Abstract
Purpose
To report long-term results in a case series of patients treated with systemic immune suppression for prevention of penetrating keratoplasty (PKP) graft rejection.
Design
Retrospective noncomparative chart review.
Participants
Three patients presented with PKP graft failure.
Methods
Patients received oral prednisone, azathioprine and cyclosporine to prevent rejection of repeat corneal transplant. Patients received repeat PKP and graft outcome was reported.
Main outcome measures
Visual acuity and graft survival were recorded.
Results
Mean age was 55 years, two male and one female. Mean follow-up period was 37 months (range 24-46). All three patients completed the treatment protocol with minimal adverse effects. All grafts remained clear over observational period.
Conclusion
Our study suggests that systemic immune suppression with 2 or more agents may be helpful to prevent corneal graft rejection in high-risk patients.
Keywords: Corneal transplant, Penetrating keratoplasty, Graft rejection, Systemic immunosuppression
One of the most prevalent tissue transplant procedures performed worldwide, penetrating keratoplasty (PKP) has an unsatisfactory long-term success rate, especially in high-risk recipients [1,2]. Irreversible allograft rejection causes 16-30% of all corneal graft failures.3-4 The prognosis decreases substantially with the number of previous grafts, and the survival rates for third and fourth regrafts are only 25% and 0%, respectively [5,6].
Management of immunogenic allograft rejection typically involves topical corticosteroids for epithelial rejection and systemic corticosteroids for endothelial rejection [7]. Systemic immunosuppression regimens are more commonly reserved for high-risk grafts [8]. Systemic calcineurin inhibitors, antimetabolites, or monoclonal antibodies, have been employed, either as monotherapy or in combination with corticosteroid, with variable success [9-14]. However, these systemic agents have significant adverse effect profile. To decrease side effects, we hypothesized that a tailored combination of systemic prednisone, azathioprine, and cyclosporine A (CsA) may be an effective regimen in high-risk regraft. The goal of multi-agent therapy is to leverage their synergistic mechanisms to achieve maximal therapeutic effects and minimal adverse effects [15].
Although the use of multi-agent therapy noninfectious uveitis has been documented [16], there exists a scarcity of literature on the utility of such regimen for keratoplasty rejection. Here, we described the visual outcome and graft survival of high-risk corneal transplant patients who received systemic immunosuppression prior to re-graft.
Institutional review board/ethics committee approvals were obtained before the study commenced. The patients are included if they present with more than one identifiable risk factors. (1) Failure to meet inclusion criteria, (2) refusal of the triple therapy, (3) lack of mental capacity to understand risks and benefits, or (4) noticeable poor compliance defines the exclusion criteria. All PKs were performed by one surgeon (S.C. Yiu) at the Doheny Eye Institute. All patients were scheduled for follow-up on postoperative day (POD) 1, postoperative week (POW) 1, and POW 3, and monthly thereafter. The systemic immunosuppressive protocols were specifically tailored to each patient depending on their comorbidities by a rheumatologist (S. Shinada). Patients were seen and examined; basic studies, such as CBC, chemistry, liver function panel, erythrocyte sedimentation rate, c-reactive protein, and urinalysis, were vigilantly monitored on a regular basis by S. Shinada.
Case Reports
Case 1
This 45-year-old male, with a history of right corneal injury and opacification since childhood, bullous keratopathy, keratoconjunctivitis sicca, and recurrent iritis, previously underwent PKP of his right eye at an outside facility. On post-operative year two, he presented to us with a culture positive Streptococcus pneumoniae corneal ulcer. Graft failure with corneal edema and neovascularization followed despite an aggressive course of topical fortified antibiotics and prednisolone acetate 1% (Pred Forte®, Allergan, Irvine, California). Subsequently, the patient failed two more PKP despite topical regimen of gatifloxacin ophthalmic 0.3% (Zymar®, Allergan, Irvine, California) 1 gtt qid, rimexolone 1 gtt q2h, and CsA 1% 1 gtt qid.
Given that the patient demonstrated all four high-risk characteristics [17] and has a reported success rate of 0% for the fourth corneal transplant [6], the multi-agent systemic immunosuppression therapy was offered to the patient and his family. On POD 1, prednisolone acetate 1% 1 gtt q2h and systemic prednisone were initiated. Oral prednisone taper schedule was as followed: 40 mg daily for 7 days, then stepped down by 10 mg each week until completion. At POW 1, oral azathioprine 50 mg daily was implemented for 7 days then increased to 50 mg bid. At POM 1, we started oral CsA 50 mg bid.
