Abstract
Previously, permeability and site of intestinal absorption of propranolol have been reported using the Ussing chamber. In the present study, the utility of Single-Pass Intestinal Perfusion to study permeability and site of intestinal absorption of propranolol was evaluated in rats. Drug permeability in different regions of rat intestine viz. duodenum, jejunum, ileum and colon was measured. Propranolol (30 μg/ml) solution was perfused in situ in each intestinal segment of rats. Effective permeability (Peff) of propranolol in each segment was calculated and site of absorption was determined. The Peff of propranolol in rat duodenum, jejunum, ileum and colon was calculated to be 0.3316×10-4 cm/s, 0.4035×10-4cm/s, 0.5092×10-4 cm/s and 0.7167×10-4 cm/s, respectively. The above results suggest that permeability of propranolol was highest through colon compared to other intestinal sites, which is in close agreement to that reported previously. In conclusion, in situ single pass intestinal perfusion can be used effectively to study intestinal permeability as well as site of intestinal absorption of compounds in rats.
Keywords: Intestine, permeability, propranolol, single-pass intestinal perfusion, site of absorption
Oral administration is the most convenient and useful route for drug delivery, which depends on factors like solubility and permeability i.e. the ability of a molecule to cross the biological membranes[1,2]. Prediction of drug permeability is very important for the design and success of an oral medication. Often formulation strategies are used to enhance bioavailability of drugs, e.g. enteric-coated tablets to prevent degradation of drug in the acidic pH of stomach. However, for such strategies to succeed, knowledge of site of absorption of drug is essential.
Although the entire gastrointestinal tract is capable of drug absorption, the small intestine (which can be subdivided into duodenum, jejunum and ileum) is the major site of absorption for most nutrients and drugs[3]. The two principal routes of drug absorption across small intestinal epithelium are paracellular and transcellular. Typically, hydrophilic drugs are slowly absorbed via the transcellular route or in some cases via the paracellular[5,6] and lipophilic drugs are absorbed by transcelluar route[7,6].
In the preclinical drug discovery stage a number of in vitro, ex vivo and in vivo experimental models are used to determine the intestinal absorption and mechanism of absorption of drugs[9–11]. Some of these are everted gut sac method, intestinal rings, Ussing chamber, Single-Pass Intestinal Perfusion (SPIP) and Caco-2 cell permeability[12]. Among these methods, the in situ SPIP technique[13] is frequently used, which provides physiological conditions closer to those encountered following oral administration[2,4]. It has however, a lower sensitivity to pH variation because of a preserved microclimate above the epithelial cell layer but maintains an intact blood supply to the intestine[2,4]. This technique offers the advantage of a better control of compound concentration and intestinal perfusion rate, which allows the study of the regional factors influencing intestinal absorption of a compound.
The objective of the present investigation was to study the utility of in situ SPIP to determine the site of absorption of a model drug propranolol, which is a clinically important beta-blocker, used orally in the treatment of disorders such as hypertension, arrhythmia and angina pectoris[1] It is a highly soluble and highly permeable drug and belongs to Class I compounds of the Biopharmaceutics Classification System[14].
Propranolol and urethane were purchased from Sigma-Aldrich, St. Louis MO, USA. Phenol red was purchased from Rankem, New Delhi, India. Na2HPO4 and NaH2PO4, acetonitrile, methanol, glacial acetic acid and triethylamine were purchased from Merck India Limited, Mumbai, India. NaCl was purchased from Qualigens Fine Chemicals, Mumbai, India. All other chemicals were of analytical grade and purchased from commercial suppliers.
Male Wistar rats (200-250 g) bred in-house at Piramal Life Sciences Limited, Goregaon, Mumbai, India, were maintained in a temperature and humidity-controlled room with a 12-h light/dark cycle with free access to standard diet and water. All animals used in this study were mature and healthy and were not subjected to any form of treatment/medication. Guidelines of committee for the purpose of control and supervision on experiments on animals (CPCSEA), Government of India, were followed and the in-house animal ethics committee approved all experimental procedures.
