Abstract
In addition to neutralization, antibodies mediate other antiviral activities including antibody-dependent cellular-phagocytosis (ADCP), antibody dependent cellular-cytotoxicity (ADCC), as well as complement deposition. While it is established that progressive HIV infection is associated with reduced ADCC and ADCP, the underlying mechanism for this loss of function is unknown. Here we report considerable changes in FcR expression over the course of HIV infection on both mDCs and monocytes, including elevated FcγRI expression in acute HIV infection and reduced expression of FcγRII and FcγRIIIa in chronic HIV infection. Furthermore, selective blockade of FcγRII alone was associated with a loss in ADCP activity, suggesting that FcγRII plays a central role in modulating ADCP. Overall, HIV infection is associated with a number of changes in FcR expression on phagocytic cells that are associated with changes in their ability to respond to antibody-opsonized targets, potentially contributing to a failure in viral clearance in progressive HIV-1 infection.
Keywords: Innate Immunity, Fc-receptors, Antibody dependent cellular phagocytosis, HIV-1
Introduction
In addition to their ability to neutralize pathogens, antibodies are able to mediate a number of additional antiviral functions following ligation of innate immune cells via an Fc receptor (fragment, crystallizable receptor, FcR) (Nimmerjahn and Ravetch, 2008; Oganesyan et al., 2008). These include stimulation of cytokine production, induction of cytolytic activity, antibody-mediated cellular-phagocytosis (ADCP), antibody dependent cellular cytotoxicity (ADCC), or complement mediated cytotoxicity (CMC).
FcRs belong to the immunoglobulin superfamily and bind to the constant regions of antibodies (Nimmerjahn and Ravetch, 2007). Five different FcRs, with distinct antibody isotype binding properties, exist in humans including FcαR (IgA), FcγR (IgG), FcμR (IgM), FcδR (IgD), and FcεR (IgE). Among the IgG binding FcRs, 3 isoforms of FcγR with distinct functions have been identified: the FcγRI (high affinity), FcγRII (a and c, medium-low affinity), and FcγRIII (a and b, medium-low affinity) are activating receptors, whereas FcγRIIb delivers inhibitory signals (Siberil et al., 2007). All cells of the innate immune system, and a few non-innate immune cells, express FcRs including monocytes/macrophages, granulocytes, DC, and mast cells that express both inhibitory and activating FcγR, natural killer (NK) cells that express the activating receptor FcγRIIIa, and B cells that only express the inhibitory receptor FcγRIIb (Anderson, 1989; Nimmerjahn and Ravetch, 2008).
Different combinations of FcRs on innate immune cells allow antibodies to induce a broad range of innate immune responses depending on the innate immune cell engaged, that may play a vital role in anti-viral or anti-tumor immune responses. Importantly, ADCC and ADCP activity have been associated with better outcome in several diseases such as chronic viral infections (Shore et al., 1974), autoimmune diseases (Laszlo et al., 1986) and particular cancers (Dall’Ozzo et al., 2004; Natsume et al., 2009). In addition to NK cells that mediate potent ADCC via FcγRIIIa, other innate immune cells, such as monocytes, DCs, and neutrophils are also able to mediate ADCC as well as ADCP, and have equally been implicated as critical responders to antibody-opsonized material. Furthermore, unlike neutralizing antibodies, ADCC-inducing antibodies have been observed early in HIV infection, and correlate with better disease outcome (Aasa-Chapman et al., 2005; Forthal et al., 2001). Furthermore, results from the RV144 Thai trial, suggest that additional antiviral humoral mechanisms, beyond neutralization, may play a critical role not only after infection but may also protect individuals from infection (Rerks-Ngarm et al., 2009).
While a great deal of work has focused on the role of ADCC activity in the control of HIV infection, little has been done to understand the role of other antibody-mediated effector functions, such as ADCP, which may also play a vital role in the effective clearance of immune complexes that are abundantly produced during progressive HIV infection (Tomaras et al., 2008). Thus robust ADCP inducing antibodies could play a critical role in the rapid clearance of antibody-opsonized viral particles from the peripheral circulation where they may contribute to immune activation and disease progression (Tomaras et al., 2008).
Given the central role of FcRs in regulating the recruitment of innate immune responses, including ADCP, we speculated that changes in FcR expression may provide clues regarding the FcRs that are involved in viral clearance at different stages of HIV infection. Here we report that acute infection was associated with an increased expression of FcγRI, whereas chronic infection was associated with a decreased FcγRII and FcγRIII expression on a variety of myeloid cell subsets (mDC, pDC and monocytes). Phagocytic activity was significantly elevated in acute HIV infection while it was reduced in chronic infection suggesting that changes in particular FcRs are associated with defective ADCP-inducing antibody function at different stages of HIV infection that may contribute to a differential capacity to clear immune complexes during HIV infection.
Results
Dramatic changes in the frequency and magnitude of FcR expression starting in acute HIV-1 infection
Chronic HIV infection is associated with an accumulation of dysfunctional NK cells (Alter et al., 2005; Alter et al., 2004; Eger and Unutmaz, 2004; Hu et al., 1995; Mavilio et al., 2003), and more recent work has shown that impaired NK-mediated ADCC activity is related to reduced expression of FcγRIIIa on the surface of these cytolytic effector cells (Liu et al., 2009). Given the profound impact of reduced FcγR expression on NK cell mediated ADCC, it is plausible that changes in FcR expression on other cell subsets over the course of HIV infection may also modulate their capacity to respond to cytolytic antibodies. Thus we compared the frequency and magnitude of the expression of the 3 main Fcγ-receptors (FcγRI, FcγRII, FcγRIIIa) as well as FcεR and FcαR at different stages of HIV infection on several innate immune cell subsets, starting in acute HIV infection.
FcγRI is the sole high affinity receptor for monomeric IgG that is constitutively expressed on monocytes, macrophages and neutrophils (Hulett and Hogarth, 1994; Ravetch and Kinet, 1991), and has been shown to play a central role in the induction of ADCC and ADCP (Diamond et al., 1978). HIV infection was associated with significant changes in the frequency of cells expressing FcγRI (FIG 1A) and in the mean fluorescence intensity (MFI) of this FcR (FIG 1B) on innate immune cells. In acute infection, we observed an increase in the frequency of FcγRI expressing mDCs (FIG 1Ai) (p<0.001) and in the MFI of FcγRI expression on monocytes (FIG 1Biii) (p<0.001) compared to chronically infected individuals and healthy controls. Similar to previous reports suggesting that pDCs do not express FcγRI (Bave et al., 2003), we did not observe FcγRI expression on pDCs (FIG 1Bii). In summary, we found that only acute HIV infection is associated with a robust increase in the frequency of mDCs expressing FcγRI and an increased expression of FcγRI on monocytes potentially suggesting that these innate immune cells may expand and/or upregulate the expression of this high affinity FcγR preferentially at this early stage of the infection in response to the large burst of viral replication and/or the early cytokine cascade.
