Abstract
OBJECTIVE
The study objective was to compare dietary patterns in their relationship with metabolic risk factors (MRFs) and the metabolic syndrome (MetS).
RESEARCH DESIGN AND METHODS
Cross-sectional analysis of 773 subjects (mean age 60 years) from the Adventist Health Study 2 was performed. Dietary pattern was derived from a food frequency questionnaire and classified as vegetarian (35%), semi-vegetarian (16%), and nonvegetarian (49%). ANCOVA was used to determine associations between dietary pattern and MRFs (HDL, triglycerides, glucose, blood pressure, and waist circumference) while controlling for relevant cofactors. Logistic regression was used in calculating odds ratios (ORs) for MetS.
RESULTS
A vegetarian dietary pattern was associated with significantly lower means for all MRFs except HDL (P for trend < 0.001 for those factors) and a lower risk of having MetS (OR 0.44, 95% CI 0.30–0.64, P < 0.001) when compared with a nonvegetarian dietary pattern.
CONCLUSIONS
A vegetarian dietary pattern is associated with a more favorable profile of MRFs and a lower risk of MetS. The relationship persists after adjusting for lifestyle and demographic factors.
The metabolic syndrome (MetS) is a cluster of disorders that are associated with a heightened risk of diabetes and cardiovascular disease (1). Previous studies have reported associations between major dietary patterns and MetS (2). However, no agreement is found as to which dietary patterns would confer the lowest risk of MetS (2). It is thus the aim of this report to analyze the relationship between dietary patterns defined by degree of animal food intake and the prevalence of MetS.
RESEARCH DESIGN AND METHODS
This report includes subjects from a substudy of the Adventist Health Study 2. In brief, subjects (n = 1,011) with a mean age of 60 years (range 30–94 years) were randomly selected from the 96,000 enrollees of the Adventist Health Study 2. Subjects were required to attend a clinic during which weight and height were measured and fasting blood samples were collected by trained staff. The methods have been described by Chan et al. (3) and Jaceldo-Siegl et al. (4). Height was measured to the nearest 1/4 inch (0.64 cm) with the subject standing erect and barefoot using a Seca 214 Portable Height Rod (Seca Corp., Hamburg, Germany), and weight was measured to the nearest 0.1 kg with the subject wearing light clothing without shoes and socks using the Tanita BF-350 (Tanita UK Ltd., Middlesex, U.K.). Waist circumference was measured with an anthropometric tape 1 inch above the navel. The measurements were repeated three times, and the mean of the values was used for further calculations. BMI was calculated as weight/height squared (kilograms/meters squared). Clinic staff obtained fasting blood glucose, cholesterol, LDL-cholesterol, HDL-cholesterol, and triglyceride concentrations via finger stick using the Cholestech LDX System (Cholestech, Hayward, CA) (5). Three blood pressure measurements were obtained using the Omron Automatic Digital Blood Pressure Monitor HEM-747IC (Omron Healthcare, Inc., Vernon Hills, IL) (6), and the mean value was used for analysis. The Adult Treatment Panel III 2001 (7) definition for identifying the MetS was used with cutoff levels for impaired fasting plasma glucose set at ≥100 mg/dL, as suggested by the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus in 2003 (8). Subjects taking antihypertensive (27%) or diabetes (8%) medication were considered to indicate the presence of the respective risk factor.
Dietary intake was obtained using a quantitative, self-administered food frequency questionnaire. Validity and detailed description of the computation of the dietary data were previously reported (4,9). For all meat, poultry, fish, and dairy validity, correlations were >0.80 and 0.58 for egg intake (10).
Type of diet was categorized by defining vegetarians as subjects who reported consuming meat, poultry, or fish <1 time/month. Semi-vegetarians were defined as consuming fish at any frequency but consuming other meats <1 time/month or total meat (with red meat and poultry ≥1 time/month and <1 time/week). Nonvegetarians were defined as consuming red meat or poultry ≥1 time/month and the total of all meats ≥1 time/week. A modification of the Blair Seven-Day Physical Activity Recall (11) was used to obtain measures of physical activity. Subjects were interviewed by telephone and asked to recall time spent in light, moderate, hard, and very hard activities during the previous 7 days. Alcohol intake was defined as consumption of any amount or none during the previous 12 months. Tobacco use was defined as ever smoking any amount of tobacco or none.