In situ permeability studies in rat were performed using established method adapted from literature[13,15]. Male Wistar rats were fasted 12-18 h prior to the start of the experiment but had free access to tap water. Anesthesia was induced and maintained for the duration of the experiment by intraperitoneal injection of urethane (1.5 g/kg). The animals were placed on a heated pad to maintain body temperature at 37°. A midline incision was made on the ventral surface (abdomen). Approximately 10 cm section of the intestinal segment (jejunum) was selected for permeability study. The segment was canulated on both sides with Portex Fine Bore Polythene tubing (1.19 mm ID and 1.70 mm OD). The intestinal segment was flushed with saline (37), until outlet solution was clear of food particles and finally the cannulated segment was connected to the perfusion assembly. Care was taken during the surgery in order to maintain an intact blood supply. The exposed segment was covered with a cotton pad soaked in normal saline to prevent evaporation of fluid. For permeability determination, propranolol was dissolved in perfusion buffer (30 μg/ml) and perfused at a flow rate of 0.2 ml/min for 50 min, and the outlet perfusate samples were collected at 20, 30, 40 and 50 min from the start of perfusion. At the end of 50 min, the length of cannulated segment was measured. Samples were stored at –70°until analysis by HPLC.
Twelve fasted rats were divided into four groups (n=3). SPIP was performed in different intestinal segments (duodenum, jejunum, ileum and colon). Each intestinal segment of rat was isolated as follows: duodenum-beginning from pylorus and about 100 mm in length; jejunum the longest part of intestine, beginning at end of duodenum with an average length of 900-1350 mm, ileum-beginning at the site above caecum, 600-700 mm in length and colon-beginning from end of caecum to descending colon. The above segments were cannulated and perfused with solution containing propranolol (30 μg/ml) as described earlier for determination of site of absorption.
Perfusion buffer of pH 6.5 was prepared by dissolving 4.47 g Na2HPO4(anhydrous), 9.52 g NaH2PO4.2H2O and 7 g NaCl in one liter of distilled water. Perfusion buffer of pH 7.2 was prepared by dissolving 3.89 g Na2HPO4(anhydrous), 10.22 g NaH2PO4.2H2O and 7 g NaCl in one liter of distilled water. Perfusion buffer of pH 6.5 was used for perfusion of duodenum, jejunum. Perfusion buffer of pH 7.2 was used for perfusion of ileum and colon.
All the samples were analyzed for propranolol concentration using HPLC. The HPLC system consisted of a Shimadzu LC-10A7 (LC), Shimadzu 10AVP (System controller), Shimadzu FCV-10AL VP (degasser), Shimadzu SPD-N10AVP (PDA), Shimadzu SIL-10ADVP (Auto sampler), Shimadzu CT010ASVP (Oven) and Class VP software. The analytical column used was Hypersil BDS C18, 240×4.6 mm, 5 μm maintained at 34. Propranolol was detected at 240 nm, using a gradient system of Solvent A (acetonitrile) and Solvent B (0.01 M ammonium acetate containing 0.5% triethylamine, pH 5.0 adjusted using glacial acetic acid). The gradient (time/%A) was 0/0, 10/65, 15/65, 15.01/0, 20/0 with a flow rate of 1.0 ml/min. For analysis, perfusate samples were thawed at room temperature, centrifuged at 2739 g for 5 min and supernatants were subjected to HPLC analysis. Propranolol concentrations in the samples were read against linearity curve of propranolol in the concentration range of 1 to 100 μg/ml.
Calculations were based on the mean outlet perfusate concentrations of propranolol achieved at 20, 30, 40 and 50 min sampling points. The steady-state intestinal effective permeability (Peff cm/s) was calculated according to following Eqn. 1[13], Peff= [-Q.ln(Cout/Cin)]/2πrL.. (1), where, Q is the flow rate (0.2 ml/min), Cin and Cout are the inlet and outlet perfusate concentrations. 2πrL is the cylindrical surface area of the individual intestinal segment with a length ‘L’ (approximately 10 cm) and ‘r’ is the intestinal radius. For duodenum, jejunum and ileum r= 0.18 cm and for colon r= 0.25 cm[16]. Values were reported as mean±SD of 3 observations.
