Abstract
Whereas the roles of proangiogenic factors in carcinogenesis are well established, those of endogenous angiogenesis inhibitors (EAIs) remain to be fully elaborated. We investigated the roles of three EAIs during de novo tumorigenesis to further test the angiogenic balance hypothesis, which suggests that blood vessel development in the tumor microenvironment can be governed by a net loss of negative regulators of angiogenesis in addition to the well-established principle of up-regulated angiogenesis inducers. In a mouse model of pancreatic neuroendocrine cancer, administration of endostatin, thrombospondin-1, and tumstatin peptides, as well as deletion of their genes, reveal neoplastic stage-specific effects on angiogenesis, tumor progression, and survival, correlating with endothelial expression of their receptors. Deletion of tumstatin and thrombospondin-1 in mice lacking the p53 tumor suppressor gene leads to increased incidence and reduced latency of angiogenic lymphomas associated with diminished overall survival. The results demonstrate that EAIs are part of a balance mechanism regulating tumor angiogenesis, serving as intrinsic microenvironmental barriers to tumorigenesis.
Keywords: integrins, cell biology
Angiogenesis, the formation of new blood vessels, is crucial during transient physiological and pathological processes in the adult. An increasing body of evidence supports the hypothesis that angiogenesis is switched on and orchestrated by balance in the local abundance of endogenous proangiogenic and antiangiogenic factors (1, 2). Proangiogenic signaling is well understood, in terms of intracellular networks and mechanistic effects (3, 4). By contrast, understanding of the roles, regulation, and effects of the counterbalancing endogenous antiangiogenic signals in regulating the angiogenic switch has lagged, due to complexities in their molecular structures and receptors (5–7). Several endogenous antiangiogenic factors are proteolytic fragments of structural proteins, including constituents of the vascular basement membrane (8). Most have several receptors or coreceptors, in several cases including heterodimeric integrins involved in anchoring endothelial cells to the vascular basement membrane (9, 10).
Among the growing list of endogenous angiogenesis inhibitors, several of the best characterized are peptide fragments of naturally occurring extracellular matrix (ECM) and basement membrane proteins (1). Endostatin, derived from the noncollagenous (NC1) domain of the α1 chain of type XVIII collagen, attenuates angiogenesis, tumor growth, and metastasis in experimental animal models (11, 12). Another such fragment, tumstatin, consisting of the NC1 domain of the α3 chain of type IV collagen, is antiangiogenic and selectively proapoptotic to proliferating endothelial cells (13). Thrombospondin-1 (TSP1) offers another example of how glycoproteins within the tumor microenvironment can serve as important functional regulators of angiogenesis (14).
In this study, we present genetic and pharmacological data, which demonstrate that endogenous protein fragments are involved in regulating the angiogenic switch and controlling de novo tumor growth and survival associated with pancreatic neuroendocrine tumorigenesis (PNET) in RIP-Tag2 (RT2) mice. Genetic testing in a second cancer model, involving deletion of tumstatin and TSP1 in p53−/− mice, generalizes the significance of these endogenous angiogenesis inhibitors in controlling cancer progression and increasing overall survival.
Results
We documented the importance of VEGF-A gene expression for the initial angiogenic switch, as illustrated in Fig. S1, assessing hyperproliferative islets in RT2 mice in which the VEGF-A gene had been specifically deleted within the oncogene-expressing islet β cells of the pancreas (RT2/VEGFRIPKO). When hyperproliferative islet lesions (hyperplasias and dysplasias) were identified in the pancreas by BrdU labeling, both nonangiogenic islets as well as angiogenic islets were detected in VEGF wild-type mice (RT2/VEGFWT), whereas the RT2/VEGFRIPKO mice had only hyperproliferative lesions without evident angiogenesis. Hypoxia was prevalent in RT2/VEGFRIPKO islets (Fig. S1F).
Although these results illustrate the importance of VEGF as an inducing signal for triggering the initial angiogenic switch, the data do not implicate a balance mechanism. A question remained as to whether there were significant negative regulatory signals counterbalancing the proangiogenic signals exemplified by VEGF, defining a balance mechanism for the initial angiogenic switch.
Endostatin, Tumstatin, and TSP1 Restrict Tumor Angiogenesis and Growth in RT2 Mice.
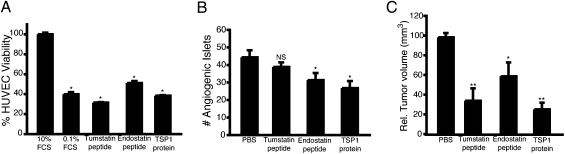
We used functionally active peptides derived from tumstatin and endostatin and a recombinant protein that includes the second type 1 repeat (TSR) of thrombospondin-1 (TSP1), to probe the capabilities of the three endogenous inhibitors to suppress angiogenesis and tumor growth via pharmacological elevation of their levels in the circulatory system (15–17). To confirm the relative efficacy of these reagents, we evaluated a 10-μM dose of each reagent (based on previous studies and chosen to ensure maximal activity while retaining solubility) in cell-culture–based endothelial viability assays (15–17). As shown in Fig. 1A, 10 μM of each peptide or recombinant protein reagent significantly inhibited endothelial cell viability with similar efficacy.
Fig. 1.