FIG. 1. Acute HIV-1 infection is associated with changes in FcγRI expression on mDCs and monocytes.
The dot plots depict changes in the frequency (A) and intensity (B) of FcγRI (CD64) expression on (i) mDCs, (ii) pDCs and (iii) monocytes derived from acute (●), chronic treated (■), chronic untreated (⬪), controllers (⬧) and HIV-1 negative controls (◆).
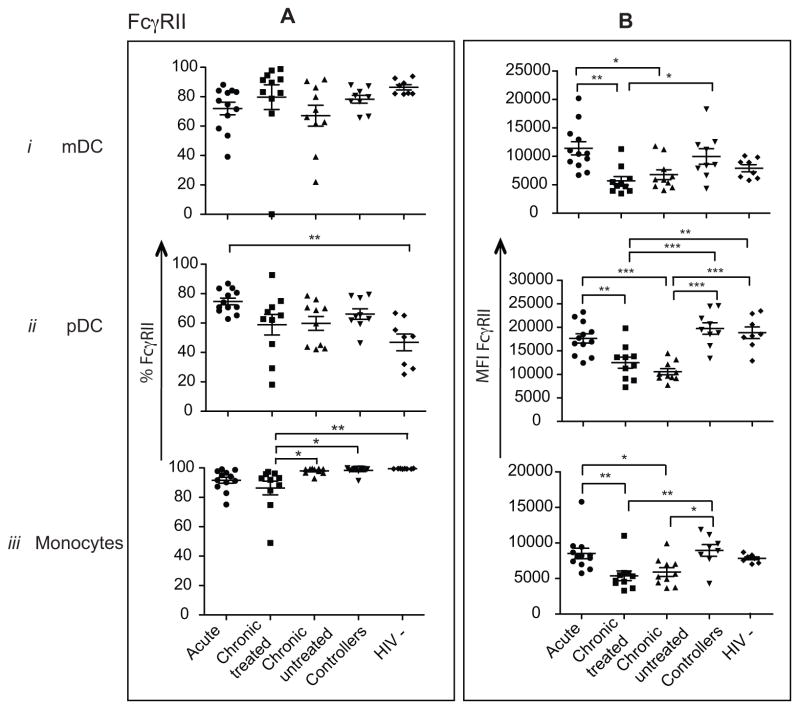
In contrast to FcγRI, FcγRII has a low affinity for monomeric IgG and typically only binds multimerized IgGs found on immune complexes (Hulett and Hogarth, 1994; Ravetch and Kinet, 1991). FcγRII+ mDCs (FIG 2Ai) and FcγRII+ monocytes (FIG 2Aiii) were slightly reduced in chronic untreated and chronic treated individuals, respectively, compared to controllers and negative controls. More interestingly, FcγRII expression was significantly reduced on monocytes in both chronic treated and untreated HIV-1 infected patients compared to controllers (p<0.01 and p<0.05, respectively, FIG 2Biii). Similar patterns of FcγRII expression were observed on mDCs (FIG 2Bi) and pDCs (FIG 2Bii) suggesting that despite a reduction in viral replication with successful antiretroviral therapy, expression of FcγRs does not return to normal levels, which potentially results in a persistent reduction in ADCC/ADCP activity. These data suggest that FcγRII is significantly downregulated on the surface of multiple innate immune cell subsets in both chronic treated and untreated HIV-1 infection, potentially resulting in irreversibly reduced ADCP activity in progressive infection even in the absence of active viral replication (Liu et al., 2009).
FIG. 2. FcγRII expression is strongly downregulated in chronic HIV infection.
The frequency (A) and the intensity (B) of FcγRII (CD32) expression was compared among acute (●), chronic treated (■), chronic untreated (⬪), controllers (⬧) and HIV-1 negative controls (◆) on (i) mDCs, (ii) pDCs and (iii) monocytes.
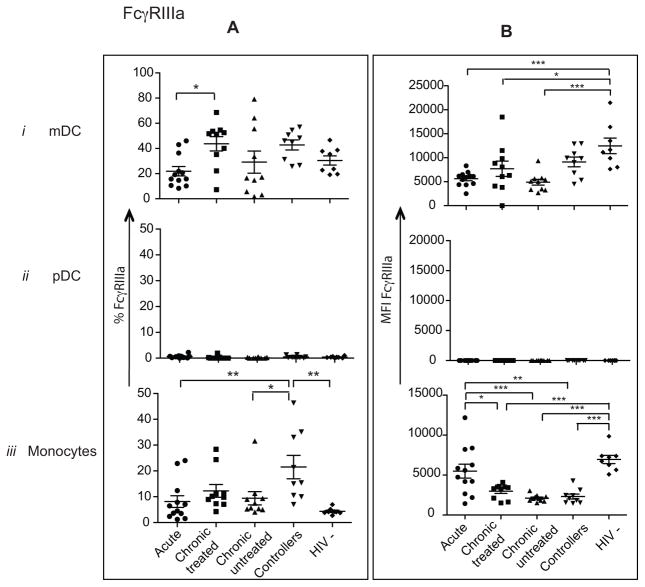
Like FcγRII, FcγRIIIa also binds antibodies with a weak affinity, and thus binds multimerized IgG complexes more readily (reviewed by (Nimmerjahn and Ravetch, 2008)). Previous work has show that FcγRIIIa expression is reduced on the surface of NK cells in chronic HIV infection, correlating with reduced ADCC activity (Liu et al., 2009). Interestingly, we also observed reduced FcγRIIIa expression on mDCs in both acute and chronic untreated HIV-1 infection compared to HIV-1 negative controls (p<0.001, for both comparisons FIG 3Bi). In addition, while FcγRIIIa+ monocytes were present at higher frequencies in controllers than in all the other groups (p<0.05 for all comparisons, FIG 3Aiii) its expression levels on monocytes was downregulated in all chronically infected individuals independent of treatment or spontaneous control compared to acute and HIV-negative controls (p<0.001, for both comparisons FIG 3Biii). Thus similar to FcγRII, FcγRIIIa expression is reduced on mDCs and monocytes in chronic HIV-1 infection, likely contributing to reduced ADCC/ADCP activity; whereas it is expressed at higher levels on innate immune cells in controllers, potentially allowing them to clear virus from the circulation more effectively.
FIG. 3. FcγRIIIa expression is downregulated on mDC and monocytes in chronic HIV-1 infection.
