Abstract
Many acute and chronic effects of ionizing radiation are mediated by reactive oxygen species and reactive nitrogen species, which deplete antioxidant stores, leading to cellular apoptosis, stem cell depletion and accelerated aging. C57BL/6NHsd mice receiving intravenous MnSOD-PL prior to 9.5 Gy total-body irradiation (TBI) show increased survival from the acute hematopoietic syndrome, and males demonstrated improved long-term survival (Epperly et al., Radiat. Res. 170, 437–444, 2008). We evaluated the effect of an antioxidant-chemopreventive diet compared to a regular diet on long-term survival in female mice. Twenty-four hours before the LD50/30 dose of 9.5 Gy TBI, subgroups of mice were injected intravenously with MnSOD-PL (100 μg plasmid DNA in 100 μl of liposomes). Mice on either diet treated with MnSOD-PL showed decreased death after irradiation compared to irradiated mice on the house diet alone (P = 0.031 for the house diet plus MnSOD-PL or 0.015 for antioxidant diet plus MnSOD-PL). The mice on the antioxidant-chemoprevention diet alone or with MnSOD-PL that survived 30 days after irradiation had a significant increase in survival compared to mice on the regular diet (P = 0.04 or 0.01, respectively). In addition, mice treated with MnSOD-PL only and surviving 30 days after radiation also had increased survival compared to those on the regular diet alone (P = 0.02). Survivors of acute ionizing radiation damage have ameliorated life shortening if they are fed an antioxidant-chemopreventive diet.
INTRODUCTION
Ionizing radiation induces nuclear DNA strand breaks that initiate a transfer to the mitochondria of both pro-apoptotic and anti-apoptotic molecules (1). The molecular events that occur early in the initiation of apoptosis originate at the mitochondrial membrane and include molecular sequelae of both oxidative and nitrosative stress, producing rapid depletion of antioxidant stores (2). Depletion of antioxidants at the mitochondria is associated with disruption of cytochrome C binding to cardiolipin, mitochondrial membrane disruption, and leakage into the cytoplasm of cytochrome C, which then initiates a cascade of molecular events leading to apoptosis (3). Administration of antioxidant agents including manganese superoxide dismutase plasmid liposome gene product and small molecule antioxidants (4, 5) has been shown to decrease radiation-induced cellular apoptosis, tissue injury and improved survival in organ-specific- and total-body-irradiated rodents (6–12). These experimental models have demonstrated the importance of antioxidant therapy in ameliorating the acute effects of radiation.
Recent evidence has supported a prominent role for antioxidants in ameliorating the chronic effects of radiation exposure. Organ-specific late radiation injury such as pulmonary fibrosis, renal failure, hepatic fibrosis and central nervous system damage resulting in neuro-cognitive impairment has been shown to be ameliorated by antioxidant therapy. The acute effects of total-body irradiation are also ameliorated by antioxidant therapies. Administration of MnSOD-PL prior to total-body irradiation not only improved survival from the LD50/30 dose of 9.5 Gy in C57BL/6HNsd mice but also ameliorated the late radiation-induced life shortening in male mice (12). Antioxidant dietary supplements have been shown to improve survival in acutely irradiated mice (13–16).
In the present study, we tested the hypothesis that a diet containing antioxidant dietary supplements as a source of continual antioxidant bioavailability results in improved acute as well as long-term survival after LD50/30 total-body irradiation. To test this hypothesis, we used a dietary supplement consisting of antioxidants and chemopreventive agents maintained continually in the diets starting 7 days prior to irradiation and continuing for over 450 days postirradiation. A subgroup of these mice also received MnSOD-PL before irradiation to test whether the antioxidant diet would complement the radioprotective effect of MnSOD-PL. The results show significant amelioration effects of the antioxidant diet in both MnSOD-PL-treated and irradiated control mice.
MATERIALS AND METHODS
Mice and Animal Care
All experimental protocols were approved by the University of Pittsburgh Institutional Animal Care and Use Committee. Veterinary care was provided by the University of Pittsburgh Division of Laboratory Animal Research. C57BL/6HNsd female mice (18 to 20 g) were housed five per cage and maintained according to protocols of the University of Pittsburgh Division of Laboratory Animal Research.
Experimental Protocol
In this study we used 160 female C57BL/6NHsd female mice (8 weeks of age) that were divided into four groups of 40 mice. Two of the groups were placed on the antioxidant-chemopreventive diet (Table 1) 7 days before irradiation and maintained on the diet until conclusion of the experiment. The other two groups were maintained on the regular or “house” diet [LabDiet rMH 3000 (5P00) with 0.12% hydrogen silicon dioxide from TestDiet, catalog no. 1812877]. The silicon dioxide is added as an inert compound to compensate for weight changes due to addition of antioxidant ingredients. The antioxidant diet consisted of a micronutrient multivitamin and trace mineral formula (AmeriSciences®/NASA Premium Multivamin Premix, AmeriSciences LP, Houston, TX) and a non-essential antioxidant and chemoprevention mixture derived primarily from natural foods (AmeriSciences®/NASA Fruit and Veggie Antioxidant Formula Premix, AmeriSciences LP, Houston TX). Of this chow serving size, 99.95% was chow mix, 0.024% was the AS/NASA Premium Multivitamin Formula (Table 1), and 0.023% was the AS/NASA Fruit/Veggie Antioxidant Formula (Table 1). The constituents of the antioxidant and chemoprevention diet supplements are shown in Table 1.
