well understood (25). Indeed, glycolysis inhibitors are currently in various stages of clinical trials as viable treatments for cancer (26) and may be of additional benefit in instances where anticancer drug resistance is an issue (56). Therefore, glycolysis inhibitors may provide similar benefits in the prophylaxis and treatment of fungal diseases such as cryptococcosis. The findings presented in this work suggest new targets for antifungal therapy that may help alleviate the problem of antifungal drug resistance by attacking slow-growing or dormant yeast cells.
MATERIALS AND METHODS
Strains and media.
The Cryptococcus neoformans strains used in this study are listed in Table S1 in the supplemental material. The C. neoformans 44-1 and HC1 strains are clinical isolates from deidentified patients with cryptococcal meningitis and were procured from the Duke University Infectious Disease Specimen Repository. All gene deletions were performed in the C. neoformans H99 strain background. Strains were grown using standard yeast media as described previously (57). The 20% lactate solution used to provide a carbon source for the pyk1∆ mutant strains was prepared by diluting concentrated d,l-lactic acid (item no. L6661; Sigma-Aldrich, St. Louis, MO) in sterile distilled H2O (dH2O) and adjusting the pH of the solution to ~4.5 by adding 5.8 g/liter NaOH. Selective media contained either nourseothricin (clonNAT; 100 mg/liter; Werner BioAgents, Jena-Cospeda, Germany) or geneticin (G418; 200 mg/liter; Invitrogen Corp., Carlsbad, CA) for selection of transformants. Capsule-inducing medium (Dulbecco’s modified Eagle’s medium [DMEM] with or without 22 mM NaHCO3 buffer, 10% NCTC 109, 10% fetal calf serum, 1% minimal essential medium [MEM] nonessential amino acid solution, 1% penicillin-streptomycin) was prepared as previously described (58), using carbon sources conducive for growth of the various mutant strains. Melanin production was visually assessed using dopamine agar (59). Urease activity was verified as previously described (60).
Gene deletions.
We identified the C. neoformans PCK1 (CNAG_04217), PYK1 (CNAG_01820), HXK1 (CNAG_05480), and MIG1 (CNAG_06327) genes by searching the C. neoformans H99 predicted-protein database available from the Broad Institute (http://www.broadinstitute.org/annotation/genome/cryptococcus_neoformans/MultiHome.html), using a BLASTp homology search with the Pck1p, Pyk1p, Hxk1p, and Mig1p protein sequences from S. cerevisiae. Genes carrying HXK2 (CNAG_03769), ACS1, and SNF1 have previously been identified and are listed in Table S1 in the supplemental material. The PCK1 and PYK1 gene homologs are present in the C. neoformans genome as single copies, with 66% and 64% identity to the corresponding S. cerevisiae protein sequences, respectively. Hxk1 shows 35% identity to S. cerevisiae Hxk1p and has previously been identified (22). C. neoformans Mig1 exhibits 80% and 89% identity to the S. cerevisiae Mig1p and A. nidulans CreA putative DNA binding domains, respectively.
After identifying the loci of these genes in the H99 genome, we constructed PCR primers to delete the genes using established methods (see Table S2 in the supplemental material) (61). Gene deletions were verified by Southern hybridization (62), and previously unpublished gene deletion strains with altered phenotypes were complemented by reintroduction of the native gene by biolistic transformation as previously described (63). A functional role for the MIG1 gene in the regulation of gluconeogenesis was established by comparison of transcription of putative Mig1-regulated genes (ACS1, PCK1, and FBP1) between the WT and mig1∆ strains as described below for real-time PCR.
Carbon utilization screen.
The WT, pyk1∆, pyk1∆ PYK1, pyk1∆/mig1∆, mig1∆, pck1∆, hxk1∆, hxk2∆, and hxk1∆/hxk2∆ strains were grown to saturation in 5 ml of either yeast extract-peptone-dextrose (YPD) or yeast extract-peptone-lactate-raffinose (YPLR; 1% yeast extract, 2% peptone, 2% lactic acid, 1% raffinose) broth, counted using a hemocytometer, and diluted to 2 × 107 CFU/ml in sterile phosphate-buffered saline (PBS). A 10-fold dilution series from 2 × 107 to 2 × 104 CFU/ml was made for each strain, and 5 µl of each dilution was spotted onto yeast nitrogen base (YNB)-2% glucose, YNB-3% glycerol, and YNB-2% lactate. The plates were incubated at 30°C for 2 to 4 days and photographed.
