Abstract
Purpose:
Graft versus host disease is the major complication of allogeneic bone marrow transplantation and limits the therapeutic efficacy of this modality. While the role of natural regulatory T cells (nTregs) in attenuating GVHD has been extensively examined, the ability of induced regulatory T cells (iTregs) to mitigate GVHD is unknown. The purpose of this study was to examine the ability of in vitro and in vivo-induced Tregs to abrogate GVHD.
Experimental Design:
We examined the ability of in vitro-differentiated and in vivo-induced Tregs to reduce the severity of GVHD in a clinically relevant mouse model of BMT. The effect of blockade of interleukin 6 signaling on the efficacy of these Treg populations was also studied.
Results:
In vitro-differentiated iTregs fail to protect mice from lethal GVHD even when administered at high Treg: effector T cell ratios. Lack of GVHD protection was associated with loss of Foxp3 expression and in vivo reversion of these cells to a proinflammatory phenotype characterized by secretion of IFN-γ. Phenotypic reversion could not be abrogated by blockade of IL-6 signaling or by in vitro exposure of iTregs to all-trans-retinoic acid. In contrast, the in vivo induction of iTregs was significantly augmented by IL-6 blockade and this resulted in reduced GVHD.
Conclusion:
Instability of Foxp3 expression limits the utility of adoptively transferred iTregs as a source of cellular therapy for the abrogation of GVHD. Blockade of IL-6 signaling augments the ability of in vivo-induced Tregs to prevent GVHD, but has no effect on in vitro-differentiated iTregs.
Keywords: regulatory T cells, GVHD, IL-6, iTregs
INTRODUCTION
Regulatory T cells (Tregs) are a critical component of the adaptive immune system and play a pivotal role in controlling inflammatory responses as well as preventing the development of autoimmunity (1) There are two subsets of Tregs, both of which are highly dependent on Foxp3 expression for their suppressive function, and have unique and overlapping characteristics. Natural Tregs (nTregs) differentiate in the thymus (2), are most commonly characterized by the constitutive expression of activation markers such as CD25, CTLA-4, CD134, CD103 and glucocorticoid-induced tumor necrosis factor receptor (GITR), and require high affinity interactions with self peptides (3-6). In contrast, induced Tregs (iTregs) arise from CD4+Foxp3− conventional T cells that upregulate Foxp3 in the periphery upon activation in the context of TGF-β (7,8) Moreover, iTregs can be generated in vitro by co-culture with TGF-β after stimulation through the T cell receptor and provision of IL-2 (9-11).
Graft versus host disease (GVHD) is the major complication of allogeneic bone marrow transplantation. GVHD is characterized by the expansion and differentiation of donor alloreactive T cells, the release of proinflammatory cytokines, and the recruitment of other effector cell populations, leading to the damage of host target organs (12-14). A number of studies have shown that the reconstitution of Tregs, which plays a pivotal role in regulating donor T cell-mediated alloresponses, is severely impaired in both acute and chronic GVHD (15-18). This premise is further supported by studies which have demonstrated that the adoptive transfer of nTregs is able to significantly attenuate GVHD severity, indicating that exogenous supplementation of these cells is an effective strategy for the re-establishment of transplantation tolerance (19-23). The clinical application of this approach, however, is constrained by the low frequency of these cells in the peripheral blood that makes these cells difficult to isolate (24). iTregs, on the other hand, can be generated in large numbers in vitro and are able to suppress alloreactive T cells responses (25), raising the possibility that these cells might also be efficacious in vivo. However, the role of these cells in GVHD biology has yet to be fully examined, and many questions remain regarding their stability and ability to protect against disease.
iTregs can also be generated in vivo in response to inflammatory events (26,27), although the specific contribution of these cells relative to nTregs is not known. Moreover, the extent to which the in vivo conversion of these cells from naïve T cell precursors is affected by the proinflammatory environment that accompanies most pathological conditions has also not been defined. We have recently shown that IL-6 is one of the proinflammatory cytokines that is induced during GVHD and inhibits the generation of Tregs (28), but the effect of inflammatory cytokines in the local microenvironment on the conversion of iTregs from conventional T cells remains uncertain. The goal of the current study therefore was to define the role of iTregs during GVHD. Specifically, we sought to determine the efficacy of iTregs in the prevention of GVHD, whether there was differential protection afforded by in vitro versus in vivo-induced Tregs, and to what extent protection or lack thereof was attributable to the stability of Foxp3 expression and the underlying proinflammatory cytokine milieu.
METHODS
Mice.
C57BL/6 (B6) (H-2b, Thy1.2+), B6.PL (H-2b, Thy 1.1+) and Balb/c (H-2d, Thy1.2+) mice were bred in the Animal Resource Center (ARC) at the Medical College of Wisconsin (MCW) or purchased from Jackson Laboratories (Bar Harbor, ME). Foxp3EGFP mice (backcrossed to the B6 background for six generations) in which the Foxp3 gene is coupled to the enhanced green fluorescent protein (EGFP) were obtained from Dr. Calvin Williams (MCW) and have been previously described (29). All animals were housed in the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC)-accredited ARC of the Medical College of Wisconsin. Experiments were all carried out under protocols approved by the MCW Institutional Animal Care and Use Committee. Mice received regular mouse chow and acidified tap water ad libitum.
Reagents.
Anti-IL-6R antibody (MR-16-1) is a rat IgG antibody that has been previously described (28). Animals received a loading dose of 2 mg intravenously on the day of transplantation and then were treated with 0.5 mg weekly by intraperitoneal injection. Antibody was re-suspended in phosphate-buffered saline (PBS) prior to injection. Rat IgG (Jackson Immunoresearch Laboratories, West Grove, PA) was used as a control and administered at the same dose and schedule as MR-16-1.
Bone Marrow Transplantation.
Bone marrow (BM) was flushed from donor femurs and tibias with Dulbecco’s modified media (DMEM) (Gibco-BRL, Carlsbad, CA) and passed through sterile mesh filters to obtain single cell suspensions. BM was T-cell depleted (TCD) in vitro with anti-Thy1.2 monoclonal antibody plus low toxicity rabbit complement (C-six Diagnostics, Mequon, WI). The hybridoma for 30-H12 (anti-Thy1.2, rat IgG2b) antibody was purchased from the American Type Culture Collection (Rockville, MD). Host mice were conditioned with total body irradiation (TBI) administered as a single exposure at a dose rate of 82 cGy using a Shepherd Mark I Cesium Irradiator (J.L. Shepherd and Associates, San Fernando, CA). Irradiated recipients received a single intravenous injection in the lateral tail vein of BM with or without added spleen cells.
Cell Sorting and Flow Cytometry.
Spleen and peripheral lymph node cells were collected from Foxp3EGFP mice and sorted on a FACSAria (Becton-Dickenson, Mountain View, CA). Sort purity for these studies consistently averaged 98-99%. Spleen, liver, lung, colon and lymph node cells from transplant recipients were labeled with monoclonal antibodies (mAb) conjugated to fluorescein isothiocyanate (FITC), phycoerythrin (PE), PE-Cy5, or APC that were obtained from BD Biosciences (San Diego, CA). Cells were analyzed on a FACSCalibur or LSRII flow cytometer with Cellquest or FACSDiva software (Becton-Dickinson). Data were analyzed using FlowJo software (Treestar, Ashland, Oregon).
Cell Culture.
To generate iTregs in vitro, sorted CD4+ EGFP− T cells from Foxp3EGFP mice were cultured in complete RPMI with immobilized anti-CD3 antibody (2.5 μg/ml), soluble anti-CD28 antibody (1 μg/ml), IL-2 (100 U/ml) and TGF-β (10 ng/ml) for three days. This typically resulted in the conversion of 60-70% of cells. GFP-expressing cells were then re-sorted for use in BMT studies. In some experiments, all-trans retinoic acid (ATRA) (Sigma-Aldrich, St Louis, MO) was added to cultures at a concentration of 10 nM for the entire three days. In other experiments, cells were labeled with Annexin-PE and 7-AAD to assess the percentage of apoptotic cells in expanded cultures.
Cell Isolation.
To isolate lamina propria lymphocytes, pooled colons were incubated in Hank’s Balanced Salt Solution (HBSS) buffer (Gibco-BRL) supplemented with 2% fetal bovine serum (FBS), EDTA (0.05 mM) and 15 μg/ml dithiothreitol (Invitrogen, Carlsbad, CA) at 37°C for 30 min and subsequently digested in a solution of collagenase D (Roche Diagnostics, Mannheim, Germany, 1 mg/ml) in DMEM with 2% FBS for 75 min at 37°C for five days. The resulting cell suspension was then layered on a 44%/67% Percoll gradient (Sigma, St. Louis, MO). Liver and lung lymphocytes were isolated by collagenase D digestion followed by layering on a Percoll gradient.
