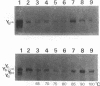
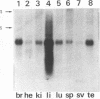
Abstract
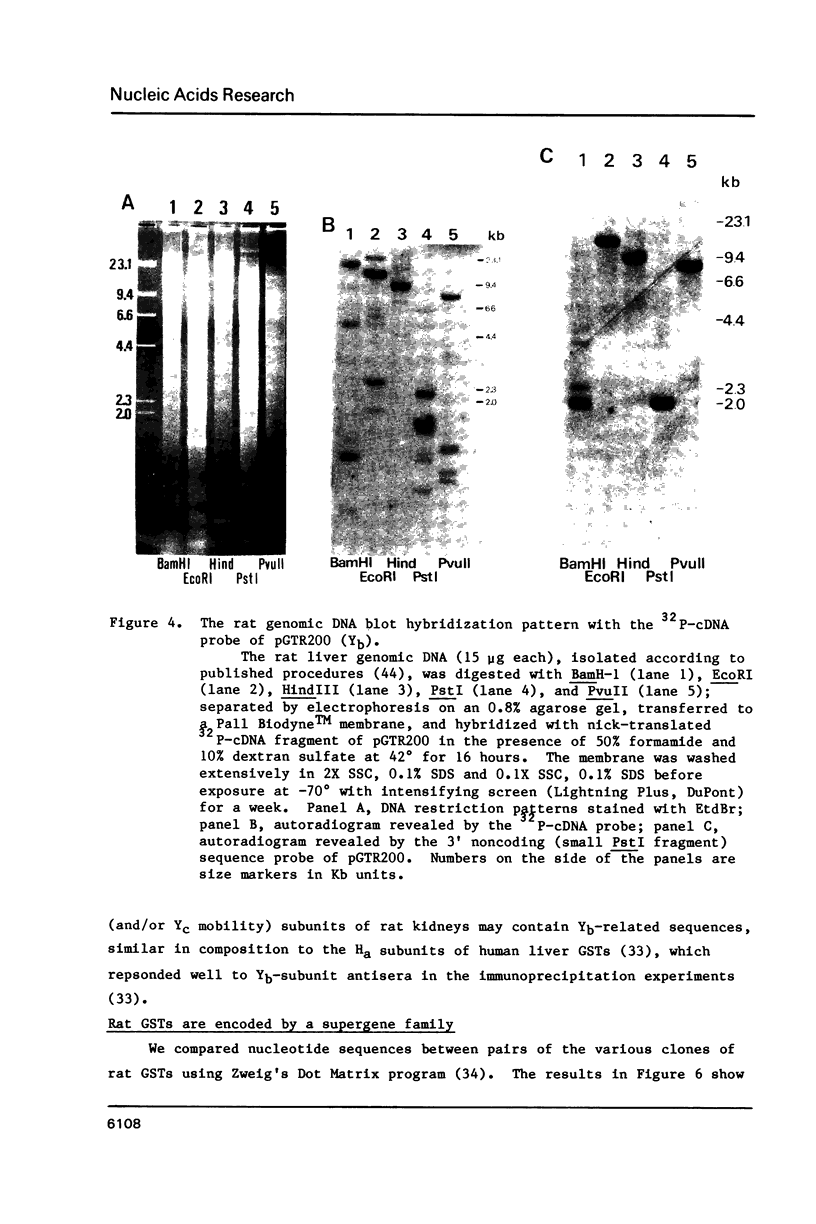
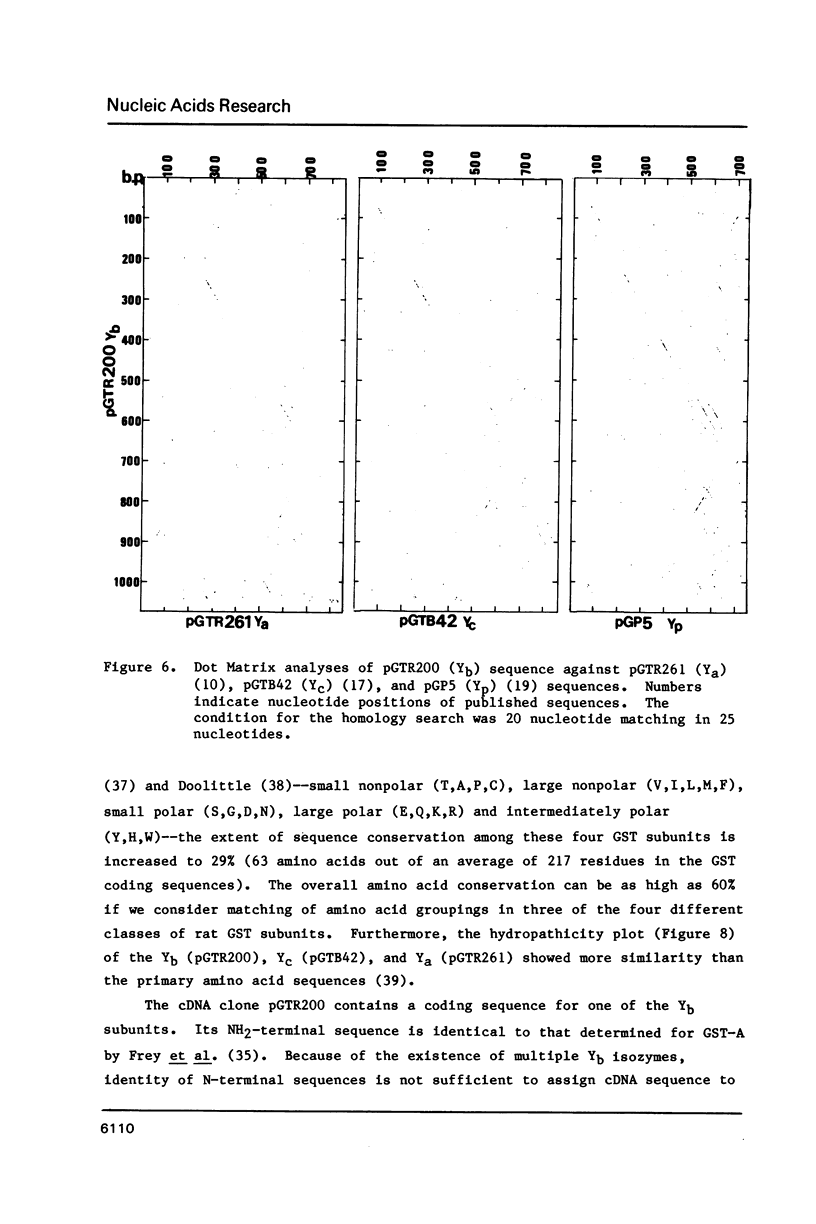
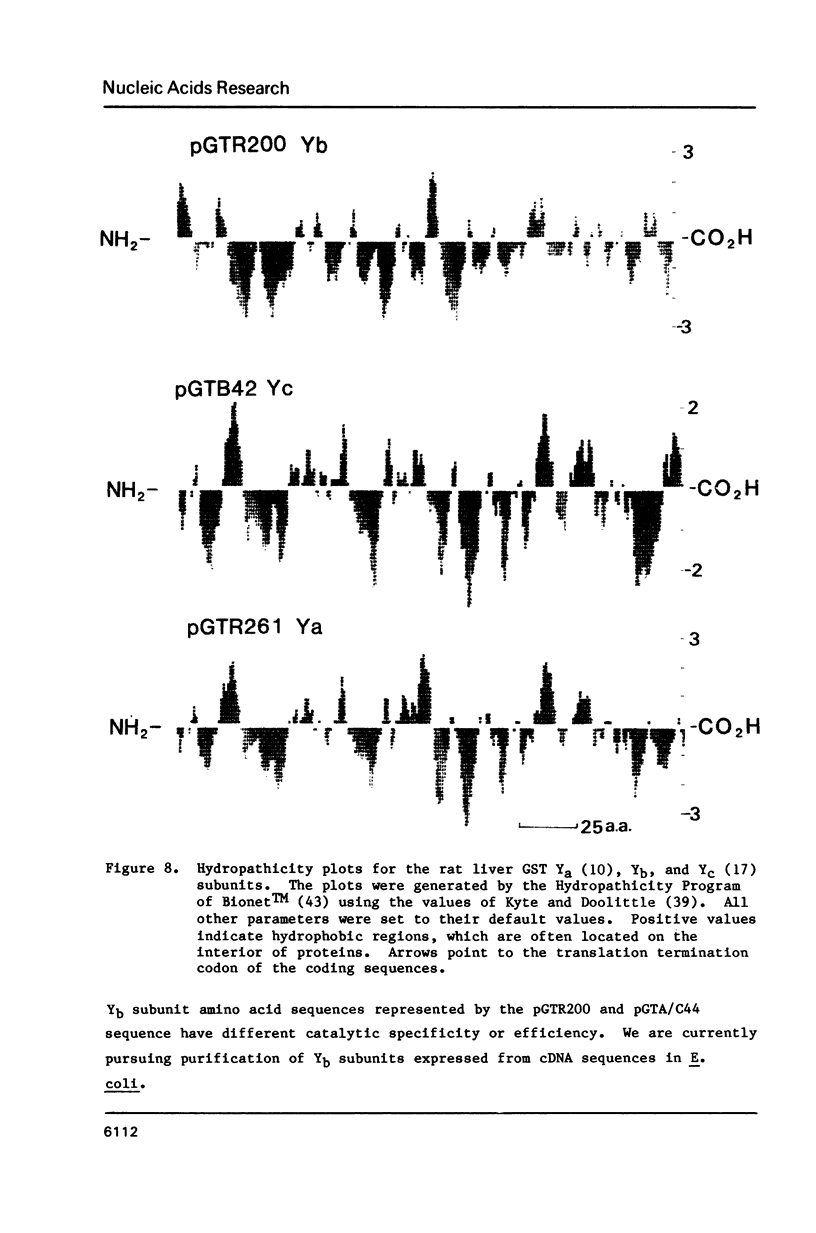
We have isolated a Yb-subunit cDNA clone from a GSH S-transferase (GST) cDNA library made from rat liver polysomal poly(A) RNAs. Sequence analysis of one of these cDNA, pGTR200, revealed an open reading frame of 218 amino acids of Mr = 25,915. The deduced sequence is in agreement with the 19 NH2-terminal residues for GST-A. The sequence of pGTR200 differs from another Yb cDNA, pGTA/C44 by four nucleotides and two amino acids in the coding region, thus revealing sequence microheterogeneity. The cDNA insert in pGTR200 also contains 36 nucleotides in the 5' noncoding region and a complete 3' noncoding region. The Yb subunit cDNA shares very limited homology with those of the Ya or Yc cDNAs, but has relatively higher sequence homology to the placental subunit Yp clone pGP5. The mRNA of pGTR200 is not expressed abundantly in rat hearts and seminal vesicles. Therefore, the GST subunit sequence of pGTR200 probably represents a basic Yb subunit. Genomic DNA hybridization patterns showed a complexity consistent with having a multigene family for Yb subunits. Comparison of the amino acid sequences of the Ya, Yb, Yc, and Yp subunits revealed significant conservation of amino acids (approximately 29%) throughout the coding sequences. These results indicate that the rat GSTs are products of at least four different genes that may constitute a supergene family.
Full text
PDF









Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aviv H., Leder P. Purification of biologically active globin messenger RNA by chromatography on oligothymidylic acid-cellulose. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1408–1412. doi: 10.1073/pnas.69.6.1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bantle J. A., Maxwell I. H., Hahn W. E. Specificity of oligo (dT)-cellulose chromatography in the isolation of polyadenylated RNA. Anal Biochem. 1976 May 7;72:413–427. doi: 10.1016/0003-2697(76)90549-2. [DOI] [PubMed] [Google Scholar]
