Abstract
Previous studies identified zebrafish foxc1a andfoxc1b as homologs of the mouse forkhead gene, Foxc1.Both genes are transcribed in the unsegmented presomitic mesoderm (PSM), newly formed somites, adaxial cells, and head mesoderm. Here, weshow that inhibiting synthesis of Foxc1a (but not Foxc1b) protein withtwo different morpholino antisense oligonucleotides blocks formation ofmorphological somites, segment boundaries, and segmented expression ofgenes normally transcribed in anterior and posterior somites andexpression of paraxis implicated in somite epithelialization.Patterning of the anterior PSM is also affected, as judged by theabsence of mesp-b, ephrinB2, and ephA4 expression, and the down-regulation of notch5 andnotch6. In contrast, the expression of other genes, includingmesp-a and papc, in the anterior of somite primordia,and the oscillating expression of deltaC and deltaD inthe PSM appear normal. Nevertheless, this expression is apparentlyinsufficient for the maturation of the presumptive somites to proceedto the stage when boundary formation occurs or for the maintenance ofanterior/posterior patterning. Mouse embryos that are compound nullmutants for Foxc1 and the closely related Foxc2 have nomorphological somites and show abnormal expression of Notch signalingpathway genes in the anterior PSM. Therefore, zebrafish foxc1a plays an essential and conserved role in somite formation, regulatingboth the expression of paraxis and the A/P patterning of somiteprimordia.
Keywords: Forkhead, somite formation, morpholinoantisense oligonucleotide, Danio rerio
Somite formation—the reiterated subdivision of paraxial mesoderminto paired, epithelial spheres of cells on eitherside of the midline—is fundamental to vertebrate development. Giventhis importance, many genetic and embryological studies have beencarried out in mouse, chick, frog, and zebrafish to gain insight intothe cellular and molecular mechanisms involved. These studies have ledto the conclusion that before morphologically discrete pairs of somitesappear there is a prepattern of presumptive somites in the anterior ofthe unsegmented or presomitic mesoderm (PSM). Moreover, cells in theanterior and posterior halves of these presumptive somite alreadyexpress genes characteristic of anterior and posterior somites, andthis polarization is necessary for boundary formation to occur (forreviews, see Pourquie 1999; Dale and Pourquie 2000; Holley andNusslein-Volhard 2000; Pourquie 2000a,b; Stickney et al. 2000).
There is at present no single model that completely accounts forthe entire process of somite formation (Meinhardt 1986; Sternand Keynes 1988; Primmett et al. 1989). The “clock and wavefront”postulated by Cooke and Zeeman (1976) is one of the first models aimedat explaining the formation of patterned segment primordia. This modelproposes that cells in the PSM oscillate from one state that ispermissive for somite formation to another that is not. The model alsorequires that neighboring cells are synchronized in their cycles andthat the oscillations are slower in the anterior of the PSM than in theposterior. Consequently, changes in gene expression associated with thealternate states manifest themselves as repeated waves that sweepthrough the PSM from posterior to anterior. All of the genes identifiedso far that show this behavior are related to the Notch pathway,although the specific genes involved vary among species. For example,dynamic expression is seen for chick c-hairy1, a vertebratehomolog of the Drosophila hairy gene (Palmeirim et al. 1997),mouse lunatic fringe (Lfng) (Forsberg et al. 1998;McGrew et al. 1998), and zebrafish her1 (Holley et al. 2000)and deltaC (Smithers et al. 2000). The synchronization ofoscillations between neighboring cells in the PSM cells also appears toinvolve the Notch pathway (Jiang et al. 2000). Opposing thesegmentation clock is a hypothetical wave front activity that proceedsposteriorly, slowing and then halting the oscillation cycles andinducing somite maturation. When anterior PSM cells receive thisputative wave front signal, they give rise to bands or cohorts of cellsone somite wide with the same gene expression profiles. Signalingwithin and between these stabilized cohorts of cells subsequentlyrefines the anterior and posterior domains of the presumptive somite.Formation of these stripes is manifested in domain-specific expressionof genes such as mouse Mesp1 and Mesp2 (Takahashi etal. 2000), zebrafish mesp-a and mesp-b (Durbin et al.2000; Sawada et al. 2000a), and mouse Delta-like1(Dll1) (Bettenhausen et al. 1995) or ephrinB2 (Holderand Klein 1999; Holder et al. 2000). The helix-loop-helix transcriptionfactor Mesp appears to be important for the anterior–posteriorregionalization of somite primordia, and this process is thought to beessential for boundary formation (Durbin et al. 2000; Sawada et al.2000b; Takahashi et al. 2000).
Formation of the boundary between the posterior of the forming somiteand the anterior of the next-to-be-formed presumptive somite involvesthe correct expression in the mesoderm of a number of evolutionarilyconserved genes, as well as signals, not yet identified, from theectoderm (Sosic et al. 1997; Correia and Conlon 2000). Among themesodermally expressed genes are members of the ephrinand Eph receptor gene families, encoding components of the Ephcell communication pathway. A role for EphA4 andephrinB2 in somite boundary formation has been demonstrated inzebrafish. Both genes are expressed in alternating anterior andposterior domains in somite primordia, and overexpression of dominantnegative constructs of Eph receptors and ephrins results in strongdefects in boundary formation (Durbin et al. 1998, 2000). Inactivationof function of the basic-helix-loop-helix (bHLH) transcription factorparaxis in the mouse embryo impairs epithelialization andformation and maintenance of morphological boundaries, but does notaffect the initial specification and patterning of the somite primordiawithin the anterior PSM (Johnson et al. 2000). Therefore, severalgenetic pathways appear to act in parallel within the PSM toorchestrate the formation of morphologically distinct somites.
Roles in somitogenesis have been postulated for several bHLH genes,both those in the Notch signaling pathway such as mesp, andhes/her1 (Takke and Campos-Ortega 1999; Takahashi et al. 2000)and Notch-independent genes such as paraxis (Johnson et al.2000). However, little is known about the function of other classes oftranscription factors. We have shown previously that genes encoding twoevolutionarily conserved forkhead/winged helix transcription factors,foxc1a and foxc1b, are expressed in the PSM andsomites. Based on this evidence, we proposed that these closely relatedgenes play a role in somite formation and differentiation (Topczewskaet al. 2001). In the present study, we tested this proposition bytaking advantage of a recently established and effective targeted genemorpholino “knockdown” technology (Nasevicius and Ekker 2000). Thedata we obtained provide the first evidence of an essential role forthe forkhead gene foxc1a in zebrafish somitogenesis.Significantly, our data indicate that Foxc1a is essential for theformation of the most anterior somites, whereas the requirement formost other genes becomes manifested later, for posterior somites.Analysis of gene expression in the anterior PSM offoxc1a–morpholino-injected embryos suggests that Foxc1aprotein is a key component of the complex molecular circuitry that iscentered on the Notch pathway, and initially establishes and thenstabilizes the anterior/posterior (A/P) domains of the presumptivesomites. In the absence of Foxc1a protein, A/P patterning of somiteprimordia is not completed, boundaries and epithelial somites are notformed, and presumptive somites fail to complete their maturation.
Results
Antisense morpholino-modified oligonucleotides specific forfoxc1a, but not foxc1b, inhibit formation of somites
To test the hypothesis that foxc1a and foxc1b arerequired for somitogenesis, we blocked translation of the respectivemRNAs using antisense morpholino-modified oligonucleotides (MOs).Recent reports have shown that MOs injected into early embryos inhibitthe translation of specific mRNAs even many hours later, mimicking anull mutant phenotype (Nasevicius and Ekker 2000). For eachfoxc1 gene we designed two different MOs, one against 5′untranslated sequences (MO-1), the other against sequences overlappingthe translation start site (MO-2). The same phenotypes were observedwhen these MOs are injected either singly or in combination, confirmingthe specificity of our results.
