Abstract
In this study, new strains were isolated from an environment with elevated arsenic levels, Sainte-Marie-aux-Mines (France), and the diversity of aoxB genes encoding the arsenite oxidase large subunit was investigated. The distribution of bacterial aoxB genes is wider than what was previously thought. AoxB subfamilies characterized by specific signatures were identified. An exhaustive analysis of AoxB sequences from this study and from public databases shows that horizontal gene transfer has likely played a role in the spreading of aoxB in prokaryotic communities.
TEXT
Arsenic, which is one of the most toxic metalloids, is distributed ubiquitously but not uniformly around the world. Levels of arsenic differ considerably from one geographical region to another, depending on the geochemical characteristics of the soil (natural contamination) and the industrial activities carried out in the vicinity (anthropogenic contamination) (22). In aquatic environments, arsenic occurs mainly in the form of the inorganic species arsenate [As(V)] and arsenite [As(III)]; the latter species, which is more bioavailable, is usually thought to have more-toxic effects on prokaryotes than As(V) (34). As(III) oxidation leads to the formation of the less available form As(V), which can either precipitate with iron [Fe(III)] or be adsorbed by ferrihydrite. The oxidation process may be mediated by microbial activities, which contribute to the natural remediation processes observed in contaminated environments (21, 26, 27, 34). Consequently, bioprocesses for the treatment of arsenic-contaminated waters have been developed based on the precipitation or adsorption of the As(V) produced by bacteria (4, 9, 21). Some well-known prokaryotes oxidize As(III) into As(V) under aerobic (e.g., Herminiimonas arsenicoxydans, Thiomonas spp., or Rhizobium sp. strain NT26) or anaerobic (e.g., Alkalilimnicola ehrlichii) conditions as part of a detoxification process (12, 17, 31, 32, 39). Some chemolithotrophs also use arsenite as an electron donor (e.g., Rhizobium sp. strain NT26 or Thiomonas arsenivorans) (5, 32). The aerobic arsenite oxidases involved in such processes are heterodimers consisting of a large subunit with a molybdenum center and a [3Fe-4S] cluster (AroA, AsoA, and AoxB) and a small subunit containing a Rieske-type [2Fe-2S] cluster (AroB, AsoB, and AoxA) (1, 13). The large subunit in these enzymes is similar to that found in other members of the dimethyl sulfoxide (DMSO) reductase family of molybdenum enzymes but is clearly phylogenetically divergent from the respiratory arsenate reductases (ArrA) or other proteins of the DMSO reductase family of molybdenum oxidoreductases, such as the new arsenite reductase described recently for Alkalilimnicola ehrlichii (25, 31, 40).
aox genes have been identified in 25 bacterial and archaeal genera isolated from various arsenic-rich environments, most of which belong to the Alpha-, Beta-, or Gammaproteobacteria phylum (7, 10, 12, 14, 23, 25, 29, 32, 37). Recent studies based on environmental DNA extracted from soils, sediments, and geothermal mats with different chemical characteristics and various levels of arsenic contamination have suggested that the distribution and the diversity of arsenite-oxidizing microorganisms may be greater than previously suggested (6, 10, 14–16, 18, 28, 29). The aim of this study was to investigate the presence of the aox gene in bacteria other than the Proteobacteria in order to improve our knowledge about the phylogeny and evolution of AoxB. With this aim, an environment with elevated arsenic levels showing considerable prokaryote diversity was chosen. The aoxB sequences studied here were amplified from the DNA of bacteria isolated from sediments collected at this site and from the DNA extracted directly from these sediments.
The study site is located in the vicinity of the Gabe-Gottes mine in the Sainte-Marie-aux-Mines valley (France) (48°12′51"N, 007°09′34"E) (15a). Water percolating from the mine walls accumulates in a creek, where sediments were collected in December 2007, 40 m downstream of the entrance to the mine, from a 30-m-long part of the creek. In these sediment samples, where the total arsenic level was 320 mg kg−1, the As(V) species concentration was 10 times greater than the As(III) species (15a). The main arsenic species observed in sedimentary interstitial water were As(III) and As(V), in concentrations of 14.6 (±0.3) μg liter−1 and 135 (±5) μg liter−1, respectively. Such concentrations correspond to moderately high arsenic levels; our samples were either more severely contaminated than or similarly contaminated relative to most of previously studied sites at which arsenic oxidase-encoding genes have been detected (see Table S1 in the supplemental material) (3, 6, 8, 10, 14–16, 18, 29, 37). Moreover, the sediments at this site were previously found to show considerable bacterial diversity (15a). These characteristics made these samples suitable for investigation of the diversity of aox genes.
