Abstract
Influenza A virus assembly is a complex process that requires the intersection of pathways involved in transporting viral glycoproteins, the matrix protein, and viral genomes, incorporated in the viral ribonucleoprotein (vRNP) complex, to plasma membrane sites of virion formation. Among these virion components, the mechanism of vRNP delivery is the most incompletely understood. Here, we reveal a functional relationship between the cellular Rab11 GTPase isoform, RAB11A, and vRNPs and show that RAB11A is indispensable for proper vRNP transport to the plasma membrane. Using an immunofluorescence-based assay with a monoclonal antibody that recognizes nucleoprotein in the form of vRNP, we demonstrate association between RAB11A and vRNPs at all stages of vRNP cytoplasmic transport. Abrogation of RAB11A expression through small interfering RNA (siRNA) treatment or disruption of RAB11A function by overexpression of dominant negative or constitutively active proteins caused aberrant vRNP intracellular accumulation, retention in the perinuclear region, and lack of accumulation at the plasma membrane. Complex formation between RAB11A and vRNPs was further established biochemically. Our results uncover a critical host factor with an essential contribution to influenza virus genome delivery and reveal a potential role for RAB11A in the transport of ribonucleoprotein cargo.
INTRODUCTION
Influenza A virus is one of the most important human pathogens, causing annual epidemics with significant worldwide mortality and sporadic pandemics in which increased human infections and mortality typically occur. In addition, highly pathogenic avian influenza (HPAI) viruses continue to cause human infections that result in exceptionally high mortality rates (∼60%) (51), and the threat of an HPAI pandemic remains an alarming possibility. For these reasons, a comprehensive understanding of the processes involved in influenza virus replication and interaction of the virus with its host is imperative as any essential process or interaction could represent a potential target for a novel intervention strategy. In this regard, we set out to clarify the mechanism of influenza virus assembly in more detail.
Infectious influenza virus production requires that each viral structural component is delivered to plasma membrane sites of virion formation (i.e., lipid raft microdomains), a process thought to commence with the clustering of the viral hemagglutinin (HA) and neuraminidase (NA) proteins (24). As integral membrane proteins, HA and NA are transported through the Golgi complex in infected cells and are directed to lipid rafts by sorting determinants within their transmembrane domains and cytoplasmic tails (5, 42, 53). The viral matrix protein (M1), the major interior virion structural component, exhibits intrinsic membrane association properties (23, 46, 52) and is recruited to lipid rafts through interactions with the HA and NA cytoplasmic tails (1, 14). M1 also interacts with viral ribonucleoproteins (vRNPs), the functional influenza virus genomic subunits (8, 36), and with the viral M2 ion channel (12). M2 may have a role in vRNP recruitment as virions produced by viruses encoding M2 that lacks its cytoplasmic tail are defective in nucleoprotein (NP) and genomic RNA incorporation (29). M2 was recently shown to require the cellular Rab11 GTPase for transport to the plasma membrane (40); however, the mechanistic basis for vRNP delivery to virion budding sites has not been clearly elucidated.
Influenza vRNPs consist of a negative-sense genomic viral RNA (vRNA) wrapped around multiple subunits of viral NP and are associated with the viral polymerase complex (PB2, PB1, and PA). Individual vRNPs assemble in the nucleus and undergo nuclear export after M1-mediated release from the nuclear matrix (9, 28, 41). vRNP-M1 complexes are thought to associate with the viral nuclear export protein (NS2/NEP), which bridges an interaction with the cellular CRM1 nuclear export machinery, and vRNPs are transported to the cytoplasm through nuclear pore complexes in an NEP-dependent manner (33, 37). Following nuclear export, vRNPs initially accumulate near the microtubule-organizing center (MTOC) and subsequently may associate with microtubules during transit to the plasma membrane (31). A previous study indicates that vRNPs associate with actin microfilaments (4); however, disruption of actin polymerization in infected cells does not impair influenza virus replication (3, 19), suggesting that actin is not essential for vRNP transport. Besides the potential association between vRNPs and cytoskeletal elements, the mechanism of vRNP movement from the MTOC through the cytoplasm and to the budding site is completely unknown.
A recent report indicated an important role for the cellular Rab11 GTPase in the budding of influenza virus virions from the plasma membrane (7). In this study, influenza virus NP colocalized with endogenous Rab11 or an overexpressed, constitutively active Rab11 mutant in influenza virus-infected 293T cells. Rab11 GTPase localizes to the pericentriolar recycling endosome compartment, which is closely associated with the MTOC, and has a well-established role in mediating vesicular trafficking toward the plasma membrane. We therefore hypothesized that Rab11 may be involved in transporting vRNPs to budding sites. Here, we developed a system to monitor vRNP trafficking in infected cells to test this hypothesis. Our results reveal that Rab11 is an essential host factor involved in the transport of influenza vRNPs from the MTOC to the plasma membrane.
MATERIALS AND METHODS
Cells, viruses, and infections.
A549, 293T, and Madin-Darby canine kidney (MDCK) cells were propagated in a 1:1 mix of Dulbecco's modified Eagle's medium (DMEM) and Ham's F12 medium containing 10% fetal bovine serum (FBS), in DMEM with 10% FBS, or in minimum essential medium (MEM) containing 5% newborn calf serum (NCS), respectively. All cell cultures were supplemented with penicillin-streptomycin (Invitrogen, Carlsbad, CA) and maintained at 37°C in an atmosphere of 5% CO2. Influenza A/WSN/1933 (H1N1) (referred to as WSN) virus was rescued in 293T cells using plasmid-based reverse genetics, as previously described (34). For immunofluorescence and immunoprecipitation studies, A549 cells were infected with WSN virus at a multiplicity of infection (MOI) of 3 PFU per cell and harvested at the times indicated on the figures. To determine the effects of specific small interfering RNAs (siRNAs) on multicycle influenza virus replication, siRNA-treated A549 cells grown in 24-well dishes were infected with 100 PFU of WSN virus. Supernatants were harvested 48 h after infection, and the amount of virus in the supernatants was quantified by plaque assay in MDCK cells.
FISH, immunofluorescence, and microscopy.
