Abstract
Previously, we showed that rhinovirus (RV), which is responsible for the majority of common colds, disrupts airway epithelial barrier function, as evidenced by reduced transepithelial resistance (RT), dissociation of zona occludins 1 (ZO-1) from the tight junction complex, and bacterial transmigration across polarized cells. We also showed that RV replication is required for barrier function disruption. However, the underlying biochemical mechanisms are not known. In the present study, we found that a double-stranded RNA (dsRNA) mimetic, poly(I:C), induced tight junction breakdown and facilitated bacterial transmigration across polarized airway epithelial cells, similar to the case with RV. We also found that RV and poly(I:C) each stimulated Rac1 activation, reactive oxygen species (ROS) generation, and Rac1-dependent NADPH oxidase 1 (NOX1) activity. Inhibitors of Rac1 (NSC23766), NOX (diphenylene iodonium), and NOX1 (small interfering RNA [siRNA]) each blocked the disruptive effects of RV and poly(I:C) on RT, as well as the dissociation of ZO-1 and occludin from the tight junction complex. Finally, we found that Toll-like receptor 3 (TLR3) is not required for either poly(I:C)- or RV-induced reductions in RT. Based on these results, we concluded that Rac1-dependent NOX1 activity is required for RV- or poly(I:C)-induced ROS generation, which in turn disrupts the barrier function of polarized airway epithelia. Furthermore, these data suggest that dsRNA generated during RV replication is sufficient to disrupt barrier function.
INTRODUCTION
Tight junctions located at the apicolateral borders of adjacent airway epithelial cells contribute significantly to epithelial barrier function. Tight junctions regulate the selective passage of ions and solutes through the paracellular space and prevent paracellular migration of pathogens and their products from lumen to interstitium. Thus, perturbation of the barrier function may increase paracellular permeability, facilitate translocation of pathogens and their soluble products, and expose basolateral receptors.
Rhinovirus (RV), which is responsible for the majority of common colds (1), also provokes acute lower respiratory symptoms in healthy individuals (7, 18) and exacerbates airway diseases in patients with asthma, chronic obstructive pulmonary disease (COPD), and cystic fibrosis (9, 32, 47, 50). In addition to stimulating production of proinflammatory cytokines (13, 34), RV infection may also promote secondary bacterial infections by interfering with host innate defense mechanisms or by increasing the adherence of bacteria to the host mucosa (2, 20, 48). Recently, we and others demonstrated that RV infection compromises barrier function and facilitates bacterial transmigration across polarized airway epithelial cells (41, 54). Furthermore, we showed that infectious RV is required for the impairment of barrier function in polarized airway epithelia. The impairment of barrier function caused by RV is independent of epithelial cell destruction, apoptosis, or virus-stimulated proinflammatory cytokines, indicating that other mechanisms play a role.
Oxidative stress has been implicated in the impairment of airway and colonic epithelial barrier function (6, 38, 51, 52). Treatment with hydrogen peroxide disrupted the barrier function of airway epithelial cells by destabilizing the actin cytoskeleton, damaging tight junctions, and inhibiting cell proliferation (53). In colonic epithelial cells, hydrogen peroxide caused tyrosine phosphorylation of occludin and dissociation of occludin and ZO-1, leading to decreased transepithelial resistance (RT) and increased epithelial permeability (39). RV has been shown to induce oxidative stress in nonpolarized airway epithelial cells by generating reactive oxygen species (ROS) (5, 23, 36), but the role of ROS in barrier function disruption has not been investigated in polarized epithelial cells.
ROS play an important role in innate host defense mechanisms. In phagocytes, ROS are necessary for the killing of invading microorganisms. In other cells, ROS act as a molecular switch to stimulate proinflammatory responses. The majority of intracellular ROS are generated from two sources: the mitochondrial electron transport chain complex and membrane-bound NADPH oxidase (NOX) enzymes. In phagocytes, the NOX holoenzyme is a multisubunit complex composed of the membrane-bound p22phox and catalytic subunit gp91phox (now known as NOX2) and the cytoplasmic regulators p47phox and p67phox. During the last decade, six functionally distinct homologs of NOX2, namely, NOX1, NOX3, NOX4, NOX5, DUOX1, and DUOX2, have been identified in a wide variety of cells, including airway epithelial cells (3, 14, 29). NOX1 to -5 generate superoxide, which converts to other ROS, whereas DUOX1 and DUOX2 produce hydrogen peroxide at the cell surface. NOX2 was recently shown to control respiratory syncytial virus-stimulated NF-κB activation in airway epithelial cells (14, 55). Similarly, RV-stimulated interleukin-8 (IL-8) responses were shown to be dependent on p47phox, though the specific identity of the NOX2 homolog was not determined (5, 23, 36). The roles of ROS and NOX in the RV-induced impairment of barrier function have not been investigated.
In the present study, we examined the biochemical mechanisms of RV-impaired barrier function in polarized airway epithelial cells, focusing on the generation of ROS by replicating RV and NOX enzyme. We demonstrate that RV-induced Rac1-dependent NOX1 activity is required to disrupt barrier function. We also show that polyinosinic-poly(C) [poly(I:C)], a synthetic analog of double-stranded RNA (dsRNA) that has been used to examine viral dsRNA-stimulated responses in airway epithelial cells (26, 30, 49), impairs barrier function by a mechanism similar to that of RV, indicating that dsRNA generated during replication contributes to the disruption of barrier function.
MATERIALS AND METHODS
Rhinovirus.
Rhinovirus 39 (RV39) was purchased from the American Type Culture Collection (Manassas, VA), and viral stocks were generated as described previously (43). Briefly, cell culture supernatants from RV-infected HeLa cells were partially purified by ultrafiltration, and 50% tissue culture infective dose (TCID50) values of viral stocks were determined by the Spearman-Karber method (22). A sham was prepared that was similar to RV but from uninfected HeLa cell supernatant. The sham served as a control for cells infected with RV or UV-irradiated RV (UV-RV).
Bacteria and growth conditions.
A clinical isolate of nontypeable Haemophilus influenzae (NTHI), 6P5H, obtained from a patient with COPD at the time of exacerbation, was kindly provided by T. Murphy (University of Buffalo) and has been described previously (41). Bacteria maintained as a glycerol stock at −80°C were subcultured on a chocolate agar plate and incubated overnight at 37°C and 5% CO2. Bacteria were scraped off the plate and suspended in serum- and antibiotic-free cell culture medium at the required concentration.
Cell culture and infection.
Immortalized human bronchial epithelial cells (16HBE14o− cells) were grown in Transwells (Corning, Lowell, MA) in minimum essential medium (MEM; Invitrogen, Carlsbad, CA) amended with 10% heat-inactivated fetal bovine serum (FBS) and 2 mM l-glutamine, as described previously (41). Briefly, 8 × 104 or 3 × 105 cells were seeded in 6.5-mm or 12-mm collagen-coated Transwells, respectively, and grown under submerged conditions until the RT reached 800 to 1,000 Ω · cm2, which usually takes 4 to 5 days. Medium in both the apical and basolateral chambers was changed every other day. In nearly all experiments, Transwells with a pore size of 0.4 μm were used. To determine bacterial transmigration, cells were grown in Transwells with a pore size of 3 μm.
On the day of infection, medium in the basolateral chamber was replaced with fresh growth medium, and the apical surface was infected with 100 μl RV diluted in growth medium at a multiplicity of infection (MOI) of 1 or with an equivalent volume of UV-RV or sham control. After 90 min of incubation, infection medium from the apical surface was replaced with fresh growth medium, and the cells were further incubated for 4 to 24 h at 33°C. In inhibition studies, N-propyl gallate, diphenylene iodonium (DPI), oxypurinol, quercetin (all from Sigma-Aldrich, St. Louis, MO), or the Rac1 inhibitor NSC23766 (EMD Chemicals, Gibbstown, NJ) was added to both the apical and basolateral chambers 90 min after RV infection and incubated for 24 h, and RT was measured with an Evom voltmeter equipped with an EndOhm 6 tissue resistance measurement chamber (World Precision Instruments, Sarasota, FL) (25, 41).
Treatment with poly(I:C).
Polarized 16HBE14o− cells were treated apically with 300 μl of medium containing 500 ng/ml of high-molecular-weight poly(I:C) (1.5 to 8 kb; Invivogen), and RT was measured at the time points indicated in Results.
Transmigration of bacteria from apical to basolateral chamber.
Transmigration of bacteria across the polarized cells was measured as described earlier (41). Briefly, 16HBE14o− cells grown in Transwells with a pore size of 3 μm were infected with RV or UV-RV at an MOI of 1 or treated with poly(I:C) as described above. Eight hours [for poly(I:C)-treated cells] or 24 h (for RV- and UV-RV-infected cells) later, 100 μl of NTHI at an MOI of 10 was added to the apical chamber and further incubated for 3 h. Medium from the basolateral chambers was sampled at 3 h and plated on chocolate agar to determine the number of bacteria that transmigrated across the polarized airway epithelial cells.
