Abstract
Present work on indole-3-acetic acid (IAA)-induced adventitious rooting in sunflower hypocotyl highlights a clear demarcation of nitric oxide (NO)-dependent and NO-independent roles of auxin in this developmental process. Of the three phases of adventitious rooting, induction is strictly auxin-dependent though initiation and extension are regulated by an interaction of IAA with NO. A vital role of auxin-efflux transporters (PIN) is also evident from 1-napthylphthalamic acid (NPA)-triggered suppression of adventitious roots (AR). Use of actin depolymerizing agent, latrunculin B (Lat B), has demonstrated the necessity of functional actin filaments in auxin-induced AR response, possibly through its effect on actin-mediated recycling of auxin transporter proteins. Thus, evidence for a linkage between IAA, NO and actin during AR formation has been established.
Key words: adventitious roots, auxin, sunflower
Adventitious roots (AR) are post-embryonic roots known to originate from stem, leaf petiole and non-pericycle tissue of old roots. In young stem, AR commonly arise from the interfascicular parenchyma while they appear from vascular rays near the cambium in older stem. Formation of AR begins with re-differentiation of predetermined cells which switch from their morphogenetic path to act as mother cells for initiation of root primordia.1 The process of AR formation consists of three physiologically interdependent phases: induction, initiation and extension.1 Induction phase comprises of various molecular and biochemical events but no morphologically visible changes appear during this phase. Formation of multilayered cells and conception of root primordia occurs during initiation phase. During expression phase, root primordia exhibit intra-stem growth and their emergence through epidermis. Various environmental and endogenous factors, such as temperature, light, hormones (particularly auxin), sugars and mineral salts, act as cues for promoting redifferentiation of predetermined cells resulting in root induction.
The three phases of AR formation are known to be regulated by alterations in the endogenous level of auxin.2 A transient increase in auxin concentration has been reported during induction phase, which is followed by a decrease and again an increase during expression phase.3 Auxin transport to and from the responding region is essential for root organogenesis. Acropetal transport of auxin occurs through the vascular cylinder and the basipetal transport takes place through the epidermal and subtending cortical cells.4 Polar transport of auxin from the shoot apical meristem to the rooting region is primarily facilitated by auxin-influx (AUX1) and -efflux (PIN) transporters. Asymmetric localization of these transporter proteins in the vascular cambium cells is responsible for differential distribution of auxin in a particular zone of cells.5 Polar auxin transport is known to be inhibited by 1-napthylphthalamic acid (NPA, a phytotropin). This inhibition is mediated through a binding of NPA molecule to putative NPA-binding protein (NBP), which is functionally associated with PIN proteins.6 Efflux transporters exhibit rapid turnover in plasma membrane.7 High affinity of NBP for actin filaments,8,9 suggests its involvement in the cycling and polar distribution of PIN proteins.10 Organization of actin filaments is known to be rapidly, reversibly and specifically disrupted by Latrunculin B (Lat B), a macrolide toxin isolated from Latrunculia magnificia, a red sea sponge.11 Lat B associates with actin monomers in 1:1 ratio, thereby preventing their repolymerization into filaments, resulting in a complete shift from F-actin to G-actin.12 Owing to its well-understood and simple mode of action and low effective dosage, Lat B has supplanted the classic actin-depolymerizing drug cytochalasin D13 in pharmacological investigations. In the past few years, significant work has been done on NO as a signaling molecule in a variety of plant developmental processes.14 Nitric oxide is known to play a crucial role in root development.15
Using sunflower as a model system, present work has been undertaken to investigate the possible role of NO during IAA-induced adventitious rooting in hypocotyl explants. Since auxin action is principally based on PIN-regulated polar transport of IAA molecules, and PIN proteins are known to exhibit actin-asssisted rapid recycling in the target cells, attempts have been made in the present work to find a correlation between auxin transport, actin and NO, using specific pharmacological agents. Additionally, effect of Cyclosporin A (CsA), an inhibitor of nitric oxide synthase (NOS) in animal systems,16 has also been investigated. These specific agents have been used to monitor root initiation at the target sites in our attempts to decipher a signaling cascade for AR.