Statistical analysis
Subjects with a complete set of clinical and nondietary covariate data were included in the analysis (n = 773). Imputation of missing dietary data used guided multiple imputation (12). In the descriptive analyses, differences in covariate values by type of dietary pattern (vegetarian, semi-vegetarian, nonvegetarian) were assessed by ANOVA and by χ2 tests for categoric data.
ANCOVA was used in comparing metabolic risk factors (MRFs) (HDL, triglycerides, glucose, blood pressure, waist circumference) and BMI stratified by types of dietary patterns. Values for triglycerides and glucose were logarithmically transformed for analysis because of skewness, and exponentiated values are shown. Logistic regression analysis was used to compute multivariable-adjusted odds ratios (ORs) and 95% CIs for the association of dietary patterns with MetS. Nonvegetarian dietary pattern was used as the reference group. Adjustments were made for age, sex, ethnicity, smoking, alcohol intake, physical activity, and dietary energy intake in the ANCOVA and logistic regression analysis.
Analysis was carried out with IBM SPSS Statistics18.01 (SPSS Inc., an IBM Company, Chicago, IL) and with R 2.10.1: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, Vienna, Austria). The type I error rate was set at 0.05.
RESULTS
Vegetarians and semi-vegetarians were on average 3 years older than nonvegetarians. Height was not significantly different among the three dietary patterns. BMI was lowest in vegetarians (25.7 kg/m2), intermediate in semi-vegetarians (27.6 kg/m2), and highest in nonvegetarians (29.9 kg/m2).
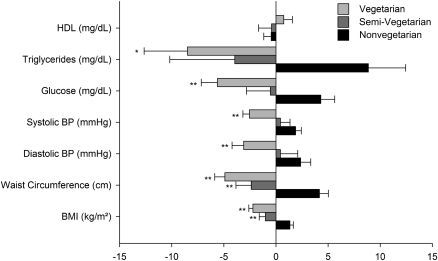
The dietary patterns had marked differences in the prevalence of MRFs that were over the risk threshold: vegetarians had 12% with three factors, 8% with four factors, and 3% with five factors. Semi-vegetarians had 19% with three factors, 10% with four factors, and 2% with five factors. Nonvegetarians had 19% with three factors, 13% with four factors, and 5% with five factors. After adjusting for age, sex, ethnicity, smoking, alcohol intake, physical activity, and dietary energy intake, the results showed (Fig. 1) that triglycerides, glucose, blood pressure levels, waist circumference, and BMI were significantly lower (P < 0.05) in vegetarians than in nonvegetarians. Semi-vegetarians had significantly lower waist circumference and BMI (P < 0.001) than nonvegetarians. Additional adjustment of these variables for BMI showed that the results remained significantly lower in vegetarians for glucose and diastolic blood pressure (P < 0.05).
Figure 1.
Dietary pattern and MRFs. Mean-centered values are shown with whiskers representing SE. ANCOVA was used in comparing risk factors stratified by dietary patterns. P values represent contrasts with nonvegetarian dietary pattern, which is the group of reference. *P < 0.05; **P < 0.001. P for trend was < 0.001 for each risk factor with the exception of HDL (P > 0.05). Adjustments were made for age, sex, ethnicity, smoking, alcohol intake, physical activity, and dietary energy intake. BP, blood pressure.
The MetS was highest in nonvegetarians (39.7%), intermediate in semi-vegetarians (37.6%), and lowest in vegetarians (25.2%) (P for trend < 0.001). Significant differences remained after adjustments for sex, ethnicity, smoking, alcohol intake, physical activity, and dietary energy intake. Those adhering to a vegetarian dietary pattern had an OR for MetS of 0.44 (95% CI 0.30–0.64, P < 0.001) when compared with nonvegetarians.
CONCLUSIONS
This report showed that a vegetarian dietary pattern is associated with a more favorable profile of MRFs and a lower risk of MetS. This favorable relationship persisted and was considerably strengthened when adjusting for possible confounding factors. Our results thus confirm and build on previous studies on vegetarian diets and metabolic risk (13–16), and suggest that a vegetarian dietary pattern can play a favorable role in lowering the risk of MetS.
Acknowledgments
This work was supported by National Institutes of Health Grant 1R01-CA-94594. N.S.R. was supported by the McClean Postdoctoral Fellowship Grant.