Viral load and cell infectivity.
Total RNA was extracted from polarized epithelial cells 90 min or 24 h after RV infection, and viral RNA (vRNA) was quantified by quantitative PCR (qPCR) as described previously (43). To quantify the percentage of cells infected with virus, cells were fixed/permeabilized with 4% paraformaldehyde after appropriate treatment, blocked with normal goat serum, and incubated with Alexa Fluor 488-conjugated antibody to VP2, one of the capsid proteins of RV (21) (kindly provided by Wai-Ming Lee, University of Wisconsin, Madison, WI). Cells were analyzed by flow cytometry. Similarly treated uninfected cells were used as negative controls.
Assay of NOX mRNA expression by qPCR.
The mRNA expression of NOX genes was assessed by qPCR using gene-specific primers (SA Biosciences, Frederick, MD). All PCRs were performed on an Eppendorf Mastercycler machine (Westbury, NY), using the comparative threshold cycle (CT) method. The expression levels were normalized to the glyceraldehyde-3-phosphate dehydrogenase (G3PDH) reference gene as described previously (43).
Confocal indirect immunofluorescence microscopy.
Distributions of the tight junction proteins ZO-1 and occludin were determined by indirect immunofluorescence microscopy as described previously (41). Briefly, after appropriate treatments, polarized 16HBE14o− cells were fixed in cold methanol, blocked with phosphate-buffered saline (PBS) containing 1% (wt/vol) bovine serum albumin (BSA), and incubated overnight at 4°C with polyclonal antibody to ZO-1 (1 μg/ml) or monoclonal antibody to occludin (both from BD Biosciences, San Jose, CA). Bound antibodies were detected by using an appropriate Alexa Fluor-conjugated secondary antibody (Invitrogen) and visualization by confocal fluorescence microscopy (Carl Zeiss, Thornwood, NY). Cells treated with normal IgG isolated from nonimmunized animals (Jackson ImmunoResearch Laboratories, West Grove, PA) instead of with primary antibody served as negative controls.
To localize poly(I:C), polarized 16HBE14o− cells were treated with rhodamine-labeled poly(I:C), incubated for 4 h, fixed in methanol, immunolabeled with anti-ZO-1, and visualized by confocal microscopy.
Determination of NOX enzymatic activity.
NOX enzymatic activity in cells treated with RV or UV-RV was determined by measuring the reduction of cytochrome c (36). Briefly, polarized 16HBE14o− cells were infected with RV or UV-RV or treated with poly(I:C) as described above. Cells were collected, homogenized in a Dounce homogenizer, and centrifuged, and supernatant was collected. Aliquots of cell supernatant containing equal amounts of total protein were mixed with NADPH and cytochrome c and incubated for exactly 10 min at 37°C in the absence or presence of a flavoprotein inhibitor (DPI) or oxypurinol (both from Sigma-Aldrich Co., St. Louis, MO), and the optical density at 550 nm (OD550) was measured.
ROS production.
ROS production was measured by using carboxy-2′,7′-dichlorodihydrofluorescein diacetate (carboxy-H2DCFDA; Invitrogen) as described previously (40). Briefly, after appropriate treatment, cells were infected with either RV or UV-RV as described above. Six hours before the measurement of ROS, medium was removed and cells were rinsed with Hanks balanced salt solution (HBSS) and then incubated in HBSS containing 5 μM carboxy-H2DCFDA without phenol red for 6 h. At this concentration, carboxy-H2DCFDA had no effect on the cells or on RV replication, as measured by lactate dehydrogenase (LDH) release and vRNA copy number. For poly(I:C) treatment, cells were preloaded with 5 μM carboxy-H2DCFDA for 1 h and then incubated with poly(I:C) for a predetermined time. Cells were then detached from the wells, washed, and resuspended in PBS. Fluorescence emission was quantified by flow cytometry (FACSCaliber; BD), and the results were analyzed using WinMDI software.
Transfection of 16HBE14o− cells growing in Transwells.
16HBE14o− cells (8 × 104 cells/well) were seeded in 6.5-mm Transwells and grown for 2 days. At this time, cells were partially polarized and showed RT values ranging from 60 to 100 Ω · cm2. Pools of double-stranded small interfering RNAs (siRNAs) specific to NOX1 (antisense sequences in siRNA pool, 5′-GGUUAGGGCUGAAUGUUUU-3′, 5′-CUGCCUACAUACAGCUAUU-3′, 5′-GACAAAUACUACUACACAA-3′, and 5′-UGAGAAAGCAAUUGGAUCA-3′) and Toll-like receptor 3 (TLR3) (antisense sequences in siRNA pool, 5′-GAACUAAAGAUCAUCGAUU-3′, 5′-CAGCAUCUGUCUUUAAUAA-3′, 5′-AGACCAAUCUCUCAAAUUU-3′, and 5′-UCACGCAAUUGGAAGAUUA-3′) and nontargeting (NT) siRNAs (sense, 5′-CGAACUCACUGGUCUGACCdtdt-3′; and antisense, 5′-GGUCAGACCAGUGAGUUCGdtdt-3′) were purchased from Dharmacon (Lafayette, CO). siRNA (5 to 10 pmol/well) was incubated with 1 μl of Lipofectamine RNAi Max (Invitrogen) in 100 μl Opti-MEM (Invitrogen) for 20 min at room temperature. Medium in the basolateral chamber was replaced with fresh medium, the apical chamber medium was removed, and 100 μl of transfecting reaction mix was added. After 6 h of incubation, the volume in the apical chamber was made up to 300 μl with fresh medium, and incubation was continued for another 18 h. Media in both the apical and basolateral chambers were changed and incubated for another 24 h. Forty-eight hours after transfection, the RT of cells was measured to confirm that cells were polarized similarly to nontransfected cells.
Western blot analysis.
After relevant treatment, cells were lysed in RIPA buffer (25 mM HEPES, pH 7.4, 150 mM NaCl, 1% Nonidet P-40 [NP-40], 0.25% sodium deoxycholate, 0.1% SDS) containing Complete protease inhibitor (Roche Diagnostics, Indianapolis, IN) and then centrifuged, and the supernatant was collected. In some experiments, cells were lysed in NP-40 solubilization buffer (25 mM HEPES [pH 7.4], 150 mM NaCl, 1% [vol/vol] NP-40, and Complete protease inhibitor) and then centrifuged (10,000 × g for 30 min at 4°C). The pellet containing NP-40-insoluble material was collected, suspended in Laemmli reducing buffer, and heated for 10 min at 100°C. Equal amounts of protein (for total cell lysates) or equal volumes (for NP-40-insoluble fractions) were subjected to SDS-PAGE, and proteins were transferred to nitrocellulose membranes by wet transfer for 1 h (for detection of occludin and NOX1) and for 90 min (for detection of ZO-1). The membranes were blocked with either 5% bovine serum albumin or fat-free milk and then probed with antibody to ZO-1 (BD Biosciences, San Diego, CA) (dilution, 1:1,000), occludin (BD Biosciences) (dilution, 1:500), NOX1 (Abcam, Cambridge, MA) (1 μg/ml), or β-actin (Sigma-Aldrich) (dilution, 1:10,000). The bound antibody was detected with an appropriate secondary antibody conjugated with horseradish peroxidase (Bio-Rad, Hercules, CA) and chemiluminescent substrate (Pierce, Rockford, IL). Specific bands were quantified by densitometry using NIH ImageJ software.
Determination of TLR3 expression by flow cytometry.
After relevant treatment, cells were fixed with 4% paraformaldehyde and permeabilized with 0.5% saponin in blocking buffer (1% BSA in PBS) on ice for 15 min. Cells were washed with blocking buffer, incubated with 1:10-diluted antibody to TLR3 (Abcam) or normal mouse IgG (Jackson ImmunoResearch Laboratories) for 1 h, washed, and incubated with 1:1,000-diluted Alexa Fluor 488-conjugated anti-mouse IgG. Cells were then analyzed by flow cytometry.
Rac1 activation assay.
Activation of Rac1 in 16HBE14o− cells was determined by using a Rac1 activation assay kit following the manufacturer's instructions (Millipore, Temecula, CA). Briefly, after relevant treatment, cells were lysed in the presence of aprotinin and leupeptin, lysates were incubated with glutathione agarose for 10 min and centrifuged, and total protein in the supernatant was determined. A volume equivalent to 500 μg of total protein was incubated with PAK-1 PBD agarose for 60 min at 4°C. Agarose beads were collected by a brief centrifugation and washed with lysis buffer. Bound Rac1-GTP was resolved by 15% SDS-PAGE, and Rac1 was detected by immunoblotting using a monoclonal antibody to Rac1. Clarified cell lysates incubated with GTPγS and GDP followed by incubation with PAK-1 PBD agarose were used as positive and negative controls, respectively. An aliquot of clarified cell lysate equivalent to 5 μg of total protein was used as a loading control. Quantification of enhanced chemiluminescence (ECL) signals was performed by densitometry using NIH ImageJ.