RNA preparation.
Total RNA was extracted from lyophilized cells using TRIzol reagent (Invitrogen Corp., Carlsbad, CA) and purified using a Qiagen RNeasy kit (Qiagen, Inc., Valencia, CA) in accordance with the manufacturer’s instructions. RNA samples were then stored at −80°C for later analysis. For the evaluation of PCK1 expression from anonymous human samples, RNA was extracted from either fresh (in vivo) or subcultured (in vitro) harvested cells.
Real-time PCR.
cDNA was prepared from RNA purified as described above, using a RETROscript kit (Applied Biosystems/Ambion, Austin, TX). Real-time PCR was performed on the prepared cDNA as previously described (64), using primers listed in Table S1 in the supplemental material. Transcript amplification for PCK1 from each strain and condition was normalized to the amplification of the constitutively expressed GPD1 gene (65). For the ex vivo CSF and in vivo rabbit expression assays, changes in transcript levels were calculated relative to the expression of PCK1 in the WT grown in YPD at 37°C in shake culture.
To verify the identity of the MIG1 gene, the WT and mig1∆ strains were grown in 50 ml YPD shake culture at 30°C overnight, washed once with sterile dH2O, and resuspended in fresh YPD the following day for 1 h at 30°C. Following this incubation, the cultures were centrifuged, and the cell pellets were frozen at −80°C for RNA preparation as described above. The transcription levels of ACS1, FBP1, and PCK1 were normalized to the level for the constitutively expressed GPD1 gene in both strains. Differences in expression (n-fold) were calculated using the Bio-Rad iCycler software system, which utilizes the comparative cycle threshold statistical methods as previously described (66).
Measurement of ATP levels.
Cellular ATP levels were measured as previously described (67), following in vitro CSF exposure. Briefly, fresh colonies of the WT, pyk1∆, pyk1∆ PYK1, pyk1∆/mig1∆, mig1∆, hxk1∆, hxk2∆, and hxk1∆/hxk2∆ strains were grown in 30 ml of YPL broth to saturation at 37°C with shaking. The cultures were centrifuged at 3,000 rpm for 5 min, washed once with PBS, resuspended in 30 ml of filter-sterilized pooled human CSF, and incubated at 37°C with shaking. A 15-ml aliquot of each culture was removed after 1 h and 24 h and processed as follows: 10 ml was centrifuged and washed once with PBS, and the pellet was stored at −80°C; 5 ml was centrifuged and washed once with PBS, and the pellet was stored overnight at −80°C in screw-cap 2-ml tubes for analysis in the ATP assay. Pellets for ATP analysis were resuspended in 1 ml of 50 mM HEPES (pH 7.7) and disrupted using a Mini BeadBeater-16 (BioSpec Products, Inc., Bartlesville, OK) with 6.3-mm stainless steel beads. Samples were disrupted using one 45-s pulse and kept on ice before and after disruption. The disrupted samples were centrifuged at 5,000 rpm at 4°C for 10 min, and the supernatants were collected to determine ATP concentrations using an ATP bioluminescence assay kit (Sigma-Aldrich, St. Louis, MO). Soluble-protein concentrations were measured for an aliquot of the supernatant from each sample using the Bradford assay (68). ATP concentrations were normalized for direct comparison using soluble-protein concentrations for each sample. The mean measurements for three separate experiments are reported.
In vivo virulence experiments.