- Bass N. M., Kirsch R. E., Tuff S. A., Marks I., Saunders S. J. Ligandin heterogeneity : evidence that the two non-identical subunits are the monomers of two distinct proteins. Biochim Biophys Acta. 1977 May 27;492(1):163–175. doi: 10.1016/0005-2795(77)90223-9. [DOI] [PubMed] [Google Scholar]
- Bhargava M. M., Ohmi N., Listowsky I., Arias I. M. Subunit composition, organic anion binding, catalytic and immunological properties of ligandin from rat testis. J Biol Chem. 1980 Jan 25;255(2):724–727. [PubMed] [Google Scholar]
- Birnboim H. C., Doly J. A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res. 1979 Nov 24;7(6):1513–1523. doi: 10.1093/nar/7.6.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chasseaud L. F. The role of glutathione and glutathione S-transferases in the metabolism of chemical carcinogens and other electrophilic agents. Adv Cancer Res. 1979;29:175–274. doi: 10.1016/s0065-230x(08)60848-9. [DOI] [PubMed] [Google Scholar]
- Cleveland D. W., Lopata M. A., MacDonald R. J., Cowan N. J., Rutter W. J., Kirschner M. W. Number and evolutionary conservation of alpha- and beta-tubulin and cytoplasmic beta- and gamma-actin genes using specific cloned cDNA probes. Cell. 1980 May;20(1):95–105. doi: 10.1016/0092-8674(80)90238-x. [DOI] [PubMed] [Google Scholar]
- Ding G. J., Lu A. Y., Pickett C. B. Rat liver glutathione S-transferases. Nucleotide sequence analysis of a Yb1 cDNA clone and prediction of the complete amino acid sequence of the Yb1 subunit. J Biol Chem. 1985 Oct 25;260(24):13268–13271. [PubMed] [Google Scholar]
- Doolittle R. F. Proteins. Sci Am. 1985 Oct;253(4):88–99. doi: 10.1038/scientificamerican1085-88. [DOI] [PubMed] [Google Scholar]
- Frey A. B., Friedberg T., Oesch F., Kreibich G. Studies on the subunit composition of rat liver glutathione S-transferases. J Biol Chem. 1983 Sep 25;258(18):11321–11325. [PubMed] [Google Scholar]
- Gubler U., Hoffman B. J. A simple and very efficient method for generating cDNA libraries. Gene. 1983 Nov;25(2-3):263–269. doi: 10.1016/0378-1119(83)90230-5. [DOI] [PubMed] [Google Scholar]
- Hanahan D. Studies on transformation of Escherichia coli with plasmids. J Mol Biol. 1983 Jun 5;166(4):557–580. doi: 10.1016/s0022-2836(83)80284-8. [DOI] [PubMed] [Google Scholar]