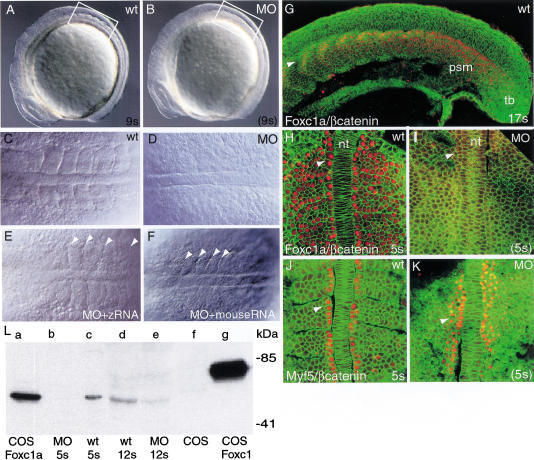
Coinjection of MO-1 and MO-2 against foxc1a at a totalconcentration of ∼6 ng/embryo inhibited the formation ofmorphologically distinct anterior somites without significantlyaffecting either the rate of development of the embryos or theiroverall size (Fig. 1A–D). Confocalmicroscopy after whole-mount staining with β-catenin antibody alsoshowed no evidence of segmental organization of the paraxial mesodermcells (Fig. 1H,I). Both the severity and penetrance of the observedphenotype depended on the dose of MOs. As shown in Table1, ∼80% of the embryos injected with∼6 ng of foxc1a-MOs lacked anterior somites. The inhibitionwas usually seen up to the stage when control embryos have developed6–7 somites, after which there was a gradual recovery of somitogenesiswith morphologically distinguishable somites forming at the anterior ofthe PSM. Embryos injected with higher doses of foxc1a-MO(∼10 ng) displayed a more extensive loss of somites (up to nine), butthey also showed a general delay in development. Lower doses causedonly partial loss of somites and a more rapid recovery ofsomitogenesis.
Figure 1.
Distribution of Foxc1 protein in zebrafish embryosand effect of inhibition of translation. (A–F) Images of liveembryos using Nomarski optics. Lateral view of wild-type (A)and Foxc1a-MO injected (B) embryos at 9-somite stage.(C and D) Dorsal views of 7-somite stage wild-type(C) and injected (D) embryos corresponding to theregions boxed in A and B. (E,F)Dorsal view of embryo injected with both foxc1a-MOs andzebrafish modified foxc1a RNA (E) and MOs togetherwith mouse synthetic Foxc1 RNA (F). Control embryoswere at the 7-somite stage. Arrowheads indicate intersomitic borders.(G–K) Confocal images of embryos after immunohistochemistry.Cell boundaries are visualized using β-catenin antibody (green).(G) Localization of Foxc1a protein (red) in nuclei of17-somite stage wild-type embryo. Arrowhead indicates migrating slowmuscle precursors. Dorsal view of wild-type (H) andMO-injected (I) embryos at 5-somite stage afterimmunohistochemistry with Foxc1 antibody (red). (J,K)Dorsal view of wild-type and MO-injected embryos afterimmunohistochemistry with Myf5 antibody (red). (L) Westernblot analysis with mouse Foxc1 specific antibody. Columns a–grepresent protein extracts from: (a) COS cells transfectedwith zebrafish pCS2–Foxc1a expression vector; (b)foxc1a-MO-injected zebrafish embryos (MO) at 5-somite stage;(c) wild-type embryos at 5-somite stage;(d,e) wild-type embryos andfoxc1a-MO-injected embryos at 12-somite stage; (f)COS cells; (g) COS cells transfected with mousepCS2–Foxc1 expression vector. In H–F arrowheadsindicate adaxial cells. nt, Notochord; psm, presomitic mesoderm; tb,tail bud.
Table 1.
The effect of foxc1a-specific MOs onthe development of wild-type embryos
| Observedphenotypes
|
Total amount of foxc1a-MOs (1 +2) injected
|
||
|---|---|---|---|
| 4 ng
|
6 ng
|
10 ng
|
|
| Partial loss ofsomites | 78% | 12% | 2% |
| Nosomites | 9% | 77% | 24% |
| Nosomites,retarded development | 0% | 2% | 59% |
| Notaffected | 13% | 9% | 15% |
| Totalembryo number | 102 | 127 | 113 |
Results from two independent experiments carried out for thepurpose of establishing dose response. All subsequent experiments forgene expression studies used 6 ng foxc1a-MOs (1 + 2) perembryo.
Different combinations of two MOs against foxc1b did notinhibit somite formation at any concentrations tested (2–10 ng/embryo,Table 2) but did affect the development ofthe head mesoderm, including the presumptive branchial arch regionwhere the gene is strongly expressed (Topczewska et al. 2001).
Table 2.
The effect of foxc1b-specific MOs onthe development of wild-type embryos
| Observedphenotype
|
foxc1b-MOsinjected
|
|||
|---|---|---|---|---|
| MO-1
|
MO-2
|
MO-1
|
MO-2
|
|
|
|
4–6ng
|
6–8 ng
|
||
| Embryos withsomites | 100% | 100% | 100% | 100% |
| Totalembryo number | 42 | 38 | 30 | 26 |
Results from two independent experiments.
Foxc1a-MOs specifically inhibit synthesis ofFoxc1a protein
To determine the effectiveness of MOs in blocking the translation offoxc1a, we used an affinity-purified, polyclonal rabbitantibody raised against a mouse Foxc1 peptide. The sequence of thepeptide used for immunization differs by two and three amino acids fromthe corresponding predicted sequences of zebrafish Foxc1a and Foxc1b,respectively (see Materials and Methods). Western blot analysis showedthat the antibody recognizes a protein made in COS cells transfectedwith a mouse Foxc1 expression vector (Fig. 1L, column g) ortransfected with zebrafish foxc1a (Fig.1L, column a) but notfoxc1b expression vector (data not shown). The antibody alsoreacts with a protein in extracts of wild-type zebrafish embryos (atthe 5-somite stage; Fig. 1L, column c) and embryos injected withzebrafish foxc1a synthetic RNA (data not shown), furtherconfirming antibody specificity. The difference in electrophoreticmobility between the zebrafish and mouse proteins may be due todifferences in their overall predicted amino acid sequences (476residues vs. 553, respectively). Moreover, the zebrafish protein lackslong stretches of alanine, glycine, and serine residues that maydecrease the mobility of the mouse protein (Topczewska et al. 2001).
To study subcellular localization of Foxc1a protein in the intactembryo, we used immunohistochemistry and confocal microscopy. As shownin Figure 1G and H, during the segmentation period Foxc1a protein islocalized in the nucleus of cells in the PSM, but not in the tail bud.Significantly, protein expression in the PSM appears uniform, even inthe anterior region where foxc1a transcript levels are higherin two distinct stripes (Fig. 2A). Thesestripes overlap with the transcription domains of mesp-b genein the most anterior cells of S-1 and S-2 (where S0 is the formingsomite), but are broader and extend more posteriorly (Fig. 2B).Foxc1a-positive nuclei were also observed in adaxial cells, which areorganized into a single epithelial-like layer on either side of thenotochord and give rise to slow muscles (Fig. 1H) (Devoto et al. 1996).The newly formed epithelial somites, including dorsal and ventrallayers, intersomitic border cells, and the few internal mesenchymalcells also accumulate Foxc1a in the nucleus. When young somites startto differentiate, immunoreactivity declines in most of the cells butremains high in migrating adaxial cells and presumptive sclerotomecells (Fig. 1G; data not shown).