Mixed lymphocyte culture (MLC).
Thy1.2+ cells (1 × 105) were purified from B6 spleens using the MACS magnetic cell separation system (Miltenyi Biotech, Auburn, CA) and co-cultured with 5 × 104 Balb/c dendritic cell-enriched stimulator cells in U-bottomed microwell plates (Becton Dickinson, Lincoln Park, NJ) at 37°C. Stimulator cells were obtained by digestion of spleens with collagenase D (1 mg/ml) followed by positive selection of CD11c+ “dendritic cells” using the MACS system. Nonirradiated stimulator cells were then seeded into microwell plates. Flow-sorted iTreg or nTreg cells were added at varying ratios to wells containing T and dendritic cells. One microcurie of 3H-thymidine was added to triplicate wells for the final 12-18 hours prior to harvest. Thymidine incorporation was assessed using a Wallac 1450 Microbeta liquid scintillation counter (Perkin Elmer, Shelton, CT). Control wells consisted of responders, stimulators, and Tregs alone.
Histological Analysis.
Representative samples of liver, colon, and lung were obtained from transplant recipients and fixed in 10% neutral-buffered formalin. Samples were then embedded in paraffin, cut into five micron thick sections and stained with hematoxylin and eosin. A semiquantitative scoring system was employed to account for histological changes in the colon, liver, and lung as previously described (30). All slides were coded and read in a blinded fashion.
Intracellular Cytokine Staining.
Lymphocytes isolated from spleen, liver, lung and colon were stimulated with 50 ng/ml phorbol 12-myristate 13-acetate (PMA) (Sigma) and 750 ng/ml ionomycin (Calbiochem, La Jolla, CA) for 1 hour and then incubated with GolgiStop (BD Pharmingen) for an additional 4 hours. Cells were surface stained with Pacific Blue anti-CD4 and APC anti-Thy1.2, and then intracellularly stained with PE-labeled antibody to IL-17 and Alexa Fluor 700-labeled antibody to IFN-γ. All antibodies were obtained from Becton Dickenson.
Statistics.
Group comparisons of Tregs and T cell populations, pathology scores, and thymidine incorporation were performed using the Mann Whitney U test. Survival curves were constructed using the Kaplan-Meier product limit estimator and compared using the log rank rest. A p value ≤ 0.05 was deemed to be significant in all experiments.
RESULTS
iTregs are equipotent to nTregs in their in vitro suppressive ability.
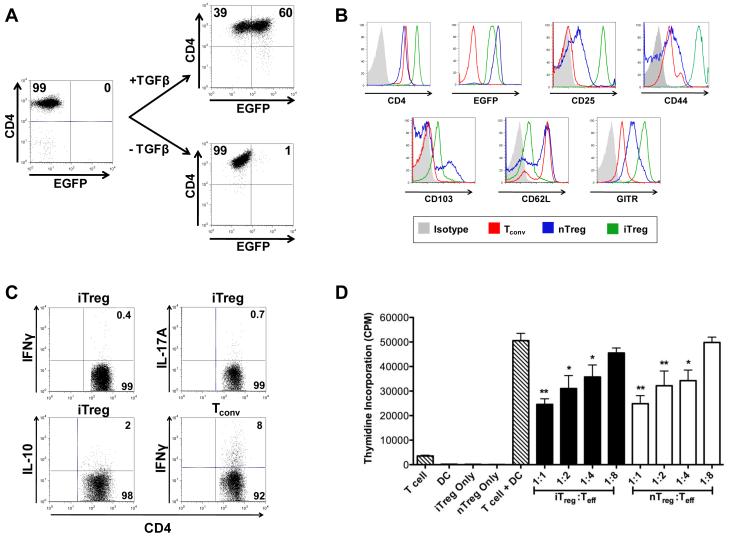
CD4+ EGFP− T cells were sorted from Foxp3EGFP mice and cultured with immobilized anti-CD3 antibody, soluble anti-CD28 antibody, IL-2 and TGF-β. After 72 hours, approximately 60% of the cells expressed EGFP (Figure 1A). Phenotypic analysis demonstrated that iTregs expressed higher levels of CD4, CD25, and CD44 on their surface as compared to nTregs. Conversely, iTregs expressed lower levels of CD62L, which is typically downregulated upon cell activation, and CD103 than nTregs (Figure 1B). Intracellular cytokine staining revealed that iTregs had negligible secretion of IFN-γ, IL-17A, or IL-10 (Figure 1C). To confirm functionality, iTregs were sorted and compared to freshly isolated nTregs from Foxp3EGFP animals for their ability to suppress an alloresponse. Sorted iTregs were stained with Annexin and 7-AAD to determine whether the cell culture process resulted in any apoptotic cells. Only 3% of cells were observed to be Annexin+ 7-AAD− indicating no significant apoptosis in the cultured cells (data not shown). nTregs were obtained by sorting CD4+ Foxp3+ T cells from Foxp3EGFP mice. FACS-sorted iTregs and nTregs were then added to cultures at varying ratios of Tregs to effector T cells (Teff). We observed that iTregs and nTregs suppressed alloreactive T cell proliferation to the same extent and in a dose-dependent manner, indicating that these cells have comparable suppressive capability in vitro (Figure 1D).
Figure 1. Induced Tregs are equipotent to nTregs in the suppression of alloreactive T cell responses.
(A). Sorted CD4+ EGFP− T cells from Foxp3EGFP mice were cultured with immobilized anti-CD3 (2.5 μg/ml), soluble anti-CD28 (1 μg/ml), and IL-2 (100 U/ml) with or without TGF-β (10 ng/ml). The percentage of CD4+ EGFP+ T cells after three days in culture with or without TGF-β is shown in the dot plots. (B). Cell surface expression of EGFP, CD25, CD44, CD103, CD62L, and GITR on in vitro-differentiated iTregs and nTregs obtained from the spleen and lymph nodes of Foxp3EGFP mice. (C). Intracellular cytokine staining for IFN-γ and IL-17 secretion in iTregs after culture in anti-CD3, anti-CD28 and TGF-β for 3-4 days. IFN-γ secretion from naïve CD4+ T cells (Tconv) stimulated with anti-CD3 and anti-CD28 antibodies is shown as a control. (D). Purified B6 Thy1.2+ T cells (1 × 105) were cultured with Balb/c CD11c+ dendritic cells (5 × 104) in the presence of varying ratios of iTregs (black bars) or nTregs (white bars) for five days in triplicate wells. Control wells are depicted as hatched bars. Data are presented as mean cpm ± SEM and are representative of one of three experiments with similar results. Statistics: * p ≤ 0.05, ** p < 0.01.
iTregs do not protect against lethal GVHD.
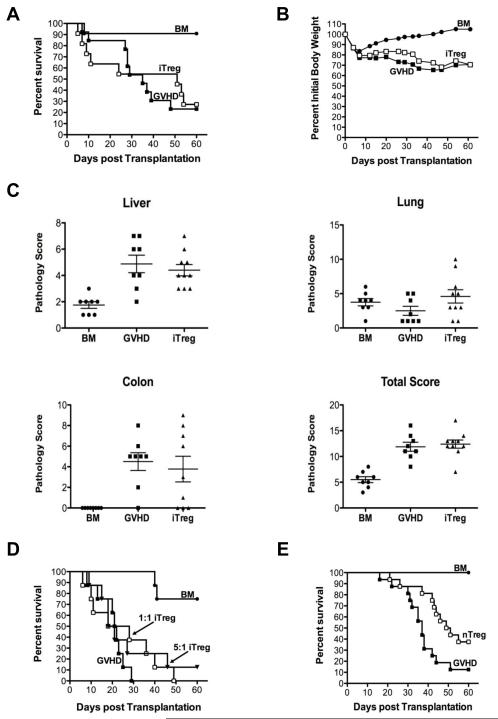
To determine in vivo functionality of in vitro-differentiated Tregs, we examined whether iTregs were able to suppress GVHD. Lethally irradiated Balb/c mice were transplanted with TCD B6 BM cells alone, TCD BM plus whole splenocytes, or TCD BM and spleen cells along with in vitro-differentiated Tregs at a 1:1 ratio. There was no difference in survival between GVHD control animals and mice that were supplemented with iTregs (Figure 2A), indicating that iTregs did not attenuate GVHD severity. We also observed no difference in serial weight curves between these two cohorts of animals (Figure 2B). In order to more fully examine the ability of iTregs to suppress GVHD, we repeated these studies with a reduced T cell dose so that mice in all groups would survive and be able to be examined for GVHD-associated pathology. Histological analysis of GVHD target organs 24 days post transplantation revealed that iTregs effected no reduction in pathological damage in the colon, liver or lung of transplant recipients (Figure 2C), further confirming that these cells had no significant protective role and that there was no differential effect on individual tissues. To assess whether lack of protection was attributable to an inadequate dose, we repeated these studies at an iTreg: effector T cell ratio of 5:1. Again, there was no significant difference in long term survival in animals that received an escalated does of iTregs (Figure 2D) indicating that the inability of these cells to abrogate GVHD did not appear to be due to an inadequate cell number. These results were in contrast to what was observed when mice were transplanted with nTregs. Specifically, transplantation of nTregs at a 1:1 ratio with conventional T cells significantly prolonged survival compared to animals that received no Tregs (Figure 2E). Collectively, these data demonstrate that, in contrast to nTregs, iTregs are unable to prevent GVHD-associated mortality and pathological damage.
