Abstract
In view of the severe immunosuppression in visceral leishmaniasis (VL), a rational approach to effectively combat the parasitic scourge would be to enhance the immune status of the host. Use of CpG oligodeoxynucleotide (CpG-ODN) against leishmaniasis has previously been reported, especially as an immunomodulator and adjuvant with various immunogens. In the present study, experiments were carried out with BALB/c mice and hamsters infected with Leishmania donovani. Immunostimulating class B bacterial CpG-ODN namely, ODN-2006, was administered at various doses by the intraperitoneal (i.p.) route. The dose of CpG-ODN-2006 (1 nM/single dose) showing the most antileishmanial activity was given as free and liposomal forms with different doses of miltefosine, namely, 5 and 10 mg/kg of body weight, for 5 days in mice and hamsters, respectively. Among the various groups, mice coadministered liposomal CpG-ODN and miltefosine (5 mg/kg) showed the best inhibitory effect (97% parasite inhibition) compared with free CpG-ODN plus miltefosine and miltefosine, free CpG-ODN, and liposomal CpG-ODN given separately. Similar responses were observed in the case of hamsters, where the combination of liposomal CpG-ODN with miltefosine (10 mg/kg) gave 96% parasite inhibition. Promising antileishmanial efficacy was observed in animals treated with liposomal CpG-ODN and miltefosine.
INTRODUCTION
Visceral leishmaniasis (VL), also known as kala-azar in the Indian subcontinent, is caused by the protozoan parasites Leishmania donovani and L. infantum (L. chagasi) and is a potentially fatal disease with a worldwide distribution, in Asia, East Africa, South America, and the Mediterranean region. The parasites are transmitted through the bite of female sand flies (Lutzomyia sp.) and in the human host are obligate intracellular parasites of the reticulo-endothelial system, surviving and multiplying in different macrophage populations.
Available antileishmanial therapy is inadequate and suffers from several drawbacks. The first-line therapy includes sodium stibogluconate (SbV), which has unfortunately developed resistance in some areas of Bihar, where failure rates of up to 65% have been reported and the use of antimony has been abandoned (20). Pentamidine also went out of favor for treatment of VL (19). The use of liposomal amphotericin B is limited due to its high market price (3). A major milestone in chemotherapy of VL is the discovery of miltefosine, an analogue of phosphatidylcholine initially developed as an anticancer agent (7). It is an effective oral drug, but its use in women of child-bearing age is restricted due to teratogenicity. In addition, it has a long half-life, which might encourage the emergence of resistance once its use becomes widespread (6).
Suppression of immune function in VL infection can compromise treatment efficacy (5). It is usually associated with a depression of Th1 cells and preferential expansion of Th2 cells, and accordingly, skewing of T helper cells toward a Th1 response is considered a promising therapeutic strategy. The findings of Zimmermann et al. (24) against L. major infection imply an important role of bacterial DNA and CpG oligodeoxynucleotide (CpG-ODN) in the instruction of adaptive immune responses. They also point to the therapeutic potential of CpG-ODN in redirecting curative Th1 responses in Th2-driven disorders. Previous work from our lab also strengthens the immunomodulatory role of CpG-ODN for treatment of VL (18). CpG-ODNs are a class of pharmacotherapeutic agents characterized by the presence of an unmethylated CG dinucleotide in specific base sequence contexts (CpG motif). These CpG motifs are not seen in eukaryotic DNA but are present in bacterial DNA, to which they confer immunostimulatory properties (16).
In the present work, we explored a combination of CpG-ODN-2006 and miltefosine at various subcurative doses for the treatment of experimental VL in rodent models. CpG-ODN-2006 contains the CpG motif 5′-GTCGTT-3′, by virtue of which it stimulates mainly human primary B cells (11). However, it has also shown immunomodulatory responses in mouse cells (23). It has been used extensively in antitumor clinical trials (14). This study involves the use of free as well as liposomal CpG-ODN-2006 in combination with different doses of miltefosine, namely, 2.5 and 5 mg/kg of body weight in mice and 5 and 10 mg/kg in hamsters.
MATERIALS AND METHODS
Parasite.
