Abstract
Background
Correlations between Educational Attainment (EA) and measures of cognitive performance are as high as 0.8. This makes EA an attractive alternative phenotype for studies wishing to map genes affecting cognition due to the ease of collecting EA data compared to other cognitive phenotypes such as IQ.
Methodology
In an Australian family sample of 9538 individuals we performed a genome-wide association scan (GWAS) using the imputed genotypes of ∼2.4 million single nucleotide polymorphisms (SNP) for a 6-point scale measure of EA. Top hits were checked for replication in an independent sample of 968 individuals. A gene-based test of association was then applied to the GWAS results. Additionally we performed prediction analyses using the GWAS results from our discovery sample to assess the percentage of EA and full scale IQ variance explained by the predicted scores.
Results
The best SNP fell short of having a genome-wide significant p-value (p = 9.77×10−7). In our independent replication sample six SNPs among the top 50 hits pruned for linkage disequilibrium (r2<0.8) had a p-value<0.05 but only one of these SNPs survived correction for multiple testing - rs7106258 (p = 9.7*10−4) located in an intergenic region of chromosome 11q14.1. The gene based test results were non-significant and our prediction analyses show that the predicted scores explained little variance in EA in our replication sample.
Conclusion
While we have identified a polymorphism chromosome 11q14.1 associated with EA, further replication is warranted. Overall, the absence of genome-wide significant p-values in our large discovery sample confirmed the high polygenic architecture of EA. Only the assembly of large samples or meta-analytic efforts will be able to assess the implication of common DNA polymorphisms in the etiology of EA.
Introduction
Access to education is considered to be a predictor for a wide range of later life outcomes such as employment [1], income [2], and health outcomes such as obesity [3], [4]. In addition to its relevance to economics and health, educational attainment (EA) is a measure of interest in the study of cognitive abilities/intelligence. The correlation between EA and measures of cognitive ability ranges from 0.45 up to 0.80 [5], [6], [7]. Three hypotheses of causality have been postulated to explain the underlying mechanisms of the relationship between EA and cognitive functioning, (i) intelligence/cognitive abilities are a cause of EA because intelligence is believed to be more biologically anchored than scholastic achievements [8], (ii) cognitive abilities are a product of scholastic achievement [9], [10] and (iii) basic cognitive processes such as reaction time, inspection time and memory recall partly determine both scholastic achievement and cognitive performance [11]. At present, none of these hypotheses have been discarded and it is likely that a mixture of all three plays a role in the link between EA and cognitive abilities. Additionally, a recent report suggested that the causal relationship between EA and intelligence varies according to an individual's level of intelligence [7].
Biological processes involved in scholastic achievement and cognitive performance are likely to be shared to some extent. Twin studies that take advantage of differences between genetically identical twins (monozygotic or MZ) and fraternal twins (dizygotic or DZ), have shown that EA and cognitive performance are influenced to a large extent by common genetic factors [12], [13], [14] and the heritability of EA has been estimated as high as 80% [6], [15]. Therefore, investigating the biological etiology of educational attainment could provide insights into the molecular basis of cognition because of its strong heritability and high correlation with cognitive performance. Moreover, EA has the advantage of being a measure that is much easier to collect than IQ and therefore it is more viable to assemble the large cohorts necessary to perform genome wide association scans (GWAS).
Molecular genetics studies of EA have been largely limited to a candidate gene approach including the Catechol-O-methyltransferase (COMT) [16], [17], the Brain-derived neurotrophic factor (BDNF) [18] and the Dopamine receptor D4 (DRD4) [19]. The choice of these genes as candidates for EA is strongly hypothesis driven due to their characterized neurobiological functions but convincing replications are lacking in support of their hypothetical role in the etiology of EA. The advances in microarray technologies in recent years have made it possible to genotype millions of single nucleotide polymorphisms (SNPs) for a low cost (about US$500 per individual). This has led to a major shift toward a hypothesis free genome-wide association study design. A lot of GWAS studies has emerged in the literature, identifying hundreds of polymorphisms and genes associated with complex traits and diseases. In this study we present the results of a GWAS for EA, using an Australian twin family discovery sample of 9538 individuals from the general population and a further 968 individuals as an independent replication sample.
Methods
Participants
The participants for our discovery sample were drawn from two cohorts of adult twin families (cohorts 1 and 2) that have taken part in a wide range of studies of health and well-being. Individuals from Cohort 1 were born before 1964 and individuals from cohort 2 were born between 1964 and 1981. Both these cohorts have participated in previous postal questionnaire and telephone interview studies, and recruitment was extended to their relatives (parents, siblings, adult children and spouses). Our total discovery sample was composed of 9538 individuals for whom both EA and genome-wide SNP genotype data were available. Our replication sample (Cohort 3) consisted of 968 individuals who are the parents of adolescent twins participating in our melanoma risk factors and cognition studies (1996-ongoing) [20]. Information for the different studies is available in Table S1.
For the prediction analysis of cognitive ability based on the EA GWAS results obtained in the discovery sample, we used one of the twin children of cohort 3. Their full scale IQ (FSIQ) was collected as part of the cognition study and genotyping data were available for 1842 adolescent twins and their siblings ranging in age from 15 to 22 years (mean = 16.28 years ±0.45 SD).
Ethical approval, for the studies from which the data drawn, was obtained from the Human Research Ethics Committee of the Queensland Institute of Medical Research. Informed written consent for all measures was obtained from each participant and their parents/guardian if participants were younger than 18 years of age.
