Abstract
Gamma irradiation is a nonthermal processing technology that has been used for the preservation of a variety of food products. This technology has been shown to effectively inactivate bacterial pathogens. Currently, the FDA has approved doses of up to 4.0 kGy to control food-borne pathogens in fresh iceberg lettuce and spinach. However, whether this dose range effectively inactivates food-borne viruses is less understood. We have performed a systematic study on the inactivation of a human norovirus surrogate (murine norovirus 1 [MNV-1]), human norovirus virus-like particles (VLPs), and vesicular stomatitis virus (VSV) by gamma irradiation. We demonstrated that MNV-1 and human norovirus VLPs were resistant to gamma irradiation. For MNV-1, only a 1.7- to 2.4-log virus reduction in fresh produce at the dose of 5.6 kGy was observed. However, VSV was more susceptible to gamma irradiation, and a 3.3-log virus reduction at a dose of 5.6 kGy in Dulbecco's modified Eagle medium (DMEM) was achieved. We further demonstrated that gamma irradiation disrupted virion structure and degraded viral proteins and genomic RNA, which resulted in virus inactivation. Using human norovirus VLPs as a model, we provide the first evidence that the capsid of human norovirus has stability similar to that of MNV-1 after exposure to gamma irradiation. Overall, our results suggest that viruses are much more resistant to irradiation than bacterial pathogens. Although gamma irradiation used to eliminate the virus contaminants in fresh produce by the FDA-approved irradiation dose limits seems impractical, this technology may be practical to inactivate viruses for other purposes, such as sterilization of medical equipment.
INTRODUCTION
Human norovirus is a major food and waterborne pathogen that causes acute gastroenteritis. The virus is transmitted primarily through the fecal-oral route, either by direct contact with fecal matter or indirect contact through contaminated food or water supplies. It is highly stable and contagious, requiring less than 10 virus particles to cause an infection (20, 35, 44, 51). Recent human volunteer study and mathematical modeling showed that the average probability of infection for a single norovirus particle is close to 0.5 (56). Norovirus outbreaks occur worldwide and have a significant impact on food safety and public health. The National Institute of Allergy and Infectious Diseases (NIAID) categorized human norovirus as a category B priority biodefense pathogen. Food-associated outbreaks occur in diverse locales, including restaurants, nursing homes, schools, cruise ships, and personal residences (1, 15, 21, 25, 31, 43). In fact, it is estimated that over 90% of nonbacterial gastroenteritis is caused by norovirus infections (21, 35, 43). This may even be an underestimation due to the large number of unreported or asymptomatic infections (21, 35). The virus has a significant economic and emotional impact on society; however, research on this biodefense agent has been impeded because it cannot be cultivated in vitro (19).
Foods at high risk of norovirus contamination include fresh produce. Norovirus contamination can occur during preharvest and postharvest stages, such as through contaminated irrigation water, septic tank runoff, and mishandling by farm workers and food handlers (2, 8, 24, 26, 39, 53). Additionally, fresh produce undergoes minimal to no processing, increasing the viral infection risk (8, 18, 26, 54). In fact, norovirus accounts for more than 40% of fresh produce-associated outbreaks (18). Berries shipped across international borders have also been known to cause outbreaks because the virus can remain stable on frozen tissue (10, 11, 48). Currently, chlorine-based sanitizers are widely used in the fresh produce industry as the primary decontamination method for pathogen removal (3, 18). However, it has been demonstrated that these sanitizers are not effective in removing human norovirus surrogates and hepatitis A virus from fresh produce (3, 53). Chlorine solution rinses can achieve only a maximum 1- to 1.5-log virus reduction in contaminated fresh produce (3, 53). More importantly, recent evidence suggests noroviruses can become internalized in fresh produce (50, 61, 63). Chemical sanitizers may reduce the viruses at the surface level of produce, but not those that have been internalized.
One potential technology for decontaminating the pathogens in fresh produce is irradiation technology (22, 59). High-frequency electromagnetic waves (gamma rays) are released during isotopic negative beta decay. The decay involves the conversion of a neutron into a proton, which releases an electron and an antineutrino; gamma rays are released during this process (17, 23). Gamma ray frequencies are in the magnitude of 1019 Hz; these high-energy electromagnetic waves may hold potential in disrupting viral pathogens (17, 23). The high-energy waves can penetrate into food products and, thus, can potentially inactivate internalized pathogens. Currently, the FDA has approved doses of up to 4.0 kGy to control food-borne pathogens in fresh iceberg lettuce and spinach (62). It has been shown that gamma irradiation effectively inactivated Escherichia coli O157:H7, Listeria monocytogenes, and Salmonella in food products, including fresh produce, at this approved dose (5, 17, 34, 45, 59).
However, the feasibility of using gamma irradiation to eliminate human norovirus, the major causative agent of food-borne illnesses, is not understood. This is due to the fact that human norovirus lacks a cell culture system (19). Thus, survival studies of human norovirus must rely on suitable surrogates such as feline calicivirus (FCV) (16, 60), canine calicivirus (CaCV) (16), and murine norovirus 1 (MNV-1) (4, 13). It was reported that a 3-log virus reduction was observed in low-protein-content solution at 0.5 kGy for FCV and 0.3 kGy for CaCV by gamma irradiation (16). It seems that these two surrogates were quite susceptible to gamma irradiation, or at least in low-protein-content solutions. However, no survival trend data have been reported for any human norovirus surrogates in food samples, including fresh produce. More importantly, recent studies found that MNV-1 is a better surrogate for human norovirus due to their biochemical, genetic, and morphological similarities (13). For example, the size (35 to 38 nm in diameter), shape (icosahedral, small rounded structure), viral capsid, and RNA genome of MNV-1 are all analogous to those of human norovirus (32, 64). Furthermore, MNV-1 is highly resistant to acidic environments and is more stable than FCV (13). To date, no study has investigated the susceptibility of MNV-1 to gamma irradiation. Moreover, the mechanism of viral inactivation by irradiation is poorly understood. Noroviruses and other caliciviruses are nonenveloped viruses, and the outer shell of these viruses possesses a highly stable capsid that protects their genetic material, the single-stranded positive-sense RNA. The effect of gamma irradiation on viral capsid and genomic RNA has not been determined.
In this study, we performed a systematic study on the inactivation of MNV-1 and human norovirus virus-like particles (VLPs) by gamma irradiation. We also selected vesicular stomatitis virus (VSV), an enveloped virus, as a comparison since the virion structure of VSV is significantly different than that of norovirus. Furthermore, we determined the mechanism of viral inactivation by gamma irradiation. We investigated the effect of gamma irradiation on viral proteins, genomic RNA, capsid integrity, and virus particles. In addition, we provide the first evidence that the capsid proteins of human norovirus VLPs and MNV-1 have similar stability after exposure to gamma irradiation.
MATERIALS AND METHODS
Viruses and cell culture.
Murine norovirus strain MNV-1 was generously provided by Herbert W. Virgin IV, Washington University School of Medicine (32). Vesicular stomatitis virus (VSV) Indiana strain was a generous gift from Sean Whelan at Harvard Medical School (36, 37). MNV-1 and VSV were propagated in confluent monolayers of murine macrophage cell line RAW 264.7 (ATCC, Manassas, VA) and baby hamster kidney cells (BHK-21 cells), respectively. Both RAW 264.7 and BHK-21 cells were cultured in high-glucose Dulbecco's modified Eagle medium (DMEM) (Invitrogen, Carlsbad, CA) supplemented with 10% fetal bovine serum (FBS) (Invitrogen) at 37°C under a 5% CO2 atmosphere. For growing MNV-1 stock, confluent RAW 264.7 cells were infected with MNV-1 at a multiplicity of infection (MOI) of 0.01. After 1 h of incubation at 37°C, 15 ml of DMEM supplemented with 2% FBS was added. The virus was harvested after 2 days postinoculation by three freeze-thaw cycles and low-speed centrifugation at 5,000 × g for 30 min. Preparation of VSV stock was performed as described previously. Briefly, confluent BHK-21 cells were infected with VSV at an MOI of 3. After 1 h of incubation at 37°C, 15 ml of DMEM supplemented with 2% FBS was added. Virus was harvested after 18 h postinoculation by low-speed centrifugation at 5,000 × g for 10 min.
Sample preparation and gamma irradiation.
