Abstract
Background
People exposed to secondhand tobacco smoke (SHS) inhale the lung carcinogen NNK which is metabolized to NNAL and its glucuronides. These urinary metabolites, termed total NNAL, can be quantified. A related compound, iso-NNAL, has been proposed as a biomarker for exposure to smoke constituent residues on surfaces (thirdhand tobacco smoke). There is limited information in the literature on levels of total NNAL in children exposed to SHS.
Methods
We recruited 79 parent child dyads from homes where the enrolled parent was a cigarette smoker, and visited their homes. Parents were asked questions, home ambient air quality was evaluated, and children provided urine samples. Urine was analyzed for total NNAL, total cotinine, total nicotine, and iso-NNAL.
Results
Ninety percent of the children had detectable total NNAL in urine; total nicotine and total cotinine were also detected in most samples. There were significant positive relationships between biomarker levels and exposure of children in the home. Levels were highest in homes with no smoking restrictions. African-American children had significantly higher levels than other children. iso-NNAL was not detected in any urine sample.
Conclusions
There was nearly universal exposure of children to the lung carcinogen NNK, due mainly to exposure to SHS from adult smokers in their homes.
Impact
Homes with adult smokers should adopt restrictions to protect their children from exposure to a potent lung carcinogen.
Keywords: NNAL, secondhand tobacco smoke, thirdhand tobacco smoke, children’s exposure
Introduction
Secondhand smoke (SHS) exposure in adulthood is an accepted cause of lung cancer in non-smokers (1,2). The literature is less clear concerning SHS exposure in childhood and the development of lung cancer later in life in non-smokers. The results of studies reviewed by the International Agency for Research on Cancer were somewhat contradictory (1). The U.S. Surgeon General found some increased risk for lung cancer but the results were inconsistent (2). Some studies published since these reviews indicate a positive association. A nested case-control study within the EPIC cohort found an association between lung cancer in adulthood and frequent exposure to SHS in childhood (3). An increased risk for lung cancer among non-smokers exposed to SHS during childhood, particularly among those possessing a haplotype corresponding to a known overactive complement pathway of the innate immune system, was reported in a recent study (4).
Urinary 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol and its glucuronides (total NNAL) has emerged as a very useful biomarker for assessing exposure to SHS in the context of the potential effects of SHS as a lung carcinogen (5–9). NNAL and its glucuronides are metabolites of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK)(Figure 1), a powerful tobacco-specific lung carcinogen in rodents (10). Advantages of total NNAL as a biomarker include its relatively long half-life, reliable quantitation by validated analytical chemistry techniques, tobacco-specificity, and direct relationship to the lung carcinogen NNK. Total NNAL has been quantified in a number of studies dealing with SHS exposure (5–9). The results of these studies demonstrate conclusively that non-smokers exposed to SHS take up and metabolize the lung carcinogen NNK. When NNAL is detected in urine, the source must have been tobacco products because NNK is found only in these products. Levels of total NNAL in the urine of non-smokers exposed to SHS are about 1–5% as great as those in smokers. Collectively, these results provide strong biochemical support for the relationship between SHS exposure and lung cancer, as observed in epidemiologic studies.
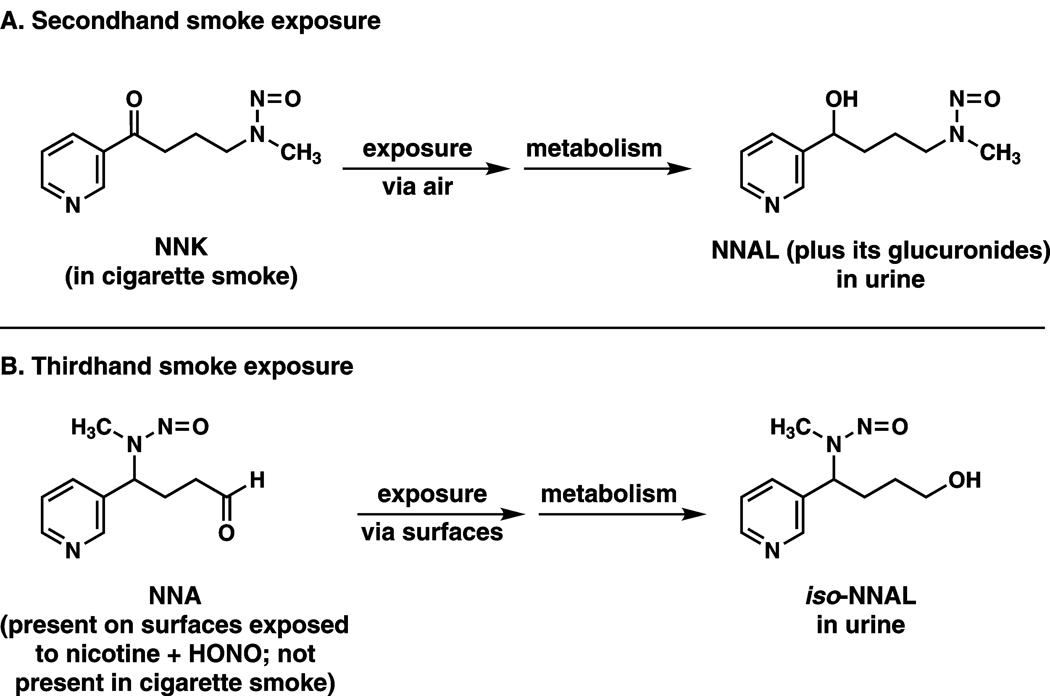
Figure 1.