Figure 2. In vitro generated iTregs fail to protect from lethal GVHD.
(A,B). Lethally irradiated (900 cGy) Balb/c mice were transplanted with TCD B6 BM alone (8 × 106) (●, n=11) or together with B6 spleen cells adjusted to yield a T cell dose of 0.4-0.6 × 106 αβ T cells. Animals transplanted with adjunctive spleen cells received either no additional cells (■, n=13) or in vitro-differentiated Tregs in a 1:1 ratio with naïve αβ T cells (□, n=11). Overall survival (A) and the percentage of initial body weight over time (B) are depicted. Data are cumulative results derived from three independent experiments. (C). Pathological damage in the colon, liver, and lung at day 24 post transplantation using a semi-quantitative scale as described in “Histologic analysis.” Mice (n= 8-10/group) were similarly transplanted as in A with the exception that that the αβ T cells dose was 0.5 × 106. Data are presented as the mean ± SEM. (D). Lethally irradiated Balb/c mice were transplanted with TCD B6 BM alone (●, n=8) or together with B6 spleen cells adjusted to yield a T cell dose of 0.6 × 106 αβ T cells. Animals transplanted with adjunctive spleen cells received either no additional cells (■, n=8) or in vitro-differentiated Tregs at a 1:1 (□, n= 8) or 5:1 (▼, n= 8) ratio with naïve αβ T cells. Data are cumulative results of two independent experiments. (E). Lethally irradiated Balb/c mice were transplanted with B6 BM alone (●, n=15), or B6 BM plus spleen cells adjusted to yield a dose of 0.6 × 106 αβ T cells. Animals transplanted with adjunctive spleen cells received no additional cells (■, n= 16) or 0.6 × 106 nTregs (□, n= 16). Overall survival is depicted. Data are cumulative results of four experiments. Statistics: * p ≤ 0.05, ** p < 0.01.
Foxp3-expressing iTregs have limited in vivo persistence in GVHD recipients.
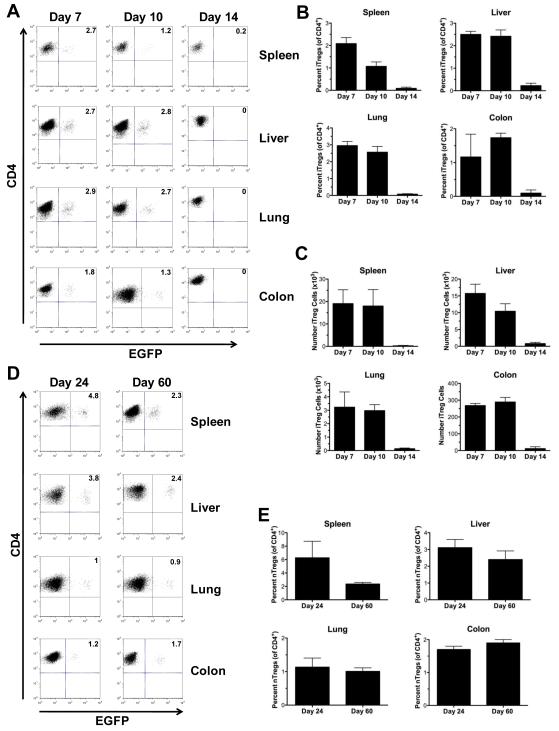
Studies were then conducted to understand why in vitro-differentiated Tregs were unable to protect mice from lethal GVHD. We reasoned that possible explanations for the lack of protection from GVHD by iTregs might be that these cells had limited survival or that down regulation of molecules such as CD62L might alter trafficking to GVHD target organs. To address this question, lethally irradiated Balb/c mice were transplanted with TCD B6 BM and spleen cells along with an equivalent number of iTregs. Cohorts of mice were sacrificed on days 7, 10 and 14 post-BMT and GVHD target organs were examined for the presence of EGFP+ cells. iTregs were detectable in the spleen, colon, liver and lung of all recipients on days 7 and 10 days post transplantation (Figure 3A). Both the percentage (Figure 3B) and absolute number (Figure 3C) of these cells was similar at both time points. Notably, iTregs were also present in the peripheral lymph nodes on day 10 indicating that, despite low CD62L expression, these cells could traffic into this tissue site early post transplantation (Supplementary Figure 1). By day 14, however, iTregs were essentially undetectable in all GVHD target organs. This was in contrast to what was observed when nTregs were transplanted into recipient mice. In these studies, examination of GVHD target organs demonstrated persistence of nTregs in all tissue sites for up to 60 days post transplantation with ~1-3% of all CD4+ T cells expressing EGFP (Figures 3D-E). Thus, Foxp3-expressing iTregs had limited in vivo persistence in GVHD recipients that were not protected from pathological damage.
Figure 3. Foxp3-expressing iTregs have limited in vivo persistence in GVHD recipients.
(A-C) Lethally irradiated Balb/c mice were transplanted with TCD B6 BM, B6 spleen cells (adjusted to yield 0.6 × 106 αβ T cells), and 0.6 × 106 iTregs. Mice (n=8-12/group) were sacrificed at 7, 10, or 14 days post transplantation. (A). Representative dot plots gated on CD4+ cells depicting EGFP+ iTregs in the specified tissue sites. (B,C). Relative and absolute number of iTregs in the spleen, liver, lung and colon at the defined time points post transplantation. Data are presented as the mean ± SEM and are the cumulative results from three independent experiments. (D,E). Lethally irradiated Balb/c mice were transplanted with TCD B6 BM, B6 spleen cells (adjusted to yield 0.6 × 106 αβ T cells), and 0.6 × 106 nTregs. Mice (n = 4-8/group) were sacrificed at 24 or 60 days post transplantation. (D). Representative dot plots gated on CD4+ cells depicting EGFP+ nTregs in the specified tissue sites. (E). The relative percentage of nTregs in the spleen, liver, lung and pooled colon samples is shown. Data are presented as the mean ± SEM and are the cumulative results from two experiments.
iTregs revert to a proinflammatory phenotype early after transplantation.