- Hayes J. D. Purification and characterization of glutathione S-transferases P, S and N. Isolation from rat liver of Yb1 Yn protein, the existence of which was predicted by subunit hybridization in vitro. Biochem J. 1984 Dec 15;224(3):839–852. doi: 10.1042/bj2240839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hood L., Kronenberg M., Hunkapiller T. T cell antigen receptors and the immunoglobulin supergene family. Cell. 1985 Feb;40(2):225–229. doi: 10.1016/0092-8674(85)90133-3. [DOI] [PubMed] [Google Scholar]
- Jakoby W. B. The glutathione S-transferases: a group of multifunctional detoxification proteins. Adv Enzymol Relat Areas Mol Biol. 1978;46:383–414. doi: 10.1002/9780470122914.ch6. [DOI] [PubMed] [Google Scholar]
- Kaplowitz N., Kuhlekamp J., Clifton G. Drug induction of hepatic glutathione S-transferases in male and female rats*. Biochem J. 1975 Feb;146(2):351–356. doi: 10.1042/bj1460351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kraus J. P., Rosenberg L. E. Purification of low-abundance messenger RNAs from rat liver by polysome immunoadsorption. Proc Natl Acad Sci U S A. 1982 Jul;79(13):4015–4019. doi: 10.1073/pnas.79.13.4015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kyte J., Doolittle R. F. A simple method for displaying the hydropathic character of a protein. J Mol Biol. 1982 May 5;157(1):105–132. doi: 10.1016/0022-2836(82)90515-0. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lai H. C., Li N., Weiss M. J., Reddy C. C., Tu C. P. The nucleotide sequence of a rat liver glutathione S-transferase subunit cDNA clone. J Biol Chem. 1984 May 10;259(9):5536–5542. [PubMed] [Google Scholar]
- Lehrach H., Diamond D., Wozney J. M., Boedtker H. RNA molecular weight determinations by gel electrophoresis under denaturing conditions, a critical reexamination. Biochemistry. 1977 Oct 18;16(21):4743–4751. doi: 10.1021/bi00640a033. [DOI] [PubMed] [Google Scholar]
- Levi A. J., Gatmaitan Z., Arias I. M. Two hepatic cytoplasmic protein fractions, Y and Z, and their possible role in the hepatic uptake of bilirubin, sulfobromophthalein, and other anions. J Clin Invest. 1969 Nov;48(11):2156–2167. doi: 10.1172/JCI106182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mannervik B. The isoenzymes of glutathione transferase. Adv Enzymol Relat Areas Mol Biol. 1985;57:357–417. doi: 10.1002/9780470123034.ch5. [DOI] [PubMed] [Google Scholar]