The intestinal net water flux (NWF, μl/h/cm) was calculated using Eqn. 2, NWF= (1-[Ph.red(out)/Ph.red(in)]×Qin)/L (2), where [Ph.red(out)/] and [Ph.red(in)] are the inlet and outlet concentrations of the non-absorbable, water flux marker phenol red. A negative net water flux indicates loss of fluid from the mucosal side (lumen) to serosal side (blood) while a positive net water flux indicates secretion of fluid into the segment[13].
Most drug molecules are either weak acids or bases. When given orally, the extent of the ionization of these drugs is dependent on their pKa and pH gradient inherent to the GI tract. The interplay between these two interrelated parameters has a major impact on the drug permeability and solubility and ultimately the rate and extent of oral drug absorption. One of the original concepts that govern the oral drug absorption is the pH partition theory. This theory states that oral drug absorption takes place mainly by a passive diffusion of the un-ionized form of the drug molecule through the lipophilic intestinal membrane[17,18]. Accordingly, the oral absorption of a weak base will be favored in the intestine rather than the stomach, as the intestine has a higher pH and, as a result, a higher proportion of the un-ionized drug fraction will be available for oral absorption. On the contrary, weak acid absorption will be favored in the stomach owing to its lower pH[18,19].
Intestinal permeability is related to the ability of a compound to move across the epithelial barrier of the intestine and represents a direct measurement of the local absorption rate and reflects the transport velocity across the epithelial barrier, expressed as centimeter per second[20]. Comparisons between human Peffdata and preclinical permeability models like in situ perfusion of rat intestine, Caco-2 cell monolayer model, and excised intestinal segments in the Ussing chamber show that they can be used to investigate and classify passive transport with a high accuracy[20]. In this study, propranolol was selected since it shows a pH dependent absorption profile, being a weak base and transported by passive diffusion. Intestinal permeability of propranolol was determined in rat jejunum and site of intestinal absorption was determined in different rat intestinal segments (duodenum, jejunum, ileum and colon) using in situ single-pass intestinal perfusion technique.
Phenol red was used as a non-absorbable marker for calculating the NWF. The reported value for intestinal NWF according to Fagerholm et al.[13] was -54 (±59) μl/h/cm. Any leakage across the jejunal mucosa results in changes in the intestinal barrier function which is probably one of the main reasons for contradictory results obtained by the -in situ model. In our results, stable water fluxes with time were observed in jejunum and colon as reported by Fagerholm et al.[13]. The Mean NWF (μl/h/cm) in different rat intestinal segments -22.60(±22.57) -4.443(±81.824) and -84.93(±74.78) μl/h/cm were observed in jejunum, duodenum and colon respectively but in the ileum, the NWF was higher Table 1. Others have reported the intestinal NWF as 28±12 μl/h/cm by the SPIP method[2], which is close to the NWF we observed in ileum. This indicates that a stable barrier function was maintained in all experiments.
TABLE 1.
EFFECTIVE PERMEABILITY OF PROPRANOLOL IN DIFFERENT RAT INTESTINAL SEGMENTS
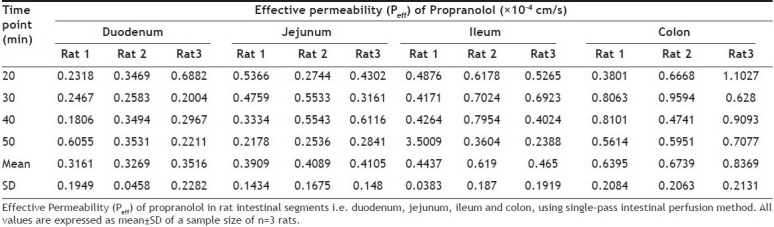
Effective permeability (Peff) of propranolol calculated in each intestinal segment of each rat is given in Table 1. Mean effective permeability values of propranolol in duodenum, jejunum, ileum and colon are shown in Table 2. Results indicate that effective permeability of propranolol was highest in colon as compared to other intestinal segments. The permeability of propranolol in rat intestinal segments was in the order duodenum<jejunum<ileum<colon and were 0.3316×10-4cm/s, 0.4035×10-4cm/s, 0.5092×10-4cm/s and 0.7167×10-4cm/s, respectively. Previously, the intestinal effective permeability of propranolol by SPIP method in rat jejunum has been reported as 0.49±0.19×10-4cm/s[2,17] which is in close agreement to our result.