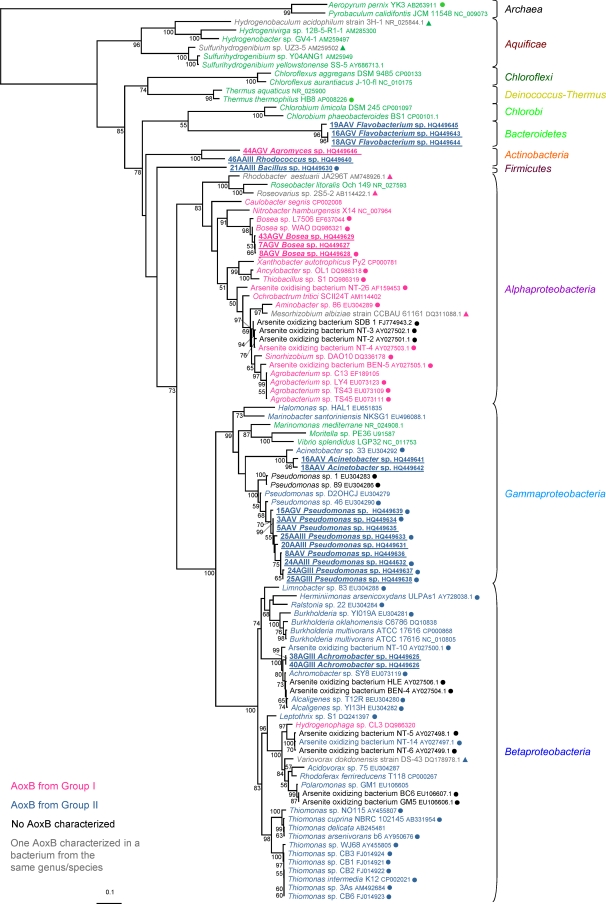
In the present study, aoxB genes were amplified from the total sediment DNA and from DNA extracted from several arsenic-resistant isolates. These bacterial isolates were grown for 18 days at 20°C on DR2A medium, for which the composition was as follows (per liter): 0.05 g each of yeast extract, peptone, acid hydrolysate of casein, dextrose, and soluble starch, 0.03 g each of dipotassium phosphate and sodium pyruvate, and 0.0024 g of magnesium sulfate (36) amended with either As(III) or As(V) [isolates named with the suffix III or V refer to the presence of As(III) or As(V), respectively] under aerobic or anaerobic conditions (isolates named with the prefix A refer to aerobic conditions) using the Anaerocult P system (Grosseron) (the middle letter, A or G, corresponds to the gelling agent, i.e., agar or gellan gum, respectively). Among the 220 isolates obtained and tested using BM1-2F/BM3-1R primers (see Table S2 in the supplemental material) to detect the presence of the aoxB gene, positive PCR amplification was obtained for 22 aerobe strains (see Table S3 in the supplemental material) and no aoxB amplification was observed for anaerobe isolates. 16S rRNA genes from these isolates were amplified and sequenced using primers W01/W02 (see Table S2). These 22 isolates belong to eight genera (Achromobacter, Pseudomonas, Agromyces, Rhodococcus, Flavobacterium, Bosea, Acinetobacter, and Bacillus), representing four bacterial phyla (Firmicutes, Actinobacteria, Bacteroidetes, and Proteobacteria) (Fig. 1 [underlined, bold names]; see also Table S3). To our knowledge, this is the first time aoxB genes have been reported to occur in Bacillus, Rhodococcus, Agromyces, and Flavobacterium spp. Positive arsenite oxidase activity was detected with the AgNO3 method (35) using H. arsenicoxydans ULPAs1 as a positive control (39) in the case of 8 isolates, 6 of which belong to the Pseudomonas genus, 1 to the Bacillus genus, and 1 to the Bosea genus (Fig. 1 [underlined, bold names highlighted with circles]). The absence of activity in the other strains may be due to a lack of aox gene expression under the conditions tested, the lack of the small subunit, or a gene coding for a nonfunctional enzyme. Arsenite oxidation activity has been observed in previous studies in the Pseudomonas, Acinetobacter, Achromobacter, and Bosea genera (7, 28–30) (Fig. 1 [indicated with circles]).
Fig. 1.
