Abstract
Background
Arterial blood pressure (BP) measurement at least every five minutes is part of the American Society of Anesthesiologists' (ASA) monitoring standard, but prolonged BP gaps in electronic anesthesia records have been noted. We undertook multicenter studies to determine the frequency of cases with at least one interval ≥ 10 minutes between successive BP measurements and then to ascertain if educational feedback via an electronic, near real-time notification system alerting providers to the presence of such gaps would reduce their incidence.
Methods
We evaluated 212,706 electronic anesthesia records from three large academic centers. We determined the fraction of cases with ≥ 10 minute BP monitoring gaps at baseline and did a root cause analysis to determine common causes for these lapses. We then designed and implemented automated systems at two of the hospitals to notify point-of-care providers immediately after such 10-minute gaps occurred and determined the subsequent impact of this feedback on BP gap incidence, compared to baseline.
Results
At Hospital A, the notification system reduced the incidence of cases with at least one BP gap (1.48% ± 0.19% SD vs 0.79% ± 0.36% SD, p<0.0001). At Hospital B, the gap incidence was not significantly altered when notification was provided after a 10-min gap had already occurred (2.72% ± 0.60% SD vs. 2.45% ± 0.48% SD, P=0.27), but the incidence was reduced when such notification was provided after 6 minutes without a BP reading (2.72% ± 0.60% SD vs 1.54% ± 0.19% SD, P<0.0001). At Hospital C, where notification was not implemented, the baseline rate of BP gaps was consistent across the preintervention and follow-up periods (7.03% ± 1.27% SD vs. 7.13% ± 0.11% SD, p=0.74). Although monitors disconnected during position change was the most common identifiable cause of BP gaps, reasons for the missing BPs were often not documented. During a week when the electronic charting system was temporarily inoperable, no BP gaps were noted on a convenience sample of 500 paper records from Hospital A (99% upper confidence limit = 0.83%).
Conclusions
BP gaps of ≥ 10 minutes were common in electronic anesthesia records, and their incidence was reduced but not eliminated by near real-time feedback to providers. The ASA standard for every 5 min BP documentation may not be achievable with current practices and technology. Anesthesia information management systems users need to be cognizant of the potential for gaps in BP measurement, take steps to minimize their occurrence, and document an explanation when such failures occur.
Background
The American Society of Anesthesiologists' (ASA) “Standards for Basic Anesthetic Monitoring”1 require measurement and evaluation of arterial blood pressures (BP) at least every five minutes except “under extenuating circumstances,”1 but acknowledge that “brief interruptions of continual monitoring may be unavoidable.” The standards apply to all patients undergoing anesthetics (general, regional, or monitored anesthesia care). Although there is no published Level I or II evidence that the frequency of BP measurement during anesthesia is related to improved outcomes, the ASA monitoring standards exist, in large part, because use of intraoperative physiologic monitors has become fundamental to anesthesiologists' efforts to prevent adverse events.2,3 While a reduction in patient mortality has been attributed to the establishment of monitoring standards,4 actual practice may not match the standards. For example, Figure 1 shows a 15-minute gap in BP monitoring after induction of general anesthesia. The gap was due to a failure to engage the automatic cycling function of a noninvasive BP cuff.
Figure 1. Case Example of a 15-Minute Gap in Arterial Blood Pressure Monitoring.
Example of a gap in blood pressure monitoring as displayed on an anesthesia information management systems (AIMS) workstation. Blood pressure readings are indicated by the “v” (systolic), “x” (mean), and “^” (diastolic). Here a 15-minute gap in blood pressure monitoring was caused by a failure to initiate the auto-cycle feature of the noninvasive blood pressure cuff.
Anesthesia information management systems (AIMS) provide more accurate intraoperative physiologic data than those recorded on manual records.5,6 In contrast to paper records, prolonged data lapses in electronic records have been reported7 and gaps in automatic recording of BPs had been anecdotally noted at all three of the authors' institutions. Our preliminary work at one hospital revealed that some patients had ≥ 10-minute gaps in BP documentation in the AIMS database.8 In our evaluation of this problem, we chose to focus on gaps that are at least double the established ASA standard1 to identify gaps in BP monitoring that are unequivocally meaningful.
To more fully understand the frequency, causes, and possible solutions to the problem of intraoperative gaps in BP monitoring, we undertook studies at three large academic institutions, each with a well established and different commercially available AIMS, to determine (1) the incidence of cases with at least one interval ≥ 10 minutes between successive BP measurements, (2) the etiology of such gaps, and (3) if AIMS-based near real-time feedback to providers when gaps occurred would lead to a subsequent reduction in their incidence.