Data analysis.
Statistical significance was assessed by analysis of variance (ANOVA) or ANOVA on ranks, as appropriate. Differences identified by ANOVA or ANOVA on ranks were pinpointed by the Tukey-Kramer or Dunn's test, respectively.
RESULTS
Poly(I:C) treatment decreases RT of polarized airway epithelial cells.
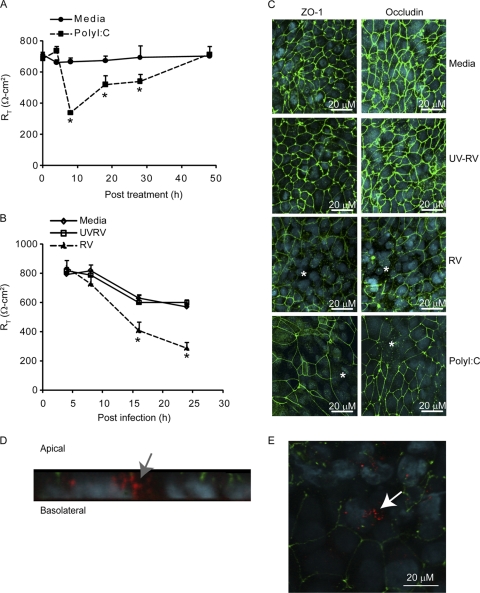
Previously, we demonstrated that RV replication is required for disruption of barrier function. RV generates dsRNA during replication. To examine whether dsRNA is sufficient to disrupt barrier function, we treated polarized 16HBE14o− cells with 500 ng/well poly(I:C) and monitored RT for up to 48 h. Poly(I:C) decreased RT, showing a significant reduction starting at 6 h and reaching a maximum reduction at 8 h. RT returned to normal by 48 h (Fig. 1A). As observed earlier, cell cultures infected with infectious RV also showed a significant reduction in RT compared to cells treated with medium or replication-deficient UV-RV (Fig. 1B).
Fig. 1.
Poly(I:C) decreases RT by dissociating ZO-1 and occludin from tight junction complexes, similar to RV. Polarized airway epithelial cells grown in Transwells were treated with 500 ng/ml poly(I:C) and incubated for up to 48 h (A) or were infected apically with RV or UV-RV for 90 min, after which the infection medium was replaced with fresh medium and the cells were incubated for up to 24 h (B). RT was measured at various time points. Results represent means ± standard errors of the means (SEM) calculated for three independent experiments carried out in triplicate. *, different from medium control or UV-RV- or sham-treated cells (P ≤ 0.05 by ANOVA). In some experiments, cells were treated with poly(I:C) (C), infected with RV or UV-RV (C), or treated with rhodamine-labeled poly(I:C) (D and E) as described above, fixed in methanol, and immunostained with antibody to ZO-1 or occludin (green). Nuclei were stained with DAPI (4′,6-diamidino-2-phenylindole) (blue). Asterisks in panel C represent dissociation of ZO-1 or occludin from the periphery of cells. Arrows in panels D and E represent rhodamine-labeled poly(I:C) below the apical surface and within the cells, respectively. Images are representative of three independent experiments.
Next, we examined the distributions of the tight junction proteins ZO-1 and occludin in poly(I:C)-treated (8 h posttreatment) and RV-infected (24 h postinfection) polarized 16HBE14o− cells by indirect immunofluorescence confocal microscopy. While medium-treated control cells showed normal distributions of both ZO-1 and occludin, poly(I:C)-treated and RV-infected cell cultures each showed dissociation of these tight junction proteins from the periphery of cells (Fig. 1C). These results indicate that dsRNA generated during RV replication may play a role in reducing RT.
To localize poly(I:C), polarized cells were treated with rhodamine-labeled poly(I:C) and visualized in context with ZO-I. In x-z section, we observed that poly(I:C) was located at and below the apical surface and also saw a loss of ZO-1 from the tight junctions of the cells that contained poly(I:C) (Fig. 1D). An x-y optical section taken in the middle of the cells showed poly(I:C) within the cells and ZO-1 in the periphery of the cells (Fig. 1E).
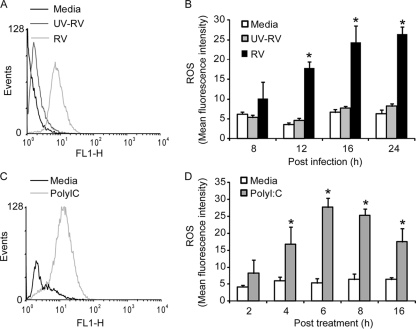
RV and poly(I:C) treatment each induce ROS generation.
RV infection may generate ROS (5, 23, 36). Therefore, we measured ROS production in 16HBE14o− polarized cells infected with either infectious RV or UV-RV for 8, 12, 16, or 24 h. Cells were also treated with medium or poly(I:C) for 2, 4, 6, 8, or 16 h. Compared to medium- or UV-RV-treated cells, RV-infected cells showed a significant increase in ROS production, starting at 12 h, which was sustained for up to 24 h (Fig. 2A and B). Similarly, poly(I:C)-treated cells showed an increase in ROS production starting at 4 h and reaching a maximum at 6 h (Fig. 2C and D). The increase in ROS production in both RV-infected cells and poly(I:C)-treated cells preceded the reduction in RT.
Fig. 2.
RV infection or poly(I:C) treatment stimulates ROS generation in polarized airway epithelial cells. Polarized 16HBE14o− cells were infected with RV or UV-RV (A and B) or treated with poly(I:C) (C and D). Generation of ROS was measured by flow cytometry using carboxy-H2DCFDA 24 h after RV infection (A) or 8 h after poly(I:C) treatment (C). The kinetics of ROS generation was measured in RV-infected (B) or poly(I:C)-treated (D) cells by flow cytometry. Histograms are representative of four independent experiments. Data in panels B and D are means ± SEM for four independent experiments carried out in duplicate. *, different from medium control or UV-RV-treated cells (P ≤ 0.05 by ANOVA).
DPI inhibits RV- or poly(I:C)-induced reductions in RT in polarized epithelial cells.
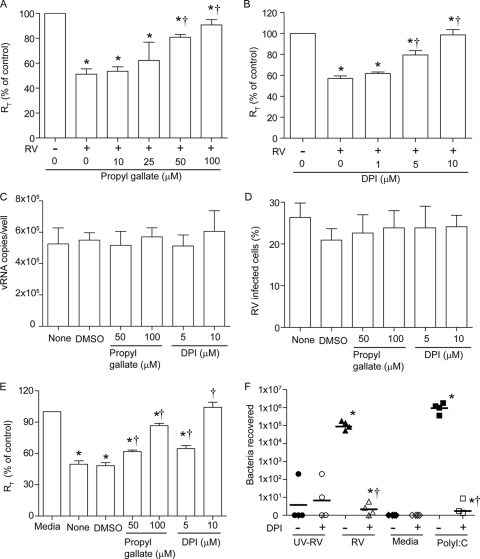
To determine the contribution of ROS to the reduction of RT caused by RV, polarized 16HBE14o− cells were infected with RV. Ninety minutes later, the ROS scavenger N-propyl gallate or the inhibitor of ROS-generating enzymes DPI (a NOX flavoprotein inhibitor) or oxypurinol (a xanthine oxidase inhibitor) was added, and RT was measured 8 or 24 h later. Propyl gallate and DPI inhibited the RV-induced reduction in RT (Fig. 3A and B). This was not due to a reduction in RV load, as there was no significant difference in vRNA levels between untreated and antioxidant-treated cells (Fig. 3C) or in the number of cells infected with RV, as determined by flow cytometry (Fig. 3D). Propyl gallate and DPI also inhibited poly(I:C)-induced reductions in RT (Fig. 3E). Oxypurinol had no effect on either RV- or poly(I:C)-induced reductions in RT (data not shown).
Fig. 3.