Maximum likelihood phylogenetic tree of the 16S rRNA sequences of our 22 isolates showing positive aoxB PCR amplification. This tree also includes 16S rRNA sequences deposited in public databases, corresponding to the organisms found (or close relatives) to carry the aoxB gene or closely related organisms. The tree was rooted in between Archaea and Bacteria domains. Phylogenetic analyses were performed on the 1,014 unambiguously aligned nucleic acid positions by using Treefinder (19). The evolutionary model GTR + Γ4 was used for this purpose, as suggested by the “propose model” tool available in Treefinder. Numbers at branches are bootstrap values obtained using the nonparametric bootstrap approach implemented in Treefinder (based on 100 replicates of the original data set). Only bootstrap values above 50% are shown. Bacteria harboring aoxB sequences corresponding to phylogenetic group I or group II or neither of these two groups are given in pink, dark blue, or green, respectively. The 22 bacterial isolates of this study are in boldface and underlined. All bacteria experimentally shown to be able to oxidize arsenite are labeled with a circle at the end of the name. Bacteria which were previously shown to be able to oxidize arsenite or harbor an aoxB gene, but for which no 16S sequence was available, were therefore not included in this tree. However, in this case, we included the closest relative of these strains for which a 16S gene was available (in gray), and the triangle color at the end of these names corresponds to the group to which the aoxB gene harbored by the strain belongs (pink, dark blue, and green corresponding to group I, group II, and neither of these groups, respectively). The names of bacteria which were described in the literature as able to oxidize arsenite but for which aoxB sequences were not available are presented in black. The scale bar represents the average number of substitutions estimated per site.
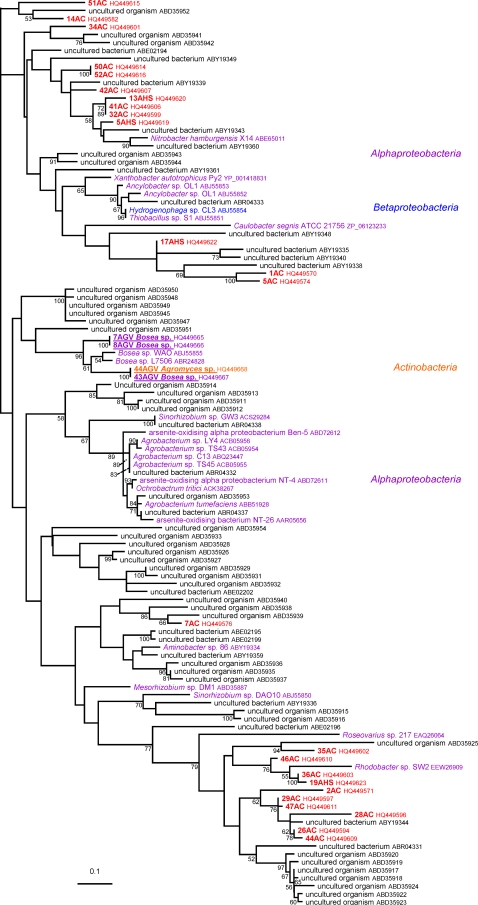
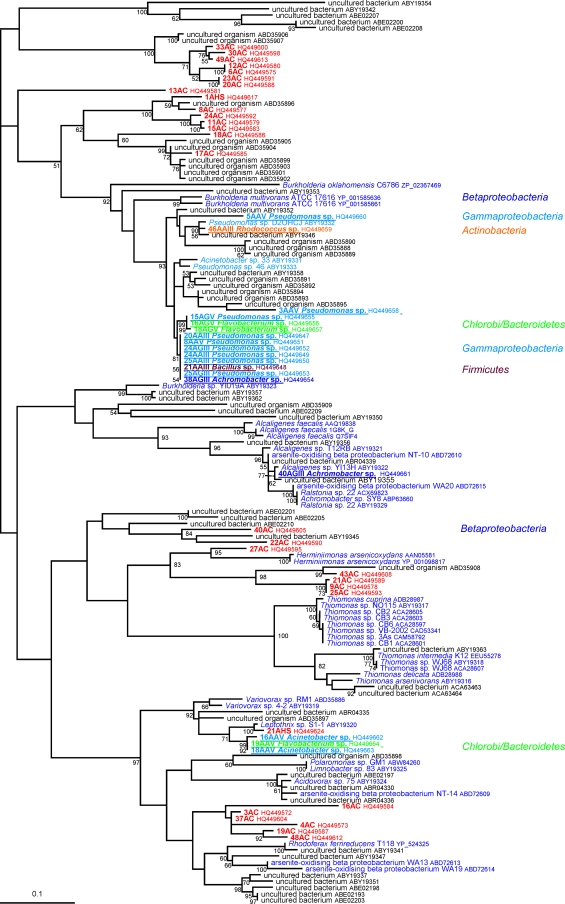
The phylogenetic analysis of the 22 deduced AoxB amino acid sequences and their homologues present in the nonredundant protein database at the NCBI was performed. A preliminary phylogenetic analysis (data not shown) helped us to select only aerobic arsenite oxydase (AoxB, AroA, or AsoA) sequences and to exclude respiratory arsenate reductases (ArrA) or other proteins of the DMSO reductase family of molybdenum oxidoreductases, such as the new arsenite reductase recently described for Alkalilimnicola ehrlichii, which are clearly phylogenetically divergent from the AoxB/AsoA/AroA proteins, as previously shown (25, 40). The phylogenetic analysis of these AoxB amino acid sequences showed that most of them belonged to two major groups (see Fig. S1 in the supplemental material), as recently described (29). Group I sequences were found mainly in Alphaproteobacteria, whereas group II was composed mainly of betaproteobacterial sequences (Fig. 2 and 3; see also Fig. S1). This pattern suggests that these two groups probably originated from these two proteobacterial divisions. AoxB sequences belonging to none of these two groups were found in a few representatives of various bacterial divisions and a few archaeal phyla (see Fig. S1). Among the 22 AoxB characterized here, four were found to belong to group I. More specifically, three of them were amplified from isolates classified as Bosea sp. (Alphaproteobacteria) and one as Agromyces sp. (Actinobacteria) (Fig. 2). The 18 remaining AoxB sequences belong to group II. These sequences were amplified from isolates affiliated with Betaproteobacteria (Achromobacter sp.), Gammaproteobacteria (Pseudomonas sp.), Actinobacteria (Rhodococcus sp.), Firmicutes (Bacillus sp.), and Bacteroidetes (Flavobacterium sp.) (Fig. 3).
Fig. 2.