- Maxam A. M., Gilbert W. Sequencing end-labeled DNA with base-specific chemical cleavages. Methods Enzymol. 1980;65(1):499–560. doi: 10.1016/s0076-6879(80)65059-9. [DOI] [PubMed] [Google Scholar]
- Okayama H., Berg P. High-efficiency cloning of full-length cDNA. Mol Cell Biol. 1982 Feb;2(2):161–170. doi: 10.1128/mcb.2.2.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelham H. R., Jackson R. J. An efficient mRNA-dependent translation system from reticulocyte lysates. Eur J Biochem. 1976 Aug 1;67(1):247–256. doi: 10.1111/j.1432-1033.1976.tb10656.x. [DOI] [PubMed] [Google Scholar]
- Pickett C. B., Telakowski-Hopkins C. A., Ding G. J., Argenbright L., Lu A. Y. Rat liver glutathione S-transferases. Complete nucleotide sequence of a glutathione S-transferase mRNA and the regulation of the Ya, Yb, and Yc mRNAs by 3-methylcholanthrene and phenobarbital. J Biol Chem. 1984 Apr 25;259(8):5182–5188. [PubMed] [Google Scholar]
- Reddy C. C., Burgess J. R., Tu C. P. Isolation and characterization of an anionic glutathione S-transferase from rat liver cytosol. Biochem Biophys Res Commun. 1983 Mar 29;111(3):840–846. doi: 10.1016/0006-291x(83)91375-x. [DOI] [PubMed] [Google Scholar]
- Reddy C. C., Li N., Tu C. P. Identification of a new glutathione S-transferase from rat liver cytosol. Biochem Biophys Res Commun. 1984 Jun 29;121(3):1014–1020. doi: 10.1016/0006-291x(84)90778-2. [DOI] [PubMed] [Google Scholar]
- Scully N. C., Mantle T. J. Tissue distribution and subunit structures of the multiple forms of glutathione S-transferase in the rat. Biochem J. 1981 Jan 1;193(1):367–370. doi: 10.1042/bj1930367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Suguoka Y., Kano T., Okuda A., Sakai M., Kitagawa T., Muramatsu M. Cloning and the nucleotide sequence of rat glutathione S-transferase P cDNA. Nucleic Acids Res. 1985 Sep 11;13(17):6049–6057. doi: 10.1093/nar/13.17.6049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swanson R. A unifying concept for the amino acid code. Bull Math Biol. 1984;46(2):187–203. doi: 10.1007/BF02460068. [DOI] [PubMed] [Google Scholar]