Pharmacological trials in RT2 mice using endogenous angiogenesis inhibitors. (A) In vitro viability assay of HUVEC cells treated with antiangiogenesis inhibitor reagents. (B) Prevention trial of RT2 mice from 5.5 to 10 wk of age. For all groups n = 6. (C) An intervention trial was conducted starting at 10 wk of age and continuing to 13.5 wk of age. Endostatin peptide (n = 9), tumstatin peptide (n = 8), and the TSP1 protein (n = 8) all significantly inhibited tumor growth compared with the PBS control group (n = 8). Results are shown as mean ± SEM; *P < 0.05, **P < 0.01.
The endostatin, tumstatin, and TSR inhibitors were administered to RT2 mice in two trials to assess their efficacy during different stages of PNET tumorigenesis (18). A prevention trial from 5.5 to 10 wk of age was designed to assess the effect of the inhibitors on the initial angiogenic switch in hyperplastic lesions. At this stage, angiogenesis is assessed by quantifying the number of neoplastic islets that have undergone the angiogenic switch (19, 20). In the prevention trial, daily treatment with endostatin peptide or TSR-based protein produced a ∼40% reduction in the number of angiogenic islets, whereas the tumstatin peptide did not exhibit significant antiangiogenic activity at this stage (Fig. 1B).
In the intervention trial, in which mice were treated from 10 to 13.5 wk, all three reagents demonstrated significant efficacy in inhibiting tumor growth (Fig. 1C). These results demonstrate that all three angiogenesis inhibitors significantly inhibited the growth of islet tumors in RT2 mice.
Notably, the tumstatin peptide was efficacious in the intervention but not the prevention trial; thus, its lack of efficacy in the prevention trial was not due to intrinsic inactivity of the peptide when used in vivo. In contrast, endostatin peptide and TSR-based protein were effective in both the prevention and intervention trials. Having established that all three inhibitory pathways could be activated by exogenous ligands, albeit with intriguing stage specificity in the case of tumstatin, we next assessed the expression of their receptors at different stages of tumorigenesis in the RT2 mice.
Tumor-Stage–Dependent Expression of Integrins and CD36 in the RT2 Mice.
We assessed expression of receptors for the three EAIs in the distinctive lesional stages of islet tumorigenesis. CD36 and the α5 and β3 integrin subunits showed distinct patterns of expression. Endothelial expression (as judged by CD31 colocalization) of α5 integrin was detected in the normal islets and was slightly increased in the angiogenic islets, albeit without statistical significance (Fig. S2 A and D). β3 integrin was barely detectable in normal islets or in the premalignant stages of cancer progression and then appeared robustly on most blood vessels of nascent solid tumors. However, as tumors became larger, expression levels decreased somewhat (Fig. S2 B and D). Colocalization of CD36 and CD31 by immunostaining revealed CD36 expression in endothelial cells of normal islets and all stages of tumorigenesis (Fig. S2C).
The mRNA levels of αV, β3, α5, and β1 integrin subunits along with CD36 were then analyzed by quantitative real-time PCR of RNA prepared from physically isolated normal pancreatic islets, hyperplastic/dysplastic islets, angiogenic islets, and tumors. The expression of the integrin subunits increased in the tumors, compared with normal islets, by 50% to over 100%, in relation to baseline CD31 coexpression in the normal islets (Fig. S3). CD36 showed a modest decrease in gene expression in the tumor setting relative to CD31 expression.
Deficiency of Endogenous Angiogenesis Inhibitors Accelerates Islet Carcinogenesis.
We next performed genetic loss-of-function studies to assess the contribution of physiological levels of these EAIs to the emergence of angiogenic islets and tumors in RT2 mice.
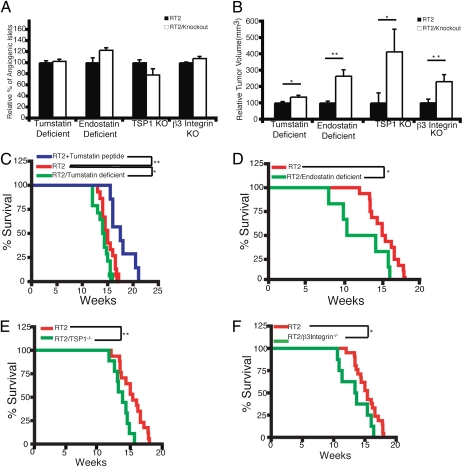
There was no significant difference in the numbers of angiogenic islets between RT2/tumstatin-deficient and WT RT2 mice at 10 wk of age (Fig. 2A). However, RT2/tumstatin-deficient mice displayed a significant increase in tumor volume at 13.5 wk, compared with WT RT2 control mice (Fig. 2B). Tumstatin deficiency significantly decreased the median survival of RT2 mice (Fig. 2C). The aforementioned intervention trial (Fig. 1) using tumstatin peptide produced the complementary result—significantly increased median survival of the RT2 mice (Fig. 2C). These results collectively substantiate the hypothesis that endogenous tumstatin plays an important role in controlling cancer progression in the RT2 mice.
Fig. 2.