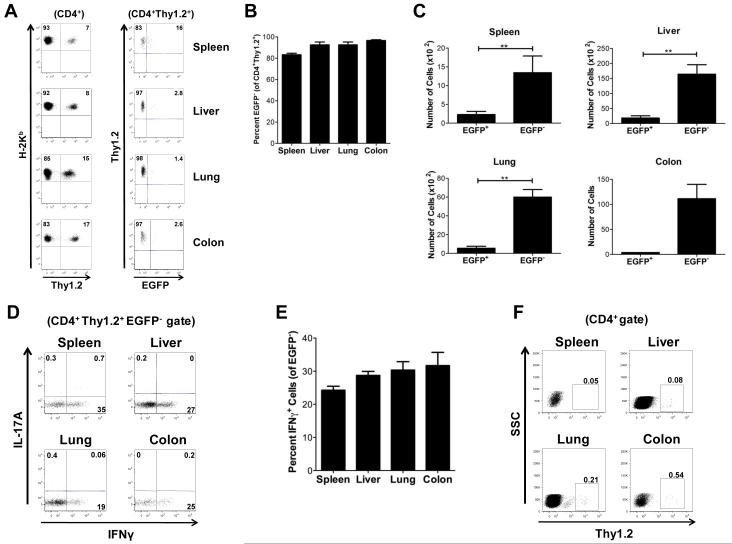
While the inability to detect iTregs early post transplantation could have been due to reduced in vivo survival, an alternative explanation was that these cells persisted in recipient animals but had lost expression of Foxp3. In fact, plasticity in the Treg lineage has been recently described whereby Foxp3-expressing cells have been shown to be capable of reverting to a proinflammatory phenotype (31-33). To determine if the latter explanation was valid, lethally irradiated Balb/c mice were transplanted with B6.PL (Thy 1.1) BM alone, B6.PL BM plus B6.PL whole splenocytes or B6.PL BM and spleen cells plus B6 Foxp3EGFP (Thy1.2+) iTregs. Using iTregs from congenic Thy1.2 mice allowed us to specifically identify the presence of these cells in otherwise fully donor-engrafted recipients and determine whether these cells had survived but lost EGFP expression. Cohorts of mice were sacrificed at days 3, 5, 7, 10, 14 and 21 and examined for the presence of Thy1.2+ cells that retained EGFP expression. We observed that at early time points (days 3, 5, and 7), there was a population of residual Thy1.2+ that were of host origin (H-2d+)(data not shown). By day 10, however, mice were completely donor T cell engrafted (Figure 4A) and we therefore selected this time point for our analysis to eliminate any potentially confounding host populations. A population of CD4+ Thy1.2+ cells was easily discernible in all of the tissue examined (Figure 4A). The majority of these cells were EGFP− (Figure 4B), although a small population of Thy1.2+ EGFP+ cells was present in all organs (Figure 4C). To determine whether these revertant cells had acquired a proinflammatory phenotype, cells were stained for secretion of IFN-γ and IL-17. Whereas these cells produced a negligible amount of IL-17A, 20-35% of the revertant iTregs from all tissue sites secreted IFN-γ (Figures 4D-E). This was noteworthy given that iTregs did not secrete either cytokine immediately after in vitro conversion (Figure 1C) and was evidence that these cells had acquired a proinflammatory phenotype in vivo. Further longitudinal analysis revealed that iTregs were undetectable by day 21 post transplantation (data not shown). To determine whether in vitro-differentiated iTregs had the potential to induce GVHD, we transplanted animals with these cells alone in the absence of conventional donor T cells. iTregs were detectable in animals 10 days post transplantation in all tissues sites (Supplementary Figure 2A). However, mice transplanted with these cells had no mortality and survival was comparable to that observed in animals transplanted with BM cells alone (Supplementary Figure 2B), indicating that these cells did not appear to contribute significantly to the induction of GVHD.
Figure 4. Lack of protection is attributable to reversion of iTregs to a proinflammatory phenotype in vivo.
(A-E) Lethally irradiated Balb/c mice (n=12) were transplanted with 8 × 106 B6.PL (Thy1.1) BM and spleen cells (adjusted to yield a dose of 0.6 × 106 αβ T cells) with 0.6 × 106 iTregs (Thy1.2). Spleen, liver, lung, and pooled colon samples were analyzed for Thy1.2+ cells to determine EGFP expression. (A) Representative dot plots gated on CD4+ T cells depicting the percentage of Thy1.2+ H-2Kb+ cells that expressed EGFP 10 days after transplantation. (B,C) The relative percentage of CD4+ Thy1.2+ EGFP− cells is shown in B, and the absolute number of EGFP+ and EGFP− cells in the specified tissue sites is depicted in C. Data are presented as the mean ± SEM and are the cumulative results of three independent experiments. (D) Representative dot plots and (E) percentages of CD4+Thy1.2+EGFP− cells that secreted IFN-γ or IL-17A as determined by intracellular cytokine staining. (F) Lethally irradiated Balb/c mice were transplanted with B6.PL BM, B6.PL spleen cells (adjusted to yield a dose of 0.6 × 106 αβ T cells) and 1.8 × 104 B6 Thy1.2+ CD4+ EGFP− cells. Representative dot plots gated on CD4+ T cells showing Thy1.2+ expressing cells in the spleen, liver, lung, and colon from animals 10 days after transplantation. Statistics: * p<0.05, **p<0.01.
A possible confounding factor in our interpretation that phenotypic reversion had occurred in vivo was that small numbers of contaminating Thy1.2+ non-GFP expressing cells may have been present in the iTreg sort and then expanded in recipient animals. Since the average sort purity was 98-99%, we assumed a worst case scenario whereby 3% of the transferred iTreg population was contaminated by non-iTreg cells. This amounted to an absolute number of 1.8 × 104 cells. To address this question, lethally irradiated Balb/c mice were transplanted with B6.PL BM and whole splenocytes plus 1.8 × 104 Thy1.2+ CD4+ EGFP− cells from Foxp3EGFP mice. On day ten post transplantation, we observed only a negligible population of these cells in the spleen or GVHD target organs (i.e. ≤ 0.5%) (Figure 4F), indicating that the revertant cells were not the result of contaminating non-iTreg cells in the sort population.
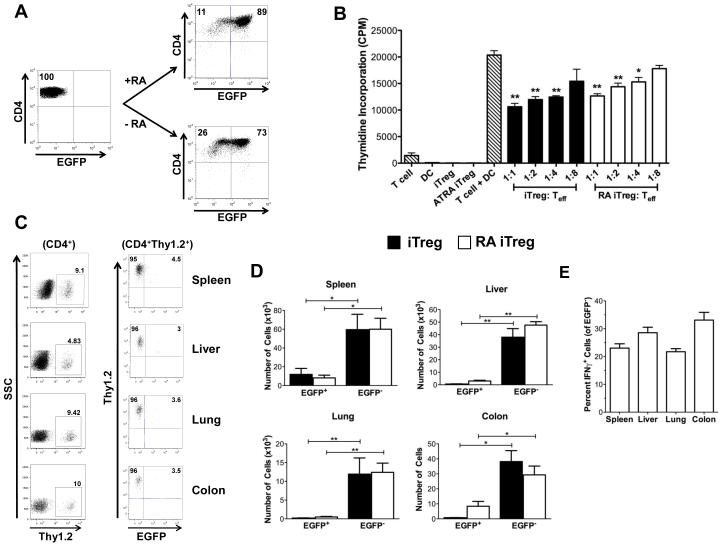
In vitro exposure of iTregs to ATRA does not prevent phenotypic reversion.
Based on prior studies that demonstrated that in vitro exposure to ATRA can enhance the induction of Foxp3 in conventional T cells (34,35), we examined whether ATRA could stabilize Foxp3 expression in iTregs after their adoptive transfer into recipient animals. To examine this question, CD4+ Foxp3EGFP− T cells were first cultured in TGF-β and IL-2 alone or in the presence of ATRA. Exposure to ATRA resulted in a modest 10-15% increase in Foxp3-expressing cells compared to cells cultured in the absence of ATRA (Figure 5A). To determine whether RA-iTregs were more effective at suppressing an alloresponse than iTregs, RA-iTregs and iTregs were added to MLCs at varying ratios of Tregs to effector T cells. Both RA-iTregs and iTregs were observed to be equally suppressive as determined by 3H-thymidine incorporation (Figure 5B). Since RA exposure had no additive effect on the suppressive function of iTregs in vitro, we examined whether these cells had any suppressive role in vivo. Lethally irradiated Balb/c mice were transplanted with B6.PL BM and whole splenocytes (0.6 × 106 αβ T cells) plus 0.6 × 106 Thy1.2+ Foxp3EGFP+ RA-iTregs or iTregs. Ten days after transplantation, the majority of RA-iTregs had lost GFP expression (Figure 5C). Notably, while the number of RA-iTreg-derived Thy1.2+EGFP− cells was significantly greater than the number of Thy1.2+EGFP+ cells, RA-iTregs had a comparable number of revertant cells as the iTreg control group (Figure 5D). Moreover, revertant cells from the RA-iTreg population produced IFN-γ to the same extent as revertant cells from the control iTreg population (Figure 5E versus Figure 4E). Thus, in vitro exposure to RA did not prevent phenotypic reversion in vivo.
Figure 5. All-trans retinoic acid does not prevent iTreg reversion.
(A) CD4+EGFP− cells were converted to iTregs in the presence or absence of 10 nM ATRA. The percentage of CD4+ EGFP+ T cells after three days in culture with (RA-iTregs) or without (iTregs) ATRA is shown. (B) B6 Thy1.2+ T cells (1 × 105) were cultured with Balb/c CD11c+ dendritic cells (5 × 104) in the presence of varying ratios of iTregs that had been converted in the absence (black bars) or presence (white bars) of ATRA for five days. Controls are depicted as hatched bars. Data are presented as mean cpm ± SEM and are representative of one of three experiments with similar results (C-E) Lethally irradiated Balb/c mice (n=8) were transplanted with B6.PL BM, B6.PL spleen cells (adjusted to yield 0.6 × 106 αβ T cells) and 0.6 × 106 B6 iTregs or RA-iTregs. (C) Representative dot plots gated on CD4+ T cells showing the percentage of Thy1.2+ cells in spleen, liver, lung, and pooled colon 10 days after transplantation. (D) Absolute numbers of CD4+Thy1.2+ cells that were EGFP+ and EGFP− cells in the specified tissue sites. (E) Percentage of CD4+ Thy1.2+ EGFP− cells that secreted IFN-γ derived from mice transplanted with RA-iTregs. Data are cumulative results of two experiments. Statistics: * p<0.05, **p<0.01.