Unrooted phylogenetic trees of the 86 group I AoxB sequences (152 unambiguously aligned positions). The 86 sequences were retrieved from the nonredundant database of the NCBI, deduced from aoxB genes from 4 isolated strains (in bold and underlined) or deduced from the 24 PCR products obtained from metagenomic DNA (in red). Phylogenetic analyses were performed using the maximum likelihood approach implemented in Treefinder (19). The LG + Γ4 model was used for this purpose, as suggested by the “propose model” tool available in Treefinder. Numbers at branches are bootstrap values determined using the nonparametric bootstrap approach implemented in Treefinder (based on 100 replicates of the original data set). The scale bar represents the average number of substitutions estimated per site.
Fig. 3.
Unrooted phylogenetic trees of the 97 group II AoxB sequences (260 unambiguously aligned positions). The 97 sequences were retrieved from the nonredundant database of the NCBI, deduced from 18 isolated strains (in bold and underlined) or deduced from the 29 PCR products obtained from metagenomic DNA (in red). Phylogenetic analyses were performed using the maximum likelihood approach implemented in Treefinder (19). The LG +Γ4 model was used for this purpose, as suggested by the “propose model” tool available in Treefinder. Numbers at branches are bootstrap values determined using the nonparametric bootstrap approach implemented in Treefinder (based on 100 replicates of the original data set). The scale bar represents the average number of substitutions estimated per site.
Comparisons between the 16S rRNA and AoxB phylogenies showed the existence of some striking inconsistencies between organism and gene evolutionary histories (Fig. 1, 2, and 3; see also Fig. S1 in the supplemental material). These can be explained by four hypotheses: (i) hidden paralogies (i.e., gene duplications followed by differential gene losses), (ii) allele sorting, (iii) horizontal gene transfer (HGT), and (iv) tree reconstruction artifacts. In the case of AoxB, a few HGT events represent the most likely hypothesis to explain the presence of identical (or nearly identical) AoxB sequences in some distant lineages, such as group I AoxB sequences found in Agromyces and Bosea, two unrelated bacteria that belong respectively to Actinobacteria and Alphaproteobacteria (Fig. 2). Moreover, in addition to including sequences from Alphaproteobacteria, group I included one betaproteobacterial AoxB sequence (Hydrogenophaga sp. strain CL3) (Fig. 2), which indicates that this bacterium has very likely acquired its aoxB gene from Alphaproteobacteria via HGT. A similar process may account for the sequence observed in the Agromyces sp. isolated in this study (Fig. 2). In group II, some Pseudomonas, Marinobacter, Halomonas, and also Firmicutes and Chlorobi/Bacteroidetes AoxB sequences were intermixed with those from Betaproteobacteria (Fig. 3). This observation strongly suggests that the former acquired their aoxB gene from the latter by HGT. More convincingly, one of the two actinobacterial sequences characterized in this study belonged to group I, whereas the second sequence belonged to group II (Fig. 2 and 3), although the strains from which these sequences were amplified are closely related (Fig. 1). This suggests that these two aoxB genes were acquired by HGT from two unrelated proteobacterial donors. The great similarity observed between some AoxB sequences, such as those from Bacillus (isolate 21AAIII), Flavobacterium (isolates 16AGV and 18AGV) and Pseudomonas, indicates that the HGT events probably occurred quite recently (Fig. 3). HGT in prokaryotic communities has been proposed previously to explain the strong similarities observed between AoxB proteins from Ralstonia spp. and Achromobacter sp. strain SY8 (GenBank accession numbers ACX69823 and ABP63660) (20). In that previous study, it was suggested that Ralstonia has acquired the corresponding gene cluster by HGT from another betaproteobacterium (20). The proximity of these AoxB proteins in the AoxB phylogenetic tree (Fig. 3) strengthens this proposal to explain the presence of nearly identical AoxB sequences in these two Betaproteobacteria. Interestingly, the present work revealed for the first time that such HGT may have occurred between bacteria belonging to different classes or phyla.
