Abstract
Cyanobacterial aldehyde decarbonylase (AD) catalyzes conversion of fatty aldehydes (R-CHO) to alka(e)nes (R-H) and formate. Curiously, although this reaction appears to be redox-neutral and formally hydrolytic, AD has a ferritin-like protein architecture and a carboxylate-bridged di-metal cofactor that are both structurally similar to those found in di-iron oxidases and oxygenases. In addition, the in vitro activity of the AD from Nostoc punctiforme (Np) was shown to require a reducing system similar to the systems employed by these O2-utilizing di-iron enzymes. Here, we resolve this conundrum by showing that aldehyde cleavage by the Np AD also requires dioxygen and results in incorporation of 18O from 18O2 into the formate product. AD thus oxygenates, without oxidizing, its substrate. We posit that (i) O2 adds to the reduced cofactor to generate a metal-bound peroxide nucleophile that attacks the substrate carbonyl and initiates a radical scission of the C1-C2 bond, and (ii) the reducing system delivers two electrons during aldehyde cleavage, ensuring a redox-neutral outcome, and two additional electrons to return an oxidized form of the cofactor back to the reduced, O2-reactive form.
Photosynthetic cyanobacteria use light to “fix” CO2 into energy-rich biomolecules, including fatty acids.1,2 A two-step pathway recently identified by Schirmer, et al. allows these organisms also to produce alkanes and alkenes from abundant saturated and unsaturated fatty acids.3 Together, these two pathways could potentially be harnessed for a bioprocess that would effectively harvest solar energy, store it as a fungible fuel, and consume an important greenhouse gas.2 The incalculable potential value of such a process has motivated a flurry of scientific,3,4 intellectual-property,5,6 and investment activity.7 A deeper understanding of the cyanobacterial alkane-biosynthetic pathway might facilitate efforts to develop such a process.
The second step in the pathway is catalyzed by the enzyme aldehyde decarbonylase (AD8). Schirmer, et al. suggested that AD converts the Cn fatty aldehyde product of the first enzyme into the corresponding Cn−1 alkane or alkene and carbon monoxide (CO),3 but we recently showed that the C1-derived co-product from the in vitro conversion of octadecanal (R-13CHO, where R = n-C17H35) to heptadecane (R-H) by the Nostoc punctiforme (Np) AD produced heterologously in Escherichia coli (Ec) is formate rather than CO.4 On paper this reaction is formally hydrolytic (redox neutral). However, the structure of the Prochlorococcus marinus (Pm) MIT9313 ortholog showed that the ADs have a ferritin-like protein architecture and cofactor site that are similar to those found in non-heme di-iron oxygenases and oxidases,3 including bacterial multicomponent monooxygenases (e.g., soluble methane monooxygenase)9,10 and plant fatty acyl-ACP desaturases.11 Moreover, the in vitro activity of the Np AD was observed (by both Schirmer, et al.3 and us4) to require a reducing system (fulfilled by NADPH and spinach ferredoxin and ferredoxin reductase; hereafter N/F/FR), just as these di-iron oxidases and oxygenases require reducing systems to convert the Fe2III/III “resting” forms of their cofactors to the O2-reactive Fe2II/II states during each reaction cycle.9–11 It appears that these analogies to known di-iron oxidases and oxygenases led to the incorrect depiction of the C1-derived co-product as CO2 (which would be an oxidative outcome) and the designation of the cyanobacterial ADs as “decarboxylative monooxygenases” in a patent application from the company Joule Unlimited (Boston, MA).6 The available data thus created a puzzle: how does an enzyme that looks and acts like an O2-utilizing oxidoreductase effect a formally hydrolytic reaction?