Genetic ablation of endogenous angiogenesis inhibitors reveals functional roles as barriers to tumor growth but not angiogenic switching. (A) Deficiency in tumstatin (n = 6; RT2 only n = 6), endostatin (n = 2; RT2 only n = 2), or TSP1 (n = 5; RT2 only n = 5) did not significantly increase the frequency of angiogenic switching compared with RT2 mice. Deficiency in β3 integrin (n = 8; RT2 only n = 9), a functional receptor for tumstatin, also did not increase the frequency of angiogenic switching. (B) Enhanced tumor growth was observed in RT2 mice deficient in tumstatin (n = 5; RT2 only n = 6), endostatin (n = 3; RT2 only n = 9), TSP1 (n = 6; RT2 only n = 8), or β3 Integrin (n = 8; RT2 only n = 7). Additionally, a decreased lifespan was observed in RT2 mice deficient in tumstatin (n = 17; RT2 only n = 17) (C), endostatin (n = 6; RT2 only n = 16) (D), TSP1 (n = 21; RT2 only n = 17) (E), and β3 Integrin (n = 12; RT2 only n = 21) (F). Administration of the tumstatin peptide was able to improve survival of WT RT2 mice (n = 7) as shown in C. Results in A and B are shown as mean ± SEM; for C–F, significant differences are indicated by *P < 0.05, **P < 0.01.
Next, the physiological function of endostatin as an endogenous angiogenesis inhibitor was assessed by crossing the RT2 mice with mice deficient in the α1 chain of type XVIII collagen (RT2/endostatin deficient). RT2/endostatin-deficient mice developed more angiogenic islets (albeit not statistically significant) at 10 wk of age (Fig. 2A), whereas the tumor volume was significantly increased at 14 wk of age (Fig. 2B). Deficiency of endostatin in these mice led to a significantly decreased lifespan (Fig. 2D). Thus, the effects of endostatin inferred from its genetic ablation correlate with the vascular expression of α5 integrin expression in the endothelium throughout the stages of the RT2 tumorigenesis pathway.
Finally, we assessed the function of physiological levels of TSP1 during RT2 tumorigenesis in RT2/TSP1−/− mice. No significant difference was observed in the number of angiogenic islets (Fig. 2A). The RT2/TSP1−/− mice subsequently displayed a fourfold greater tumor burden compared with the littermate RT2 mice at 14 wk of age (Fig. 2B). Consistent with this enhanced tumorigenesis, there was a statistically significant decrease in the survival of RT2/TSP1−/− mice (Fig. 2E).
Evaluation of β3 Integrin as a Functional Receptor for Tumstatin.
The results of the gene knockout crosses and of the therapeutic trials suggest that the efficacy of these endogenous inhibitors correlate with expression of their putative receptors. In the case of tumstatin, there is little impact on the emergence of angiogenic islets, but a significant effect on the growth of tumors. These results correlate with the expression levels of integrin αVβ3 on the vasculature of the islets (vide supra) and are consistent with the hypothesis that a deficiency in this tumstatin receptor would affect tumor growth but not angiogenic switching. To address the stage-specific functional contribution of αVβ3 integrin in antiangiogenic signaling, we deleted β3 integrin in RT2 mice by crossbreeding to β3 integrin-null mice and examined the kinetics of tumorigenesis in the compound RT2/β3integrin−/− mice. As in the case of RT2/tumstatin-deficient mice, the number of angiogenic islets in RT2/β3integrin−/− mice at 10 wk of age remained similar to that of the littermate RT2 control mice (Fig. 2A), whereas the tumor volume of RT2/β3integrin−/− mice at 14 wk of age was significantly increased (Fig. 2B). The Kaplan–Meier survival curve reveals that lack of β3 integrin led to a significant reduction in the lifespan of RT2 mice (Fig. 2F).
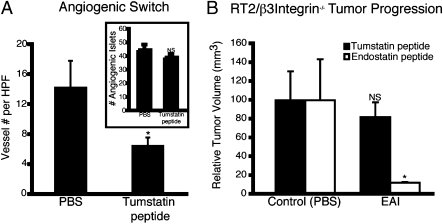
To further assess the postulate that low levels of αVβ3 integrin underlie the lack of tumstatin efficacy in 10-wk-old RT2 mice, we performed a trial targeting two sites of neovascularization in RT2 mice: (i) s.c. Matrigel plugs embedded with VEGF and (ii) hyperplastic/dysplastic pancreatic islets. We implanted Matrigel plugs infused with VEGF165 and FGF2 into 9-wk-old RT2 mice and analyzed the s.c. Matrigel plug 1 wk later. The data presented in Fig. 1B (and reproduced in Fig. 3A for ease of comparison) demonstrate a lack of effect on angiogenic switching in pancreatic neoplasias in tumstatin-treated RT2 mice. In contrast, the vascularization of s.c. Matrigel plugs implanted on RT2 mice was significantly inhibited by 1 wk of tumstatin peptide treatment (Fig. 3A). Previous analysis of Matrigel plugs demonstrated robust expression of β3 integrin on the VEGF-induced vasculature in the Matrigel (21), a vasculature presumably derived from the skin. β3 integrin expression is only minimally observed in the VEGF-associated vasculature of the angiogenic islets of the pancreatic bed (Fig. S2B).
Fig. 3.
Genetic deletion of β3 integrin abrogates activity of tumstatin peptide. (A) RT2 mice were implanted s.c. with Matrigel plugs at 9 wk of age and treated with tumstatin daily as described above for 1 wk. The Matrigel plugs were then harvested; six plugs were assayed in each group. (B) An intervention trial comparing tumstatin and endostatin peptides in RT2/β3 Integrin−/− mice treated from 10 to 13.5 wk of age. The tumstatin peptide (control, n = 7; tumstatin peptide, n = 7) did not prevent tumor growth in the absence of β3 integrin, whereas the endostatin peptide (control, n = 3; endostatin peptide, n = 4) significantly inhibited tumor growth in RT2/β3 integrin−/− mice. Results are shown as mean ± SEM; *P < 0.05.