Summary of existing and proposed tobacco-specific carcinogen biomarkers for secondhand and thirdhand smoke exposure. Total NNAL (NNAL plus its glucuronides) is an established biomarker for uptake of the tobacco-specific lung carcinogen NNK in non-smokers exposed to secondhand smoke (SHS). The NNK comes from the sidestream smoke of a cigarette and the non-smoker is exposed to it by breathing air that contains sidestream smoke. iso-NNAL has been proposed as a biomarker of thirdhand smoke exposure because it is an expected metabolite of NNA, a compound formed on surfaces containing nicotine and exposed to nitrous acid (HONO). NNA is not known to be present in cigarette mainstream or sidestream smoke (11).
Recently, an additional mode of involuntary exposure to tobacco constituents, given the term “thirdhand smoke”, has been proposed (11). Thirdhand smoke exposure would occur by exposure to anything left after a cigarette is extinguished, and would predominantly be due to contact with residues remaining on surfaces. It could be particularly relevant to the exposure of young children because they are routinely in contact with rugs, furniture, and other objects in the household. This concept received substantial publicity when a recent paper reported that carcinogenic tobacco-specific nitrosamines could readily form when nitrous acid was passed over surfaces containing nicotine (12). The authors proposed a specific biomarker for this process: 1-(methylnitrosamino)-1-(3-pyridyl)butan-4-ol (iso-NNAL) which could result from uptake and metabolism of 4-(methylnitrosamino)-4-(3-pyridyl)butanal (NNA), the major tobacco-specific nitrosamine observed on surfaces in their study, but which is not present in tobacco smoke. These relationships are summarized in Figure 1.
There have been only five published studies on total NNAL in the urine of children exposed to SHS, and none have focused specifically on exposure in the home. The first two demonstrated widespread exposure of elementary school children to SHS, but the sources of exposure were not conclusively identified (13,14). Another similar study demonstrated the presence of NNAL and its glucuronides in Moldovan children (15). A fourth study focused on infants less than one year old (7). A recent study documented relatively high levels of exposure of children in the U.S. population (16). In the investigation reported here, we have determined levels of total NNAL, total nicotine, and total cotinine in the urine of children known to be exposed to SHS in their homes. We have also evaluated the concept of thirdhand smoke exposure in these children by investigating the presence of iso-NNAL in their urine.
Materials and methods
Subjects and data collection
This study was approved by the University of Minnesota Research Subjects’ Protection Program Institutional Review Board: Human Subjects Committee. The participating parent-child dyads were part of the “Healthy Homes for Healthy Kids” study, a randomized clinical trial designed to test the efficacy of “biomarker feedback” (i.e., providing parents who smoke with laboratory results documenting their child’s exposure to SHS) on increasing the implementation of complete home smoking restrictions.
Participants were recruited through flyers posted at WIC (Women, Infants, and Children) clinics, serving low income women and young children in the Twin Cities metropolitan area. To be eligible for WIC, applicants must have income at or below a standard set by the state, tied closely to Federal poverty guidelines. All participants were receiving foodstamps. Eligible participants included adults who were the parent or legal guardian of a child ≤ age 10 who lived in their home, smoked at least 100 cigarettes in their lifetime and had smoked at least one cigarette per day on at least 25 of the past 30 days. Participants were excluded if they were currently pregnant, planning to become pregnant or planning to move in the next three months, and/or were receiving treatment for smoking cessation.
A total of 258 people were screened. Of those, 37 were ineligible because they did not have a child less than 10 years of age residing in their home, 18 were not interested in participating, and 123 were ineligible for a variety of other reasons including current pregnancy, plans to move in the next three months, not the legal guardian of the child in their home, smoked less than 25 of the last 30 days, or was a non-smoker. A total of 80 parent-child dyads completed the baseline assessment; however, we were unable to collect a sufficient amount of urine from one child. Therefore, this dyad was excluded from further study. Our final sample size included 79 parent-child dyads.
For the 79 who enrolled in the study, study staff met with each parent/child dyad in their homes to obtain verbal and written consent and assent as appropriate and to administer the baseline questionnaire. Baseline visits occurred between March and August, 2009. For parents who had more than one qualified child living in their home, the youngest, toilet trained child in the home was enrolled.
Enrolled parents were asked a series of questions about their smoking and other smoking-related behaviors, as well as questions about socioeconomic status, residential characteristics, and health. For example, they were asked “On average, how many hours a day do you spend with (child enrolled in study)?”; “To the best of your knowledge has your child been exposed to tobacco smoke in your home in the past week?”; “Not including you, how many people in your home smoke?”; “In the past week, on average, how many cigarettes did you smoke in your home (in your car), in a day?”; “How soon after waking do you smoke your first cigarette?”(proxy measure of nicotine dependence); “Which of the following statements best describes the rules about smoking in your home (in your car)?” Response options included, a) No one is allowed to smoke anywhere, ever; b) Smoking is allowed in some places or at some times; c) Smoking is permitted anywhere, there are no rules. Given the potential for social desirability bias and differential levels of enforcement of smoking restrictions, parents who endorsed complete home smoking bans were eligible to participate in this trial.
A TSI SidePak AM510 Personal Aerosol Monitor (TSI, Inc., St. Paul, MN) was used to monitor home air quality by recording the level of fine particulate matter (PM 2.5) in the home environment. During the baseline visit, a monitor was set up in the main room of the participant’s home, which was most frequently the living room. The monitor was operated for a minimum of 30 min and the mean score was recorded. The SidePak uses a built-in sampling pump to draw air through the device where the particulate matter in the air scatters light from a laser to assess real-time concentration of small particulate matter (<2.5 µm), expressed in mg/m3. Passive air nicotine dosimeters were also used to assess SHS exposure in the home. The dosimeter consists of a filter treated with sodium bisulfate. The filter is contained in a 4-cm polystyrene cassette. Nicotine passively diffuses to the dosimeter and binds to the filter. The dosimeter is housed in a plastic compartment on a HEPA carbon-permanganate-zeolite air cleaner. At the baseline visit, the dosimeter was hung above reach in an unobtrusive location, away from sources of air circulation, in the room in which the child reportedly spent the most time. The badge was subsequently retrieved approximately 3 weeks after placement. Badges were analyzed by gas chromatography in the laboratory of S. Katharine Hammond (University of California, Berkeley) using a standardized procedure (17).