Educational attainment
Self reported educational attainment (EA) was collected as part as of questionnaires and telephone interviews. In the adult cohorts (1 and 2) three similar education scales were used to collect EA depending on the study in which an individual participated (fig. 1). Each individual score was transformed to create a new 6-level EA scale harmonised across the studies, with 1 = 7 years or less of schooling, 2 = 8 to 10 years of schooling, 3 = 8 to 10 years of schooling + apprenticeship or 11 to 12 years of schooling or 12 years of schooling + apprenticeship, 4 = teacher college or technical college, 5 = university undergraduate training and 6 = university postgraduate training (fig. 1). For a number of individuals (n = 5314) multiple reports of EA were available and the highest education level reported was selected for analysis. In cohort 3, EA was also assessed as part of a questionnaire that the parents of the adolescent twins answered while their children underwent cognitive testing. EA was recorded with one of the scales previously used in the adult cohorts (Figure 1). All individuals that were included in the GWAS analysis were at least 21 years of age (the standard age of first degree graduation). Descriptive statistics of the age and educational attainment of the participants according to their study of origin can be found in Table S1.
Figure 1. Transformation applied to the original education scales (1 to 3) to obtain a new harmonized scale across studies.
Cognitive abilities
Cognitive abilities were measured by full scale IQ (FSIQ) that was assessed using the Multi-dimensional Aptitude Battery (MAB) [21]. The MAB is a general intelligence test designed to mirror the WAIS-R [22] and it is presented in a multiple-choice format. Participants completed three verbal (information, arithmetic, vocabulary) and two performance subtests (spatial, object assembly) which were combined here to form a full-scale IQ score. Twins completed the MAB as close as possible to their 16th birthday when they came to participate in the cognition study. Further details of the IQ testing procedure have been previously published [23], [24].
Genotyping and Imputation
The genotypic data used in the current study come from a large genotyping project involving nine waves of genotyping that used three different Illumina SNP chips (Human610-Quad, HumanCNV370-Quadv3 and Human 317K) [25]. For each genotyping wave, rigorous quality control (QC) steps were applied to ensure the highest standard of the pre imputation data. Details of the QC steps have been described elsewhere [25]. Imputation of the autosomal chromosomes was performed in two stages with MACH [26] using a set of SNPs (N = 269840) common to the different Illumina chips. These data were screened for ancestry outliers. Full siblings and offspring of individuals who had been identified as ancestry outliers were excluded from the reference set used in MACH stage 1. In the first stage, the data from a set of 450 reference individuals from our set were compared to the phased haplotype data from the HapMap samples of european ancestry (CEU I+II) (release 22, build 36). These 450 reference individuals were made up of fifty unrelated individuals (with the lowest missingness) from each of the nine subsamples. Stage 1 generated recombination and error files that describe how our data relate to the HapMap data, in effect allowing us to customise the HapMap data for our population. In the second stage, data were imputed for the 17,862 individuals using the HapMap data (release 22, build 36) as the reference panel and the recombination and error files generated in stage 1 to customise the imputation. SNPs with a minor allele frequency <.01, SNPs with an Rsq imputation quality score <.3 were excluded. A panel of 2,480,163 autosomal SNPs and a panel of 13783 genotyped X-chromosome SNPs were use for association analysis.
Data analysis
Genome wide association analysis using dosage scores was performed in MERLIN offline [27] to account for family structure. The association analysis of genotyped markers on the X-chromosome were conducted in Minx (as implemented in MERLIN). Sex, year of birth (YOB), age, YOB×sex, YOB2, YOB2×sex, age2, Age×sex and Age2×Sex were used as covariates. Both YOB and age were used as covariates in this study: YOB in order to account for changes in terms of access to education that have occurred since the early 1900's and age in order to account for the fact that the older an individual is, the less likely he/she is to undertake further education. Visualisation and annotation of the GWAS results was conducted using WGAViewer [28] and the pruning for linkage desquilibrium (LD) (r2<0.8) of the top 200 SNPs was performed in SNAP [29] based on the HapMap release 22 CEU panel.
Additionally, The VEGAS gene-based test [30] that can be used with related individuals was performed using the GWAS output data of our discovery sample. The test summarizes evidence for association on a per gene basis by considering the full set of SNPs within the gene (determined by SNPs lying within ±50 kb of a gene's 5′ and 3′ UTRs) and the LD between them.
Power analyses
It is expected that many genes of very small effect size contribute to the genetic variance of complex traits. We estimated the empirical power our discovery sample provides to detect genetic variants explaining 1%, 0.5% and 0.2% of the phenotypic variance by running association tests on simulated datasets in Merlin. The simulated datasets are similar to the original data in terms of marker informativeness, spacing, allele frequency, trait distribution, and missing data patterns, but they are simulated such that a selected SNP accounts for a specified proportion of the variance. The selected SNP had minor allele frequency 0.25. Association analysis was conducted on 1000 data sets generated by the simulation procedure. The empirical power is estimated as that proportion of the 1000 association analyses in which a genome-wide significant association (α = 5*10−8) was detected. Results indicated that our sample provides 100%, 80% (799 out of the 1000 simulations) and 13% (130 out of the 1000 simulations) power to detect SNPs that explain 1%, 0.5% and 0.2% of the variance in EA, respectively.
Prediction analyses
Prediction analyses were conducted in two stages. In stage 1, the effect sizes of the 2,480,163 SNPs from GWAS for EA, as well as 5 sub-panels of SNPs (based on p-value thresholds of p<0.5, p<0.4, p<0.3, p<0.2 and p<0.1), were extracted from the MERLIN [27] output of our discovery sample. Based on the effect sizes for these panels and the imputed SNP data of our adolescent twin families we generated 6 sets of prediction scores using the PLINK [31] scoring routine for cohort 3 (968 parents and only one of their twin children (n = 799) in order to have a sample of independent individuals). In stage 2, we compared the predicted scores of the parents to their EA by fitting EA as a function of the predicted scores in a linear model (EA∼predicted scores) using R (http://cran.r-project.org/) to evaluate to percentage of variance explained (R2) by the predicted score and its level of significance. Similarly, we fitted the FSIQ of one of the twin children as a function of their predicted scores in a linear model (FSIQ∼predicted scores).