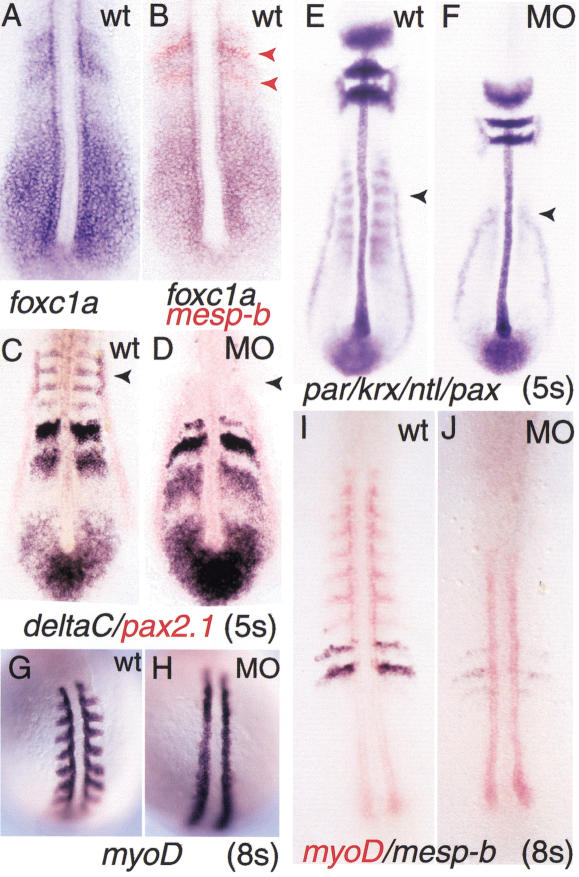
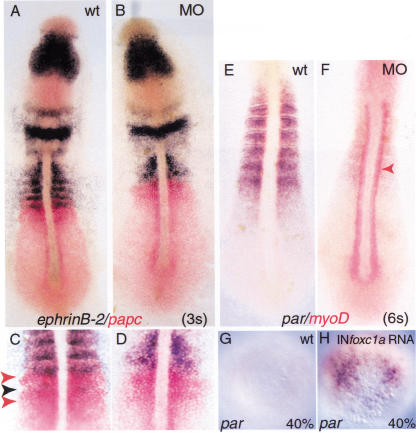
Figure 2.
Gene expression in wild-type andfoxc1a-MO-injected embryos revealed by whole-mount in situhybridization. (A) foxc1a RNA in wild-type embryo.(B) Double color in situ hybridization of foxc1a(purple) and mesp-b expression (red) in wild-type embryo at5-somite stage. (C,D) deltaC (purple) andpax2.1 (red) expression in 5-somite stage of wild-type andMO-injected embryos. (E,F) In situ hybridization withparaxis (par), krox20 (krox),no tail (ntl), and pax2.1 probes inwild-type and MO-injected embryos. (G–J) In situhybridization with probe for myoD (G,H) anddouble color in situ hybridization with probes for myoD (red)and mesp-b (blue) at the 8-somite stage. Arrowheads inC and E indicate pax2.1- anddeltaC-positive pronephros primordium present in wild-type butnot MO-injected embryos.
Immunohistochemistry showed the complete absence of Foxc1 protein inMO-injected embryos at the time when uninjected embryos are at the5-somite stage (Fig. 1I). In contrast, adaxial cells of injectedembryos stained strongly with antibody against Myf5, indicating thatthe MOs do not inhibit expression of an unrelated protein (Fig. 1J,K).Interestingly, in MO-injected embryos there appears to be somewhat moreMyf5 protein-positive cells than in the wild-type, and they extend morethan one cell diameter from the notochord (Fig. 1J,K), but this effecthas not been quantitated or explored further.
The absence of Foxc1a protein at the 5-somite stage in MO-injectedembryos was also confirmed by Western blot analysis (Fig. 1L, columnb). However, protein was detected in extracts of MO-injected embryoscollected at the stage when wild-type embryos have reached the12-somite stage (Fig. 1L, columns d and e). Taken together, theseresults indicate that foxc1a-MOs antisense oligonucleotidesspecifically inhibit the translation of Foxc1a. Moreover, the failureof early somites formation is accompanied by loss of Foxc1a protein,and the subsequent recovery of somitogenesis is associated with itsresynthesis.
The inhibitory effect of foxc1a-MO on segmentationis selective
To test whether the effect of foxc1a-MO is specific forsomitogenesis, we assayed the expression of a number of genes normallyexpressed in somites and other tissues. As shown in Figure 2C–F, theexpression of no tail (ntl) in the forming notochord,krox20 in rhombomeres 3 and 5, and pax2.1 in themidbrain–hindbrain boundary, otic primordia, and pronephric ducts wereall unaffected compared to wild-type embryos at the 5-somite stage.However, expression domains of deltaC and pax2.1 inthe pronephric primordia, which are located lateral to somites 2 to 4(Smithers et al. 2000) were missing in MO-injected embryos, even thoughthe duct primordia were unaffected (Fig. 2C,D). This is consistent withthe fact that only primordia of the pronephros and not the ductsexpress foxc1a (Topczewska et al. 2001) and suggests that thegene is specifically required for the development of the pronephros. Inaddition, the expression of myoD was strongly reduced in thesomite region of MO-injected embryos, but not in the adaxial cells(Fig. 2G–J).
The foxc1a-MO induced phenotype can be rescued
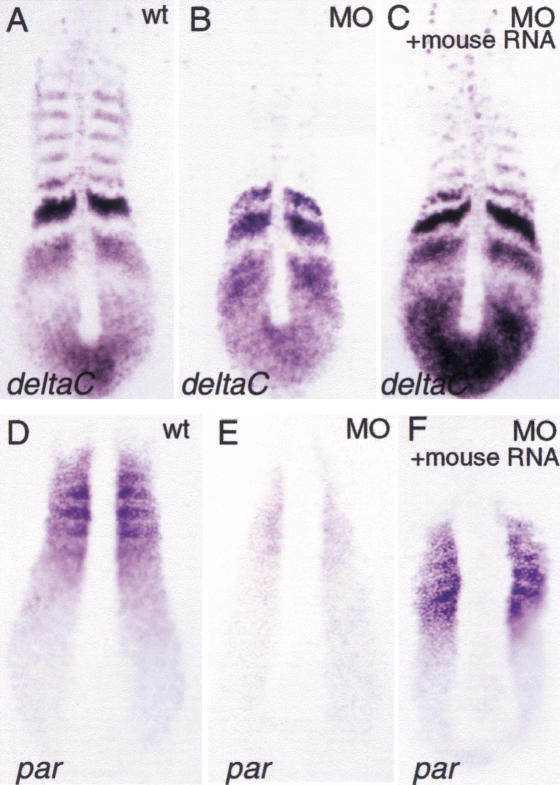
Further evidence of the specificity of the foxc1a-MOphenotype comes from rescue experiments with the mouse homolog,Foxc1, which differs from zebrafish foxc1a in thenucleotide sequence targeted by MO. Preliminary experiments indicatedthat zebrafish embryos are very sensitive to the dose of zebrafish ormouse Foxc1 RNA injected and that high levels impairgastrulation (data not shown). This finding is consistent with theobservation that mouse embryos are sensitive to the gene dosage ofFoxc1 and Foxc2 (Kume et al. 2001). A series ofdifferent concentrations of mouse Foxc1 RNA was thereforeinjected together with 6 ng of foxc1a-MOs. At a dose of about10 pg of Foxc1 RNA, partial rescue of morphological somitesand recovery of expression of deltaC and paraxis wasobserved compared to embryos injected with foxc1a-MO alone(Figs. 1E, 3; Table3). Because mouse synthetic Foxc1RNA only weakly suppressed the MO phenotype, another construct wasprepared in which the 5′ UTR sequences of zebrafish foxc1amRNA recognized by foxc1a-MOs was deleted. Injection of thismodified foxc1a RNA together with foxc1a-MO (10 pg ofRNA and 8 ng of MOs) increased by 60% the frequency of embryos withmorphologically distinguishable somites compared to MOs injection alone(n = 84; Fig. 1D,E).