A Micro Smokerlyzer (Bedfont Scientific, Ltd) was used to record the baseline carbon monoxide level of the enrolled parent. Toilet trained children were asked to provide a 40 ml urine sample in a sterile commode specimen collection hat. Families with eligible children who were not toilet trained were mailed two NatraCare 100% cotton natural pads (NatraCare Products) and vinyl medical examination gloves prior to the baseline visit. Parents were provided with written and verbal instructions to place a pad in the child’s diaper the night before the baseline home visit and another on the morning of the visit. They were instructed to place the soaked pad(s) in an accompanying biohazard bag. Parents were strongly encouraged to wear the gloves provided when placing and removing the pad to avoid the possibility of third-hand nicotine contamination from their hands. At the baseline visit, either the urine sample or the soaked pad(s) were collected and returned to the laboratory at the Masonic Cancer Center.
Biomarker analyses
Total NNAL, total cotinine, and total nicotine were quantified essentially as described (7). In each case, the urine was treated with β-glucuronidase and the reported amount represents the total of the unconjugated compound plus its glucuronide. Limits of quantitation for total NNAL, total cotinine and total nicotine were approximately 0.01 pmol/ml, 0.5 ng/ml, and 0.5 ng/ml, respectively. Data on precision (CV = <10%) and accuracy of these assays have been described (18,19). Analysis for iso-NNAL was carried out using the same method as employed for total NNAL, except that its gas chromatographic retention time was different. Ten urine samples were also analyzed for iso-NNAL by liquid chromatography-electrospray ionization-tandem mass spectrometry (LC-ESI-MS/MS) using a Waters Associates nanoAcquity Ultra Performance instrument fitted with a 100 mm × 0.5 mm, 3µ Hypercarb column (Thermo Scientific) eluted with 40% MeOH in H2O for 30 min at 10 µl/min and interfaced with a Thermo Scientific TSQ Quantum Ultra instrument. The transitions m/z 210 → m/z 180 and m/z 210 → m/z 149 were monitored.
Hepatocyte Incubations
In order to investigate the metabolism of NNA to iso-NNAL, primary human hepatocytes were purchased from Cellzdirect (St. Louis, MO), and prepared as described (20). Hepatocytes (approximately 0.12 mg protein per well) were incubated with 10 µM NNA or NNK, as a positive control. The nitrosamines were dissolved in 20 µl H2O and added to the 2 ml incubation mixtures. One ml of media was collected at 24 h. Samples were stored at −20 °C until analysis.
Statistical analysis and related considerations
Descriptive statistics of child and parent demographics, parent smoking characteristics and child exposure variables were calculated for the study sample. Categorical variables were summarized by frequencies and percentages, and continuous variables were summarized by means or geometric means. The Wilcoxon rank sum tests or Kruskal-Wallis tests were used to compare the geometric means of total NNAL, total cotinine, total nicotine, and sum of total cotinine and total nicotine by the categorical demographics, smoking characteristics and exposure variables. Spearman correlations were calculated to assess association among the biomarkers and continuous demographics, smoking characteristics and exposure variables. Samples below the detection limit were assigned a value of 0.0035 pmol/ml for total NNAL and 0.1 ng/ml for total cotinine and total nicotine.
Results
Parent-child demographic characteristics and parents’ smoking characteristics of the 79 parent-child dyads are summarized in Table 1. Children ranged in age from one month to 10 years (mean ± S.D.: 3.82 ± 2.55); 49% were male. Approximately 56% of parents were African American and 20% were White. Twenty-seven percent of parents had less than a high school education, 67% were unemployed and 76% reported a monthly income of <$1800. The average number of cigarettes smoked per day was 9.5 ± 5.3; 64% reported smoking their first cigarette within 30 min of waking and the average number of smokers living in the home was 1.9 ± 0.96. Seventy-two percent of parents reported that their child had been exposed to SHS in the past week and 70.9% reported having at least some home smoking restrictions in their home.
Table 1.
Demographic and smoking characteristics of parent-child dyads (N = 79)
| Characteristic | N (%) | Mean ± S.D. or Geometric mean (95% CI) |
|---|---|---|
| Child age (years) | 3.82 ± 2.55 | |
| Child Gender | ||
| Female | 40 (50.6) | |
| Male | 39 (49.4) | |
| Parent Race | ||
| African American | 44 (55.7) | |
| White | 16 (20.3) | |
| Other | 19 (24.0) | |
| Income, | ||
| <$1800/month | 60 (76.0) | |
| >=$1800/month | 19 (24.0) | |
| Education | ||
| <12 grades | 21 (26.6) | |
| >=12 grades | 58 (73.4) | |
| Employment Status | ||
| Not employed | 53 (67.2) | |
| Full or part time employed | 26 (32.9) | |
| Housing Type | ||
| Townhouse or duplex | 47 (59.5) | |
| Single family house | 32 (40.5) | |
| Number of children under 18 in home | 2.44 ± 1.63 | |
| Min to first cigarette | ||
| 0–30 | 51 (64.6) | |
| 31+ | 28 (35.4) | |
| Parent CO (ppm) | 10.2 (8.7–11.9) | |
| PM2.5, home air quality (mg/m3) | 51.4 (39.5–66.9) | |
| Cigarettes per day (last 7 days) | 9.5 ± 5.3 | |
| Number of smokers living in home | 1.9 ± 0.96 | |
| Number of cigarettes smoked in home per day | 6.9 ± 7.2 | |
| Number of cigarettes smoked in car per day | 4.3 ± 3.4 | |
| Time spent per day with child (h) | 15.6 ± 7.5 | |
| Child exposed to SHS in home past week | 57 (72.3) | |
| Child exposed to SHS outside home past week | 38 (48.7) | |
| Home smoking restrictions | ||
| Complete restrictions | 17 (21.5) | |
| Some restrictions | 39 (49.4) | |
| No restrictions | 23 (29.1) | |
| Car smoking restrictions | ||
| Complete restrictions | 10 (12.7) | |
| Some restrictions | 15 (19.0) | |
| No restrictions | 13 (16.5) | |
| Do not own a car | 41 (51.9) |
Levels of total NNAL, total cotinine, total nicotine, and total cotinine plus total nicotine are summarized in Table 2. Geometric mean levels of total NNAL for the entire sample were 0.08 pmol/ml (95% CI 0.06 – 0.10), whereas those of total cotinine, total nicotine, and total cotinine plus total nicotine were 11.88 ng/ml (95% CI 8.42 – 16.77), 6.90 ng/ml (95% CI 4.57 – 10.43), and 0.13 nmol/ml (95% CI 0.09 – 0.18), respectively. Ninety percent of the children had detectable NNAL in their urine; the corresponding figures for detectable total cotinine and total nicotine were 95% and 90%, respectively. The distributions of total NNAL, total cotinine, and total nicotine values are illustrated in Figure 2A–C.