Probes for fluorescence in situ hybridization (FISH) were generated using a digoxigenin (DIG) RNA labeling kit (Roche, Indianapolis, IN). Briefly, approximately 300 bp of the WSN virus positive-sense PB2 genome segment was PCR amplified from pPol1-PB2 (34), and products were purified using a QIAquick PCR purification kit (Qiagen, Valencia, CA). One microgram of the purified PCR product was added to an RNA labeling mixture consisting of 5 μl of transcription buffer (10×; Roche), 5 μl of RNA labeling mix (10×;10 mM ATP, CTP, and GTP; 6.5 mM UTP; and 3.5 mM DIG-11-UTP [Roche]), and 5 μl of T7 enzyme mix (Promega, Madison, WI) in a final volume of 50 μl and incubated for 2 h at 37°C. Labeled RNA probes were subsequently treated with 5 units of RQ1 RNase-Free DNase I (Promega) for 15 min at 37°C. The primer sequences used for the amplification of the positive-sense PB2 probe are as follows: T7_WSNseg1_1021F, GGATCCTAATACGACTCACTATAGGGAGAACAAGCGGATCATCAGTCAA; WSNseg1_1320R, CATGGGATTCAATCGCTGATTCGCC.
For coimmunofluorescence and FISH analysis, influenza virus-infected MDCK cells grown on 35-mm glass-bottomed dishes (diameter of glass coverslip, 12 mm; Iwaki, Chiba, Japan) were fixed with 4% paraformaldehyde in 0.1 M phosphate-buffer (pH 7.4) for 15 min and permeabilized with 0.5% Triton X-100 for 5 min at room temperature. Indirect immunofluorescence staining of NP was performed before probe hybridization by incubating cells with the mouse monoclonal anti-WSN virus NP antibody 3/1 (1:1,000 dilution; a kind gift from Robert Webster, St. Jude Children's Research Hospital, Memphis, TN) in 1% bovine serum albumin (BSA) in phosphate-buffered saline (PBS) for 40 min at 37°C, followed by goat anti-mouse conjugated with Alexa-Fluor (AF) 594 (1:250; Invitrogen) for 40 min at 37°C. To prevent RNA degradation, 1 U/μl of RNasin Plus (Promega) was added to the antibody solutions. Subsequently, cells were sequentially washed with 2× SSC (1× SSC is 150 mM NaCl and 15 mM sodium citrate) and 0.1× SSC, incubated in 95% formamide in 0.1× SSC for 15 min at 65°C, and immediately chilled on ice. Cells were then blocked with prehybridization buffer (50% formamide, 2× SSC, 5× Denhardt's solution, 20 μg/ml of salmon sperm DNA [sonicated to 300- to 700-bp pieces; BioDynamics Laboratory, Tokyo, Japan]) for 1 h at room temperature and then incubated with 200 ng/ml of the DIG-labeled PB2 segment RNA probe diluted in prehybridization buffer for 40 h at 50°C. After hybridization, cells were washed extensively in wash solution 1 consisting of 50% formamide and 2× SSC and wash solution 2 consisting of 0.1× SSC (three washes with each buffer for 20 min/wash at 60°C). Finally, cells were incubated in in situ hybridization blocking solution (Vector Laboratories, Burlingame, CA) for 30 min at room temperature, and probes were detected by indirect immunofluorescence using polyclonal sheep anti-DIG (2 μg/ml; catalog number 1333089; Roche) and AF 488 donkey anti-sheep (1:250; Invitrogen) antibodies. Nuclei were stained by incubation with 1 μg/ml Hoechst 33342 (Invitrogen).
For immunofluorescence studies without FISH, A549 cells grown on coverslips were fixed with 4% electron microscopy-grade paraformaldehyde (Electron Microscopy Sciences, Hatfield, PA) in phosphate-buffered saline for 15 min. Cells were permeabilized with 0.1% Triton X-100 for 5 min, and nonspecific protein binding was blocked by incubation with PBS containing 10% normal goat serum and 1% fraction V bovine serum albumin (blocking solution). Primary and secondary antibody dilutions were prepared in blocking solution and were incubated with coverslips for 16 h at 4°C and for 45 min at room temperature, respectively. Nuclei were stained by incubation with 0.4 μg/ml Hoechst 33258 (Invitrogen) for 30 min at room temperature, where indicated in figure legends. Extensive washing with PBS was performed between all staining steps. The primary antibodies used were monoclonal antibody (MAb) 3/1 (1:1,000) and polyclonal rabbit anti-RAB11A (1:100; catalog number 71-5300; Invitrogen). Secondary antibodies included AF 546 goat anti-mouse and AF 488 goat anti-rabbit (1:1,000; Invitrogen). Stained coverslips were mounted to glass slides using Immu-Mount (Shandon, Pittsburgh, PA).
All fluorescence images were captured with a Zeiss LSM 510 Meta point-scan laser confocal microscope system using argon/2, HeNe543, and diode lasers and either a 63× LCI Plan NeoFluar objective for coimmunofluorescence and FISH analysis or a 40× Enhanced Contrast Plan NeoFluar objective for indirect immunofluorescence alone. Both objectives have numerical apertures of 1.3. Individual fluorescence channels were acquired independently at a frame size of 1,024 by 1,024 pixels, with additional zooming where indicated. Raw images were exported as TIFF files and cropped as necessary by using Adobe Photoshop CS4 but were otherwise unaltered.
siRNAs, plasmids, and transfections.
The following siRNAs were used: a highly validated nontargeting siRNA known to be loaded into the RNA-induced silencing complex (AllStars Neg, catalog number 1027281; Qiagen), a previously described siRNA targeting influenza virus NP mRNA (NP-1496; synthesized by Qiagen) (17), and an siRNA targeting RAB11A (catalog number SI04437881; Qiagen). For siRNA transfections, A549 cells were seeded into 24-well plates (5 × 104 cells/well), and 2 h later, each well was transfected with 20 nM siRNA (final concentration) and 4.5 μl of HiPerfect transfection reagent (Qiagen), according to the manufacturer's recommendations. Cells were incubated with transfection complexes for 48 h to allow protein knockdown before additional manipulations were performed.