The frequency (A) of FcγRIII+ cells and the expression (B) of FcγRIII on mDCs, pDCs and monocytes was compared in subjects in HIV-infected subjects in acute (●), chronic treated ■), chronic untreated (⬪), controllers (⬧) and HIV-1 negative controls (◆).
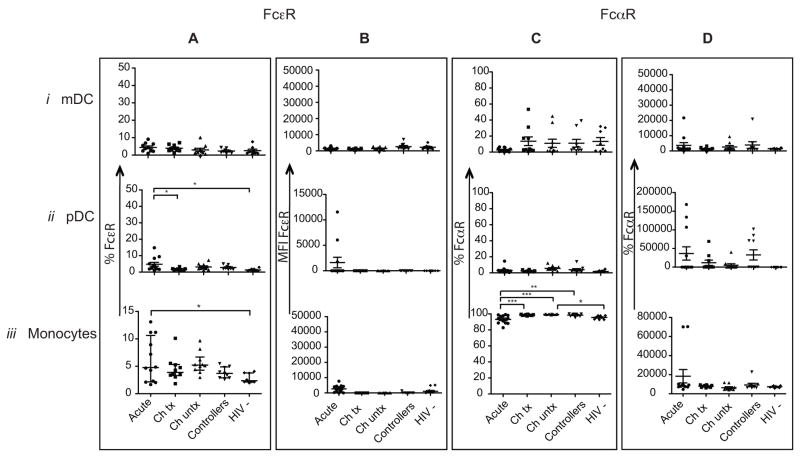
Finally we examined changes in FcαR and FcεR expression, which bind to IgA and IgE, respectively. While the frequency of mDCs and pDCs expressing FcαR and FcεR was extremely low, we observed an enrichment in the frequency of FcαR+ monocytes at all stages of HIV-1 infection (FIG 4). Acute HIV infection was associated with a trend towards an increase in the frequency of FcεR+ monocytes (FIG 4Aiii), elevated FcεR+ pDCs (FIG 4Aii), and a decreased frequency of FcαR+ monocytes compared to all groups (p<0.001 FIG 4Ciii). Overall, both FcεR and FcαR were weakly expressed on innate immune cells, however acute HIV infection was associated with a higher frequency of FcεR+ pDCs and monocytes suggesting that FcεR expression may also be upregulated to contribute to clearance of immune complexes bound by antibodies of other isotypes during early HIV-1 infection.
FIG. 4. Elevated frequencies of FcεR+ monocytes in acute HIV-1 infection.
The percentage and intensity FcεR (CD23) (A, B) and FcαR (CD89) (C, D) was compared on mDCs, pDCs and monocytes derived from patients at different stages of the infection including cells derived from acute (●), chronic treated (■), chronic untreated (⬪), controllers (⬧) and HIV-1 negative controls (◆).
Impairment of phagocytic activity in untreated chronically HIV infected individuals
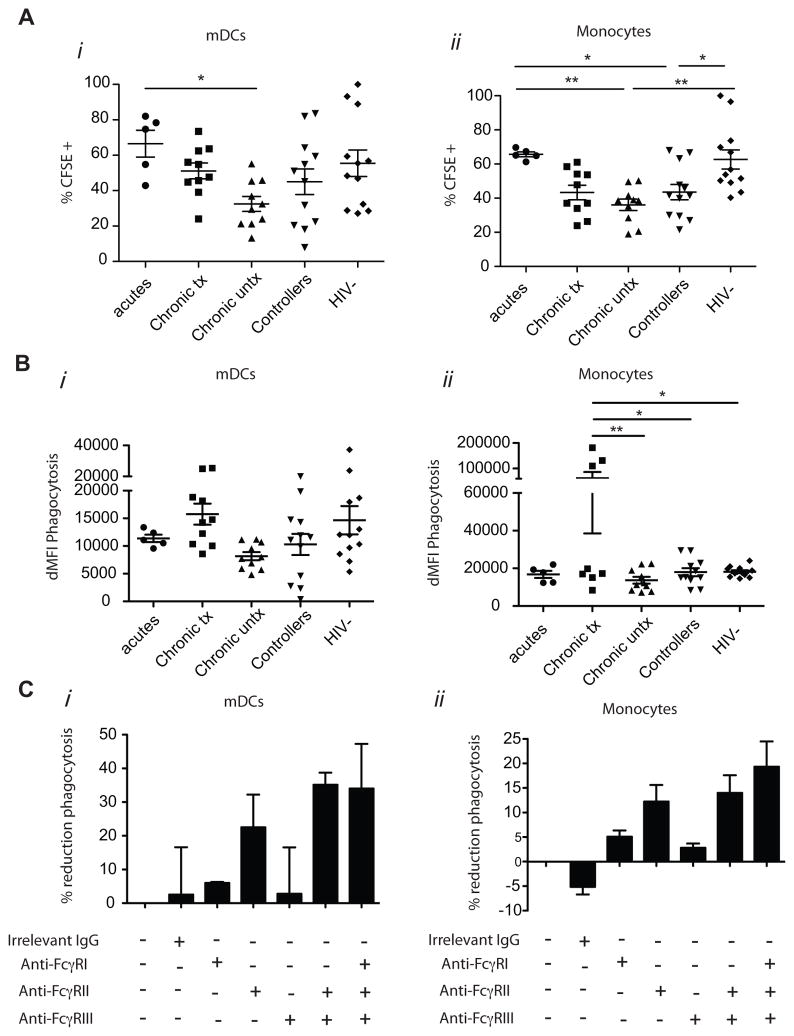
Previous reports that aimed at studying the underlying mechanism(s) accounting for alterations in phagocytic activity in progressive HIV infection (Kedzierska et al., 2002; Kedzierska et al., 2001) (and reviewed by Kedzierska et al (Kedzierska et al., 2003)) have shown that compromised ADCP activity is associated with a reduction in FcγRIIIa mobilization to the cell surface potentially due to shedding (Webster et al., 2006), or due to reduced expression of the γ signaling subunit of FcγRIIIa in HIV-infected phagocytes (Kedzierska et al., 2002; Leeansyah et al., 2007). We did not observed any correlation between ADCP activity and CD4 counts or viral loads (data not shown), thus we sought to determine whether changes in FcR expression at different stages of HIV infection on mDCs and monocytes may be associated with differences in their phagocytic activity. Thus we employed an opsonized, CFSE-labeled cell line, p815 cells, coated with p815-specific antibodies, to determine FcR-mediated phagocytic activity by the different innate immune effector cells by measuring CFSE-cell labeled uptake by flow cytometry in the presence or absence of antibody-coating. Our data demonstrate that mDCs and monocytes from acutely infected individuals possess an enhanced capacity to mediate ADCP, as these cells were able to take up elevated frequencies of antibody-coated CFSE+ p815 cells compared to cells from chronically infected individuals off therapy (p<0.1 and p<0.05 respectively, Fig 5Ai), suggesting that elevated FcγRI expression observed on mDCs and monocytes in acute infection may contribute to an enhanced capacity to clear immune complexes early in infection. We also observed a reduced frequency of mDC mediated-phagocytosis among untreated chronically infected patients compared to controllers, chronic treated and uninfected controls (Fig 5Ai). This compromised frequency of phagocytic cells was also observed in monocytes in chronic untreated patients compared to uninfected controls (p<0.05, FIG. 5Aii). These data suggest that changes in FcR expression on innate immune cells over the course of HIV-1 infection modulate ADCP activity.