Table 2.
Geometric mean (95% C.I.) children’s urine total NNAL, total cotinine, total nicotine and total nicotine + total cotinine levels by demographic and smoking characteristics.
| Total NNAL (pmol/ml) |
Total cotinine (ng/ml) |
Total nicotine (ng/ml) |
Total nicotine + total cotinine (nmol/ml) |
|||||
|---|---|---|---|---|---|---|---|---|
| All children | 0.08 (0.06 – 0.10) | 11.88 (8.42–16.77) | 6.90 (4.57–10.43) | 0.13 (0.09–0.18) | ||||
| Race | † | † | † | † | ||||
| African American | 0.11 (0.08–0.17) | 20.01 (13.82–28.95) | 12.01 (7.59–18.99) | 0.21 (0.15–0.31) | ||||
| White | 0.03 (0.02–0.06) | 4.06 (1.51–10.89) | 1.70 (.47–6.16) | 0.04 (0.02–0.11) | ||||
| Others | 0.07 (0.03–0.12) | 8.78 (4.37–17.64) | 6.21 (3.18–12.16) | 0.10 (0.04–0.19) | ||||
| Income | ns | * | † | † | ||||
| <$1800/month | 0.09 (0.07–0.13) | 45.36 (10.49–22.48) | 8.87 (5.69–13.84) | 0.16 (0.11–0.24) | ||||
| >=$1800/month | 0.04 (0.02–0.09) | 5.28 (2.58–10.82) | 3.12 (1.17–8.31) | 0.06 (0.03–0.12) | ||||
| Education | ns | ns | ns | ns | ||||
| <12 years | 0.11 (0.07–0.18) | 17.44 (10.45–30.27) | 10.74 (5.52–20.91) | 0.19 (0.11–0.32) | ||||
| ≥12 years | 0.07 (0.05–0.10) | 10.34 (6.74–15.87) | 5.88 (3.52–9.87) | 0.11 (0.07–0.17) | ||||
| Employment Status | * | * | * | * | ||||
| Not employed | 0.10 (0.07–0.14) | 15.90 (11.10–22.77) | 9.61 (6.41–13.39) | 0.17 (0.12–0.24) | ||||
| Full or part time | 0.05 (0.03–0.09) | 6.56 (3.14–13.69) | 3.52 (1.37–8.99) | 0.07 (0.03–0.15) | ||||
| Housing Type | ns | ns | ns | ns | ||||
| Multi-unit dwelling | 0.09 (0.06–0.13) | 11.64 (7.37–18.40) | 7.51 (4.23–13.33) | 0.13 (0.08–0.21) | ||||
| Single family house | 0.07 (0.04–0.11) | 12.24 (7.04–21.28) | 6.09 (3.32–11.20) | 0.12 (0.07–0.21) | ||||
| Child Gender | ns | ns | ns | ns | ||||
| Female | 0.07(0.04–0.11) | 12.46 (7.99–19.43) | 8.45 (5.34–13.37) | 0.14 (0.09–0.21) | ||||
| Male | 0.09 (0.06–0.13) | 11.31 (6.53–19.60) | 5.61 (2.76–11.38) | 0.12 (0.07–0.20) | ||||
| Time to first cigarette | † | † | § | § | ||||
| 0–30 min | 0.11 (0.09–0.15) | 18.21 (13.36–24.82) | 11.99 (8.03–17.92) | 0.20 (0.15–0.27) | ||||
| 31+ min | 0.04 (0.02–0.07) | 5.45 (2.61–11.43) | 2.52 (1.12–5.69) | 0.05 (0.03–0.11) | ||||
| Child exposed in home | § | § | § | § | ||||
| Yes | 0.11 (0.09–0.15) | 19.53 (14.78–25.81) | 10.99 (7.46–16.22 | 0.20(0.16–0.27) | ||||
| No | 0.03 (0.01–0.06) | 3.28(1.44–7.47) | 2.06 (0.78–5.44) | 0.04(0.02–0.09) | ||||
| Child exposed outside home | ns | ns | ns | ns | ||||
| Yes | 0.08 (0.05–0.13) | 10.42 (5.83–18.63) | 7.14 (3.84–13.28) | 0.11(0.06––0.20) | ||||
| No | 0.07(0.05–0.11) | 13.43(8.81–20.48) | 6.49 (3.61–11.68) | 0.14 (0.09–0.21) | ||||
| Home smoking restrictions | § | § | § | § | ||||
| Complete restrictions | 0.02 (0.0085–0.040) | 2.08 (.86–5.03) | 1.70 (.57–5.11) | 0.03 (0.01–0.07) | ||||
| Some/no restrictions | 0.11 (0.087–0.15) | 19.16 (14.54–25.24) | 10.13 (6.81–15.07) | 0.20 (0.15–0.26) | ||||
| Car Smoking Restrictions | ns | ns | ns | ns | ||||
| Complete restrictions | 0.07 (0.02–0.23) | 13.26 (4.05–43.37) | 8.05 (1.84–35.27) | 0.15 (0.05–0.47) | ||||
| Some/ no restrictions | 0.05 (0.03–0.08) | 5.78 (2.73–12.21) | 3.20 (1.45–7.02) | 0.06 (0.03–0.12) | ||||
p<0.05
p<0.01
p<0.001
not significant
Figure 2.