If αVβ3 integrin is indeed the main antiangiogenic signaling receptor for tumstatin, then β3 integrin knockout mice should be refractory to therapeutic tumstatin. Therefore, we tested both tumstatin and endostatin peptides in therapeutic trials of RT2/β3integrin−/− mice. The endostatin peptide inhibited tumor growth in the RT2/β3integrin−/− mice (Fig. 3B), consistent with previous studies indicating that αVβ3 integrin is not an endostatin receptor. The tumstatin peptide had little effect in RT2/β3integrin−/− mice (Fig. 3B), in contrast with its effects in WT RT2 mice (Fig. 1B; reshown for ease of comparison in Fig. 3A) indicating that this integrin is a key component in the antiangiogenic activity of tumstatin.
Translational Applications of the Balance Hypothesis for Angiogenic Switching.
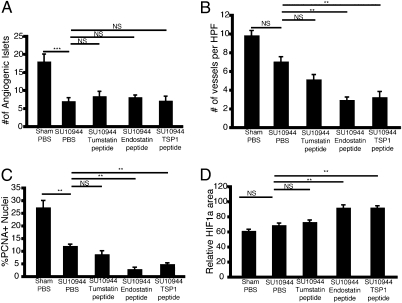
Together with the previous functional validation of multiple proangiogenic signals (22–24) it is evident that a balance mechanism involving inducer and inhibitor signals exists in the RT2 model of de novo multistage carcinogenesis, involved both in initial angiogenic switching and in the persistence of tumor angiogenesis during tumor growth and progression. This knowledge of mechanism raises the question of applicability to therapeutic targeting. We began exploring this prospect in a pharmacological combination trial. A prevention trial targeting the initial angiogenic switch was performed, in which the VEGF receptor inhibitor SU10944 (25) alone or in combinations with tumstatin peptide, endostatin peptide, or TSP1 peptide were used to treat RT2 mice. SU10944 proved to be a potent inhibitor of the angiogenic switch as indicated by a substantial decrease in the number of angiogenic islets (Fig. 4A). The combination of the various EAIs with SU10944 did not further reduce the onset of angiogenesis (Fig. 4A); notably, however, differences were observed in the vasculature associated with angiogenic islets compared with SU10944 treatment alone. Angiogenic islets of mice treated with SU10944 and endostatin peptide or TSP1 peptide were significantly less vascularized, displayed decreased cellular proliferation, and increased hypoxic response as shown by an up-regulation of hypoxia inducible factor 1 alpha (HIF-1α) in comparison with islets isolated from SU10944 alone or in combination with tumstatin peptide (Fig. 4 B–D and Fig. S4). Future therapeutic trials assessing survival and effects on tumor burden and histopathology in tumor-bearing animals are warranted.
Fig. 4.
Dual targeting of the angiogenic balance and angiogenic switching. Angiogenic switching was assessed by isolating and counting hemorrhagic pancreatic islets. (A) VEGFR1/2 inhibition with the small molecule SU10944 significantly decreased the number of islets undergoing the angiogenic switch compared with the sham-treated group. Analysis of isolated angiogenic islets revealed that the combination of endostatin or thrombospodin peptides with SU10944 reduced the number of vessels in hyperplastic/angiogenic islets (B), and the rate of tumor cell proliferation (C) as determined by the number of PCNA positive nuclei (DAPI) per high-powered (400×) field. (D) An increase in HIF-1α expression, as determined by the total area stained positive per high-powered (400×) field was observed. For all groups, n = 8. Results are shown as mean ± SEM; **P < 0.01, ***P < 0.001. See also Fig. S2.
Deletion of Tumstatin and TSP1 in p53−/− Mice Leads to Increased Tumor Burden and Reduced Survival.
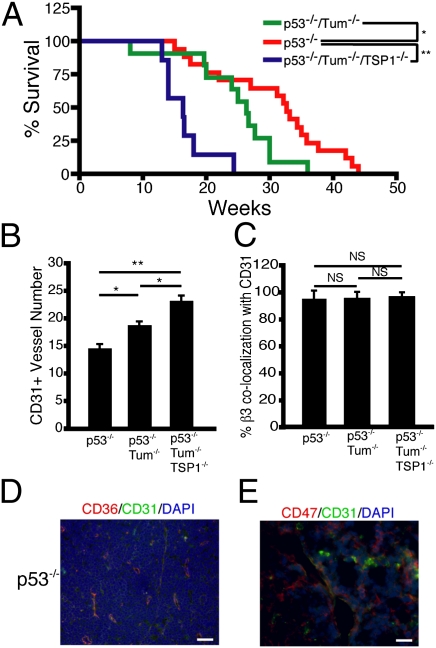
Seeking to continue generalizing the importance of endogenous angiogenesis inhibitors for tumor development and progression, we crossed the tumstatin−/− mice with p53−/− mice. Mice lacking the p53 tumor suppressor develop lymphomas and, to a lesser extent, angiosarcomas and sarcomas (26). Mice that were doubly deficient in p53 and tumstatin developed more lymphomas and angiosarcomas, and the mice died earlier than the littermate control p53−/− mice (Fig. 5A and Table S1). Analysis of the tumor spectrum in these mice at 3 mo of age indicates that tumstatin deficiency led to an increased occurrence of lymphoma: 81.8% in p53−/−/tumstatin−/− mice versus 66.7% in p53−/− mice (Table S1).