Because automated systems of process monitoring, exception detection and annunciation are acknowledged to out-perform those based on human vigilance,9–12 we expected that automatically notifying providers about the presence of BP gaps would call attention to this monitoring problem. Although noninvasive BP monitors are automated systems (i.e., the provider sets the cycling frequency and the machine automatically measures BP), they remain under supervisory control of the operator, and therefore we hypothesized that immediate feedback when monitoring gaps occurred would improve subsequent performance.13 We chose this AIMS-based methodology because we and others have previously shown the effectiveness of our general method of automatically detecting and notifying clinicians about documentation errors.14–19
Materials and Methods
The study protocol was approved or exempted from review by the IRBs of all three hospitals (A, B, and C) involved in the study.
To assess baseline performance before implementation of a BP gap notification system, we interrogated the AIMS databases at each of the three hospitals using structured query language for the values and timestamps of all invasive and noninvasive BP variables (i.e., systolic, mean, and diastolic). We also noted the internal identification of the anesthesia provider logged into the AIMS at the time of the start of each gap. At Hospital B, attribution could be made for all gaps, because the identification of the currently logged-in provider is recorded with each vital sign. At Hospital A, due to software differences in the AIMS, such determinations could only be made unambiguously when a single care provider was logged into the AIMS for the entire case. For the purpose of this study, we characterized a case as being noncompliant with the ASA standard if there were at least one gap ≥ 10 minutes between successive BP measurements during the interval from induction of anesthesia to the end of surgery. At Hospitals A and B, the default BP monitor cycle frequency interval was 3 min, whereas at Hospital C, it was either 3 or 5 minutes, depending on the procedure location.
We defined induction of anesthesia as the earliest of (1) documented time of first IV induction drug administration, (2) manually documented induction of anesthesia time, or (3) first time 0.20 minimum alveolar concentration* of volatile anesthetic was detected in the breathing circuit. We defined the end of surgery as the documented time of the end of surgery event, or the time of tracheal extubation if the surgery end event was missing.20
To remove temporal variation as a potential confounder (e.g., the arrival of new anesthesia residents in July), we chose baseline comparison periods that corresponded to our intervention period, but in the prior calendar year(s) as outlined in the study timeline shown in Figure 2.
Figure 2. Data Collection Periods (Baseline and Intervention).
Schema showing the data collection periods for the three hospitals included in the study. Hospital A provided baseline data from two prior calendar years (9/06–4/07, 9/07–4/08), data from the intervention period (9/08–4/09), and data from the follow-up period (8/09 to 10/09). Hospital B provided baseline data from the prior calendar year (5/07 to 5/08) and data from after the initial intervention (7/08 to 4/09). A second intervention was also undertaken at Hospital B (8/09 to 10/09). Hospital C provided baseline data for five years (9/04–7/05, 9/05–7/06, 9/06–7/07, 9/07–7/08, 9/08–7/09) and from the follow-up period (8/09 to 10/09). Note: during the follow-up period the intervention at Hospital A was unchanged, but at hospital B the allowed gap duration was shortened (from 10 minutes to 6 minutes) in a deliberate attempt to improve performance. Hospital C continued to serve as our control, and had no intervention during the follow-up period.
We included all patients ≥ 16 years old who underwent anesthesia (general, regional, or monitored anesthesia care) and whose cases were recorded electronically in the AIMS during the periods of interest. Cases involving cardiopulmonary bypass were excluded because of the occasional use of circulatory arrest and expected missing BP values.
At Hospital A we also studied a convenience sample of 500 cases that were documented manually on paper records due to the AIMS being unavailable for technical reasons. This serendipitous event allowed us to evaluate the authors' impression that gaps on manually prepared anesthesia records are seldom found. The providers completing the manual records were the same as those using the AIMS, and the system outage occurred in the same timeframe as the study interval. The 99% upper confidence limit for the incidence of BP gaps was determined using the method of Blyth-Still-Casella (StatExact® v9, Cytel, Inc, Cambridge, MA).
At Hospitals A and B, computer software was developed that queried the AIMS database at 1-minute intervals for currently running cases in which a new BP was not recorded within 10 minutes after a prior measurement. At both hospitals, a notification was sent to the clinician at the point-of-care via an on-screen pop-up message. At Hospital A a text message was also sent to the clinician's alphanumeric pager (Figure 3). At Hospital A the only end-user education was a one-time general announcement at grand rounds as to how the notification messages would be provided. At Hospital B there was no advance notice, because the method used the same pathway in place for other documentation reminders and notifications. A notification system was not implemented at Hospital C, which served as an internal control for changes in practice over time and independent of the notification systems. Thus, improvement in performance could be attributed to the notification process itself, not to an intensive educational effort about the importance of monitoring BP.
Figure 3.