Frequency distributions of levels of urinary A) total NNAL, B) total cotinine, and C) total nicotine among the children in this study.
There were significant positive relationships between biomarker levels and reported levels of child SHS exposure in the home, with exposed children having higher levels (Tables 2 and 3). All biomarker levels were significantly higher in homes endorsing no restrictions (e.g., total NNAL 0.13 [0.09 – 0.19]) or among those reporting some smoking restriction (0.10 [.07 – 0.15]) versus those with complete restrictions against smoking (0.02 [0.01 – 0.04]) (p <0.001). There were also significant correlations between total NNAL and parents’ CO levels (r = 0.29, p <0.05); home air quality as measured by the level of fine particulate matter (PM 2.5) (r = 0.28, p < 0.05); number of smokers residing in the household (r = 0.23, p <0.01); and cigarettes per day smoked in the home (r = 0.30, p <0.01). Similar trends were observed for total cotinine, total nicotine, and total cotinine plus total nicotine. Exposure outside of the home was not related to biomarker levels. Exposures in cars were inconsistent in their relationship with biomarker levels and less than 50% of our subjects owned a car.
Table 3.
Spearman correlation coefficients between adult demographic and tobacco-related characteristics and total NNAL, total cotinine, total nicotine and total nicotine plus total cotinine levels in children’s urine.
| Total NNAL |
Total cotinine | Total nicotine | Total cotinine plus total nicotine |
|
|---|---|---|---|---|
| Children’s age | −0.1 | −0.16 | −0.27* | −0.22* |
| Number of children under 18 in household | 0.04 | 0.04 | 0.03 | 0.02 |
| Smokers’ CO | 0.29* | 0.31† | 0.23* | 0.31† |
| Home air quality | 0.28* | 0.33† | 0.27* | 0.32† |
| Nicotine dosimeter badge | 0.39§ | 0.44§ | 0.39§ | 0.44§ |
| Smokers’ cigarettes per day | −0.13 | −0.0008 | −0.14 | −0.06 |
| Number of smokers in household | 0.23* | 0.16 | 0.06 | 0.12 |
| Cigarettes per day smoked in home | 0.30† | 0.37§ | 0.27* | 0.33† |
| Cigarettes per day smoked in car | −0.33 | −0.15 | −0.09 | −0.12 |
| Hours spent with child per day | 0.15 | 0.19 | 0.13 | 0.17 |
| Total NNAL | 1.00 | 0.81§ | 0.64§ | 0.77§ |
| Total cotinine | 0.81§ | 1.00 | 0.80§ | 0.96§ |
| Total nicotine | 0.64§ | 0.80§ | 1.00 | 0.93§ |
| Total nicotine plus total cotinine | 0.77§ | 0.96§ | 0.93§ | 1.00 |
p<0.05
p<0.01
p<0.001
All biomarker levels were significantly different across ethnic groups with African-American children having higher levels than both White and “Other” children (p<0.01). Nicotine dosimeter values were significantly higher in African-American homes [0.48 µg/m3 (95% CI 0.29–0.79)] than in non-African-American homes [0.15 µg/m3 (0.074 – 0.30)], p<0.0051. When African-American homes were compared to "Others", more African-American parents endorsed child exposure to cigarette smoke in the past week (82% vs. 60%, p = 0.03); and fewer restrictions regarding home smoking (7% vs. 40%, p = 0.001) or rules against smoking in the car (9% vs. 17%, p = 0.03). No other significant demographic or tobacco related differences between African-American and other children were found.
The biomarker levels were also significantly different by parental employment status such that children of unemployed parents had higher amounts than those of employed parents (p<0.05). The association between total NNAL levels and monthly income approached significance, with lower income (< $1800 monthly) associated with higher total NNAL levels when compared to those with higher income (>$1800 month) (p = 0.052), while levels of the other biomarkers were significantly different between these groups (p < 0.05).
Total NNAL was significantly different by nicotine dependence level such that children of parents who smoked their first cigarette of the day within 30 min of waking had higher levels of total NNAL in their urine when compared to children of parents who smoked their first cigarette later (0.11, 95% CI 0.09 – 0.15 vs. 0.04, 95% CI 0.02 – 0.07, p < 0.01). Similar results were observed for levels of total cotinine, total nicotine, and their sum.
The associations among the biomarkers are also summarized in Table 3. There were strong and significant correlations among all biomarkers (0.64 – 0.96, p<0.001). .
