Abstract
This review encompasses the extant literature on West Nile virus (WNV)-associated neurologic complications and also includes some of the author's experiences with Louisiana patients infected with WNV in the Summer of 2002 who sustained neurologic sequelae. In the appropriate clinical context, patients should be screened for WNV.
West Nile virus (WNV) infection may induce encephalitis, meningitis, and acute flaccid paralysis (AFP), which may occur concomitantly or in isolation (1). In the initial North American outbreak of WNV infections, the AFP was diagnosed as Guillain-Barre syndrome (GBS) (2). However, more recent reports have attributed the AFP to poliomyelitis, which has both important therapeutic and prognostic implications (3, 4).
Effects of WNV infection span the range from asymptionatic to severe neurologic complications and death (3). Fortunately, most WNV infections are subclinical, and even symptomatic patients usually experience merely a mild, febrile illness (5). The diagnosis of acute WNV infection is verified by demonstrating WNV-derived RNA in serum, cerebrospinal fluid (CSF), or other tissue (5).
Meningitis or encephalitis develops in less than 1% of infected individuals, is more likely to occur in those over the age of 50 years, and is frequently associated with flaccid muscle weakness (5). During a Romanian WNV epidemic in 1996, 15% of patients with encephalitis died and those with meningitis had a 1.8% mortality rate (6). WNV-associated neurologic illness was first recognized in North America in 1999 (1). In this initial outbreak, patients who sustained flaccid paralysis were diagnosed with GBS (2).
GBS stems from an inflammatory response against peripheral nerve myelin or axons, which is often induced by a preceding diarrheal illness (7). Typical additional features include symmetric, flaccid weakness; sensory symptoms, such as paresthesias; and “albuminocytologic dissociation” in the CSF (i.e., an increased protein level in the absence of a pleocytosis) (7). Electromyography (EMG) typically reveals evidence of demyelination or axonal damage to both sensory and motor nerves. GBS may respond favorably to immunomodulatory therapy, such as plasmapheresis (PLEX) or intravenous immunoglobulin (IVIG) (7).
Viral poliomyelitis results from destruction of spinal cord or brainstem (bulbar) motor neurons. Given that these are CNS neurons, poliomyelitis unfortunately has no effective therapy (7). Though wild-type poliovirus has been essentially eradicated in North America due to effective immunization programs, other viruses, including coxsackievirus and echovirus, may induce poliomyelitis (7). Poliomyelitis is typically associated with asymmetric limb weakness, absent sensory symptoms, and EMG findings of motor axon dysfunction (absolute distinction between damage to motor axons and neurons is not possible on routine EMG) (7, 8).
During the WNV outbreak in the Summer of 2002, six patients in Louisiana and Mississippi with WNV-induced AFP were systematically evaluated (3). Asymmetric weakness, absent sensory complaints, CSF pleocytosis, and EMG features consistent with active motor axonal dysfunction were typical (3). These findings were attributed to acute poliomyelitis, which was later also posited by other clinicians encountering WNV-induced AFP (4). This is congruous with the extant neuropathological data pertaining to WNV infections in horses (9) and humans (Sejvar J, personal communication). Spinal cord MRI was unrevealing, as is sometimes the case in poliomyelitis (10).
Additional clinical features observed in Louisiana patients with WNV encephalitis during 2002 included a variety of movement disorders (dyskinesias). Interestingly, seizures were quite rare. Parkinsonian signs including reduced facial expression (hypomimia), limb bradykinesia, muscle rigidity, and postural instability (a tendency to fall backwards) were common. Though rest tremors were not observed, postural and action tremors were. Myoclonus was also noted in a number of patients. One patient had vertical supranuclear gaze palsy. Cranial MRIs in these patients with various dyskinesias often revealed focal lesions in subcortical grey matter, including the basal ganglia and thalamus (Figure).
Figure 1.
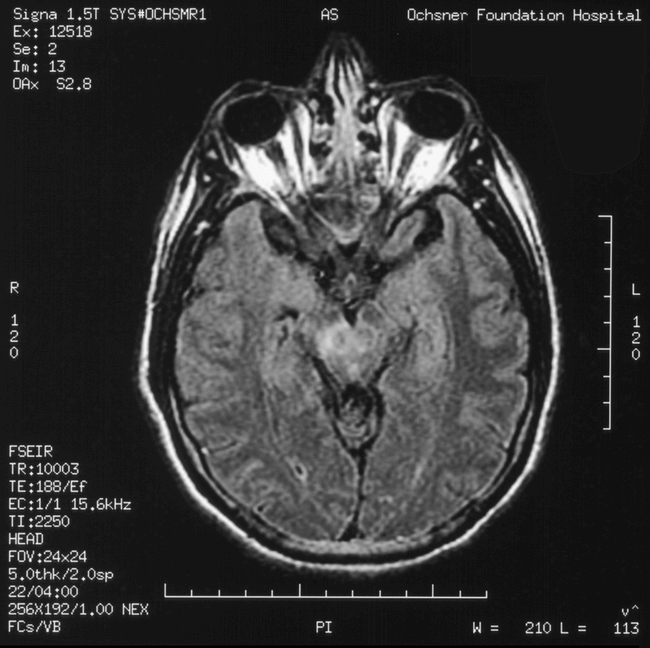
Hyperintensity of the right substantia nigra is evident on this axial (FLAIR or fluid-attenuated inversion recovery) MRI.