TABLE 2.
MEAN EFFECTIVE PERMEABILITY (PEFF) OF PROPRANOLOL AND MEAN NWF IN DIFFERENT RAT INTESTINAL SEGMENTS
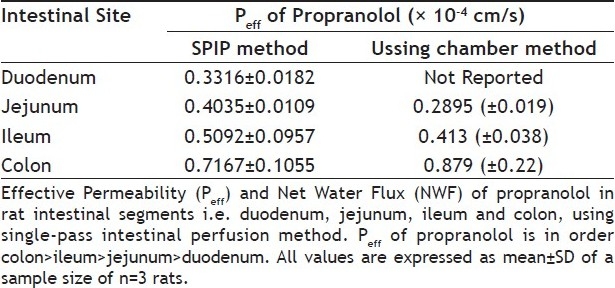
The intestinal site of absorption studies for propranolol has been reported previously using Ussing chamber method[17]. In the present study, we studied the Peff and site of intestinal absorption of propranolol using the SPIP technique with slight modification i.e. pH of perfusion buffers was kept close to physiological pH in the respective segments, to test if the intestinal absorption of propranolol exhibited site and pH dependent changes and to correlate the results obtained with those reported by the in vitro method. Our results indicate that propranolol can be absorbed along the whole intestine with the main absorption site being colon, which is in close agreement to that reported previously Table 2 by the Ussing chamber method.
In conclusion, in situ single-pass intestinal perfusion in rats gave a good estimation of the effective permeability and site of intestinal absorption of propranolol and can be successfully used to predict absorption of new chemical entities.
Footnotes
Nagare, et al.: Determination of Site of Propranolol Absorption in Rat Gut
REFERENCES
- 1.Amidon GL, Lennernäs H, Shah VP, Crison JR. A theoretical basis for a biopharmaceutic drug classification: the correlation of in vitro drug product dissolution and in vivo bioavailability. Pharm Res. 1995;12:413–20. doi: 10.1023/a:1016212804288. [DOI] [PubMed] [Google Scholar]
- 2.Parvin Z, Valizadeh H, Tajerzadeh H, Azarmi Y, Barzegar-Jalali M. Simultaneous determination of metoprolol, propranolol and phenol red in sample from rat in situ intestinal perfusion studies. DARU. 2006;14:102–7. [Google Scholar]
- 3.Friend D. Drug delivery to the small intestine. Curr Gastroenterol Rep. 2004;6:371–76. doi: 10.1007/s11894-004-0052-z. [DOI] [PubMed] [Google Scholar]
- 4.Zakeri-Milani P, Valizadeh H, Tajerzadeh H, Azarmi Y, Islambolchilar Z, Barzegar S, et al. Predicting human intestinal permeability using Single-pass Intestinal Perfusion in rat. J Pharm Pharm Sci. 2007;10:368–79. [PubMed] [Google Scholar]
- 5.Ungell A. Current status on targeted drug delivery to the gastrointestinal tract. Greenwood, SC: Capsugel Americas; 1993. [Google Scholar]
- 6.Schwartz RM, Furne JK, Levitt MD. Paracellular intestinal transport of six-carbon sugar is negligible in the rat. Gastroenterology. 1995;109:1206–13. doi: 10.1016/0016-5085(95)90580-4. [DOI] [PubMed] [Google Scholar]
- 7.Palm K, Luthman K, Ungell AL, Strandlund G, Artursson P. Correlation of drug absorption with molecular surface properties. J Pharm Sci. 1996;85 Suppl 1:32–9. doi: 10.1021/js950285r. [DOI] [PubMed] [Google Scholar]
- 8.Waterbeemd H, Camenisch J, Folkers G, Raevsky O. Estimation of Caco-2 cell permeability using calculated molecular descriptors. QSAR Comb Sci. 1996;15:480–90. [Google Scholar]