Panel A shows a schematic overview of the function of the automatic chart review system. The dashed line indicates a recursive loop that runs every minute. This system was implemented independently and using different software resources at two centers (Hospitals A and B). An example of the pop-up output is shown in Panel B. This appeared on the anesthesia information management system workstation in the provider's operating room. Additionally, at Hospital A an alphanumeric text page was sent to the individual who had entered their name as the anesthetist for the case.
After an eight-month intervention period of electronic notifications, the fraction of cases with at least one BP gap, grouped in 4-week epochs, was compared to a corresponding time interval before the intervention. A nonpaired Student's T-test was performed following a Freeman-Tukey Double Arcsine Transformation of the fractional data, with P<0.05 required to claim statistical significance, as previously described.21 To account for any external practice pattern changes over time that might have impacted our results, we also compared the percentage of cases with gaps during intervals corresponding to the pre and postintervention periods at Hospitals A and B at our control site (Hospital C) (Figure 2).
To exclude the potential influence of a small number of individual providers on the overall occurrence of gaps in BP monitoring, we determined at both Hospital A and B the number of cases with gaps by each provider divided by the total number of cases where a responsible provider could be assigned to the gap.
To evaluate possible causes for gaps in BP monitoring, we performed a post hoc root cause analysis on a random sample of 200 charts with BP gaps from each hospital in the intervention group (Hospital A and B). Each record was manually examined by two reviewers who agreed by consensus on a cause for each gap, where discernible, from either explicit documentation or from surrounding events in the AIMS record. Etiologies were then categorized and the five most common causes were tabulated and reported.
Finally, at Hospital B, after analysis of the gaps before and after implementation of the 10-minute BP gap notification, the interval of gap notification was reduced to 6 minutes with no follow-up BP. Since the outcome measurement was still the incidence of 10-minute gaps, this represented a preemptive approach, rather than modification of behavior through education. Before making this notification interval change, the incidence of BP gaps by provider was presented at grand rounds as part of a quality assurance presentation about the ASA monitoring standards, and the report was also distributed via email to all anesthesia care providers. Ten weeks of data during this follow-up period between 8/09 and 10/09, grouped in 2-week bins, were then re-evaluated for the incidence of intraoperative gaps of at least 10 minutes. Data on the incidence of anesthetics with at least one 10-minute BP gap from this follow-up period were then compared to both the original baseline data, as well as the postintervention data. Again, we applied the Student's t-test to the transformed percentages, to determine statistical significance. We also obtained data from Hospitals A and C for the same time period, and report all of the data from this follow-up period.
Results
The manual review of 500 paper anesthesia records from Hospital A did not reveal a single case in which a 10-minute gap in BP recording was present (99% upper confidence limit for BP gaps of 0.83%). However, the baseline (preintervention) incidence of gaps in the electronic records among the 212,706 cases analyzed at hospitals A, B, and C was 1.48%, 2.72%, and 7.13%, respectively (Table 1).
Table 1.
Analysis of Gaps by Institution
| Hospital A | Preintervention | Postintervention | Follow-Up† | |
|---|---|---|---|---|
| Number of Cases | 32,520 | 20,747 | 5,542 | |
| Mean % of Cases with BP Gaps ≥ 10 min | 1.48% | 1.21% | 0.79% | |
| Std Deviation | 0.19% | 0.30% | 0.36% | |
| P = 0.03 | P=<0.00011 | P=<0.022 | ||
| Hospital B | Preintervention | Postintervention | Follow-Up† | |
|---|---|---|---|---|
| Number of Cases | 17,252 | 17,241 | 4,489 | |
| Mean % of Cases with BP Gaps ≥ 10 min | 2.72% | 2.45% | 1.54% | |
| Std Deviation | 0.60% | 0.48% | 0.19% | |
| P = 0.27 | P<=0.00011 | P=0.00142 | ||
| Hospital C | Preintervention | Follow-Up† | ||
|---|---|---|---|---|
| Number of Cases | 111,438 | 6,477 | ||
| Mean % of Cases with BP Gaps ≥ 10 min | 7.03% | 7.13% | ||
| Std Deviation | 1.27% | 0.11% | ||
| P=0.74 | ||||
Follow-Up vs. Preintervention
Follow-Up vs. Postintervention
Note: The follow-up period consisted of five 2-week epochs of data from all three hospitals. During the follow-up period a second intervention was undertaken at Hospital B (notifications after 6-minute blood pressure (BP) gaps), in a deliberate, renewed attempt to reduce gap incidence. During the follow-up period the original intervention (notifications after 10-minute blood pressure gaps) was continued at Hospital A, and no intervention occurred at Hospital C.
Percentage of cases with gaps in blood pressure monitoring of ≥ 10 minutes from induction of anesthesia to extubation / end of surgery by hospital before and after the notifications were instituted.