Seeds of Sunflower (Helianthus annuus L. cv. Morden) were germinated on moist germination sheets at 25 ± 2°C under continuous illumination of 4.3 Wm−2. Hypocotyls from 4 d old seedlings and with similar growth rate, were excised up to 6 cm below the cotyledonary node. Hypocotyl explants with apical meristem intact but cotyledons excised were selected for the present work with a view to provide a continuity of the endogenous auxin source. Similar explants were also recently used by Huang et al.17 to investigate indole-3-butyric acid (IBA)-induced AR formation in mung bean (Phaseolus radiatus L.). Using IAA instead of IBA for such investigations was preferred for the present work keeping in view that the two auxins seem to employ different transport proteins for their polar transport.18 Freshly harvested explants were put upright in glass vials with their proximal cut ends dipped in 1 ml of different concentrations (1, 5, 10 and 15 µM) of IAA, thus bathing the hypocotyls up to 5 mm of their lower ends. Explants were maintained in dark during the course of experiments. The number of AR visible on hypocotyl surface was recorded daily up to 4 days of incubation. Concentration of IAA thus observed optimal for rooting (10 µM) was used for all further experiments. Similarly, various concentrations of other test solutions, namely NPA (auxin efflux blocker; 1 and 10 µM), Lat B (an inducer of actin depolymerization; 25, 50 and 100 nM), CsA (an inhibitor of cyclophilins; 1, 5 and 10 µM), sodium nitroprusside (SNP; NO donor; 1, 5, 10 and 100 µM) and 2-phenyl-4,4,5,5-tetramethyllimidazoline-1-oxyl-3-oxide (PTIO; NO scavenger; 1 and 1.5 mM), were initially used to select their respective optimal concentrations. Based on these preliminary experiments NPA, Lat B, CsA, SNP and PTIO were used at 10 µM, 100 nM, 10 µM, 100 µM and 1.5 mM, respectively, for all subsequent experiments. Some other treatment combinations, namely NPA (10 µM) + IAA (10 µM); LatB (100 nM) + IAA (10 µM); CsA (10 µM) + IAA (10 µM); SNP (100 µM) + NPA (10 µM) and PTIO (1.5 mM) + IAA (10 µM) were also used to investigate their effects on adventitious rooting. Hypocotyl explants incubated in distilled water served as control. Morphological observations of rooting response were imaged after 7 days of incubation, using Nikon digital camera fitted on a stereomicroscope (Stemi 2000, Zeiss, Germany). Detailed evaluation of root initiation was observed after clearing by immersing the explants in a 3:1 solution of ethanol: acetic acid overnight. They were then transferred to 2 N NaOH solution, left overnight, washed once with distilled water and stained with safranin solution for 2–3 min. Excess stain was removed by repeated washing in distilled water. The lower 2 cm region of hypocotyl explants was then cut and mounted on a glass slide to examine endogenous root initials, using a stereomicroscope (Stemi 2000, Zeiss, Germany) fitted with a Nikon camera.
Root initiation and extension in the basal region of hypocotyl explants maintained in distilled water indicates the expected basipetal transport of the inducing factor (endogenous IAA) from the intact meristem, as also reported earlier.19 Treatment with IAA (10 µM) elicited two effects on hypocotyl explants in comparison to those subjected to distilled water treatment: (1) Formation of greater number of root initials, (2) Greater extension of the initiated roots (Figs. 1 and 2). A response similar to that evoked by IAA is also evident in hypocotyl explants treated with 100 µM of SNP (Figs. 1 and 2). Recently SNP (NO donor) has been reported to evoke dose-dependent response on AR formation in marigold.20 In the present work, treatment with variable concentrations of SNP, ranging from 1–100 µM, lead to a gradual increase in the number and extension growth of AR till 100 µM. Pagnussat et al.21 and Liao et al.20 have used 10 µM and 50 µM as effective SNP concentration in cucumber and marigold, respectively. Thus, optimal concentration of SNP for AR formation is species-dependent. In presence of PTIO (1.5 mM; a specific NO scavenger), complete suppression of AR was evident in sunflower, as also reported earlier in mung bean.17 Combination of PTIO with IAA lead to root initiation only (no extension growth). NPA (10 µM) blocked AR initiation by endogenous (distilled water treatment) and exogenous IAA (Fig. 1). Application of NPA inhibits polar auxin transport, thus reducing the optimal concentration of IAA required for AR formation at the hypocotyl base (zone of AR formation). Thus, no evidence of root initials was evident in presence of NPA, which is also reported in cucumber22 and loblolly pine,23 respectively. Though NO is expected to act downstream of IAA24 but a treatment of SNP in combination with NPA (present work) lead to complete suppression of AR formation. Our unpublished observations have indicated the expression of NO in the interfascicular cells after induction phase (i.e., during AR iniation and extension). Thus, it can be proposed that IAA is involved in induction phase of adventitious rooting independent of NO, while initiation and extension phases appear to involve IAA-NO interaction. CsA-cyclophilin complex is known to inhibit calcineurin (a protein phosphatase) and NOS activity in animal systems.25 Treatment of hypocotyl explants with CsA (10 µM) lead to formation of fewer number of roots which exhibited extension growth. Oh et al.26 reported a significant reduction in the number of roots in the presence of CsA in hypocotyl explants from tomato. Subjecting hypocotyl explants with a combination of CsA and IAA lead to formation of fewer number of root initials, reaffirming the involvement of NO in auxin action in the developmental process under investigation (AR). However, further investigations on the role of cyclophilins and NOS in auxin-modulated AR formation are required to pinpoint their specific sites of action in this developmental process. Treatment with Lat B (+ and − IAA) lead to complete AR suppression in sunflower hypocotyl explants. Actin-mediated polar localization of PIN proteins is responsible for polar auxin transport and disruption of microfilaments by Lat-B would thus, directly affect IAA transport leading to the observed AR suppression. These observations indicate a convergence of the effect of IAA with that of NO and a role of a well organized actin in the responding cells.