To generate RAB11A dominant negative and constitutively active mutants, full-length human RAB11A cDNA was obtained from OriGene (Rockville, MD). Mutations were directly introduced using oligonucleotide primers encoding a mutation of serine 25 to asparagine (i.e., S25N) or glutamine 70 to leucine (i.e., Q70L) using site-directed mutagenesis (Stratagene, La Jolla, CA). Resultant full-length RAB11A mutants were amplified by PCR using 5′ BglII and 3′ EcoRI overhangs and inserted C-terminally in frame with enhanced green fluorescent protein (EGFP) in the pEGFP-C1 vector (Clontech, Mountain View, CA), resulting in pEGFP-RAB11A-Q70L and pEGFP-RAB11A-S25N. The presence of the appropriate mutations was confirmed by sequencing. Primer sequences are available upon request. A549 cells were transfected by electroporation with a Neon Transfection System (Invitrogen) 24 h before superinfection with WSN virus. One microgram of each plasmid was used per 2 × 105 cells, and electroporations were performed using two pulses (1200 V, 30 ms). Electroporated cells were immediately transferred to prewarmed medium that lacked antibiotics.
Immunoprecipitation.
For immunoprecipitation studies, A549 cells were seeded in 60-mm dishes and 16 h later were mock infected or infected with WSN virus. At the times indicated on the figure, cells were harvested by scraping them into ice-cold PBS, pelleted by centrifugation at 1,800 × g, and stored at −80°C until further processing. All pellets were thawed together, dispersed in TNE lysis buffer (10 mM Tris-HCl, pH 7.5, 150 mM NaCl, 1 mM EDTA, 1% Igepal CA-630) containing a protease inhibitor cocktail (Complete Mini; Roche), and lysed for 30 min on ice. Insoluble material was removed by centrifugation at 14,000 × g for 1 h at 4°C. Cleared lysates were divided into two aliquots and incubated with antibody-conjugated Dynabeads (Invitrogen), prepared according to the manufacturer's instructions, for 30 min at room temperature. One aliquot was incubated with an RAB11A-specific antibody (2 μg; Invitrogen), and the other aliquot was incubated with an equivalent amount of normal rabbit IgG (Santa Cruz Biotechnology, Santa Cruz, CA). Resultant immunocomplexes were washed three times in PBS and released from the Dynabeads by incubation in SDS-PAGE sample buffer (160 mM Tris-HCl, pH 6.8, 4% SDS, 20% glycerol, 10% 2-mercaptoethanol, and 0.04% bromophenol blue) for 5 min at 95°C.
Immunoblotting.
SDS-PAGE was performed using a Criterion system and 4% to 20% gradient gels (Bio-Rad, Hercules, CA), according to the manufacturer's recommendations. Proteins were transferred to nitrocellulose membranes using an iBlot Dry Blotting System (Invitrogen). Following transfer, membranes were immediately placed in 5% milk in PBS-Tween (PBS-T; PBS and 0.5% Tween 20) and blocked for 1 h at room temperature. Primary and secondary antibodies were diluted in 3% milk in PBS-T, and incubations were performed at 4°C for 16 h and at room temperature for 1 h, respectively. Membranes were washed extensively with PBS-T between each incubation step. To detect influenza virus NP, we used mouse MAb clone 5/1 (1:2,000; a kind gift from Robert Webster). RAB11A in siRNA-treated cells was detected with rabbit anti-RAB11A (1:500; Invitrogen), and protein loading was assessed with rabbit anti-calnexin (CNX) (1:5,000; catalog number 11397; Santa Cruz Biotechnology). To minimize background recognition of IgG in immunoblots of immunoprecipitation reactions, we used an alternate RAB11A monoclonal antibody (1:500; catalog number ab78337; Abcam, Cambridge, MA). GFP-tagged RAB11A fusions were detected with mouse anti-GFP antibody (1:20,000; catalog number 632375; Clontech). Horseradish peroxidase-conjugated secondary antibodies included goat anti-rabbit (1:2,000; catalog number G21040; Invitrogen) and goat anti-mouse (1:2,000; catalog number 31430; Thermo Scientific, Rockford, IL). Proteins were detected using Lumi-Light PLUS Western blotting substrate (Roche), Kodak Biomax XAR film, and a Konica Minolta SRX-101A X-ray film developer.
RESULTS
A system for assessing genome transport in influenza virus-infected cells.
Although colocalization between influenza virus NP and endogenous Rab11 was previously observed (7), it is not clear whether the NP recognized in these assays represents authentic vRNP complexes containing genomic RNA. Moreover, no information about the temporal relationship between Rab11 and NP was reported. Since little is known about the mechanism of vRNP transport to the plasma membrane and since Rab11 vesicles could represent a pathway for vRNP delivery to assembling virions, we considered it essential to determine whether vRNPs associate with Rab11 in infected cells. We, therefore, established a system to study the spatiotemporal distribution of influenza virus vRNPs following infection.
vRNPs exhibit a punctate cytoplasmic staining pattern following export from the nucleus that is detectable with a specific monoclonal antibody (MAb) (22, 31). We screened several in-house antibodies and identified one, raised against influenza WSN virus NP (MAb clone 3/1), that recognized vRNP-like puncta in the cytoplasm of infected cells. To determine whether this antibody could recognize authentic vRNPs, we performed a combined immunofluorescence/FISH analysis of WSN virus-infected MDCK cells with MAb 3/1 and a vRNA-specific probe. We found that the MAb 3/1 signal almost completely overlapped with that of the vRNA, exhibiting exclusive localization to the nucleus at early time points postinfection, accumulation in punctate foci in a juxtanuclear structure after exit from the nucleus, and dispersal throughout the cytoplasm and near the plasma membrane at later time points (Fig. 1). This distribution pattern is consistent with that previously reported for influenza virus vRNPs (22, 31). Moreover, nearly identical MAb 3/1 staining patterns were observed in 293, A549, and HeLa cells infected with WSN virus and after infection with influenza A/Kawasaki/173/2001 (H1N1) or influenza A/Puerto Rico/8/1934 (H1N1) virus (data not shown). This argues against a cell-type- or virus-specific phenotype and suggests that MAb 3/1 recognizes bona fide influenza virus vRNP complexes.
Fig. 1.
Monoclonal antibody 3/1 recognizes vRNP in the cytoplasm of influenza virus-infected cells. MDCK cells grown on coverslips were infected with WSN virus (MOI of 3) and fixed at 3, 5, and 7 hpi before being subjected to coimmunofluorescence and FISH staining. MAb 3/1, which recognizes influenza virus NP, is indicated in red, and the PB2 negative-sense vRNA is shown in green. (A) Individual staining profiles are shown for NP and vRNA, together with a merged image, for each time point. Merged images show Hoechst nuclear stains. (B) Enlarged portions of the boxed sections in merged frames of panel A, highlighting colocalization of vRNA and NP.