Results
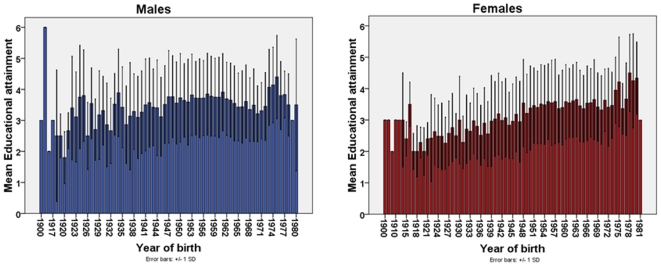
We examined educational attainment for over 9538 individuals born between 1900 and 1981 from 3764 families. A similar distribution of the mean educational attainment was observed between males and females (figure 2). We observed a gradual increase of the mean EA from 8–10 years of schooling to above 11 to 12 years of schooling for individuals born during the first half of the 20th century (Figure 2). The mean EA of individuals born between the late 40's up to the mid 70's stayed constant around 3.5 before increasing close to 4 (4 = teacher college or technical college) for individuals born after the early to mid 70's (figure 2).
Figure 2. Mean educational attainment (EA) by year of birth in male and female.
Using DNA previously collected for these individuals we performed a GWAS for EA. The heritability of EA was estimated 63.5% by MERLIN and is comparable to the 57% previously reported in our earlier variance components analysis of EA in cohort 1 [6]. Table 1 reports the results for the top 50 most associated SNPs after pruning for LD (r2<0.8), with the best-associated SNP rs2680324 having a p-value of 9.77×10−7 (the Q-Q plot is presented in Figure S1 and the manhattan plot in Figure S2). We then tested the association of these 50 SNPs with EA in an independent replication sample of 968 individuals. Five of these SNPs had an association p = value below 0.05 and an effect in the same direction: rs226039 (p = 0.040), rs976928 (p = 0.0036), rs4298928 (p = 0.004), rs7106258 (p = 0.00097) and rs226047 (p = 0.039). However, only rs7106258 survived correction for multiple testing (p = 0.05/50 = 0.001).
Table 1. Top 50 most associated SNPs in 9538 individuals after pruning for LD (r2<0.8).
| rank | SNP | Chr. | Position in bp | SNP type | Closest gene | p-value | SNP in LD* |
| 1 | rs2680324 | 10 | 56970071 | INTRONIC | PCDH15 | 9.77E-07 | 2 |
| 2 | rs10079475 | 5 | 179585240 | INTRONIC | RASGEF1C | 1.36E-06 | 3 |
| 3 | rs8085689 | 18 | 21656634 | INTRONIC | TTC39C | 5.20E-06 | 2 |
| 4 | rs13092348 | 3 | 116580097 | INTRONIC | LSAMP | 5.40E-06 | 17 |
| 5 | rs1521417 | 3 | 116761113 | INTRONIC | LSAMP | 5.55E-06 | 2 |
| 6 | rs11004778 | 10 | 56974190 | INTRONIC | PCDH15 | 5.58E-06 | 9 |
| 7 | rs2246477 | 5 | 172208454 | UPSTREAM | Y_RNA.479 | 7.27E-06 | 1 |
| 8 | rs914925 | 9 | 93584793 | INTRONIC | SYK | 8.16E-06 | 2 |
| 9 | rs226039 | 16 | 21288101 | INTRONIC | CRYM; | 8.98E-06 | 0 |
| 10 | rs976928 | 11 | 81479043 | INTERGENIC | RP11-179A16.1 | 9.60E-06 | 6 |
| 11 | rs7191909 | 16 | 21262897 | 3PRIME UTR | ANKS4B | 1.02E-05 | 15 |
| 12 | rs4298928 | 11 | 81470414 | INTERGENIC | RP11-179A16.1 | 1.11E-05 | 0 |
| 13 | rs6767669 | 3 | 116599500 | INTRONIC | LSAMP | 1.19E-05 | 0 |
| 14 | rs6581191 | 12 | 58875698 | INTERGENIC | RP11-652N17.1 | 1.23E-05 | 0 |
| 15 | rs7281948 | 21 | 32970928 | INTERGENIC | FBXW11P1 | 1.23E-05 | 2 |
| 16 | rs1517635 | 2 | 224203115 | INTERGENIC | AC068035.1 | 1.32E-05 | 0 |
| 17 | rs8082313 | 17 | 17976932 | INTERGENIC | C17orf39 | 1.40E-05 | 0 |