The WHO reference strain of L. donovani (MHOM/IN/80/Dd8), obtained from Imperial College, London, United Kingdom, was maintained in this laboratory as promastigotes in vitro and as amastigotes in golden hamsters (10).
Animals.
BALB/c mice (18 to 20 g) and inbred hamsters (40 to 45 g) of both sexes were used for the study. All experiments were conducted in compliance with the Institutional Animal Ethics Committee (IAEC) guidelines for the use and handling of animals. Throughout the study, the animals were housed in climate-controlled (23 ± 2°C; relative humidity, 60%) and photoperiod-controlled (12-h light-dark cycles) animal quarters. They were fed standard rodent pellet supplemented with grain and had free access to drinking water.
CpG-ODN.
A fully phosphothioated class B oligonucleotide, CpG-ODN-2006, was used in this work. It shows specificity for human Toll-like receptor 9 (TLR9), as it has the optimal human motif 5′-GTCGTT-3′ (24-mer). It was purchased from Trilink Biotechnologies Inc., San Diego, CA. It has the sequence 5′-TCG TCG TTT TGT CGT TTT GTC GTT-3′ and a molecular weight of 7,698 g/mol.
Miltefosine.
Miltefosine was purchased from SynphaBase AG (Switzerland). For the in vivo study, miltefosine was dissolved in deionized water.
Liposome preparation.
Liposomes containing CpG-ODN-2006 were prepared by the dehydration-rehydration vesicle (DRV) method (13, 15, 18).The lipid phase consisted of 1,2-dipalmitoyl-sn-glycero-3-phosphocholine (DPPC) (6 μM) and cholesterol (2 μM) dissolved in chloroform-methanol (2:1 [vol/vol]) in a flat-bottom tube. The solvent was removed by slow evaporation under nitrogen for deposition of a thin film of lipid on the tube surface. The tube was dried to remove any trace of solvent. The lipid film was then hydrated and dispersed in a solution with an appropriate quantity of CpG-ODN in distilled water and processed to obtain the desired preparation of liposomal CpG-ODN. The percent incorporation of CpG into the liposomes was determined using the UV absorption at 260 nm, and the concentration of CpG-ODN in the liposomes was adjusted to the desired concentration prior to administration.
Infection of mice and antileishmanial efficacy evaluation.
BALB/c mice were infected intracardially with 2 × 107 L. donovani amastigotes and randomly sorted into groups of five or six animals each. The same number of mice was kept as an untreated control group. Mice were dosed by the intraperitoneal (i.p.) and oral (p.o.) routes at 7 days postinfection for 5 consecutive days. Animals were sacrificed on day 3 posttreatment (day 14 postinfection). Impression smears of livers were prepared, fixed in methanol, and stained with 10% Giemsa stain in phosphate-buffered saline. The number of amastigotes per 500 liver cell nuclei was determined. The percent inhibition (PI) was calculated for all drug-treated groups in relation to the untreated group.
Infection of hamsters and antileishmanial efficacy evaluation.
The evaluation with hamsters was carried out in accordance with the method described by Gupta et al. (10). Five or six animals were used for each group, and the same numbers were kept as untreated controls. The drug treatment was given by the i.p. and p.o. routes. To assess the effects of drugs, spleen biopsy was performed on each animal on day 7 posttreatment, and amastigote counts were assessed by Giemsa staining (10).The PI was calculated for all drug-treated groups in relation to the untreated group.
Optimization of CpG-ODN and miltefosine dose regimens.
Each group consisted of five or six infected animals in two replicates for each species. CpG-ODN was administered at doses of 0.5, 1, 2, and 5 nM prepared in deionized water by the i.p. route for dose optimization. A 1 nM dose of CpG-ODN was found to be most appropriate for combination studies with both species. Miltefosine was given orally at doses ranging from 1.25 mg/kg to 20 mg/kg for mice and from 2.5 mg/kg to 40 mg/kg for hamsters for 5 days by the p.o. route to select subcurative doses. The subcurative doses selected were 2.5 and 5 mg/kg for mice and 5 and 10 mg/kg for hamsters.