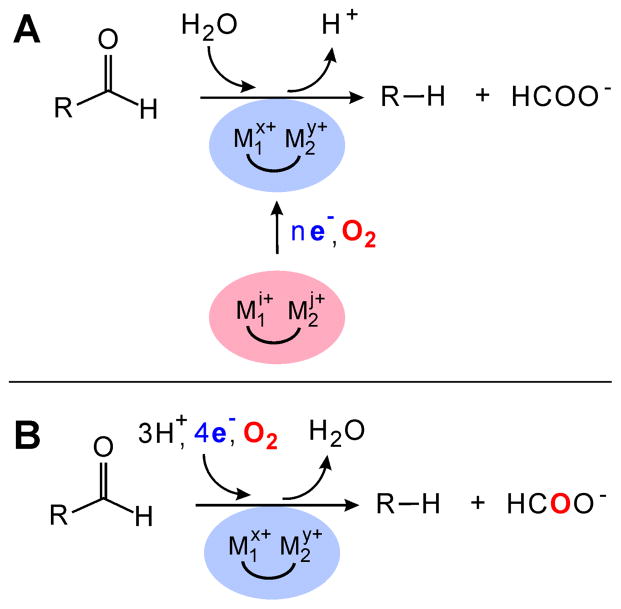
We suggested two possible resolutions to this conundrum.4,12 First, the Np AD might emerge from expression and purification with its cofactor in an inactive oxidation state, requiring reduction of the cofactor and reaction with O2 to regenerate the active state (Scheme 1A). This idea was founded on our understanding of class I ribonucleotide reductases, which (i) are structurally similar to the ADs,13,14 (ii) contain stable, catalytically essential oxidants (a tyrosyl radical in class Ia15–17 and Ib18,19 and the MnIV ion of a Mn/Fe cluster in class Ic20–22), (iii) can be inactivated by reduction of their essential oxidants,15,23,24 and (iv) in the case of the class Ia enzymes, can then be reactivated by reduction of their Fe2III/III clusters and subsequent reaction of the Fe2II/II forms with O2.25 Second, O2 might be an obligatory co-substrate in some sort of (heretofore unprecedented) cryptically redox pathway for the formally hydrolytic aldehyde cleavage. This case would require that four electrons be delivered by the reducing system in each turnover for redox balance (Scheme 1B).
Scheme 1. Two Alternative Explanations for the Similarity of Np AD to Di-iron Oxidases and Oxygenases and its Requirement for a Reducing System to Promote an Apparently Hydrolytic Reactiona.
a(A) The reducing system provides n electrons (e−), and the most reduced form of the cofactor reacts with O2 to generate the active state, which is more oxidized than the as-isolated form by 4 − n units (x + y − i − j = 4 − n); (B) O2 is reduced in every cycle by four electrons from the reducing system.
Both potential resolutions imply that O2 should be required for AD activity, a prediction not tested to date. More diagnostically, if the N/F/FR reducing system and possibly O2 should be required only for reactivation of the AD, but not for turnover, then it might be possible to observe activity after first exposing the enzyme to both in the absence of substrate and then removing one or both prior to initiating the reaction by addition of the substrate. By contrast, the second scenario would imply that omission of O2 or any component of the reducing system should prevent turnover, irrespective of any pre-treatment. To clarify the nature and mechanism of the novel AD reaction, we experimentally evaluated these predictions.
We first assessed whether O2 is required for activity. One set of reaction samples (see Figure 1 legend for their composition) was constituted with O2-free components inside an anoxic chamber (MBraun, Peabody, MA) by adding a pre-mixed solution of the N/F/FR reducing system and the Np AD (the affinity-tagged enzyme used in our previous study and purified from Ec as described therein4) to a solution containing the R-13CHO substrate and Triton x-100 detergent (0.2% in the complete sample). A matched set of samples was prepared by removing the pre-mixed solution of N/F/FR reducing system and Np AD from the anoxic chamber and adding it under ambient atmosphere to an air-saturated solution of the substrate and detergent. These two sets of samples were incubated at ambient temperature (23 ± 1 °C) for between 1.5 and 20 h and then analyzed for [13C]-formate by conversion to its 2-nitrophenylhydrazide (2NPH) derivative and quantification by liquid chromatography and mass spectrometry (LC-MS), as previously described.4 Mass spectra of the samples incubated under air exhibit intense peaks at m/z = 181 (Figure 1A, black bars), indicating that, as expected, formate was produced in these reactions (145 μM in Figure 1A). Note that the peak at m/z = 180 in the mass spectra of all samples arises from contaminating environmental formate, which also contributes weakly at m/z = 181 due to the presence of natural-abundance M+1 isotopic species (~ 8.9% of the m/z = 180 species) in its 2NPH derivative. By contrast, spectra of the samples prepared and incubated in the absence of O2 have much weaker peaks at m/z = 181 (Figure 1A, gray bars), implying that much less formate (9 μM) was produced in these reactions. This result demonstrates that the Np AD requires O2 in some capacity. To assess whether O2 is required for activation (Scheme 1A) or catalysis (Scheme 1B), an additional matched sample was prepared by removing the solution of N/F/FR system and Np AD from the anoxic chamber and exposing it to air for 30 min, with periodic mixing to ensure permeation of O2 into the enzyme solution. The air-exposed (N/F/FR + Np AD) solution was then returned to the anoxic chamber and mixed therein with 9 equivalent volumes of an O2-free solution of substrate and detergent. The sample was placed in a sealed vessel, incubated for 20 h, and analyzed as above. The weak peak at m/z = 181 (Figure 1A, orange bars) implies that prior exposure of the Np AD to O2 in the presence of the reducing system does not activate it for subsequent O2-independent turnover. These results suggest that O2 must be continuously present for activity.