Fig. 5.
Tumstatin and/or TSP1 deficiency affect the phenotype and the tumor spectrum of p53-deficienct mice. (A) Survival curves of p53−/− (n = 14), p53−/−/tumstatin−/− (n = 11), and p53−/−/tumstatin−/−/TSP1−/− (n = 7) mice. Deficiency of tumstatin and TSP1 significantly decreased the lifespan of p53-deficient mice. (B) Lymphomas from p53−/−, p53−/−/tumstatin−/−, and p53−/−/tumstatin−/−/TSP1−/− were examined for vessel number by CD31 immunofluorescence. (C) Vessels from the lymphomas were also analyzed for colocalization of CD31 with β3 integrin. Expression of the thrombospondin receptors CD36 (D; 200× magnification) and CD47 (E; 400× magnification) were also observed in lymphomas of p53−/− mice. Results are shown as mean ± SEM; *P < 0.05, **P < 0.01. (Scale bar, 20 μM.) See also Fig. S3 and Table S1.
To begin assessing the possibility that multiple EAIs are serving as nonredundant barriers to tumorigenesis in this model, we intercrossed the p53−/−/tumstatin−/− mice with TSP1−/− mice to generate triple knockout mice. The p53−/−/tumstatin−/−/TSP1−/− mice had more lymphomas as well as a significantly reduced survival (Fig. 5A and Table S1).
The number of blood vessels increased in the tumstatin-deficient and the tumstatin + TSP1-deficient lymphomas compared with p53−/− lymphomas (Fig. 5B and Fig. S3). Quantification of CD31 and β3 integrin colocalization in the lymphomas revealed that most vessels expressed β3 integrin (Fig. 5C and Fig. S5). Expression of the thrombospondin receptors CD36 (Fig. 5D) and CD47 (Fig. 5E) was also observed in the endothelium of p53−/− lymphomas.
Discussion
We sought in this study to assess the balance hypothesis for the angiogenic switch and the persistence of angiogenesis during tumorigenesis in a prototypical mouse model of cancer. We investigated the role of the proangiogenic inducer VEGF-A for initial angiogenic switching, by analyzing incipient neoplasias in mice that are deficient of the VEGF-A gene in the oncogene-expressing β cells. In contrast to RT2/VEGFWT mice, the hyperproliferative lesions in RT2/VEGFRIPKO mice did not undergo angiogenic switching and were typified by regions of necrosis and hypoxia and increased apoptosis. This functional genetic test clearly confirms the importance of the inducing component of the angiogenic switch.
We then assessed the roles of angiogenesis inhibitors for the angiogenic switch, focusing on three: tumstatin, endostatin, and thrombospondin-1. Putative receptors for the antiangiogenic signals of endostatin and TSP1 were expressed throughout the stages of islet tumorigenesis, whereas a key component of the tumstatin receptor, the β3 integrin, was poorly expressed in incipient neoplasias and variably induced in solid tumors. We focused on the most well-established receptors for these three EAIs in regard to their function of inhibiting angiogenesis, recognizing that we may not have addressed all of the possible receptor candidates for these proteins.
Having established the presence of the receptors and hence the possibility for antiangiogenic signaling, we asked whether the initial angiogenic switch and the maintenance of subsequent tumor angiogenesis were responsive to these endogenous angiogenesis inhibitors, by elevating their levels pharmacologically. A peptide constituting endostatin activity and a core fragment of TSP1 both inhibited angiogenic switching as well as subsequent tumor angiogenesis and tumor growth, revealing the potential activity of their signals in counterbalancing VEGF-A signaling in vivo. Tumstatin did not affect initial angiogenesis in incipient neoplasias, but was able to impair angiogenesis and growth of solid tumors, consistent with the expression profile of the αvβ3 receptor. Next, we asked whether the endogenous forms of these inhibitors serve to regulate the angiogenic switch. Although the number of vessels was increased in the angiogenic islets in both collagen gene knockout settings but not in tumstatin-deficient RT2 mice (Fig. S6), none of these genetic deficiencies affected the initial angiogenic switch in premalignant lesions. In contrast, the absence of each EAI (and of the αvß3 integrin receptor) increased the growth rate of the solid tumors and reduced survival.
Our analyses of integrins α5 and β3 revealed that, in RT2 mice, integrin β3 is up-regulated from undetectable levels in the normal islets and premalignant tumor stages to elevated and then variable levels in small versus large tumors, as is also reported in some human cancers (27). We further evaluated the functional importance of β3 integrin in this situation by deleting it in the context of the RT2 mice. Deficiency of the β3 integrin, either early in tumor development or after genetic ablation, makes the tumors resistant to tumstatin treatment. The fact that pharmacological tumstatin inhibits the neovascularization of the Matrigel plugs in RT2 mice, but without concurrently impacting angiogenic switching in premalignant lesions in the pancreas (Figs. 1B and 3), further suggests that differential expression of vascular integrins in different states and/or bodily locations correlates with the susceptibility of that endothelium to particular endogenous inhibitors. Whereas β3 integrin-null RT2 tumors are resistant to tumstatin treatment, the ability of endostatin to inhibit tumor growth in the absence of αvβ3, suggests that the observed insensitivity to tumstatin is not a reflection of global vascular resistance. Interestingly, hypoxia is more prevalent in the tumor stages than in the pretumor stages of islet tumorigenesis, concordant with up-regulation of β3 (Fig. S7), which suggests that the HIF-dependent hypoxia response program might be capable of directly or indirectly inducing expression of β3 in endothelial cells and rendering the later tumors susceptible to inhibition by tumstatin. The overall functions of αvβ3 integrin in angiogenesis remain incompletely understood, with some results suggesting that αvβ3 and αvβ5 integrins can promote angiogenesis and that their inhibition can be antiangiogenic (28–30), whereas others suggest that these two integrins are not necessary for angiogenesis and may even act as negative regulators (10, 31–36). Both interpretations may be correct in different contexts; but the results presented here and elsewhere (21, 37) show clearly that inhibition of angiogenesis by tumstatin requires αvβ3 integrin.