Figure 3.
Mouse synthetic Foxc1 RNA can partiallyrescue deltaC and par expression in MO-injectedembryos. (A–C) deltaC expression; (D–F)par expression. (A,D) wild-type embryos;(B,E) foxc1a-MO-injected embryos;(C,F) foxc1a-MO-injected embryos coinjectedwith mouse Foxc1 RNA.
Table 3.
The effect of mouse synthetic foxc1 RNA and rescue, modifiedfoxc1a RNA on thefoxc1a-MO phenotype
| Gene analyzed
|
No expressiona
|
Partial expressionb
|
Wild-type expressionc
|
Totalno. of injected embryos
|
|---|---|---|---|---|
| Injection of 6 ngfoxc1a-MO
| ||||
| deltaC | 86% | 8% | 6% | 140 |
|
paraxis
|
87%
|
5%
|
8%
|
117
|
| Coinjectionof 6 ng of foxc1a-MO and 10 pg of mouse foxc1syntheticRNA | ||||
| deltaC | 45% | 51% | 5% | 164 |
| paraxis | 58% | 38% | 3% | 178 |
Results from three independent experiments. Embryos were fixed atthe 5 somite stage and examined by whole amount in situhybridization for deltaC and paraxis.
No expression in region of first 5 somites.
One to three stripes of deltaC or segments ofparaxis expression.
5 bands of expression.
Loss of morphological somites in MO-injected embryos is accompaniedby loss of molecular markers in paraxial mesoderm
Although microscopy analyses (Nomarski and confocal) have shown thatthere is no morphological segmentation in foxc1a-MO-injectedembryos, we nevertheless asked whether genes associated with anterioror posterior somite identity are expressed in the anterior of the PSMof MO-injected embryos. Anterior markers tested were deltaD,notch6, ephA4, and papc, and the posteriormarkers were deltaC, notch5, ephrinB2, andmyoD (Figs.2,4,5; data notshown). In all cases, no stripes of RNA expression were seen infoxc1a-MO-injected embryos in the region where somites shouldhave been present. These results suggest that Foxc1a is required forsomitogenesis and functions prior to the formation of morphologicalsomites.
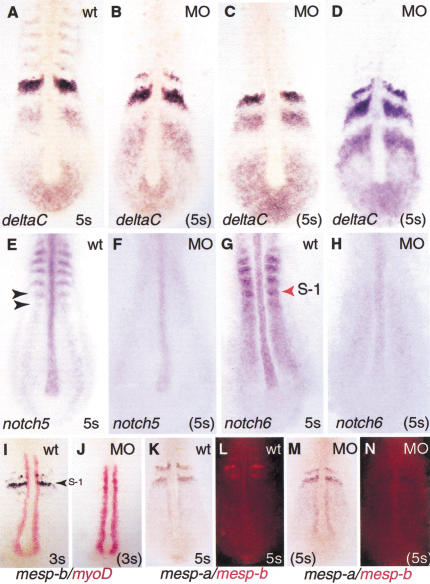
Figure 4.
Effect of inhibiting Foxc1a protein synthesis onexpression of deltaC, notch 5, notch 6,mesp-a, and mesp-b. (A) Wild-type embryohybridized with probe for deltaC. Only one example of thevariable pattern of expression is shown for the wild-type.(B–D) Representative foxc1a-MO-injected embryosshowing normal variation in pattern of deltaC expression.(E,F) Wild-type at 5-somite stage and MO-injectedembryos hybridized with probe for notch5 and(G,H) for notch6. Arrowheads indicate PSMdomains of notch genes; S-1 indicates presumptive somiteposterior to forming somite. (I,J) Double color insitu hybridization with myoD and mesp-b probe inwild-type and MO embryos at 3-somite stage, and (K–N) thesame with mesp-a and mesp-b probes at 5-somite stage.(K,M) Viewed under visible light.(L,N) The same embryos viewed with rhodamine filter.Note strong expression of myoD in the adaxial cells andcomplete absence of mesp-b transcripts in MO-injectedembryos.
Figure 5.
Inhibition of Foxc1a translation byfoxc1a-MOs selectively reduces expression of ephrinB2in PSM and region where somites should be present. (A–D)Double color in situ hybridization with ephrinB2 (purple) andpapc (red) probes at 3-somite stage in wild-type and MOsembryos. (C,D) The same embryos at highermagnification. Arrowheads indicate PSM domains ofpapc (red) and ephrinB2 (purple).(E,F) Double in situ hybridization with parand myoD probes at 6-somite stage showing that expression ofpar is strongly down-regulated in MO-injected embryos.Arrowhead indicates myoD expressing adaxial cells.(G,H) Foxc1a synthetic RNA induces ectopicexpression of par at 40% epiboly. In situ hybridization withpar in wild-type embryos (G) and embryos injectedwith Foxc1a RNA (H). Animal pole views.
Delta genes show normal expression in the presomitic mesoderm offoxc1a-MO injected embryos
The models of somitogenesis postulate that cells in the PSMoscillate from one mutually exclusive state to another. This ismanifested in the zebrafish embryo as periodic waves of expression ofher1, deltaC, and deltaD that appear tosweep anteriorly from the tail bud (Jiang et al. 2000; Keller 2000;Kerszberg and Wolpert 2000; Pourquie 2000b). An example of this dynamicbehavior is the characteristic pattern of broad and narrow stripesof deltaC gene expression seen in the PSM of a populationof wild-type embryos all examined at the same stage. As shown in Figure4A–D, the wild-type pattern of deltaC expression is also seenin a group of MO-injected embryos, and transcripts did become localizedinto tight bands in the presumptive somite region. Similar dynamicexpression in the PSM of MO-injected embryos was observed forher1 and deltaD (data not shown). However asmentioned before, the striped expression of deltaC was notmaintained in the region where the somites should have been present.Taken together, these results suggest that the first step ofsegmentation, establishment of segmental prepattern, is not affected byFoxc1a inhibition and that synchronized oscillatory behavior does occurin the PSM of MO-injected embryos.
Some aspects of the anterior/posterior identity of cells in theanterior presomitic mesoderm are affected in MO-injected embryos
Before epithelial somites are formed, precise A and P domains areestablished in the somite primordia, a step that is thought to requireNotch-dependent signaling (Buchberger et al. 2000; Johnson et al. 2000;Sawada et al. 2000a; Takahashi et al. 2000). Studies in both mouse andzebrafish have identified Mesp proteins as specific bHLH transcriptionfactors required for effecting this Notch signaling in the anteriorPSM. Both mesp-a and mesp-b genes are expressed inoverlapping domains in the anterior of the somite primordia (S-1 andS-2) (Fig. 4I–N; Durbin et al. 2000; Sawada et al. 2000a). Inaddition, a transient band of mesp-b expression is seen in theforming somite, S0. We have consistently observed that the expressionof mesp-b is strongly reduced in MO-injected embryos in S0,S-1, and S-2 (Fig. 4I,J). In contrast, the expression ofmesp-a appears to be unaffected. This result is clearlyillustrated in experiments in which the same MO-injected embryos werehybridized simultaneously with probes for the two genes, one labeledwith DIG and the other with fluorescein (Fig. 4K,N). Our data suggest adifferential requirement of Foxc1a protein for expression ofmesp-a and mesp-b. Additionally, the expressionof protocadherin C (papc), which is localized in thesame regions of the PSM (S-1 and S-2) as the mesp genes wasnot affected in MO-injected embryos (Fig. 5; data not shown).