Evaluation of free CpG-ODN and liposomal CpG-ODN alone and in combination with miltefosine.
Groups of mice consisting of five or six animals in two replicates were used for these experiments. Group I received free CpG-ODN (1 nM single dose given i.p.), group II received free CpG-ODN (1 nM single dose given i.p.) plus miltefosine (2.5 mg/kg for 5 days, given p.o.), group III received liposomal CpG-ODN (1 nM single dose given i.p.), group IV received liposomal CpG-ODN (1 nM single dose given i.p.) plus miltefosine (2.5 mg/kg for 5 days, given p.o.), group V received liposomal CpG-ODN (1 nM single dose given i.p.) plus miltefosine (5 mg/kg for 5 days, given p.o.), group VI received miltefosine at 2.5 mg/kg for 5 days (p.o.), group VII received miltefosine at 5 mg/kg for 5 days (p.o.), and group VIII received curative miltefosine (20 mg/kg for 5 days, given p.o.). Group IX received deionized water as a control. The same protocol was adopted for hamsters, where miltefosine was given orally at subcurative doses of 5 and 10 mg/kg and a curative dose of 40 mg/kg for 5 days. The PI was calculated for all drug-treated groups in relation to a nontreated group.
Statistical analysis.
Results are presented as means ± standard deviations (SD) for two experiments, and analysis of data was carried out by Bonferroni's multiple comparison tests and Dunnett's multiple comparison tests. Differences with P values of <0.05 were considered significant. The subcurative dose of miltefosine was determined by probit analysis (9).
RESULTS
Dose optimization of CpG-ODN-2006.
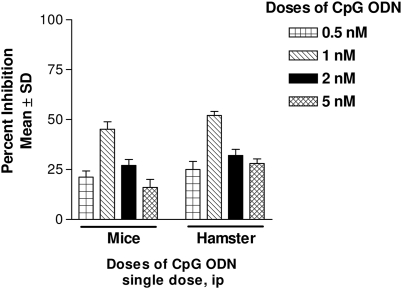
The results of CpG-ODN dose optimization in mice and hamsters are displayed in Fig. 1.
Fig. 1.
Dose optimization of CpG-ODN-2006. Leishmania donovani infection was given intracardially to BALB/c mice (2 × 107 amastigotes/animal) and hamsters (1 × 107 amastigotes/animal). Mice and hamsters were dosed at 7 and 15 days postinfection, respectively, with single shots of various dosages of CpG-ODN by the i.p. route. Mice were sacrificed 3 days after the completion of treatment, whereas hamsters were sacrificed 7 days after the completion of treatment. Mean PI ± SD were calculated by comparing parasitic burdens of treated groups to those of control animals.
(i) Mouse model.
Of the various doses of CpG-ODN-2006 tried, the best antileishmanial efficacy was witnessed with a single dose of 1 nM given i.p. (45.1% inhibition of parasite multiplication). This was followed by gradually decreasing efficacies with 2 and 5 nM doses, namely, 27% and 16% inhibition of parasite multiplication, respectively. A 0.5 nM dose was also tested, but it showed no efficacy (21.2%). Based on the results, a 1 nM single dose given i.p. was selected for the combination trial.
(ii) Hamster model.
The highest antileishmanial efficacy of CpG-ODN-2006 was observed with a single dose of 1 nM given i.p., giving 52% inhibition of parasite multiplication, followed by decreased efficacies at 0.5, 2, and 5 nM doses, namely, 25%, 32%, and 28% inhibition of parasite multiplication, respectively.
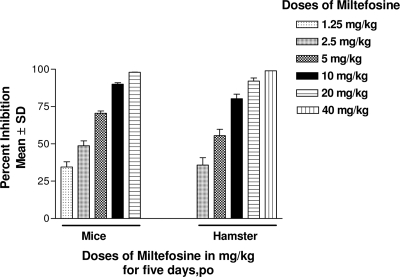
Dose optimization of miltefosine.
The results of miltefosine dose optimization in mice and hamsters are presented in Fig. 2.
Fig. 2.