FIG. 5. Impact of FcγRI, FcγRII and FcγRIIIa expression in FcR-mediated phagocytosis.
The dot plots represent differences in FcR mediated phagocytosis, measured as the proportion of phagocytic cells that have taken up an antibody coated target cell (A) or the intensity of phagocytosis (B), in mDCs (FIG. 5i) and monocytes (FIG. 5ii) among a sub-group of acutely infected patients, chronic treated, chronic untreated, controllers and HIV-1 negative individuals. (C) The bars represent the percentage of reduction in phagocytosis in mDCs (i) or monocytes (ii) in 3 different donors in the presence of FcγR-blocking antibodies.
While the frequency of phagocytic cells provide insights into the ability of the total population of innate immune cells to mediate ADCP, we also aimed to determine whether on a per cell level, ADCP differed among the patient populations. Thus we compared the intensity of phagocytosis on a per cell level by determining the difference in the mean fluorescence intensity (dMFI) of CFSE+ mDCs or monocytes in the presence of non-coated or antibody-coated p815 cells. No difference was observed in phagocytosis of the uncoated-815 cells among the patient populations, demonstrating that this ADCP activity is specific to the presence of the antibody. Moreover, the level of antibody-mediated phagocytosis (dMFI) on a per cell level was reduced in chronic untreated subjects compared to the chronically treated individuals in mDCs (Fig 5Bi) as well as in monocytes (p<0.05, Fig 5Bii), suggesting that not only there are fewer phagocytic innate immune cells, but on a per cell level, phagocytic cells also mediated ADCP inefficiently.
Given the dramatic changes in FcγR expression in HIV infection and their associated effect on compromising ADCP, we next sought to define whether changes in all or individual FcγRs modulated ADCP differentially using selective blocking reagents (FIG. 5C). As previously described in tumor cells (Richards et al., 2008), neither 10ug/mL of an irrelevant murine IgG nor FcγRIIIa blocking antibody had an impact on the capacity of mDCs or monocytes to induce ADCP. However selective blockade of either FcγRII alone or in combination with FcγRIIIa decreased ADCP in mDCs (FIG. 5Ci) and to a lesser extent in monocytes from healthy donors (FIG. 5Cii). Furthermore, the selective blockade of FcγRI alone also reduced ADCP activity in monocytes, albeit to a lesser extend then FcγRII. Moreover, triple blockade of FcγRI, FcγRII, and FcγRIIIA resulted in more robust inhibition of ADCP, suggesting that FcγRI may also contribute to the induction of phagocytosis (FIG. 5Cii). Thus overall, our data confirm that FcγRI and FcγRII expression on phagocytic cells play a central role in modulating ADCP over the course of HIV-1 infection. These data newly suggest that changes in FcγR expression over the course of HIV-1 infection, including an increase in FcγRI in acute infection and a decline in FcγRII expression in chronic infection, may differentially contribute to changes in ADCP function at these different stages of disease, altering the capacity of innate immune cells to clear and/or control HIV infection (Richards et al., 2008).
Discussion
Whereas ADCC has been shown to play a crucial role in better disease outcome in HIV-1 infection (Aasa-Chapman et al., 2005) (Forthal et al., 2001), little is known about additional non-neutralizing antiviral antibody functions, including ADCP which may play a central role in antiviral control through the rapid clearance of immune complexes. Furthermore, the recent results from the Thai trial suggest that non-neutralizing antibodies with the capacity to recruit the innate immune system through FcγR may have contributed to protection from HIV infection (Rerks-Ngarm et al., 2009), however the precise effector mechanism of these non-neutralizing antibodies is not known. In this study, we show that HIV-1 infection is associated with significant alterations in the frequency of FcγR expressing cells, and that these changes occur as early as acute HIV-1 infection. In fact, acute infection was strongly associated with an increase in the frequency of FcγRI+ mDCs as well as an increase in FcγRI expression on monocytes. In contrast, as opposed to acute infection, chronic HIV infection was associated with a significant and persistent downregulation of FcγRII expression, even following a reduction in viral replication with anti-retroviral therapy. Interestingly, we did not observe any statistical differences in the FcγRII expression on innate immune cells between chronics and HIV-negative individuals, suggesting that additional factors, independent of the receptor expression, may also contribute to alterations in ADCP activity. Furthermore, selective blockade of either FcγRI or FcγRII alone or in combination with FcγRIIIa on monocytes resulted in decreased phagocytosis, suggesting that alterations in these specific FcRs may play a central role in modulating ADCP activity. These data strongly suggest that increased expression of FcγRI on monocytes in acute infection may help promote viral clearance of immune complexes whereas decreased FcγRII expression in chronically infected individuals may dampen ADCP, resulting in compromised clearance of immune complexes in later disease. Overall, these results demonstrate that HIV-1 infection is associated with changes in the expression of specific FcγRs on innate immune cells that are responsible for alterations in the capacity of these cells to mediate ADCP in chronic infection potentially resulting in a failure of innate immune cells to efficiently clear immune complexes and therefore help control viral replication.
Several lines of evidence suggest that a number of myeloid cell activities are impaired following HIV-1 infection in vivo as well as in vitro including chemotaxis (Tas et al., 1988; Wahl et al., 1989), phagocytosis (Kedzierska et al., 2002; Kedzierska et al., 2000; Leeansyah et al., 2007; Webster et al., 2006), intracellular killing (Biggs et al., 1995), and cytokine production (Kedzierska et al., 2001). Several groups have speculated that the compromised phagocytic activity following HIV infection may partially contribute to the AIDS-associated pathogenesis, as the loss of these functions could result in poor antibody-mediated recruitment of innate immune cell associated viral control (Crowe, 1995; Crowe and Sonza, 2000; Kedzierska et al., 2003), however the mechanism by which this activity is lost is unknown. Previous reports have demonstrated that HIV-1 infection of monocyte derived macrophages (MDM) results in a defect in phagocytic activity (Leeansyah et al., 2007) in the absence of changes in the surface expression of FcγRs in vitro (Kedzierska et al., 2002). However only a small fraction of monocytes and mDCs are infected in vivo, and alterations in innate immune cell mediated phagocytic activity extend far beyond this small subset of infected cells. Thus additional mechanisms must account for changes in ADCP activity in the larger majority of phagocytic cells. Here we show that changes in the frequency and expression levels of particular FcγRs over the course of HIV-1 infection, starting in acute infection, may contribute to variation in the capacity of these cells to mediate ADCP.