TABLE 1.
Antioxidant Diet Composition
| Daily dose per mousea | Equivalent human daily dose | Human ULb (19–70 years old) | Human NOAELc | |
|---|---|---|---|---|
| Micronutrient components | ||||
| Vitamin A (30% as vitamin A palmitate and 70% as beta-carotene) | 0.2451 IU | 750 IU | 10,000 IU | 10,000 IU |
| Beta-carotene (part of vitamin A total) | 0.3431 μg | 1.05 mg | NEa | 25 mg |
| Vitamin C (as ascorbic acid) | 0.0817 mg | 250 mg | 2000 mg | >1000 mg |
| Vitamin D (as cholecalciferol) | 0.3921 IU | 1200 IU | 4000 IU | 800 IU |
| Vitamin E (as d-alpha tocopheryl succinate and mixed tocopherols) | 0.0653 IU | 200 IU | 1490 IU | 1200 IU |
| Vitamin K (as phytonadione) | 0.0261 μg | 80 μg | NE | 30 μg |
| Thiamine (vitamin B1) (as thiamine mononitrate) | 0.7352 μg | 2.25 mg | NE | 50 mg |
| Riboflavin (vitamin B2) | 0.8332 μg | 2.55 mg | NE | 200 mg |
| Niacin (as inositol hexanicotinate) | 9.802 μg | 30 mg | 35 mg | 500 mg |
| Vitamin B6 (as pyridoxine hydrochloride) | 0.9802 μg | 3 mg | 100 mg | 200 mg |
| Folate (as folic acid) | 0.1960 μg | 600 μg | 1000 μg | 1000 μg |
| Vitamin B12 (as cyanocobalamin) | 0.0029 μg | 9 μg | NE | 3000 μg |
| Biotin | 0.1470 μg | 450 μg | NE | 2500 μg |
| Pantothenic acid (as d-calcium pantothenate) | 4.901 μg | 15 mg | NE | 1000 mg |
| Calcium (as calcium carbonate, dicalcium phosphate) | 0.1634 mg | 500 mg | 2500 mg | 1500 mg |
| Iodine (from kelp) | 0.0098 μg | 30 μg | 1100 μg | 1000 μg |
| Magnesium (as magnesiumoxide and chelate) | 65.35 μg | 200 mg | 350 mg | 700 mg |
| Zinc (as zinc chelate [monomethionine]) | 4.901 μg | 15 mg | 40 mg | 30 mg |
| Selenium (as L-selenomethionine) | 0.0327 μg | 100 μg | 400 μg | 200 μg |
| Copper (as copper amino acid chelate) | 0.0588 μg | 0.18 mg | 10 mg | 9 mg |
| Manganese (as manganese amino acid chelate) | 0.6535 μg | 2 mg | 11 mg | 10 mg |
| Chromium (as chromium polynicotinate) | 0.0653 μg | 200 μg | NE | 1000 μg |
| Molybdenum (as molybdenum amino acid chelate) | 0.0183 μg | 56 μg | 2000 μg | 350 μg |
| Potassium (as potassium citrate) | 94.75 μg | 290 mg | NE | NE |
| Choline (as choline bitartrate) | 16.34 μg | 50 mg | 3500 mg | NE |
| Inositol (as inositol and inositol hexanicotinate) | 16.34 μg | 50 mg | NE | NE |
| Boron (as boron chelate) | 0.3267 μg | 1 mg | 20 mg | NE |
| Vanadium (as vanadyl sulfate) | 0.0163 μg | 50 μg | 1800 μg | NE |
| Non-essential natural antioxidant and chemoprevention agents: | ||||
| Rutin | 8.036 μg | 25 mg | ||
| Quercetin | 257.1 μg | 800 mg | ||
| Hesperidin | 1.607 μg | 5 mg | ||
| Alpha lipoic acid | 128.6 μg | 400 mg | ||
| N-Acetyl-L-cysteine (NAC) | 192.9 μg | 00 mg | ||
| Lutein | 3.214 μg | 10 mg | ||
| Lycopene | 1.607 μg | 5 mg | ||
| Astaxanthin | 0.3214 μg | 1 mg | ||
| Plant sterols | 80.36 μg | 250 mg | ||
| Isoflavones (from soy extract) | 8.036 μg | 25 mg | ||
| Garlic extract (bulb) | 88.39 μg | 275 mg | ||
| Green tea extract (leaf) [standardized to 95% polyphenols and 50% epigallocatechin gallate (EGCG)] | 80.36 μg | 250 mg | ||
| Cruciferous vegetable extract (Brassica spp.) (plant) | 32.14 μg | 100 mg | ||
| Fruit blend (strawberry, escobillo, blueberry, blackberry, cranberry, grape, pomegranate) | 32.14 μg | 100 mg | ||
| Ginkgo biloba extract (leaf) | 19.29 μg | 60 mg | ||
| Coenzyme Q-10 | 32.14 μg | 100 mg | ||
| Resveratrol | 1.607 μg | 5 mg | ||
Each mouse weighed an average of 22.5 g.