We next examined the effect of absence of Foxc1a protein on theexpression of two genes encoding Delta receptors, notch5 andnotch6 (Fig. 4E–H). Both genes are segmentally expressed inthe PSM of wild-type embryos. In addition, notch5 andnotch6 transcripts are present in the posterior andanteromedial parts of the formed somites, respectively (Bierkamp andCampos-Ortega 1993; Westlin and Lardelli 1997). This segmentedexpression was strongly down-regulated in MO-injected embryos, butweak, uniform expression of notch6 persisted throughout thePSM. These observations indicate that only an incomplete A/Ppolarization of somite primordia is established in the absence ofFoxc1a protein.
Expression of ephrin2B and eph4A, required forformation of somite boundaries, is abnormal in MO-injected embryos
Because the formation of intersomitic boundaries requiresintercellular signaling mediated by cell surface molecules of theEph/ephrin family (Durbin et al. 1998, 2000), we examined theexpression of ephrinB2 and ephA4 in MO-injectedembryos. As described earlier, stripes of expression of both genes wereabsent from the region where somites should be present (Fig. 5A,B; datanot shown). To assess expression in the anterior PSM, double stainingusing papc and ephrin-B2 probes was carried out atthe 3–4-somite stage (Fig. 5C,D). While the papc expressiondomains located in S-1 and S-2 were still present in MO-injectedembryos, the stripes of ephrin-B2 RNA were lost. This analysisshows that the expression of Eph/ephrin signaling molecules isperturbed in MO-injected embryos, and this may account, at least inpart, for the defect in somite border formation.
Expression of paraxis depends on foxc1
Mouse embryos lacking the bHLH gene paraxis (par)fail to form epithelial somites, although segmentation and initialanteroposterior specification of somite primordia do occur (Burgess etal. 1996). As shown in Figures 2E,F and 5E,F, par expressionis severely down-regulated in both the PSM and paraxial mesoderm ofMO-injected embryos. As in the case of mesp-a, this effect isselective, because myoD expression in adaxial cells ofinjected embryos was normal or even elevated (Fig. 5E,F).
The down-regulation of par expression in MO-injected embryosraised the possibility that Foxc1a protein is a positive regulator ofpar gene. To test this hypothesis, we injected cappedsynthetic foxc1a RNA (40–60 pg) into one cell stage zebrafishembryos. As shown in Figure 5G,H, the overexpression of foxc1ainduces premature transcription of par at 40% epiboly,whereas during normal development, expression of par RNA wasfirst detected at about 65% epiboly (Shanmugalingam and Wilson 1998).These results show that foxc1a is both essential andsufficient for par expression.
The phenotype of foxc1a-MO-injected embryos is more severethan that of fss mutants
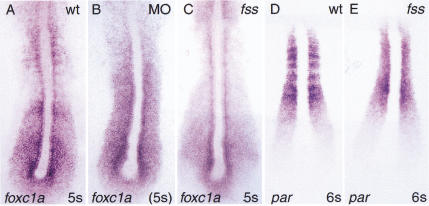
Several zebrafish mutants have been described that are defective insomitogenesis. In every case except fused somites(fss), the anterior somites develop normally, before defectsare seen in the formation of more posterior somites (van Eeden et al.1996). In contrast, fss mutants, likefoxc1a-MO-injected embryos, lack all anterior somites. Despitethis similarity, fss and foxc1a affect somitogenesisdifferently, because par is not down-regulated in fssas observed for MO-injected embryos (Fig.6). We also compared the expression offoxc1a in fss and MO-injected embryos. Our dataindicate that the striped pattern of foxc1a expression is lostin the anterior PSM of fss mutants (Fig. 6A,C). Similarly, nostriped expression of foxc1a was seen in MO embryos, whereinhibition of foxc1a translation resulted in uniformexpression of the gene throughout the PSM and into the region wheresomites should have been present. We conclude that foxc1a actsin parallel to the fss genetic pathway as an essential factorfor the complete anterior–posterior patterning of the PSM and theformation of epithelial somites.
Figure 6.
Phenotypic differences between MO-injected embryosand fss mutants. (A,D) Wild-type embryos;(B) foxc1a-MO injected embryo, and(C,D) fss embryos. (A–C) In situhybridization with foxc1a probe. Note loss of stripedexpression of foxc1a in the anterior PSM of fssmutant (D,E) par probe. Note that expressionof par is not down-regulated in fss embryos.
Discussion
The findings reported here establish the forkhead/winged helixtranscription factor, Foxc1a, as a novel and necessary component of themolecular circuitry underlying somitogenesis in the zebrafish.Moreover, taken together with the new findings in the mouse describedin the accompanying paper (Kume et al. 2001), it appears that thisfunction of Foxc1, the requirement of the gene for parexpression, and its involvement with elements of the Notch signalingpathway have all been evolutionarily conserved.
The conclusion that Foxc1a is required for zebrafish somitogenesis isbased on findings with embryos in which synthesis of the protein isspecifically inhibited by morpholino antisense oligonucleotides(Nasevicius and Ekker 2000). These embryos lack, at least up to aboutthe 6–7-somite stage, morphological somites, segmented paraxialmesoderm, and expression in the somite region of genes characteristicof anterior and posterior somite cell fates. Several lines of evidenceargue that the inhibitory effect of the foxc1a-MOs is specificand not due to a general toxicity. First, MOs block the synthesis ofFoxc1a protein but not that of Myf5. Second, the inhibition of somiteformation is dose-dependent, and a similar phenotype is observed withtwo separate foxc1a-MOs, each targeted against differentregions of the foxc1a RNA. In contrast, somitogenesis proceedsnormally in embryos injected with MOs targeted to the closely relatedgene foxc1b. Third, differentiation of other tissues continueson schedule in the absence of somite formation. Fourth, spontaneousrecovery of somitogenesis in MO-injected embryos at around the 7-somitestage is accompanied by reexpression of the Foxc1a protein. Oneexplanation for this recovery is that, as shown in Figure 6, the levelof foxc1a RNA increases significantly infoxc1a-MO-injected embryos, probably due to negative feedbackby Foxc1a protein on the transcription of its own gene. Consequently,MO oligonucleotides may become saturated. Finally, the phenotype ofMO-injected embryos can be reversed by injection of either mouseFoxc1 or zebrafish foxc1a synthetic RNA, which lackthe sequences against which the MOs are directed.
Foxc1a protein is not required for the segmentation clock
Analysis of gene expression in foxc1a-MO-injected embryosshows that the dynamic expression of deltaC, deltaD,and her1 genes in the PSM resembles that of wild-type embryos.Moreover, the anterior boundaries of the delta gene expressionstripes are sharp and not diffuse, as seen in embryos with mutations inthe Notch signaling pathway (Jiang et al. 1998; Holley et al. 2000).These observations suggest that the absence of Foxc1a protein does notaffect the oscillation of cells in the PSM from one state to another.Nor does it affect the proposed synchronization of these oscillationsbetween neighboring cells. Although notch6 expression (Westlinand Lardelli 1997) is strongly down-regulated in MO-injected embryos,and is no longer expressed in stripes in the anterior region, there isstill weak uniform expression throughout the PSM. This weak expressionmay be sufficient to enable synchronization of oscillations in the PSMto occur. Alternatively, other Notch genes such as notch1a maybe engaged in this process and be unaffected by the absence of Foxc1a.