Changes in FcR expression at different stages of HIV infection may provide critical insights into the receptors that may be centrally involved in the clearance of antibody-opsonized material. Along these lines, we observed a unique upregulation of FcγRI on monocytes in acute HIV infection, suggesting that at this early stage of infection, FcγRI expression may be critical for early clearance of immune complexes. Moreover, selective blockade of FcγRI on monocytes decreased ADCP activity, confirming its role in ADCP mediated clearance of antibody-opsonized material. In fact, FcγRI is the only high-affinity FcγR and is able to bind monomeric antibodies (Hulett and Hogarth, 1994; Ravetch and Kinet, 1991). Upregulation of FcγRI expressing monocytes may provide these phagocytic cells with the capacity to respond to antibody-opsonized material with higher sensitivity, to aide in viral control. Previous studies have shown that FcγR expression is altered on innate immune cells in other viral infections, driven by interferon-γ (IFN-γ) (Okayama et al., 2000), tumor growth factor-β (TGF-β) (Tridandapani et al., 2003) or other stimuli including LPS, associated with chronic immune activation in HIV as well as in SIV infection (Arend et al., 1987; Pricop et al., 2001) (Brenchley et al., 2006) supporting the fact that changes in the inflammatory milieu, due to cytokine cascades following infection may drive alterations in FcR expression at different stages of HIV infection, potentially modulating the capacity of innate immune cells to respond to antibody-opsonized material (Arend et al., 1987; Boruchov et al., 2005; Okayama et al., 2000; Tridandapani et al., 2003). Moreover, we show that FcγRII expression on mDCs and monocytes was reduced in chronic HIV infection and that alterations in the expression of this particular receptor contributed centrally to the reduced ADCP observed in chronic infection. Similarly, previous reports from the SLE model, suggest that FcγRIIa expression also declines on monocytes in this inflammatory disease contributing to impaired ADCP clearance of immune complexes in the sera of these patients (Szucs et al., 1994) strongly suggesting that FcγRIIa expression is required for phagocytic clearance of immune complexes in autoimmunity as well as during infection.
While a great deal of research has focused on cross talk between inhibitory and activating FcRs (Holl et al., 2004), little has been done in the way of understanding how different activating FcRs may synergize with one another to modulate effector functions. Moreover, while the selective blockade of FcγRI and/or FcγRII alone or in combination with FcγRIIIa inhibited ADCP activity, the selective blockade of FcγRIIIa alone did not modify ADCP (Figure 5Ci and Cii). These data suggest that both FcγRI and II independently promote ADCP activity, but also may synergize to additively mediate more robust ADCP when co-expressed on the same innate cell.
Finally, although the antibody-opsonized p815 assay does not directly measure the HIV-specific capacity of antibodies to trigger ADCP activity, it offers an indirect measure of the ADCP capacity of different innate immune cell subsets (Kondo et al., 1981)(Caligiuri et al., 1993)(Grazia Cifone et al., 1990). Thus this assay offers an indirect measure of the potential capacity of innate immune cells to mediate clearance of antibody-opsonized material during infection, that can be antibody-opsonized viral particles, HIV-infected cells, or other antibody-opsonized material. Thus it is also plausible that reduced ADCP activity in chronic HIV infection may also result in compromised clearance of other antibody-opsonized material that may contribute to reduced control/clearance of other opportunistic pathogens.
In conclusion, we show here that HIV-associated changes in ADCP activity in progressive infection may be directly related to changes in FcR expression. We highlight the potential involvement of particular FcRs in ADCP to help control viral infection at different stages of HIV infection, FcγRI in acute infection and FcγRII in viral control of infection. These data provide new insights into the mechanism of protective FcR mediated ADCP activity and its loss over the course of HIV infection that may be critical for efficient antibody-mediated control that may make important new targets to enhance protection mediated by HIV-specific antibodies through vaccination.
Materials and methods
Subjects
A total of 101 subjects were recruited for this study, including 23 healthy HIV-1 negative control subjects; 24 untreated viremic HIV-1-infected subjects with an average viral load of 41, 562 copies HIV-1 mRNA per ml of plasma (range, 1210 to 229,000 copies per ml) and an average of CD4 count of 538 cells per mm3 (range, 29 to 1041 cells per mm3); 20 HIV-1-infected subjects receiving highly active antiretroviral therapy (HAART), with undetectable viral loads (<50 copies) and an average of CD4 count of 716 cells per mm3 (range, 193 to 1534 cells per mm3); 17 Controllers recruited from the Controllers cohort at the Ragon institute (Pereyra et al., 2008) with an average viral load of 710 copies of RNA per ml plasma (range, 49 to 1662) and an average of CD4 count of 819 cells per mm3 (range, 445 to 1297 cells per mm3); and 17 acutely HIV-1-infected individuals recruited from our Boston Acute Infection cohort. Acute samples were in a Fiebig stage IV–V, with an average viral load of 18,066,300 copies per ml (range, 200,000 to 66,000,000 copies) and an average of CD4 count of 460 cells per mm3 (range, 310 to 517 cells per mm3). All samples were cryopreserved prior to usage. The MGH institutional review board approved the study, and each subject gave written informed consent for participation in the study (Pereyra et al., 2008).
Phenotyping
Peripheral blood mononuclear cells (PBMCs) were obtained after Ficoll-Hypaque density gradient centrifugation of whole blood. FcR expression was assessed on three different cell subsets from cryopreserved PBMCs: mDCs (myeloid dendritic cells), defined as CD3neg CD14neg CD123neg and CD11cpos; pDCs (plasmacytoid dendritic cells), defined as CD3neg CD14neg CD123pos and CD11cneg; and monocytes, defined as CD3neg and CD14pos. PBMC were stained with blue viability dye (Invitrogen), and then with CD64-FITC, CD23-biotin, CD16-APCCy7, CD89-PE, CD3-alexa700, CD123-PECy5, CD14-PB (BD biosciences), CD32-APC (Biolegend), CD11c-PECy5.5 (Invitrogen) and streptavidin Cascade Yellow (Invitrogen). An average of 6.7×105 PBMCs events (range 3×105-1×106 cells), were acquired for each individual on a BD LSRII, and data were analyzed with FlowJo software (version 8.7.1).