RAB11A associates with influenza virus genomes en route to the plasma membrane.
We next utilized MAb 3/1 to assess vRNP trafficking and association with Rab11 in a time course infection of human lung epithelial cells (A549). Here, it should be noted that influenza virus replication in A549 cells is slower than what is observed in MDCK cells, leading to some cell-type-specific differences in the kinetics of influenza virus vRNP nuclear export (compare Fig. 1 and 2). Despite these differences, the overall pattern of vRNP nuclear and cytoplasmic distributions is similar between the two cell types. The cellular pool of Rab11 consists of two closely related but unique isoforms, RAB11A and RAB11B. For the following studies, we focused on RAB11A, which could be abundantly detected in A549 cells.
Fig. 2.
Cytoplasmic vRNPs colocalize with RAB11A during transport from the MTOC to the plasma membrane. A549 cells grown on coverslips were mock infected or infected with WSN virus and stained with MAb 3/1 (vRNP; red) and a polyclonal rabbit antibody against RAB11A (green). Mock-infected cells were counter-stained with Hoechst 33258 to identify nuclei (blue). Representative spatiotemporal distribution patterns are shown for each staining condition at each time point. For each panel, frame i depicts merged images of vRNP, RAB11A, and Hoechst-stained nuclei, as indicated. Arrows point to the plasma membrane boundaries; N indicates the nucleus. Individual vRNP and RAB11A staining profiles are shown in frames ii and iii, respectively. Enlarged (3×) images of individual and merged vRNP and RAB11A distributions from the boxed regions in frames i are shown in the in frames iv for vRNP, v for RAB11A, and vi for merged images. The time point is indicated at the top of each panel, and a staining key is shown in the box in the bottom right corner. To facilitate observation of the details of vRNP and RAB11A localization, original images were acquired with a 40× objective and a 3× zoom. All images are representative of several independent experiments.
RAB11A staining in mock-infected cells was dominated by accumulation in a juxtanuclear region, with fine punctate staining in the peripheral cytoplasm (Fig. 2A). The juxtanuclear staining pattern is associated with the MTOC (47), the trans-Golgi network (13, 48), and the apical recycling compartment (10); here, we will refer to this region as the MTOC. At 5 h postinfection (hpi), when vRNP staining was restricted to the nucleus, RAB11A distribution was similar to that of mock-infected cells (Fig. 2B). At 7 hpi, vRNPs, which initially accumulate at the MTOC upon nuclear export (31), overlapped with the RAB11A staining at the MTOC and completely colocalized with RAB11A in punctate foci emanating from the MTOC into the peripheral cytoplasmic region (Fig. 2C). Interestingly, RAB11A accumulation in peripheral structures was more prominent than that observed in uninfected cells, suggesting specific recruitment (compare frames v in Fig. 2A and C). Extensive colocalization between vRNPs and RAB11A persisted at 9 hpi, and vRNPs and RAB11A accumulated together in structures adjacent to the plasma membrane, which were larger than the trafficking intermediates observed at earlier time points (Fig. 2D; compare frame v with the corresponding frames in A and C). Finally, by 11 hpi, vRNPs prominently localized to the plasma membrane, while RAB11A remained essentially intracellular and no longer colocalized with vRNPs (Fig. 2E). These observations clearly demonstrate an intimate, spatiotemporal relationship between vRNPs and RAB11A during vRNP delivery to the plasma membrane and strongly imply a role for RAB11A in vRNP cytoplasmic trafficking in late-stage influenza virus-infected cells.
Perturbation of RAB11A expression or function impairs influenza virus RNP transport.
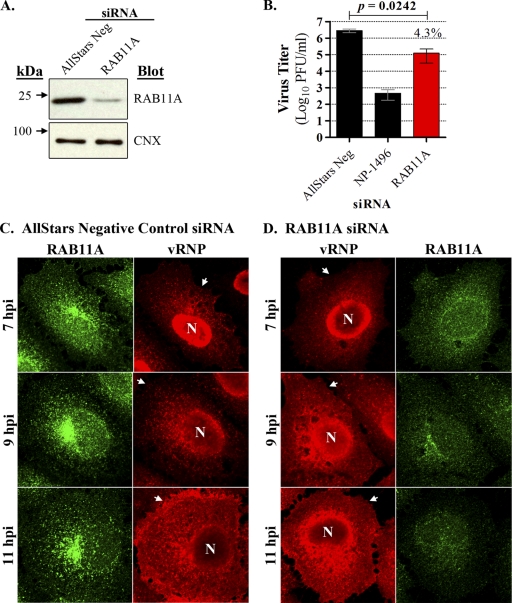
To address the hypothesis that RAB11A is involved in vRNP cytoplasmic transport, we used two approaches to perturb RAB11A function in A549 cells: siRNA to downregulate protein expression or overexpression of dominant negative or constitutively active mutants to disrupt endogenous Rab11 function. Initial siRNA experiments indicated that RAB11A-specific siRNA suppressed RAB11A expression in A549 cells without a concomitant reduction in the level of a nontargeted cellular protein, calnexin (CNX) (Fig. 3A). Further, RAB11A siRNA-treated cells infected with WSN virus exhibited a 23-fold reduction in virus titer relative to nontargeting siRNA-treated control cells after multicycle replication (Fig. 3B). These results are consistent with previous observations (7). We next used the same transfection conditions to repress RAB11A protein expression and then infected A549 cells with WSN virus and assessed vRNP trafficking at the single-cell level, using an immunofluorescence time course with MAb 3/1. Since RAB11A knockdown was incomplete (Fig. 3A), we also costained the cells with Rab11-specific antibodies to evaluate RAB11A expression levels after siRNA treatment. Using this approach, we identified differences in vRNP distribution between nontargeting siRNA-treated control cells and cells expressing only low levels of RAB11A by 9 hpi and observed even greater disparity by 11 hpi (Fig. 3C and D). Specifically, control cells exhibited vRNP accumulation at the MTOC and punctate vRNP foci throughout the cytoplasm and near the plasma membrane at 9 hpi, while at 11 hpi heavy vRNP accumulation was observed at or near the plasma membrane. In contrast, RAB11A siRNA-treated cells exhibited aberrant accumulation of vRNPs adjacent to the nucleus, with occasional punctate vRNP foci in the peripheral cytoplasm at both time points. Importantly, little or no vRNPs accumulated at the plasma membrane at 11 hpi in RAB11A siRNA-treated cells. Therefore, in cells with significantly reduced RAB11A expression, vRNP transport to the plasma membrane appears to be defective.