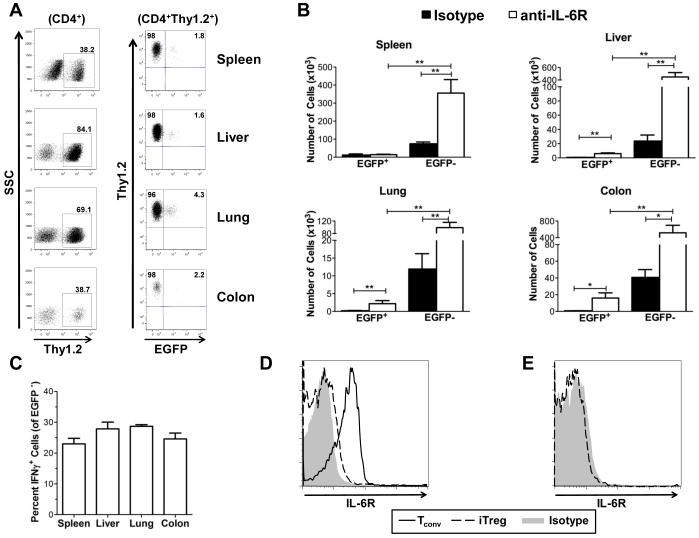
Blockade of IL-6 signaling does not prevent phenotypic reversion in iTregs.
A defining characteristic of GVHD is the overproduction of inflammatory cytokines that are able to modulate the intensity of the disease (36-38). We previously have shown that IL-6 is one inflammatory cytokine that has a critical role in modulating the balance between effector and regulatory T cells during GVHD (28). In fact, blockade of IL-6 signaling is able to significantly reduce GVHD-associated pathological damage by increasing nTreg numbers and decreasing the absolute number of proinflammatory T cells. Based on these prior data, we considered that over production of IL-6 might be a factor that contributes to the phenotypic reversion of iTregs in vivo. To test this hypothesis, animals were transplanted with B6.PL BM and spleen cells along with B6 iTregs. Cohorts of mice were then administered either an anti-IL-6R or an isotype control antibody as described in Methods. On day 10 after transplantation, mice were sacrificed and the percentage of revertant iTregs was determined. Compared to mice treated with isotype antibody, there was a striking increase in the number and percentage of Thy1.2+ cells in animals treated with anti-IL-6R antibody (Figures 6A and 6B). This resulted in a significant increase in the absolute number of both EGFP+ and EGFP− cells compared to isotype control-treated mice. Notably, however, the majority of these Thy1.2+ cells also lacked expression of EGFP (Figure 6A) and a similar percentage secreted IFN-γ (Figure 6C), indicating that blockade of IL-6 signaling did not prevent phenotypic reversion in vivo.
Figure 6. Blockade of IL-6R signaling does not prevent iTreg reversion but increases Thy1.2+ cell numbers.
(A-C) Lethally irradiated Balb/c mice were transplanted with B6.PL BM, B6.PL spleen cells (adjusted to yield 0.6 × 106 αβ T cells) and 0.6 × 106 B6 in vitro-differentiated iTregs. Mice were then administered anti-IL-6R (n=11) or isotype control (n=9) antibody on days 0 and 7 as described in Methods. Data are cumulative results of three experiments. (A) Representative dot plots gated on CD4+ T cells showing the percentage of Thy1.2+ cells that expressed EGFP in spleen, liver, lung, and colon samples 10 days after transplantation. (B) Absolute number of CD4+ Thy1.2+ EGFP+ and EGFP− cells in the specified tissue sites from animals treated with isotype (black bars) or anti-IL-6R antibody (white bars). (C) Percentage of CD4+ Thy1.2+ EGFP− cells that secreted IFN-γ in each tissue. (D) Representative histograms showing IL-6R expression on naïve CD4+ T cells (solid line) and in vitro-differentiated iTregs (dashed line) prior to transplantation. Isotype control is in gray. (E). IL-6R expression on iTregs obtained from the spleen of mice ten days post transplantation. Isotype control is in gray. Statistics: * p<0.05, **p<0.01.
To understand why treatment with anti-IL-6R antibody resulted in the preferential expansion of Thy1.2+ T cells, we examined IL-6R expression on naïve CD4+ T cells and iTregs pre and post transplantation. We observed that the IL-6R was expressed on naïve CD4+ T cells from donor animals but was not present on in vitro-differentiated iTregs that had been cultured in IL-2 and TGF-β (Figure 6D). Similarly, cells obtained from the spleen of animals 10 days post transplantation revealed a lack of IL-6R expression on revertant cells which resulted in a significant increase in the number of these cells in all tissues (Figure 6E). Thus, the expansion of revertant iTregs relative to donor-derived Thy1.1+ CD4+ T cells appeared to be due to a lack of receptor expression that made these cells resistant to antibody blockade.
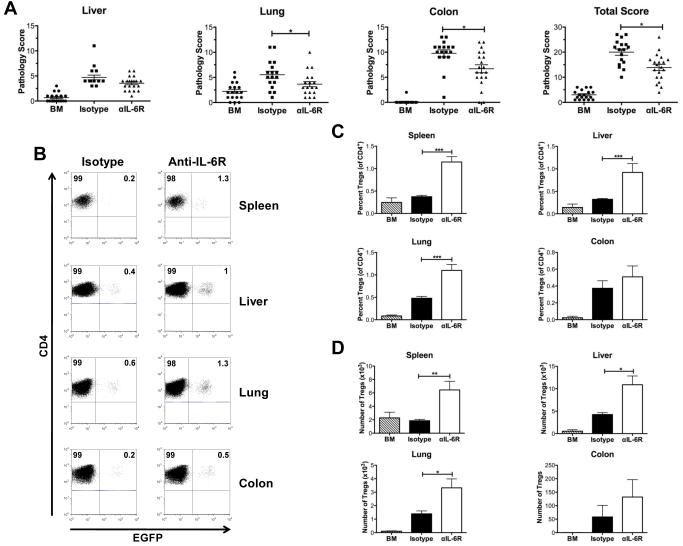
Antibody blockade of the IL-6R augments the in vivo induction of iTregs.
In vivo, conventional T cells are able to differentiate into either proinflammatory or regulatory cells depending upon the cytokine milieu to which they are exposed. During GVHD, there is an inflammatory environment that directs differentiation along TH1 and TH17 cytokine phenotypes and inhibits the in vivo induction of Tregs from CD4+ Foxp3− T cells (28). Since treatment with anti-IL-6R antibody was unable to prevent phenotypic reversion of in vitro-differentiated iTregs, we examined whether in vivo induced Tregs were also resistant to IL-6 blockade. To address this question, lethally irradiated Balb/c mice were transplanted with B6 Rag-1 BM cells and sorted CD4+ Foxp3EGFP− T cells, and then treated with either anti-IL-6R or control antibody. Anti-IL-6R antibody-treated animals had a significant overall reduction in pathological damage when compared to control-treated mice (Figure 7A). Specifically, there was reduced pathology in the lung and colon in treated animals, with a trend for reduced damage observed in the liver. Commensurate with the attenuation of GVHD-related pathological damage, there was a significant increase in the relative and absolute number of iTregs in GVHD tissue sites when examined 4 weeks post transplantation (Figures 7B-7D). Collectively, these data demonstrated that, in contrast to what was observed with in vitro-differentiated iTregs, blockade of IL-6 signaling was able to augment the in vivo induction of iTregs and this was associated with a significant reduction in the severity of GVHD.
Figure 7. Antibody blockade of the IL-6R augments generation of iTregs in vivo and attenuates GVHD.