Figure 1.

Reconstructed mass spectra illustrating the catalytic requirement for O2 and the incorporation of 18O from 18O2 into the formate product in the Np AD reaction. (A) Reactions in the continuous presence of O2 (under air atmosphere, black bars), the absence of O2 (gray), and the presence of O2 during pre-incubation with the N/F/FR system (without substrate) but the absence of O2 during the reaction (orange). (B) Reactions under an atmosphere of natural-abundance O2 (red) or 18O2 (99% isotopic purity; blue). (C) Control reactions in which the 2NPH derivative of either natural-abundance formate (green) or [13C]-formate (purple) was generated in H218O (70% enrichment) to quantify the extent of exchange of the oxygen atoms during the coupling reaction. The reactions in A were carried out at 21 °C for 20 h and contained, in a final volume of 0.40 mL, 0.10 mM Np AD, 0.5 mM R-13CHO substrate, 2 mM NADPH, and 100 μg/mL each of spinach ferredoxin and ferredoxin reductase in air-saturated 100 mM HEPES buffer, pH 7.4, containing 0.2% triton X-100. The reactions in B had the same composition but were carried out for 2 h.
In addition to scission of the C1-C2 bond and formation of a new C2-H bond, a second O-atom is added to C1 in conversion of R-CHO to R-H and HCO2−. The requirement for O2 raised the possibility that it could be the source of this O-atom, as occurs in other oxygenase reactions. Because substrate oxygenation without net oxidation is, to the best of our knowledge, unprecedented, we considered this possibility to be unlikely but tested for it nevertheless. Surprisingly, the mass spectrum of a sample in which the reaction was carried out under an atmosphere of 18O2 (Figure 1B, blue bars) exhibits a strong peak at m/z = 183 (an increase of two mass units) that is absent in the spectrum of a control sample prepared identically but with natural-abundance O2 (red bars). It is important to note here that the analysis for formate, which involves its prior conversion to formyl-2NPH, leads to loss of one of two equivalent oxygen atoms, and so quantitative incorporation of a single 18O atom into the formate product should result in precisely 50% of the derivative having m/z = 183 (the other 50% having m/z = 181). Integration of the peaks in Figure 1B and analogous spectra from two other trials gave only (34 ± 3)% (mean and standard deviation) of the m/z = 183 species. However, abortive coupling in the derivatization (i.e., carboxylate attack on the activating carbodiimide, EDC, followed by hydrolysis of the adduct) would lead to further loss of 18O and diminution of the m/z = 183 peak relative to the m/z = 181 peak. To assess whether the loss of ~ 16% of the m/z = 183 species in Figure 1B can be explained this way or reflects partial exchange of the O2-derived atom with solvent during the AD reaction, control samples were constituted in H218O (70% enrichment), subjected to the 2NPH coupling procedure, and analyzed by LC-MS (Figure 1C). They contained all reaction components except the substrate and were amended with either natural-abundance formate or [13C]-formate. Prominent peaks at m/z = 182 for the sample containing natural-abundance formate (green bars) and m/z = 183 for the sample with H13CO2− (purple bars) demonstrate that, as expected, the coupling reaction does result in partial exchange of the formate O-atoms with solvent. Comparison of the integrated intensities of the peaks corresponding to the 16O- and 18O-containing formyl-2NPH isotopologues gave 15% of the latter species, which corresponds to 20% when the 18O content of 70% is taken into account. This extent of exchange fully accounts for the diminution of the heavier isotope from the theoretical maximum of 50% in the 18O2 experiments in Figure 1B. These results show that (i) O2 is the source of the incorporated oxygen and (ii) to the limit of our analysis, there is no demonstrable exchange with solvent during the enzyme reaction. The AD reaction is thus the first example (of which we are aware) of substrate oxygenation without net oxidation.