Propyl gallate and DPI inhibit RV- or poly(I:C)-induced barrier disruption in polarized airway epithelial cells. Polarized 16HBE14o− cells were infected with RV or UV-RV for 90 min, and the infection medium was replaced with medium containing propyl gallate (A) or DPI (B). RT was measured after 24 h and expressed as a percentage relative to UV-RV-treated controls. (C) RNAs were extracted from selected groups of RV-infected cells at 24 h postinfection, and the number of vRNA copies was determined. (D) Percentage of cells infected with RV, assessed by flow cytometry. (E) For poly(I:C)-treated cells, propyl gallate and DPI were added 30 min after adding poly(I:C), and RT was measured after 8 h and expressed as a percentage relative to similarly treated medium controls. Data represent means ± SEM. (F) Polarized cells were treated with RV, UV-RV, or poly(I:C) as described above in the presence or absence of 10 μM DPI for 8 h [for poly(I:C)-treated cells] or 24 h (for RV-infected cells), NTHI was added to the apical chamber, and the number of bacteria in the basolateral medium after 3 h was determined by plating. Data represent ranges and medians. *, different from respective UV-RV- or medium-treated controls (P ≤ 0.05); †, different from RV-infected or poly(I:C)-treated cells in the absence of propyl gallate or DPI (P ≤ 0.05 by ANOVA [A to E] or ANOVA on ranks [F]). DMSO, dimethyl sulfoxide.
DPI inhibits transmigration of bacteria in RV-infected polarized airway epithelial cells.
Next, we examined the effect of DPI on the transmigration of bacteria across the polarized 16HBE14o− cells infected with RV. As previously observed (41), we recovered bacteria from the basolateral medium of RV-infected cells (Fig. 3F). On the other hand, cells infected with RV and incubated in the presence of DPI did not show bacteria in the basolateral chamber, indicating that DPI abrogates RV-induced changes in the paracellular permeability of polarized airway epithelial cells. A similar abrogation in the transmigration of bacteria was observed in cells treated with poly(I:C) in the presence of DPI. Together, these results indicate that ROS generation mediated by NOX is required for RV- or poly(I:C)-induced barrier function disruption.
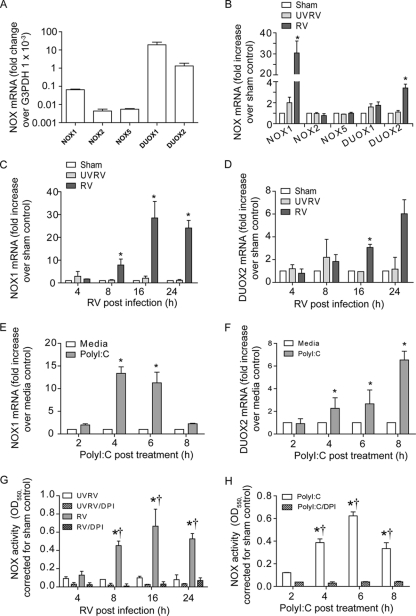
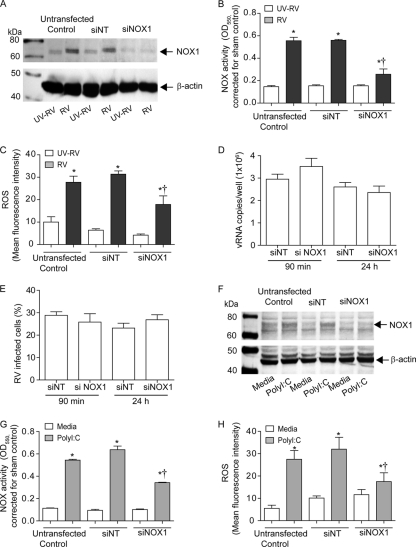
RV and poly(I:C) stimulate NOX1 expression and activity.
So far, at least seven NOXs have been described. We examined the effects of RV infection on the expression of all 7 NOXs in 16HBE14o− cells by qPCR. NOX3 and NOX4 were undetectable in our cell culture model at baseline (untreated cells) as well as after RV or poly(I:C) treatment (data not shown). Baseline expression of DUOX1 and DUOX2 was higher than that of NOX1, NOX2, and NOX5 (Fig. 4A). DUOX2 expression was increased further (4-fold) in RV-infected cells (Fig. 4B). On the other hand, NOX1 expression increased almost 30-fold in RV-infected cells compared to that in sham- or UV-RV-infected cells. RV infection did not alter expression of either NOX2 or NOX5. These results indicate that DUOX2 and NOX1 may contribute to RV-induced ROS generation. Next, we determined the kinetics of NOX1 and DUOX2 expression in cells treated with infectious RV or UV-RV (Fig. 4C and D). In cells incubated with intact RV, but not those incubated with UV-RV, NOX1 expression increased as early as 8 h and reached the maximum 16 h after infection, and this coincided with the increased ROS generation caused by RV infection (Fig. 2B). In contrast, DUOX2 expression did not show a significant increase until 16 h postinfection and was increased further at 24 h postinfection. Expression of NOX1 was much higher than that of DUOX2 at all time points examined. Poly(I:C)-treated cells also showed increased mRNA expression of both NOX1 and DUOX2, and again, NOX1 expression was higher than DUOX2 expression, reaching the maximum at 6 h (Fig. 4E and F), which again coincided with poly(I:C)-induced ROS generation (Fig. 2D).
Fig. 4.
RV and poly(I:C) increase mRNA expression of NOX1 and DUOX2 and enhance NOX activity. Total RNAs were isolated from medium-treated cells (A) and from sham-, RV-, and UV-RV-infected cells (B), and expression of NOX1, NOX2, NOX5, DUOX1, and DUOX2 was measured by qPCR. The kinetics of NOX1 and DUOX2 mRNA expression was determined for RV-infected cells (C and D) and poly(I:C)-treated cells (E and F). The kinetics of NOX enzymatic activity was measured at various time points after RV infection (G) or poly(I:C) treatment (H) by cytochrome c reduction assay in the presence or absence of DPI. Data represent means ± SEM calculated for 3 or 4 independent experiments carried out in duplicate. *, different from sham- or medium-treated controls (P ≤ 0.05); †, different from NOX enzymatic activity determined in the presence of DPI (P ≤ 0.05 by ANOVA).
We then examined the total NOX activity in RV- or poly(I:C)-treated cells by measuring the reduction of cytochrome c (36). This assay measures the activities of both NOX and xanthine oxidase. Therefore, we measured the cytochrome c reduction in the presence or absence of DPI (an inhibitor of NOX) or oxypurinol (an inhibitor of xanthine oxidase). Compared to cells treated with UV-RV, cells infected with RV showed significant increases in the reduction of cytochrome c at 8, 12, and 16 h postinfection (Fig. 4G), coinciding with expression of NOX1 and with ROS generation. DPI, but not oxypurinol, abrogated the reduction of cytochrome c in RV-infected cells, indicating that cytochrome c reduction is due to the activity of NOX, not xanthine oxidase. Poly(I:C)-treated cells also showed a time-dependent increase in NOX activity, which peaked at 6 h (Fig. 4H), coinciding with NOX1 expression and ROS production.
Inhibition of NOX1 abrogates RV-induced NOX activity and ROS generation.
To determine the contribution of NOX1 to RV-induced NOX activity, 16HBE14o− cells transfected with NOX1-specific (siNOX1) or nontargeting (siNT) siRNA were infected with intact RV or UV-RV. After 16 h of incubation, expression of NOX1, NOX activity, and ROS generation were determined. Both untransfected cells and cells transfected with siNT followed by RV infection showed increased NOX1 expression (Fig. 5A), NOX activity (Fig. 5B), and ROS generation (Fig. 5C) compared to cells infected with UV-RV or sham. In contrast, cells transfected with siNOX1 prior to RV infection showed no increase in NOX1 expression, as well as reduced NOX activity and ROS generation. This was not due to decreased RV infection or replication in siNOX1-transfected cells (Fig. 5D and E). These results indicate that RV-induced NOX1 activity contributes to total NOX activity and ROS generation. Poly(I:C)-induced NOX activity and ROS generation were also found to be partially dependent on NOX1 (Fig. 5F to H).
Fig. 5.
RV- or poly(I:C)-induced NOX1 expression contributes to increased total NOX activity and ROS generation. 16HBE14o− cells were transfected with siNT or siNOX1. Two days later, cells were infected with RV or treated with poly(I:C) as described in the text. Cells were harvested 16 h (for RV-infected cells) or 8 h [for poly(I:C)-treated cells] after treatment and assessed for expression of NOX1 by Western blot analysis (A and F), for total NOX activity by measuring cytochrome c reduction (B and G), and for ROS generation by flow cytometry using carboxy-H2DCFDA (C and H). The vRNA copy number (D) and % of RV-infected cells (E) were determined for siNT- and siNOX1-transfected cells 24 h after RV infection. Data represent means ± SEM calculated for 3 or 4 independent experiments carried out in duplicate. *, different from respective controls (P ≤ 0.05); †, different from respective siNT-treated controls (P ≤ 0.05 by ANOVA).