Dietary intake’s tolerable upper intake levels. The maximum level of daily nutrient intake that is likely to pose no risk of adverse effects. Food and Nutrition Board, Institute of Medicine, National Academies of Science.
“No Observed Adverse Event Level” is a level that should be considered safe and requires no application of a safety factor to determine a safe intake, based on the most sensitive subgroup.
None established.
The material and specifications of micronutrients such as the vitamins (e.g. ascorbic acid, cholecalciferol, etc.), minerals (e.g. potassium, zinc, etc.), and other coenzyme and non-botanical constituents of the formulae (e.g. coenzyme Q-10, choline bitartrate, N-acetyl cysteine, etc.) have been documented extensively in pharmacopeial compendia such as the USP (17). Certain ingredients of a botanical nature that may not be subject to a pharmacopeial monograph (e.g. quercetin, astaxanthin, fruit extracts, etc.) have nevertheless been extracted, isolated and/or produced to standardized protocols in accordance with the supplier’s proprietary specifications and current Good Manufacturing Practices (cGMP). Although proprietary and commercially available though the referenced supplier, the following is a brief description of these non-compendial ingredients.
Quercetin, rutin and hesperedin are flavonols characterized by a phenyl benzo(γ)pyrone-derived structure (18, 19). Commercially, quercetin is produced by extraction of the quercetin glycosides, primarily rutin, from plants. Rutin is subsequently subjected to hydrolysis to yield the aglycone. A variety of botanical species may be used for the isolation and synthesis of quercetin, such as citrus peel, apples, onions and/or Uncaria leaves, but immature sun-dried Fava d’Anta beans (Dimorphandra mollis or Dimorphandra gardeneriana) are preferred and principally used as the starting material for the flavonols in the antioxidant diet. The manufacturing process for quercetin essentially consists of the aqueous extraction of rutin from the above-identified plant source(s) and release of the aglycone via hydrolysis by the addition of an acidic aqueous solution common to food ingredient manufacturing processes and subsequent neutralization to produce a crude crystalline quercetin product. Next, the resultant quercetin product is subjected to several purification processes to yield purified quercetin crystals. All starting, intermediate and finished materials involved in the manufacturing process are appropriate for food use and are listed as GRAS (Generally Recognized as Safe) by the U.S. Food and Drug Administration. Green tea extract is standardized from the leaves of Camellia sinensis, which are gently washed, dried, shivered, compacted and kept at controlled room temperature under low humidity conditions prior to processing. Extraction takes place in a reactor using purified water as the solvent at 90°C. This intermediate is then concentrated under high pressure at lower temperatures prior to being filtered and crystallized with additional solvents that are appropriate for use in food processing. Finally, the crystallized extract is dried and powdered to specification.
The antioxidant diet contains a proprietary blend of fruit concentrates and extracts known for their antioxidant values, as described in the literature, including the U.S. Department of Agriculture’s Database for the Oxygen Radical Absorbance Capacity (ORAC) (20). The whole fruits of F. ananassa (strawberry), E. vaccinium (blueberry), R. rubus (blackberry) and E. vaccinium (cranberry) are washed and treated solely with water, then dried and blended into powdered fruit concentrates. Meanwhile, M. glabra (Escobillo), V. vinifera (grape) and P. granatum (pomegranate) are subjected to extractions by way of percolation using water, ethanol or both as solvents. The mixture of these extracts is homogenized in a two-stage process with heated transfer lines before proceeding to a spray dry tower, where they are powdered. All material undergoes metal detection scanning before being blended and combined.
Brassica oleracea italia seed is known for its perceived health benefits as well as its high antioxidant values, primarily attributed to its content of sulforaphane (20). Collection of such seeds is used as the precursor for growing broccoli sprouts. Sprouts are cultivated in pesticide-free conditions. Florets of young broccoli are harvested at conditions of maximal glucosinolate content. Proprietary processing technology by the supplier ensures that sulforaphane is not digested by endogenous myrosinase enzymes. No solvents are used in processing. Approximately 20 lb. of broccoli sprouts yield 1 lb. of end material (20:1 concentration).
Resveratrol (3,4′,5-trihydroxystilbene) belongs to a class of polyphenolic compounds called stilbenes (21). Some types of plants produce resveratrol and other stilbenes in response to stress, injury, fungal infection or ultraviolet (UV) irradiation (22). Resveratrol-3-O-beta-glucoside is called piceid (23). Scientists became interested in exploring potential health benefits of resveratrol in 1992 when its presence was first reported in red wine, leading to speculation that resveratrol might help explain the “French Paradox” (24). More recently, reports on the potential for resveratrol to inhibit the development of cancer and extend life span in cell culture and animal models have continued to generate scientific interest (25, 26). Vitis vinifera, Carignane and Cinsault varieties are whole red grapes from the Rhone Valley in Southern France. Grape seeds and skins are collected after wine fermentation from wine vessels, then purified and concentrated for extraction. A multistep process involving water extraction and purification of polyphenols on adsorbent resin ensures high purity and reproducibility. As with other extracts, standardization, quality assurance testing and metal detection scanning are employed prior to blending and release. Approximately 500–750 lb. of red grapes yield 1 lb. of the standardized extract.