The metal ions in the AD cofactor have not yet been identified. Nevertheless, the vast majority of proteins in this structural family that have been studied to date have di-iron clusters. In the reactions of several of these enzymes, (μ-peroxo)-Fe2III/III intermediates have been identified.26–31 These intermediates either undergo O-O-bond cleavage to give high-valent (Fe2III/IV or Fe2IV/IV) complexes that effect oxidation reactions or directly react as electrophiles (e.g., via attack of the π-electrons of an olefinic or aromatic substrate or the nitrogen of an amine or hydroxylamine), also leading to oxidative outcomes. However, studies of heme enzymes (cytochromes P-450) and inorganic complexes have shown that metal-bound peroxides can also act as nucleophiles to attack (among other electrophiles) aldehydes, leading to production of formate and oxidized co-products.32–38 Moreover, recent computational studies by Hirao and Morokuma on myo-inositol oxygenase and hydroxyethylphosphonate dioxygenase suggest that, in each reaction, the nucleophilic attack of an Fe-coordinated peroxide on the carbonyl of a reaction intermediate may be a key step in effecting C-C-bond cleavage.39,40 These reactions would provide precedent for the early steps of the hypothetical AD mechanism shown in Scheme 2. It involves addition of O2 to the reduced Fe2II/II cofactor (A) to form a peroxide intermediate (B) and attack of the peroxide on the C1 carbonyl of the substrate to form a peroxyhemiacetal complex (C). Breakdown of this intermediate leading to O-O and C1-C2 scission would explain production of formate with one O-atom from O2. The intriguing and (to date) unprecedented aspect of this hypothetical mechanism would be breakdown of the peroxyhemiacetal intermediate to convert C2 of the substrate into a fully reduced (methyl) rather than partially oxidized (e.g, alcohol or olefinic methylene) center in the R-H product. One possibility, shown in Scheme 2, is that the reducing system would deliver an electron during cleavage of the O-O bond, forming a gem-diolyl radical and (μ-oxo)-Fe2III/III cluster (D). The gem-diolyl intermediate could undergo radical fragmentation of the C1-C2 bond, generating formate and the R• radical (E). Transfer of a hydrogen atom, either from the cofactor (as depicted) or from an amino acid in the active site, would produce R-H and either a (μ-oxo)-Fe2III/IV complex (analogous to the X intermediate that generates the tyrosyl radical in a class I RNR41,42) or an amino acid radical. The reducing system would then complete the reaction by quenching this remaining oxidizing equivalent, and subsequently deliver two more electrons to convert the Fe2III/III “product” form of the cofactor (F) back to the O2-reactive Fe2II/II “reactant” state (A) in preparation for the next turnover.
Scheme 2.
Hypothetical Mechanism for the Np AD Reaction
The mechanism in Scheme 2 accounts for all the available data of which we are aware, including the requirement for a reducing system and the continuous presence of O2, the identity of the C1-derived co-product (formate),4 the origin of the new O-atom in the formate, and the origins of the hydrogen atoms in both formate and R-H products.4 It also predicts an NADPH:formate (NADPH:R-H) reaction stoichiometry of 2:1 in order to account for the four electrons needed in each turnover for complete reduction of O2 (Scheme 1B). We tested this prediction by spectrophotometric determination of NADPH consumption and parallel quantification of formate at two reaction times (Figure S1). Experimental NADPH:formate ratios of 5.6 after 1 h and 6.7 after 2 h were calculated from the results. These ratios are sufficiently large to be consistent with the mechanism of Scheme 2 and also imply considerable uncoupling of NADPH consumption from R-CHO cleavage. This uncoupling is unsurprising, given that the reaction is so slow and that we are employing a heterologous reducing system (ferredoxin and ferredoxin reductase from spinach) to transfer the electrons from the reduced nicotinamide to the AD cofactor. It should be more informative to re-determine this ratio with the native reducing system, once it has been identified.
The mechanism of Scheme 2 implies that the cyanobacterial AD cofactor could indeed be a Fe2 cluster, as originally suggested by Schirmer, et al.3 However, the unprecedented aspects of the mechanism leave room for the possibility that a different transition metal might be present at one or both sites. Such a substitution might, for example, permit formation of a peroxyhemiacetal intermediate less disposed toward an oxidative outcome than the depicted Fe2 complex is expected to be on the basis of the cited precedents. Similarly, mechanisms in which a metal-bound superoxide attacks C1 can also be formulated. In this case, a mixed-metal cluster might disfavor further reduction of the O2 unit to the peroxide state and give the superoxo complex sufficient time to add to the carbonyl. Clearly, identification of the metals in the cofactor is urgently needed for a deeper understanding of this novel reaction.