At POM 3, the patient reported no constitutional symptoms, and laboratory studies were in normal range. At POM 11, no signs of graft rejection were observed and his VA improved to 20/30-1. The patient reported doing well on cyclosporine A 50 mg po bid, azathioprine 50 mg po bid, and prednisolone acetate 0.125% 1 gtt (Econopred®, Alcon Laboratories, United Kingdom) qid. We tapered CsA by 25 mg each week and discontinue it at POM 12, while azathioprine is maintained at 100 mg daily. Azathioprine was tapered to 50 mg daily on POM 15 and discontinued on POM 17. The extended course was necessary in this particular four-time PK case. On most recent evaluation at POM 41, graft status was viable and VA 20/50+1 on prednisolone acetate 0.125% 1 gtt BID.
Case 2
A 55-year-old female with a history of Axenfeld-Rieger syndrome, 28-year history of glaucoma, recurrent iritis, and multiple prior ocular surgeries, such as Baerveldt tube placement and revision, cataract extraction and intraocular lens placement, who received a PKP for pseudophakic bullous keratopathy. The graft was rejected while patient was on topical management with prednisolone acetate 1 gtt qid and CsA 2% 1 gtt q4h.
In preparation for the repeat PK, we instituted azathioprine step-up at (50 mg po qd for 1 week then 50 mg po bid) and CsA step-up after one month of azathioprine (50 mg po qd for 1 week then 50 mg po bid), two months prior to transplant procedure. After the PKP procedure, oral prednisone was started at 40 mg for 1 week then tapered by 10 mg per week until completion. Laboratory studies at POM 1 were within normal range, except for an asymptomatic pyuria (10-14 WBC). At POM 4, urinalysis showed 3+ blood and many RBCs on microscopic examination, with normal chem-20 and CBC panels. At this time, her prednisone protocol had completed. CsA was discontinued and the patient remained on only azathioprine 50 mg bid. The hematuria subsequently resolved. At POM 9, early signs of graft rejection became evident and the patient was urgently started on prednisolone acetate q3h, CsA q4h, and oral prednisone, in combination with azathioprine. Systemic CsA was not started due to the history of microscopic hematuria. The oral prednisone course was 60 mg daily for 2 weeks, 40 mg daily for 2 weeks, 20 mg daily for 2 weeks, 10 mg daily for 2 weeks, 5 mg daily for 2 weeks, and then discontinue. Topical prednisolone acetate 1% and CsA 1% were subsequently increased to 8 times per day and 6 times per day, respectively. The patient tolerated the regimen well.
At POM 14, resolution of graft rejection was seen and azathioprine was tapered over a period of 3 months, as followed: 100 mg alternating with 50 mg every other day for 1 month, then 50 mg daily for 1 month, and then 25 mg daily for 1 month. At POM 17, complete resolution of graft rejection was achieved and the systemic immunosuppression regimen concluded. The patient continued to be on topical CsA 1% 1 gtt qid and prednisolone acetate 1% 1 gtt 6 times per day. Graft remained viable at POM 27, and topical CsA was tapered to 1 gtt bid and prednisolone acetate to 1 gtt bid. At POM 46, no rejection was observed.
Case 3
This 60-year-old male patient has an ocular history of trabeculectomy and aqueous shunt placement for advanced glaucoma, cataract extraction and intraocular lens placement, and corneal transplant for pseudophakic bullous keratopathy in the same eye. The graft subsequently failed.
In preparation for a repeat PKP, we conducted a preoperative discussion with the patient about the risks and benefits of the multi-agent immunosuppression therapy. Considering his medical history of poorly controlled hypertension (170s/100s), cyclosporine was deferred as it is known to cause elevation of blood pressure. Oral prednisone and azathioprine were started first, on POD 1. The oral prednisone tapering schedule was as followed: 60 mg daily for one week, 50 mg daily for one week, then 40 mg daily for one week, then 20 mg daily for one week, then 10 mg daily for one week then 5 mg daily for one week then discontinue. Azathioprine started at 50 mg daily for 7 days, then 50 mg BID. At POM 2, our patient had elevated renal function (BUN 47 and creatinine 1.6) but other laboratory studies were normal and he tolerated the regimen well. His vision only improved to counting fingers at six feet from preoperative vision of HM, secondary to advanced glaucoma.
Case 3 has a comorbidity of JNC-7 stage 1 hypertension, requiring vigilant surveillance of his blood pressures. CsA has been demonstrated to exacerbate hypertension with rapid development of end-organ decompensation and damage via multiple mechanisms such as nephrotoxicity and inhibition of endothelial NO activity [17-22]. No exacerbation of blood pressures was observed during the follow-up period.