Isoflavones are polyphenolic compounds commonly found in legumes such as soybeans. The most common and abundant soy isoflavone aglycone is genistein, followed by daidzein and glycitein (27). The soy isoflavone isolate starts off with non-GMO soybeans that undergo extraction with water and ethanol, filtration, elution with a resin, concentration and a second round of filtration. The extract is then dried, pulverized, assayed and finally diluted and blended to achieve standardization specification.
Astaxanthin is a carotenoid with known antioxidant properties, with documented effects on immunology, muscular endurance, visual acuity, reduced rate of macular degeneration, and reactive oxygen species (ROS) (28). The starting material from which astaxanthin is extracted is the algae Haematococcus pluvialis, cultivated in Hawaii. After harvesting, the microalgae are washed, dried and pulverized. The dried biomass intermediate is then extracted by means of effused supercritical CO2. The resulting oleoresin extract intermediate is subsequently mixed with stabilizing ingredients that are generally recognized as safe by the Food and Drug Administration and spray-dried. Finally, the end product is milled or chilsonated in accordance with the specified mesh size.
All of the ingredients are mixed in a V-type blender in accordance with proprietary specifications of order, time and speed. The blend is then subjected to verification of homogeneity, assay, particle size, microbial specifications, density, humidity and other applicable measures of quality as determined by the supplier.
The chow portion per mouse per day was 5000 mg. This diet then yielded the following serving sizes: 1.22 mg per day of AS/NASA Premium Multivitamin Formula and 1.13 mg per day of AS/NASA Fruit/Veggie Antioxidant Formula. There were no additional ingredients other than those listed. Additives were combined with the control house diet by Purina Corporation in the concentrations shown above, then formed in pellets of size and shape identical to the non-additive control house diet. One of the two antioxidant diet groups (40 mice) and one of the two house diet groups (40 mice) was injected intravenously with MnSOD-PL (100 μg plasma in 100 μl of liposomes) 24 h before irradiation according to published methods (12). All mice were exposed to 9.5 Gy total-body radiation using a J. L. Shepherd Mark I cesium irradiator at 70 cGy/min.
Statistical Evaluation
All mice were evaluated for survival, overall survival and conditional survival as described previously (12). Overall survival is defined as the time from the date of irradiation to the date of death for all mice under study. Conditional survival is defined as the time from the date of irradiation to the date of death for all mice who survived 31 days or longer after irradiation. Mouse 30-day mortality was compared between any two groups with the two-sided Fisher’s exact test. For mice surviving 31 days or longer, survival was compared between any two groups with the two-sided log-rank test. These analyses were performed in SAS software. P values less than 0.050 were regarded as significant.
RESULTS
MnSOD-PL Administration Improves Survival after LD50/30 Total-Body Irradiation
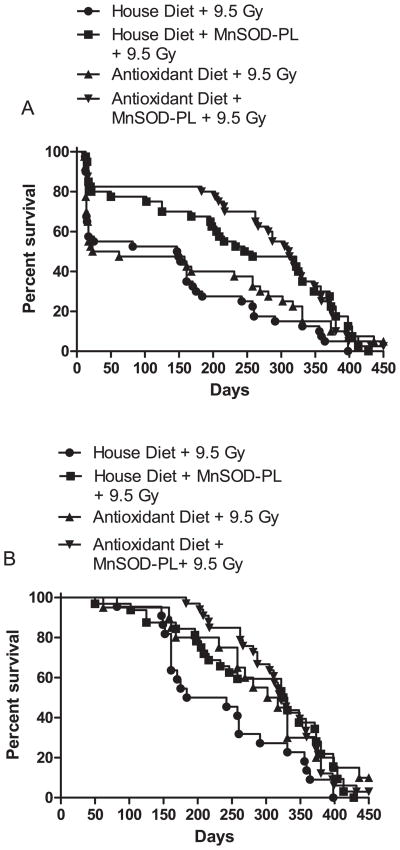
Mice that received intravenous administration of 100 μg of plasmid DNA in 100 μl of liposomes showed improved survival compared to mice in the control group after 9.5 Gy TBI. MnSOD-PL administration resulted in increased survival from the acute effects of 9.5 Gy TBI (Fig. 1A, P = 0.031). With respect to mice on the house diet, these data confirm and extend those of a previous publication (12). Mice receiving the antioxidant diet did not show an improvement in survival at 30 days, with a mortality of 50% compared to 45% for the control diet (P = 0.82, Table 2, Fig. 1A). The data confirmed and extended our previous publication (12) and demonstrated decreased 30-day mortality in the MnSOD-PL group compared to the control: 20% mortality in the MnSOD-PL group compared 45% in the control (P = 0.031, Table 2). Thirty-day mortality was significantly lower in the antioxidant diet + MnSOD-PL group compared to the control house diet or antioxidant diet only: 17.5% for the antioxidant diet + MnSOD-PL group compared to 45% mortality in irradiated house diet controls and 50% in the antioxidant diet group (P = 0.015 and 0.004, respectively, Table 2). These results establish that the antioxidant diet did not negatively influence the radioprotective effect of MnSOD-PL against total-body irradiation.