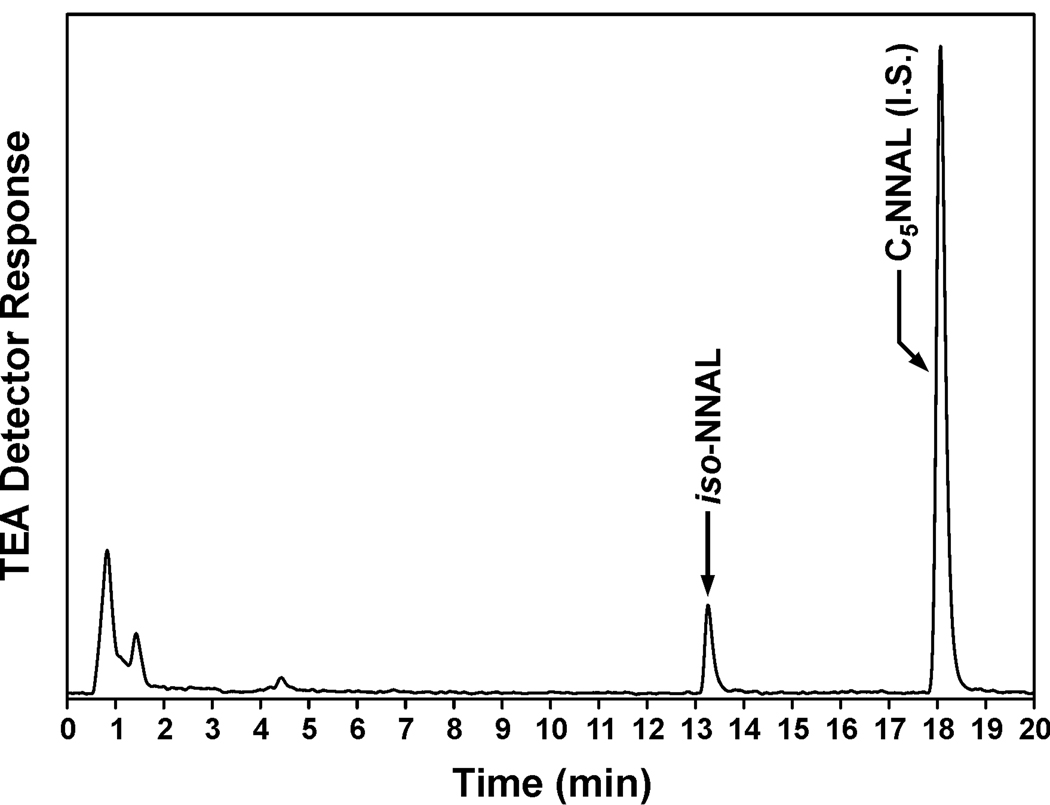
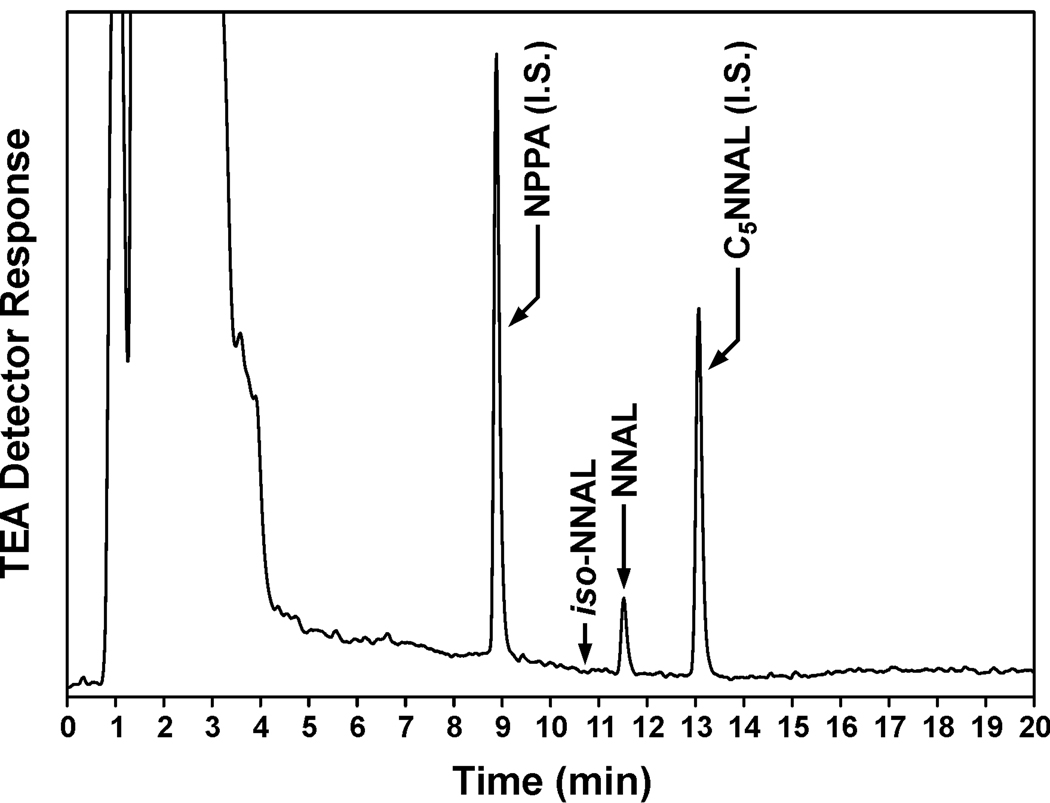
We investigated the possible exposure of the children in this study to thirdhand smoke, using the proposed iso-NNAL biomarker. Although iso-NNAL is a suggested metabolite of NNA (Figure 1), this has not been documented previously in the literature. We incubated human hepatocytes with 10 µM NNA, and detected iso-NNAL as a metabolite (0.57 ± 0.079 µM, N = 4) (Figure 3). This established that iso-NNAL could be formed metabolically from NNA in humans and is therefore a potential biomarker for NNA exposure, as proposed. The amount formed was about 9 % as great as the amount of NNAL formed from NNK in human hepatocytes cultured under the same conditions. Using gas chromatography – nitrosamine selective detection (the same method used for the NNAL analyses), we did not detect iso-NNAL in any of the urine samples from the children in this study (detection limit, 0.008 pmol/ml). A representative chromatogram is shown in Figure 4. We also carried out the analysis for iso-NNAL in 10 of these urine samples by LC-ESI-MS/MS. It was not detected in any sample (detection limit, 0.002 pmol/ml).