- 9.Amidon GL, Sinko PJ, Fleisher D. Estimating human oral fraction dose absorbed: a correlation using rat intestinal membrane permeability for passive and carrier-mediated compounds. Pharm Res. 1988;5:651–4. doi: 10.1023/a:1015927004752. [DOI] [PubMed] [Google Scholar]
- 10.Cook T, Shenoy SS. Intestinal permeability of chlorpyrifos using a single-pass intestinal perfusion method in the rat. Toxicology. 2003;184:125–33. doi: 10.1016/s0300-483x(02)00555-3. [DOI] [PubMed] [Google Scholar]
- 11.Salphati L, Childers K, Pan L, Tsutsui K, Takahashi L. Evaluation of a single-pass intestinal perfusion method in rat for the prediction of absorption in man. J Pharm Pharmacol. 2001;53:1007–13. doi: 10.1211/0022357011776252. [DOI] [PubMed] [Google Scholar]
- 12.Barthe L, Woodley J, Houin G. Gastrointestinal absorption of drugs: Methods and studies. Fundam Clin Pharmacol. 1999;13:154–68. doi: 10.1111/j.1472-8206.1999.tb00334.x. [DOI] [PubMed] [Google Scholar]
- 13.Fagerholm U, Johansson M, Lennernas H. Comparison between permeability coefficients in rat and human jejunum. Pharm Res. 1996;13:1336–42. doi: 10.1023/a:1016065715308. [DOI] [PubMed] [Google Scholar]
- 14.Kim JS, Mitchell S, Kijek P, Tsume Y, Hilfinger Y, Amidon GL. The suitability of an in situ perfusion model for permeability determinations: Utility for BCS Class I Biowaiver requests. Mol Pharm. 2006;3:686–94. doi: 10.1021/mp060042f. [DOI] [PubMed] [Google Scholar]
- 15.Sinko PJ, Hu P, Waclawski AP, Patel NR. Oral absorption of anti-AIDS nucleoside analogues. 1. Intestinal transport of didanosine in rat and rabbit preparations. J Pharm Sci. 1995;84:959. doi: 10.1002/jps.2600840811. [DOI] [PubMed] [Google Scholar]
- 16.Komiya I, Park J, Kamani A, Ho N, Higuchi W. Quantitative mechanistic studies in simultaneous fluid flow and intestinal absorption using steroids as model solutes. Int J Pharm. 1980;4:249–62. [Google Scholar]
- 17.Ungell AL, Nylander S, Bergstrand S, SjoBerg A, Lennernas H. Membrane transport of drugs in different regions of the intestinal tract of the rat. J Pharm Sci. 1998;87:360–6. doi: 10.1021/js970218s. [DOI] [PubMed] [Google Scholar]
- 18.Palm K, Luthman K, Ros J, Grasjo J, Artursson P. Effect of molecular charge on intestinal epithelial drug transport: pH-dependent transport of cationic drugs. J Pharmacol Exp Ther. 1999;291:435–43. [PubMed] [Google Scholar]
- 19.Hurst S, Loi CM, Brodfuehrer J, El-Kattan A. Impact of physiological, physicochemical and biopharmaceutical factors in absorption and metabolism mechanisms on the drug oral bioavailability of rats and humans. Expert Opin Drug Metab Toxicol. 2007;4:469–89. doi: 10.1517/17425225.3.4.469. [DOI] [PubMed] [Google Scholar]
- 20.Song NN, Li QS, Liu CX. Intestinal permeability of metformin using single-pass intestinal perfusion in rats. World J Gastroenterol. 2006;12:4064–70. doi: 10.3748/wjg.v12.i25.4064. [DOI] [PMC free article] [PubMed] [Google Scholar]