Prior to 1996, few cases of severe neurologic sequelae associated with WNV infections in humans were reported (11). Unfortunately, with increasing WNV outbreaks this is changing (11). Clinicians should maintain a high index of suspicion for WNV infection in patients with meningitis or encephalitis especially if associated with asymmetric AFP. On those with AFP, an EMG should be obtained expeditiously to more precisely localize the disease process (e.g., peripheral nerve versus motor neuron). Also, patients with AFP should undergo a lumbar puncture, even in the absence of nuchal rigidity, headache, or altered mental status. A CSF pleocytosis should suggest a diagnosis other than GBS. A caveat to this is that patients with GBS secondary to HIV infection may have a CSF pleocytosis (12), but the EMG will facilitate sorting this out. Such an approach will minimize the chance that inappropriate therapy (e.g., IVIG) is initiated.

Dr. Van Gerpen is a neurophysiology and movement disorders specialist on staff in Ochsner's Department of Neurology.
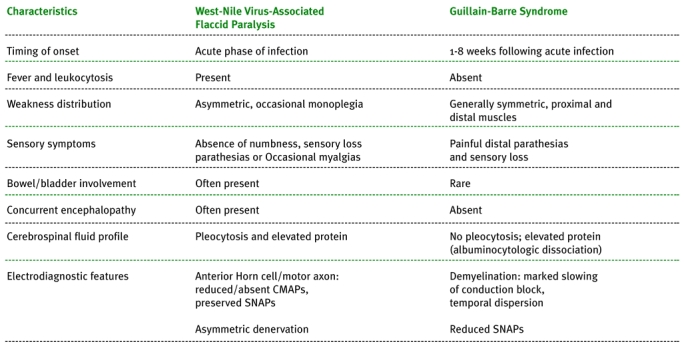
Table. Clinical characteristics of patients with West Nile virus-associated acute flaccid paralysis compared to typical Guillain-Barre syndrome (7). CMAP = compound muscle action potential, SNAP = sensory nerve action potential
REFERENCES
- Nash D., Mostashari F., Fine A. The outbreak of West Nile virus infection in the New York City area in 1999. N Engl J Med. 2001;344:1807–1814. doi: 10.1056/NEJM200106143442401. [DOI] [PubMed] [Google Scholar]
- Ahmed S., Libman R., Wesson K. Guillain-Barre syndrome: an unusual presentation of West Nile virus infection. Neurology. 2000;55:144–146. doi: 10.1212/wnl.55.1.144. [DOI] [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention. Acute flaccid paralysis syndrome associated with West Nile virus infection - Mississippi and Louisiana, July–August 2002. MMWR Morb Mortal Wkly Rep. 2002;51:825–828. [PubMed] [Google Scholar]
- Glass J. D., Samuels O., Rich M. M. Poliomyelitis due to West Nile virus (letter) N Engl J Med. 2002;347:1280–1281. doi: 10.1056/NEJM200210173471616. [DOI] [PubMed] [Google Scholar]
- Petersen L. R., Roehrig J. T., Hughes J. M. West Nile virus encephalitis. N Engl J Med. 2002;347:1225–1226. doi: 10.1056/NEJMo020128. [DOI] [PubMed] [Google Scholar]
- Ceausu E., Erscoiu S., Calistru P. Clinical manifestations in the West Nile virus outbreak. Rom J Virol. 1997;48:3–11. [PubMed] [Google Scholar]
- Gorson K. C., Ropper A. H. Nonpoliovirus poliomyelitis simulating Guillain-Barre syndrome. Arch Neurol. 2001;58:1460–1464. doi: 10.1001/archneur.58.9.1460. [DOI] [PubMed] [Google Scholar]
- Kimura J. Electrodiagnosis in Diseases of Nerve and Muscle: Principles and Practice. 2nd ed. 1989:249–274. Philadelphia, F.A. Davis Co. [Google Scholar]
- Cantile C., DiGuardo G., Eleni C., Arispici M. Clinical and neuropathological features of West Nile virus equine encephalomyelitis in Italy. Equine Vet J. 2000;32:31–35. doi: 10.2746/042516400777612080. [DOI] [PubMed] [Google Scholar]
- Horiuchi I., Yamasaki K., Osoegawa M. Acute myelitis after asthma attacks with onset after puberty. J Neurol Neurosurg Psychiatry. 2000;68:665–668. doi: 10.1136/jnnp.68.5.665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell G. L., Marfin A. A., Lanciotti R. S., Gubler D. J. West Nile virus. Lancet Infect Dis. 2002;2:519–529. doi: 10.1016/s1473-3099(02)00368-7. [DOI] [PubMed] [Google Scholar]
- Brannagen T. H., III, Zhou Y. HIV-associated Guillain-Barre syndrome. J Neurol Sci. 2003;208:39–42. doi: 10.1016/s0022-510x(02)00418-5. [DOI] [PubMed] [Google Scholar]