- Swanstrom R., Shank P. R. X-Ray Intensifying Screens Greatly Enhance the Detection by Autoradiography of the Radioactive Isotopes 32P and 125I. Anal Biochem. 1978 May;86(1):184–192. doi: 10.1016/0003-2697(78)90333-0. [DOI] [PubMed] [Google Scholar]
- Telakowski-Hopkins C. A., Rodkey J. A., Bennett C. D., Lu A. Y., Pickett C. B. Rat liver glutathione S-transferases. Construction of a cDNA clone complementary to a Yc mRNA and prediction of the complete amino acid sequence of a Yc subunit. J Biol Chem. 1985 May 10;260(9):5820–5825. [PubMed] [Google Scholar]
- Tu C. P., Chang M., Reddy C. C. The major rat heart glutathione S-transferases are anionic isozymes composed of Yb size subunits. Biochem Biophys Res Commun. 1984 Sep 28;123(3):981–988. doi: 10.1016/s0006-291x(84)80230-2. [DOI] [PubMed] [Google Scholar]
- Tu C. P., Lai H. C., Li N. Q., Weiss M. J., Reddy C. C. The Yc and Ya subunits of rat liver glutathione S-transferases are the products of separate genes. J Biol Chem. 1984 Aug 10;259(15):9434–9439. [PubMed] [Google Scholar]
- Tu C. P., Matsushima A., Li N. Q., Rhoads D. M., Srikumar K., Reddy A. P., Reddy C. C. Immunological and sequence interrelationships between multiple human liver and rat glutathione S-transferases. J Biol Chem. 1986 Jul 15;261(20):9540–9545. [PubMed] [Google Scholar]
- Tu C. P., Reddy C. C. On the multiplicity of rat liver glutathione S-transferases. J Biol Chem. 1985 Aug 25;260(18):9961–9964. [PubMed] [Google Scholar]
- Tu C. P., Weiss M. J., Karakawa W. W., Reddy C. C. Cloning and sequence analysis of a cDNA plasmid for one of the rat liver glutathione S-transferase subunits. Nucleic Acids Res. 1982 Sep 25;10(18):5407–5419. doi: 10.1093/nar/10.18.5407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tu C. P., Weiss M. J., Li N. Q., Reddy C. C. Tissue-specific expression of the rat glutathione S-transferases. J Biol Chem. 1983 Apr 25;258(8):4659–4662. [PubMed] [Google Scholar]
- Zweig S. E. Analysis of large nucleic acid dot matrices on small computers. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 2):767–776. doi: 10.1093/nar/12.1part2.767. [DOI] [PMC free article] [PubMed] [Google Scholar]