Virulence of the C. neoformans strains was assessed using both a murine inhalation model and a rabbit CSF model of cryptococcosis as previously described (42, 69). For the murine inhalation model, female A/Jcr mice were intranasally inoculated with 105 CFU/ml of each C. neoformans strain (10 mice per strain in survival studies; 3 mice for the WT and 27 mice per mutant strain for the fungal persistence study). For the rabbit CSF model, male New Zealand White (NZW) rabbits were treated with cortisone via daily injection and intrathecal inoculation into the subarachnoid space with 108 CFU of each C. neoformans strain (3 rabbits per group). Animals were evaluated daily for signs of morbidity and were sacrificed at predetermined clinical endpoints correlating with mortality per approval from the Duke Institutional Animal Care and Use Committee Guidelines. Differences between strains in the murine inhalation model were assessed using the log rank test in JMP version 8 (SAS Institute, Inc., Cary, NC). Differences between strains in the rabbit CSF model were determined by an analysis-of-variance (ANOVA) comparison using the Fit Model process in JMP version 8.
SUPPLEMENTAL MATERIAL
Supplemental methods. Download Text S1, DOC file, 0.088 MB.
Overview of core carbon metabolism in C. neoformans. Proteins encoded by genes examined in this study are highlighted in red at the enzymatic steps or the regulatory processes that they perform. *, gene regulation relationships are shown for the situation in S. cerevisiae; similar regulation may occur in C. neoformans, but more-detailed analyses are needed. Download Figure S1, PDF file, 0.044 MB.
Examples of carbon utilization capability for glycolysis/gluconeogenesis mutants. The WT, pyk1Δ, pyk1Δ PYK1, pyk1Δ/mig1Δ, mig1Δ, pck1Δ, hxk1Δ, hxk2Δ, and hxk1Δ/hxk2Δ strains were grown to saturation in 5 ml of either YPD or YPLR broth, counted using a hemocytometer, and diluted to 2 × 107 CFU/ml in sterile PBS. A 10-fold dilution series from 2 × 107 to 2 × 104 CFU/ml was made for each strain, and 5 µl of each dilution was spotted onto YNB-2% glucose, YNB-3% glycerol, and YNB-2% lactate. These carbon sources are assimilated at early, middle, and late steps in glycolysis. The plates were incubated at 30°C for 2 to 4 days and photographed. Download Figure S2, PDF file, 0.423 MB.
Glycolysis defects reduce survival and proliferation of C. neoformans in macrophages. Activated J774A.1 macrophages were challenged with either the WT, the pyk1Δ, the pyk1Δ-PYK1, the hxk1Δ, the hxk2Δ, or the hxk1Δ/hxk2Δ strain of C. neoformans (multiplicity of infection, 1:1). After 60 min of coincubation at 37°C in 5% CO2, extracellular yeasts were removed and the cocultures were incubated at 37°C in 5% CO2 overnight. Viable C. neoformans organisms were plated on YPL agar plates and incubated at 30°C for 2 days for CFU quantification. Strains were assayed in triplicate, and error bars represent standard error. Differences in numbers of CFU between strains were assessed using Student’s t test (*, P < 0.001 for comparison to the WT; **, P < 0.01 for comparison to all strains). Download Figure S3, PDF file, 0.033 MB.
List of strains used in this study.
DNA PCR primers used in this study.
ACKNOWLEDGMENTS
We thank Connie Nichols and Andrew Alspaugh for thoughtful discussions of the data and its implications.
This work was supported by NIH PHS grants AI73896 and AI28388 (J.R.P.).
Footnotes
Citation Price MS, et al. 2011. Cryptococcus neoformans requires a functional glycolytic pathway for disease but not persistence in the host. mBio 2(3):e00103-11. doi:10.1128/mBio.00103-11.