Foxc1a is required for correct A/P patterning of presumptivesomites in the anterior presomitic mesoderm
For morphological boundaries to be generated between formingsomites, it appears necessary that each presumptive somite besubdivided into stable anterior and posterior domains (Durbin et al.2000). Observations initially made in the mouse have indicated acrucial role for Notch/Delta signaling and the basic-loop-helixtranscription factor, Mesp2, in establishing this A/P patterning(Takahashi et al. 2000). Thus, Mesp2 in the anterior half of thepresumptive somite is thought to inhibit the up-regulation of theDll1 gene in response to Notch activation, while the absenceof Mesp2 expression in the posterior domain allowsDll1 induction to proceed. Moreover, Mesp2 is thought to actin an autoregulatory loop with Notch, being both up-regulated by Notchsignaling and required for Notch gene expression. A similar role hasbeen suggested for zebrafish mesp-b gene (Sawada et al.2000a). We found that the expression of one mesp gene,mesp-a, is unaffected in MO-injected embryos, in both theposterior PSM where it is expressed diffusely and in the stripe ofhigh-level expression in the anterior region of the presumptive somites(S-1 and S-2) (Fig. 4). In contrast, transcripts for themesp-b gene, normally localized exclusively in the mostanterior part of S-1 and S-2, are completely absent in MO-injectedembryos. This deficit is accompanied by the absence of the stripedexpression of notch6 and notch5 genes. We concludethat in zebrafish, the activity of mesp-b is required forcorrect patterning of the presumptive somites. Moreover, the loss ofexpression of only one mesp gene, in this case mesp-bdue to the absence of Foxc1a protein, is enough to prevent thecompletion of somite formation, possibly in part because of thedown-regulation of notch 5 and notch6.
Among the several zebrafish mutations with defects in somitogenesis,fss has the closest phenotype to that offoxc1a-MO-injected embryos, because in both sets of embryosall anterior somites are missing (van Eeden et al. 1996). However,there are significant differences between the two phenotypes. Forexample, the fss mutation completely disrupts normalexpression of both mesp-a and mesp-b during thesegmentation period (Sawada et al. 2000a). In other fss-typemutants such as aei, mesp-b is expressed in a diffuse“salt and pepper” pattern in the paraxial mesoderm similar to thatseen for deltaC in the same mutants (Jiang et al. 2000),whereas expression of mesp-a is very reduced and limitedmostly to adaxial cells. Based on these results, Sawada et al. (2000a)proposed that the two mesp genes are differentially regulated,a conclusion supported by our own results. Another difference betweenthe phenotype of fss and foxc1a-MO-injected embryosis seen in the expression of the protocadherin gene,papc. In fss mutants, the down-regulation of bothmesp genes is associated with strongly reduced expression ofpapc in the anterior PSM (Jiang et al. 2000). In contrast,papc is still expressed normally infoxc1a-MO-injected embryos in the PSM, suggesting that theremaining activity of mesp-a is sufficient to drivepapc expression even though segmentation does not proceed tocompletion. Expression of mesp-a and papc in theanterior of the presumptive somites also suggests that the hypotheticalwave front activity, postulated to be disrupted in fssmutants, functions in the absence of Foxc1a activity, and is able topartially stabilize the anterioposterior patterning of the anteriorPSM.
Foxc1a is required for expression of ephrinB2 and itsreceptor ephA4 during somite formation
The formation of intersomitic boundaries requires the expression ofEphrins and their receptors, and manipulation of ephrin signaling genesin zebrafish disturbs somite differentiation (Durbin et al. 1998). Wefound that expression domains of ephrinB2 and its receptorephA4 are strongly down-regulated in the anterior PSM offoxc1a-MO-injected embryos. This result is significantlydifferent from that seen in fss mutant embryos, where theposteriorly expressed ephrinB2 is expanded, andfss-like mutants in which ephrinB2 and ephA4are expressed in a salt and pepper pattern (Durbin et al. 2000). Thestrong phenotype of MO-injected embryos compared to that offss-type mutants raises the possibility that Ephrins and Ephreceptors are regulated by Foxc1a directly, or that completed A/Pspecification of somite primordia is required for ephrinB2 andephA4 expression. The observed lack of ephrinB2 andephA4 expression in our experiments may be one of the reasonswhy somite boundaries are not formed and further somitic mesodermdifferentiation is arrested.
Foxc1a is required for paraxis expression
Analysis of mouse mutants has shown that the gene paraxis,encoding a bHLH transcription factor, is required for the formation ofepithelial somites but not for segmentation of paraxial mesoderm(Burgess et al. 1995, 1996). Recent evidence suggests that in theabsence of Par in the mouse, Notch signaling and expression ofMesp2, EphrinB2, and EphA4 in the PSM areinitially normal. However, intersomitic boundaries fail to form, andthe A/P polarity of the anterior PSM is not maintained in the segmentedmesoderm (Johnson et al. 2000). Paraxis expression does notdepend on the Notch signaling pathway in the mouse (Johnson et al.2000), and is maintained in fss mutant embryos (Fig. 6E). Incontrast, in foxc1a-MO-injected embryos (and in mouse embryoslacking both Foxc1 and Foxc2) paraxisexpression is strongly down-regulated. Further, we have shown thatinjection of foxc1a RNA into the zebrafish embryo leads toectopic and premature par expression, suggesting thatpar is a direct or early downstream target of the Foxc1atranscription factor. Nevertheless, the phenotype offoxc1a-MO-injected embryos is more severe than that ofpar null embryos and affects the A/P patterning polarity ofthe anterior PSM. We conclude that loss of Paraxis function cannot bethe primary and sole defect in MO-injected embryos, although it maystill contribute to the lack of border formation and epithelializationof somites.
Comparison of the roles of foxc1 genes in mouse andzebrafish somitogenesis
Recent evidence in the mouse presented in the accompanying paperindicates that the two closely related winged helix transcriptionfactors, Foxc1 and Foxc2, function combinatorily, and that inactivationof both sets of alleles is required to completely disrupt somiteformation and the prepatterning of the anterior PSM (Kume et al. 2001).However, our present results suggest that there are differences in therole of foxc1 genes in somitogenesis between mice andzebrafish. First, foxc1b, although transcribed in the samepattern as foxc1a in paraxial mesoderm and probably requiredfor head mesoderm development, cannot substitute for the absence offoxc1a in somite formation. Second, if a homolog of mouseFoxc2 exists in the zebrafish, then it, too, does not compensate forthe absence of Foxc1a. It therefore appears that the completion ofsomite formation in the zebrafish embryo is more dependent on the levelof foxc1 gene expression compared to the mouse embryo.Inhibiting Foxc1a protein synthesis alone is sufficient to uncover anevolutionarily conserved requirement for this class of transcriptionfactor in the maturation of the presumptive somites.
Materials and methods
Fish embryos
Fish were maintained as described (Solnica-Krezel et al. 1994). Themutant allele used was fused somites(fssm774; Driever and Fishman 1996).
In situ hybridization
Whole-mount in situ hybridization was performed essentially asdescribed (Thisse et al. 1993). For two-color in situ hybridization,the digoxigenin- and fluorescein-labeled antisense probes were usedsimultaneously. After the first color reaction, the alkalinephosphatase-conjugated antibody was inactivated by washing three timesfor 5 min at room temperature first with 0.1 M glycine-HCl (pH 2.2),0.1% Tween 20 and then with PBST (PBS at pH 7.4, 0.1% Tween 20).Embryos were then incubated with antifluorescein antibody for 2 h atroom temperature or overnight at 4°C. After intense washing in PBST(6 times every 15 min at room temperature), followed by two more washeswith 0.1 M Tris (pH 8.2), 0.1% Tween, the second color reaction wasperformed with Fast Red (Roche) in 0.1 M Tris (pH 8.2), 0.1% Tween,with the addition of 0.4 M NaCl. Depending on the probe, the reactionwas continued for a few hours to overnight. For photomicrography, theembryos were mounted flat in 50% glycerol.
Antibody against mouse Foxc1 protein
A peptide from the C-terminal domain of mouse Foxc1 protein wasused to raise a polyclonal rabbit antiserum, followed bypeptide-affinity purification (Research Genetics). The differencesbetween the predicted mouse and zebrafish protein sequences areunderlined: Foxc1, AYPGQQQNFHSVREM FESQRI; Foxc1a,ATPAQQQNFHSVREMFESQRI; Foxc1b,AS PGQQQNFHAVREMFETQRI.