Fresh produce (strawberries, romaine lettuce, and spinach) was purchased from a local supermarket. One milliliter of MNV-1 virus stock (108 PFU/ml) was inoculated into 10 g of each produce to achieve an inoculation level of 107 PFU/g. Samples were prepared in triplicates. The samples were heat sealed in plastic compartments using an AIE-200 impulse sealer (American International Electric, Whittier, CA) and mixed thoroughly with a shaker for 30 min. All samples were stored in an icebox and delivered to the Nuclear Reactor Laboratory (NRL) at The Ohio State University. Irradiation treatment took place in a 6-in diameter cylinder in the NRL Cobalt-60 irradiator. Samples were irradiated at doses of 0.175 kGy, 0.35 kGy, 0.525 kGy, 0.7 kGy, 2.8 kGy, 5.6 kGy, 11.2 kGy, 16.8 kGy, and 22.4 kGy. After treatment, 5 ml of phosphate-buffered saline (PBS; 137 mM NaCl, 10 mM Na2HPO4, 2 mM NaH2PO4; pH 7.4) was added to each bag, and the samples were stomached for 2 min. Virus was eluted by low-speed centrifugation at 5,000 × g for 10 min. The sensitivities of VSV and MNV-1 to irradiation in DMEM were also tested. One milliliter each of the MNV-1 (108 PFU/ml) and VSV (109 PFU/ml) stocks was inoculated into 10 ml of DMEM to achieve inoculation levels of 107 PFU/ml and 108 PFU/ml, respectively. To study the mechanism of viral inactivation, 20 μg of highly purified MNV-1 and VSV (1 μg/μl) was prepared for gamma irradiation. All samples were heat sealed in plastic compartments and irradiated for 2.8 kGy, 5.6 kGy, 11.2 kGy, 16.8 kGy, and 22.4 kGy. The viral survivors were determined by plaque assay.
MNV-1 and VSV plaque assays.
MNV-1 and VSV plaque assays were performed in RAW 264.7 and Vero cells, respectively. Briefly, cells were seeded into six-well plates (Corning Life Sciences, Wilkes-Barre, PA) at a density of 2 × 106 cells per well. After 24 h of incubation, RAW 264.7 and Vero cell monolayers were infected with 400 μl of a 10-fold dilution series of MNV-1 and VSV, respectively, and the plates were incubated for 1 h at 37°C, with agitation every 10 min. The cells were overlaid with 3 ml of Eagle minimum essential medium (MEM) containing 1% agarose, 2% FBS, 1% sodium bicarbonate, 0.1 mg of kanamycin/ml, 0.05 mg of gentamicin/ml, 15 mM HEPES (pH 7.7), and 2 mM l-glutamine. After incubation at 37°C and 5% CO2 for 24 h, the plates were fixed in 10% formaldehyde, and the plaques were visualized by staining with 0.05% (wt/vol) crystal violet.
Purification of MNV-1 and VSV.
To grow a large stock of MNV-1, 18 confluent T150 flasks of RAW 267.1 cells were infected with MNV-1 at an MOI of 0.01 in a volume of 3 ml of DMEM. At 1 h postabsorption, 15 ml of DMEM with 2% FBS was added to the flasks, and infected cells were incubated at 37°C for 48 h. When extensive cytopathic effect (CPE) was observed, cell culture fluid was harvested and subjected to three freeze-thaw cycles to release virus particles. The purification of MNV-1 was performed using the method described by Katpally et al. (2008), with minor modifications (33). Briefly, virus suspension was centrifuged at 8,000 × g for 15 min to remove cellular debris. The supernatant was digested with DNase I (10 μg/ml) and MgCl2 (5 mM) at room temperature. After 1 h of incubation, 10 mM EDTA and 1% lauryl sarcosine were added to stop nuclease activity. Virus was concentrated by centrifugation at 82,000 × g for 6 h at 4°C in a Ty 50.2 rotor (Beckman, Brea, CA). The pellet was resuspended in PBS and further purified by centrifugation at 175,000 × g for 6 h at 4°C through a sucrose gradient (7.5 to 45%) in an SW55 Ti rotor (Beckman). The final virus-containing pellets were resuspended in 100 μl PBS. The virus titer was determined by plaque assay using RAW 264.7 cells. The amount of viral protein was measured with Bradford reagent (Sigma Chemical Co., St. Louis, MO). The purified virus stock was diluted at the final concentration of 1 μg/μl. Approximately 1 mg of purified MNV-1 was obtained.
Purification of VSV was performed by the method described in our previous publications (36, 37). Briefly, 10 confluent T150 flasks of BHK-21 cells were infected by VSV at an MOI of 0.01. At 1 h postabsorption, 15 ml of DMEM (supplemented with 2% FBS) was added to the cultures, and infected cells were incubated at 37°C. After 24 h postinfection, cell culture fluid was harvested by centrifugation at 3,000 × g for 5 min. Virus was concentrated by centrifugation at 40,000 × g for 90 min at 4°C in a Ty 50.2 rotor. The pellet was resuspended in NTE buffer (100 mM NaCl, 10 mM Tris, 1 mM EDTA [pH 7.4]) and further purified through 10% sucrose-NTE by centrifugation at 150,000 × g for 1 h at 4°C in an SW50.1 rotor. The final pellet was resuspended in 0.3 ml of NTE buffer. The virus titer was determined by plaque assay using Vero cells, and the protein content was measured by Bradford reagent (Sigma Chemical Co.). The purified virus stock was diluted at the final concentration of 1 μg/μl. Approximately 5 mg of purified MNV-1 was obtained.
Expression and purification of human norovirus VLPs.
The capsid VP1 gene of human norovirus genogroup II.4 strain HS66 (GenBank accession no. EU105469; kindly provided by Linda Saif, The Ohio State University) was amplified by high-fidelity PCR and cloned into a pFastBac-Dual expression vector (Invitrogen) at the SmaI and XhoI sites under the control of the p10 promoter. The resultant plasmid pFastBac-Dual-VP1 was transformed into DH10Bac competent cells. Baculovirus-expressing VP1 protein was generated by transfection of bacmids into Spodoptera frugiperda (Sf9) cells with a Cellfectin transfection kit (Invitrogen), according to the instructions from the manufacturer. Purification of human norovirus virus-like particles (VLPs) from insect cells was described in our previous publication (40). Briefly, Sf9 cells were infected with baculovirus at an MOI of 10, and the infected Sf9 cells and cell culture supernatants were harvested at 6 days postinoculation. The VLPs were purified from cell culture supernatants and cell lysates by ultracentrifugation through a 40% (wt/vol) sucrose cushion, followed by CsCl isopycnic gradient (0.39-g/cm3) ultracentrifugation. Purified VLPs were analyzed by SDS-PAGE, Western blotting, and electron microscopy (EM). The protein concentration of the VLPs was measured by Bradford reagent (Sigma Chemical Co.).
Reverse transcription-PCR (RT-PCR).
Viral genomic RNA was extracted from MNV-1 and VSV suspensions (either gamma irradiation treated or untreated) using an RNeasy minikit (Qiagen, Valencia, CA), according to the manufacturer's instructions. A total of 100 μl of unpurified virus suspension (108 PFU/ml) or 10 μl of purified virus stock (total viral protein concentration at 1 μg/μl) was used for RNA extraction. RT-PCR was performed using a One-Step RT-PCR kit (Qiagen). Two primers (5′-ATGAGGATGAGTGATGGCGC-3′ and 5′-TTATTGTTTGAGCATTCGGCC-3′) were designed to target the capsid MNV-1 VP1 gene. Two primers (5′-ATGTCTGTTACAGTCAAGAG-3′ and 5′-TCATTTGTCAAATTCTGAC-3′) were designed to target the VSV nucleocapsid (N) gene. All the primers were purchased from Sigma. One-Step RT-PCR was performed in a 50-μl reaction mixture containing 400 μM each deoxynucleoside triphosphate (dNTP), 0.6 μM each primer, 4 μl of RNA template, 5 units of RNase inhibitor, and 2 μl of RT-PCR enzyme mix. The amplified products were analyzed by 1% agarose gel electrophoresis.
Analysis of viral proteins by SDS-PAGE.
A total of 20 μl of highly purified viruses (viral protein concentration is 1 μg/μl, and viral titer is approximately 1011 PFU/ml) was treated by gamma irradiation. Two micrograms of highly purified MNV-1 and VSV suspensions (either gamma irradiation treated or untreated) were analyzed by SDS-PAGE. Samples were boiled for 5 min in loading buffer containing 1% SDS, 2.5% β-mercaptoethanol, 6.25 mM Tris-HCl (pH 6.8), and 5% glycerol and loaded into 12% polyacrylamide gel. Viral proteins were visualized by Coomassie blue staining.
Western blotting.