Lethally irradiated Balb/c mice were transplanted with B6 Rag-1 BM (8 × 106) alone (n= 18) (hatched bars) or together with sorted CD4+ EGFP− T cells (0.2 × 106) from Foxp3EGFP mice. Cohorts of mice transplanted with CD4+ EGFP− T cells were then administered rat IgG isotype control (n= 17) (black bars) or anti-IL-6R antibody (n= 20) (white bars) once weekly for four weeks as described in Methods. Mice in both groups were sacrificed 27-28 days post transplantation. Data are cumulative results from four independent experiments. (A). Pathological damage in the colon, liver and lung using a semiquantitative scoring system as detailed in Methods. (B). Representative dot plots showing percentage of in vivo-induced iTregs in the gated CD4+ T cell population from transplant recipients treated with either isotype control or anti-IL-6R antibody. (C). Percentage and (D) absolute number of iTregs in the spleen, liver, lung and colon of animals administered Rag-1 BM alone (hatched bars, n=14), or Rag-1 BM and CD4+ EGFP− T cells and then treated with control (black bars, n=14) or anti-IL-6R antibody (white bars, n=16). Data are presented as the mean ± SEM and are the cumulative results from three experiments. Statistics: * p ≤ 0.05, ** p < 0.01.
DISCUSSION
The use of Tregs to facilitate transplantation tolerance is an emerging area in clinical allogeneic stem cell transplantation. Based on preclinical murine studies that demonstrated that the adoptive transfer of Tregs could mitigate GVHD, several groups of investigators have begun clinical trials to determine if this approach will be efficacious in humans (39,40). Obtaining sufficient numbers of these cells for transfer into individual patients as well as insuring that the acquired cell population is not contaminated by potentially alloreactive non-Treg cells have been ongoing challenges with this strategy (24). An alternative approach to circumvent these potential obstacles is predicated on the in vitro expansion of Tregs that can be converted from conventional CD4+ T cells in the presence of IL-2 and TGF-β. Under these conditions, large numbers of in vitro-derived iTregs can be obtained for the subsequent administration into recipients. This strategy, however, is contingent upon the underlying assumption that these cells will function in a similar manner to nTregs with respect to suppression of GVH reactivity. In the current study, we examined this question to determine the functional capability of these cells to ameliorate GVHD in vivo. These studies demonstrated that in vitro-differentiated iTregs were not effective for the prevention of GVHD despite their ability to suppress alloreactive T cells responses in vitro. The failure to mitigate GVHD was attributable to limited in vivo survival, instability of Foxp3 expression and the subsequent reversion of these cells to a TH1 cytokine phenotype.
Our results, along with those of other investigators, suggest that the ability of in vitro-differentiated iTregs to suppress disease may be a function of the magnitude of the inflammatory milieu. For example, Haribhai et al (26) showed that these cells could protect mice from colitis in a T cell transfer model. Similarly, Zheng et al (41) demonstrated that in vitro-differentiated iTregs could prolong survival in an autoimmune model characterized by a lupus-like syndrome. Both models, however, are characterized by less inflammation than is observed in GVHD where the administration of a lethal TBI conditioning regimen exacerbates the inflammatory milieu induced by a T cell-mediated alloresponse. In that regard, our results are consistent with those of Koenecke et al (42) who recently showed that alloantigen-specific iTregs generated in vitro after co-culture with activated dendritic cells had a negligible effect on protection from lethal GVHD. The current study, however, extends these findings by demonstrating that the loss of Foxp3 expression is associated with the acquisition of an inflammatory phenotype characterized by IFN-γ secretion with no evidence of IL-17 production. Thus, in the presence of an inflammatory environment induced by GVHD, there is a reversion of a suppressive T cell population to one with a TH1 cytokine phenotype.
One of the primary observations of these studies was the limited in vivo survival of iTregs which were essentially undetectable after two weeks, even after accounting for revertant cells. This was observed in all GVHD target organs examined indicating no preferential localization of these cells at sites of pathological damage. This finding is consistent with an earlier report by Selvaraj and colleagues (25) who demonstrated that only a small percentage of iTregs were able to survive in vivo after the adoptive transfer into immunocompetent animals. In that study, iTregs disappeared from nearly all organs within the first two weeks with the exception of the bone marrow and lymph nodes. This was distinctly different from what we observed when nTregs were transferred into recipient animals where Foxp3+ cells were observed for up to eight weeks post-BMT. In the latter case, the stability of Foxp3 expression correlated with protection from lethal GVHD.
Prior studies have shown that RA is able to induce differentiation of Tregs from conventional T cells through both direct and indirect effects. Hill and colleagues reported that RA promoted iTreg generation by relieving the inhibition that was mediated indirectly by CD4+ CD44hi T cells (41). Conversely, others have noted that RA can directly enhance TGF-β-mediated Foxp3 induction while inhibiting the generation of TH17 cells (34,35). Based on these studies, we examined whether RA could enhance the stability of Foxp3-expressing iTregs in vivo and prevent phenotypic reversion in lethally irradiated transplant recipients. Although treatment with RA increased iTreg conversion, the in vitro exposure to this agent did not prevent phenotypic reversion in vivo after adoptive transfer. These results differ from what was observed in a murine model where animals immunized with ovalbumin had less reversion if they received RA-iTregs (44). The degree of inflammation and the extent of reversion in control animals, however, were notably less in that model and suggest that other cytokines and or environmental factors are responsible for the phenotypic reversion which occurs after adoptive transfer of iTregs during GVHD.
We reasoned that IL-6 might be one cytokine that contributed to instability of Foxp3 expression given prior studies which have shown that IL-6 deleteriously affects the generation of iTregs induced by TGF-β by blocking Foxp3 expression (45). This has been shown to occur by up regulation of the TGF-β inhibitor SMAD7 (8). IL-6 has also been shown to play a pivotal role in driving the differentiation of naïve T cells to become TH17 cells and in inhibiting the generation of CD4+ Foxp3+ from CD4+ Foxp3− T cells (46). Moreover, Horwitz and colleagues (47) have reported that iTregs are more resistant to the inhibitory effects of IL-6 compared to nTregs. Thus, we considered that transfer of these cells might be more effective if the effects of IL-6 which is significantly increased during GVHD (28) could be blunted. Blockade of IL-6 signaling, however, had no effect on stabilization of Foxp3 expression or preventing phenotypic reversion. Further analysis revealed that in vitro-differentiated iTregs did not express the IL-6R after either in vitro conversion or in vivo isolation of this same population after transfer. Down regulation of the IL-6R on iTregs has been shown to inhibit signaling though IL-6 (41), providing an explanation for why these cells were not affected by antibody administration. Down regulation of the receptor is also a potential explanation for the absence of IL-17 secretion in revertant iTregs as the inability to respond to IL-6 would prevent TH17 differentiation (41).
One of the main findings was the observation that in vitro and in vivo-generated iTregs appear to be differentially regulated by IL-6. Specifically, blockade of IL-6 signaling was able to augment the in vivo induction of Tregs from the conventional CD4+ T cell pool. Under these conditions, mice treated with antibody blockade had an increased number of Foxp3+ cells in all tissue sites due to enhanced in vivo conversion which correlated with augmented GVHD protection. Since naive T cells express the IL-6R, we surmise that, in contrast to in vitro-differentiated iTregs, antibody administration was able to direct the differentiation of these cells towards a regulatory pathway due to their susceptibility to IL-6 signaling blockade. These results have direct clinical relevance given the availability of a tocilizumab which is a humanized version of this antibody and has been FDA-approved for the treatment of patients with rheumatoid arthritis. Thus, administration of this antibody may be a means to augment Treg development in vivo without the need for more time consuming and costly cellular therapy approaches.
In summary, these studies demonstrate that in vitro-differentiated iTregs are ineffective at attenuating the severity of GVHD. This lack of protection is characterized by instability of Foxp3 expression which results in reversion to an inflammatory TH1 cytokine phenotype. Reversion is not due to increased production of IL-6 nor is in vitro exposure to retinoic acid able to stabilize iTregs, indicating that other mechanisms are responsible for this unstable phenotype. These results have implications for the potential use of in vitro-differentiated iTregs for the abrogation of GVHD and suggest that additional studies that are focused on augmenting the stability of Foxp3 expression in these cells is warranted before their use is contemplated in the clinical setting. Finally, blockade of IL-6 signaling is an effective approach to augment the generation of iTregs in vivo which may be a clinically feasible strategy to enhance Treg-mediated suppression and reduce GVHD severity.
TRANSLATIONAL RELEVANCE.
Allogeneic bone marrow transplantation (BMT) is the most potent form of immunotherapy directed against hematological malignancies. However, the success of this therapeutic modality is limited by the development of graft versus host disease (GVHD) which is the major complication of allogeneic BMT. The adoptive transfer of regulatory T cells is an emerging clinical strategy to prevent GVHD, but the relative role of natural (nTregs) versus induced Tregs (iTregs) is not defined. This has implications for the use of these cells as cellular therapy for GVHD prevention. In this study, we demonstrate that in vitro-expanded iTregs are much less potent than nTregs at preventing GVHD. This is due to the loss of Foxp3 expression and reversion to a proinflammatory phenotype after in vivo transfer. These cells, however, can be induced in vivo after blockade of IL-6 signaling and potently suppress GVHD. These results indicate that the conditions under which iTregs are generated as well as the corresponding proinflammatory cytokine milieu in which they function are critical factors in their ability to mitigate GVHD.