At Hospital A, there was a statistically significant reduction in the percentage of cases with at least one BP gap ≥ 10 minutes after implementation of the notification system (1.48% vs 1.21%, p<0.03). The reduction in cases with BP gaps at Hospital B was not statistically different (2.72% vs 2.45%, p=0.27). At Hospital C where no intervention was undertaken, the baseline rate of BP gaps was consistent across the preintervention and follow-up periods (Hospital C – 7.03% vs. 7.13%, p=0.74).
During the follow-up period in which the notification interval was reduced from 10 minutes to 6 minutes at Hospital B, but maintained at 10 minutes at Hospital A, there was a further reduction in BP gaps at Hospital A (1.21% vs 0.79%, p=0.02) and improvement at Hospital B (2.45% vs. 1.54%, p=0.0014), when compared to the initial postintervention period (Table 1).
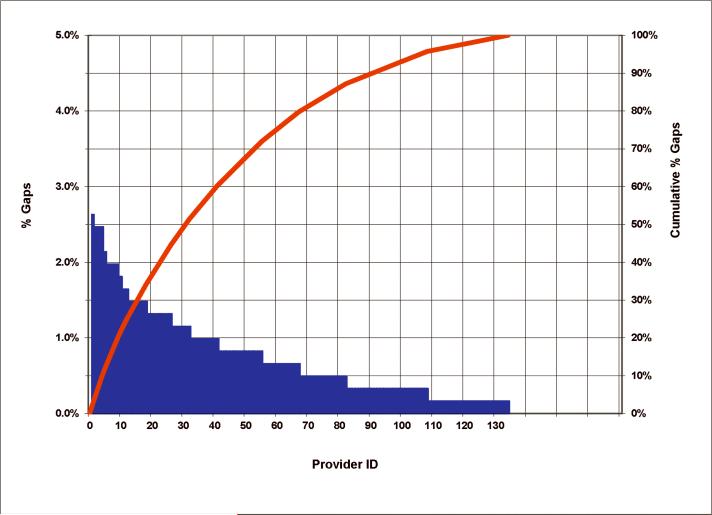
The gaps were attributable to a large number of providers, with no one provider at either Hospital A or B contributing more than 3.7% of the cases with gaps (Figure 4).
Figure 4. Distribution of Cases with Gaps by Provider at Hospital A and Hospital B.
Distribution of all blood pressure gaps at Hospital A and Hospital B by provider. A blood pressure gap was considered to be present if there was at least one 10 min interval without a blood pressure recorded in the anesthesia information management systems, during the period from induction of anesthesia to the end of surgery / extubation. The number of gaps for each provider with at least one case with a blood pressure gap during the study period (blue bars) is plotted as a percentage of the total number of gaps. Providers were numbered sequentially starting with 1 so that the cumulative number of staff is indicated by the x intercept of each point along the cumulative percentage curve (red line).
The timing of BP gaps at one of the study sites (Hospital A) during the intraoperative portion of the case (induction to end of surgery) is shown in Table 2. We excluded gaps occurring before induction or after the end of surgery because these intervals included times when the ASA monitoring standard does not apply (e.g., checking the patient into the room, waiting for the surgeon to arrive, waiting for a postanesthesia care unit (PACU) slot after tracheal extubation, etc.). The data reveal that 90% of BP gaps occurred during the surgical phase of care (start to end of surgery), comprising 78% of the intraoperative time. Only 10% of gaps occurred between induction and the start of surgery, which accounted for 22% of the intraoperative time. The relative timing of BP gaps did not differ significantly before and after the notification intervention was applied.
Table 2.
Timing of Gaps in Blood Pressure (BP) Monitoring at Hospital A
| Hospital A | Induction – Start of Surgery | Start of Surgery – Extubation or End of Surgery | ||
|---|---|---|---|---|
| Preintervention | Postintervention | Preintervention | Postintervention | |
| % of all BP Gaps ‡ | 11.9% | 8.6% | 88.1% | 91.4% |
| 10.3% (average) | 89.7% (average) | |||
Percentage of all cases with gaps in blood pressure monitoring stratified by case phase (induction – start of surgery; start of surgery – extubation / end of surgery).
Note: this analysis examined only BP gaps occurring between induction and the end of surgery (not the entire intraoperative period)
A root cause analysis of BP gaps at Hospital A and B is shown in Figure 5. Where a reason for the BP gap could be assigned, the five most common causes were (1) monitors disconnected during position change (e.g. supine to prone), (2) patient awake in operating room, waiting for PACU bed, (3) BP monitor failure, (4) AIMS communication failure, and (5) BP cuff turned off while placing an indwelling arterial catheter. However, at both hospitals, the most common cause of a gap was “indeterminate,” i.e., could not be determined from a retrospective evaluation of the anesthesia record.†
Figure 5. Root Cause Analysis of Arterial Blood Pressure Gaps in 400 Cases at Hospital A and B.