Dose optimization of miltefosine. Leishmania donovani infection was given intracardially to BALB/c mice (2 × 107 amastigotes/animal) and hamsters (1 × 107 amastigotes/animal). Mice and hamsters were dosed at 7 and 15 days postinfection, respectively, with various dosages of miltefosine by the p.o. route for 5 days. Mice were sacrificed 3 days after the completion of treatment, whereas hamsters were sacrificed 7 days after the completion of treatment. Mean PI ± SD were calculated by comparing parasitic burdens of treated groups to those of control animals.
(i) Mouse model.
Miltefosine was evaluated at various doses, ranging from 1.25 mg/kg to 20 mg/kg, for 5 days by the p.o. route in order to select a subcurative dose. The parasite inhibition observed at 20 mg/kg was 98%, followed by 90%, 70.5%, 48.8%, and 34.5% at doses of 10, 5, 2.5, and 1.25 mg/kg, respectively. The dose selected for the combination trial was 2.5 mg/kg.
(ii) Hamster model.
Miltefosine was tested at various doses, ranging from 2.5 mg/kg to 40 mg/kg, for 5 days by the p.o. route. The parasite inhibition observed at 40 mg/kg was 99.8%, followed by 92%, 80.3%, 55.5%, and 35.8% at doses of 20, 10, 5, and 2.5 mg/kg, respectively. The dose selected for the combination trial was 5 mg/kg, as it was subcurative and nontoxic (18).
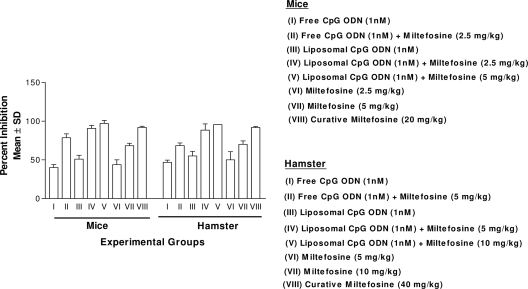
Combination therapy (CpG-ODN plus miltefosine).
The results of combination therapy with free or liposomal CpG-ODN and miltefosine are displayed in Fig. 3.
Fig. 3.
Combination therapy (CpG-ODN-2006 plus miltefosine). Mean PI ± SD were calculated by comparing parasitic burdens of treated groups to those of control animals. Significance among different groups was calculated by Bonferroni's multiple comparison tests. The significance levels for groupwise comparisons of mice were as follows: I versus II, P < 0.001; I versus III, P < 0.01; II versus IV, P < 0.001; V versus II, P < 0.01; and V versus IV, P < 0.001. The significance levels for groupwise comparisons of hamsters were as follows: I versus II, P < 0.001; I versus III, P < 0.01; II versus IV, P < 0.001; V versus II, P < 0.01; and V versus IV, P < 0.001.
(i) Mouse model.
Free CpG-ODN showed an efficacy of 40%, which was moderately enhanced to 51% by liposomal encapsulation (P < 0.01). The percentages of parasite inhibition (PI) of miltefosine at 2.5 mg/kg and 5 mg/kg were 44% and 68.3%, respectively. However, when free CpG-ODN was given with a suboptimal dose (2.5 mg/kg) of miltefosine, parasite inhibition increased from 44% to 72.4% (P < 0.001), and liposomal CpG-ODN further enhanced this efficacy, to 90.6% (P < 0.001). Parasitic inhibition was markedly increased, to 97%, when liposomal CpG-ODN was administered with 5 mg/kg of miltefosine. The efficacy of this combination was comparable with the efficacy of a curative dose (20 mg/kg for 5 days) of miltefosine (98%).
(ii) Hamster model.
Free CpG-ODN showed an efficacy of 46.8%, which was moderately enhanced to 55.1% by liposomal encapsulation (P < 0.01). The PI of miltefosine at 5 mg/kg and 10 mg/kg were 50% and 70.1%, respectively. However, when free CpG-ODN was given with a subcurative dose (5 mg/kg) of miltefosine, parasite inhibition increased from 50% to 68.5% (P < 0.01), and liposomal CpG-ODN further enhanced the efficacy, to 88.5% (P < 0.001). Interestingly, the effectiveness of this combination was further enhanced to 96% when liposomal CpG-ODN was coadministered with 10 mg/kg of miltefosine. This efficacy was close to the curative dose (40 mg/kg for 5 days) of miltefosine (99%).