MNV-1 and VSV proteins were separated by 12% SDS-PAGE and transferred onto a Hybond ECL nitrocellulose membrane (Amersham, Piscataway, NJ) in a Mini Trans-Blot electrophoretic transfer cell (Bio-Rad, Hercules, CA). For MNV-1, the blot was probed with rabbit polyclonal MNV-1 antibody (a generous gift from Herbert Virgin) at a dilution of 1:10,000 in blocking buffer (5% skim milk), followed by horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG secondary antibody (Santa Cruz Biotechnology, Santa Cruz, CA) at a dilution of 1:20,000. Western blotting of human norovirus VLPs was essentially identical to that of MNV-1 VP1, with the exception of using polyclonal antibody against VP1 protein of the human norovirus genogroup II.4 strain (a generous gift from Xi Jiang, Cincinnati Children's Hospital Medical Center). For VSV, the blot was probed with mouse monoclonal anti-VSV glycoprotein antibody (Sigma-Aldrich) at a dilution of 1:5,000 in blocking buffer, followed by incubation with HRP-conjugated anti-mouse IgG secondary antibody at a dilution of 1:100,000. Afterwards, the membranes were washed three times with PBS plus 0.02% Tween for 10 min each time. The blots were developed with SuperSignal West Pico chemiluminescent substrate (Thermo Scientific, Pittsburgh, PA) and exposed to Kodak BioMax MR film (Kodak, Rochester, NY).
Transmission electron microscopy.
To determine whether gamma irradiation damages the virus particles, negative staining electron microscopy of purified virions or human norovirus VLPs was performed using the method described in our previous publications (38, 40). Briefly, 20-μl aliquots of either gamma irradiation-treated or untreated samples were fixed in copper grids (Electron Microscopy Sciences, Inc., Hatfield, PA) and negatively stained with 1% ammonium molybdate. Virus particles were visualized by a FEI Tecnai G2 Spirit transmission electron microscope (TEM) at 80 kV at the Microscopy and Imaging Facility at The Ohio State University. Images were captured on a MegaView III side-mounted CCD camera (Soft Imaging System, Lakewood, CO), and figures were processed using Adobe Photoshop software (Adobe Systems, San Jose, CA).
Statistical analysis.
All experiments were carried out in triplicate. Virus survival was expressed as the mean log titer ± standard deviation. Quantitative analysis was performed by densitometric scanning of autoradiographs and ImageQuant TL software (GE Healthcare, Piscataway, NJ). Statistical analysis was performed by one-way multiple comparisons using SPSS 8.0 statistical analysis software (SPSS Inc., Chicago, IL). A P value of <0.05 was considered statistically significant.
RESULTS
Gamma irradiation inactivation of MNV-1 in fresh produce.
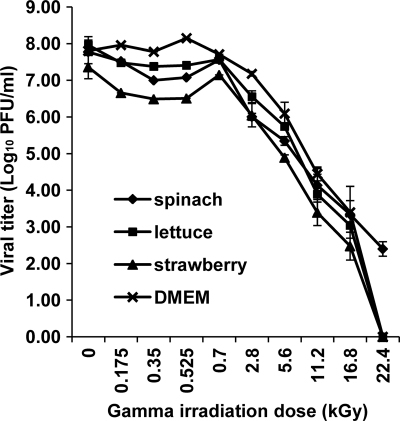
Using cultivable murine norovirus as a surrogate, we have now systematically investigated the survival of the virus in fresh produce after gamma irradiation treatment. As shown in Fig. 1, MNV-1 was gradually inactivated in fresh produce in a dose-dependent manner. However, MNV-1 is resistant to gamma irradiation. At low irradiation doses (0.175 kGy, 0.35 kGy, 0.525 kGy, and 0.7 kGy), there was no significant virus reduction (less than 0.28 logs) in all fresh produce samples (P > 0.05). At a dose of 2.8 kGy, only 1.77-log, 1.40-log, and 1.31-log reductions were observed in spinach, romaine lettuce, and strawberries, respectively. At a dose of 5.6 kGy, only a 1.7- to 2.4-log reduction was observed. Virus inactivation was significantly enhanced at a high dose of gamma irradiation (P < 0.05). At a dose of 11.2 kGy, 3.6- to 4.1-log virus reductions were achieved in all fresh produce samples. At a dose of 22.4 kGy, MNV-1 was completely inactivated in romaine lettuce and strawberries, while 2.4 logs of the virus were detected within the spinach sample. This suggested that food matrices may play a role in protecting virus from inactivation. Taken together, these results demonstrated that MNV-1 was more resistant to gamma irradiation than most bacterial pathogens. A <2-log virus reduction was achieved in all three fresh produce samples at the FDA-approved dose (4 kGy). Therefore, it seems impractical to use gamma irradiation as a means to inactivate virus in fresh produce.
Fig. 1.
Gamma irradiation of MNV-1 in fresh produce and cell culture medium. MNV-1 stock solutions (108 PFU/ml) were inoculated into spinach, lettuce, strawberries, and DMEM to achieve an inoculation level of 107 PFU/g or 107 PFU/ml. Prepared samples were irradiated with up to 22.4 kGy and were stomached for 2 min. The survival plot was determined by plaque assays. Data points were averages of three replicates. Error bars represent ±1 standard deviations.
VSV, an enveloped virus, is more sensitive to gamma irradiation.
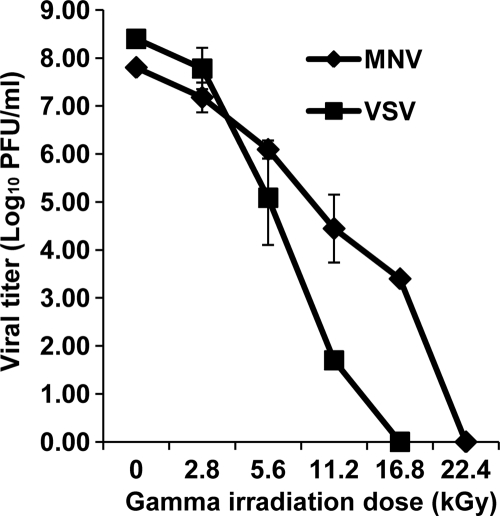
We compared the stability levels after exposure to gamma irradiation of VSV (an enveloped virus) and MNV-1 (a nonenveloped virus). As shown in Fig. 2, VSV was much more sensitive to gamma irradiation than MNV-1. Both VSV and MNV-1 decreased in virus titer as the irradiation dose increased in intensity. However, VSV was reduced at a greater concentration starting from 5.6 kGy. At this dose, a 3.3-log virus reduction was observed in VSV, while only a 1.7-log virus reduction was observed for MNV-1. At the dose of 16.8 kGy, VSV was completely inactivated, as confirmed by plaque assays. However, there were still 3.4-log virus survivors of MNV-1. Actually, an additional 5.6 kGy was required to completely inactive MNV-1.
Fig. 2.
Comparison of the sensitivities of MNV-1 and VSV to gamma irradiation. MNV-1 stock (108 PFU/ml) and VSV stock (109 PFU/ml) were inoculated in DMEM and exposed to irradiation up to 22.4 kGy. The survival plot was determined by plaque assays. Data points were averages of three replicates. Error bars represent ±1 standard deviations.
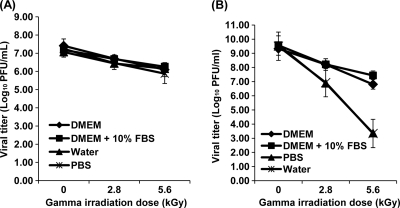
We further tested the stability of MNV-1 and VSV in different buffers. Briefly, MNV-1 and VSV were diluted into four different solutions (water, PBS, DMEM, and DMEM plus 10% FBS) and were treated by two different irradiation doses (2.8 and 5.6 kGy, respectively). As shown in Fig. 3 A, MNV-1 was stable in all four solutions when treated by gamma irradiation. There was no significant difference in virus reduction among these solutions (P > 0.05). Approximately less-than-1.0- and 2.0-log virus reductions were achieved at 2.8 and 5.6 kGy, respectively. In sharp contrast, the stability levels of VSV in these four solutions were different (Fig. 3B). Specifically, VSV was highly susceptible to gamma irradiation when it was diluted in water and PBS. At 2.8 kGy, 2.7-log virus reductions were observed. At 5.6 kGy, 6.3-log virus reductions were observed. However, when VSV was diluted in DMEM and DMEM plus 10% FBS, only 1.1- to 2.5-log and 1.3- to 2.1-log virus reductions were observed at irradiation doses of 2.8 and 5.6 kGy, respectively. These results demonstrated that the matrix of solution does not affect the stability of MNV-1, but it has a dramatic impact on the stability of VSV. Again, these data demonstrated that VSV was much more susceptible to gamma irradiation than MNV-1.