Fig. 3.
RAB11A knockdown impairs vRNP transport in influenza virus-infected cells. (A) A549 cells were transfected with nontargeting siRNA (AllStars Neg) or siRNA targeting RAB11A, and cell lysates were subjected to immunoblot analysis 48 h after transfection. siRNA treatments are indicated at the top, molecular mass standards are shown to the left, and detected proteins are indicated to the right of each panel. (B) Parallel A549 cultures were transfected with AllStars Neg siRNA, siRNA targeting influenza virus NP mRNA (NP-1496), or siRNA targeting RAB11A, and after 48 h cultures were directly inoculated with WSN virus. Supernatants were harvested at 48 h postinfection, and titers were determined by plaque assay in MDCK cells. Titers for each condition are represented by an average of triplicate infections, and variation is indicated by standard deviation. A paired Student's t test was performed to compare the AllStars Neg siRNA treatment with that of RAB11A siRNA, and the resultant P value is indicated at the top. The percentage of replication observed in RAB11A siRNA-transfected cells relative to that in cells transfected with the AllStars Neg control is indicated above the RAB11A bar. (C and D) A549 cells grown on coverslips were transfected with AllStars Neg siRNA or siRNA targeting RAB11A and then infected with WSN virus for vRNP trafficking analysis. Panel C shows the vRNP (red) and RAB11A (green) staining profiles for nontargeting siRNA while panel D shows the same for siRNA targeting RAB11A. Time points are indicated to the left. Arrows indicate the plasma membrane boundaries. Data are representative of two independent experiments. N, nucleus.
To further support this conclusion, we also assessed the effects of overexpression of dominant negative or constitutively active RAB11A-GFP fusion proteins on vRNP trafficking. The RAB11A dominant negative mutant possesses an S25N mutation, exists preferentially in the GDP-bound state (47), and exhibits predominantly juxtanuclear distribution in uninfected A549 cells (Fig. 4A); the constitutively active mutant possesses a Q70L mutation, exists preferentially in the GTP-bound state (47), and exhibits localization similar to that seen with endogenous RAB11A (compare Fig. 4A and 2A). Both mutants are known to disrupt cellular Rab11-dependent trafficking (30). A549 cells that expressed the constitutively active mutant exhibited nearly complete retention of vRNPs in perinuclear structures that completely overlapped with the constitutively active mutant, and almost no vRNPs were observed near the plasma membrane (Fig. 4C). This was in stark contrast with vRNP distribution and trafficking in cells expressing GFP alone, where vRNPs were observed en route to and accumulating at the plasma membrane (Fig. 4B). The dominant negative RAB11A mutant also disrupted vRNP trafficking, inducing robust accumulation in the perinuclear region by 12 hpi (Fig. 4D). Notably, vRNP distribution in dominant negative RAB11A-expressing cells differed from that observed with constitutively active RAB11A overexpression, more closely resembling vRNP distribution in cells treated with RAB11A siRNA (compare vRNP profiles from Fig. 4D with vRNP profiles at 9 and 11 hpi in Fig. 3C and D). Interestingly, the dominant negative mutant exhibited localization that was nearly entirely exclusive of vRNPs, whereas the constitutively active mutant exclusively localized with vRNPs, suggesting that vRNPs may preferentially associate with GTP-loaded RAB11A.
Fig. 4.
RAB11A mutant proteins disrupt vRNP trafficking during the late-stage of influenza virus infection. (A) RAB11A GFP fusion protein behavior in plasmid-transfected, uninfected cells. The upper left portion of this panel shows immunoblots of the GFP fusion proteins. The specific overexpressed protein is indicated above each lane and molecular mass markers (kDa) are indicated to the left. Images show the localization patterns for GFP alone, GFP-RAB11A-Q70L (constitutively active), and GFP-RAB11A-S25N (dominant negative), as indicated on the figure, in A549 cells. Cells were counterstained with Hoechst 33258. (B to D) vRNP distribution in WSN virus-infected A549 cells expressing RAB11A GFP fusion proteins. A549 cells were transfected with plasmids expressing GFP alone (B), GFP-RAB11A-Q70L (C), or GFP-RAB11A-S25N (D) and were subsequently superinfected with WSN virus. vRNP localization was assessed at 12 or 21 h after infection using immunofluorescence staining with monoclonal antibody 3/1. Individual GFP (green) and vRNP (red) profiles are shown for all conditions, and merged GFP/vRNP profiles are shown for GFP-RAB11A-Q70L and GFP-RAB11A-S25N transfections. Arrows point to the plasma membrane boundaries. Data are representative of two independent experiments. N, nucleus.
Biochemical validation of NP-RAB11A complex formation in influenza virus-infected cells.
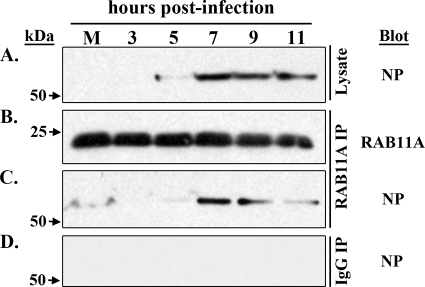
The association between RAB11A and vRNP in A549 cells was further assessed by using coimmunoprecipitation analysis at different times after WSN virus infection. Cell lysates derived from mock-infected or infected cells were immunoprecipitated with antibodies specific for RAB11A or with normal rabbit IgG (control), and immunoprecipitates were assayed for the presence of NP by immunoblotting. NP expression was detected in cell lysates by 5 hpi, and NP accumulation increased at all subsequent time points (Fig. 5A). Similar amounts of RAB11A protein were efficiently precipitated from mock-infected and infected cells at all time points (Fig. 5B), indicating that RAB11A expression was not affected by influenza virus infection. Significant levels of NP coimmunoprecipitated with RAB11A at 7 and 9 hpi (Fig. 5C), consistent with times when prominent cytoplasmic colocalization was observed at the single-cell level (Fig. 2). NP coprecipitated slightly less efficiently with RAB11A at 11 hpi. Importantly, we did not observe any nonspecific NP coimmunoprecipitation in control reaction mixtures containing normal rabbit IgG (Fig. 5D). These results indicate an association between influenza virus NP and RAB11A, which occurs in parallel with vRNP and RAB11A colocalization in the cytoplasm of infected cells. Based on these findings, we suggest that vRNPs physically associate with RAB11A-containing vesicles during transport to the plasma membrane.