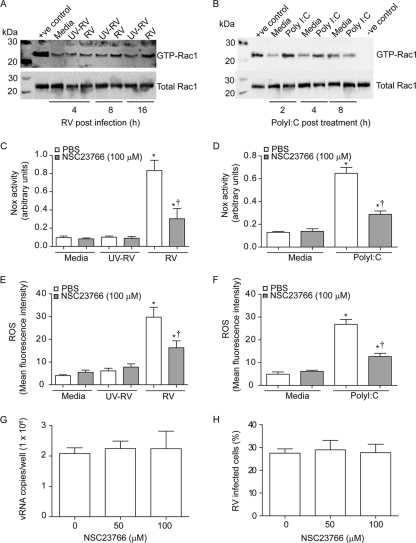
RV infection stimulates Rac1 activation in 16HBE14o− cells.
Rac1 activation is required for assembly of the holoenzyme and for maximal activity of NOX1 (8). Therefore, we investigated whether RV stimulates Rac1 activity in 16HBE14o− cells by pulldown assay. Compared to cells infected with UV-RV, cells infected with infectious RV showed increased Rac1 activity 8 h after infection, which further increased at 16 h (Fig. 6A). In contrast, poly(I:C)-treated cells showed the highest activity 2 h after treatment, which slowly returned to normal by 8 h (Fig. 6B).
Fig. 6.
RV or poly(I:C)-induced Rac1 activity partially contributes to total NOX activity and ROS generation. 16HBE14o− cells were infected with RV (A) or treated with poly(I:C) (B), and Rac1 activity was determined by pulldown assay at various time points. Medium- and UV-RV-treated cells were used as controls. +ve control and −ve control, cell lysates from medium-treated cells incubated with GTPγS and GDP, respectively. Images are representative of 3 experiments. Cells were then infected with RV or UV-RV (C and E) or treated with medium or poly(I:C) (D and F) and incubated in the presence or absence of NSC27366. Cells were harvested after 8 h [for poly(I:C)-treated cells] or 24 h (for RV-infected cells) and then examined for NOX activity (C and D) and ROS generation (E and F). The vRNA copy number (G) and % of RV-infected cells (H) were determined for siNT- and siNOX-transfected cells 24 h after RV infection. Data represent means ± SEM calculated for 3 or 4 independent experiments carried out in duplicate. *, different from respective controls (P ≤ 0.05); †, different from RV-infected or poly(I:C)-treated cells in the absence of NSC23766 (P ≤ 0.05 by ANOVA).
We then examined the requirement of Rac1 activation for RV-induced NOX1 activity. 16HBE14o− cells were infected with intact or UV-irradiated RV and incubated for 1 h. Infection medium was replaced with fresh medium containing 50 μM NSC23766, a chemical inhibitor of Rac1 (17), and NOX activity was determined 16 h after incubation. NSC23766 (50 μM) reduced RV-induced NOX activity by 71% and ROS production by 51% (Fig. 6C and E). NSC23766 also partially inhibited poly(I:C)-induced NOX activity in 16HBE14o− cells (Fig. 6D and F). Sixteen hours after RV infection, there was no difference in levels of vRNA or percentages of infected cells between untreated and NSC23766-treated cells (Fig. 6G and H). Together, these results indicate that both RV and poly(I:C) stimulate Rac1 activation and that Rac1 activity is required for maximal NOX activity and ROS production induced by these agents.
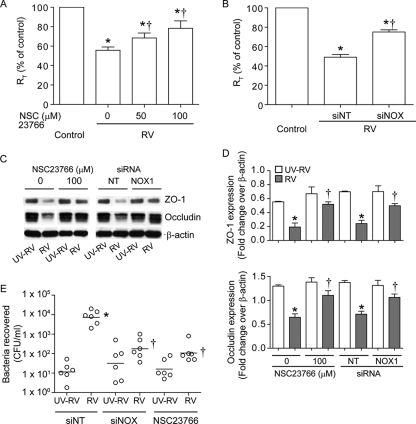
RV-induced disruptions in barrier function require Rac1 and NOX1.
To determine the requirement of Rac1 activation for the observed reductions in RT, polarized 16HBE14o− cells were infected with RV for 90 min to allow endocytosis, the medium was replaced with fresh medium containing NSC23766, and RT was measured at the end of 24 h. The Rac1 inhibitor attenuated the RV-induced reduction in RT (Fig. 7A). Cells transfected with siRNA specific to NOX1 were resistant to RV-induced reductions in RT compared to cells transfected with siNT (Fig. 7B). These results suggest that both Rac1 and NOX1 are required for RV-induced reductions in RT.
Fig. 7.
Rac1 and NOX1 are required for RV-induced disruption of barrier function. (A) Polarized airway epithelial cells grown in Transwells were infected with RV or UV-RV as described in the text and incubated for 24 h in the presence or absence of various concentrations of NSC23766. RT was expressed as a percentage relative to UV-RV-infected controls. (B) Cells growing in Transwells were transfected with NT or NOX1 siRNA, and 2 days later, cells were infected with RV or UV-RV. RT was measured after 24 h and expressed as a percentage relative to the respective UV-RV controls. (C) The cytoskeleton fraction was subjected to Western blot analysis with antibody to ZO-1 or occludin, and the image is representative of 3 independent experiments. (D) Quantification of the ratio of occludin or ZO-1 to β-actin. (E) Twenty-four hours after RV or UV-RV infection, nontypeable H. influenzae was added to the apical chamber and bacteria in the basolateral chamber were quantified to assess bacterial transmigration across polarized airway epithelial cells. Data represent means ± SEM (A, B, and D) or ranges with medians (E) calculated for 3 or 4 independent experiments carried out in duplicate. *, different from respective controls (P ≤ 0.05); †, different from RV-infected cells in the absence of NSC23766 or from NT siRNA-transfected cells infected with RV (P ≤ 0.05 by ANOVA or ANOVA on ranks).
In cells with intact tight junctions, most of the cellular ZO-1 and occludin partitions with the cytoskeletal fraction. We have previously shown that RV infection reduces the amount of ZO-1 in the cytoskeletal fraction (41). To assess whether inhibition of Rac1 and NOX1 improves RT by increasing the retention of ZO-1 and occludin with the cytoskeleton, we treated cells with NSC23766 and siNOX1 and subjected cytoskeletal fractions to Western blot analysis. RV-infected cells showed decreased amounts of both ZO-1 and occludin in the cytoskeletal fraction compared to cells infected with UV-RV (Fig. 7C and D). The Rac1 inhibitor partially inhibited the loss of both ZO-1 and occludin from the cytoskeletal fraction of RV-infected cells. RV-infected cells transfected with siNOX1 also showed increased amounts of ZO-1 and occludin in cytoskeletal fractions compared to cells transfected with siNT. Consistent with these findings, confocal microscopy revealed increased retention of ZO-1 and occludin in tight junctions of cells infected with RV in the presence of the Rac1 inhibitor and also in RV-infected siNOX1-transfected cells (data not shown). These results imply that inhibition of Rac1 activity and NOX1 expression improves RT in RV-infected cells by reducing dissociation of ZO-1 and occludin from the cytoskeleton.
Next, we examined whether inhibition of Rac1 and NOX1 improves the barrier function in RV-infected cells. As observed earlier, RV-infected cells showed increased transmigration of bacteria. Transmigration was significantly inhibited by NSC23766 and siNOX1 but not by siNT (Fig. 7E).
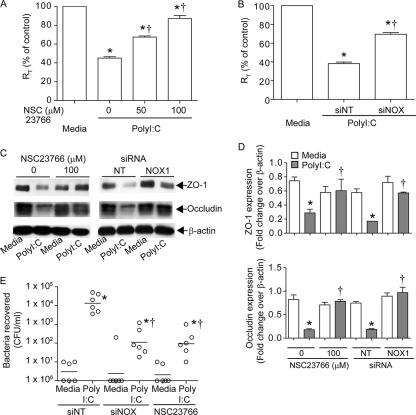
Rac1 and NOX1 are also required for the disruption of barrier function induced by poly(I:C).
Polarized 16HBE14o− cells were treated with poly(I:C) in the presence or absence of NSC23766, and RT and the association of ZO-1 and occludin with the cytoskeleton were determined. The Rac1 inhibitor attenuated poly(I:C)-induced reductions in RT, and this was associated with increased retention of both ZO-1 and occludin with the cytoskeleton fraction (Fig. 8A, C, and D). Consistent with this, transmigration of bacteria from the apical to the basolateral surface was decreased in cells treated with poly(I:C) in the presence of the Rac1 inhibitor (Fig. 8E). Similarly, cells transfected with siNOX1 and then treated with poly(I:C) showed a higher RT, more ZO-1 and occludin in the cytoskeletal fraction, and fewer bacteria in the basolateral chamber than similarly treated cells transfected with siNT (Fig. 8B to E). Collectively, these results suggest that Rac1-dependent NOX1 activity is required for RV- and poly(I:C)-induced reductions in tight junction function in polarized airway epithelial cells.
Fig. 8.