Genomic islands (GEI) or plasmids may be involved in such transfers. This is, for instance, the case for aoxB genes found in Thiomonas (Fig. 3) that are localized in one genomic island (2). We searched for other examples of genomic islands containing aoxB belonging to group I (Nitrobacter hamburgensis X14 [Alphaproteobacteria]) (Fig. 2) or group II (Thiomonas strain 3As, Thiomonas intermedia K12, and H. arsenicoxydans [three Betaproteobacteria]) (Fig. 3) or outside these two groups (Vibrio splendicus [Gammaproteobacteria], Aeropyrum pernix, and Pyrobaculum calidifontis [Archaea], and Thermus thermophilus [Thermus/Deinococcus]) (see Fig. S1 in the supplemental material). With this aim, we used the RGPfinder (for regions of genomic plasticity [RGP]) tool of the Microscope platform (https://www.genoscope.cns.fr/agc/microscope/) (38). Interestingly, aoxB genes were present in RGP in H. arsenicoxydans as previously proposed (24), in V. splendidus and P. calidifontis (see Fig. S2 in the supplemental material). In addition, aox genes were carried on plasmids in N. hamburgensis X14 (plasmid 2, NC_007960) and T. thermophilus HB8 (pTT27, NC_006462). In the case of this microorganism, a transposase-encoding gene was found in the vicinity of the aoxAB genes (in green in Fig. S2 in the supplemental material). Finally, whereas the aox operon is present in a GEI in Thiomonas strain 3As (2), the orthologous genes were not found in an RGP in Thiomonas intermedia. In fact, the synteny between these two strains is restricted to the aoxABD and ptxB-aoxRS operons. More generally, the synteny of aoxAB genes is conserved among all bacteria for which a genome sequence was available (see Fig. S2). In contrast, synteny with the other aox genes, aoxC and aoxD or aoxRS, was observed only in few bacteria belonging to group II (Thiomonas sp. strain 3As and H. arsenicoxydans), whose aoxB genes are related (Fig. 3), and in Xanthobacter autotrophicus. Synteny of aoxAB with aoxR or aoxS was conserved in several bacteria belonging to group II or to group I. These observations suggest that the synteny conservation is not linked to the aoxB phylogeny observed or to a specific energy/carbon metabolism (heterotrophs versus autotrophs).
The phylogenic analyses support the hypothesis that HGTs have played a role in the widespread distribution of some aoxB genes in prokaryotes, meaning that the aoxB gene is not an efficient phylogenetic marker. Indeed, because of the discrepancies found to exist between the 16S rRNA and aoxB phylogenies, it is not possible to predict the taxonomic affiliation of strains based on the aoxB sequences alone. This precludes the use of approaches involving the direct amplification of aoxB in order to characterize the arsenite oxidizers from environmental samples. This is the case for the 56 aoxB sequences obtained in this study from the total DNA (shown in red in Fig. 2 and 3). As in the case of the sequences obtained from cultivated isolates, most of these deduced AoxB proteins belonged to group I (24 sequences) and group II (29 sequences). In contrast, only three of the characterized sequences were similar to sequences from Chloroflexi and Thermus, which branched outside these two groups (data not shown). Interestingly, most of our AoxB sequences belonging to groups I and II were found to be closely related to sequences from hitherto uncultured and/or unidentified bacteria or to constitute new subgroups (Fig. 2 and 3). This situation clearly shows that the diversity of AoxB is far from having been exhaustively investigated.