Supplementary Material
Acknowledgments
This work was supported by the National Institutes of Health (GM-63847 to S.J.B.) and the Dreyfus Foundation (Teacher-Scholar Award to C.K.).
Footnotes
Supporting Information Available: Figure illustrating determination of the ratio of NADPH oxidized to formate produced in the Np AD reaction. This material is available free of charge via the internet at http://pubs.acs.org
References
- 1.Bryant DA. The Molecular Biology of Cyanobacteria. Kluwer Academic Publishers; Dordrecht: 1994. [Google Scholar]
- 2.Ducat DC, Way JC, Silver PA. Trends in Biotechnology. 2011;29:95–103. doi: 10.1016/j.tibtech.2010.12.003. [DOI] [PubMed] [Google Scholar]
- 3.Schirmer A, Rude MA, Li X, Popova E, del Cardayre SB. Science. 2010;329:559–562. doi: 10.1126/science.1187936. [DOI] [PubMed] [Google Scholar]
- 4.Warui DM, Li N, Nørgaard H, Krebs C, Bolllinger JM, Jr, Booker SJ. J Am Chem Soc. 2011;133:3316–3319. doi: 10.1021/ja111607x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schirmer A, Rude M, Brubaker S. LS9. WO/2009/140696 patent application. 2009
- 6.Reppas NB, Ridley CP. Joule Unlimited Inc; US: 7794969 patent. 2010
- 7.Service RF. Science. 2009;325:379. doi: 10.1126/science.325_379a. [DOI] [PubMed] [Google Scholar]
- 8.Abbreviations: 2NPH, 2-nitrophenyl-hydrazide; AD, aldehyde decarbonylase; Ec, Escherichia coli; EDC, 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide; GC, gas chromatography; LC, liquid chromatography; MS, mass spectrometry; N/F/FR, NADPH, ferredoxin, ferredoxin reductase reducing system; Np, Nostoc punctiforme; Pm, Prochlorococcus marinus MIT9313; R, n-C17H35; SIM, single ion monitoring.
- 9.Merkx M, Kopp DA, Sazinsky MH, Blazyk JL, Müller J, Lippard SJ. Angew Chem Int Ed. 2001;40:2782–2807. doi: 10.1002/1521-3773(20010803)40:15<2782::AID-ANIE2782>3.0.CO;2-P. [DOI] [PubMed] [Google Scholar]
- 10.Wallar BJ, Lipscomb JD. Chem Rev. 1996;96:2625–2657. doi: 10.1021/cr9500489. [DOI] [PubMed] [Google Scholar]
- 11.Fox BG, Lyle KS, Rogge CE. Acc Chem Res. 2004;37:421–429. doi: 10.1021/ar030186h. [DOI] [PubMed] [Google Scholar]
- 12.Krebs C, Bollinger JM, Jr, Booker SJ. Curr Opin Chem Biol. 2011 doi: 10.1016/j.cbpa.2011.02.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nordlund P, Sjöberg BM, Eklund H. Nature. 1990;345:593–598. doi: 10.1038/345593a0. [DOI] [PubMed] [Google Scholar]
- 14.Nordlund P, Eklund H. Curr Opin Struct Biol. 1995;5:758–66. doi: 10.1016/0959-440x(95)80008-5. [DOI] [PubMed] [Google Scholar]
- 15.Atkin CL, Thelander L, Reichard P, Lang G. J Biol Chem. 1973;248:7464–7472. [PubMed] [Google Scholar]
- 16.Sjöberg BM, Reichard P, Gräslund A, Ehrenberg A. J Biol Chem. 1977;252:536–541. [PubMed] [Google Scholar]
- 17.Stubbe J. Curr Opin Chem Biol. 2003;7:183–188. doi: 10.1016/s1367-5931(03)00025-5. [DOI] [PubMed] [Google Scholar]
- 18.Cotruvo JA, Stubbe J. Biochemistry. 2010;49:1297–1309. doi: 10.1021/bi902106n. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cox N, Ogata H, Stolle P, Reijerse E, Auling G, Lubitz W. J Am Chem Soc. 2010;132:11197–11213. doi: 10.1021/ja1036995. [DOI] [PubMed] [Google Scholar]
- 20.Jiang W, Yun D, Saleh L, Barr EW, Xing G, Hoffart LM, Maslak MA, Krebs C, Bollinger JM., Jr Science. 2007;316:1188–1191. doi: 10.1126/science.1141179. [DOI] [PubMed] [Google Scholar]