DISCUSSION
In view of the severe immunosuppression in VL, a rational approach to effectively combat this parasitic scourge would be to enhance the immune status of the host (1, 17). The use of CpG-ODN against leishmaniasis has been reported, especially its use as an immunomodulator or adjuvant with various immunogens (2, 8, 12, 22). In our previous communication, we demonstrated that CpG-ODN-1826 in combination with miltefosine exhibits potential for the treatment of visceral leishmaniasis. Encouraged by these results, we decided to explore the use of another oligonucleotide, CpG-ODN-2006, in combination therapy. Its sequence is 5′-TCG TCG TTT TGT CGT TTT GTC GTT-3′ (21), and it is known for having better efficacy in human models (14). Since natural oligonucleotides are known to be susceptible to nucleases, we used a fully thioated oligonucleotide to enhance metabolic stability. In addition, the CpG oligonucleotide was also used as a liposomal preparation to further enhance its bioavailability. CpG-ODN-2006 was evaluated in combination with subcurative doses (2.5 mg/kg for mice and 5 mg/kg for hamsters, for 5 days) and slightly higher subcurative doses (5 mg/kg and 10 mg/kg for mice and hamsters, respectively, for 5 days) of miltefosine. The results clearly showed that liposomal encapsulation enhanced the antileishmanial efficacy of free CpG-ODN. Coadministration of liposomal CpG-ODN (1 nM) with subcurative doses of miltefosine showed more of an inhibitory effect than administration of either free CpG-ODN or miltefosine alone. The efficacy of this combination was comparable to the efficacy of the curative dose of miltefosine. It was observed that the efficacy profile of CpG-ODN-2006 corroborated with the efficacy of CpG-ODN-1826 observed in a previous study from our lab (18). In order to explore the effect of liposomal lipids on parasitic inhibition, an empty liposome preparation was also administered along with free and liposomal CpG-ODN (1 nM) in a separate experiment. We did not observe any improvement over the untreated infected control groups. This is in agreement with a previous report of Badiee et al. (2). The present work clearly supports the application of liposomal CpG-ODN-2006 in combination with miltefosine as a novel therapeutic approach for safer treatment of VL.
ACKNOWLEDGMENTS
We thank T. K. Chakraborty, CDRI, Lucknow, India, for his encouragement and for the use of facilities.
This is CDRI communication no. 8066.
Footnotes
Published ahead of print on 2 May 2011.
REFERENCES
- 1. Badaró R., et al. 1996. rK39: a cloned antigen of Leishmania chagasi that predicts active visceral leishmaniasis. J. Infect. Dis. 173:758–761 [DOI] [PubMed] [Google Scholar]
- 2. Badiee A., Jaafari M. R., Samiei A., Soroush D., Khamesipour A. 2008. Coencapsulation of CpG oligodeoxynucleotides with recombinant Leishmania major stress-inducible protein 1 in liposome enhances immune response and protection against leishmaniasis in immunized BALB/c mice. Clin. Vaccine Immunol. 15:668–674 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Bern C., et al. 2006. Liposomal amphotericin B for the treatment of visceral leishmaniasis. Clin. Infect. Dis. 43:917–924 [DOI] [PubMed] [Google Scholar]
- 4.Reference deleted.