The comparisons between 78 AoxB proteins characterized in this study, including the most divergent sequences, showed that residues present in the catalytic site [21C-X2-24C-X3-28C-X70-99S] according to the betaproteobacterium Alcaligenes faecalis NCIB 8687 protein sequence numbering (13) (UniProt accession number Q7SIF4), and the hydrophilic channel near the Mo centers that was proposed to be the substrate binding site (from amino acid positions 195 to 203 according to the A. faecalis AoxB) were conserved in 76 of the 78 sequences studied (data not shown; see also Fig. S3 in the supplemental material). This suggested that when they are expressed, these AoxB proteins might function like the A. faecalis arsenite oxidase, for which a crystal structure was determined (13). Close comparisons between all the AoxB sequences belonging to groups I and II showed that four motifs differ between these two groups (Fig. 4; see also Fig. S3 in the supplemental material). These four regions extend from amino acid positions 65 to 70, 76 to 84, 133 to 145, and 182 to 191 (according to A. faecalis AoxB protein sequence numbering). They contain the motifs W-[YF]-[SAP]-[PA]-[SA]-M-[YH] and [MLVA]-[TS]-X-[AT]-X2, Q-X-G-X-[DN]-[VM]-[HNQ]-X-V and X2-G-X4-[IV]-[ML], P-T-X-W-X-D-A-[LFP]-[DS]-L-V-[AT]-X and X3-W-X2-A-[LM]-X2-Y-X-G or L-Y-F-X-[AS]-[MIL]-[KR]-[VI]-[KR]-[NH] and L-[MIVT]-[FS]-X3-[QKR]-T-[PQT]-X in the case of group I sequences and group II sequences, respectively (according to the PROSITE pattern nomenclature) (Fig. 4). These motifs were not detected in other AoxB sequences and are therefore specific to each of these groups.
Fig. 4.
Group I and group II-specific motifs deduced from AoxB sequence alignment with the WebLogo software program via the WebLogo website (http://weblogo.berkeley.edu/) (11, 33). Residues with which these motifs could be unambiguously defined are in bold. The absence of these motifs in AoxB sequences belonging to neither group I nor group II was checked visually. Amino acid numbering was based on the Alcaligenes faecalis protein sequence (UniProt accession number Q7SIF4).
In conclusion, the results of these studies on a mildly arsenic-contaminated environment by using cell culture-dependent and -independent approaches show that the aoxB gene diversity is wider than described in previous studies. Comparisons between 78 AoxB bacterial sequences obtained from the Sainte-Marie-aux-Mines creek sedimentary microbial community and homologues previously deposited in public databases confirmed that most of the aoxB sequences detected so far belong to two subfamilies (groups I and II) harboring specific signatures. It is worth noting that the pattern of aox gene evolution was not strictly correlated with organism evolution and that AoxB was not found to be a suitable phylogenetic marker for studying the microbial diversity of arsenic-contaminated environments.
Nucleotide sequence accession numbers.
Sequences were submitted to the GenBank database with the following accession numbers: HQ449625 to HQ449646 for 16S rRNA genes, HQ449647 to HQ449668 for aoxB from isolates, and HQ449570 to HQ449624 for aoxB from metagenomic DNA.
Supplementary Material
Acknowledgments
Audrey Cordi was supported by grants of the French Ministry of Research and the Lorraine Region (the Ecotoxicology Platform). Audrey Heinrich-Salmeron and David Halter were supported by grants of the French Ministry of Research and the University of Strasbourg. This study was financed by the EC2CO program (Institut National des Sciences de l'Univers, CNRS). It was performed in the framework of the Groupement de Recherche Métabolisme de l'Arsenic Chez les Micro-organismes: de la Résistance à la Détoxication and supported by the Centre National de la Recherche Scientifique (CNRS) (GDR2909). Céline Brochier-Armanet is supported by the CNRS (Action Thématique et Incitative sur Programme [ATIP]).
Footnotes
Supplemental material for this article may be found at http://aem.asm.org/.
Published ahead of print on 13 May 2011.