FcγR-mediated phagocytosis
p815 cells (a mouse leukemic cell line) were stained with 0.25 uM carboxy-fluorescein succinimidyl ester (CFSE; Molecular Probes) and incubated with a p815-specific rabbit monoclonal antibody (10ug/mL) (Accurate Chemical), previously used widely to measure human Fc-receptor function (Caligiuri et al., 1993; Grazia Cifone et al., 1990; Kondo et al., 1981). Coated and uncoated p815 cells were then cocultured with previously cryopreserved PBMC from 5 recently acute infected individuals, 10 chronic untreated, 10 chronic treated, 12 controllers and 10 negative controls at an effector:target ratio of 10:1 in 10% fetal calf serum (Sigma), penicillin, glutamine and streptomycin (CellGro). The percentage of effector cells that took-up CFSE labeled-target cells was monitored by flow cytometry using the following markers: CD3-Alexa 700, CD14-APC, CD123-PECy5 (BD Biosciences) and CD11c-PECy5.5 (Invitrogen). Then the delta-Mean Fluorescence Intensity (dMFI) was calculated as the percentage of effector cells that phagocytosed CFSE-labeled uncoated-target cells subtracted from the percentage of effector cells that took-up CFSE-positive target cells from antibody-coated p815 cells. A minimum of 300 000 cells were acquired on a BD LSRII, and analysis was then performed using FlowJo software.
FcγR blockade assay
Blocking antibodies FcγRI (10ug/mL, clone 10-1; BD pharmingen), FcγRII (10ug/mL, clone AT10; R and D systems) and/or FcγRIIIa (10ug/ml, clone 3G8; BD pharmingen) were added to PBMCs for 1 hour at 37 degrees before the addition of either antibody coated or non-coated CFSE-labeled p815 cells for 3 hours. Phagocytosis was evaluated as the proportion of mDCs and monocytes that took up fluorescent antibody coated p815 cells by flow cytometry using the following markers: CD3-Alexa 700, CD14-APC, CD123-PECy5 (BD Biosciences) and CD11c-PECy5.5 (Invitrogen). Cells were acquired on a BD LSRII, and analysis was then performed using FlowJo software.
Statistics
Statistical analysis was performed using Graph Pad Prism. Statistical comparisons of marker expression levels were made using one-way ANOVA. P-values of pairwise comparisons were adjusted for multiple comparisons using Tukey’s method. Significant differences were noted when p<0.05. (* represents p < 0.05; ** represents p < 0.001 and *** represents p < 0.0001).
Table 1.
Patient characteristics
| Groups | n | Viral Load (RNA copies/mL) | CD4 T cells (/uL) |
|---|---|---|---|
| HIV-negative controls | 23 | ||
| Controllers | 17 | 710 | 819 |
| Acute | 17 | 18,066,300 | 460 |
| Chronic treated | 20 | <50 | 716 |
| Chronic untreated | 24 | 41562 | 538 |
Acknowledgments
This work was supported by the National Institute of Health (R01 AI080289).
Financial report: National Institute of Health (R01 AI080289)
Footnotes
Potential conflict of interest: none reported
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Aasa-Chapman MM, Holuigue S, Aubin K, Wong M, Jones NA, Cornforth D, Pellegrino P, Newton P, Williams I, Borrow P, McKnight A. Detection of antibody-dependent complement-mediated inactivation of both autologous and heterologous virus in primary human immunodeficiency virus type 1 infection. J Virol. 2005;79:2823–2830. doi: 10.1128/JVI.79.5.2823-2830.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alter G, Teigen N, Davis BT, Addo MM, Suscovich TJ, Waring MT, Streeck H, Johnston MN, Staller KD, Zaman MT, Yu XG, Lichterfeld M, Basgoz N, Rosenberg ES, Altfeld M. Sequential deregulation of NK cell subset distribution and function starting in acute HIV-1 infection. Blood. 2005;106:3366–3369. doi: 10.1182/blood-2005-03-1100. [DOI] [PubMed] [Google Scholar]
- Alter G, Tsoukas CM, Rouleau D, Cote P, Routy JP, Sekaly RP, Bernard NF. Assessment of longitudinal changes in HIV-specific effector activity in subjects undergoing untreated primary HIV infection. AIDS. 2004;18:1979–1989. doi: 10.1097/00002030-200410210-00002. [DOI] [PubMed] [Google Scholar]
- Anderson CL. Structural and functional polymorphism of human Fc receptors for IgG. Chem Immunol. 1989;47:1–20. doi: 10.1159/000318901. [DOI] [PubMed] [Google Scholar]
- Arend WP, Ammons JT, Kotzin BL. Lipopolysaccharide and interleukin 1 inhibit interferon-gamma-induced Fc receptor expression on human monocytes. J Immunol. 1987;139:1873–1879. [PubMed] [Google Scholar]
- Bave U, Magnusson M, Eloranta ML, Perers A, Alm GV, Ronnblom L. Fc gamma RIIa is expressed on natural IFN-alpha-producing cells (plasmacytoid dendritic cells) and is required for the IFN-alpha production induced by apoptotic cells combined with lupus IgG. J Immunol. 2003;171:3296–3302. doi: 10.4049/jimmunol.171.6.3296. [DOI] [PubMed] [Google Scholar]
- Biggs BA, Hewish M, Kent S, Hayes K, Crowe SM. HIV-1 infection of human macrophages impairs phagocytosis and killing of Toxoplasma gondii. J Immunol. 1995;154:6132–6139. [PubMed] [Google Scholar]
- Boruchov AM, Heller G, Veri MC, Bonvini E, Ravetch JV, Young JW. Activating and inhibitory IgG Fc receptors on human DCs mediate opposing functions. J Clin Invest. 2005;115:2914–2923. doi: 10.1172/JCI24772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenchley JM, Price DA, Schacker TW, Asher TE, Silvestri G, Rao S, Kazzaz Z, Bornstein E, Lambotte O, Altmann D, Blazar BR, Rodriguez B, Teixeira-Johnson L, Landay A, Martin JN, Hecht FM, Picker LJ, Lederman MM, Deeks SG, Douek DC. Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nat Med. 2006;12:1365–1371. doi: 10.1038/nm1511. [DOI] [PubMed] [Google Scholar]