Figure 1.
Effect of IAA and various other pharmacological agents on adventitious rooting in hypocotyl explants. Morphological observations of rooting response (A and C). Evaluation of endogenous root initiation and elongation observed in cleared explants stained with safranin (B and D). Scale bar represents 3 mm.
Figure 2.
Quantitative analysis of AR initiation in presence of distilled water, IAA and various pharmacological agents in hypocotyl explants of sunflower. Each datum presents a mean and standard error from at least three observations.
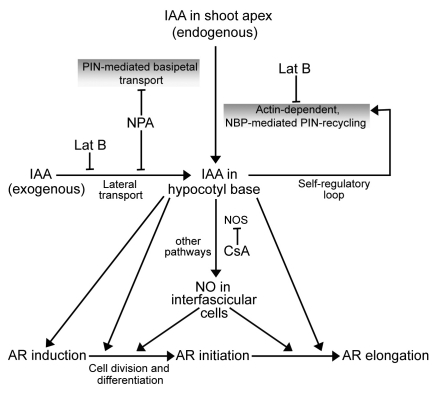
To sum up, present investigations provide evidence for a linkage between auxin-induced AR response in seedling hypocotyls and NO (Fig. 3). Both endogenous and exogenous IAA-mediated AR induction seem to depend on actin. Significance of actin in this developmental response has become evident via its role in the cycling of auxin efflux proteins (PIN). The three phases of AR formation can be differentiated from each other in terms of their sensitivities to IAA and NO. AR induction phase seems to be governed by auxin alone, independent of NO. NO seems to become operative in this auxin-modulated response (AR) during initiation and extension phase only. Investigations are being undertaken in the author's laboratory to visualize and quantitate the NO signal in the IAA-responding hypocotyl explants so that the phasing of the role of NO during AR formation can be precisely predicted.
Figure 3.
Schematic presentation of the probable events associated with NO-mediated adventitious rooting.
Acknowledgements
The photomicroscopy work was undertaken on the microscope provided by Alexander von Humboldt Foundation to S.C.B. Authors are thankful to Mr. S.K. Das for digital photography. This work was supported by CSIR, New Delhi in the form of Junior Research Fellowship to S.Y. and A.D.
Abbreviations
- AR
adventitious roots
- NPA
1-napthylphthalamic acid
- NO
nitric oxide
- NOS
nitric oxide synthase
- IAA
indole-3-acetic acid
- CsA
cyclosporin A
- SNP
sodium nitroprusside
- PTIO
2-phenyl-4,4,5,5-tetramethyllimidazoline-1-oxyl-3-oxide
- Lat B
latrunculin B
Footnotes
Previously published online: www.landesbioscience.com/journals/psb/article/12159
References
- 1.Li SW, Xue L, Xu S, Feng H, An L. Mediators, genes and signalling in adventitious rooting. Bot Rev. 2009;75:230–247. [Google Scholar]
- 2.Bellamine J, Penel C, Greppin H, Gaspar T. Confirmation of the role of auxin and calcium in the late phases of adventitious root formation. Plant Growth Regul. 1998;26:191–194. [Google Scholar]
- 3.Nag S, Saha K, Choudhuri MA. Role of auxin and polyamines in adventitious root formation in relation to changes in compounds involved in rooting. J Plant Growth Regul. 2001;20:182–194. [Google Scholar]
- 4.Jones AM. Auxin transport: down and out and up again. Science. 1998;282:2201–2203. doi: 10.1126/science.282.5397.2201. [DOI] [PubMed] [Google Scholar]
- 5.Taiz L, Zeiger E. Plant Physiology. Fourth Edition. Massachusetts: Sinauer Associates Inc., Publishers; 2006. pp. 467–507. [Google Scholar]
- 6.Morris DA, Friml J, Zažímalová E. Auxin transport. In: Davies PJ, editor. Plant Hormones. Dordrecht, AA: Kluwer Academic Publisher; 2004. pp. 437–470. [Google Scholar]
- 7.Morris DA. Transmembrane auxin carrier systems—dynamic regulators of polar auxin transport. Plant Growth Regul. 2000;32:161–172. doi: 10.1023/a:1010701527848. [DOI] [PubMed] [Google Scholar]
- 8.Muday GK, DeLong A. Polar auxin transport: controlling where and how much. Trends Plant Sci. 2001;6:535–542. doi: 10.1016/s1360-1385(01)02101-x. [DOI] [PubMed] [Google Scholar]