Fig. 5.
NP and RAB11 form a complex in infected cells. A549 cells were mock infected or infected with WSN virus and then immunoprecipitated with antibodies against RAB11A or with normal rabbit IgG (control). Cell lysates or immunoprecipitates were subjected to immunoblot analysis as follows: total soluble influenza virus nucleoprotein (A), immunoprecipitated RAB11A (B), coimmunoprecipitated NP (C), and control IgG immunoprecipitates stained with antibodies against influenza virus NP (D). Time points are indicated at the top of the panel, and molecular mass standards are indicated to the left. M, mock infection.
DISCUSSION
Although many aspects of influenza virus assembly have been well studied, the mechanisms of vRNP transport through the cytoplasm to plasma membrane sites of virion budding remain largely mysterious. Here, we identified a critical, noncytoskeletal host protein with an essential function in influenza vRNP trafficking: the Rab11 GTPase. RAB11A colocalized with vRNP in transit to the plasma membrane, and disruption of RAB11A protein expression or function impaired vRNP transport from the MTOC. When considered with previous reports of a role for Rab11 in delivering M2 to the plasma membrane and in virion membrane scission and release (7, 40), our results suggest an integral, multifactorial role for Rab11 in the influenza virus life cycle.
Upon export from the nucleus, vRNPs initially accumulated at the MTOC and were observed in punctate foci in the peripheral cytoplasm. At later times after infection (e.g., 9 hpi), vRNP foci accumulated in larger structures adjacent to the plasma membrane, suggesting the possible coalescence of multiple smaller transport intermediates. Still later in the infection process (i.e., at 11 hpi), vRNPs were transferred to the plasma membrane, as indicated by the loss of cytoplasmic puncta and enhanced plasma membrane localization. RAB11A was tightly associated with vRNPs at the MTOC, in punctate cytoplasmic foci, and in regions of vRNP coalescence near the plasma membrane but did not accumulate with vRNPs at the plasma membrane. These observations suggest that RAB11A is involved in vRNP delivery to staging areas prior to vRNP incorporation into budding virions and that the vRNP-RAB11A interaction is released before the vRNPs are incorporated into virus particles. Consistent with this notion, RAB11A was not among the cellular proteins detected in purified virions in a previous study (44). Although vRNP puncta have been observed previously en route to the plasma membrane (22, 31), the coalescence of vRNP puncta near the plasma membrane has not been described. With this new finding, we can now hypothesize that vRNP cytoplasmic trafficking consists of at least four steps: (i) accumulation at the MTOC following nuclear export, (ii) association with RAB11A and release from the MTOC for cytoplasmic transport, (iii) coalescence in a staging area near the plasma membrane, and (iv) selection for incorporation into budding virions (Fig. 6).
Fig. 6.
Overview of influenza virus vRNP cytoplasmic trafficking. Our data suggest a four-step influenza virus vRNP trafficking process that includes the following: vRNP (indicated by the green coils) colocalization with Rab11-positive recycling endosomes (RE; shown in light green) following nuclear export (A); vRNP association with Rab11 vesicles (light orange, with green coils) and release from the perinuclear region for transport toward the plasma membrane (B); merging of Rab11/vRNP transport intermediates near the plasma membrane (light purple, with green coils) (C); and (D) vRNP selection for incorporation into budding virions. Viral glycoproteins at the plasma membrane are indicated in coral, yellow, and blue. Following vRNP selection for incorporation into budding virions, Rab11-positive vesicles may recycle back to the MTOC.
The accumulation of vRNPs in the putative staging area near the plasma membrane could have implications for the formation of influenza virus virions containing a complement of eight unique genomic segments. We previously reported that virions that bud from infected cells generally contain no more than eight individual genome segments (35). In addition, segment-specific packaging signals have been identified for all eight vRNA segments (15, 16, 18, 20, 21, 25–27, 32, 38, 39, 50), and hierarchical incorporation of those segments into budding virions has been observed (27, 32). These findings imply that vRNP-vRNP interactions may be responsible for the association between individual genome segments; however, the mechanism by which this occurs is not clear. One recent report suggests that individual genome segments may associate with each other in the cytoplasm after nuclear export (45). It may be that the putative staging area adjacent to the plasma membrane is the site at which influenza virus genome segments interact. Additional experiments to investigate the trafficking dynamics of individual vRNPs and their temporal association with staging areas are needed.
Influenza vRNPs preferentially associated with the predominantly GTP-loaded, constitutively active RAB11A mutant and did not colocalize with the GDP-bound dominant negative version. The GTP-bound forms of Rab GTPases are bound to the cytosolic surface of membranes and associate with specific effector proteins involved in vesicle formation, transport, docking, and fusion; GDP-bound forms, on the other hand, may be dissociated from the membrane (43). Therefore, our data imply that vRNPs associate with GTP-loaded RAB11A at membrane structures. Given the lack of association between vRNPs and GDP-loaded dominant negative RAB11A and the aberrant accumulation of vRNPs with constitutively active RAB11A in enlarged structures in the perinuclear region, we further suggest that GTP hydrolysis may be required for the release of RAB11A-vRNP complexes from the MTOC and that an active GTP hydrolysis and GDP-GTP exchange cycle may be required for RAB11A-vRNP complex transport to the plasma membrane.
How do influenza vRNPs associate with the Rab11 vesicular compartment? One possibility is that the vRNPs interact directly with Rab11 or one of its effectors. However, vRNPs may associate with M1 in the cytoplasm of infected cells since M1 can prevent reimport of nuclear exported vRNPs (28). Given the intrinsic membrane-binding properties of M1, we suggest that vRNPs could associate with RAB11A via M1 for transport to the plasma membrane. Alternatively, since Rab11 is important for the cell surface delivery of M2 (40) and since the M2 cytoplasmic tail likely interacts with vRNPs (29), vRNP association with Rab11 transport vesicles may involve M2. Additional experiments are needed to dissect the specific mechanism that regulates vRNP association with the Rab11 compartment.