Figure 1 illustrates the effectiveness of the multi-agent immunosuppression therapy in management of graft failure. Photo (A) shows complete graft failure with microcystic edema, endothelial failure, conjunctival hyperemia, and some corneal vascularization. Photo (B) demonstrates clear graft after a complete course of systemic suppression therapy.
Figure 1.
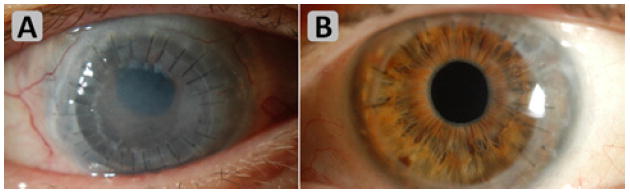
Regraft status after 9 months of immunosuppressive protocol. Complete graft failure is shown in (A), demonstrating microcystic edema, endothelial failure, conjunctival hyperemia, and some corneal vascularization. (B) demonstrates clear graft after a complete course of triple therapy.
Conclusion
Over an average follow-up period of 37 months (range: 24–46 months), the multi-agent systemic immunosuppression therapy demonstrated success in both preventing and reversing graft failure in high-risk recipients. Our patients have complicated medical and surgical histories with multiple risk factors for graft rejection. Risk factors for allograft rejection include degree of vascularization, preoperative inflammation of the anterior segment, previous ocular surgical interventions, pseudophakia, anterior or posterior synechiae, large graft size, presence of limbus in graft proximity, loosening of sutures, and corneal vascularization [1,5-6,23,24]. Any factors that may emancipate soluble antigens may predispose the patients to immunologic graft rejection.
Some mild side effects were observed. The majority of these resolved satisfactorily. No graft failure was observed during this follow-up period, compared to one study that reported almost 13% of the regrafts failed within 6 months [25,26] after surgery.
Previously, some success [6,9,12] in extending graft longevity and reversing graft rejection with the combined intravenous pulse methylprednisolone, topical prednisolone, and oral CsA regime has been reported. However, systemic CsA monotherapy has very limited benefit in repeated corneal transplantation in a highly vascularized bed [13]. A case control study [27] reported no statistically significant benefit of systemic CsA over conventional therapy in terms of managing graft failure, whereas many side effects of systemic CsA were observed.
Other immunosuppressive agents may also be employed in high-risk transplants. One prospective randomized clinical trial [11] suggests comparable efficacies between mycophenolate mofetil (MMF) and CsA for treatment of high-risk patients. Another study showed some success of an 18-24 month course systemic tacrolimus therapy after high-risk corneal transplantation [14]. In murine model, combination therapy of CsA and MMF appeared to be superior than monotherapy [28], without higher incidence of adverse effects. Topical tacrolimus had been shown to be effective in prevention of corneal allograft rejection in a murine corneal graft rejection model [29]. Utility of these agents in toxicity-sparing protocols for organ transplant recipients had been proposed [29,30].
Evidently, a cautious approach to aggressive immunosuppressant therapies for corneal immunologic rejection is rational. Any particular immunosuppressant can precipitate a range of serious adverse effects. A case of sudden death following high-dose intravenous methylprednisolone has been reported [31]. CsA side effects have been reported in corneal graft rejection patients [6,26]. A rare association between azathioprine-induced aplastic anemia and bilateral macular hemorrhage in one corneal graft recipient has been reported [32]. MMF may produce gastrointestinal distress, hepatotoxicity, aplastic anemia, lymphoma and skin carcinoma.
Evidently, the weakness of this investigation is limited number participants. Further studies with this and other multi-agent combinations, especially the toxicity sparing regimens, are enthusiastically encouraged. Preoperative systemic management may be necessary in case of prior multiple regrafts. However, given the realistic adverse effect profile and the fact that 12 or more months [13,33] of systemic immunosuppression may be required, we advocate a systematic and comprehensive discussion with the patients before initiation of the triple therapy. A multidisciplinary patient-centric approach is advantageous for optimization of the treatment protocol to each individual patient.
Acknowledgments
Financial Support: NEI Core Grant EY03040
Unrestricted grant from Research to Prevent Blindness.