REFERENCES
- 1. Anderson G. L., Williams J., Hille R. 1992. The purification and characterization of arsenite oxidase from Alcaligenes faecalis, a molybdenum-containing hydroxylase. J. Biol. Chem. 267:23674–23682 [PubMed] [Google Scholar]
- 2. Arsène-Ploetze F., et al. 2010. Structure, function, and evolution of the Thiomonas spp. genome. PLoS Genet. 6:e1000859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Bruneel O., Duran R., Casiot C., Elbaz-Poulichet F., Personné J.-C. 2006. Diversity of microorganisms in Fe-As-rich acid mine drainage waters of Carnoulès, France. Appl. Environ. Microbiol. 72:551–556 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Bruneel O., et al. 2003. Mediation of arsenic oxidation by Thiomonas sp. in acid-mine drainage (Carnoulès, France). J. Appl. Microbiol. 95:492–499 [DOI] [PubMed] [Google Scholar]
- 5. Bryan C. G., et al. 2009. Carbon and arsenic metabolism in Thiomonas strains: differences revealed diverse adaptation processes. BMC Microbiol. 9:127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Cai L., Liu G., Rensing C., Wang G. 2009. Genes involved in arsenic transformation and resistance associated with different levels of arsenic-contaminated soils. BMC Microbiol. 9:4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Cai L., Rensing C., Li X., Wang G. 2009. Novel gene clusters involved in arsenite oxidation and resistance in two arsenite oxidizers: Achromobacter sp. SY8 and Pseudomonas sp. TS44. Appl. Microbiol. Biotechnol. 83:715–725 [DOI] [PubMed] [Google Scholar]
- 8. Casiot C., et al. 2005. Sorption and redox processes controlling arsenic fate and transport in a stream impacted by acid mine drainage. Sci. Total Environ. 347:122–130 [DOI] [PubMed] [Google Scholar]
- 9. Casiot C., et al. 2003. Bacterial immobilization and oxidation of arsenic in acid mine drainage (Carnoulès Creek, France). Water Res. 37:2929–2936 [DOI] [PubMed] [Google Scholar]
- 10. Chang J.-S., et al. 2010. Arsenic detoxification potential of aox genes in arsenite-oxidizing bacteria isolated from natural and constructed wetlands in the Republic of Korea. Environ. Geochem. Health 32:95–105 [DOI] [PubMed] [Google Scholar]
- 11. Crooks G. E., Hon G., Chandonia J.-M., Brenner S. E. 2004. WebLogo: a sequence logo generator. Genome Res. 14:1188–1190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Duquesne K., et al. 2008. Arsenite oxidation by a chemoautotrophic moderately acidophilic Thiomonas sp.: from the strain isolation to the gene study. Environ. Microbiol. 10:228–237 [DOI] [PubMed] [Google Scholar]
- 13. Ellis P. J., Conrads T., Hille R., Kuhn P. 2001. Crystal structure of the 100 kDa arsenite oxidase from Alcaligenes faecalis in two crystal forms at 1.64 Å and 2.03 Å. Structure 9:125–132 [DOI] [PubMed] [Google Scholar]
- 14. Escalante G., et al. 2009. Arsenic resistant bacteria isolated from arsenic contaminated river in the Atacama Desert (Chile). Bull. Environ. Contam Toxicol. 83:657–661 [DOI] [PubMed] [Google Scholar]
- 15. Fan H., et al. 2008. Sedimentary arsenite-oxidizing and arsenate-reducing bacteria associated with high arsenic groundwater from Shanyin, northwestern China. J. Appl. Microbiol. 105:529–539 [DOI] [PubMed] [Google Scholar]
- 15a. Halter D., et al. Taxonomic and functional prokaryote diversity in mildly arsenic contaminated sediments. Res. Microbiol., in press [DOI] [PubMed] [Google Scholar]
- 16. Hamamura N., et al. 2009. Linking microbial oxidation of arsenic with detection and phylogenetic analysis of arsenite oxidase genes in diverse geothermal environments. Environ. Microbiol. 11:421–431 [DOI] [PubMed] [Google Scholar]
- 17. Hoeft S. E., et al. 2007. Alkalilimnicola ehrlichii sp. nov., a novel, arsenite-oxidizing haloalkaliphilic gammaproteobacterium capable of chemo autotrophic or heterotrophic growth with nitrate or oxygen as the electron acceptor. Int. J. Syst. Evol. Microbiol. 57:504–512 [DOI] [PubMed] [Google Scholar]
- 18. Inskeep W. P., et al. 2007. Detection, diversity and expression of aerobic bacterial arsenite oxidase genes. Environ. Microbiol. 9:934–943 [DOI] [PubMed] [Google Scholar]