Whole-mount immunocytochemistry
Embryos were fixed in 4% paraformaldehyde, 4% sucrose, 3 mMCaCl2, and PBS (pH 7.4) overnight at 4°C and permeabilizedin acetone for 7 min at −20°C. Blocking was performed using PBSTbuffer with 5% goat serum, 0.2% BSA, and 2% DMSO. Incubation withprimary antibody was overnight at 4°C followed by intense washing inPBST buffer at room temperature. For immunofluorescence microscopy, thesecondary antibody was conjugated with Cy-3 (Jackson Labs) or AlexaFluor 488 (Molecular Probe). Incubation was performed at roomtemperature for 2–3 h. In double staining experiments, the sampleswere incubated with the Foxc1 antibody diluted 1:200 overnight at4°C and subsequently with β-catenin monoclonal antibody (Sigma)diluted in blocking solution 1:500 for 4 h at room temperature.
Transient transfection of COS cells
This procedure was performed using lipofectamine reagent accordingto the manufacturer's protocol (Life/Technologies-GIBCO BRL).
Western blot
Embryos were dechorionated and washed in Danieau buffer, and theyolk was removed manually. Tissues from 60 embryos were boiled in 60μL of Laemmli sample buffer (Laemmli 1990) for 5 min. Followingcentrifugation, the supernatant equivalent of 20 embryos waselectrophoresed on a 10% polyacrylamide gel according to Sambrook atal. (1989). Proteins were electroblotted onto Zeta-Probe membranes (BioRad), and antigenic proteins were detected using affinity-purifiedFoxc1 antiserum at 1:1000 dilution and an ECL kit (Amersham).
Confocal microscopy
Flat mounted embryos were observed initially using epifluorescencewith a Zeiss Axiophot microscope and subsequently using a Zeiss LSM410Confocal Laser Scanning Inverted Microscope with 20× or 40× Neofluarobjectives (facility supported by NIH grants CA68485 and DK20593). Forthe Alexa 488 and Cy-3 labels, excitation was at 488 and 543 nm,respectively.
Microinjection of zebrafish embryos
Capped mRNA was synthesized by in vitro transcription of linearizedplasmid (MessageMachine kit, Ambion). RNA (10–100 pg) was injectedinto the yolk of one- to two-cell stage embryos.
Morpholino
Morpholino antisense oligonucleotides (Gene-Tools) were designedaccording to the manufacturer's suggestions. Two differentoligonucleotides specific for foxc1a and two forfoxc1b were used for injections, as follows:foxc1a-MO-1, 5′-GT CAAGAAGACTGAAGCAATCCACA-3′;foxc1a-MO-2, 5′-CCTGCATGACTGCTCTCCAAAACGG-3′;foxc1b-MO-1, 5′-GCATCGTACCCCTTTCTTCGGTACA-3′;foxc1b-MO-2, 5′-AAGTGAAATGAAGACTATGCAGACG-3′.
Morpholino oligonucleotides were diluted in 1× Danieau buffer, andbetween 2 and 10 ng was injected into the yolk of embryos at the 1–8cell stage.
Acknowledgments
We acknowledge E.S. Weinberg, S.W. Wilson, B. Appel, H. Takeda, andS. Amacher for the myoD, paraxis, notch, anddelta, mesp, and papc probes, respectively.C. Brenner, J. Lewis, Randy Johnson, and members of our labs kindlyprovided helpful comments and discussion. We thank J. Clanton forexcellent fish care and A. Land-Dedrick for help preparing themanuscript. The work was supported in part by grants from the NIH (GM62283) and the Pew Scholars Program for Biomedical Research to L. S-K.J.M.T is an Associate and B.L.M.H. an Investigator of the Howard HughesMedical Institute.
The publication costs of this article were defrayed in part by paymentof page charges. This article must therefore be hereby marked“advertisement” in accordance with 18 USC section 1734 solely toindicate this fact.
Footnotes
E-MAIL brigid.hogan@mcmail.vanderbilt.edu; FAX (615) 343-2033.
Article and publication are athttp://www.genesdev.org/cgi/doi/10.1101/gad.907401.
References
- Bettenhausen B, Hrabe de Angelis M, Simon D, Guenet JL, Gossler A. Transient and restricted expression during mouse embryogenesis of Dll1, a murine gene closely related to Drosophila Delta. Development. 1995;121:2407–2418. doi: 10.1242/dev.121.8.2407. [DOI] [PubMed] [Google Scholar]
- Bierkamp C, Campos-Ortega JA. A zebrafish homologue of the Drosophila neurogenic gene Notch and its pattern of transcription during early embryogenesis. Mech Dev. 1993;43:87–100. doi: 10.1016/0925-4773(93)90027-u. [DOI] [PubMed] [Google Scholar]
- Buchberger A, Bonneick S, Arnold H. Expression of the novel basic-helix-loop-helix transcription factor cMespo in presomitic mesoderm of chicken embryos. Mech Dev. 2000;97:223–226. doi: 10.1016/s0925-4773(00)00424-x. [DOI] [PubMed] [Google Scholar]
- Burgess R, Cserjesi P, Ligon KL, Olson EN. Paraxis: A basic helix-loop-helix protein expressed in paraxial mesoderm and developing somites. Dev Biol. 1995;168:296–306. doi: 10.1006/dbio.1995.1081. [DOI] [PubMed] [Google Scholar]
- Burgess R, Rawls A, Brown D, Bradley A, Olson E. Requirement of the paraxis gene for somite formation and musculoskeletal patterning. Nature. 1996;384:570–573. doi: 10.1038/384570a0. [DOI] [PubMed] [Google Scholar]
- Cooke J, Zeeman EC. A clock and wavefront model for control of the number of repeated structures during animal morphogenesis. J Theor Biol. 1976;58:455–476. doi: 10.1016/s0022-5193(76)80131-2. [DOI] [PubMed] [Google Scholar]
- Correia KM, Conlon RA. Surface ectoderm is necessary for the morphogenesis of somites. Mech Dev. 2000;91:19–30. doi: 10.1016/s0925-4773(99)00260-9. [DOI] [PubMed] [Google Scholar]
- Dale KJ, Pourquie O. A clock-work somite. Bioessays. 2000;22:72–83. doi: 10.1002/(SICI)1521-1878(200001)22:1<72::AID-BIES12>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- Devoto SH, Melancon E, Eisen JS, Westerfield M. Identification of separate slow and fast muscle precursor cells in vivo, prior to somite formation. Development. 1996;122:3371–3380. doi: 10.1242/dev.122.11.3371. [DOI] [PubMed] [Google Scholar]
- Driever W, Fishman MC. The zebrafish: Heritable disorders in transparent embryos. J Clin Invest. 1996;97:1788–1794. doi: 10.1172/JCI118608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Durbin L, Brennan C, Shiomi K, Cooke J, Barrios A, Shanmugalingam S, Guthrie B, Lindberg R, Holder N. Eph signaling is required for segmentation and differentiation of the somites. Genes & Dev. 1998;12:3096–3109. doi: 10.1101/gad.12.19.3096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Durbin L, Sordino P, Barrios A, Gering M, Thisse C, Thisse B, Brennan C, Green A, Wilson S, Holder N. Anteroposterior patterning is required within segments for somite boundary formation in developing zebrafish. Development. 2000;127:1703–1713. doi: 10.1242/dev.127.8.1703. [DOI] [PubMed] [Google Scholar]