| 18 | rs2431664 | 5 | 172203911 | WITHIN NON CODING GENE | RP11-779O18.3 | 1.50E-05 | 0 |
| 19 | rs17157509 | 7 | 109881775 | INTERGENIC | AC003088.1 | 1.52E-05 | 6 |
| 20 | rs12497655 | 3 | 116764010 | INTRONIC | LSAMP | 1.82E-05 | 1 |
| 21 | rs13402289 | 2 | 100659389 | INTRONIC | AFF3 | 1.89E-05 | 6 |
| 22 | rs7106258 | 11 | 81484019 | INTERGENIC | RP11-179A16.1 | 1.92E-05 | 14 |
| 23 | rs10068846 | 5 | 179588797 | INTRONIC | RASGEF1C | 2.01E-05 | 0 |
| 24 | rs5009524 | 20 | 38894887 | INTERGENIC | RP1-191L6.2 | 2.17E-05 | 0 |
| 25 | rs226047 | 16 | 21301472 | INTRONIC | CRYM | 2.30E-05 | 3 |
| 26 | rs744684 | 7 | 154196001 | INTRONIC | DPP6 | 2.32E-05 | 0 |
| 27 | rs226038 | 16 | 21288338 | INTRONIC | CRYM | 2.37E-05 | 0 |
| 28 | rs6691053 | 1 | 173868955 | DOWNSTREAM | ZBTB37 | 2.37E-05 | 0 |
| 29 | rs1011689 | 10 | 97760476 | INTRONIC | CC2D2B | 2.38E-05 | 6 |
| 30 | rs4876153 | 8 | 2304334 | INTERGENIC | AC133633.2 | 2.38E-05 | 0 |
| 31 | rs7672521 | 4 | 175302173 | UPSTREAM | RP11-51M24.1 | 2.58E-05 | 0 |
| 32 | rs2029238 | 18 | 21702106 | INTRONIC | AC090772.1 | 2.63E-05 | 2 |
| 33 | rs9903961 | 17 | 10732872 | UPSTREAM | PIRT | 2.75E-05 | 0 |
| 34 | rs1122208 | 15 | 58623525 | INTERGENIC | LIPC | 2.82E-05 | 3 |
| 35 | rs11194701 | 10 | 111292456 | UPSTREAM | BTF3P15 | 2.92E-05 | 0 |
| 36 | rs2297679 | 1 | 32157009 | INTRONIC | COL16A1 | 3.00E-05 | 1 |
| 37 | rs2044541 | 11 | 41343185 | INTRONIC | LRRC4C | 3.36E-05 | 1 |
| 38 | rs1793791 | 11 | 131111013 | WITHIN NON CODING GENE | AP002856.7 | 3.61E-05 | 0 |
| 39 | rs6011899 | 20 | 61356257 | INTRONIC | NTSR1 | 3.76E-05 | 0 |
| 40 | rs472975 | 1 | 185534476 | WITHIN NON CODING GENE | GS1-204I12.2 | 3.83E-05 | 12 |
| 41 | rs256940 | 5 | 114866191 | INTRONIC | FEM1C | 3.84E-05 | 0 |
| 42 | rs4514783 | 18 | 61918306 | INTERGENIC | AC100848.1 | 3.85E-05 | 4 |
| 43 | rs11876820 | 18 | 61891408 | INTERGENIC | AC100848.1 | 4.12E-05 | 0 |
| 44 | rs2275998 | 11 | 66326581 | INTRONIC | CTD-3074O7.2 | 4.15E-05 | 0 |
| 45 | rs7936167 | 11 | 40156059 | INTRONIC | LRRC4C | 4.40E-05 | 4 |
| 46 | rs6986402 | 8 | 17630589 | INTRONIC | MTUS1 | 4.43E-05 | 1 |
| 47 | rs10057387 | 5 | 172230003 | INTERGENIC | RP11-536N17.1 | 4.49E-05 | 0 |
| 48 | rs6938281 | 6 | 25312488 | INTRONIC | LRRC16A | 4.54E-05 | 0 |
| 49 | rs2257055 | 9 | 130311404 | INTRONIC | FAM129B | 4.57E-05 | 0 |
| 50 | rs6584475 | 10 | 103806793 | INTRONIC | C10orf76 | 4.68E-05 | 1 |
*number of SNPs in LD (r2≥0.8) among the top 200 GWAS hits.
To completely utilise the GWAS results, we then performed a gene-based test that maps SNPs to their respective genes if they are located within 50 kb of a gene locus. The gene-based test produced an empirical p-value for 17549 autosomal genes. The top 50 most associated genes are reported in table 2 and the best result was obtained for FLJ41766 (p = 1.4×10−5), however, it does not reach the multiple-testing correction threshold (p = 0.05/17549 = 2.8*10−6).
Table 2. Top 50 most associated genes with the EA in 9538 individuals.