Fig. 3.
Stability of MNV-1 and VSV in different buffers treated by gamma irradiation. MNV-1 and VSV stocks were inoculated into four different buffers (water, PBS, DMEM, and DMEM plus 10% FBS) at final concentrations of 107 and 108 PFU/ml, respectively. The samples were exposed to 2.8 and 5.6 kGy of irradiation. Virus survivors were determined by plaque assays. Data points were averages of three replicates. (A) Stability of MNV-1 in different buffer; (B) stability of VSV in different buffer. The inactivation kinetics of VSV in water and PBS were indistinguishable.
Gamma irradiation degrades viral proteins.
To gain mechanistic insight into viral inactivation, we selected three specific irradiation doses, 2.8 kGy, 5.6 kGy, and 22.4 kGy. We chose 2.8 kGy and 5.6 kGy because less-than-1-log and 2.4-log virus reductions were observed, respectively. A mixture of infectious and noninfectious virus particles would be present at these two doses. In addition, we chose 22.4 kGy because viruses were completely inactivated, which allowed us to precisely determine what happens to virus particles and viral components when no infectious virus particles remained.
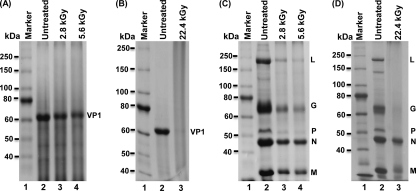
It would be lethal to the virus if viral proteins became damaged by gamma irradiation. The structure of MNV-1 is relatively simple. The surface of MNV-1 possesses a highly stable viral capsid protein, VP1, that surrounds and protects the viral genomic RNA (33, 64). The structure of VSV is significantly different from that of MNV-1. The viral genomic RNA is completely encapsulated by the nucleocapsid (N) protein, forming the N-RNA complex (36, 37). In addition, the viral RNA-dependent RNA polymerase complex (large polymerase protein [L] and phosphoprotein [P]) is tightly bound to N-RNA, which results in the formation of the RNP complex. This RNP complex is further surrounded by a matrix (M) protein and viral envelope. The viral glycoprotein (G) is anchored in the envelope. To gain mechanistic insight into viral inactivation by gamma irradiation, we first determined whether viral proteins were degraded. Briefly, 20 μg of highly purified MNV-1 and VSV was treated by gamma irradiation at three different doses, 2.8, 5.6, and 22.4 kGy. After treatment, 2 μg of either treated or untreated samples were analyzed by SDS-PAGE, followed by Coomassie blue staining. As shown in Fig. 4, the abundance of MNV-1 and VSV proteins gradually decreased when the irradiation dose increased. For MNV-1, the major capsid protein VP1 (molecular mass of approximately 58 kDa) was visualized on SDS-PAGE (Fig. 4A). After 2.8- and 5.6-kGy treatments, the abundance of VP1 proteins decreased to 50% and 30% compared to that of the untreated control, respectively (Fig. 4A). At 22.4 kGy, the VP1 protein was undetectable by SDS-PAGE analysis, suggesting that the MNV-1 VP1 protein was completely degraded (Fig. 4B). For VSV, five structural proteins, L, G, P, N, and M, were observed using SDS-PAGE. However, each viral protein exhibited a different sensitivity to gamma irradiation. The VSV polymerase complex (L and P proteins) and G protein were highly sensitive to gamma irradiation and rapidly degraded after 2.8- and 5.6-kGy doses of irradiation (Fig. 4C). These viral proteins were completely degraded after 22.4-kGy treatment (Fig. 4D). VSV M proteins displayed moderate sensitivity. There were approximately 30%, 20%, and 5% of M protein visualized after treatment with the 2.8-kGy, 5.6-kGy, and 22.4-kGy doses, respectively. The N protein was the VSV protein most resistant to irradiation treatment. There was 45% and 35% of N protein detected after the 2.8-kGy and 5.6-kGy doses, respectively (Fig. 4C). Even after 22.4-kGy treatment, there was approximately 25% of N protein remaining (Fig. 4D). In summary, these data clearly demonstrated that gamma irradiation degraded viral proteins. However, the sensitivity to irradiation of each viral protein varied.
Fig. 4.
Gamma irradiation degrades MNV-1 and VSV structural proteins. (A) SDS-PAGE analysis of purified MNV-1 irradiated at 2.8 kGy and 5.6 kGy. Total viral proteins were analyzed by 12% SDS-PAGE, followed by Coomassie staining. VP1 = MNV-1 capsid protein. (B) SDS-PAGE analysis of purified MNV-1 irradiated at 22.4 kGy. No VP1 protein was present after the treatment. (C) SDS-PAGE analysis of purified VSV irradiated at 2.8 kGy and 5.6 kGy. Five structural proteins of VSV, L, G, P, N, and M, were visualized after Coomassie blue staining. (D) SDS-PAGE analysis of purified VSV irradiated at 22.4 kGy. Only the VSV N and M proteins were visualized after the treatment.
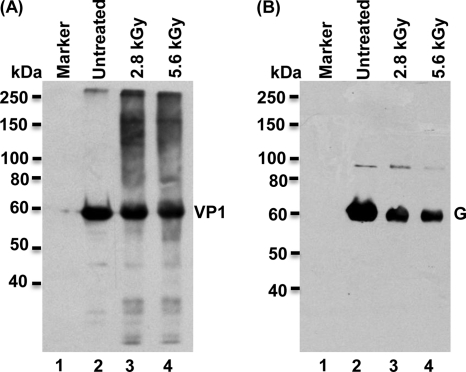
Subsequently, we determined whether the remaining proteins from the 2.8- and 5.6-kGy treatments were antigenic. To address this question, Western blotting was performed using antibody against MNV-1 VP1 or VSV G protein. As shown in Fig. 5, the abundance of MNV-1 VP1 and VSV G proteins detected by Western blotting essentially correlated with the amount of proteins obtained using Coomassie blue staining. Specifically, 50% and 30% of MNV-1 VP1 protein were detected by Coomassie blue staining after the 2.8- and 5.6-kGy treatments, respectively. Based on the size of the bands observed in Western blotting, 65% and 40% of MNV-1 VP1 protein were detected (Fig. 5A). For VSV, 15% and 10% of G protein were detected by Coomassie blue staining after the 2.8 and 5.6-kGy treatments (Fig. 4C). In the Western blot, 20% and 13% of G protein were detected (Fig. 5B). Therefore, these results demonstrated that the undegraded viral proteins still reacted with monoclonal and polyclonal antibodies and perhaps retained correct primary amino acid sequences.
Fig. 5.
Western blot analysis of the MNV-1 capsid protein and VSV G protein after gamma irradiation. (A) Western blot analysis of the MNV-1 capsid protein. Purified MNV-1 was irradiated at doses of 2.8 and 5.6 kGy. Total proteins were separated by SDS-PAGE and subjected to Western blotting using rabbit anti-MNV VP1 polyclonal antibody. (B) Western blot analysis of VSV G protein. Purified VSV was irradiated at doses of 2.8 and 5.6 kGy. Total proteins were separated by SDS-PAGE and subjected to Western blotting using monoclonal antibody against VSV G protein.
Gamma irradiation damages virus particles.
To determine whether gamma irradiation directly damages the virus particles, we analyzed the virus particles by electron microscopy. For the untreated control, MNV-1 particles were small spherical structured virions of 30 to 38 nm in diameter (Fig. 6 A). After 2.8- and 5.6-kGy irradiation treatments, the number of virus particles was significantly reduced (Fig. 6B and C). Clearly, this was due to the fact that the viral capsid protein was degraded by gamma irradiation. At the 22.4-kGy treatment dose, we failed to detect any small round structured virions (Fig. 6D), suggesting that virus particles were completely disrupted. There were some debris materials observed by EM, which perhaps was the degraded viral protein or a mixture of degraded protein and RNA. For VSV, the virion is a bullet-shaped particle that is approximately 70 nm in diameter and 140 nm in length (Fig. 6E). After 2.8-kGy treatment, VSV exhibited some morphological changes (Fig. 6F). Some virions became rounder and more ambiguous rather than having the traditional bullet shape (Fig. 6F). In the 5.6-kGy treated sample, less intact virions were observed. A large number of VSV virions were damaged and thus did not retain their original geometry (Fig. 6G). The viral envelope was not uniform and less defined. After 22.4-kGy treatment, there was no intact bullet-shaped VSV particle observed under EM. Instead, a large concentration of damaged viruses clumped together (Fig. 6H). These clumped materials contained a few particles with severe damage and physical distortions. The viral envelopes were lost, and the viral N-RNA complex was spilling out of the damaged particles. Taken together, these results demonstrated that gamma irradiation damaged virus particles, which in turn resulted in the inactivation of the viruses.