- 9.Muday GK, Murphy AS. An emerging model of auxin transport regulation. Plant Cell. 2002;14:293–299. doi: 10.1105/tpc.140230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Geldner N, Friml J, Stierhof YD, Jürgens G, Palme K. Auxin transport inhibitors block PIN1 cycling and vesicle trafficking. Nature. 2001;413:425–428. doi: 10.1038/35096571. [DOI] [PubMed] [Google Scholar]
- 11.Spector I, Shochet NR, Kashman Y, Groweiss A. Latrunculins: novel marine toxins that disrupt microfilament organization in cultured cells. Science. 1983;219:493–495. doi: 10.1126/science.6681676. [DOI] [PubMed] [Google Scholar]
- 12.Walter MM, Kathryn RA, Paul JM. Latrunculin alters the actin-monomer subunit interface to prevent polymerization. Nat Cell Biol. 2000;2:376–378. doi: 10.1038/35014075. [DOI] [PubMed] [Google Scholar]
- 13.Gibbon BC, Kovar DR, Staiger CJ. Latrunculin B has different effects on pollen germination and tube growth. Plant Cell. 1999;11:2349–2363. doi: 10.1105/tpc.11.12.2349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Moreau M, Lindermayr C, Durner J, Klessig DF. NO synthesis and signaling in plants—where do we stand? Physiol Plant. 2010;138:372–383. doi: 10.1111/j.1399-3054.2009.01308.x. [DOI] [PubMed] [Google Scholar]
- 15.Stöhr C, Stremlau S. Formation and possible roles of nitric oxide in plant roots. J Exp Bot. 2006;57:463–470. doi: 10.1093/jxb/erj058. [DOI] [PubMed] [Google Scholar]
- 16.Kleinert H, Schwarz PM, Förstermann U. Regulation of the expression of inducible nitric oxide synthase. Biol Chem. 2005;384:1343–1364. doi: 10.1515/BC.2003.152. [DOI] [PubMed] [Google Scholar]
- 17.Huang AX, She XP, Huang C, Song TS. The dynamic distribution of NO and NADPH-diaphorase activity during IBA-induced adventitious root formation. Physiol Plant. 2007;130:240–249. [Google Scholar]
- 18.Rashotte AM, Poupart J, Waddell CS, Muday GK. Transport of the two natural auxins, indole-3-butyric acid and indole-3-acetic acid, in Arabidopsis. Plant Physiol. 2003;133:761–772. doi: 10.1104/pp.103.022582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Neuman DS, Hansberry J. Adventitious roots. In: Roberts K, editor. Handbook of Plant Science. West Sussex: John Wiley & sons Ltd; 2007. pp. 72–74. [Google Scholar]
- 20.Liao W, Xiao H, Zhang M. Role and relationship of nitric oxide and hydrogen peroxide in adventitious root development of marigold. Acta Physiol Plant. 2009;31:1279–1289. [Google Scholar]
- 21.Pagnussat GC, Simontacchi M, Puntarulo S, Lamattina L. Nitric oxide is required for root organogenesis. Plant Physiol. 2002;129:954–956. doi: 10.1104/pp.004036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Xuan W, Zhu FY, Xu S, Huang BK, Ling TF, Qi JY, et al. The heme oxygenase/carbon monoxide system is involved in the auxin-induced cucumber adventitious rooting process. Plant Physiol. 2008;148:881–893. doi: 10.1104/pp.108.125567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Diaz-Sala C, Singer PB, Hutchinson KW, Greenwood MS. Molecular approaches to maturation-caused decline in adventitious rooting in loblolly pine (Pinus taeda L.) In: Ahuja MR, Boerjan W, Neale DB, editors. Somatic Cell Genetics and Molecular Genetics of Trees. Dordrecht: Kluwer Academic Publishers; 1996. pp. 57–62. [Google Scholar]
- 24.Pagnussat GC, Lanteri ML, Lamattina L. Nitric oxide and cyclic GMP are messengers in the indole acetic acid-induced adventitious rooting process. Plant Physiol. 2003;132:1241–1248. doi: 10.1104/pp.103.022228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wang P, Heitman J. The cyclophilins. Genome Biol. 2005;6:226. doi: 10.1186/gb-2005-6-7-226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Oh KC, Ivanchenko MG, White TJ, Lomax TL. The diageotropica gene of tomato encodes a cyclophilin: a novel player in auxin signalling. Planta. 2006;224:133–144. doi: 10.1007/s00425-005-0202-z. [DOI] [PubMed] [Google Scholar]