| rank | Chr. | Gene | Number of SNPs | Start position | Stop position | P-value |
| 1 | 16 | FLJ41766 | 35 | 21219670 | 21237413 | 1.40E-05 |
| 2 | 16 | CRYM | 64 | 21177342 | 21221918 | 2.10E-05 |
| 3 | 16 | ANKS4B | 82 | 21152516 | 21171251 | 2.80E-05 |
| 4 | 10 | NPM3 | 24 | 103531071 | 103533148 | 6.10E-05 |
| 5 | 10 | FGF8 | 22 | 103519876 | 103525817 | 8.80E-05 |
| 6 | 10 | MGEA5 | 31 | 103534198 | 103568165 | 0.000117 |
| 7 | 14 | LTB4R2 | 110 | 23848000 | 23850798 | 0.000154 |
| 8 | 10 | HPS6 | 12 | 103815136 | 103817783 | 0.000155 |
| 9 | 14 | C14orf21 | 115 | 23838937 | 23844214 | 0.000171 |
| 10 | 14 | ADCY4 | 122 | 23857409 | 23873704 | 0.000175 |
| 11 | 19 | ZNF100 | 83 | 21698682 | 21742270 | 0.000232 |
| 12 | 14 | RABGGTA | 76 | 23804583 | 23810643 | 0.000237 |
| 13 | 5 | DUSP1 | 108 | 172127706 | 172130809 | 0.000249 |
| 14 | 10 | CC2D2B | 80 | 97749872 | 97782431 | 0.000263 |
| 15 | 14 | TGM1 | 80 | 23788159 | 23802256 | 0.000319 |
| 16 | 10 | KCNIP2 | 29 | 103575720 | 103593667 | 0.000538 |
| 17 | 1 | ZBTB37 | 53 | 172104115 | 172122397 | 0.000553 |
| 18 | 1 | DARS2 | 45 | 172060580 | 172094305 | 0.000617 |
| 19 | 18 | CABYR | 76 | 19972952 | 19995562 | 0.000629 |
| 20 | 10 | CCNJ | 84 | 97793143 | 97810612 | 0.000723 |
| 21 | 17 | DYNLL2 | 49 | 53515797 | 53521810 | 0.000783 |
| 22 | 11 | FLRT1 | 99 | 63627937 | 63643221 | 0.000844 |
| 23 | 10 | C10orf76 | 89 | 103595345 | 103805922 | 0.000847 |
| 24 | 7 | C7orf59 | 44 | 99584465 | 99589769 | 0.000872 |
| 25 | 11 | BBS1 | 64 | 66034694 | 66057660 | 0.000953 |
| 26 | 19 | ZNF99 | 88 | 22730846 | 22744624 | 0.000961 |
| 27 | 11 | ACTN3 | 56 | 66070966 | 66087373 | 0.001017 |
| 28 | 7 | C7orf43 | 41 | 99589978 | 99594238 | 0.00102 |
| 29 | 17 | CCDC43 | 45 | 40110330 | 40122691 | 0.001079 |
| 30 | 11 | CTSF | 50 | 66087510 | 66092623 | 0.001108 |
| 31 | 14 | NFATC4 | 104 | 23907093 | 23918650 | 0.001127 |
| 32 | 11 | DPP3 | 54 | 66004455 | 66033706 | 0.00114 |
| 33 | 11 | ZDHHC24 | 57 | 66063310 | 66070247 | 0.00117 |
| 34 | 15 | RHCG | 89 | 87815643 | 87840803 | 0.001346 |
| 35 | 3 | APEH | 52 | 49686438 | 49695938 | 0.00135 |
| 36 | 7 | GAL3ST4 | 52 | 99594800 | 99604309 | 0.001364 |
| 37 | 1 | SERPINC1 | 57 | 172139564 | 172153096 | 0.001451 |
| 38 | 7 | BET1 | 79 | 93458935 | 93471626 | 0.001466 |
| 39 | 3 | RHOA | 51 | 49371582 | 49424530 | 0.001497 |
| 40 | 17 | SPHK1 | 52 | 71892296 | 71895536 | 0.001549 |
| 41 | 3 | MST1 | 48 | 49696391 | 49701099 | 0.001573 |
| 42 | 18 | CD226 | 199 | 65681172 | 65775212 | 0.00158 |
| 43 | 3 | RNF123 | 53 | 49701993 | 49733966 | 0.00163 |
| 44 | 5 | C5orf39 | 57 | 43074938 | 43076098 | 0.00166 |
| 45 | 11 | PELI3 | 39 | 65990911 | 66001384 | 0.001683 |
| 46 | 6 | LY6G6F | 110 | 31782662 | 31786351 | 0.00177 |
| 47 | 3 | BSN | 87 | 49566925 | 49683986 | 0.001818 |
| 48 | 7 | STAG3 | 50 | 99613473 | 99649946 | 0.00182 |
| 49 | 6 | BAT5 | 131 | 31762714 | 31779067 | 0.00186 |
| 50 | 6 | LY6G6C | 97 | 31794403 | 31797489 | 0.00188 |
Our Prediction analyses of EA in our replication sample using predicted scores for different thresholds of GWAS significance (from p<0.1 to the whole genome) showed a regression r2 ranging between 0.0011 to 0.0023 (non-significant: p≥0.14) between the predicted scores and EA in the independent sample of adults in all 6 analyses. Similarly, in the children of these individuals (n = 799) when we attempted to predict FSIQ as a function of the predicted scores across the different thresholds of GWAS significance the regression r2 were non-significant (p≥0.15) and varied between 9.0*10−5 to 0.0026.
Discussion
In the current study we observed that Educational attainment (EA) has increased constantly during the 20th century from a mean EA of 8–10 years of schooling to around 12 years of schooling followed by apprenticeship, teacher college or technical college. This trend was the same for males and females. However, it should be noted that only 2% of our sample was born before 1926 and 31% before 1951. Thus the data are too sparse to comment on the changes of mean EA in relation to time specific events such as World War II. The majority of our sample was born post World War II and the mean EA for these individuals confirms that pursuing a tertiary education was much more common for these generations than it was for the previous generations. The mean EA for individuals born in this period remained fairly constant and major events such as the Vietnam War (Australian conscription: 1965–1975) or free access to university education (1974–1988) do not appear to have affected the level of EA in our sample.
Using SNP genotyping data for these individuals, we performed a large GWAS study for EA. The strength of our study was its large sample size that conferred 100% and 80% power to detect polymorphisms (MAF = 0.25) explaining 1% and 0.5% of the phenotypic variance respectively. However, despite a good power, no SNP had a p-value significant at the genome wide level of 5×10−8. Therefore, we tested the robustness of the top 50 most associated SNPs pruned for LD (r2<0.8) in a replication sample. Five of these 50 SNPs reached a p-value smaller than p = 0.05 and an effect in the same direction. Among these SNPs only one had a p-value surviving multiple correction, rs7106258 (p = 0.00097) located in an intergenic region of chromosome 11q14.1 (combined p-value was 3.71*10−7). This region deserves future attention as it has been suggested to be involved in other brain phenotypes. This region contains the GAB2 gene, a candidate gene for Alzheimer's disease [32] and copy number variations in this region have also been linked with mental retardation [33].