- 21.Jiang W, Yun D, Saleh L, Bollinger JM, Jr, Krebs C. Biochemistry. 2008;47:13736–13744. doi: 10.1021/bi8017625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bollinger JM, Jr, Jiang W, Green MT, Krebs C. Curr Opin Struct Biol. 2008;18:650–657. doi: 10.1016/j.sbi.2008.11.007. [DOI] [PubMed] [Google Scholar]
- 23.Stubbe J. Advances in enzymology and related areas of molecular biology. Vol. 63. Wiley; 1990. pp. 349–419. [DOI] [PubMed] [Google Scholar]
- 24.Jiang W, Xie J, Varano PT, Krebs C, Bollinger JM., Jr Biochemistry. 2010;49:5340–5349. doi: 10.1021/bi100037b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wu CH, Jiang W, Krebs C, Stubbe J. Biochemistry. 2007;46:11577–11588. doi: 10.1021/bi7012454. [DOI] [PubMed] [Google Scholar]
- 26.Liu KE, Wang D, Huynh BH, Edmondson DE, Salifoglou A, Lippard SJ. J Am Chem Soc. 1994;116:7465–7466. [Google Scholar]
- 27.Tong WH, Chen S, Lloyd SG, Edmondson DE, Huynh BH, Stubbe J. J Am Chem Soc. 1996;118:2107–2108. [Google Scholar]
- 28.Yun D, García-Serres R, Chicalese BM, An YH, Huynh BH, Bollinger JM., Jr Biochemistry. 2007;46:1925–1932. doi: 10.1021/bi061717n. [DOI] [PubMed] [Google Scholar]
- 29.Murray LJ, Naik SG, Ortillo DO, García-Serres R, Lee JK, Huynh BH, Lippard SJ. J Am Chem Soc. 2007;129:14500–14510. doi: 10.1021/ja076121h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Korboukh VK, Li N, Barr EW, Bollinger JM, Jr, Krebs C. J Am Chem Soc. 2009;131:13608–13609. doi: 10.1021/ja9064969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Li N, Korboukh VK, Krebs C, Bollinger JM., Jr Proc Natl Acad Sci USA. 2010;107:15722–15727. doi: 10.1073/pnas.1002785107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.LeCloux DD, Barrios AM, Lippard SJ. Bioorg Med Chem. 1999;7:763–772. doi: 10.1016/s0968-0896(98)00270-3. [DOI] [PubMed] [Google Scholar]
- 33.Wertz DL, Sisemore MF, Selke M, Driscoll J, Valentine JS. J Am Chem Soc. 1998;120:5331–5332. [Google Scholar]
- 34.Wertz DL, Valentine JS. Metal-Oxo and Metal-Peroxo Species in Catalytic Oxidations. Vol. 97. Springer-Verlag Berlin; Berlin: 2000. pp. 37–60. [Google Scholar]
- 35.Annaraj J, Suh Y, Seo MS, Kim SO, Nam W. Chem Comm. 2005:4529–4531. doi: 10.1039/b505562h. [DOI] [PubMed] [Google Scholar]
- 36.Cho J, Sarangi R, Annaraj J, Kim SY, Kubo M, Ogura T, Solomon EI, Nam W. Nature Chemistry. 2009;1:568–572. doi: 10.1038/nchem.366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Annaraj J, Cho JH, Lee YM, Kim SY, Latifi R, de Visser SP, Nam W. Angew Chem - Int Ed. 2009;48:4150–4153. doi: 10.1002/anie.200900118. [DOI] [PubMed] [Google Scholar]
- 38.Cho J, Sarangi R, Kang HY, Lee JY, Kubo M, Ogura T, Solomon EI, Nam W. J Am Chem Soc. 2010;132:16977–16986. doi: 10.1021/ja107177m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hirao H, Morokuma K. J Am Chem Soc. 2009;131:17206–17214. doi: 10.1021/ja905296w. [DOI] [PubMed] [Google Scholar]
- 40.Hirao H, Morokuma K. J Am Chem Soc. 2010;132:17901–17909. doi: 10.1021/ja108174d. [DOI] [PubMed] [Google Scholar]
- 41.Bollinger JM, Jr, Edmondson DE, Huynh BH, Filley J, Norton JR, Stubbe J. Science. 1991;253:292–298. doi: 10.1126/science.1650033. [DOI] [PubMed] [Google Scholar]
- 42.Sturgeon BE, Burdi D, Chen S, Huynh BH, Edmondson DE, Stubbe J, Hoffman BM. J Am Chem Soc. 1996;118:7551–7557. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.