- Caligiuri MA, Murray C, Robertson MJ, Wang E, Cochran K, Cameron C, Schow P, Ross ME, Klumpp TR, Soiffer RJ, et al. Selective modulation of human natural killer cells in vivo after prolonged infusion of low dose recombinant interleukin 2. J Clin Invest. 1993;91:123–132. doi: 10.1172/JCI116161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crowe SM. Role of macrophages in the pathogenesis of human immunodeficiency virus (HIV) infection. Aust N Z J Med. 1995;25:777–783. doi: 10.1111/j.1445-5994.1995.tb02881.x. [DOI] [PubMed] [Google Scholar]
- Crowe SM, Sonza S. HIV-1 can be recovered from a variety of cells including peripheral blood monocytes of patients receiving highly active antiretroviral therapy: a further obstacle to eradication. J Leukoc Biol. 2000;68:345–350. [PubMed] [Google Scholar]
- Dall’Ozzo S, Tartas S, Paintaud G, Cartron G, Colombat P, Bardos P, Watier H, Thibault G. Rituximab-dependent cytotoxicity by natural killer cells: influence of FCGR3A polymorphism on the concentration-effect relationship. Cancer Res. 2004;64:4664–4669. doi: 10.1158/0008-5472.CAN-03-2862. [DOI] [PubMed] [Google Scholar]
- Diamond B, Bloom BR, Scharff MD. The Fc receptors of primary and cultured phagocytic cells studied with homogeneous antibodies. J Immunol. 1978;121:1329–1333. [PubMed] [Google Scholar]
- Eger KA, Unutmaz D. Perturbation of natural killer cell function and receptors during HIV infection. Trends Microbiol. 2004;12:301–303. doi: 10.1016/j.tim.2004.05.006. [DOI] [PubMed] [Google Scholar]
- Forthal DN, Landucci G, Daar ES. Antibody from patients with acute human immunodeficiency virus (HIV) infection inhibits primary strains of HIV type 1 in the presence of natural-killer effector cells. J Virol. 2001;75:6953–6961. doi: 10.1128/JVI.75.15.6953-6961.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grazia Cifone M, Giacomelli R, Famularo G, Paolini R, Danese C, Napolitano T, Procopio A, Perego AM, Santoni A, Tonietti G. Natural killer activity and antibody-dependent cellular cytotoxicity in progressive systemic sclerosis. Clin Exp Immunol. 1990;80:360–365. doi: 10.1111/j.1365-2249.1990.tb03293.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holl V, Hemmerter S, Burrer R, Schmidt S, Bohbot A, Aubertin AM, Moog C. Involvement of Fc gamma RI (CD64) in the mechanism of HIV-1 inhibition by polyclonal IgG purified from infected patients in cultured monocyte-derived macrophages. J Immunol. 2004;173:6274–6283. doi: 10.4049/jimmunol.173.10.6274. [DOI] [PubMed] [Google Scholar]
- Hu PF, Hultin LE, Hultin P, Hausner MA, Hirji K, Jewett A, Bonavida B, Detels R, Giorgi JV. Natural killer cell immunodeficiency in HIV disease is manifest by profoundly decreased numbers of CD16+CD56+ cells and expansion of a population of CD16dimCD56- cells with low lytic activity. J Acquir Immune Defic Syndr Hum Retrovirol. 1995;10:331–340. [PubMed] [Google Scholar]
- Hulett MD, Hogarth PM. Molecular basis of Fc receptor function. Adv Immunol. 1994;57:1–127. doi: 10.1016/s0065-2776(08)60671-9. [DOI] [PubMed] [Google Scholar]
- Kedzierska K, Azzam R, Ellery P, Mak J, Jaworowski A, Crowe SM. Defective phagocytosis by human monocyte/macrophages following HIV-1 infection: underlying mechanisms and modulation by adjunctive cytokine therapy. J Clin Virol. 2003;26:247–263. doi: 10.1016/s1386-6532(02)00123-3. [DOI] [PubMed] [Google Scholar]
- Kedzierska K, Ellery P, Mak J, Lewin SR, Crowe SM, Jaworowski A. HIV-1 down-modulates gamma signaling chain of Fc gamma R in human macrophages: a possible mechanism for inhibition of phagocytosis. J Immunol. 2002;168:2895–2903. doi: 10.4049/jimmunol.168.6.2895. [DOI] [PubMed] [Google Scholar]
- Kedzierska K, Mak J, Jaworowski A, Greenway A, Violo A, Chan HT, Hocking J, Purcell D, Sullivan JS, Mills J, Crowe S. nef-deleted HIV-1 inhibits phagocytosis by monocyte-derived macrophages in vitro but not by peripheral blood monocytes in vivo. AIDS. 2001;15:945–955. doi: 10.1097/00002030-200105250-00002. [DOI] [PubMed] [Google Scholar]
- Kedzierska K, Mak J, Mijch A, Cooke I, Rainbird M, Roberts S, Paukovics G, Jolley D, Lopez A, Crowe SM. Granulocyte-macrophage colony-stimulating factor augments phagocytosis of Mycobacterium avium complex by human immunodeficiency virus type 1-infected monocytes/macrophages in vitro and in vivo. J Infect Dis. 2000;181:390–394. doi: 10.1086/315191. [DOI] [PubMed] [Google Scholar]
- Kondo LL, Rosenau W, Wara DW. Role of lymphotoxin in antibody-dependent cell-mediated cytotoxicity (ADCC) J Immunol. 1981;126:1131–1133. [PubMed] [Google Scholar]
- Laszlo A, Petri I, Ilyes M. Antibody dependent cellular cytotoxicity (ADCC)-reaction and an in vitro steroid sensitivity test of peripheral lymphocytes in children with malignant haematological and autoimmune diseases. Acta Paediatr Hung. 1986;27:23–29. [PubMed] [Google Scholar]
- Leeansyah E, Wines BD, Crowe SM, Jaworowski A. The mechanism underlying defective Fcgamma receptor-mediated phagocytosis by HIV-1-infected human monocyte-derived macrophages. J Immunol. 2007;178:1096–1104. doi: 10.4049/jimmunol.178.2.1096. [DOI] [PubMed] [Google Scholar]