Overall our GWAS findings are comparable to the latest GWAS report for general cognitive abilities in that no SNPs survived multiple testing correction for genome wide significance [34]. The estimated heritability of EA in our sample was 63.2% and comparable to the 57% found in our behavior genetic analysis of EA [6]. However, we also know from our previous study that the heritability of EA could be as high as 82% in Australia once corrected for assortative mating [6]. So why can we not find SNPs with genome wide significant p-values for a highly heritable trait such as EA? Some elements of an answer can be drawn from what we have learned from other human complex traits such as IQ [34] and height [35] in which common variants of large effect size were not found. Common explanations for the missing phenotypic variance due to genetic variation might be (i) a large number of common variants of small effect sizes (<1%), (ii) rare variants of large effect sizes, (iii) structural variants or (iv) low power to detect epistasis or gene-environment interactions [36]. These questions are being addressed with the formation of a new consortium and meta-analytical efforts that assemble large samples to examine if a large number of common variants of small effect sizes contribute to the current lack of robust association signals. Some new elements of an answer to the above question are already available with Yang and colleagues [37] recently showing that 45% of variance for human height can be explained when all SNP effects from a panel of nearly 300 000 SNPs were considered simultaneously. On the other hand, the potential role of rare and structural variants in the etiology of EA and other complex traits may be answered by the next-generation of association studies [38]. An example of an innovative study design was recently described by Holm et al [39] and was successful to detect low frequency SNPs associated with sick sinus syndrome. Their multi step approach consisted of a classic GWAS followed by whole genome sequencing for a small number of cases and controls, before imputing the newly discovered variants into their original sample. Although the above example was applied to a case-control setting this could be adapted to quantitative traits by sequencing a small number of individuals at the extremes of the distribution.
Another noticeable feature of our GWAS results is that none of the top 50 SNPs fall into exons. This is not surprising as more than 80% of associated variants detected by GWAS were found in non-coding regions [36] which further supports the importance of including these regions in gene mapping studies of complex traits [40].
Moving from a traditional single marker analysis, we performed the VEGAS gene-based test [30]. This is an attractive approach that gives a second life to GWAS data and it might ease the current frustration of the millions of dollars spent in large genotyping projects that cannot produce replicated associations. FLJ 41766 was the best hit, however, there is no evidence for this gene to suggest a role in neurobiology. A similar observation was found when looking at the genes among the top hits one at the time. Additionally, both the gene-based test and GWAS showed no evidence of association for BDNF (rank = 16132 out of 17549 genes tested by VEGAS), COMT (rank = 197), and DRD4 (rank = 17451), which have been previously associated with EA [16], [17], [18], [19]. However, these candidate gene studies were conducted on small samples that may have not produced genuine association signals. If true, these association signals previously reported should have been detected in our large sample (>9 500 individuals).
One of the major goals of genetic epidemiologists is to identify genetic variants that can be used to predict complex traits and susceptibility risk to diseases. So far, this has been harder than originally thought due to the difficulty of mapping loci of small effect that reflect the highly polygenic architecture of common diseases and complex traits. One recent approach that has been used in this exercise is to generate a predicted score based on a genome wide profile of SNPs effects [41], [42]. Here, we generated an EA predicted score based on different level of p-values obtained in the GWAS of our large discovery sample to see whether or not we could predict EA in an independent sample. The EA variance explained by the six sets of predicted scores was low and non significant. When a similar prediction analysis was performed with FSIQ instead of EA, this percentage of variance was lower than with EA as expected and also non-significant. These low correlations between the predicted phenotypes scores might arise from a low accuracy of the genome wide SNP effects that were used to generate these scores. In the future, it will be most interesting to see the evolution of these results if these analyses were to be repeated once genome wide SNP effects from a larger sample or from a meta-analysis are available.
In the present study we performed one of the largest genome wide association scans to examine the molecular genetics of educational attainment. Despite our large discovery sample and good genome coverage of the genome, no SNP reached genome wide evidence of association. As for many other complex traits (e.g. Human heights [43]), our results confirmed the high polygenic architecture of EA. Future large consortiums combined with sequencing efforts will hopefully bring more insights into the molecular architecture of EA and shed light on whether it is common variants of small effects or rare variants of large effect that contribute to the biological blue print of EA.
Supporting Information
Quantile-Quantile plot from the GWAS result of educational attainment in 9538 individuals. (Lambda = 1.0229). The grey shade area represents the 95% confidence intervals.
(TIFF)
Manhattan plot of GWAS for educational attainment for 9538 individuals.
X-axis represents the chromosomal location for each SNPs, and Y-axis the −log10 P-value for association with educational attainment.
(TIF)
Descriptive statistics for the different cohorts from which the educational attainment data originated.
(DOC)
Acknowledgments
We thank the twins and their families for their participation. We also thank Dixie Statham, Ann Eldridge, Marlene Grace, Kerrie McAloney (sample collection); Lisa Bowdler, Steven Crooks (DNA processing); David Smyth, Harry Beeby, and Daniel Park (IT support).
Footnotes
Competing Interests: The authors have declared that no competing interests exist.