Root cause analysis of 400 randomly selected blood pressure gaps (200 at Hospital A, 200 at Hospital B). Three causes accounted for at least 10% of all gaps during the intraoperative period (95% lower confidence limit): indeterminate, position change, and waiting for postanesthesia care unit. *See footnote in the results section.
Discussion
Summary of Findings
At three large, academic institutions with well established AIMS, we found an incidence of 1.5% – 7.0% of anesthetics in which there were one or more gaps in recording successive BPs that were at least twice the current 5-minute ASA monitoring standard. While the impact of these gaps on patient outcomes is unknown, there may be medico-legal implications of such lapses.7 Education, as implemented by an immediate feedback mechanism after the occurrence of a minute BP gap, was successful in reducing the incidence of such gaps at Hospital A where both on-screen pop-ups and text pages were used, but not at Hospital B, where only pop-ups were used. At the latter hospital, performance improved only when the notification interval was reduced from 10 minutes to 6 minutes and an educational program on the ASA monitoring standards, including public disclosure of deficiencies, was made. It is unlikely that improvement in performance over time represented heightened awareness in the anesthesia community at large about the importance of BP monitoring, because there was no change in Hospital C (where a BP gap notification system was not implemented) between the intervals corresponding to the pre and postimplementation periods.
Comparison with Previous Reports
Previous reports have demonstrated that AIMS-based notifications improve process of care variables related to documentation. These variables have included timely documentation of patient drug allergies,14 placement of intraoperative arterial catheters,17 attending compliance statements,15 and required events for billing.22 None of these reminder systems was completely effective, even when overt financial penalties or in-person follow-up visits from an authority figure were used. It is probably not reasonable to expect that the incidence of cases with gaps in monitoring BP can be reduced to zero, and the ASA standard recognizes that brief interruptions in continual measurements may be unavoidable.1 Furthermore, monitoring device and AIMS failures cannot be eliminated. However, our analysis was conservative in that we only considered gaps of 10 minutes of longer, an interruption which we consider to be quite long.
Root Cause Analysis
The root cause analysis suggested that many providers choose to omit monitoring at certain points in the case (Figure 5). However, the rationale for the choice was rarely documented in the record. Given that it is relatively simple to arrange monitor connections so that they are maintained during patient repositioning, and repositioning is a time of potential hemodynamic instability,23 we suggest that the apparently common practice of disconnecting monitors during repositioning be reconsidered. Likewise, it is simple to continue to monitor patients in the operating room while waiting for a PACU bed, and to switch the BP cuff to a different extremity when attempting to place an arterial catheter. In any case, we consider it prudent to document an explanation for a failure to obtain a BP according to the ASA standard if it occurs (e.g., surgeon leaning on the BP cuff, device failure, AIMS failure, etc.). Alternatively, both use errors and technical failures may be thought of as representing a failure of the clinician to perform adequate supervisory control over the automatic device.24
Our data regarding the overall baseline incidence of gaps in monitoring and their root causes are consistent with prior work. For example, a published pilot study evaluating BP gaps revealed a similar incidence approximately a decade ago.‡ Similarly, a study of pulse oximetry gaps by Reich et al. revealed a 9% incidence of 10 minute gaps in SpO2 value recording in an AIMS, attributed largely to signal acquisition problems related to technical limitations of the then available pulse oximeters.25 These prior results suggest that the data collected from the three academic centers involved in our study of BP gaps will be widely applicable to any facility that has implemented an AIMS. Indeed, one institution (where the standard BP cycle interval was 5 minutes in all locations) reported a 39% incidence of cases with at least one BP gap >10 min.§
Finally, our study found zero 10-minute BP gaps in a cohort of 500 paper charts which were created during the study period. “Smoothing” of high and low BPs during the recording of manual anesthesia records is well known.6 We strongly suspect the absence of gaps in paper anesthesia records reflects a similar process of “interpolation” of missing values. Given that the providers who created these manual records were the same providers who were using the AIMS, we consider it highly unlikely that they immediately changed their monitoring practices as a result of the AIMS unexpectedly becoming unavailable.
Implications
Cycle frequency for automatic BP measurement is likely to affect the incidence of BP gaps. With a cycle frequency of every 5 minutes, missing two consecutive determinations leads to a 10-minute monitoring interruption. In contrast, when the cycle frequency is every 3 minutes, four complete measurement cycles must be missed before such a gap occurs. The longer average cycle interval at Hospital C was likely the cause of its higher baseline incidence of BP gaps, compared to the other two hospitals. Since a single failure to acquire a reading if the cycle interval is 5 minutes will cause one to be out of compliance with ASA standards, an interval of 2 ½ – 3 minutes might be a better choice, especially during procedures where hemodynamic instability is common. However, tissue injury due to repeated BP cuff cycling at intervals even longer than 5 minutes has been reported,26,27 so the risk-to-benefit ratio of more frequent cycling needs to be considered.