Figure 3.
Chromatogram obtained upon analysis by gas chromatography-nitrosamine selective detection of the medium from human hepatocytes which had been incubated with NNA. The indicated peak is silylated iso-NNAL. The internal standard (I.S.) for the analysis was 5-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (C5NNAL)
Figure 4.
Typical chromatogram obtained upon analysis by gas chromatography-nitrosamine selective detection of urine from a child in this study. Arrow: retention time of silylated iso-NNAL. Peaks with arrows correspond to NNAL and two internal standards (I.S.) used in the analysis, C5NNAL and N-nitrosopentyl-3-picolylamine (NPPA). Gas chromatography conditions are slightly different from those used for Figure 3.
Discussion
This is the first study to examine levels of total NNAL in the urine of children with documented exposure to cigarette smoke mainly from adult smokers in their homes. Ninety percent of the children had detectable total NNAL in their urine, demonstrating that exposure to the potent lung carcinogen NNK is virtually universal in these homes. Levels of total NNAL were significantly lower in homes which had smoking restrictions, and there was a correlation between cigarettes smoked per day in the home and total NNAL levels in childrens’ urine. Levels of total NNAL in the urine of children living in homes with only some or no restrictions (geometric mean, 0.113 pmol/ml) are the highest reported to date due to SHS exposure (7). These results clearly demonstrate that adult smoking in the home is an important source of NNK exposure in children. Similar results were observed for urinary total nicotine and total cotinine in these children.
The overall mean level of total NNAL found in the urine of the children in this study (0.08 pmol/ml) was similar to those reported in previous studies of children, as were the levels of total cotinine and total nicotine. In a study of elementary school children in Minneapolis, the mean level of total NNAL was 0.056 pmol/ml (13). Children in Moldova had a mean total NNAL level of 0.09 pmol/ml (15). Infants less than one year old exposed to their parents’ smoking had a mean total NNAL level of 0.083 pmol/ml (7). In previous reports, the levels of total NNAL in the urine of children exposed to SHS have generally been higher than those in adults, and this study provides further substantiation of this observation (7). Our results indicate that the higher levels of total NNAL found in the urine of children result mainly from smoking by others in their homes.
We observed significantly higher levels of total NNAL, total nicotine, and total cotinine in the urine of African-American children compared to children of other ethnicities. This apparently relates to exposure factors, since additional analyses demonstrated that African-American homes had significantly less smoking restrictions than other homes and the children were exposed significantly more frequently to SHS. Furthermore, nicotine dosimeter results demonstrated significantly higher levels in African-American homes than in others. Ethnic differences in nicotine metabolism leading to higher cotinine levels in African-Americans could contribute to these results (21), but are unlikely to contribute to the higher levels of nicotine and total NNAL which we observed. Our results are consistent with previous reports from the NHANES study that non-Hispanic black children have higher serum cotinine than non-Hispanic white children (2). African-American adults have a higher risk for lung cancer than do Caucasian adults who smoke the same number of cigarettes (22). One can speculate that exposure during childhood may contribute to this difference, perhaps by providing initiating events in cells that later are exposed to tumor promoters and other toxicants in cigarette smoke, completing the progression to lung cancer. The initiating events could be higher in African-American children, based on our results.
Thirdhand smoke has been proposed as a potentially significant route of exposure to tobacco-specific nitrosamines (12). This could be highly relevant to children who would likely have contact with residues in the household. We found no evidence for thirdhand smoke exposure in this study. We demonstrated that iso-NNAL is a metabolite of NNA in human hepatocytes, consistent with the proposal of Sleiman et al that it could be a useful biomarker for thirdhand smoke exposure (12), but iso-NNAL was not detected in the urine of the children in this study. This conclusion could be limited by the fact that iso-NNAL appears to be less readily formed metabolically from NNA, than is NNAL from NNK, at least in human hepatocytes. The lower extent of formation could limit our ability to detect it in urine.
Exposure in this study, as measured by urinary total NNAL, total cotinine, and total nicotine was clearly related to typical variables associated with cigarette smoking such as time to first cigarette in the morning (a measure of nicotine dependence), parent CO level, home air quality, number of smokers residing in the household and cigarettes per day smoked in the home. Based on these results, there is little doubt that total NNAL levels in the urine of children could be substantially reduced by home smoking restrictions, as our data also indicate.
In summary, this study demonstrates substantial and widespread exposure to the lung carcinogen NNK, as well as to nicotine, in children who live in homes where smoking is allowed. Exposure apparently occurs via secondhand smoke, not thirdhand smoke. The results clearly indicate that home smoking restrictions could reduce childrens’ exposure to a powerful lung carcinogen as well as to nicotine, an addictive compound.
Acknowledgments
Grant support: This study was supported by grant CA-81301 from the National Cancer Institute and American Lung Association award SB-82592-N. Janet L. Thomas was supported in part by grant 1P60MD003422 from the National Institute of Minority Health and Health Disparities and the American Lung Association.