To begin generalizing the conclusion from the RT2 model and from traditional xenotransplant models (21, 38) that EAIs serve as intrinsic barriers to tumorigenesis, we assessed the relationship between tumstatin and its receptor αVβ3 integrin in the p53−/− mouse (26, 39). We found that, in the absence of tumstatin, p53−/− mice had increased incidence and growth of lymphomas, consistent with the fact that most of the vessels in lymphomas expressed αVβ3; deletion of both tumstatin and TSP1 in the p53 null mice led to even more aggressive lymphomas and a further decrease in lifespan.
Our data suggest a biphasic regulation of the angiogenic switch. The initial activation of angiogenesis in incipient neoplasias is largely dependent on inductive signaling by VEGF-A. Only one of the three potentially counterbalancing inhibitory circuits was present and active; the other two do not serve as natural barriers to initial VEGF-dependent induction of angiogenesis, as revealed by the gene knockouts. The second phase, operative in solid tumors, involves the maintenance and intensification of angiogenesis in support of expansive tumor growth. In this phase, all three EAIs function as components of the angiogenic regulatory mechanism, counterbalancing the proangiogenic signals, as evidenced by the effects both of their pharmacological elevation, which impair tumor growth, and of their ablation, which accelerates tumor growth and diminishes survival of cancer-bearing mice.
Materials and Methods
Additional details are provided in SI Materials and Methods.
Transgenic Mice and Drug Treatment.
All mice were on a C57BL/6 background and were maintained at the Beth Israel Deaconess Medical Center, Massachusetts Institute of Technology animal facilities, and Osaka Medical Center for Cancer and Cardiovascular Diseases under standard conditions. Animal studies were approved by the institutional animal care and use committees of Beth Israel Deaconess Medical Center, Massachusetts Institute of Technology, and Osaka Medical Center for Cancer and Cardiovascular Diseases.
Peptides of human endostatin, human tumstatin, and TSP1 were used for prevention trials (5.5–10 wk) and intervention trials (10–13.5 wk) with RT2 transgenic mice. To provide a circulating concentration of 10 μM,a dose of 1 mg/gram/day, 0.5 mg/gram/day, and 1.1 mg/gram/day for the tumstatin, endostatin, and TSP1 inhibitors, respectively, was used in all of the pharmacological intervention experiments. At least six mice were used for each group.
For combination trials, SU10944 was prepared as previously described (40) and EAI reagents were given as described above. Mice received treatment twice a week by gavage starting at 6.5 wk of age and ending at 8.5 wk of age. Eight mice were used in each treatment group.
Assessment of the Angiogenic Islets and Tumor Burden.
Angiogenic islets were isolated at 10 wk by collagenase digestion of the excised pancreas followed by Ficoll gradient separation and counted under a dissecting microscope (41). Tumors were microdissected from freshly excised pancreata at 13.5 wk. Tumor volume was measured and the formula [volume = 0.52 × (width)2 × length] was applied. Tumor burden per mouse equals the average sum of the total tumor volume. Comparisons between groups were analyzed by t test. P values less than 0.05 were considered statistically significant.
Supplementary Material
Acknowledgments
We thank James Christensen for the VEGF receptor inhibitor SU10944. This research was supported by grants from the National Institutes of Health (NIH) DK55001 (to R.K.), CA125550 (to R.K.), RO1CA17007 (to R.O.H), U54CA126515 (to R.O.H), and CA92644 (to J.L.) and the Champalimaud Metastasis Program (R.K.), and by a fellowship from the Stop and Shop Pediatric Tumor Foundation (to L.X.). M.D. was a 2007 United Negro College Fund/Merck Postdoctoral Fellow and supported by National Cancer Institute Training Grant 5T32CA081156-08 from the NIH. R.O.H. is also supported by the Howard Hughes Medical Institute. T.M. was supported by German Research Foundation (DFG) MU 2298/2-2. Work performed at Univeristy of California, San Francisco was supported by grants from the National Cancer Institute (to D.H.) and the William K. Bowes Jr. Charitable Foundation, and in Osaka by a Grant-in-Aid for Scientific Research from the Japanese Society for the Promotion of Science (to M.I.).
Footnotes
The authors declare no conflict of interest.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1105041108/-/DCSupplemental.