While we chose to use near-real time notifications as a method for enhancing supervisory control of BP monitoring during surgery, there may be other strategies which could also be successful. These could include providing notifications when the noninvasive BP cuff is placed in standby mode while an anesthetic is in progress and an alternative source of pressure measurement is not being provided. This is a clear use case for integration of state monitoring either across devices (e.g., anesthesia machine and physiologic monitor), or under the aegis of an AIMS. Because an AIMS can easily determine if an anesthetic is underway in a given patient, and does not have a requirement for competing vendors to cooperate or agree to common standards, this approach may be much simpler to implement. Indeed, some vendors have already implemented rudimentary support for such processes in their systems (as demonstrated by the implementation at Hospital A). The human factors literature demonstrates that while automation can relieve clinicians of the burden of repeatedly performing specific tasks, automation tends to increase the requirements for attention to end-user training, interface design, and interaction design.24,28
Limitations
First, the notifications provided in the initial intervention at Hospital A and B occurred after the BP gap had already taken place, so they could not have prevented the inciting event. Thus, improvement at Hospital A must have resulted from user education over time to avoid the gaps in the first place.14 Why the same effect was not shown at Hospital B after initial implementation is unclear, but the dual notification method at Hospital A might have been responsible for the better result. Learning effects may be reduced in situations where a barrage of alarms causes users to suffer from alert fatigue.29 However, given the relatively low frequency of the BP gap alert (1.5–2.7% of cases), this was unlikely to have been a major factor during our study.
Second, the latency of the notification systems (i.e., the time between gap detection and when the anesthesia care provider noticed the reminder message) was not formally measured and likely differed between the two hospitals.30 However, since impact of the notifications was primarily one of changing future behavior, this issue is not of major importance.
A third limitation is that the hospitals involved in the study each have their own BP measurement practices, including different default BP cycling frequencies (e.g., 3 minutes vs 5 minutes). In addition, it was discovered during manuscript revision that the BP monitors at Hospitals A and C, but not B, are configured to turn off automatic cycling if a failure to obtain a BP occurs, and that an obvious warning is not provided by the monitor when this occurred. Thus, if the surgeon leaned on the BP cuff during a measurement and caused a failure to obtain a pressure, subsequent pressures would not be taken until the anesthesia care provider noticed that a new BP had not been obtained and reset automatic recycling. These differences likely reduce the generalizability of any one approach to reducing BP measurement gaps, because the implementation of a particular strategy (e.g. send a page after 10 minutes) might fail to have the intended effect at another institution with different practice characteristics. However, configuring the BP monitor not to disable automated cycling in the event of a measurement failure could potentially reduce the incidence of seemingly random intraoperative BP gaps. Unfortunately, such a modification is not currently possible for the devices in use at Hospitals A and C, because this default behavior cannot be overridden. At the least, monitors configured to terminate automated BP cycling in the event of acquisition failure should provide a strong alert to the provider if such an event occurs.
Fourth, our root cause analysis was limited by the frequent absence of practitioner-entered event comments in situations where a BP gap occurred, undermining the ability to determine the cause of the BP gap.
Conclusions
Despite the lack of Level I or II evidence that the frequency of measuring BP influences patient outcomes, recording such measurements at least every 5 minutes is an ASA standard of care during anesthetics.1 However, our data reveal that BP gaps of > 10 minutes are common in AIMS records. Given the similar incidence of such lapses at three large academic centers with three different, well-established AIMS, it is likely that this represents common performance. Although in many cases the source of the gaps is related to addressable user errors, technical failures by the monitors and the AIMS also occur. Providing notifications to users about the occurrence of gaps in BP monitoring was successful in reducing the incidence of this problem, but did not eliminate gaps completely, and is not likely to do so. Regulatory standards need to recognize device and software problems leading to interruptions of continual monitoring to a greater extent than they do currently. However, AIMS users also need to be more cognizant of potential failures to acquire BP measurements, to take steps to minimize their occurrence, and to document an explanation when such inevitable failures occur.
Acknowledgments
Funding: Financial support for the preparation of this manuscript was provided from a Research Fellowship Grant from the Foundation for Anesthesia Education and Research, 5T32GM007592 from the National Institute of Health, as well as by department funds of the Department of Anesthesia, Critical Care, and Pain Medicine, Massachusetts General Hospital.