Funding: Funding was provided by the Australian National Health and Medical Research Council (241944, 339462, 389927, 389875, 389891, 389892, 389938, 442915, 442981, 496739, 552485, 552498), the Australian Research Council (A7960034, A79906588, A79801419, DP0770096, DP0212016, DP0343921), the FP-5 GenomEUtwin Project (QLG2-CT-2002-01254), and the U.S. National Institutes of Health (NIH grants AA07535, AA10248, AA13320, AA13321, AA13326, AA14041, MH66206). A portion of the genotyping on which this study was based (Illumina 370K scans) was carried out at the Center for Inherited Disease Research, Baltimore (CIDR), through an access award to the authors' late colleague Dr. Richard Todd (Psychiatry, Washington University School of Medicine, St Louis). Imputation was carried out on the Genetic Cluster Computer, which is financially supported by the Netherlands Scientific Organization (NWO 480-05-003). N.W.M and K.J.H.V. are supported by a PhD scholarship from the ANZ trust. S.E.M., D.R.N., and G.W.M. are supported by the National Health and Medical Research Council (NHMRC) Fellowship Scheme. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Inglis PA, Stromback T. Migrants unemployment: the determinants of employment success. The Economic Record. 1986;62:310–324. [Google Scholar]
- 2.Chiswick BR, Miller PW. Immigrant generation and in Australia. The Economic Record. 1986;61:540–553. [Google Scholar]
- 3.Swinburn B, Egger G. Preventive strategies against weight gain and obesity. Obes Rev. 2002;3:289–301. doi: 10.1046/j.1467-789x.2002.00082.x. [DOI] [PubMed] [Google Scholar]
- 4.Webbink D, Martin NG, Visscher PM. Does education reduce the probability of being overweight? J Health Econ. 2010;29:29–38. doi: 10.1016/j.jhealeco.2009.11.013. [DOI] [PubMed] [Google Scholar]
- 5.Lynn R, Mikk J. National differences in intelligence and educational attainment. Intelligence. 2007:115–121. [Google Scholar]
- 6.Baker LA, Treloar SA, Reynolds CA, Heath AC, Martin NG. Genetics of educational attainment in Australian twins: sex differences and secular changes. Behav Genet. 1996;26:89–102. doi: 10.1007/BF02359887. [DOI] [PubMed] [Google Scholar]
- 7.Johnson W, Deary IJ, Iacono WG. Genetic and Environmental Transactions Underlying Educational Attainment. Intelligence. 2009;37:466–478. doi: 10.1016/j.intell.2009.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jensen AR. The g factor: the science of mental ability. Westport: Praeger Publishers; 1998. [Google Scholar]
- 9.Ceci SJ. How much does schooloing influence development and its cognitive components? A reassessment of the evidence. Developmental Psychology. 1991;27:703–722. [Google Scholar]
- 10.Ceci SJ. Schooling and intelligence. Psychological Science Agenda. 1992;5:7–9. [Google Scholar]
- 11.Luo D, Thompson LA, Detterman DK. The causal factor underlying the correlation between psychometric g and scholastic performance. Intelligence. 2003;31:67–83. [Google Scholar]
- 12.Wainwright MA, Wright MJ, Luciano M, Geffen GM, Martin NG. Multivariate genetic analysis of academic skills of the Queensland core skills test and IQ highlight the importance of genetic g. Twin Res Hum Genet. 2005;8:602–608. doi: 10.1375/183242705774860259. [DOI] [PubMed] [Google Scholar]
- 13.Bartels M, Rietveld MJ, Van Baal GC, Boomsma DI. Heritability of educational achievement in 12-year-olds and the overlap with cognitive ability. Twin Res. 2002;5:544–553. doi: 10.1375/136905202762342017. [DOI] [PubMed] [Google Scholar]
- 14.Johnson W, McGue M, Iacono WG. Genetic and environmental influences on academic achievement trajectories during adolescence. Developmental Psychology. 2005;42:514–532. doi: 10.1037/0012-1649.42.3.514. [DOI] [PubMed] [Google Scholar]
- 15.Szanton SL, Johnson B, Thorpe RJ, Whitfield K. Education in time: cohort differences in educational attainment in African-American twins. PLoS One. 2009;4:e7664. doi: 10.1371/journal.pone.0007664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Enoch MA, Waheed JF, Harris CR, Albaugh B, Goldman D. COMT Val158Met and cognition: main effects and interaction with educational attainment. Genes Brain Behav. 2009;8:36–42. doi: 10.1111/j.1601-183X.2008.00441.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yeh TK, Chang CY, Hu CY, Yeh TC, Lin MY. Association of catechol-O-methyltransferase (COMT) polymorphism and academic achievement in a Chinese cohort. Brain Cogn. 2009;71:300–305. doi: 10.1016/j.bandc.2009.07.011. [DOI] [PubMed] [Google Scholar]
- 18.Zdanys KF, Kleiman TG, Zhang H, Ozbay F, Macavoy MG, et al. BDNF Variants, Premorbid Educational Attainment, and Disease Characteristics in Alzheimer's Disease: An Exploratory Study. J Alzheimers Dis. 2009;17:887–898. doi: 10.3233/JAD-2009-1106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ham BJ, Lee YM, Kim MK, Lee J, Ahn DS, et al. Personality, dopamine receptor D4 exon III polymorphisms, and academic achievement in medical students. Neuropsychobiology. 2006;53:203–209. doi: 10.1159/000094729. [DOI] [PubMed] [Google Scholar]
- 20.Luciano M, Wright MJ, Geffen GM, Geffen LB, Smith GA, et al. A genetic investigation of the covariation among inspection time, choice reaction time, and IQ subtest scores. Behav Genet. 2004;34:41–50. doi: 10.1023/B:BEGE.0000009475.35287.9d. [DOI] [PubMed] [Google Scholar]
- 21.Jackson DN. Multidimentional Aptitude Battery Test: Manual. London, Canada: Research Psychologist Press; 1984. [Google Scholar]