REFERENCES
- 1. Casadevall A, Perfect JR. 1998. Cryptococcus neoformans. ASM Press, Washington, DC [Google Scholar]
- 2. Park BJ, et al. 2009. Estimation of the current global burden of cryptococcal meningitis among persons living with HIV/AIDS. AIDS 23:525–530 [DOI] [PubMed] [Google Scholar]
- 3. Fraser JA, et al. 2005. Same-sex mating and the origin of the Vancouver Island Cryptococcus gattii outbreak. Nature 437:1360–1364 [DOI] [PubMed] [Google Scholar]
- 4. Fraser JA, Subaran RL, Nichols CB, Heitman J. 2003. Recapitulation of the sexual cycle of the primary fungal pathogen Cryptococcus neoformans var. gattii: implications for an outbreak on Vancouver Island, Canada. Eukaryot. Cell 2:1036–1045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Kidd S, et al. 2004. A rare genotype of Cryptococcus gattii caused the cryptococcosis outbreak on Vancouver Island (British Columbia, Canada). Proc. Natl. Acad. Sci. U. S. A. 101:17258–17263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Perfect JR, Casadevall A. 2002. Cryptococcosis. Infect. Dis. Clin. North Am. 16:837–874 [DOI] [PubMed] [Google Scholar]
- 7. Diamond RD, Bennett JE. 1973. Growth of Cryptococcus neoformans within human macrophages in vitro. Infect. Immun. 7:231–236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Feldmesser M, Kress Y, Novikoff P, Casadevall A. 2000. Cryptococcus neoformans is a facultative intracellular pathogen in murine pulmonary infection. Infect. Immun. 68:4225–4237 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Feldmesser M, Tucker S, Casadevall A. 2001. Intracellular parasitism of macrophages by Cryptococcus neoformans. Trends Microbiol. 9:273–278 [DOI] [PubMed] [Google Scholar]
- 10. Levitz SM, et al. 1999. Cryptococcus neoformans resides in an acidic phagolysosome of human macrophages. Infect. Immun. 67:885–890 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Ma H, Croudace JE, Lammas DA, May RC. 2006. Expulsion of live pathogenic yeast by macrophages. Curr. Biol. 16:2156–2160 [DOI] [PubMed] [Google Scholar]
- 12. Perfect JR. 2006. Cryptococcus neoformans: a sugar-coated killer, p. 281–303 In Heitman J, Filler SG, Edwards JE, Mitchell AP, Molecular principles of fungal pathogenesis. ASM Press, Washington, DC [Google Scholar]
- 13. Buchanan KL, Murphy JW. 1998. What makes Cryptococcus neoformans a pathogen? Emerg. Infect. Dis. 4:71–83 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Chayakulkeeree M, Perfect JR. 2006. Cryptococcosis. Infect. Dis. Clin. North Am. 20:507–544 [DOI] [PubMed] [Google Scholar]
- 15. Moyrand F, Klaproth B, Himmelreich U, Dromer F, Janbon G. 2002. Isolation and characterization of capsule structure mutant strains of Cryptococcus neoformans. Mol. Microbiol. 45:837–849 [DOI] [PubMed] [Google Scholar]
- 16. Barelle CJ, et al. 2006. Niche-specific regulation of central metabolic pathways in a fungal pathogen. Cell. Microbiol. 8:961–971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Lorenz MC, Bender JA, Fink GR. 2004. Transcriptional response of Candida albicans upon internalization by macrophages. Eukaryot. Cell 3:1076–1087 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Lorenz MC, Fink GR. 2001. The glyoxylate cycle is required for fungal virulence. Nature 412:83–86 [DOI] [PubMed] [Google Scholar]
- 19. Yin Z, et al. 2003. Glucose triggers different global responses in yeast, depending on the strength of the signal, and transiently stabilizes ribosomal protein mRNAs. Mol. Microbiol. 48:713–724 [DOI] [PubMed] [Google Scholar]