Poly(I:C)-induced barrier disruption requires both Rac1 and NOX1. (A) Polarized epithelial cells were treated with medium or poly(I:C) for 8 h in the presence or absence of NSC23766, and RT was measured. (B) Cells transfected with NT or NOX1 siRNA were treated with poly(I:C), and RT was measured after 8 h. RT is expressed as a percentage relative to the respective medium controls. (C and D) ZO-1 and occludin protein levels in the cytoskeleton fraction were determined by Western blot analysis followed by densitometry and are expressed as the ratio of ZO-1 or occludin to β-actin. The Western blot is representative of 3 experiments. (E) Eight hours after poly(I:C) treatment, bacterial transmigration across polarized airway epithelial cells was measured as described in the text. Data represent means ± SEM (A, B, and D) or ranges with medians (E) calculated for 3 or 4 independent experiments carried out in duplicate. *, different from respective controls (P ≤ 0.05); †, different from RV-infected cells in the absence of NSC23766 or from NT siRNA-transfected cells infected with RV (P ≤ 0.05 by ANOVA or ANOVA on ranks).
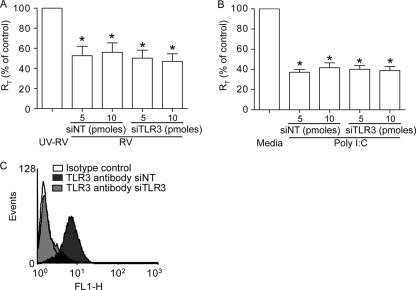
TLR3 is not required for either RV- or poly(I:C)-induced reductions in RT.
TLR3 recognizes both poly(I:C) and RV dsRNA (31, 49). To determine the role of TLR3 in RV- and poly(I:C)-induced reductions in RT, polarized cells transfected with siNT or TLR3-specific siRNA (siTLR3) were either infected with RV or treated with poly(I:C) as described above, and RT was measured 24 h after RV infection or 8 h after poly(I:C) treatment. Both RV and poly(I:C) caused reductions in RT in siTLR3-transfected cells that were similar to those observed in cells transfected with siNT (Fig. 9A and B). Flow cytometry indicated reduced TLR3 expression in cells transfected with siTLR3 (Fig. 9C). Together, these results suggest that TLR3 is not required for either RV- or poly(I:C)-induced reductions in RT.
Fig. 9.
TLR3 is not required for either RV- or poly(I:C)-induced reduction in RT. Cells transfected with NT or TLR3 siRNA were treated with RV (A) or poly(I:C) (B), and RT was measured after 24 or 8 h, respectively. Data are expressed as percentages relative to the respective UV-RV- or medium-treated controls and represent means ± SEM calculated for 3 or 4 independent experiments carried out in duplicate. *, different from respective controls (P ≤ 0.05 by ANOVA). (C) Expression of TLR3 in siNT- or siTLR3-transfected cells was determined by flow cytometry. The histogram shown is representative of 3 experiments.
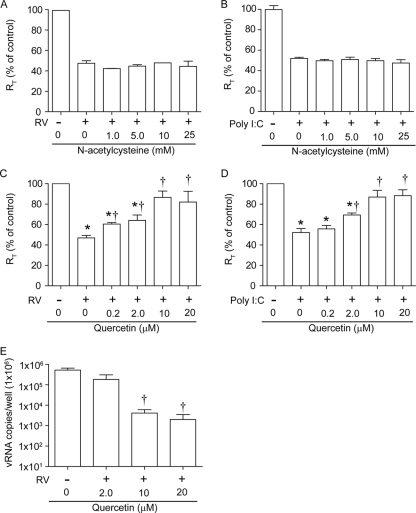
Quercetin, but not N-acetylcysteine, blocks the disruptive effects of RV or poly(I:C) on airway epithelial cell barrier function.
We examined the effects of N-acetylcysteine, an antioxidant that has been used in human trials (10, 27, 46), and quercetin, a flavonoid with both antioxidant and anti-inflammatory properties that may slow the progression of inflammatory lung disease (16, 33, 37), on RV- and poly(I:C)-induced responses. N-Acetylcysteine had no effect on either RV- or poly(I:C)-induced reductions in RT (Fig. 10A and B) and did not affect the viral load (data not shown). In contrast, quercetin blocked the effects of RV and poly(I:C) in a dose-dependent manner, showing maximum effects at 10 μM (Fig. 10C and D). However, quercetin also decreased the vRNA copy number (Fig. 10E). These results suggest that quercetin may block RV-induced effects not only by scavenging ROS but also by decreasing the RV load.
Fig. 10.
Quercetin, but not N-acetylcysteine, blocks disruptive effects of RV and poly(I:C) on RT. Polarized 16HBE14o− cells were infected with RV or UV-RV for 90 min, the infection medium was replaced with medium containing N-acetylcysteine (A) or quercetin (C), and RT was measured 24 h later. Cells were also treated with poly(I:C) in the presence of N-acetylcysteine (B) or quercetin (D), and RT was measured after 8 h. Data are expressed as percentages relative to medium- or UV-RV-treated controls, as appropriate. (E) Total RNA was isolated from cells infected with RV in the presence of quercetin, and the number of copies of vRNA was determined by qPCR. Data represent means ± SEM calculated for 2 or 3 independent experiments carried out in duplicate. *, different from respective UV-RV- or medium-treated controls (P ≤ 0.05); †, different from RV-infected or poly(I:C)-treated cells in the absence of quercetin (P ≤ 0.05 by ANOVA).
DISCUSSION
Unlike influenza virus, RV does not extensively damage the airway epithelium. But RV compromises barrier function (41, 54), potentially increasing permeability of the airway epithelium to allergens and susceptibility to secondary bacterial infections. However, the mechanisms by which RV compromises barrier function are not well understood. In the present study, we demonstrate that ROS generation is required for RV-induced disruptions of barrier function in polarized airway epithelial cells. We also show that RV-induced Rac1-dependent NOX1 activity is required for maximal ROS generation. Finally, we demonstrate for the first time that poly(I:C), a synthetic dsRNA, also disrupts barrier function by inducing ROS generation via activation of NOX1, similar to RV. Together, these results imply that dsRNA, an intermediate formed during RV replication, contributes to ROS generation and the disruption of barrier function.
Previously, we demonstrated that replication of RV is required for the disruption of barrier function (41). dsRNA is one of the intermediates that accumulates during RV replication. A synthetic dsRNA, poly(I:C), has been used extensively as a surrogate for viral dsRNA to study host responses. It provokes inflammatory and interferon (IFN) responses in airway epithelial cells, similar to viral dsRNA (4, 30, 49). Recently, dsRNA was also shown to decrease RT in immortalized nasal epithelial cells (35), but the underlying mechanism was not elucidated. Therefore, to investigate whether dsRNA generated during RV replication contributes to the RV-induced disruption of barrier function, we used poly(I:C) (1.5 to 8 kb) as a surrogate for RV dsRNA. We found that poly(I:C) disrupts the barrier function of polarized airway epithelial cells, similar to RV. We also noted the presence of poly(I:C) within the cells. Since the complete RV replication cycle occurs in the cytoplasm (45), it is reasonable to speculate that dsRNA generated during RV replication will also be in the cytoplasm, similar to poly(I:C). Therefore, it is conceivable that dsRNA generated during RV replication is sufficient to cause disruption of barrier function. However, one cannot rule out the involvement of structural or nonstructural proteins of RV.
Ligation of dsRNA to specific cellular proteins is required for stimulation of IFN or proinflammatory responses. The pattern recognition receptor TLR3 is located primarily in the endosomal membrane and, to some extent, in the plasma membrane. The RNA helicases retinoic acid-inducible gene I (RIG-I), melanoma differentiation-associated gene 5 (MDA5), and LGP2 and the serine/threonine kinase protein kinase R (PKR) are located within the cytoplasm. Previously, we demonstrated with cultured airway epithelial cells that both TLR3 and MDA5 are required for maximal RV-stimulated IFN responses (49) and that TLR3 is partially required for RV-induced IL-8 expression (42). On the other hand, poly(I:C)-induced IFN and cytokine responses were found to be dependent on TLR3 (31). Together, these observations suggest that TLR3 recognizes both poly(I:C) and RV dsRNA, and therefore it is reasonable to postulate that TLR3 may be required for RV- or poly(I:C)-induced disruption of barrier function. Surprisingly, knockdown of TLR3 had no effect on either the RV or poly(I:C) effect on RT, suggesting the involvement of a dsRNA recognition receptor other than TLR3 in this process. Further studies to identify the receptor for dsRNA responsible for barrier disruption are under way.
Since our previous studies indicated that cytokines produced in response to RV infection do not contribute to RV-induced disruption of barrier function, we investigated other mechanisms, in particular the role of ROS. ROS have previously been implicated in the disruption of barrier function in polarized epithelial cells. For instance, ROS generation stimulated by exogenous H2O2 impaired the barrier function of both airway and colonic epithelial cells by causing reorganization of the actin cytoskeleton and dissociation of occludin and ZO-1 from the tight junction complex (6, 38). Since RV has been shown to stimulate ROS generation (5, 23, 36), we reasoned that ROS contribute to RV-induced disruption of tight junctions in polarized airway epithelial cell cultures. Consistent with this notion, we found that the antioxidants DPI and propyl gallate each blocked the disruptive effects of RV on barrier function.