FIG. 1.
Groups of 40 C57BL/6HNsd female mice were untreated or were treated with MnSOD-PL (100 μl/100 μg plasmid) 24 h prior to irradiation. All designated for the antioxidant diet were fed the antioxidant-chemopreventive diet starting 7 days prior to total-body irradiation. Mice received 9.5 Gy and then were followed for survival and maintained on each representative diet. Panel A shows overall survival. Panel B shows conditional survival.
TABLE 2.
Mortality after 9.5 Gy Total-Body Irradiation of Mice in Relation to Antioxidant-Chemopreventive Diet and MnSOD-PL Treatment Compared to House Diet (Control)
| Group | n | 30-day mortality
|
Survival > 30 daysa |
||
|---|---|---|---|---|---|
| % | Pb | Median (95% CI) | Pc | ||
| Control | 40 | 45 | 213 (161–291) | ||
| MnSOD-PL | 40 | 20 | 0.031 (compared to group 1) | 328 (216–373) | 0.020 (compared to group 1) |
| Antioxidant diet | 40 | 50 | 0.82 (compared to group 1) | 309.5 (231–373) | 0.040 (compared to group 1) |
| Antioxidant diet + MnSOD-PL | 0.015 (compared to group 1) | 0.010 (compared to group 1) | |||
| 1.00 (compared to group 2) | 0.95 (compared to group 2) | ||||
| 40 | 17.5 | 0.0041 (compared to group 3) | 322 (287–358) | 0.87 (compared to group 3) | |
Analysis for animals surviving more than 30 days.
Fisher’s exact test.
Log-rank test.
Antioxidant Diet Improves Conditional Survival and Ameliorates Radiation-Induced Life Shortening
Mice surviving at 30 days after 9.5 Gy total-body irradiation were followed for evaluation of the late effects of radiation (conditional survival). As shown in Fig. 1B and Table 2, the conditional survival of mice on the antioxidant diet was significantly improved over the 450 days of observation compared to that of those on the house diet (P = 0.040, Table 2). Mice on the house diet that also received MnSOD-PL showed an improvement in conditional survival compared to irradiated house diet controls (P = 0.020, Fig. 1B). Mice on the antioxidant diet that received MnSOD-PL in addition also showed an improvement in conditional survival compared to those on the house diet alone (P = 0.010, Fig. 1B). There was no significant difference in conditional survival between the antioxidant diet or the mice treated with MnSOD-PL and the mice receiving MnSOD-PL alone or on the antioxidant diet alone. Among the irradiated mice surviving 31 days or longer, the conditional median survival time was 213 days for the house diet controls and was longer in the MnSOD-PL house diet group (328 days), antioxidant diet group (309.5 days), and antioxidant diet plus MnSOD-PL group (322 days, Table 2). These results establish that antioxidant diet supplements ameliorate radiation-induced life shortening and provide support for the concept of continuing oxidative stress in the postirradiation cellular microenvironment of tissues, organs and organ systems.
DISCUSSION
There is accumulating evidence for a role of oxidative stress in both the acute and chronic effects of ionizing radiation (3, 29). Administration of organ-specific targeted antioxidant therapies including MnSOD-PL has shown improvement in increased survival due to a decrease in acute and chronic toxicities of single-fraction and fractionated irradiation (4–7), as has systemic administration of some antioxidant agents, including Amifostine, GS-nitroxide and SOD mimic small molecules (29). With respect to late effects of ionizing radiation, two categories of studies have been published. Prior studies have reported improved conditional survival of MnSOD-PL-treated high-dose-irradiated animals after passage of the time for acute radiation events (12). Other studies have described improved conditional survival effects of antioxidants in low-dose-or partial-body-irradiated animals (13–16). In both categories of studies, antioxidant therapy ameliorated late radiation effects.
It was postulated that a formula mixing lower levels of each of the most effective chemoprotection molecules would allow for a broad range of cellular protection and bioavailability without the toxicity associated with high-dose single agents. The supplement formulas consist of (1) a multivitamin and trace mineral formula with commonly accepted doses as used in industry with most components at levels of the U.S. Recommended Daily Allowance, but some with antioxidant capacity at slightly higher but safe levels (well below levels of any adverse effect) (Table 1, top panel), and (2) a chemoprevention mixture, derived mostly from natural food and herbal sources (Table 1, bottom panel), which have demonstrated antioxidant effects, as demonstrated by previous studies published in scientific peer-reviewed journals at doses found to be safe by the NIH consensus conference on dietary supplements, or recommended by the National Cancer Institute/Chemoprevention Branch for possible reductions in cancer development risk, based on epidemiological reviews or by testing in randomized, placebo-controlled studies (17–28).