Fig. 6.
Gamma irradiation damages MNV-1 and VSV. Purified MNV-1 and VSV were irradiated at doses of 2.8, 5.6, and 22.4 kGy. Treated and untreated virus particles were negatively stained with 1% ammonium molybdate and visualized by transmission electron microscopy. (A) Untreated MNV-1 virion; (B) MNV-1 particles treated with 2.8 kGy; (C) MNV-1 particles treated with 5.6 kGy; (D) MNV-1 particles treated with 22.4 kGy; (E) untreated VSV virion; (F) VSV particles treated with 2.8 kGy; (G) VSV particles treated with 5.6 kGy; (H) VSV particles treated with 22.4 kGy.
Gamma irradiation degrades viral genomic RNA.
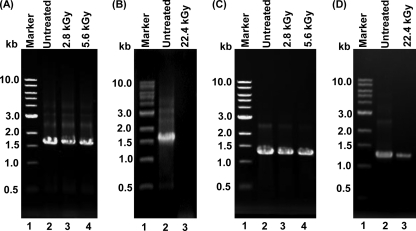
MNV-1 is a positive-sense RNA virus, whereas VSV is a nonsegmented negative-sense RNA virus. It would be lethal to the virus if the viral RNA genome became damaged and/or degraded. Briefly, highly purified MNV-1 and VSV were treated with gamma irradiation at three doses, 2.8, 5.6, and 22.4 kGy. After treatment, RNA was extracted from each sample, followed by RT-PCR to amplify the MNV-1 VP1 and VSV N genes, and the products were visualized by gel electrophoresis on 1% agarose gel. As shown in Fig. 7 A, the VP1 gene was amplified in RNA samples extracted from MNV-1 treated by 2.8- and 5.6-kGy irradiation doses, although the abundance of the VP1 gene decreased compared with those of the untreated controls. This was not surprising since there were significant amounts of virus survivors at both doses. However, the VP1 gene was not detectable in the RNA sample from MNV-1 stock treated by 22.4-kGy irradiation in which MNV-1 was completely inactivated (Fig. 7B), suggesting that MNV-1 genomic RNA was completely degraded after this dose of irradiation exposure. Similarly, the amount of the VSV N gene decreased when VSV was treated by 2.8- and 5.6-kGy irradiation (Fig. 7C). Interestingly, a significant amount of the VSV N gene was still detected in the RNA sample from VSV that was treated with 22.4 kGy (Fig. 7D). It should be emphasized that VSV was completely inactivated at a dose of 16.8 kGy (Fig. 2). Since VSV genomic RNA was completely encapsidated by the N protein, intact RNA may still present if the N protein was not completely degraded. Indeed, approximately 25% of the VSV N protein remained after 22.4-kGy treatment (Fig. 4D). Therefore, the VSV genome was still detectable by RT-PCR at this irradiation dose.
Fig. 7.
RT-PCR analysis of MNV-1 and VSV after gamma irradiation. (A) Detection of the VP1 gene from MNV-1 irradiated with 2.8 and 5.6 kGy. Viral genomic RNA was extracted from either treated or untreated MNV-1. The VP1 gene of MNV-1 was amplified by one-step RT-PCR, and PCR products were visualized on 1% agarose gel electrophoresis. (B) Detection of the VP1 gene from MNV-1 irradiated with 22.4 kGy. (C) Detection of the N gene from VSV irradiated with 2.8 and 5.6 kGy. Viral genomic RNA was extracted from either treated or untreated VSV. The VSV N gene was amplified by one-step RT-PCR. (D) Detection of the N gene from VSV irradiated with 22.4 kGy.
Next, we directly treated the viral genomic RNA by gamma irradiation. Briefly, viral genomic RNA was extracted from MNV-1 and VSV and treated by three irradiation doses (2.8, 5.6, and 11.2 kGy), followed by RT-PCR. At doses of 5.6 and 11.2 kGy, MNV-1 and VSV genomic RNA was undetectable by RT-PCR, respectively (data not shown). Taken together, these data demonstrated that gamma irradiation degraded viral genomic RNA.
Gamma irradiation damages human norovirus VLPs.
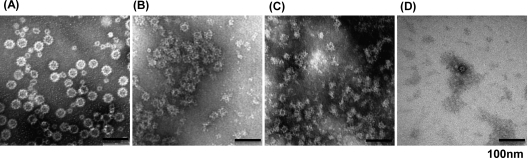
Unfortunately, the study of the survival of human norovirus is hampered because it cannot be grown in cell culture systems (19). However, it is known that expression of human norovirus VP1 protein in insect cells results in self-assembly of VLPs that are structurally and antigenically similar to native virions (6, 14, 30). Therefore, VLPs have been used as a tool to understand the biology of human norovirus (6. 14, 30). In fact, our above-described experiments demonstrated that the disruption of viral proteins and structure is one of the mechanisms of virus inactivation. Thus, we wanted to demonstrate the sensitivity of human norovirus VLPs to gamma irradiation. Briefly, the VP1 capsid gene of human norovirus strain HS66 was cloned into the baculovirus expression system and expressed in insect cells, and the VLPs were purified as described in Materials and Methods. The purified VLPs were negatively stained with ammonium molybdate and analyzed by EM. As shown in Fig. 8A, the expressed VP1 protein formed small round structured particles, which were similar to the native human norovirus virions. Consistent with previous observations (6), two sizes of particles were found in EM analysis (Fig. 8A). The size of the larger particles was between 30 to 38 nm, and the smaller size was between 18 to 20 nm. To determine whether gamma irradiation damages the capsid, human norovirus VLPs were treated with three doses of irradiation. At a dose of 2.8 kGy, VLPs were clumped together, and the morphology of VLPs was altered (Fig. 8B). At a dose of 5.6 kGy, we observed a large concentration of protein debris, and the structure of VLPs disappeared (Fig. 8C). After 22.4-kGy irradiation, we could not find any small spherically structured VLPs (Fig. 8D), suggesting that the VLPs were completely disrupted by gamma irradiation.
Fig. 8.
Gamma irradiation damages human norovirus VLPs. Human norovirus VLPs were expressed and purified from insect cells using a baculovirus expression system. The VLPs were irradiated at three doses, 2.8, 5.6, and 22.4 kGy. Treated and untreated VLPs were negatively stained with 1% ammonium molybdate and visualized by transmission electron microscopy. (A) Untreated human norovirus VLPs; (B) VLPs treated with 2.8 kGy; (C) VLPs treated with 5.6 kGy; (D) VLPs treated with 22.4 kGy.
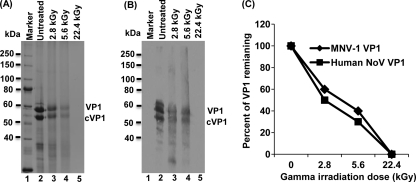
To demonstrate whether gamma irradiation degrades the capsid protein, the irradiated VLPs were analyzed by SDS-PAGE. For the untreated control, two protein bands with molecular masses of 58 and 55 kDa were observed (Fig. 9 A, lane 2). The 58-kDa protein represented the native full-length VP1 protein, and the 55-kDa protein was the cleaved form of VP1 protein (cVP1). This is consistent with the previous observation that baculovirus-expressed VP1 is cleaved in insect cells (6, 30). After irradiation, the abundances of both of the protein bands were significantly diminished. Approximately 40% and 25% of human norovirus VP1 protein remained after treatment at doses of 2.8 and 5.6 kGy, respectively (Fig. 9A, lanes 3 and 4), and VP1 protein was not visualized at all after 22.4 kGy of irradiation (Fig. 9A, lane 5). For MNV-1 VP1 protein, 50% and 30% of VP1 remained after 2.8- and 5.8-kGy treatments, respectively. Furthermore, similar to MNV-1 VP1, the remaining human norovirus VP1 from irradiation still reacted with the polyclonal antibody (Fig. 9B), suggesting that the undegraded protein was still antigenic. Figure 9C shows the direct comparison of the stability levels after exposure to gamma irradiation of the capsid proteins of MNV-1 and human norovirus. Indeed, there was no significant difference between these two capsid proteins (P > 0.05). Therefore, it seems that the irradiation dose that degraded MNV-1 VP1 may be sufficient to degrade human norovirus VP1.
Fig. 9.