DPI is an inhibitor of ROS-generating flavoenzymes, including NOX enzymes (24), suggesting that one or more NOX isoforms contribute to the RV-induced disruption of barrier function. Previously, RV and dsRNA have each been shown to stimulate mRNA expression of DUOX2, an isoform of NOX, in mucociliary differentiated airway epithelial cells (19). We also observed increased expression of DUOX2 in both RV- and poly(I:C)-treated polarized airway epithelial cells. DUOX2, which is located on the plasma membrane, secretes H2O2 into the extracellular milieu (15); hence, we expected that treatment with catalase would block RV- and poly(I:C)-induced reductions in RT. However, catalase had no effect (data not shown). On the other hand, both RV and poly(I:C) induced robust mRNA expression of NOX1 compared to the other isoforms. Furthermore, inhibition of NOX1 by use of a gene-specific siRNA blocked the reductions in RT by more than 50%. In addition, inhibition of Rac1 also blocked RV- and poly(I:C)-induced disruptions of barrier function. Rac1 is required for the assembly of the NOX1 holoenzyme and for maximal NOX1 activity (8, 29). Finally, inhibition of NOX1 and Rac1 each partially attenuated the generation of ROS stimulated by RV and poly(I:C). Together, our results suggest that both RV infection and poly(I:C) treatment increase expression of NOX1 and assembly of the NOX1 holoenzyme, which requires Rac1 activity, and that these effects are responsible, at least in part, for RV- and poly(I:C)-induced disruptions of barrier function in polarized airway epithelial cell cultures.
In addition to NOX, ROS are also produced by other enzymes, such as the mitochondrial respiratory chain enzymes and xanthine oxidase. In nonpolarized airway epithelial cells, RV was shown to stimulate ROS by activating xanthine oxidase (36). In the latter study, ROS increased within 20 min of infection and reached a maximum by 60 min, suggesting that binding of virus was sufficient for ROS generation. This effect was completely inhibited by pretreatment of cells with oxypurinol, an inhibitor of xanthine oxidase, but not by pretreatment with DPI. In the present study, a significant increase in ROS production was not observed in polarized airway epithelial cells until 8 h after RV infection. Furthermore, UV-RV, which stimulates host responses associated with binding and endocytosis similarly to intact RV, did not stimulate ROS generation. In addition, oxypurinol had no effect on either RV-induced ROS generation or the observed reduction in RT. These results suggest that RV may not stimulate the activity of xanthine oxidase in polarized airway epithelial cells. Finally, it should be noted that DPI inhibits the activity not only of NOX but also of enzymes comprising the mitochondrial respiratory chain (28). Since DPI completely blocks both ROS generation and disruption of barrier function caused by RV, it is possible that ROS generated by the mitochondrial respiratory chain also contribute to the effects of RV on barrier function.
ROS generated by professional phagocytes in response to infection are beneficial for the host because they participate in direct killing of infecting microbes. However, in nonphagocytic cells, ROS regulate a variety of physiological responses, including cell proliferation, apoptosis, and immune and proinflammatory responses (reviewed in reference 12). ROS generation may not always be beneficial to the host, particularly during viral infections. Here we show that ROS produced by NOX1 disrupt the barrier function of RV-infected airway epithelial cells. On a similar note, ROS production via NOX2 was proposed to cause excessive inflammation during respiratory syncytial virus (RSV) infection (14). In influenza virus-infected mice, an absence of NOX2 led to improved viral clearance, an increased Th1 response, and decreased airway inflammation (44). ROS also inhibit IFN-α-induced antiviral gene expression in human hepatoma cells infected with hepatitis C virus (11). In light of these observations, we speculate that increased ROS generation during viral infection may compromise innate immune defense mechanisms, and therefore treatment with antioxidants may prevent oxidative damage and reduce susceptibility to secondary bacterial infections and permeability to allergens. Indeed, our preliminary studies showed that quercetin, a potent free radical scavenger, blocked the disruptive effect of RV on airway epithelial barrier function. On the other hand, N-acetylcysteine, a precursor of glutathione that has been used in clinical trials to inhibit influenza symptoms and to treat chronic bronchitis (10, 27, 46), had no effect on RV-induced reductions in barrier function. Further studies are required to assess the potential beneficial effects of antioxidants in vivo.
ACKNOWLEDGMENTS
This work was supported by National Institutes of Health grants HL897720 and AT4793 (to U.S.S.).
Footnotes
Published ahead of print on 20 April 2011.
REFERENCES
- 1. Arruda E., Pitkaranta A., Witek T. J., Jr., Doyle C. A., Hayden F. G. 1997. Frequency and natural history of rhinovirus infections in adults during autumn. J. Clin. Microbiol. 35:2864–2868 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Avadhanula V., et al. 2006. Respiratory viruses augment the adhesion of bacterial pathogens to respiratory epithelium in a viral species- and cell type-dependent manner. J. Virol. 80:1629–1636 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Bedard K., Krause K. H. 2007. The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol. Rev. 87:245–313 [DOI] [PubMed] [Google Scholar]
- 4. Berube J., Bourdon C., Yao Y., Rousseau S. 2009. Distinct intracellular signaling pathways control the synthesis of IL-8 and RANTES in TLR1/TLR2, TLR3 or NOD1 activated human airway epithelial cells. Cell. Signal. 21:448–456 [DOI] [PubMed] [Google Scholar]
- 5. Biagioli M. C., Kaul P., Singh I., Turner R. B. 1999. The role of oxidative stress in rhinovirus induced elaboration of IL-8 by respiratory epithelial cells. Free Radic. Biol. Med. 26:454–462 [DOI] [PubMed] [Google Scholar]
- 6. Boardman K. C., Aryal A. M., Miller W. M., Waters C. M. 2004. Actin re-distribution in response to hydrogen peroxide in airway epithelial cells. J. Cell. Physiol. 199:57–66 [DOI] [PubMed] [Google Scholar]
- 7. Calvo C., et al. 2007. Role of rhinovirus in hospitalized infants with respiratory tract infections in Spain. Pediatr. Infect. Dis. J. 26:904–908 [DOI] [PubMed] [Google Scholar]
- 8. Cheng G., Diebold B. A., Hughes Y., Lambeth J. D. 2006. Nox1-dependent reactive oxygen generation is regulated by Rac1. J. Biol. Chem. 281:17718–17726 [DOI] [PubMed] [Google Scholar]
- 9. Contoli M., et al. 2009. Viral infections in exacerbations of asthma and chronic obstructive pulmonary disease. Minerva Med. 100:467–478 [PubMed] [Google Scholar]
- 10. De Flora S., Grassi C., Carati L. 1997. Attenuation of influenza-like symptomatology and improvement of cell-mediated immunity with long-term N-acetylcysteine treatment. Eur. Respir. J. 10:1535–1541 [DOI] [PubMed] [Google Scholar]
- 11. Di Bona D., et al. 2006. Oxidative stress inhibits IFN-alpha-induced antiviral gene expression by blocking the JAK-STAT pathway. J. Hepatol. 45:271–279 [DOI] [PubMed] [Google Scholar]
- 12. Droge W. 2002. Free radicals in the physiological control of cell function. Physiol. Rev. 82:47–95 [DOI] [PubMed] [Google Scholar]
- 13. Edwards M. R., et al. 2007. Protein kinase R, IkappaB kinase-beta and NF-kappaB are required for human rhinovirus induced pro-inflammatory cytokine production in bronchial epithelial cells. Mol. Immunol. 44:1587–1597 [DOI] [PubMed] [Google Scholar]
- 14. Fink K., Duval A., Martel A., Soucy-Faulkner A., Grandvaux N. 2008. Dual role of NOX2 in respiratory syncytial virus- and Sendai virus-induced activation of NF-kappaB in airway epithelial cells. J. Immunol. 180:6911–6922 [DOI] [PubMed] [Google Scholar]
- 15. Forteza R., Salathe M., Miot F., Conner G. E. 2005. Regulated hydrogen peroxide production by Duox in human airway epithelial cells. Am. J. Respir. Cell Mol. Biol. 32:462–469 [DOI] [PubMed] [Google Scholar]