Footnotes
For the purposes of our study, we used the following values to represent approximate equivalents of 0.20 MAC: N2O = 30%, Isoflurane = 0.2%, Desflurane = 1.2%, Sevoflurane = 0.4%
Note: In most of the cases where a gap was attributed to a positioning change that occurred after induction, there was also a corresponding change before tracheal extubation. Whether the position change was attributed to the surgical period (start to end of surgery) depended on when the provider noted the “End of Surgery” event. Unfortunately, this is not a reliable time stamp as providers tend to have different views of what constitutes the “End of Surgery” (e.g. the last stitch being placed, the dressings going on, after the patient is repositioned for wake-up, or another time point).
Salgar V, Hartmannsgruber M, Gillerman R, Alterman M, Feldman J. Manual Analysis of 110 Consecutive Automated Anesthesia Records- A Pilot Study to Determine the Incidence of NonContinual Blood Pressure Monitoring. Society for Technology in Anesthesia Annual Meeting, 2001.
Epstein, RH, Ehrenfeld, JM, Sandberg, WS, Vigoda, MM. Frequency Of Prolonged Gaps In Blood Pressure Documentation In Anesthesia Information Management Systems. IARS Annual meeting, S-99, 2009.
The authors declare no conflicts of interest.
Reprints will not be available from the authors.
This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.American Society of Anesthesiologists Standards of the American Society of Anesthesiologists: Standards for Basic Anesthetic Monitoring. 2004 http://www.asahq.org/publicationsAndServices/standards/02.pdf.
- 2.Eichhorn JH. Prevention of intraoperative anesthesia accidents and related severe injury through safety monitoring. Anesthesiology. 1989;70:572–7. doi: 10.1097/00000542-198904000-00002. [DOI] [PubMed] [Google Scholar]
- 3.Tinker JH, Dull DL, Caplan RA, Ward RJ, Cheney FW. Role of monitoring devices in prevention of anesthetic mishaps: a closed claims analysis. Anesthesiology. 1989;71:541–6. doi: 10.1097/00000542-198910000-00010. [DOI] [PubMed] [Google Scholar]
- 4.Mayfield JB. The impact of intraoperative monitoring on patient safety. Anesthesiol Clin. 2006;24:407–17. doi: 10.1016/j.atc.2006.03.001. [DOI] [PubMed] [Google Scholar]
- 5.Muravchick S, Caldwell JE, Epstein RH, Galati M, Levy WJ, O'Reilly M, Plagenhoef JS, Rehman M, Reich DL, Vigoda MM. Anesthesia information management system implementation: a practical guide. Anesth Analg. 2008;107:1598–608. doi: 10.1213/ane.0b013e318187bc8f. [DOI] [PubMed] [Google Scholar]
- 6.Reich DL, Wood RK, Jr., Mattar R, Krol M, Adams DC, Hossain S, Bodian CA. Arterial blood pressure and heart rate discrepancies between handwritten and computerized anesthesia records. Anesth Analg. 2000;91:612–6. doi: 10.1097/00000539-200009000-00022. [DOI] [PubMed] [Google Scholar]
- 7.Vigoda MM, Lubarsky DA. Failure to recognize loss of incoming data in an anesthesia record-keeping system may have increased medical liability. Anesth Analg. 2006;102:1798–802. doi: 10.1213/01.ane.0000217235.25350.5e. [DOI] [PubMed] [Google Scholar]
- 8.Ehrenfeld JM, Sandberg WS. Incidence of Intraoperative Gaps in Patient Monitoring During Anesthesia. Anesthesiology. 2006;105 [Google Scholar]
- 9.Allnutt MF. Human factors in accidents. Br J Anaesth. 1987;59:856–64. doi: 10.1093/bja/59.7.856. [DOI] [PubMed] [Google Scholar]
- 10.Frankmann JP, Adams JA. Psychological bulletin. Psychol Bull. 1962;59:257–72. [PubMed] [Google Scholar]
- 11.Salvendy G. Handbook of human factors. Wiley; New York: 1987. [Google Scholar]
- 12.Reason JT. Human error Cambridge [England] Cambridge University Press; New York: 1990. [Google Scholar]
- 13.Sarter NBW, David D. How in the World Did We Ever Get into That Mode? Mode Error and Awareness in Supervisory Control Human Factors: The Journal of the Human Factors and Ergonomics Society. 1995;37:5–19. [Google Scholar]
- 14.Sandberg WS, Sandberg EH, Seim AR, Anupama S, Ehrenfeld JM, Spring SF, Walsh JL. Real-time checking of electronic anesthesia records for documentation errors and automatically text messaging clinicians improves quality of documentation. Anesth Analg. 2008;106:192–201. doi: 10.1213/01.ane.0000289640.38523.bc. [DOI] [PubMed] [Google Scholar]