Gamma irradiation degrades the capsid protein of human norovirus. (A) Visualization of human norovirus capsid protein by 12% SDS-PAGE. The purified VLPs were irradiated with 2.8, 5.6, and 22.4 kGy. Total viral proteins were analyzed by 12% SDS-PAGE, followed by Coomassie staining. VP1 = human norovirus capsid protein; cVP1 = cleaved VP1 protein. (B) Western blot analysis of human norovirus VP1 protein. Samples identical to those shown in panel A were separated by SDS-PAGE and subjected to Western blotting using a polyclonal antibody against VP1 protein. (C) Comparison of the stability levels of the capsid proteins of MNV-1 and human norovirus after exposure to gamma irradiation. Two micrograms of MNV-1 and human norovirus VLPs was treated with 2.8, 5.6, and 22.4 kGy. Total proteins were separated by SDS-PAGE, followed by Coomassie staining. The remaining proteins from gamma irradiation were quantified by ImageQuant TL software. Data points were averages of three replicates.
DISCUSSION
Irradiation was patented for food preservation in 1905 in France. Since then, irradiation has been found to be an effective food processing technology to eliminate bacteria, insects, fungi, and pests and poses no significant risks to human health or the environment (5, 17, 34, 45, 59). However, the feasibility of gamma irradiation to eliminate viruses in foods is less understood. In this study, we found that MNV-1, a human norovirus surrogate, is resistant to gamma irradiation. Only 1.7- to 2.4-log virus reductions in fresh produce samples at an irradiation dose of 5.6 kGy were observed. Although VSV, an enveloped virus, is more susceptible than MNV-1, only 3.3-log reductions at an irradiation dose of 5.6 kGy in DMEM were achieved. Overall, viruses are much more resistant to irradiation than bacterial pathogens. Furthermore, we found that gamma irradiation degraded viral structural proteins and genetic materials and damaged viral particles, which resulted in viral inactivation. Although gamma irradiation seems impractical to eliminate the virus contaminants in fresh produce by the FDA-approved irradiation dose limits, this technology may be practical to inactivate viruses in other foods and to sterilize medical devices, ophthalmic solutions, pharmaceuticals, tissue culture sera, animal waste, and municipal sewage (17, 28, 29, 55, 57, 58).
Gamma irradiation is not effective in eliminating the human norovirus surrogate in fresh produce.
A large amount of outbreak data showed that fresh produce has become one of the major vehicles of transmitting food-borne viruses, particularly human norovirus (2, 8, 26, 39, 53). Recent evidence showed that human norovirus not only tightly binds to fresh produce but also can become internalized (50, 61, 63). Traditional sanitization strategy is thus not effective in removing human norovirus from produce, particularly for internalized virions. To improve the safety of fresh produce, the FDA has approved food irradiation at doses of up to 4 kGy to inactivate bacterial pathogens. It has been demonstrated that irradiation doses of 4 to 5 kGy almost completely inactivated major food-borne bacterial pathogens such as Escherichia coli O157:H7, Staphylococcus aureus, Listeria monocytogenes, Salmonella enterica serovar Typhimurium, and mycotoxin-producing Aspergillus spp. and Fusarium spp. (5, 17, 34, 45, 52, 59).
Our initial objective was to determine whether food-borne viruses can be inactivated at the approved irradiation dose. Unfortunately, according to our results, a 5.6-kGy dose eliminated only MNV-1 at a range between 2.2 and 2.4 logs in the produce samples. Thus, the irradiation dose that normally would eliminate all pathogenic bacteria would not be sufficient to inactivate the norovirus surrogate. Previously, gamma irradiation of the other two human norovirus surrogates, feline calicivirus (FCV) and canine calicivirus (CaCV), has been reported (16). Surprisingly, a 3-log virus reduction at a very low dose of gamma irradiation (0.5 kGy for FCV and 0.3 kGy for CaCV) was observed (16). Based on our results, MNV-1 is much more resistant than FCV and CaCV to gamma irradiation. Irradiation of other food-borne and waterborne viruses, such as hepatitis A virus (HAV) (9), rotavirus (41), and poliovirus (27), has been reported. It was found that the D10 values (dose required to reduce infectivity by 1 log10) for HAV in lettuce and strawberries were 2.72 ± 0.05 and 2.97 ± 0.18 kGy, respectively (9), while the D10 value for gamma irradiating HAV in clams and oysters was 2 kGy (41). It was also reported that the D10 value for rotavirus required 2.4 kGy (41). Poliovirus inoculated into fish fillets required a dose of 6 kGy to achieve a 2-log reduction (27). Overall, major food-borne and waterborne viruses are highly resistant to gamma irradiation. It seems impractical to utilize gamma irradiation to target viruses in fresh produce for the food processing industry at the approved doses. However, it is possible that food irradiation can be used for inactivating viruses in other food products. According to FDA code 21CFR179.26 (62), a medium dose of irradiation (up to 10 kGy) can be used for killing pathogenic and spoilage microorganisms within fresh fish, mushrooms, and frozen poultry. Medium-dose irradiation is also employed for reducing the cooking time of dehydrated vegetables and extending the shelf life of food products. High-dose (10- to 50-kGy) irradiation is reserved for the sterilization of spices, herbs, seasonings, meats, and other prepared foods in combination with heat in order to inactivate enzymes.
Mechanism of viral inactivation by gamma irradiation.
In early studies, it is thought that the damage of genetic material was the primary mechanism of microorganism inactivation by gamma irradiation (55, 57, 58). Gamma rays can directly “hit” the genetic material and indirectly react with the nucleic acid via the free radicals generated when gamma rays strike water molecules, which results in single- or double-strand breaks, cross-linkage breaks, and nucleotide degradation. Consistent with this, the amount of the MNV-1 VP1 gene decreased as the irradiation dose increased. For MNV-1, genomic RNA was undetectable when the virus was treated by 22.4-kGy irradiation, suggesting that RNA was degraded. However, genetic material may not be completely degraded when a virus becomes inactivated. For example, VSV was completely killed after the 16.8-kGy dose, but a significant amount of the VSV N gene was amplified even after 22.4-kGy treatment (Fig. 7D), a dose well beyond its inactivation limit. This was consistent with the fact that there was a significant amount of viral N protein remaining (Fig. 4D), which acted as a shield for the genetic material. It is also possible that RNA damage from irradiation was fortuitously not in the segment that was amplified by RT-PCR.
In addition to RNA degradation, we found that gamma irradiation also damaged the virion structure and viral proteins. The number of intact virus particles and the concentration of viral proteins gradually decreased as the gamma irradiation dose increased. The damage caused by gamma irradiation included breaking the viral envelope, disrupting the viral capsid, and physically distorting the virion geometry. Gamma irradiation may disrupt not only the covalent bonds but also noncovalent interactions such as hydrogen bonds, ionic bonds, van der Waals forces, and hydrophobic interactions, which are responsible for the secondary, tertiary, and quaternary structure of a protein. The full-length viral proteins were physically eroded without observation of intermediate products, such as small peptides or protein fragments. This suggested that gamma irradiation not only disrupted the quaternary and tertiary structures of a protein but also broke down secondary structure and primary amino acid sequences. Although virus inactivation is essentially a one-hit event, some viral proteins may entail many more hits at high gamma irradiation doses. It was likely that these peptides, fragments, or amino acids were too small to be resolved by SDS-PAGE. Interestingly, the remaining viral proteins after 2.8 and 5.6 kGy of irradiation still reacted with antibody, suggesting that these proteins retained correct primary amino acid sequences. Previously, it was shown that UV irradiation affected the functions of the capsid proteins of HAV and FCV (46, 47). It will be interesting to determine whether UV also affects the integrity of virion structure.
We found that VSV was much more sensitive to gamma irradiation than MNV-1. There may be many factors contributing to irradiation sensitivity. First, the genome size of VSV (11 kb) is larger than that of MNV-1 (7.4 kb). Indeed, early studies suggest there is an inverse relationship between inactivation dose and viral genome size (55, 57, 58). Second, enveloped viruses are likely to be more sensitive to gamma irradiation since the envelope may be easily damaged by irradiation. Third, VSV is structurally more complicated than MNV-1. VSV possesses five structural proteins, and damage to any of these structural proteins would be lethal to the virus. Finally, the size of virus particle may also contribute to the irradiation sensitivity. Larger particles would be more likely to be hit by gamma rays due to the larger exposed surface area.