- 22.Wechsler D. Manual for the Wechsler Adult Inteligence Scale-Revised (WAIS-R) San Antonio, TX: The Psychological Corporation; 1981. [Google Scholar]
- 23.Luciano M, Wright M, Smith GA, Geffen GM, Geffen LB, et al. Genetic covariance among measures of information processing speed, working memory, and IQ. Behav Genet. 2001;31:581–592. doi: 10.1023/a:1013397428612. [DOI] [PubMed] [Google Scholar]
- 24.Wainwright M, Wright MJ, Geffen GM, Geffen LB, Luciano M, et al. Genetic and environmental sources of covariance between reading tests used in neuropsychological assessment and IQ subtests. Behav Genet. 2004;34:365–376. doi: 10.1023/B:BEGE.0000023642.34853.cb. [DOI] [PubMed] [Google Scholar]
- 25.Medland SE, Nyholt DR, Painter JN, McEvoy BP, McRae AF, et al. Common variants in the trichohyalin gene are associated with straight hair in Europeans. Am J Hum Genet. 2009;85:750–755. doi: 10.1016/j.ajhg.2009.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li Y, Abecasis GR. Mach 1.0: Rapid haplotype reconstruction and missing genotype inference. Am J Hum Genet. 2006:2290. [Google Scholar]
- 27.Chen WM, Abecasis GR. Family-based association tests for genomewide association scans. Am J Hum Genet. 2007;81:913–926. doi: 10.1086/521580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ge D, Zhang K, Need AC, Martin O, Fellay J, et al. WGAViewer: software for genomic annotation of whole genome association studies. Genome Res. 2008;18:640–643. doi: 10.1101/gr.071571.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Johnson AD, Handsaker RE, Pulit SL, Nizzari MM, O'Donnell CJ, et al. SNAP: a web-based tool for identification and annotation of proxy SNPs using HapMap. Bioinformatics. 2008;24:2938–2939. doi: 10.1093/bioinformatics/btn564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liu JZ, McRae AF, Nyholt DR, Medland SE, Wray NR, et al. A versatile gene-based test for genome-wide association studies. 2010. Submitted. [DOI] [PMC free article] [PubMed]
- 31.Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet. 2007;81:559–575. doi: 10.1086/519795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Reiman EM, Webster JA, Myers AJ, Hardy J, Dunckley T, et al. GAB2 alleles modify Alzheimer's risk in APOE epsilon4 carriers. Neuron. 2007;54:713–720. doi: 10.1016/j.neuron.2007.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Koolen DA, Sistermans EA, Nilessen W, Knight SJ, Regan R, et al. Identification of non-recurrent submicroscopic genome imbalances: the advantage of genome-wide microarrays over targeted approaches. Eur J Hum Genet. 2008;16:395–400. doi: 10.1038/sj.ejhg.5201975. [DOI] [PubMed] [Google Scholar]
- 34.Davis OS, Butcher LM, Docherty SJ, Meaburn EL, Curtis CJ, et al. A Three-Stage Genome-Wide Association Study of General Cognitive Ability: Hunting the Small Effects. Behav Genet. 2010;40:759–767. doi: 10.1007/s10519-010-9350-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Visscher PM. Sizing up human height variation. Nat Genet. 2008;40:489–490. doi: 10.1038/ng0508-489. [DOI] [PubMed] [Google Scholar]
- 36.Manolio TA, Collins FS, Cox NJ, Goldstein DB, Hindorff LA, et al. Finding the missing heritability of complex diseases. Nature. 2009;461:747–753. doi: 10.1038/nature08494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yang J, Benyamin B, McEvoy BP, Gordon S, Henders AK, et al. Common SNPs explain a large proportion of the heritability for human height. Nat Genet. 2010;42:565–569. doi: 10.1038/ng.608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zeggini E. Next-generation association studies for complex traits. Nat Genet. 2011;43:287–288. doi: 10.1038/ng0411-287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Holm H, Gudbjartsson DF, Sulem P, Masson G, Helgadottir HT, et al. A rare variant in MYH6 is associated with high risk of sick sinus syndrome. Nat Genet. 2011;43:316–320. doi: 10.1038/ng.781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hindorff LA, Sethupathy P, Junkins HA, Ramos EM, Mehta JP, et al. Potential etiologic and functional implications of genome-wide association loci for human diseases and traits. Proc Natl Acad Sci U S A. 2009;106:9362–9367. doi: 10.1073/pnas.0903103106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wray NR, Goddard ME, Visscher PM. Prediction of individual genetic risk to disease from genome-wide association studies. Genome Res. 2007;17:1520–1528. doi: 10.1101/gr.6665407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lee SH, van der Werf JH, Hayes BJ, Goddard ME, Visscher PM. Predicting unobserved phenotypes for complex traits from whole-genome SNP data. PLoS Genet. 2008;4:e1000231. doi: 10.1371/journal.pgen.1000231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lango Allen H, Estrada K, Lettre G, Berndt SI, Weedon MN, et al. Hundreds of variants clustered in genomic loci and biological pathways affect human height. Nature. 2010;467:832–838. doi: 10.1038/nature09410. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Quantile-Quantile plot from the GWAS result of educational attainment in 9538 individuals. (Lambda = 1.0229). The grey shade area represents the 95% confidence intervals.
(TIFF)
Manhattan plot of GWAS for educational attainment for 9538 individuals.
X-axis represents the chromosomal location for each SNPs, and Y-axis the −log10 P-value for association with educational attainment.
(TIF)
Descriptive statistics for the different cohorts from which the educational attainment data originated.
(DOC)