Supplementary Material
Supplementary Figure 1: In vitro-differentiated iTregs are detectable in the peripheral lymph nodes early post transplantation. Lethally irradiated Balb/c mice (n=5) were transplanted with B6.PL BM, B6.PL spleen cells (adjusted to yield 0.6 × 106 αβ T cells) and 0.6 × 106 B6 in vitro-differentiated iTregs. Mice were sacrificed 10 days post transplantation, and axillary and inguinal lymph nodes were obtained for pooled analysis. (A). Representative dot plot gated on CD4+ cells depicting the percentage of EGFP+ cells. (B,C). The percentage and absolute number of CD4+ T cells that expressed EGFP in the peripheral draining lymph nodes. Data are shown as the mean ± SEM and represent replicate mice from one experiment.
Supplementary Figure 2: In vitro-differentiated iTregs do not induce lethal GVHD in the absence of conventional T cells. Lethally irradiated Balb/c mice were transplanted with B6.PL BM and 0.6 × 106 B6 in vitro-differentiated iTregs. (A). Representative dot plot showing the percentage of H-2Kb Thy 1.2+ and Thy1.2+ EGFP+ cells present in the spleen, liver, and lung from animals sacrificed 10 days post transplantation. (B). Overall survival from mice transplanted with B6.PL BM cells alone (□, n=5) or together with 0.6 × 106 B6 in vitro-differentiated iTregs (●, n=5).
Acknowledgments
Grant Information This research was supported by grants from the National Institutes of Health (HL064603, HL081650 and DK083358) (W.R.D) and by an award from the Midwest Athletes Against Childhood Cancer Fund (Milwaukee, WI) (W.R.D.).
REFERENCES
- 1.Wing K, Sakaguchi S. Regulatory T cells exert checks and balances on self tolerance and autoimmunity. Nat Immunol. 2010;11:7–13. doi: 10.1038/ni.1818. [DOI] [PubMed] [Google Scholar]
- 2.Itoh M, Takahashi T, Sakaguchi N, et al. Thymus and autoimmunity: production of CD4+ CD25+ naturally anergic and suppressive T cells as a key function of the thymus in maintaining immunologioc self-tolerance. J Immunol. 1999;162:5317–5326. [PubMed] [Google Scholar]
- 3.Sakaguchi S, Sakaguchi N, Asano M, Itoh M, Toda M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor a chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J Immunol. 1995;160:1151–1164. [PubMed] [Google Scholar]
- 4.McHugh RS, Whitters MJ, Piccirillo CA, et al. CD4+ CD25+ immunoregulatory T cells: gene expression analysis reveals a functional role for the glucocorticoid-induced TNF receptor. Immunity. 2002;16:311–323. doi: 10.1016/s1074-7613(02)00280-7. [DOI] [PubMed] [Google Scholar]
- 5.Jordan MS, Boesteanu A, Reed AJ, et al. Thymic selection of CD4+ CD25+ regulatory T cells induced by an agonist self-peptide. Nat Immunol. 2001;2:301–306. doi: 10.1038/86302. [DOI] [PubMed] [Google Scholar]
- 6.Pacholczyk R, Ignatowicz H, Kraj P, Ignatowicz L. Origin and T cell receptor diversity of Foxp3+ CD4+ CD25+ T cells. Immunity. 2006;25:249–259. doi: 10.1016/j.immuni.2006.05.016. [DOI] [PubMed] [Google Scholar]
- 7.Fu S, Zhang N, Yopp A, et al. TGF-β induces Foxp3+ T regulatory cells from CD4+ CD25− precursors. Am J Transplant. 2004;4:1614–1627. doi: 10.1111/j.1600-6143.2004.00566.x. [DOI] [PubMed] [Google Scholar]
- 8.Fantini M, Becker C, Monteleone G, Pallone F, Galle P, Neurath M. Cutting edge: TGF-β induces a regulatory phenotype in CD4+ CD25− T cells through Foxp3 induction and down regulation of Smad7. J Immunol. 2004;172:5149–5153. doi: 10.4049/jimmunol.172.9.5149. [DOI] [PubMed] [Google Scholar]
- 9.Yamagiwa S, Gray JD, Hashimoto S, Horwitz DA. A role for TGF-beta in the generation and expansion of CD4+ CD25+ regulatory T cells from human peripheral blood. J Immunol. 2001;166:7282–7289. doi: 10.4049/jimmunol.166.12.7282. [DOI] [PubMed] [Google Scholar]
- 10.Chen W, Jin W, Hardegen N, et al. Conversion of peripheral CD4+ CD25− naïve T cells to CD4+ CD25+ regulatory T cells by TGF-beta induction of transcription factor Foxp3. J Exp Med. 2003;198:1875–1886. doi: 10.1084/jem.20030152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zheng SG, Wang J, Wang P, Gray JD, Horwitz DA. IL-2 is essential for TGF-β to convert naïve CD4+ CD25− cells to CD25+ Foxp3+ regulatory T cells and for expansion of these cells. J Immunol. 2007;178:2018–2027. doi: 10.4049/jimmunol.178.4.2018. [DOI] [PubMed] [Google Scholar]
- 12.Hill GR, Crawford JM, Cooke KR, Brinson YS, Pan L, Ferrara JL. Total body irradiation and acute graft versus host disease: the role of gastrointestinal damage and inflammatory cytokines. Blood. 1997;90:3204–3213. [PubMed] [Google Scholar]
- 13.Xun CQ, Thompson JS, Jennings CD, Brown SA, Widmer MB. Effect of total body irradiation, busulfan-cyclophosphamide, or cyclophosphamide conditioning on inflammatory cytokine release and development of acute and chronic graft versus host disease in H-2 incompatible transplanted SCID mice. Blood. 1994;83:2360–2367. [PubMed] [Google Scholar]
- 14.Ferrara JL, Reddy P. Pathophysiology of graft versus host disease. Semin Hematol. 2006;43:3–10. doi: 10.1053/j.seminhematol.2005.09.001. [DOI] [PubMed] [Google Scholar]
- 15.Miura Y, Thoburn CJ, Bright EC, et al. Association of Foxp3 regulatory gene expression with graft versus host disease. Blood. 2004;104:2187–2193. doi: 10.1182/blood-2004-03-1040. [DOI] [PubMed] [Google Scholar]
- 16.Zorn E, Kim HT, Lee SJ, et al. Reduced frequency of Foxp3+ CD4+ CD25+ regulatory T cells in patients with chronic graft versus host disease. Blood. 2005;106:2903–2911. doi: 10.1182/blood-2005-03-1257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rieger K, Loddenkemper C, Maul J, et al. Mucosal Foxp3+ regulatory T cells are numerically deficient in acute and chronic GVHD. Blood. 2006;107:1717–1723. doi: 10.1182/blood-2005-06-2529. [DOI] [PubMed] [Google Scholar]
- 18.Chen X, Vodanovic-Jankovic S, Johnson B, Keller M, Komorowski R, Drobyski WR. Absence of regulatory T cell control of TH1 and TH17 cells is responsible for the autoimmune-mediated pathology in chronic graft versus host disease. Blood. 2007;110:3804–3813. doi: 10.1182/blood-2007-05-091074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hoffmann P, Ermann J, Edinger M, Fathman CG, Strober S. Donor-type CD4(+)CD25(+) regulatory T cells suppress lethal acute graft-versus-host disease after allogeneic bone marrow transplantation. J Exp Med. 2002;196:389–399. doi: 10.1084/jem.20020399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Edinger M, Hoffmann P, Ermann J, Drago K, Fathman CG, Strober S, Negrin RS. CD4+CD25+ regulatory T cells preserve graft-versus-tumor activity while inhibiting graft-versus-host disease after bone marrow transplantation. Nat Med. 2003;9:1144–1150. doi: 10.1038/nm915. [DOI] [PubMed] [Google Scholar]
- 21.Taylor PA, Panoskaltsis-Mortari A, Swedin JM, et al. L-selectionhi but not the L-selectinlo CD4+ CD25+ T regulatory cells are potent inhibitors of GVHD and BM graft rejection. Blood. 2004;104:3804–3812. doi: 10.1182/blood-2004-05-1850. [DOI] [PubMed] [Google Scholar]
- 22.Nguyen VH, Shashidar S, Chang DS, et al. The impact of regulatory T cells on T cell immunity following hematopoietic cell transplantation. Blood. 2008;111:945–953. doi: 10.1182/blood-2007-07-103895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nguyen VH, Zeiser R, daSilva DL, et al. In vivo dynamics of regulatory T cell trafficking and survival predict effective strategies to control graft versus host disease following allogeneic transplantation. Blood. 2007;109:2649–2656. doi: 10.1182/blood-2006-08-044529. [DOI] [PubMed] [Google Scholar]