In summary, our work has identified an essential, noncytoskeletal host cofactor involved in the transport of influenza vRNPs to plasma membrane sites of virion formation. The Rab11 pathway previously has been implicated in the budding of respiratory syncytial virus from the apical plasma membrane of infected cells (6, 49). Moreover, Rab11 has been reported to colocalize with Sendai virus (SeV) vRNPs (11) although the requirement for Rab11 expression and function in their transport has not been explored. Here, we reveal an integral role for Rab11 in RNA virus genome transport in the cytoplasm. Thus, we have not only further clarified the mechanisms of influenza virus assembly but also uncovered a potentially novel cytoplasmic trafficking mechanism for ribonucleoprotein complexes.
While this work was in preparation, Amorim et al. (2) reported that influenza virus NP and vRNA colocalize with Rab11 during influenza virus infection and that siRNA treatment with RAB11A and RAB11B in combination altered vRNA and NP cytoplasmic staining at late times after infection. In support of the data presented here, they also reported that the interaction between influenza virus vRNP proteins and a Rab11 constitutively active mutant was 10-fold more efficient than interaction with a dominant negative mutant. This study thus corroborates our hypothesis that vRNPs preferentially interact with Rab11 in its GTP-bound form.
ACKNOWLEDGMENTS
We thank Susan Watson for editing the manuscript and Yuko Kawaoka for the graphic illustration.
This work was supported by the National Institute of Allergy and Infectious Diseases, Public Health Service research grants, by ERATO (Japan Science and Technology Agency), by a grant-in-aid for Specially Promoted Research from the Ministries of Education, Culture, Sports, Science, and Technology, and by grants-in-aid from Health, Labor, and Welfare of Japan.
Footnotes
Published ahead of print on 27 April 2011.
REFERENCES
- 1. Ali A., Avalos R. T., Ponimaskin E., Nayak D. P. 2000. Influenza virus assembly: effect of influenza virus glycoproteins on the membrane association of M1 protein. J. Virol. 74:8709–8719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Amorim M. J., et al. 9 February 2011. A Rab11 and microtubule dependent mechanism for cytoplasmic transport of influenza A virus vRNA. J. Virol. doi:10.1128/JVI.02606-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Arcangeletti M. C., et al. 1997. Modification of cytoskeleton and prosome networks in relation to protein synthesis in influenza A virus-infected LLC-MK2 cells. Virus Res. 51:19–34 [DOI] [PubMed] [Google Scholar]
- 4. Avalos R. T., Yu Z., Nayak D. P. 1997. Association of influenza virus NP and M1 proteins with cellular cytoskeletal elements in influenza virus-infected cells. J. Virol. 71:2947–2958 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Barman S., et al. 2004. Role of transmembrane domain and cytoplasmic tail amino acid sequences of influenza a virus neuraminidase in raft association and virus budding. J. Virol. 78:5258–5269 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Brock S. C., Goldenring J. R., Crowe J. E., Jr 2003. Apical recycling systems regulate directional budding of respiratory syncytial virus from polarized epithelial cells. Proc. Natl. Acad. Sci. U. S. A. 100:15143–15148 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Bruce E. A., Digard P., Stuart A. D. 2010. The Rab11 pathway is required for influenza A virus budding and filament formation. J. Virol. 84:5848–5859 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Bui M., Whittaker G., Helenius A. 1996. Effect of M1 protein and low pH on nuclear transport of influenza virus ribonucleoproteins. J. Virol. 70:8391–8401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Bui M., Wills E. G., Helenius A., Whittaker G. R. 2000. Role of the influenza virus M1 protein in nuclear export of viral ribonucleoproteins. J. Virol. 74:1781–1786 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Casanova J. E., et al. 1999. Association of Rab25 and Rab11a with the apical recycling system of polarized Madin-Darby canine kidney cells. Mol. Biol. Cell 10:47–61 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Chambers R., Takimoto T. 2010. Trafficking of Sendai virus nucleocapsids is mediated by intracellular vesicles. PLoS One 5:e10994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Chen B. J., Leser G. P., Jackson D., Lamb R. A. 2008. The influenza virus M2 protein cytoplasmic tail interacts with the M1 protein and influences virus assembly at the site of virus budding. J. Virol. 82:10059–10070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Chen W., Feng Y., Chen D., Wandinger-Ness A. 1998. Rab11 is required for trans-Golgi network-to-plasma membrane transport and a preferential target for GDP dissociation inhibitor. Mol. Biol. Cell 9:3241–3257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Enami M., Enami K. 1996. Influenza virus hemagglutinin and neuraminidase glycoproteins stimulate the membrane association of the matrix protein. J. Virol. 70:6653–6657 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Fujii K., et al. 2005. Importance of both the coding and the segment-specific noncoding regions of the influenza A virus NS segment for its efficient incorporation into virions. J. Virol. 79:3766–3774 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Fujii Y., Goto H., Watanabe T., Yoshida T., Kawaoka Y. 2003. Selective incorporation of influenza virus RNA segments into virions. Proc. Natl. Acad. Sci. U. S. A. 100:2002–2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Ge Q., et al. 2003. RNA interference of influenza virus production by directly targeting mRNA for degradation and indirectly inhibiting all viral RNA transcription. Proc. Natl. Acad. Sci. U. S. A. 100:2718–2723 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Gog J. R., et al. 2007. Codon conservation in the influenza A virus genome defines RNA packaging signals. Nucleic Acids Res. 35:1897–1907 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Griffin J. A., Compans R. W. 1979. Effect of cytochalasin B on the maturation of enveloped viruses. J. Exp. Med. 150:379–391 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Hutchinson E. C., Curran M. D., Read E. K., Gog J. R., Digard P. 2008. Mutational analysis of cis-acting RNA signals in segment 7 of influenza A virus. J. Virol. 82:11869–11879 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Hutchinson E. C., Wise H. M., Kudryavtseva K., Curran M. D., Digard P. 2009. Characterisation of influenza A viruses with mutations in segment 5 packaging signals. Vaccine 27:6270–6275 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Jo S., et al. 2010. Involvement of vesicular trafficking system in membrane targeting of the progeny influenza virus genome. Microbes Infect. 12:1079–1084 [DOI] [PubMed] [Google Scholar]