In our studies of conditional survival, mice receiving MnSOD-PL prior to irradiation and demonstrating improved survival from the LD50/30 dose also showed amelioration of radiation-induced late effects (12). These results were attributed to a decrease in radiation-induced aging in a non-specific sense, rather than a decrease in the frequency or type of radiation-induced tumors or evidence of neurodegenerative disease (12). Since radiation-induced life shortening is associated with biomarkers of aging including fur-graying in rodent models, organ failure, osteoporosis and fibrosis, many animals in these previous studies did not show a specific cause of death (12). Previous antioxidant dietary supplement programs have been tested with respect to low-dose total-body irradiation in rodents, dogs, rabbits and other small animal models (13–16, 29). Specific antioxidants have been tested for amelioration of late radiation effects. These include studies of Bowman-Birk inhibitor (30), administration of specific lipid-enriched diets, and continuous administration of small molecule antioxidants (13–16). There has been uniform agreement that antioxidant treatments did not adversely affect long-term survival, and there has been a suggestion of improved survival by amelioration of late effects. Rodent models of late radiation effects often increase in tumorigenesis, specifically thymic lymphoma, which has been shown in several mouse strains. Antioxidant MnSOD-PL treatment did not increase tumor frequency or lethality in our prior studies (12).
In the present studies, an antioxidant-chemopreventive diet was shown to improve conditional survival in total-body-irradiated female mice. A significant therapeutic effect of the diet above was seen in conditional survival but not in immediate 30-day survival after the LD50/30 dose, indicating that 9.5 Gy, or the LD50/30 dose associated with acute cellular, tissue and organ-damaging effects, could not be ameliorated by dietary supplements. However, in animals surviving the acute effects of radiation, the antioxidant-chemopreventive diet ameliorated radiation-induced life shortening (12).
We did not measure oxidative stress levels or depletion of antioxidant stores in mice. Other in vitro studies have shown that supplementation with antioxidants decreases radiation-induced depletion of glutathione and other cellular antioxidants and prevents nitrosative stress that can neutralize the biological effectiveness of endogenous MnSOD (31, 32). We did not measure levels of uptake of specific micronutrients in the antioxidant diet in the present studies. These studies have been carried out previously and have shown that 7–10 days administration of an antioxidant diet is associated with a significant elevation in levels of micronutrients (33, 34); thus we started the diet 7 days before 9.5 Gy irradiation and kept mice on that diet for the entire 450 days of the experiment. Mice on the house diet compared to antioxidant diet did not show any differences in body weight over the 450 days, indicating that the antioxidant-chemopreventive diet was not less palatable to the mice.
We also did not include a negative control such as a plasmid containing a scrambled sequence or a gene such as LacZ. In previous experiments it has been demonstrated that the use of plasmid complexes containing these negative controls or using a cytoplasmic CuSOD transgene did not significantly increase survival (1, 35, 36). Due to the number of mice used in this experiment and length of the experiment, the cost of including these negative controls was prohibitive.
The present studies support continued research and development of an antioxidant dietary supplement for survivors of radiological terrorism and nuclear accidents and potentially for use in astronauts exposed to galactic cosmic radiation (33, 34, 37). In the latter group, late effects of radiation including cataract formation, carcinogenesis, neurological degeneration and other biomarkers of radiation-induced aging have been implicated as issues of potential concern (37).
Acknowledgments
This research was supported by grant U19A1068021 from the NIAID/NIH.
References
- 1.Epperly MW, Gretton JE, Sikora CA, Jefferson M, Bernarding M, Nie S, Greenberger JS. Mitochondrial localization of superoxide dismutase is required for decreasing radiation-induced cellular damage. Radiat Res. 2003;160:568–578. doi: 10.1667/rr3081. [DOI] [PubMed] [Google Scholar]