References
- 1.Nyberg P, Xie L, Kalluri R. Endogenous inhibitors of angiogenesis. Cancer Res. 2005;65:3967–3979. doi: 10.1158/0008-5472.CAN-04-2427. [DOI] [PubMed] [Google Scholar]
- 2.Hanahan D, Folkman J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell. 1996;86:353–364. doi: 10.1016/s0092-8674(00)80108-7. [DOI] [PubMed] [Google Scholar]
- 3.Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. 2003;9:669–676. doi: 10.1038/nm0603-669. [DOI] [PubMed] [Google Scholar]
- 4.Lundkvist A, Lee S, Iruela-Arispe L, Betsholtz C, Gerhardt H. Growth factor gradients in vascular patterning. Novartis Found Symp. 2007;283:194–201. doi: 10.1002/9780470319413.ch15. discussion 201–206, 238–241. [DOI] [PubMed] [Google Scholar]
- 5.Grant MA, Kalluri R. Structural basis for the functions of endogenous angiogenesis inhibitors. Cold Spring Harb Symp Quant Biol. 2005;70:399–410. doi: 10.1101/sqb.2005.70.017. [DOI] [PubMed] [Google Scholar]
- 6.Karumanchi SA, et al. Cell surface glypicans are low-affinity endostatin receptors. Mol Cell. 2001;7:811–822. doi: 10.1016/s1097-2765(01)00225-8. [DOI] [PubMed] [Google Scholar]
- 7.Sudhakar A, et al. Human tumstatin and human endostatin exhibit distinct antiangiogenic activities mediated by alpha v beta 3 and alpha 5 beta 1 integrins. Proc Natl Acad Sci USA. 2003;100:4766–4771. doi: 10.1073/pnas.0730882100. [DOI] [PMC free article] [PubMed] [Google Scholar] [Research Misconduct Found]
- 8.Kalluri R. Basement membranes: Structure, assembly and role in tumour angiogenesis. Nat Rev Cancer. 2003;3:422–433. doi: 10.1038/nrc1094. [DOI] [PubMed] [Google Scholar]
- 9.Silva R, D'Amico G, Hodivala-Dilke KM, Reynolds LE. Integrins: The keys to unlocking angiogenesis. Arterioscler Thromb Vasc Biol. 2008;28:1703–1713. doi: 10.1161/ATVBAHA.108.172015. [DOI] [PubMed] [Google Scholar]
- 10.Hodivala-Dilke K. alphavbeta3 integrin and angiogenesis: A moody integrin in a changing environment. Curr Opin Cell Biol. 2008;20:514–519. doi: 10.1016/j.ceb.2008.06.007. [DOI] [PubMed] [Google Scholar]
- 11.O'Reilly MS, et al. Endostatin: An endogenous inhibitor of angiogenesis and tumor growth. Cell. 1997;88:277–285. doi: 10.1016/s0092-8674(00)81848-6. [DOI] [PubMed] [Google Scholar]
- 12.Marneros AG, Olsen BR. The role of collagen-derived proteolytic fragments in angiogenesis. Matrix Biol. 2001;20:337–345. doi: 10.1016/s0945-053x(01)00151-2. [DOI] [PubMed] [Google Scholar]
- 13.Maeshima Y, et al. Distinct antitumor properties of a type IV collagen domain derived from basement membrane. J Biol Chem. 2000;275:21340–21348. doi: 10.1074/jbc.M001956200. [DOI] [PubMed] [Google Scholar]
- 14.Ren B, Yee KO, Lawler J, Khosravi-Far R. Regulation of tumor angiogenesis by thrombospondin-1. Biochim Biophys Acta. 2006;1765:178–188. doi: 10.1016/j.bbcan.2005.11.002. [DOI] [PubMed] [Google Scholar]
- 15.Maeshima Y, et al. Extracellular matrix-derived peptide binds to alpha(v)beta(3) integrin and inhibits angiogenesis. J Biol Chem. 2001;276:31959–31968. doi: 10.1074/jbc.M103024200. [DOI] [PubMed] [Google Scholar]
- 16.Miao WM, et al. Thrombospondin-1 type 1 repeat recombinant proteins inhibit tumor growth through transforming growth factor-beta-dependent and -independent mechanisms. Cancer Res. 2001;61:7830–7839. [PubMed] [Google Scholar]
- 17.Wickstrom SA, Alitalo K, Keski-Oja J. An endostatin-derived peptide interacts with integrins and regulates actin cytoskeleton and migration of endothelial cells. J Biol Chem. 2004;279:20178–20185. doi: 10.1074/jbc.M312921200. [DOI] [PubMed] [Google Scholar]
- 18.Bergers G, Javaherian K, Lo KM, Folkman J, Hanahan D. Effects of angiogenesis inhibitors on multistage carcinogenesis in mice. Science. 1999;284:808–812. doi: 10.1126/science.284.5415.808. [DOI] [PubMed] [Google Scholar]
- 19.Folkman J, Watson K, Ingber D, Hanahan D. Induction of angiogenesis during the transition from hyperplasia to neoplasia. Nature. 1989;339:58–61. doi: 10.1038/339058a0. [DOI] [PubMed] [Google Scholar]
- 20.Astrof S, et al. Direct test of potential roles of EIIIA and EIIIB alternatively spliced segments of fibronectin in physiological and tumor angiogenesis. Mol Cell Biol. 2004;24:8662–8670. doi: 10.1128/MCB.24.19.8662-8670.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hamano Y, et al. Physiological levels of tumstatin, a fragment of collagen IV alpha3 chain, are generated by MMP-9 proteolysis and suppress angiogenesis via alphaV beta3 integrin. Cancer Cell. 2003;3:589–601. doi: 10.1016/s1535-6108(03)00133-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Inoue M, Hager JH, Ferrara N, Gerber HP, Hanahan D. VEGF-A has a critical, nonredundant role in angiogenic switching and pancreatic beta cell carcinogenesis. Cancer Cell. 2002;1:193–202. doi: 10.1016/s1535-6108(02)00031-4. [DOI] [PubMed] [Google Scholar]