- 5. Bogdon C. 2008. Mechanisms and consequences of persistence of intracellular pathogens: leishmaniasis as an example. Cell. Microbiol. 10:1221–1234 [DOI] [PubMed] [Google Scholar]
- 6. Bryceson A. 2001. A policy for leishmaniasis with respect to the prevention and control of drug resistance. Trop. Med. Int. Health 6:928–934 [DOI] [PubMed] [Google Scholar]
- 7. Croft S., Neal R., Pendergast W., Chan J. 1987. The activity of alkyl phosphocholines and related derivates against Leishmania donovani. Biochem. Pharmacol. 36:2633–2636 [DOI] [PubMed] [Google Scholar]
- 8. Datta N., Mukherjee S., Das L., Das P. K. 2003. Targeting of immunostimulatory DNA cure experimental visceral leishmaniasis through nitric oxide up-regulation and T cell activation. Eur. J. Immunol. 33:1508–1518 [DOI] [PubMed] [Google Scholar]
- 9. Finney D. J. 1971. Probit analysis, 3rd ed. Cambridge University Press, Cambridge, United Kingdom [Google Scholar]
- 10. Gupta S., Ramesh S. C., Shrivastava V. M. L. 2005. Efficacy of picroliv combination with miltefosine, an orally effective antileishmanial drug against experimental visceral leishmaniasis. Acta Trop. 94:41–47 [DOI] [PubMed] [Google Scholar]
- 11. Hartmann G., Noll B., Shen W., Schetter C., Krieg A. M. 2000. Mechanism and function of a newly identified CpG DNA motif in human primary B cells. J. Immunol. 164:944–953 [DOI] [PubMed] [Google Scholar]
- 12. Iborra S., et al. 2008. Vaccination with the Leishmania major ribosomal proteins plus CpG oligodeoxynucleotides induces protection against experimental cutaneous leishmaniasis in mice. Microbes Infect. 10:1133–1141 [DOI] [PubMed] [Google Scholar]
- 13. Jaafari M. R., et al. 2007. The role of CpG ODN in enhancement of immune response and protection in BALB/c mice immunized with recombinant major surface glycoprotein of Leishmania (rgp63) encapsulated in cationic liposome. Vaccine 25:6107–6117 [DOI] [PubMed] [Google Scholar]
- 14. Jahrsdörfer B., Weiner G. J. 2008. CpG oligodeoxynucleotides as immunotherapy in cancer. Update Cancer Ther. 3:27–32 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Kirby C., Gregoriadis G. 1984. Dehydration-rehydration vesicles: a simple method for high yield drug entrapment in liposomes. Biotechnology 2:979–984 [Google Scholar]
- 16. Krieg A. M., et al. 1995. CpG motifs in bacterial DNA trigger direct B-cell activation. Nature 374:546–549 [DOI] [PubMed] [Google Scholar]
- 17. Murray H. W. 1988. Survival of intracellular pathogens within human mononuclear phagocytes. Semin. Hematol. 25:101–111 [PubMed] [Google Scholar]
- 18. Sane S. A., Shakya N., Haq W., Gupta S. 2010. CpG oligodeoxynucleotide augments the antileishmanial activity of miltefosine against experimental visceral leishmaniasis. J. Antimicrob. Chemother. 65:1448–1454 [DOI] [PubMed] [Google Scholar]
- 19. Sundar S., Chatterjee M. 2006. Visceral leishmaniasis—current therapeutic modalities. Indian J. Med. Res. 123:345–352 [PubMed] [Google Scholar]
- 20. Sundar S. 2001. Drug resistance in Indian visceral leishmaniasis. Trop. Med. Int. Health 6:849–854 [DOI] [PubMed] [Google Scholar]
- 21. Utaisincharoen P., Anuntagool N., Chaisuriya P., Pichyangkul S., Sirisinha S. 2002. CpG ODN activates NO and iNOS production in mouse macrophage cell line RAW 264.7. Clin. Exp. Immunol. 128:467–473 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Verthelyi D., Klinman D. M. 2003. Immunoregulatory activity of CpG oligonucleotides in humans and nonhuman primates. Clin. Immunol. 109:64–71 [DOI] [PubMed] [Google Scholar]
- 23. Weeratna R. D., Makinen S. R., McCluskie M. J., Davis H. L. 2005. TLR agonists as vaccine adjuvants: comparison of CpG ODN and Resiquimod (R-848). Vaccine 23:5263–5270 [DOI] [PubMed] [Google Scholar]
- 24. Zimmermann S., et al. 1998. Cutting edge: CpG oligodeoxynucleotides trigger protective and curative Th1 responses in lethal murine leishmaniasis. J. Immunol. 160:3627–3630 [PubMed] [Google Scholar]