- Liu Q, Sun Y, Rihn S, Nolting A, Tsoukas PN, Jost S, Cohen K, Walker B, Alter G. Matrix Metalloprotease inhibitors restore impaired NK cell mediated Antibody dependent cellular cytotoxicity in human immunodeficiency virus-1 infection. J Virol. 2009 doi: 10.1128/JVI.02666-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mavilio D, Benjamin J, Daucher M, Lombardo G, Kottilil S, Planta MA, Marcenaro E, Bottino C, Moretta L, Moretta A, Fauci AS. Natural killer cells in HIV-1 infection: dichotomous effects of viremia on inhibitory and activating receptors and their functional correlates. Proc Natl Acad Sci U S A. 2003;100:15011–15016. doi: 10.1073/pnas.2336091100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Natsume A, Niwa R, Satoh M. Improving effector functions of antibodies for cancer treatment: Enhancing ADCC and CDC. Drug Des Devel Ther. 2009;3:7–16. [PMC free article] [PubMed] [Google Scholar]
- Nimmerjahn F, Ravetch JV. Fc-receptors as regulators of immunity. Adv Immunol. 2007;96:179–204. doi: 10.1016/S0065-2776(07)96005-8. [DOI] [PubMed] [Google Scholar]
- Nimmerjahn F, Ravetch JV. Fcgamma receptors as regulators of immune responses. Nat Rev Immunol. 2008;8:34–47. doi: 10.1038/nri2206. [DOI] [PubMed] [Google Scholar]
- Oganesyan V, Damschroder MM, Leach W, Wu H, Dall’Acqua WF. Structural characterization of a mutated, ADCC-enhanced human Fc fragment. Mol Immunol. 2008;45:1872–1882. doi: 10.1016/j.molimm.2007.10.042. [DOI] [PubMed] [Google Scholar]
- Okayama Y, Kirshenbaum AS, Metcalfe DD. Expression of a functional high-affinity IgG receptor, Fc gamma RI, on human mast cells: Up-regulation by IFN-gamma. J Immunol. 2000;164:4332–4339. doi: 10.4049/jimmunol.164.8.4332. [DOI] [PubMed] [Google Scholar]
- Pereyra F, Addo MM, Kaufmann DE, Liu Y, Miura T, Rathod A, Baker B, Trocha A, Rosenberg R, Mackey E, Ueda P, Lu Z, Cohen D, Wrin T, Petropoulos CJ, Rosenberg ES, Walker BD. Genetic and immunologic heterogeneity among persons who control HIV infection in the absence of therapy. J Infect Dis. 2008;197:563–571. doi: 10.1086/526786. [DOI] [PubMed] [Google Scholar]
- Pricop L, Redecha P, Teillaud JL, Frey J, Fridman WH, Sautes-Fridman C, Salmon JE. Differential modulation of stimulatory and inhibitory Fc gamma receptors on human monocytes by Th1 and Th2 cytokines. J Immunol. 2001;166:531–537. doi: 10.4049/jimmunol.166.1.531. [DOI] [PubMed] [Google Scholar]
- Ravetch JV, Kinet JP. Fc receptors. Annu Rev Immunol. 1991;9:457–492. doi: 10.1146/annurev.iy.09.040191.002325. [DOI] [PubMed] [Google Scholar]
- Rerks-Ngarm S, Pitisuttithum P, Nitayaphan S, Kaewkungwal J, Chiu J, Paris R, Premsri N, Namwat C, de Souza M, Adams E, Benenson M, Gurunathan S, Tartaglia J, McNeil JG, Francis DP, Stablein D, Birx DL, Chunsuttiwat S, Khamboonruang C, Thongcharoen P, Robb ML, Michael NL, Kunasol P, Kim JH. Vaccination with ALVAC and AIDSVAX to Prevent HIV-1 Infection in Thailand. N Engl J Med. 2009 doi: 10.1056/NEJMoa0908492. [DOI] [PubMed] [Google Scholar]
- Richards JO, Karki S, Lazar GA, Chen H, Dang W, Desjarlais JR. Optimization of antibody binding to FcgammaRIIa enhances macrophage phagocytosis of tumor cells. Mol Cancer Ther. 2008;7:2517–2527. doi: 10.1158/1535-7163.MCT-08-0201. [DOI] [PubMed] [Google Scholar]
- Shore SL, Nahmias AJ, Starr SE, Wood PA, McFarlin DE. Detection of cell-dependent cytotoxic antibody to cells infected with herpes simplex virus. Nature. 1974;251:350–352. doi: 10.1038/251350a0. [DOI] [PubMed] [Google Scholar]
- Siberil S, Dutertre CA, Fridman WH, Teillaud JL. FcgammaR: The key to optimize therapeutic antibodies? Crit Rev Oncol Hematol. 2007;62:26–33. doi: 10.1016/j.critrevonc.2006.12.003. [DOI] [PubMed] [Google Scholar]
- Szucs G, Kavai M, Suranyi P, Kiss E, Csipo I, Szegedi G. Correlations of monocyte phagocytic receptor expressions with serum immune complex level in systemic lupus erythematosus. Scand J Immunol. 1994;40:481–484. doi: 10.1111/j.1365-3083.1994.tb03493.x. [DOI] [PubMed] [Google Scholar]
- Tas M, Drexhage HA, Goudsmit J. A monocyte chemotaxis inhibiting factor in serum of HIV infected men shares epitopes with the HIV transmembrane protein gp41. Clin Exp Immunol. 1988;71:13–18. [PMC free article] [PubMed] [Google Scholar]
- Tomaras GD, Yates NL, Liu P, Qin L, Fouda GG, Chavez LL, Decamp AC, Parks RJ, Ashley VC, Lucas JT, Cohen M, Eron J, Hicks CB, Liao HX, Self SG, Landucci G, Forthal DN, Weinhold KJ, Keele BF, Hahn BH, Greenberg ML, Morris L, Karim SS, Blattner WA, Montefiori DC, Shaw GM, Perelson AS, Haynes BF. Initial B-cell responses to transmitted human immunodeficiency virus type 1: virion-binding immunoglobulin M (IgM) and IgG antibodies followed by plasma anti-gp41 antibodies with ineffective control of initial viremia. J Virol. 2008;82:12449–12463. doi: 10.1128/JVI.01708-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tridandapani S, Wardrop R, Baran CP, Wang Y, Opalek JM, Caligiuri MA, Marsh CB. TGF-beta 1 suppresses [correction of supresses] myeloid Fc gamma receptor function by regulating the expression and function of the common gamma-subunit. J Immunol. 2003;170:4572–4577. doi: 10.4049/jimmunol.170.9.4572. [DOI] [PubMed] [Google Scholar]
- Wahl SM, Allen JB, Gartner S, Orenstein JM, Popovic M, Chenoweth DE, Arthur LO, Farrar WL, Wahl LM. HIV-1 and its envelope glycoprotein down-regulate chemotactic ligand receptors and chemotactic function of peripheral blood monocytes. J Immunol. 1989;142:3553–3559. [PubMed] [Google Scholar]
- Webster NL, Kedzierska K, Azzam R, Paukovics G, Wilson J, Crowe SM, Jaworowski A. Phagocytosis stimulates mobilization and shedding of intracellular CD16A in human monocytes and macrophages: inhibition by HIV-1 infection. J Leukoc Biol. 2006;79:294–302. doi: 10.1189/jlb.0705382. [DOI] [PubMed] [Google Scholar]