- 16. Ganesan S., et al. 2010. Quercetin prevents progression of disease in elastase/LPS-exposed mice by negatively regulating MMP expression. Respir. Res. 11:131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Gao Y., Dickerson J. B., Guo F., Zheng J., Zheng Y. 2004. Rational design and characterization of a Rac GTPase-specific small molecule inhibitor. Proc. Natl. Acad. Sci. U. S. A. 101:7618–7623 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Gerna G., et al. 2009. Correlation of rhinovirus load in the respiratory tract and clinical symptoms in hospitalized immunocompetent and immunocompromised patients. J. Med. Virol. 81:1498–1507 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Harper R. W., et al. 2005. Differential regulation of dual NADPH oxidases/peroxidases, Duox1 and Duox2, by Th1 and Th2 cytokines in respiratory tract epithelium. FEBS Lett. 579:4911–4917 [DOI] [PubMed] [Google Scholar]
- 20. Ishizuka S., et al. 2003. Effects of rhinovirus infection on the adherence of Streptococcus pneumoniae to cultured human airway epithelial cells. J. Infect. Dis. 188:1928–1939 [DOI] [PubMed] [Google Scholar]
- 21. Jakiela B., Brockman-Schneider R., Amineva S., Lee W. M., Gern J. E. 2008. Basal cells of differentiated bronchial epithelium are more susceptible to rhinovirus infection. Am. J. Respir. Cell Mol. Biol. 38:517–523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Johnston S. L., Tyrrell D. A. J. 1997. Rhinoviruses, p. 533–563 In Lennette E. H., Schmidt N. J. (ed.), Diagnostic procedures for viral, rickettsial, and chlamydial infections. American Public Health Association, Washington, DC [Google Scholar]
- 23. Kaul P., Biagioli M. C., Singh I., Turner R. B. 2000. Rhinovirus-induced oxidative stress and interleukin-8 elaboration involves p47-phox but is independent of attachment to intercellular adhesion molecule-1 and viral replication. J. Infect. Dis. 181:1885–1890 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Kim H., et al. 2008. TNF-alpha-induced up-regulation of intercellular adhesion molecule-1 is regulated by a Rac-ROS-dependent cascade in human airway epithelial cells. Exp. Mol. Med. 40:167–175 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Kim J. Y., Sajjan U. S., Krasan G. P., LiPuma J. J. 2005. Disruption of tight junctions during traversal of the respiratory epithelium by Burkholderia cenocepacia. Infect. Immun. 73:7107–7112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Kuo C., et al. 2011. Rhinovirus infection induces expression of airway remodelling factors in vitro and in vivo. Respirology 16:367–377 [DOI] [PubMed] [Google Scholar]
- 27. Lai K. Y., Ng W. Y., Osburga Chan P. K., Wong K. F., Cheng F. 2010. High-dose N-acetylcysteine therapy for novel H1N1 influenza pneumonia. Ann. Intern. Med. 152:687–688 [DOI] [PubMed] [Google Scholar]
- 28. Lambert A. J., Buckingham J. A., Boysen H. M., Brand M. D. 2008. Diphenyleneiodonium acutely inhibits reactive oxygen species production by mitochondrial complex I during reverse, but not forward electron transport. Biochim. Biophys. Acta 1777:397–403 [DOI] [PubMed] [Google Scholar]
- 29. Leto T. L., Morand S., Hurt D., Ueyama T. 2009. Targeting and regulation of reactive oxygen species generation by nox family NADPH oxidases. Antioxid. Redox Signal. 11:2607–2619 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Matsukura S., et al. 2007. Role of RIG-I, MDA-5, and PKR on the expression of inflammatory chemokines induced by synthetic dsRNA in airway epithelial cells. Int. Arch. Allergy Immunol. 143(Suppl. 1):80–83 [DOI] [PubMed] [Google Scholar]
- 31. Matsumoto M., Seya T. 2008. TLR3: interferon induction by double-stranded RNA including poly(I:C). Adv. Drug Deliv. Rev. 60:805–812 [DOI] [PubMed] [Google Scholar]
- 32. McManus T. E., et al. 2008. Respiratory viral infection in exacerbations of COPD. Respir. Med. 102:1575–1580 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Nanua S., et al. 2006. Quercetin blocks airway epithelial cell chemokine expression. Am. J. Respir. Cell Mol. Biol. 35:602–610 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Newcomb D. C., et al. 2005. Phosphatidylinositol 3-kinase is required for rhinovirus-induced airway epithelial cell interleukin-8 expression. J. Biol. Chem. 280:36952–36961 [DOI] [PubMed] [Google Scholar]
- 35. Ohkuni T., et al. 2011. Poly(I:C) reduces expression of JAM-A and induces secretion of IL-8 and TNF-alpha via distinct NF-kappaB pathways in human nasal epithelial cells. Toxicol. Appl. Pharmacol. 250:29–38 [DOI] [PubMed] [Google Scholar]
- 36. Papi A., et al. 2008. Role of xanthine oxidase activation and reduced glutathione depletion in rhinovirus induction of inflammation in respiratory epithelial cells. J. Biol. Chem. 283:28595–28606 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Rahman I. 2008. Antioxidant therapeutic advances in COPD. Ther. Adv. Respir. Dis. 2:351–374 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Rao R. 2008. Oxidative stress-induced disruption of epithelial and endothelial tight junctions. Front. Biosci. 13:7210–7226 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Rao R. K., Baker R. D., Baker S. S., Gupta A., Holycross M. 1997. Oxidant-induced disruption of intestinal epithelial barrier function: role of protein tyrosine phosphorylation. Am. J. Physiol. 273:G812–G823 [DOI] [PubMed] [Google Scholar]
- 40. Saenz-Morales D., et al. 2006. Requirements for proximal tubule epithelial cell detachment in response to ischemia: role of oxidative stress. Exp. Cell Res. 312:3711–3727 [DOI] [PubMed] [Google Scholar]
- 41. Sajjan U., Wang Q., Zhao Y., Gruenert D. C., Hershenson M. B. 2008. Rhinovirus disrupts the barrier function of polarized airway epithelial cells. Am. J. Respir. Crit. Care Med. 178:1271–1281 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Sajjan U. S., et al. 2006. H. influenzae potentiates airway epithelial cell responses to rhinovirus by increasing ICAM-1 and TLR3 expression. FASEB J. 20:2121–2123 [DOI] [PubMed] [Google Scholar]
- 43. Schneider D., et al. 2010. Increased cytokine response of rhinovirus-infected airway epithelial cells in chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 182:332–340 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Snelgrove R. J., Edwards L., Rae A. J., Hussell T. 2006. An absence of reactive oxygen species improves the resolution of lung influenza infection. Eur. J. Immunol. 36:1364–1373 [DOI] [PubMed] [Google Scholar]
- 45. Steil B. P., Barton D. J. 2009. Cis-active RNA elements (CREs) and picornavirus RNA replication. Virus Res. 139:240–252 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Stey C., Steurer J., Bachmann S., Medici T. C., Tramer M. R. 2000. The effect of oral N-acetylcysteine in chronic bronchitis: a quantitative systematic review. Eur. Respir. J. 16:253–262 [DOI] [PubMed] [Google Scholar]
- 47. van Ewijk B. E., et al. 2008. Prevalence and impact of respiratory viral infections in young children with cystic fibrosis: prospective cohort study. Pediatrics 122:1171–1176 [DOI] [PubMed] [Google Scholar]
- 48. Wang J. H., Kwon H. J., Jang Y. J. 2009. Rhinovirus enhances various bacterial adhesions to nasal epithelial cells simultaneously. Laryngoscope 119:1406–1411 [DOI] [PubMed] [Google Scholar]
- 49. Wang Q., et al. 2009. Role of double-stranded RNA pattern recognition receptors in rhinovirus-induced airway epithelial cell responses. J. Immunol. 183:6989–6997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Wat D., et al. 2008. The role of respiratory viruses in cystic fibrosis. J. Cyst. Fibros. 7:320–328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Waters C. M., Savla U., Panos R. J. 1997. KGF prevents hydrogen peroxide-induced increases in airway epithelial cell permeability. Am. J. Physiol. 272:L681–L689 [DOI] [PubMed] [Google Scholar]
- 52. Welsh M. J., Shasby D. M., Husted R. M. 1985. Oxidants increase paracellular permeability in a cultured epithelial cell line. J. Clin. Invest. 76:1155–1168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Yamaya M., et al. 1995. Oxidants affect permeability and repair of the cultured human tracheal epithelium. Am. J. Physiol. 268:L284–L293 [DOI] [PubMed] [Google Scholar]
- 54. Yeo N. K., Jang Y. J. 2010. Rhinovirus infection-induced alteration of tight junction and adherens junction components in human nasal epithelial cells. Laryngoscope 120:346–352 [DOI] [PubMed] [Google Scholar]
- 55. Yoboua F., Martel A., Duval A., Mukawera E., Grandvaux N. 2010. Respiratory syncytial virus-mediated NF-kappa B p65 phosphorylation at serine 536 is dependent on RIG-I, TRAF6, and IKK beta. J. Virol. 84:7267–7277 [DOI] [PMC free article] [PubMed] [Google Scholar]