- 15.Spring SF, Sandberg WS, Anupama S, Walsh JL, Driscoll WD, Raines DE. Automated documentation error detection and notification improves anesthesia billing performance. Anesthesiology. 2007;106:157–63. doi: 10.1097/00000542-200701000-00025. [DOI] [PubMed] [Google Scholar]
- 16.St Jacques P, Sanders N, Patel N, Talbot TR, Deshpande JK, Higgins M. Improving timely surgical antibiotic prophylaxis redosing administration using computerized record prompts. Surg Infect. 2005;6:215–21. doi: 10.1089/sur.2005.6.215. [DOI] [PubMed] [Google Scholar]
- 17.Kheterpal S, Gupta R, Blum JM, Tremper KK, O'Reilly M, Kazanjian PE. Electronic reminders improve procedure documentation compliance and professional fee reimbursement. Anesth Analg. 2007;104:592–7. doi: 10.1213/01.ane.0000255707.98268.96. [DOI] [PubMed] [Google Scholar]
- 18.Wax DB, Beilin Y, Levin M, Chadha N, Krol M, Reich DL. The effect of an interactive visual reminder in an anesthesia information management system on timeliness of prophylactic antibiotic administration. Anesth Analg. 2007;104:1462–6. doi: 10.1213/01.ane.0000263043.56372.5f. [DOI] [PubMed] [Google Scholar]
- 19.O'Reilly M, Talsma A, VanRiper S, Kheterpal S, Burney R. An anesthesia information system designed to provide physician-specific feedback improves timely administration of prophylactic antibiotics. Anesth Analg. 2006;103:908–12. doi: 10.1213/01.ane.0000237272.77090.a2. [DOI] [PubMed] [Google Scholar]
- 20.Dexter F, Epstein RH, Lee JD, Ledolter J. Automatic updating of times remaining in surgical cases using bayesian analysis of historical case duration data and “instant messaging” updates from anesthesia providers. Anesth Analg. 2009;108:929–40. doi: 10.1213/ane.0b013e3181921c37. [DOI] [PubMed] [Google Scholar]
- 21.Dexter F, Marcon E, Epstein RH, Ledolter J. Validation of statistical methods to compare cancellation rates on the day of surgery. Anesth Analg. 2005;101:465–73. doi: 10.1213/01.ANE.0000154536.34258.A8. [DOI] [PubMed] [Google Scholar]
- 22.Vigoda MM, Lubarsky DA. The medicolegal importance of enhancing timeliness of documentation when using an anesthesia information system and the response to automated feedback in an academic practice. Anesth Analg. 2006;103:131–6. doi: 10.1213/01.ane.0000221602.90315.49. [DOI] [PubMed] [Google Scholar]
- 23.Sudheer PS, Logan SW, Ateleanu B, Hall JE. Haemodynamic effects of the prone position: a comparison of propofol total intravenous and inhalation anaesthesia. Anaesthesia. 2006;61:138–41. doi: 10.1111/j.1365-2044.2005.04464.x. [DOI] [PubMed] [Google Scholar]
- 24.Lee JD. Review of a pivotal Human Factors article: “Humans and automation: use, misuse, disuse, abuse”. Hum Factors. 2008;50:404–10. doi: 10.1518/001872008X288547. [DOI] [PubMed] [Google Scholar]
- 25.Reich DL, Timcenko A, Bodian CA, Kraidin J, Hofman J, DePerio M, Konstadt SN, Kurki T, Eisenkraft JB. Predictors of pulse oximetry data failure. Anesthesiology. 1996;84:859–64. doi: 10.1097/00000542-199604000-00013. [DOI] [PubMed] [Google Scholar]
- 26.Celoria G, Dawson JA, Teres D. Compartment syndrome in a patient monitored with an automated blood pressure cuff. J Clin Monit. 1987;3:139–41. doi: 10.1007/BF00858363. [DOI] [PubMed] [Google Scholar]
- 27.Lin CC, Jawan B, de Villa MV, Chen FC, Liu PP. Blood pressure cuff compression injury of the radial nerve. J Clin Anesth. 2001;13:306–8. doi: 10.1016/s0952-8180(01)00262-8. [DOI] [PubMed] [Google Scholar]
- 28.Parasuraman R, Riley V. Humans and Automation: Use, Misuse, Disuse, Abuse. Human Factors. 1997;39 [Google Scholar]
- 29.Dixon SR, Wickens CD, McCarley JS. On the independence of compliance and reliance: are automation false alarms worse than misses? Hum Factors. 2007;49:564–72. doi: 10.1518/001872007X215656. [DOI] [PubMed] [Google Scholar]
- 30.Epstein RH, Dexter F, Ehrenfeld JM, Sandberg WS. Implications of event entry latency on anesthesia information management decision support systems. Anesth Analg. 2009;108:941–7. doi: 10.1213/ane.0b013e3181949ae6. [DOI] [PubMed] [Google Scholar]