- 20. Lee A, et al. 2010. Survival defects of Cryptococcus neoformans mutants in human cerebrospinal fluid results in attenuated virulence in an experimental model of meningitis. Infect. Immun. 78:4213–4225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Hu G, Cheng PY, Sham A, Perfect JR, Kronstad JW. 2008. Metabolic adaptation in Cryptococcus neoformans during early murine pulmonary infection. Mol. Microbiol. 69:1456–1475 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Idnurm A, Giles SS, Perfect JR, Heitman J. 2007. Peroxisome function regulates growth on glucose in the basidiomycete fungus Cryptococcus neoformans. Eukaryot. Cell 6:60–72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Panepinto J, et al. 2005. The DEAD-box RNA helicase Vad1 regulates multiple virulence-associated genes in Cryptococcus neoformans. J. Clin. Invest. 115:632–641 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Rude TH, Toffaletti DL, Cox GM, Perfect JR. 2002. Relationship of the glyoxylate pathway to the pathogenesis of Cryptococcus neoformans. Infect. Immun. 70:5684–5694 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Vander Heiden MG, Cantley LC, Thompson CB. 2009. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324:1029–1033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Pelicano H, Martin DS, Xu RH, Huang P. 2006. Glycolysis inhibition for anticancer treatment. Oncogene 25:4633–4646 [DOI] [PubMed] [Google Scholar]
- 27. Haarasilta S, Oura E. 1975. On the activity and regulation of anaplerotic and gluconeogenetic enzymes during the growth process of Baker’s yeast. The biphasic growth. Eur. J. Biochem. 52:1–7 [DOI] [PubMed] [Google Scholar]
- 28. Yang J, et al. 2010. Regulation of virulence factors, carbon 1 utilization and virulence by SNF1 in Cryptococcus neoformans JEC21 and divergent actions of SNF1 between cryptococcal strains. Fungal Genet. Biol. 47:994–1000 [DOI] [PubMed] [Google Scholar]
- 29. Ronne H. 1995. Glucose repression in fungi. Trends Genet. 11:12–17 [DOI] [PubMed] [Google Scholar]
- 30. Yin Z, Smith RJ, Brown AJ. 1996. Multiple signalling pathways trigger the exquisite sensitivity of yeast gluconeogenic mRNAs to glucose. Mol. Microbiol. 20:751–764 [DOI] [PubMed] [Google Scholar]
- 31. Wishart DS, et al. 2008. The human cerebrospinal fluid metabolome. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 871:164–173 [DOI] [PubMed] [Google Scholar]
- 32. Flipphi M, et al. 2003. Onset of carbon catabolite repression in Aspergillus nidulans. Parallel involvement of hexokinase and glucokinase in sugar signaling. J. Biol. Chem. 278:11849–11857 [DOI] [PubMed] [Google Scholar]
- 33. Fernández E, Moreno F, Rodicio R. 1992. The ICL1 gene from Saccharomyces cerevisiae. Eur. J. Biochem. 204:983–990 [DOI] [PubMed] [Google Scholar]
- 34. Kornberg HL. 1966. The role and control of the glyoxylate cycle in Escherichia coli. Biochem. J. 99:1–11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Cánovas D, Andrianopoulos A. 2006. Developmental regulation of the glyoxylate cycle in the human pathogen Penicillium marneffei. Mol. Microbiol. 62:1725–1738 [DOI] [PubMed] [Google Scholar]
- 36. Idnurm A, Howlett BJ. 2002. Isocitrate lyase is essential for pathogenicity of the fungus Leptosphaeria maculans to canola (Brassica napus). Eukaryot. Cell 1:719–724 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Solomon PS, Lee RC, Wilson TJ, Oliver RP. 2004. Pathogenicity of Stagonospora nodorum requires malate synthase. Mol. Microbiol. 53:1065–1073 [DOI] [PubMed] [Google Scholar]
- 38. Wang ZY, Thornton CR, Kershaw MJ, Debao L, Talbot NJ. 2003. The glyoxylate cycle is required for temporal regulation of virulence by the plant pathogenic fungus Magnaporthe grisea. Mol. Microbiol. 47:1601–1612 [DOI] [PubMed] [Google Scholar]
- 39. Olivas I, et al. 2008. Ability to grow on lipids accounts for the fully virulent phenotype in neutropenic mice of Aspergillus fumigatus null mutants in the key glyoxylate cycle enzymes. Fungal Genet. Biol. 45:45–60 [DOI] [PubMed] [Google Scholar]
- 40. Schöbel F, et al. 2007. Aspergillus fumigatus does not require fatty acid metabolism via isocitrate lyase for development of invasive aspergillosis. Infect. Immun. 75:1237–1244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Lim TS, Murphy JW, Cauley LK. 1980. Host-etiological agent interactions in intranasally and intraperitoneally induced cryptococcosis in mice. Infect. Immun. 29:633–641 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Perfect JR, Lang SD, Durack DT. 1980. Chronic cryptococcal meningitis: a new experimental model in rabbits. Am. J. Pathol. 101:177–194 [PMC free article] [PubMed] [Google Scholar]