Footnotes
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
References
- 1.Inoue K, Amano S, Oshika T, Tsuru T. Risk factors for corneal graft failure and rejection in penetrating keratoplasty. Acta Ophthalmol Scand. 2001;79:251–255. doi: 10.1034/j.1600-0420.2001.790308.x. [DOI] [PubMed] [Google Scholar]
- 2.Waldock A, Cook SD. Corneal transplantation: how successful are we? Br J Ophthalmol. 2000;84:813–815. doi: 10.1136/bjo.84.8.813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Williams KA, Esterman AJ, Bartlett C, Holland H, Hornsby NB, et al. How effective is penetrating corneal transplantation? Factors influencing long-term outcome in multivariate analysis. Transplantation. 2006;81:836–901. doi: 10.1097/01.tp.0000185197.37824.35. [DOI] [PubMed] [Google Scholar]
- 4.The Australian Corneal Graft Registry, 1990-1992 REPORT. Aust NZ J Ophthalmol. 1993;21:1–48. [PubMed] [Google Scholar]
- 5.Vail A, Gore SM, Bradley BA, Easty DL, Rogers CA, et al. Conclusions of the corneal transplant follow up study. Collaborating Surgeons. Br J Ophthalmol. 1997;81:631–636. doi: 10.1136/bjo.81.8.631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bertelmann E, Jaroszewski J, Pleyer U. Corneal allograft rejection: current understanding. 2. Clinical implications. Opthalmologica. 2002;216:2–12. doi: 10.1159/000048289. [DOI] [PubMed] [Google Scholar]
- 7.Barker NH, Henderson TR, Ross CA, Coster DJ, Williams KA, et al. Current Australian practice in the prevention and management of corneal allograft rejection. Clin Experiment Ophthalmol. 2000;28:357–360. doi: 10.1046/j.1442-9071.2000.00335.x. [DOI] [PubMed] [Google Scholar]
- 8.Randleman JB, Stulting RD. Prevention and treatment of corneal graft rejection: current practice patterns (2004) Cornea. 2006;25:286–290. doi: 10.1097/01.ico.0000178731.42187.46. [DOI] [PubMed] [Google Scholar]
- 9.Young AL, Rao SK, Cheng LL, Wong AK, Leung AT, et al. Combined intravenous pulse methylprednisolone and oral cyclosporine A in the treatment of corneal graft rejection: 5-year experience. Eye. 2002;16:304–308. doi: 10.1038/sj.eye.6700144. [DOI] [PubMed] [Google Scholar]
- 10.Inoue K, Kimura C, Amano S, Sato T, Fujita N, et al. Long term outcome of systemic cyclosporine treatment following penetrating keratoplasty. Jpn J Ophthalmol. 2001;45:378–383. doi: 10.1016/s0021-5155(01)00339-2. [DOI] [PubMed] [Google Scholar]
- 11.Reis A, Reinhard T, Voiculescu A, Kutkuhn B, Godehardt E, et al. Mycophenolate mofetil versus cyclosporin A in high risk keratoplasty patients: a prospectively randomised clinical trial. Br J Ophthalmol. 1999;83:1268–1271. doi: 10.1136/bjo.83.11.1268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lam DS, Wong AK, Tham CC, Leung AT. The use of combined intravenous pulse methylprednisolone and oral cyclosporine A in the treatment of corneal graft rejection: a preliminary study. Eye. 1998;12:615–618. doi: 10.1038/eye.1998.154. [DOI] [PubMed] [Google Scholar]
- 13.Rumelt S, Bersudsky V, Blum-Hareuveni T, Rehany U. Systemic cyclosporin A in high failure risk, repeated corneal transplantation. Br J Ophthalmol. 2002;86:988–992. doi: 10.1136/bjo.86.9.988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Joseph A, Raj D, Shanmuganathan V, Powell RJ, Dua HS. Tacrolimus immunosuppression in high-risk corneal grafts. Br J Ophthalmol. 2007;91:51–55. doi: 10.1136/bjo.2006.097428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nguyen P, Yiu SC. Multi-agent pharmaceutical therapy for modulation of corneal allograft immunologic rejection. Curr Insights Online publication 2007 [Google Scholar]
- 16.Lustig MJ, Cunningham ET., Jr Use of immunosuppressive agents in uveitis. Curr Opin Ophthalmol. 2003;14:399–412. doi: 10.1097/00055735-200312000-00014. [DOI] [PubMed] [Google Scholar]