- 19. Jobb G., von Haeseler A., Strimmer K. 2004. TREEFINDER: a powerful graphical analysis environment for molecular phylogenetics. BMC Evol. Biol. 4:18. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 20. Lieutaud A., et al. 2010. Arsenite oxidase from Ralstonia sp. 22. J. Biol. Chem. 285:20433–20441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Lièvremont D., Bertin P. N., Lett M.-C. 2009. Arsenic in contaminated waters: biogeochemical cycle, microbial metabolism and biotreatment processes. Biochimie 91:1229–1237 [DOI] [PubMed] [Google Scholar]
- 22. Mandal B. K., Suzuki K. T. 2002. Arsenic round the world: a review. Talanta 58:201–235 [PubMed] [Google Scholar]
- 23. Muller D., Lièvremont D., Simeonova D. D., Hubert J.-C., Lett M.-C. 2003. Arsenite oxidase aox genes from a metal-resistant beta-proteobacterium. J. Bacteriol. 185:135–141 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Muller D., et al. 2007. A tale of two oxidation states: bacterial colonization of arsenic-rich environments. PLoS Genet. 3:e53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Oremland R. S., Saltikov C. W., Wolfe-Simon F., Stolz J. F. 2009. Arsenic in the evolution of earth and extraterrestrial ecosystems. Geomicrobiol. J. 26:522 [Google Scholar]
- 26. Oremland R. S., Stolz J. F. 2005. Arsenic, microbes and contaminated aquifers. Trends Microbiol. 13:45–49 [DOI] [PubMed] [Google Scholar]
- 27. Oremland R. S., Stolz J. F. 2003. The ecology of arsenic. Science 300:939–944 [DOI] [PubMed] [Google Scholar]
- 28. Quéméneur M., et al. 2010. Population structure and abundance of arsenite-oxidizing bacteria along an arsenic pollution gradient in waters of the Upper Isle River Basin, France. Appl. Environ. Microbiol. 76:4566–4570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Quéméneur M., et al. 2008. Diversity surveys and evolutionary relationships of aoxB genes in aerobic arsenite-oxidizing bacteria. Appl. Environ. Microbiol. 74:4567–4573 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Rhine E. D., Ní Chadhain S. M., Zylstra G. J., Young L. Y. 2007. The arsenite oxidase genes (aroAB) in novel chemoautotrophic arsenite oxidizers. Biochem. Biophys. Res. Commun. 354:662–667 [DOI] [PubMed] [Google Scholar]
- 31. Richey C., et al. 2009. Respiratory arsenate reductase as a bidirectional enzyme. Biochem. Biophys. Res. Commun. 382:298–302 [DOI] [PubMed] [Google Scholar]
- 32. Santini J. M., vanden Hoven R. N. 2004. Molybdenum-containing arsenite oxidase of the chemolithoautotrophic arsenite oxidizer NT-26. J. Bacteriol. 186:1614–1619 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Schneider T. D., Stephens R. M. 1990. Sequence logos: a new way to display consensus sequences. Nucleic Acids Res. 18:6097–6100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Sharma V. K., Sohn M. 2009. Aquatic arsenic: toxicity, speciation, transformations, and remediation. Environ. Int. 35:743–759 [DOI] [PubMed] [Google Scholar]
- 35. Simeonova D. D., et al. 2004. Microplate screening assay for the detection of arsenite-oxidizing and arsenate-reducing bacteria. FEMS Microbiol. Lett. 237:249–253 [DOI] [PubMed] [Google Scholar]
- 36. Tamaki H., et al. 2005. Comparative analysis of bacterial diversity in freshwater sediment of a shallow eutrophic lake by molecular and improved cultivation-based techniques. Appl. Environ. Microbiol. 71:2162–2169 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Valenzuela C., Campos V. L., Yañez J., Zaror C. A., Mondaca M. A. 2009. Isolation of arsenite-oxidizing bacteria from arsenic-enriched sediments from Camarones River, northern Chile. Bull. Environ. Contam. Toxicol. 82:593–596 [DOI] [PubMed] [Google Scholar]
- 38. Vallenet D., et al. 2009. MicroScope: a platform for microbial genome annotation and comparative genomics. Database (Oxford) 2009:bap021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Weeger W., et al. 1999. Oxidation of arsenite to arsenate by a bacterium isolated from an aquatic environment. Biometals 12:141–149 [DOI] [PubMed] [Google Scholar]
- 40. Zargar K., Hoeft S., Oremland R., Saltikov C. W. 2010. Identification of a novel arsenite oxidase gene, arxA, in the haloalkaliphilic, arsenite-oxidizing bacterium Alkalilimnicola ehrlichii strain MLHE-1. J. Bacteriol. 192:3755–3762 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.