References
- 1.International Agency for Research on Cancer. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. vol. 83. Lyon, FR: IARC; 2004. Tobacco Smoke and Involuntary Smoking; pp. 1191–1413. [PMC free article] [PubMed] [Google Scholar]
- 2.U.S.Department of Health and Human Services. Washington, DC: U.S.: Dept. of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2006. The Health Consequences of Involuntary Exposure to Tobacco Smoke: A Report of the Surgeon General. [PubMed] [Google Scholar]
- 3.Vineis P, Airoldi L, Veglia P, et al. Environmental tobacco smoke and risk of respiratory cancer and chronic obstructive pulmonary disease in former smokers and never smokers in the EPIC prospective study. BMJ. 2005;330:277. doi: 10.1136/bmj.38327.648472.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Olivo-Marston SE, Yang P, Mechanic LE, et al. Childhood exposure to secondhand smoke and functional mannose binding lectin polymorphisms are associated with increased lung cancer risk. Cancer Epidemiol Biomarkers Prev. 2009;18:3375–3383. doi: 10.1158/1055-9965.EPI-09-0986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hecht SS. Human urinary carcinogen metabolites: biomarkers for investigating tobacco and cancer. Carcinogenesis. 2002;23:907–922. doi: 10.1093/carcin/23.6.907. [DOI] [PubMed] [Google Scholar]
- 6.Hecht SS, Carmella SG, Murphy SE, Akerkar S, Brunnemann KD, Hoffmann D. A tobacco-specific lung carcinogen in the urine of men exposed to cigarette smoke. N Engl J Med. 1993;329:1543–1546. doi: 10.1056/NEJM199311183292105. [DOI] [PubMed] [Google Scholar]
- 7.Hecht SS, Carmella SG, Le K, et al. 4-(Methylnitrosamino)-1-(3-pyridyl)-1-butanol and its glucuronides in the urine of infants exposed to environmental tobacco smoke. Cancer Epidemiol Biomarkers & Prev. 2006;15:988–992. doi: 10.1158/1055-9965.EPI-05-0596. [DOI] [PubMed] [Google Scholar]
- 8.Jensen JA, Schillo BA, Moilanen MM, et al. Tobacco smoke exposure in nonsmoking hospitality workers before and after a state smoking ban. Cancer Epidemiol Biomarkers Prev. 2010;19:1016–1021. doi: 10.1158/1055-9965.EPI-09-0969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Eisner MD, Jacob P, III, Benowitz NL, Balmes J, Blanc PD. Longer term exposure to secondhand smoke and health outcomes in COPD: impact of urine 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol. Nicotine Tob Res. 2009;11:945–953. doi: 10.1093/ntr/ntp091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hecht SS. Biochemistry, biology, and carcinogenicity of tobacco-specific N-nitrosamines. Chem Res Toxicol. 1998;11:559–603. doi: 10.1021/tx980005y. [DOI] [PubMed] [Google Scholar]
- 11.Winickoff JP, Friebely J, Tanski SE, et al. Beliefs about the health effects of "thirdhand" smoke and home smoking bans. Pediatrics. 2009;123:e74–e79. doi: 10.1542/peds.2008-2184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sleiman M, Gundel LA, Pankow JF, Jacob P, III, Singer BC, Destaillats H. Formation of carcinogens indoors by surface-mediated reactions of nicotine with nitrous acid, leading to potential thirdhand smoke hazards. Proc Natl Acad Sci U S A. 2010;107:6576–6581. doi: 10.1073/pnas.0912820107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hecht SS, Ye M, Carmella SG, et al. Metabolites of a tobacco-specific lung carcinogen in the urine of elementary school-aged children. Cancer Epidemiol Biomarkers & Prev. 2001;10:1109–1116. [PubMed] [Google Scholar]
- 14.Sexton K, Adgate JL, Church TR, et al. Children's exposure to environmental tobacco smoke: using diverse exposure metrics to document ethnic/racial differences. Environ Health Perspect. 2004;112:392–397. doi: 10.1289/ehp.6473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Stepanov I, Hecht SS, Duca G, Mardari I. Uptake of the tobacco-specific lung carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) by Moldovan children. Cancer Epidemiol Biomarkers & Prev. 2006;15:7–11. doi: 10.1158/1055-9965.EPI-05-0293. [DOI] [PubMed] [Google Scholar]
- 16.Bernert JT, Pirkle JL, Xia Y, Jain RB, Ashley DL, Sampson EJ. Urine concentrations of a tobacco-specific nitrosamine carcinogen in the U.S. population from secondhand smoke exposure. Cancer Epidemiol Biomarkers Prev. 2010;19:2969–2977. doi: 10.1158/1055-9965.EPI-10-0711. [DOI] [PubMed] [Google Scholar]
- 17.Eisner MD, Katz PP, Yelin EH, Hammond SK, Blanc PD. Measurement of environmental tobacco smoke exposure among adults with asthma. Environ Health Perspect. 2001;109:809–814. doi: 10.1289/ehp.01109809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jacob P, III, Yu L, Wilson M, Benowitz NL. Selected ion monitoring method for determination of nicotine, cotinine and deuterium-labeled analogs. Absence of an isotope effect in the clearance of (S)-nicotine-3',3'-d2 in humans. Biol Mass Spectrom. 1991;20:247–252. doi: 10.1002/bms.1200200503. [DOI] [PubMed] [Google Scholar]
- 19.Carmella SG, Han S, Fristad A, Yang Y, Hecht SS. Analysis of total 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL) in human urine. Cancer Epidemiol Biomarkers & Prev. 2003;12:1257–1261. [PubMed] [Google Scholar]
- 20.Upadhyaya P, Hochalter JB, Balbo S, McIntee EJ, Hecht SS. Preferential glutathione conjugation of a reverse diol epoxide compared with a bay region diol epoxide of benzo[a]pyrene in human hepatocytes. Drug Metab Dispos. 2010;38:1397–1402. doi: 10.1124/dmd.110.034181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Berg JZ, Mason J, Boettcher AJ, Hatsukami DK, Murphy SE. Nicotine metabolism in African Americans and European Americans: variation in glucuronidation by ethnicity and UGT2B10 haplotype. J Pharmacol Exp Ther. 2010;332:202–209. doi: 10.1124/jpet.109.159855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Haiman CA, Stram DO, Wilkens LR, et al. Ethnic and racial differences in the smoking-related risk of lung cancer. N Engl J Med. 2006;354:333–342. doi: 10.1056/NEJMoa033250. [DOI] [PubMed] [Google Scholar]