A clearer understanding of the mechanism of viral inactivation would also guide us to properly use gamma irradiation processing. Prior to our study, it was thought that damage of viral genetic material is the major target for virus inactivation (28, 29, 55, 57, 58). Therefore, gamma irradiation is widely used as means to prepare inactivated viral vaccines (12, 42). However, our mechanistic studies strongly suggest that gamma irradiation is not an ideal tool for preparing inactivated vaccines. First of all, a low dose of gamma irradiation is not able to completely inactivate the viruses, which will compromise the safety of the vaccine. More importantly, a high dose of gamma irradiation disrupts the structure of the virus particle and physically reduces the number of total viral proteins, including the immunogenic antigens (such as MNV-1 VP1 and VSV G), which are responsible for triggering an effective immune response. Thus, the efficacy of the vaccine would likely be impaired because the antigens would be damaged or severely diminished.
Stability of the capsid proteins of MNV-1 and human norovirus after exposure to gamma irradiation.
Because human norovirus cannot be grown in cell culture systems (19), no survival data are currently available for this important pathogen that has dramatic impacts on food safety and public health. Using human norovirus VLPs as a model, we demonstrated that the capsid protein of human norovirus was degraded by gamma irradiation. For calicivirus, it is widely accepted that VLPs are structurally, morphologically, and antigenically similar to native virion (6, 7, 14, 30, 49). Three-dimensional structure imaging revealed that human norovirus VLPs are composed of 90 dimers of the capsid protein, and each of which forms an arch-like capsomere (14, 49). Such structure is highly stable and thus can protect genomic RNA. For MNV-1, there was 50% and 30% of VP1 protein remaining after 2.8 and 5.6 kGy of irradiation, respectively. However, there was 40% and 25% of human norovirus VP1 remaining after 2.8 and 5.6 kGy of irradiation, respectively. Similar to MNV-1, human norovirus VLPs were completely degraded at 22.4 kGy of irradiation. This suggests that the capsid proteins of human norovirus and MNV-1 may have equivalent levels of stability after exposure to gamma irradiation. EM analysis showed that gamma irradiation altered the morphology of VLPs, which resulted in the disruption of VLPs. Although our study was based on human norovirus VLPs, it is the first evidence that the degradation kinetics of human norovirus capsid by gamma irradiation is similar to that of MNV-1. It is worthy to emphasize that human norovirus also encodes a minor capsid protein, VP2, a 18-kDa protein that plays an important role in stabilizing the major capsid protein (VP1) by preventing the virus particle from undergoing degradation and disassembly (7). The native human norovirus possesses both VP1 and VP2, which is likely more stable than VLPs that contain only VP1 alone. Taking account of this factor, it is possible that human norovirus may be even more stable after exposure to gamma irradiation than MNV-1.
In summary, we demonstrated that the human norovirus surrogate is resistant to gamma irradiation. We provided the first evidence that the capsid of human norovirus has an stability equivalent to that of its surrogate. Furthermore, we provided new mechanistic insights into viral inactivation by gamma irradiation. A better understanding of the mechanism of viral inactivation will guide the proper application of irradiation in industry.
ACKNOWLEDGMENTS
This study was partially supported by a special emphasis grant (2010-01498) from the USDA National Integrated Food Safety Initiative (NIFSI) and a food safety challenge grant (2010-04443) from the USDA Agriculture and Food Research Initiative (AFRI) to J.L.
We thank Herbert W. Virgin for his generous gifts of MNV-1 and antibody. We acknowledge the OSU Nuclear Reactor Laboratory for the gamma irradiation treatment. We are grateful to Linda Saif for providing the human norovirus VP1 gene and Xi Jiang for providing human norovirus VP1 antibody. We thank Sean Whelan for providing VSV. We thank the members of the Li laboratory for critical reviews of the manuscript.
Footnotes
Published ahead of print on 25 March 2011.
REFERENCES
- 1. Adler J. L., Zickl R. 1969. Winter vomiting disease. J. Infect. Dis. 119:668–673 [DOI] [PubMed] [Google Scholar]
- 2. Allwood P. B., et al. 2004. Occurrence of Escherichia coli, noroviruses, and F-specific coliphages in fresh market-ready produce. J. Food Prot. 67:2387–2390 [DOI] [PubMed] [Google Scholar]
- 3. Baert L., Debevere J., Uyttendaele M. 2009. The efficacy of preservation methods to inactivate foodborne viruses. Int. J. Food Microbiol. 131:83–94 [DOI] [PubMed] [Google Scholar]
- 4. Baert L., Uyttendaele M., Vermeersch M., Van Coillie E., Debevere J. 2008. Survival and transfer of murine norovirus 1, a surrogate for human noroviruses, during the production process of deep-frozen onions and spinach. J. Food Prot. 71:1590–1597 [DOI] [PubMed] [Google Scholar]
- 5. Bari M. L., et al. 2005. Effectiveness of irradiation treatments in inactivating Listeria monocytogenes on fresh vegetables at refrigeration temperature. J. Food Prot. 68:318–323 [DOI] [PubMed] [Google Scholar]
- 6. Bertolotti-Ciarlet A., White L. J., Chen R., Venkataram Prasad B. V., Estes M. K. 2002. Structural requirements for the assembly of Norwalk virus-like particles. J. Virol. 76:4044–4055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Bertolotti-Ciarlet A., Crawford S. E., Hutson A. M., Estes M. K. 2003. The 3′ end of Norwalk virus mRNA contains determinants that regulate the expression and stability of the viral capsid protein VP1: a novel function for the VP2 protein. J. Virol. 77:11603–11615 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Beuchat L. R. 1996. Pathogenic microorganisms associated with fresh produce. J. Food Prot. 59:204–216 [DOI] [PubMed] [Google Scholar]
- 9. Bidawid S., Farber J. M., Sattar S. A. 2000. Inactivation of hepatitis A virus (HAV) in fruits and vegetables by gamma irradiation. Int. J. Food Microbiol. 57:91–97 [Google Scholar]
- 10. Butot S., Putallaz T., Sánchez G. 2007. Procedure for rapid concentration and detection of enteric viruses from berries and vegetables. Appl. Environ. Microbiol. 73:186–192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Butot S., Putallaz T., Amoroso R., Sánchez G. 2009. Inactivation of enteric viruses in minimally processed berries and herbs. Appl. Environ. Microbiol. 75:4155–4161 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Campbell C. H. 1985. Immunogenicity of bluetongue virus inactivated by gamma irradiation. Vaccine 3:401–406 [DOI] [PubMed] [Google Scholar]
- 13. Cannon J. L., et al. 2006. Surrogates for the study of norovirus stability and inactivation in the environment: a comparison of murine norovirus and feline calicivirus. J. Food Prot. 69:2761–2765 [DOI] [PubMed] [Google Scholar]
- 14. Chen R., Neill J. D., Estes M. K., Prasad B. V. 2006. X-ray structure of a native calicivirus: structural insights into antigenic diversity and host specificity. Proc. Natl. Acad. Sci. U. S. A. 103:8048–8053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Cliver D. O. 1997. Virus transmission via food. Food Technol. 51:71–78 [PubMed] [Google Scholar]
- 16. De Roda Husman A. M., et al. 2004. Calicivirus inactivation by nonionizing (253.7-nanometer-wavelength [UV]) and ionizing (gamma) radiation. Appl. Environ. Microbiol. 70:5089–5093 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Dickson J. S. 2001. Radiation inactivation of microorganisms, p. 23–35In Molins R. (ed.), Food irradiation: principles and applications. Wiley-Interscience, New York, NY [Google Scholar]
- 18. Doyle M. P., Erickson M. C. 2008. Summer meeting 2007—the problems with fresh produce: an overview. J. Appl. Microbiol. 105:317–330 [DOI] [PubMed] [Google Scholar]
- 19. Duizer E., et al. 2004. Laboratory efforts to cultivate noroviruses. J. Gen. Virol. 85:79–87 [DOI] [PubMed] [Google Scholar]
- 20. Duizer E., et al. 2004. Inactivation of caliciviruses. Appl. Environ. Microbiol. 70:4538–4543 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Estes M. K., Prasad B. V., Atmar R. L. 2006. Noroviruses everywhere: has something changed? Curr. Opin. Infect. Dis. 19:467–474 [DOI] [PubMed] [Google Scholar]
- 22. Fan X., Niemira B. A., Prakash A. 2008. Irradiation of fresh fruits and vegetables. Food Technol. 62:36–43 [Google Scholar]