- 43. Ngamskulrungroj P, et al. 2009. The trehalose synthesis pathway is an integral part of the virulence composite for Cryptococcus gattii. Infect. Immun. 77:4584–4596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Petzold EW, et al. 2006. Characterization and regulation of the trehalose synthesis pathway and its importance in the pathogenicity of Cryptococcus neoformans. Infect. Immun. 74:5877–5887 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Gancedo JM. 2008. The early steps of glucose signalling in yeast. FEMS Microbiol. Rev. 32:673–704 [DOI] [PubMed] [Google Scholar]
- 46. Breslow DK, et al. 2008. A comprehensive strategy enabling high-resolution functional analysis of the yeast genome. Nat. Methods 5:711–718 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. David H, Krogh AM, Roca C, Akesson M, Nielsen J. 2005. CreA influences the metabolic fluxes of Aspergillus nidulans during growth on glucose and xylose. Microbiology (Reading, Engl.) 151:2209–2221 [DOI] [PubMed] [Google Scholar]
- 48. Shroff RA, O’Connor SM, Hynes MJ, Lockington RA, Kelly JM. 1997. Null alleles of creA, the regulator of carbon catabolite repression in Aspergillus nidulans. Fungal Genet. Biol. 22:28–38 [DOI] [PubMed] [Google Scholar]
- 49. Kümmel A, et al. 2010. Differential glucose repression in common yeast strains in response to HXK2 deletion. FEMS Yeast Res. 10:322–332 [DOI] [PubMed] [Google Scholar]
- 50. Brown SM, Upadhya R, Shoemaker JD, Lodge JK. 2010. Isocitrate dehydrogenase is important for nitrosative stress resistance in Cryptococcus neoformans, but oxidative stress is not dependent on glucose-6-phosphate dehydrogenase. Eukaryot. Cell 9:971–980 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Juhnke H, Krems B, Kötter P, Entian KD. 1996. Mutants that show increased sensitivity to hydrogen peroxide reveal an important role for the pentose phosphate pathway in protection of yeast against oxidative stress. Mol. Gen. Genet. 252:456–464 [DOI] [PubMed] [Google Scholar]
- 52. Granger DL, Perfect JR, Durack DT. 1986. Macrophage-mediated fungistasis in vitro: requirements for intracellular and extracellular cytotoxicity. J. Immunol. 136:672–680 [PubMed] [Google Scholar]
- 53. Granger DL, Lehninger AL. 1982. Sites of inhibition of mitochondrial electron transport in macrophage-injured neoplastic cells. J. Cell Biol. 95:527–535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Granger DL, Taintor RR, Cook JL, Hibbs JB. 1980. Injury of neoplastic cells by murine macrophages leads to inhibition of mitochondrial respiration. J. Clin. Invest. 65:357–370 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Garcia-Hermoso D, Janbon G, Dromer F. 1999. Epidemiological evidence for dormant Cryptococcus neoformans infection. J. Clin. Microbiol. 37:3204–3209 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Xu RH, et al. 2005. Inhibition of glycolysis in cancer cells: a novel strategy to overcome drug resistance associated with mitochondrial respiratory defect and hypoxia. Cancer Res. 65:613–621 [PubMed] [Google Scholar]
- 57. Sherman F. 1991. Getting started with yeast. Methods Enzymol. 194:3–21 [DOI] [PubMed] [Google Scholar]
- 58. Alspaugh J, Perfect J, Heitman J. 1998. Cryptococcus neoformans mating and virulence are regulated by the G-protein alpha subunit GPA1 and cAMP. Genes Dev. 11:3206–3217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Chaskes S, Tyndall RL. 1975. Pigment production by Cryptococcus neoformans from para- and ortho-diphenols: effect of the nitrogen source. J. Clin. Microbiol. 1:509–514 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Price MS, Nichols CB, Alspaugh JA. 2008. The Cryptococcus neoformans Rho-GDP dissociation inhibitor mediates intracellular survival and virulence. Infect. Immun. 76:5729–5737 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Davidson RC, et al. 2002. A PCR-based strategy to generate integrative targeting alleles with large regions of homology. Microbiology 148:2607–2615 [DOI] [PubMed] [Google Scholar]