- 24.Riley JL, June CH, Blazar BR. Human T regulatory cell therapy: Take a billion or so and call me in the morning. Immunity. 2009;30:656–665. doi: 10.1016/j.immuni.2009.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Selvaraj RK, Geiger TL. A kinetic and dynamic analysis of Foxp3 induced in T cells by TGF-β. J Immunol. 2007;178:7667–7677. doi: 10.4049/jimmunol.178.12.7667. [DOI] [PubMed] [Google Scholar]
- 26.Haribhai D, Lin W, Edwards B, et al. A central role for induced regulatory T cells in tolerance induction in experimental colitis. J Immunol. 2009;182:3461–3468. doi: 10.4049/jimmunol.0802535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Curotto de Lafaille MA, Kutchukhidze N, Shen S, Ding Y, Yee H, Lafaille JJ. Adaptive Foxp3+ regulatory T cell dependent and independent control of allergic inflammation. Immunity. 2008;29:114–126. doi: 10.1016/j.immuni.2008.05.010. [DOI] [PubMed] [Google Scholar]
- 28.Chen X, Das R, Komorowski R, Beres A, Hessner MJ, Mihara M, Drobyski WR. Blockade of interleukin 6 signaling augments regulatory T cell reconstitution and attenuates the severity of graft versus host disease. Blood. 2009;114:891–900. doi: 10.1182/blood-2009-01-197178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Haribhai D, Lin W, Relland L, Truong N, Williams CB, Chatila TA. Regulatory T cells dynamically control the primary immune response to foreign antigen. J Immunol. 2007;178:2961–2972. doi: 10.4049/jimmunol.178.5.2961. [DOI] [PubMed] [Google Scholar]
- 30.Das R, Chen X, Komorowski R, Hessner MJ, Drobyski WR. Interleukin 23 secretion by donor antigen presenting cells is critical for organ-specific pathology in graft versus host disease. Blood. 2009;113:2352–2362. doi: 10.1182/blood-2008-08-175448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yang XO, Nurieva R, Martinez GJ, et al. Molecular antagonism and plasticity of regulatory and inflammatory T cell programs. Immunity. 2008;29:44–56. doi: 10.1016/j.immuni.2008.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhou L, Chong MMW, Littman DR. Plasticity of CD4+ T cell lineage differentiation. Immunity. 2009;30:646–655. doi: 10.1016/j.immuni.2009.05.001. [DOI] [PubMed] [Google Scholar]
- 33.Zhou X, Bailey-Bucktrout SL, Jeker LT, et al. Instability of the transcription factor Foxp3 leads to the generation of pathogenic memory T cells in vivo. Nat Immunol. 2009;10:1000–10007. doi: 10.1038/ni.1774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Schambach F, Schupp M, Lazar MA, Reiner SL. Activation of retinoic acid receptor-α favours regulatory T cell induction at the expense of IL-17-secreting T helper differentiation. Eur J Immunol. 2007;37:2396–2399. doi: 10.1002/eji.200737621. [DOI] [PubMed] [Google Scholar]
- 35.Mucida D, Pino-Lagos K, Kim G, et al. Retinoic acid can directly promote TGF-β-mediated Foxp3+ Treg cell conversion of naïve T cells. Immunity. 2009;30:471–472. doi: 10.1016/j.immuni.2009.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hill GR, Crawford JM, Cooke KR, Brinson YS, Pan L, Ferrara JL. Total body irradiation and acute graft versus host disease: the role of gastrointestinal damage and inflammatory cytokines. Blood. 1997;90:3204–3213. [PubMed] [Google Scholar]
- 37.Abu-Ghosh A, Goldman S, Slone V, et al. Immunological reconstitution and correlation of circulating serum inflammatory mediators/cytokines with the incidence of acute graft versus host disease during the first 100 days following unrelated umbilical cord blood transplantation. Bone Marrow Transplant. 1999;24:535–544. doi: 10.1038/sj.bmt.1701921. [DOI] [PubMed] [Google Scholar]
- 38.Imamura M, Hashino S, Kobayashi H, et al. Serum cytokine levels in bone marrow transplantation: synergistic interaction of interleukin 6, interferon gamma, and tumor necrosis factor alpha in graft versus host disease. Bone Marrow Transplant. 1994;13:745–751. [PubMed] [Google Scholar]
- 39.Trzonkowski P, Bieniaszewska M, Juscinska J, et al. First-in-man clinical results of the treatment of patients with graft versus host disease with human ex vivo expanded CD4+ CD25+ CD127− T regulatory cells. Clin Immunol. 2009;133:22–26. doi: 10.1016/j.clim.2009.06.001. [DOI] [PubMed] [Google Scholar]
- 40.Brunstein CG, Miller JS, Cao Q, et al. Infusion of ex vivo expanded T regulatory cells in adults transplanted with umbilical cord blood: safety profile and detection kinetics. Blood. 2010 doi: 10.1182/blood-2010-07-293795. epub. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zheng SG, Wang J, Horwitz DA. Cutting Edge: Foxp3+ CD4+ CD25+ regulatory T cells induced by IL-2 and TGF-β are resistant to TH17 conversion by IL-6. J Immunol. 2008;180:7112–7116. doi: 10.4049/jimmunol.180.11.7112. [DOI] [PubMed] [Google Scholar]
- 42.Koenecke C, Czeloth N, Bubke A, et al. Alloantigen-specific de novo-induced Foxp3+ Treg revert in vivo and do not protect from experimental GVHD. Eur J Immunol. 2009;39:3091–3096. doi: 10.1002/eji.200939432. [DOI] [PubMed] [Google Scholar]
- 43.Hill JA, Hall JA, Sun CM, et al. Retinoic acid enhances Foxp3 induction indirectly by relieving inhibition from CD4+ CD44hi cells. Immunity. 2008;29:758–770. doi: 10.1016/j.immuni.2008.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Benson MJ, Pino-Lagos K, Rosemblatt M, Noelle RJ. All-trans retinoic acid mediates enhanced T reg cell growth, differentiation, and gut homing in the face of high levels of co-stimulation. J Exp Med. 2007;204:1765–1774. doi: 10.1084/jem.20070719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Dominitzki S, Fantini MC, Neufert C, et al. Cutting Edge: Trans-signaling via the soluble IL-6R abrogates the induction of foxp3 in naïve CD4+ CD25− T cells. J Immunol. 2007;179:2041–2045. doi: 10.4049/jimmunol.179.4.2041. [DOI] [PubMed] [Google Scholar]
- 46.Bettelli E, Carrier Y, Gao W, et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature. 2006;441:235–238. doi: 10.1038/nature04753. [DOI] [PubMed] [Google Scholar]
- 47.Horwitz DA, Zheng SG, Gray JD. Natural and TGF-B-induced Foxp3+ CD4+ CD25+ regulatory T cells are not mirror images of each other. Trends Immunol. 2008;29:429–435. doi: 10.1016/j.it.2008.06.005. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Figure 1: In vitro-differentiated iTregs are detectable in the peripheral lymph nodes early post transplantation. Lethally irradiated Balb/c mice (n=5) were transplanted with B6.PL BM, B6.PL spleen cells (adjusted to yield 0.6 × 106 αβ T cells) and 0.6 × 106 B6 in vitro-differentiated iTregs. Mice were sacrificed 10 days post transplantation, and axillary and inguinal lymph nodes were obtained for pooled analysis. (A). Representative dot plot gated on CD4+ cells depicting the percentage of EGFP+ cells. (B,C). The percentage and absolute number of CD4+ T cells that expressed EGFP in the peripheral draining lymph nodes. Data are shown as the mean ± SEM and represent replicate mice from one experiment.
Supplementary Figure 2: In vitro-differentiated iTregs do not induce lethal GVHD in the absence of conventional T cells. Lethally irradiated Balb/c mice were transplanted with B6.PL BM and 0.6 × 106 B6 in vitro-differentiated iTregs. (A). Representative dot plot showing the percentage of H-2Kb Thy 1.2+ and Thy1.2+ EGFP+ cells present in the spleen, liver, and lung from animals sacrificed 10 days post transplantation. (B). Overall survival from mice transplanted with B6.PL BM cells alone (□, n=5) or together with 0.6 × 106 B6 in vitro-differentiated iTregs (●, n=5).