- 2.Guo HL, Seixas-Silva JA, Epperly MW, Gretton JE, Shin DM, Greenberger JS. Prevention of radiation-induced oral cavity mucositis by plasmid/liposome delivery of the human manganese superoxide dismutase (MnSOD) transgene. Radiat Res. 2003;159:361–370. doi: 10.1667/0033-7587(2003)159[0361:porioc]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 3.Kagan VE, Bayir HA, Belikova NA, Kapralov O, Tyurina YY, Tyurin VA, Jiang J, Stoyanovsky DA, Wipf P, Borisenko G. Cytochrome c/cardiolipin relations in mitochondria: a kiss of death. Free Radic Biol Med. 2009;46:1439–1453. doi: 10.1016/j.freeradbiomed.2009.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jiang J, Belikova NA, Xiao J, Zhao Q, Greenberger JS, Wipf P, Kagan VE. A mitochondria-targeted nitroxide/hemi-gramicidin S conjugate protects mouse embryonic cells against γ-irradiation. Int J Radiat Oncol Biol Phys. 2008;70:816–825. doi: 10.1016/j.ijrobp.2007.10.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rajagopalan MS, Gupta K, Epperly MW, Franicola D, Zhang X, Wang H, Zhao H, Tyurin VA, Kagan VE, Greenberger JS. The mitochondria-targeted nitroxide JP4-039 augments potentially lethal irradiation damage repair. In Vivo. 2009;23:717–726. [PMC free article] [PubMed] [Google Scholar]
- 6.Epperly MW, Bray JA, Kraeger S, Zwacka R, Engelhardt J, Travis E, Greenberger JS. Prevention of late effects of irradiation lung damage by manganese superoxide dismutase gene therapy. Gene Therapy. 1998;5:196–208. doi: 10.1038/sj.gt.3300580. [DOI] [PubMed] [Google Scholar]
- 7.Stickle RL, Epperly MW, Klein E, Bray JA, Greenberger JS. Prevention of irradiation-induced esophagitis by plasmid/liposome delivery of the human manganese superoxide dismutase (MnSOD) transgene. Radiat Oncol Invest Clin Basic Res. 1999;7:204–217. doi: 10.1002/(SICI)1520-6823(1999)7:4<204::AID-ROI2>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- 8.Epperly MW, Gretton JA, DeFilippi SJ, Sikora CA, Liggitt D, Koe G, Greenberger JS. Modulation of radiation-induced cytokine elevation associated with esophagitis and esophageal stricture by manganese superoxide dismutase-plasmid liposome (SOD-PL) gene therapy. Radiat Res. 2001;155:2–14. doi: 10.1667/0033-7587(2001)155[0002:morice]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 9.Guo HL, Seixas-Silva JA, Epperly MW, Gretton JE, Shin DM, Greenberger JS. Prevention of radiation-induced oral cavity mucositis by plasmid/liposome delivery of the human manganese superoxide dismutase (MnSOD) transgene. Radiat Res. 2003;159:361–370. doi: 10.1667/0033-7587(2003)159[0361:porioc]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 10.Guo HL, Wolfe D, Epperly MW, Huang S, Liu K, Glorioso JC, Greenberger J, Blumberg D. Gene transfer of human manganese superoxide dismutase protects small intestinal villi from radiation injury. J Gastrointest Surg. 2003;7:229–236. doi: 10.1016/s1091-255x(02)00186-5. [DOI] [PubMed] [Google Scholar]
- 11.Greenberger JS, Epperly MW. Radioprotective antioxidant gene therapy: potential mechanisms of action. Gene Ther Mol Biol (GTMB) 2004;8:31–44. [Google Scholar]
- 12.Epperly MW, Smith T, Wang H, Schlesselman J, Franicola D, Greenberger JS. Modulation of radiation-induced life shortening by systemic intravenous MnSOD-plasmid liposome gene therapy. Radiat Res. 2008;170:437–444. doi: 10.1667/rr1286.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kennedy AR, Zhou Z, Donahue JJ, Ware JH. Protection against adverse biological effects induced by space radiation by the Bowman-Birk inhibitor and antioxidants. Radiat Res. 2006;166:327–332. doi: 10.1667/RR3599.1. [DOI] [PubMed] [Google Scholar]
- 14.Guan J, Stewart J, Ware JH, Zhou Z, Donahue JJ, Kennedy AR. Effects of dietary supplements on the space radiation-induced reduction in total antioxidant status in CBA mice. Radiat Res. 2006;165:373–378. doi: 10.1667/rr3523.1. [DOI] [PubMed] [Google Scholar]
- 15.Lee JC, Kinniry PA, Arguiri E, Serota M, Kanterakis S, Chatterjee S, Solomides CC, Javvadi P, Koumenis C, Christofidou-Solomidou M. Dietary curcumin increases antioxidant defenses in lung, ameliorates radiation-induced pulmonary fibrosis, and improves survival in mice. Radiat Res. 2010;173:590–601. doi: 10.1667/RR1522.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Davis JG, Wan XS, Ware JH, Kennedy AR. Dietary supplements reduce the cataractogenic potential of proton and HZE-particle radiation in mice. Radiat Res. 2010;173:353–361. doi: 10.1667/RR1398.1. [DOI] [PubMed] [Google Scholar]
- 17.United States Pharmacopeia and National Formulary (USP 32-NF 27) United States Pharmacopeia Convention; Rockville, MD: 2009. [Google Scholar]
- 18.Kuhnau J. The flavonoids: A class of semi-essential food components: Their role in human nutrition. World Rev Nutr Diet. 1976;24:117–191. [PubMed] [Google Scholar]