- 23.Compagni A, Wilgenbus P, Impagnatiello MA, Cotten M, Christofori G. Fibroblast growth factors are required for efficient tumor angiogenesis. Cancer Res. 2000;60:7163–7169. [PubMed] [Google Scholar]
- 24.Brantley DM, et al. Soluble Eph A receptors inhibit tumor angiogenesis and progression in vivo. Oncogene. 2002;21:7011–7026. doi: 10.1038/sj.onc.1205679. [DOI] [PubMed] [Google Scholar]
- 25.Patel N, et al. A selective and oral small molecule inhibitor of vascular epithelial growth factor receptor (VEGFR)-2 and VEGFR-1 inhibits neovascularization and vascular permeability. J Pharmacol Exp Ther. 2003;306:838–845. doi: 10.1124/jpet.103.052167. [DOI] [PubMed] [Google Scholar]
- 26.Donehower LA, et al. Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature. 1992;356:215–221. doi: 10.1038/356215a0. [DOI] [PubMed] [Google Scholar]
- 27.Felding-Habermann B, et al. Involvement of tumor cell integrin alpha v beta 3 in hematogenous metastasis of human melanoma cells. Clin Exp Metastasis. 2002;19:427–436. doi: 10.1023/a:1016377114119. [DOI] [PubMed] [Google Scholar]
- 28.Brooks PC, et al. Integrin alpha v beta 3 antagonists promote tumor regression by inducing apoptosis of angiogenic blood vessels. Cell. 1994;79:1157–1164. doi: 10.1016/0092-8674(94)90007-8. [DOI] [PubMed] [Google Scholar]
- 29.Mahabeleshwar GH, Feng W, Phillips DR, Byzova TV. Integrin signaling is critical for pathological angiogenesis. J Exp Med. 2006;203:2495–2507. doi: 10.1084/jem.20060807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Desgrosellier JS, Cheresh DA. Integrins in cancer: Biological implications and therapeutic opportunities. Nat Rev Cancer. 2010;10:9–22. doi: 10.1038/nrc2748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hynes RO. A reevaluation of integrins as regulators of angiogenesis. Nat Med. 2002;8:918–921. doi: 10.1038/nm0902-918. [DOI] [PubMed] [Google Scholar]
- 32.Hynes RO. Cell-matrix adhesion in vascular development. J Thromb Haemost. 2007;5(Suppl 1):32–40. doi: 10.1111/j.1538-7836.2007.02569.x. [DOI] [PubMed] [Google Scholar]
- 33.Reynolds AR, et al. Stimulation of tumor growth and angiogenesis by low concentrations of RGD-mimetic integrin inhibitors. Nat Med. 2009;15:392–400. doi: 10.1038/nm.1941. [DOI] [PubMed] [Google Scholar]
- 34.Reynolds AR, et al. Elevated Flk1 (vascular endothelial growth factor receptor 2) signaling mediates enhanced angiogenesis in beta3-integrin-deficient mice. Cancer Res. 2004;64:8643–8650. doi: 10.1158/0008-5472.CAN-04-2760. [DOI] [PubMed] [Google Scholar]
- 35.Reynolds LE, et al. Enhanced pathological angiogenesis in mice lacking beta3 integrin or beta3 and beta5 integrins. Nat Med. 2002;8:27–34. doi: 10.1038/nm0102-27. [DOI] [PubMed] [Google Scholar]
- 36.van der Flier A, et al. Endothelial alpha5 and alphav integrins cooperate in remodeling of the vasculature during development. Development. 2010;137:2439–2449. doi: 10.1242/dev.049551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Maeshima Y, et al. Tumstatin, an endothelial cell-specific inhibitor of protein synthesis. Science. 2002;295:140–143. doi: 10.1126/science.1065298. [DOI] [PubMed] [Google Scholar]
- 38.Sund M, et al. Function of endogenous inhibitors of angiogenesis as endothelium-specific tumor suppressors. Proc Natl Acad Sci USA. 2005;102:2934–2939. doi: 10.1073/pnas.0500180102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jacks T, et al. Tumor spectrum analysis in p53-mutant mice. Curr Biol. 1994;4:1–7. doi: 10.1016/s0960-9822(00)00002-6. [DOI] [PubMed] [Google Scholar]
- 40.Pietras K, Hanahan D. A multitargeted, metronomic, and maximum-tolerated dose “chemo-switch” regimen is antiangiogenic, producing objective responses and survival benefit in a mouse model of cancer. J Clin Oncol. 2005;23:939–952. doi: 10.1200/JCO.2005.07.093. [DOI] [PubMed] [Google Scholar]
- 41.Parangi S, Dietrich W, Christofori G, Lander ES, Hanahan D. Tumor suppressor loci on mouse chromosomes 9 and 16 are lost at distinct stages of tumorigenesis in a transgenic model of islet cell carcinoma. Cancer Res. 1995;55:6071–6076. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.