No potential conflicts of interest relevant to this article were reported.
N.S.R. conceived the hypothesis, conducted statistical analyses, and wrote the article. J.S. reviewed the article and contributed to discussion. K.J.-S. contributed to data collection and the research design and methods section. G.E.F. reviewed the article and contributed to discussion.
References
- 1.Ford ES. Risks for all-cause mortality, cardiovascular disease, and diabetes associated with the metabolic syndrome: a summary of the evidence. Diabetes Care 2005;28:1769–1778 [DOI] [PubMed] [Google Scholar]
- 2.Djoussé L, Padilla H, Nelson TL, Gaziano JM, Mukamal KJ. Diet and metabolic syndrome. Endocr Metab Immune Disord Drug Targets 2010;10:124–137 [DOI] [PubMed] [Google Scholar]
- 3.Chan J, Knutsen SF, Sabate J, Haddad E, Yan R, Fraser GE. Feasibility of running clinics to collect biological specimens in a nationwide cohort study—Adventist Health Study-2. Ann Epidemiol 2007;17:454–457 [DOI] [PubMed] [Google Scholar]
- 4.Jaceldo-Siegl K, Knutsen SF, Sabaté J, et al. Validation of nutrient intake using an FFQ and repeated 24 h recalls in black and white subjects of the Adventist Health Study-2 (AHS-2). Public Health Nutr 2010;13:812–819 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bachorik PS, Rock R, Cloey T, Treciak E, Becker D, Sigmund W. Cholesterol screening: comparative evaluation of on-site and laboratory-based measurements. Clin Chem 1990;36:255–260 [PubMed] [Google Scholar]
- 6.Anwar YA, Giacco S, McCabe EJ, Tendler BE, White WB. Evaluation of the efficacy of the Omron HEM-737 IntelliSense device for use on adults according to the recommendations of the Association for the Advancement of Medical Instrumentation. Blood Press Monit 1998;3:261–265 [PubMed] [Google Scholar]
- 7.Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III). JAMA 2001;285:2486–2497 [DOI] [PubMed] [Google Scholar]
- 8.Genuth S, Alberti KG, Bennett P, et al. ; Expert Committee on the Diagnosis and Classification of Diabetes Mellitus Follow-up report on the diagnosis of diabetes mellitus. Diabetes Care 2003;26:3160–3167 [DOI] [PubMed] [Google Scholar]
- 9.Tonstad S, Butler T, Yan R, Fraser GE. Type of vegetarian diet, body weight, and prevalence of type 2 diabetes. Diabetes Care 2009;32:791–796 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jaceldo-Siegl K, Fan J, Sabaté J, et al. Race-specific validation of food intake obtained from a comprehensive food frequency questionnaire: Adventist Health Study-2. Public Health Nutr. In press [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Blair SN, Haskell WL, Ho P, et al. Assessment of habitual physical activity by a seven-day recall in a community survey and controlled experiments. Am J Epidemiol 1985;122:794–804 [DOI] [PubMed] [Google Scholar]
- 12.Fraser G, Yan R. Guided multiple imputation of missing data: using a subsample to strengthen the missing-at-random assumption. Epidemiology 2007;18:246–252 [DOI] [PubMed] [Google Scholar]
- 13.De Biase SG, Fernandes SF, Gianini RJ, Duarte JL. Vegetarian diet and cholesterol and triglycerides levels. Arq Bras Cardiol 2007;88:35–39 [DOI] [PubMed] [Google Scholar]
- 14.Sebeková K, Boor P, Valachovicová M, et al. Association of metabolic syndrome risk factors with selected markers of oxidative status and microinflammation in healthy omnivores and vegetarians. Mol Nutr Food Res 2006;50:858–868 [DOI] [PubMed] [Google Scholar]
- 15.Teixeira Rde C, Molina Mdel C, Zandonade E, Mill JG. Cardiovascular risk in vegetarians and omnivores: a comparative study. Arq Bras Cardiol 2007;89:237–244 [DOI] [PubMed] [Google Scholar]
- 16.Toohey ML, Harris MA, DeWitt W, Foster G, Schmidt WD, Melby CL. Cardiovascular disease risk factors are lower in African-American vegans compared to lacto-ovo-vegetarians. J Am Coll Nutr 1998;17:425–434 [DOI] [PubMed] [Google Scholar]