- 23. Kretzschmar E., Bui M., Rose J. K. 1996. Membrane association of influenza virus matrix protein does not require specific hydrophobic domains or the viral glycoproteins. Virology 220:37–45 [DOI] [PubMed] [Google Scholar]
- 24. Leser G. P., Lamb R. A. 2005. Influenza virus assembly and budding in raft-derived microdomains: a quantitative analysis of the surface distribution of HA, NA and M2 proteins. Virology 342:215–227 [DOI] [PubMed] [Google Scholar]
- 25. Liang Y., Hong Y., Parslow T. G. 2005. cis-Acting packaging signals in the influenza virus PB1, PB2, and PA genomic RNA segments. J. Virol. 79:10348–10355 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Liang Y., Huang T., Ly H., Parslow T. G. 2008. Mutational analyses of packaging signals in influenza virus PA, PB1, and PB2 genomic RNA segments. J. Virol. 82:229–236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Marsh G. A., Rabadan R., Levine A. J., Palese P. 2008. Highly conserved regions of influenza a virus polymerase gene segments are critical for efficient viral RNA packaging. J. Virol. 82:2295–2304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Martin K., Helenius A. 1991. Nuclear transport of influenza virus ribonucleoproteins: the viral matrix protein (M1) promotes export and inhibits import. Cell 67:117–130 [DOI] [PubMed] [Google Scholar]
- 29. McCown M. F., Pekosz A. 2005. The influenza A virus M2 cytoplasmic tail is required for infectious virus production and efficient genome packaging. J. Virol. 79:3595–3605 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Mohrmann K., van der Sluijs P. 1999. Regulation of membrane transport through the endocytic pathway by rabGTPases. Mol. Membr. Biol. 16:81–87 [DOI] [PubMed] [Google Scholar]
- 31. Momose F., Kikuchi Y., Komase K., Morikawa Y. 2007. Visualization of microtubule-mediated transport of influenza viral progeny ribonucleoprotein. Microbes Infect. 9:1422–1433 [DOI] [PubMed] [Google Scholar]
- 32. Muramoto Y., et al. 2006. Hierarchy among viral RNA (vRNA) segments in their role in vRNA incorporation into influenza A virions. J. Virol. 80:2318–2325 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Neumann G., Hughes M. T., Kawaoka Y. 2000. Influenza A virus NS2 protein mediates vRNP nuclear export through NES-independent interaction with hCRM1. EMBO J. 19:6751–6758 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Neumann G., et al. 1999. Generation of influenza A viruses entirely from cloned cDNAs. Proc. Natl. Acad. Sci. U. S. A. 96:9345–9350 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Noda T., et al. 2006. Architecture of ribonucleoprotein complexes in influenza A virus particles. Nature 439:490–492 [DOI] [PubMed] [Google Scholar]
- 36. Noton S. L., et al. 2007. Identification of the domains of the influenza A virus M1 matrix protein required for NP binding, oligomerization and incorporation into virions. J. Gen. Virol. 88:2280–2290 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. O'Neill R. E., Talon J., Palese P. 1998. The influenza virus NEP (NS2 protein) mediates the nuclear export of viral ribonucleoproteins. EMBO J. 17:288–296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Ozawa M., et al. 2007. Contributions of two nuclear localization signals of influenza A virus nucleoprotein to viral replication. J. Virol. 81:30–41 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Ozawa M., et al. 2009. Nucleotide sequence requirements at the 5′ end of the influenza A virus M RNA segment for efficient virus replication. J. Virol. 83:3384–3388 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Rossman J. S., Jing X., Leser G. P., Lamb R. A. 2010. Influenza virus M2 protein mediates ESCRT-independent membrane scission. Cell 142:902–913 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Sakaguchi A., Hirayama E., Hiraki A., Ishida Y., Kim J. 2003. Nuclear export of influenza viral ribonucleoprotein is temperature-dependently inhibited by dissociation of viral matrix protein. Virology 306:244–253 [DOI] [PubMed] [Google Scholar]
- 42. Scheiffele P., Roth M. G., Simons K. 1997. Interaction of influenza virus haemagglutinin with sphingolipid-cholesterol membrane domains via its transmembrane domain. EMBO J. 16:5501–5508 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Seabra M. C., Wasmeier C. 2004. Controlling the location and activation of Rab GTPases. Curr. Opin. Cell Biol. 16:451–457 [DOI] [PubMed] [Google Scholar]
- 44. Shaw M. L., Stone K. L., Colangelo C. M., Gulcicek E. E., Palese P. 2008. Cellular proteins in influenza virus particles. PLoS Pathog. 4:e1000085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Takizawa N., Kumakura M., Takeuchi K., Kobayashi N., Nagata K. 2010. Sorting of influenza A virus RNA genome segments after nuclear export. Virology 401:248–256 [DOI] [PubMed] [Google Scholar]
- 46. Thaa B., Herrmann A., Veit M. 2009. The polybasic region is not essential for membrane binding of the matrix protein M1 of influenza virus. Virology 383:150–155 [DOI] [PubMed] [Google Scholar]
- 47. Ullrich O., Reinsch S., Urbe S., Zerial M., Parton R. G. 1996. Rab11 regulates recycling through the pericentriolar recycling endosome. J. Cell Biol. 135:913–924 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Urbe S., Huber L. A., Zerial M., Tooze S. A., Parton R. G. 1993. Rab11, a small GTPase associated with both constitutive and regulated secretory pathways in PC12 cells. FEBS Lett. 334:175–182 [DOI] [PubMed] [Google Scholar]
- 49. Utley T. J., et al. 2008. Respiratory syncytial virus uses a Vps4-independent budding mechanism controlled by Rab11-FIP2. Proc. Natl. Acad. Sci. U. S. A. 105:10209–10214 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Watanabe T., Watanabe S., Noda T., Fujii Y., Kawaoka Y. 2003. Exploitation of nucleic acid packaging signals to generate a novel influenza virus-based vector stably expressing two foreign genes. J. Virol. 77:10575–10583 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. World Health Organization 20 January 20 2011, posting date Global alert and response: cumulative number of confirmed human cases of avian influenza A/(H5N1) reported to WHO. World Health Organization, Geneva, Switzerland: http://www.who.int/csr/disease/avian_influenza/country/cases_table_2011_01_20/en/index.html [Google Scholar]
- 52. Zhang J., Lamb R. A. 1996. Characterization of the membrane association of the influenza virus matrix protein in living cells. Virology 225:255–266 [DOI] [PubMed] [Google Scholar]
- 53. Zhang J., Pekosz A., Lamb R. A. 2000. Influenza virus assembly and lipid raft microdomains: a role for the cytoplasmic tails of the spike glycoproteins. J. Virol. 74:4634–4644 [DOI] [PMC free article] [PubMed] [Google Scholar]