- 19.Morand C, Crespy V, Manach C, Besson C, Demigne C, Remesy C. Plasma metabolites of quercetin and their antioxidant properties. Am J Physiol Regul Integr Comp Physiol. 1998;275:R212–R219. doi: 10.1152/ajpregu.1998.275.1.R212. [DOI] [PubMed] [Google Scholar]
- 20.USDA Database for the Oxygen Radical Absorbance Capacity (ORAC) of Selected Foods, Release 2. U.S. Department of Agriculture, Agricultural Research Service, Beltsville Human Nutrition Research Center, Nutrient Data Laboratory; Beltsville, MD: May, 2010. [Google Scholar]
- 21.Soleas GJ, Diamandis EP, Goldberg DM. Resveratrol: a molecule whose time has come? And gone? Clin Biochem. 1997;30:91–113. doi: 10.1016/s0009-9120(96)00155-5. [DOI] [PubMed] [Google Scholar]
- 22.Aggarwal BB, Bhardwaj A, Aggarwal RS, Seeram NP, Shishodia S, Takada Y. Role of resveratrol in prevention and therapy of cancer: preclinical and clinical studies. Anticancer Res. 2004;24:2783–2840. [PubMed] [Google Scholar]
- 23.Romero-Perez AI, Ibern-Gomez M, Lamuela-Raventos RM, de La Torre-Boronat MC. Piceid, the major resveratrol derivative in grape juices. J Agric Food Chem. 1999;47:1533–1536. doi: 10.1021/jf981024g. [DOI] [PubMed] [Google Scholar]
- 24.Siemann EH, Creasey LL. Concentration of the phytoalexin resveratrol in wine. Am J Enol Vitic. 1992;43:49–52. [Google Scholar]
- 25.Juhasz B, Varga B, Gesztelyi R, Kemeny-Beke A, Zsuga J, Tosaki A. Resveratrol: a multifunctional cytoprotective molecule. Curr Pharm Biotechnol. 2010;11:810–8. doi: 10.2174/138920110793262079. [DOI] [PubMed] [Google Scholar]
- 26.Mukherjee S, Dudley JI, Das DK. Dose-dependency of resveratrol in providing health benefits. Dose Response. 2010;8:478–500. doi: 10.2203/dose-response.09-015.Mukherjee. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lampe JW. Isoflavonoid and lignan phytoestrogens as dietary biomarkers. J Nutr. 2003;133(Suppl 3):956S–964S. doi: 10.1093/jn/133.3.956S. [DOI] [PubMed] [Google Scholar]
- 28.Park JS, Chyun JH, Kim YK, Line LL, Chew BP. Astaxanthin decreased oxidative stress and inflammation and enhanced immune response in humans. Nutr Metab (Lond) 2010;7:18. doi: 10.1186/1743-7075-7-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Greenberger JS. Radioprotection. In Vivo. 2009;23:323–336. [PMC free article] [PubMed] [Google Scholar]
- 30.Wambi CO, Sanzari JK, Sayers CM, Nuth M, Zhou Z, Davis J, Finnberg N, Lewis-Wambi JS, Ware JH, Kennedy AR. Protective effects of dietary antioxidants on proton total-body irradiation-mediated hematopoietic cell and animal survival. Radiat Res. 2009;172:175–186. doi: 10.1667/RR1708.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bayir H, Kagan VE, Tyurina YY, Tyurin V, Ruppel RA, Adelson PD, Graham SH, Janesko K, Clark RSB, Kochanek PM. Assessment of antioxidant reserves and oxidative stress in cerebrospinal fluid after severe traumatic brain injury in infants and children. Pediatr Res. 2002;51:571–578. doi: 10.1203/00006450-200205000-00005. [DOI] [PubMed] [Google Scholar]
- 32.Epperly MW, Osipov AN, Martin I, Kawai K, Borisenko GG, Jefferson M, Bernarding M, Greenberger JS, Kagan VE. Ascorbate as a “redox-sensor” and protector against irradiation-induced oxidative stress in 32D cl 3 hematopoietic cells and subclones overexpressing human manganese superoxide dismutase. Int J Radiat Oncol Biol Phys. 2004;58:851–861. doi: 10.1016/j.ijrobp.2003.10.022. [DOI] [PubMed] [Google Scholar]
- 33.Matuszczak Y, Farid M, Jones J, Landsdowne S, Taylor A, Reid M. N-Acetylcysteine inhibits muscle fatigue and glutathione oxidation during handgrip exercise. Muscle Nerve. 2005;32:633–638. doi: 10.1002/mus.20385. [DOI] [PubMed] [Google Scholar]
- 34.Jones JA, Riggs PK, Yang T, Pedemonte CH, Clarke M, Feedback D, Au W. Ionizing radiation-induced bioeffects in space and strategies to reduce cellular injury and carcinogenesis. Aviat Space Environ Med. 2007;78(Suppl 1):A67–A78. [PubMed] [Google Scholar]
- 35.Epperly MW, Defilippi S, Sikora C, Gretton J, Kalend K, Greenberger JS. Intratracheal injection of manganese superoxide dismutase (MnSOD) plasmid/liposomes protects normal lung but not orthotopic tumors from irradiation. Gene Ther. 2000;7:1011–1018. doi: 10.1038/sj.gt.3301207. [DOI] [PubMed] [Google Scholar]
- 36.Epperly MW, Kagan VE, Sikora CA, Gretton JE, DeFilippi SJ, Bar-Sagi D, Greenberger JS. Manganese superoxide dismutase-plasmid/liposome (MnSOD-PL) administration protects mice from esophagitis associated with fractionated irradiation. Int J Cancer (Radiat Oncol Invest) 2001;96:221–233. doi: 10.1002/ijc.1023. [DOI] [PubMed] [Google Scholar]
- 37.Stanford M, Jones JA. Space radiation concerns for manned exploration. Acta Astronaut. 1999;45:39–47. doi: 10.1016/s0094-5765(99)00055-7. [DOI] [PubMed] [Google Scholar]