- 62. Southern EM. 1975. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J. Mol. Biol. 98:503–517 [DOI] [PubMed] [Google Scholar]
- 63. Toffaletti DL, Rude TH, Johnston SA, Durack DT, Perfect JR. 1993. Gene transfer in Cryptococcus neoformans by use of biolistic delivery of DNA. J. Bacteriol. 175:1405–1411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Cramer KL, Gerrald QD, Nichols CB, Price MS, Alspaugh JA. 2006. Transcription factor Nrg1 mediates capsule formation, stress response, and pathogenesis in Cryptococcus neoformans. Eukaryot. Cell 5:1147–1156 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Varma A, Kwon-Chung KJ. 1999. Characterization of the glyceraldehyde-3-phosphate dehydrogenase gene [correction of glyceraldehyde-3-phosphate gene] and the use of its promoter for heterologous expression in Cryptococcus neoformans, a human pathogen. Gene 232:155–163 [DOI] [PubMed] [Google Scholar]
- 66. Vandesompele J, et al. 2002. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 3:RESEARCH0034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Garcia J, et al. 2008. Mathematical modeling of pathogenicity of Cryptococcus neoformans. Mol. Syst. Biol. 4:183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Bradford MM. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248–254 [DOI] [PubMed] [Google Scholar]
- 69. Cox GM, Mukherjee J, Cole GT, Casadevall A, Perfect JR. 2000. Urease as a virulence factor in experimental cryptococcosis. Infect. Immun. 68:443–448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Nielsen K, et al. 2005. Cryptococcus neoformans {alpha} strains preferentially disseminate to the central nervous system during coinfection. Infect. Immun. 73:4922–4933 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental methods. Download Text S1, DOC file, 0.088 MB.
Overview of core carbon metabolism in C. neoformans. Proteins encoded by genes examined in this study are highlighted in red at the enzymatic steps or the regulatory processes that they perform. *, gene regulation relationships are shown for the situation in S. cerevisiae; similar regulation may occur in C. neoformans, but more-detailed analyses are needed. Download Figure S1, PDF file, 0.044 MB.
Examples of carbon utilization capability for glycolysis/gluconeogenesis mutants. The WT, pyk1Δ, pyk1Δ PYK1, pyk1Δ/mig1Δ, mig1Δ, pck1Δ, hxk1Δ, hxk2Δ, and hxk1Δ/hxk2Δ strains were grown to saturation in 5 ml of either YPD or YPLR broth, counted using a hemocytometer, and diluted to 2 × 107 CFU/ml in sterile PBS. A 10-fold dilution series from 2 × 107 to 2 × 104 CFU/ml was made for each strain, and 5 µl of each dilution was spotted onto YNB-2% glucose, YNB-3% glycerol, and YNB-2% lactate. These carbon sources are assimilated at early, middle, and late steps in glycolysis. The plates were incubated at 30°C for 2 to 4 days and photographed. Download Figure S2, PDF file, 0.423 MB.
Glycolysis defects reduce survival and proliferation of C. neoformans in macrophages. Activated J774A.1 macrophages were challenged with either the WT, the pyk1Δ, the pyk1Δ-PYK1, the hxk1Δ, the hxk2Δ, or the hxk1Δ/hxk2Δ strain of C. neoformans (multiplicity of infection, 1:1). After 60 min of coincubation at 37°C in 5% CO2, extracellular yeasts were removed and the cocultures were incubated at 37°C in 5% CO2 overnight. Viable C. neoformans organisms were plated on YPL agar plates and incubated at 30°C for 2 days for CFU quantification. Strains were assayed in triplicate, and error bars represent standard error. Differences in numbers of CFU between strains were assessed using Student’s t test (*, P < 0.001 for comparison to the WT; **, P < 0.01 for comparison to all strains). Download Figure S3, PDF file, 0.033 MB.
List of strains used in this study.
DNA PCR primers used in this study.