- 17.Poon AC, Forbes JE, Dart JK, Subramaniam S, Bunce C, et al. Systemic cyclosporin A in high risk penetrating keratoplasties: a case-control study. Br J Ophthalmol. 2001;85:1464–1469. doi: 10.1136/bjo.85.12.1464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, et al. The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: the JNC 7 report. JAMA. 2003;289:2560–2572. doi: 10.1001/jama.289.19.2560. [DOI] [PubMed] [Google Scholar]
- 19.Taler SJ, Textor SC, Canzanello VJ, Schwartz L. Cyclosporin-induced hypertension: incidence, pathogenesis and management. Drug Safety. 1990;20:437–449. doi: 10.2165/00002018-199920050-00004. [DOI] [PubMed] [Google Scholar]
- 20.Kaye D, Thompson J, Jennings G, Esler M. Cyclosporine therapy after cardiac transplantation causes hypertension and renal vasoconstriction without sympathetic activation. Circulation. 1993;88:1101–1109. doi: 10.1161/01.cir.88.3.1101. [DOI] [PubMed] [Google Scholar]
- 21.Morales JM. Influence of the new immunosuppressive combinations on arterial hypertension after renal transplantation. Kidney Int Suppl. 2002:S81–S87. doi: 10.1046/j.1523-1755.62.s82.16.x. [DOI] [PubMed] [Google Scholar]
- 22.Oriji GK, Keiser HR. Role of nitric oxide in cyclosporine a–induced hypertension. Hypertension. 1998;32:849–855. doi: 10.1161/01.hyp.32.5.849. [DOI] [PubMed] [Google Scholar]
- 23.Pleyer U, Dannowski H, Volk HD, Ritter T. Corneal allograft rejection: current understanding. 1. Immunobiology and basic mechanisms. Opthalmologica. 2001;215:254–262. doi: 10.1159/000050870. [DOI] [PubMed] [Google Scholar]
- 24.Jonas JB, Rank RM, Budde WM. Immunologic graft reactions after allogenic penetrating keratoplasty. Am J Ophthalmol. 2002;113:437–443. doi: 10.1016/s0002-9394(01)01426-x. [DOI] [PubMed] [Google Scholar]
- 25.Patel NP, Kim T, Rapuano CJ, Cohen EJ, Laibson PR. Indications for and outcomes of repeat penetrating keratoplasty, 1989-1995. Ophthalmology. 2000;107:719–724. doi: 10.1016/s0161-6420(00)00003-8. [DOI] [PubMed] [Google Scholar]
- 26.Rapuano CJ, Cohen EJ, Brady SE, Arentsen JJ, Laibson PR. Indications for and outcomes of repeat penetrating keratoplasty. Am J Ophthalmol. 1990;109:689–695. doi: 10.1016/s0002-9394(14)72437-7. [DOI] [PubMed] [Google Scholar]
- 27.Reis A, Reinhard T, Sundmacher R, Braunstein C, Godehardt E. Effect of mycophenolate mofetil, cyclosporin A, and both in combination in a murine corneal graft rejection model. Br J Ophthalmol. 1998;82:700–703. doi: 10.1136/bjo.82.6.700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hikita N, Lopez JS, Chan CC, Mochizuki M, Nussenblatt RB, et al. Use of topical FK506 in a corneal graft rejection model in Lewis rats. Invest Ophthalmol Vis Sci. 1997;38:901–909. [PubMed] [Google Scholar]
- 29.Land W, Vincenti F. Toxicity-sparing protocols using mycophenolate mofetil in renal transplantation. Transplantation. 2005;80:S221–S223. doi: 10.1097/01.tp.0000186386.13597.cb. [DOI] [PubMed] [Google Scholar]
- 30.Vincenti F, Brennan TV, Fuller TF, Feng S. Optimizing the use of cyclosporine (neoral) for recipients of living donor kidneys. Transplant Proc. 2004;36:50S–53S. doi: 10.1016/j.transproceed.2004.01.050. [DOI] [PubMed] [Google Scholar]
- 31.Thompson JF, Chalmers DH, Wood RF, Kirkham SR, Morris PJ. Sudden death following high-dose intravenous methylprednisolone. Transplantation. 1983;36:594–596. [PubMed] [Google Scholar]
- 32.Sudhir RR, Rao SK, Shanmugam MP, Padmanabhan P. Bilateral macular hemorrhage caused by azathioprine-induced aplastic anemia in a corneal graft recipient. Cornea. 2002;21:712–714. doi: 10.1097/00003226-200210000-00016. [DOI] [PubMed] [Google Scholar]
- 33.Hill JC. Systemic cyclosporine in high-risk keratoplasty. Short- versus long-term therapy. Ophthalmology. 1994;101:128–133. doi: 10.1016/s0161-6420(13)31253-6. [DOI] [PubMed] [Google Scholar]