- 23. Farkas J. 1998. Irradiation as a method for decontaminating food: a review. Int. J. Food Microbiol. 44:189–204 [DOI] [PubMed] [Google Scholar]
- 24. Grove S. F., et al. 2006. Inactivation of foodborne viruses of significance by high pressure and other processes. J. Food Prot. 69:957–968 [DOI] [PubMed] [Google Scholar]
- 25. Harris J. P., Edmunds W. J., Pebody R., Brown D. W., Lopman B. A. 2008. Deaths from norovirus among the elderly, England and Wales. Emerg. Infect. Dis. 14:1546–1552 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Heaton J. C., Jones K. 2008. Microbial contamination of fruit and vegetables and the behaviour of enteropathogens in the phyllosphere: a review. J. Appl. Microbiol. 104:613–626 [DOI] [PubMed] [Google Scholar]
- 27. Heidelbaugh N. D., Giron D. J. 1969. Effect of processing on recovery of polio virus from inoculated foods. J. Food Sci. 34:239–241 [Google Scholar]
- 28. Hiemstra H., et al. 1991. Inactivation of human immunodeficiency virus by gamma radiation and its effect on plasma and coagulation factors. Transfusion 31:32–39 [DOI] [PubMed] [Google Scholar]
- 29. House C., House J. A., Yedloutschnig R. J. 1990. Inactivation of viral agents in bovine serum by gamma irradiation. Can. J. Microbiol. 36:737–740 [DOI] [PubMed] [Google Scholar]
- 30. Jiang X., Wang M., Graham D. Y., Estes M. K. 1992. Expression, self-assembly, and antigenicity of the Norwalk virus capsid protein. J. Virol. 66:6527–6532 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Kapikian A. Z., et al. 1972. Visualization by immune electron microscopy of a 27-nm particle associated with acute infectious nonbacterial gastroenteritis. J. Virol. 10:1075–1081 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Karst S. M., Wobus C. E., Lay M., Davidson J., Virgin H. W., IV 2003. STAT 1-dependent innate immunity to a Norwalk-like virus. Science 299:1575–1578 [DOI] [PubMed] [Google Scholar]
- 33. Katpally U., Wobus C. E., Dryden K., Virgin H. W., IV, Smith T. J. 2008. Structure of antibody-neutralized murine norovirus and unexpected differences from virus-like particles. J. Virol. 82:2079–2088 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Khattak A. B., et al. 2005. Shelf life extension of minimally processed cabbage and cucumber through gamma irradiation. J. Food Prot. 68:105–110 [DOI] [PubMed] [Google Scholar]
- 35. Koopmans M., Duizer E. 2004. Foodborne viruses: an emerging problem. Int. J. Food Microbiol. 90:23–41 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Li J., Fontaine-Rodriguez E. C., Whelan S. P. 2005. Amino acid residues within conserved domain VI of the vesicular stomatitis virus large polymerase protein essential for mRNA cap methyltransferase activity. J. Virol. 79:13373–13384 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Li J., Wang J. T., Whelan S. P. 2006. A unique strategy for mRNA cap methylation used by vesicular stomatitis virus. Proc. Natl. Acad. Sci. U. S. A. 103:8493–8498 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Lou F., Needoo H., Chen H., Li J. 2011. Inactivation of human norovirus surrogate by high pressure processing: effectiveness, mechanism and potential application in fresh produce industry. Appl. Environ. Microbiol. 77:1862–1871 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Lynch M. F., Tauxe R. V., Hedberg C. W. 2009. The growing burden of foodborne outbreaks due to contaminated fresh produce: risks and opportunities. Epidemiol. Infect. 137:307–315 [DOI] [PubMed] [Google Scholar]
- 40. Ma Y., Li J. 2011. Vesicular stomatitis virus as a vector to deliver human norovirus virus-like particles: a new vaccine candidate against an important non-cultivable virus. J. Virol. 85:2942–2952 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Mallett J. C., Beghian L. E., Metcalf T. G., Kaylor J. D. 1991. Potential of irradiation technology for improved shellfish sanitation. J. Food Saf. 11:231–245 [Google Scholar]
- 42. Martin S. S., et al. 2010. Comparison of the immunological responses and efficacy of gamma-irradiated V3526 vaccine formulations against subcutaneous and aerosol challenge with Venezuelan equine encephalitis virus subtype IAB. Vaccine 28:1031–1040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Mead P. S., et al. 1999. Food-related illness and death in the United States. Emerg. Infect. Dis. 5:607–625 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Mormann S., Dabisch M., Becker B. 2010. Effects of technological processes on the tenacity and inactivation of norovirus genogroup II in experimentally contaminated foods. Appl. Environ. Microbiol. 76:536–545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Neal J. A., Cabrera-Diaz E., Marquez-Gonzalez M., Maxim J. E., Castillo A. 2008. Reduction of Escherichia coli O157:H7 and Salmonella on baby spinach, using electron beam radiation. J. Food Prot. 71:2415–2420 [DOI] [PubMed] [Google Scholar]
- 46. Nuanualsuwan S., Cliver D. O. 2003. Capsid functions of inactivated human picornaviruses and feline calicivirus. Appl. Environ. Microbiol. 69:350–357 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Nuanualsuwan S., Mariam T., Himathongkham S., Cliver D. O. 2002. Ultraviolet inactivation of feline calicivirus, human enteric viruses and coliphages. Photochem. Photobiol. 76:406–410 [DOI] [PubMed] [Google Scholar]
- 48. Pönkä A., Maunula L., von Bonsdorff C. H., Lyytikäinen O. 1999. An outbreak of calicivirus associated with consumption of frozen raspberries. Epidemiol. Infect. 123:469–474 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Prasad B. V., et al. 1999. X-ray crystallographic structure of the Norwalk virus capsid. Science 286:287–290 [DOI] [PubMed] [Google Scholar]
- 50. Rawsthorne H., Phister T. G., Jaykus L. A. 2009. Development of a fluorescent in situ method for visualization of enteric viruses. Appl. Environ. Microbiol. 75:7822–7827 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Rzezutka A., Cook N. 2004. Survival of human enteric viruses in the environment and food. FEMS Microbiol. Rev. 28:441–453 [DOI] [PubMed] [Google Scholar]
- 52. Schmidt H. M., Palekar M. P., Maxim J. E., Castillo A. 2006. Improving the microbiological quality and safety of fresh-cut tomatoes by low-dose electron beam irradiation. J. Food Prot. 69:575–581 [DOI] [PubMed] [Google Scholar]
- 53. Seymour I. J., Appleton H. 2001. Foodborne viruses and fresh produce. J. Appl. Microbiol. 91:759–773 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Sivapalasingam S., Friedman C. R., Cohen L., Tauxe R. V. 2004. Fresh produce: a growing cause of outbreaks of foodborne illness in the United States, 1973 through 1997. J. Food Prot. 67:2342–2353 [DOI] [PubMed] [Google Scholar]
- 55. Sullivan R., Fassolitis A. C., Larkin E. P., Read R. B., Jr., Peeler J. T. 1971. Inactivation of thirty viruses by gamma radiation. Appl. Microbiol. 22:61–65 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Teunis P. F., et al. 2008. Norwalk virus: how infectious is it? J. Med. Virol. 80:1468–1476 [DOI] [PubMed] [Google Scholar]
- 57. Thomas F. C., et al. 1981. Gamma ray inactivation of some animal viruses. Can. J. Comp. Med. 45:397–399 [PMC free article] [PubMed] [Google Scholar]
- 58. Thomas F. C., Ouwerkerk T., McKercher P. 1982. Inactivation by gamma irradiation of animal viruses in simulated laboratory effluent. Appl. Environ. Microbiol. 43:1051–1056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Thomas P. 2001. Irradiation of fruits and vegetables, p. 213–240In Molins R. (ed.), Food irradiation: principles and applications. Wiley-Interscience, New York, NY [Google Scholar]
- 60. Tree J. A., Adams M. R., Lees D. N. 2005. Disinfection of feline calicivirus (a surrogate for Norovirus) in wastewaters. J. Appl. Microbiol. 98:155–162 [DOI] [PubMed] [Google Scholar]
- 61. Urbanucci A., Myrmel M., Berg I., von Bonsdorff C. H., Maunula L. 2009. Potential internalisation of caliciviruses in lettuce. Int. J. Food Microbiol. 135:175–178 [DOI] [PubMed] [Google Scholar]
- 62. U.S. FDA 1999. Irradiation in the production, processing and handling of food. Ionizing radiation for the treatment of food. Code of Federal Regulations 21CFR179.26 [Google Scholar]
- 63. Wei J., Jin Y., Sims T., Kniel K. E. 2010. Manure- and biosolids-resident murine norovirus 1 attachment to and internalization by Romaine lettuce. Appl. Environ. Microbiol. 76:578–583 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Wobus C. E., Thackray L. B., Virgin H. W., IV 2006. Murine norovirus: a model system to study norovirus biology and pathogenesis. J. Virol. 80:5104–5112 [DOI] [PMC free article] [PubMed] [Google Scholar]