- Forsberg H, Crozet F, Brown NA. Waves of mouse Lunatic fringe expression, in four-hour cycles at two-hour intervals, precede somite boundary formation. Curr Biol. 1998;8:1027–1030. doi: 10.1016/s0960-9822(07)00424-1. [DOI] [PubMed] [Google Scholar]
- Holder N, Durbin L, Cooke J. Eph receptors and ephrins are key regulators of morphogenesis. Ernst Schering Res Found Workshop. 2000;29:123–147. doi: 10.1007/978-3-662-04264-9_8. [DOI] [PubMed] [Google Scholar]
- Holder N, Klein R. Eph receptors and ephrins: Effectors of morphogenesis. Development. 1999;126:2033–2044. doi: 10.1242/dev.126.10.2033. [DOI] [PubMed] [Google Scholar]
- Holley SA, Nusslein-Volhard C. Somitogenesis in zebrafish. Curr Top Dev Biol. 2000;47:247–277. doi: 10.1016/s0070-2153(08)60727-9. [DOI] [PubMed] [Google Scholar]
- Holley SA, Geisler R, Nusslein-Volhard C. Control of her1 expression during zebrafish somitogenesis by a delta-dependent oscillator and an independent wave-front activity. Genes & Dev. 2000;14:1678–1690. [PMC free article] [PubMed] [Google Scholar]
- Jiang YJ, Aerne BL, Smithers L, Haddon C, Ish-Horowicz D, Lewis J. Notch signalling and the synchronization of the somite segmentation clock. Nature. 2000;408:475–479. doi: 10.1038/35044091. [DOI] [PubMed] [Google Scholar]
- Jiang YJ, Smithers L, Lewis J. Vertebrate segmentation: The clock is linked to Notch signalling. Curr Biol. 1998;8:R868–R871. doi: 10.1016/s0960-9822(07)00547-7. [DOI] [PubMed] [Google Scholar]
- Johnson J, Rhee J, Parsons SM, Brown D, Olson EN, Rawls A. The anterior/posterior polarity of somites is disrupted in paraxis-deficient mice. Dev Biol. 2000;229:1–12. doi: 10.1006/dbio.2000.9969. [DOI] [PubMed] [Google Scholar]
- Keller R. The origin and morphogenesis of amphibian somites. Curr Top Dev Biol. 2000;47:183–246. doi: 10.1016/s0070-2153(08)60726-7. [DOI] [PubMed] [Google Scholar]
- Kerszberg M, Wolpert L. A clock and trail model for somite formation, specialization and polarization. J Theor Biol. 2000;205:505–510. doi: 10.1006/jtbi.2000.2085. [DOI] [PubMed] [Google Scholar]
- Kume T, Jiang HY, Topczewska JM, Hogan BLM. The murine winged helix transcription factors, Foxc1 and Foxc2, are both required for cardiovascular development and somitogenesis. Genes & Dev. 2001;15:2470–2482. doi: 10.1101/gad.907301. (this issue). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227:680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- McGrew MJ, Dale JK, Fraboulet S, Pourquie O. The lunatic fringe gene is a target of the molecular clock linked to somite segmentation in avian embryos. Curr Biol. 1998;8:979–982. doi: 10.1016/s0960-9822(98)70401-4. [DOI] [PubMed] [Google Scholar]
- Meinhardt H. Hierarchical inductions of cell states: A model for segmentation in Drosophila. J Cell Sci Suppl. 1986;4:357–381. doi: 10.1242/jcs.1986.supplement_4.20. [DOI] [PubMed] [Google Scholar]
- Nasevicius A, Ekker SC. Effective targeted gene ‘knockdown’ in zebrafish. Nat Genet. 2000;26:216–220. doi: 10.1038/79951. [DOI] [PubMed] [Google Scholar]
- Palmeirim I, Henrique D, D. Ish-Horowicz D, Pourquie O. Avian hairy gene expression identifies a molecular clock linked to vertebrate segmentation and somitogenesis. Cell. 1997;91:639–648. doi: 10.1016/s0092-8674(00)80451-1. [DOI] [PubMed] [Google Scholar]
- Pourquie O. Notch around the clock. Curr Opin Genet Dev. 1999;9:559–565. doi: 10.1016/s0959-437x(99)00011-8. [DOI] [PubMed] [Google Scholar]
- Pourquie O. Segmentation of the paraxial mesoderm and vertebrate somitogenesis. Curr Top Dev Biol. 2000a;47:81–105. doi: 10.1016/s0070-2153(08)60722-x. [DOI] [PubMed] [Google Scholar]
- Pourquie O. Vertebrate segmentation: Is cycling the rule? Curr Opin Cell Biol. 2000b;12:747–751. doi: 10.1016/s0955-0674(00)00163-0. [DOI] [PubMed] [Google Scholar]
- Primmett DR, Norris WE, Carlson GJ, Keynes RJ, Stern CD. Periodic segmental anomalies induced by heat shock in the chick embryo are associated with the cell cycle. Development. 1989;105:119–130. doi: 10.1242/dev.105.1.119. [DOI] [PubMed] [Google Scholar]
- Sambrook J, Fritsch EF, Maniatis T. Molecular cloning: A laboratory manual. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press; 1989. [Google Scholar]
- Sawada A, Fritz A, Jiang Y, Yamamoto A, Yamasu K, Kuroiwa A, Saga Y, Takeda H. Zebrafish Mesp family genes, mesp-a and mesp-b are segmentally expressed in the presomitic mesoderm, and Mesp-b confers the anterior identity to the developing somites. Development. 2000a;127:1691–1702. doi: 10.1242/dev.127.8.1691. [DOI] [PubMed] [Google Scholar]
- Sawada A, Saga Y, Takeda H. Zebrafish somitogenesis-roles of mesp- and hairy-related genes. Tanpakushitsu Kakusan Koso. 2000b;45:2738–2744. [PubMed] [Google Scholar]
- Shanmugalingam S, Wilson SW. Isolation, expression and regulation of a zebrafish paraxis homologue. Mech Dev. 1998;78:85–89. doi: 10.1016/s0925-4773(98)00150-6. [DOI] [PubMed] [Google Scholar]
- Smithers L, Haddon C, Jiang Y, Lewis J. Sequence and embryonic expression of deltaC in the zebrafish. Mech Dev. 2000;90:119–123. doi: 10.1016/s0925-4773(99)00231-2. [DOI] [PubMed] [Google Scholar]
- Solnica-Krezel L, Schier AF, Driever W. Efficient recovery of ENU-induced mutations from the zebrafish germline. Genetics. 1994;136:1401–1420. doi: 10.1093/genetics/136.4.1401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sosic D, Brand-Saberi B, Schmidt C, Christ B, Olson E. Regulation of paraxis expression and somite formation by ectoderm- and neural tube-derived signals. Dev Biol. 1997;185:229–243. doi: 10.1006/dbio.1997.8561. [DOI] [PubMed] [Google Scholar]
- Stern CD, Keynes RJ. Interactions between somite cells: The formation and maintenance of segment boundaries in the chick embryo. Development. 1988;99:261–272. doi: 10.1242/dev.99.2.261. [DOI] [PubMed] [Google Scholar]
- Stickney HL, Barresi MJ, Devoto SH. Somite development in zebrafish. Dev Dyn. 2000;219:287–303. doi: 10.1002/1097-0177(2000)9999:9999<::AID-DVDY1065>3.0.CO;2-A. [DOI] [PubMed] [Google Scholar]
- Takahashi Y, Koizumi K, Takagi A, Kitajima S, Inoue T, Koseki H, Saga Y. Mesp2 initiates somite segmentation through the Notch signalling pathway. Nat Genet. 2000;25:390–396. doi: 10.1038/78062. [DOI] [PubMed] [Google Scholar]
- Takke C, J.A. Campos-Ortega JA. her1, a zebrafish pair-rule like gene, acts downstream of Notch signalling to control somite development. Development. 1999;126:3005–3014. doi: 10.1242/dev.126.13.3005. [DOI] [PubMed] [Google Scholar]
- Thisse C, Thisse B, Schilling TF, Postlethwait JH. Structure of the zebrafish snail1 gene and its expression in wild-type, spadetail and no tail mutant embryos. Development. 1993;119:1203–1215. doi: 10.1242/dev.119.4.1203. [DOI] [PubMed] [Google Scholar]
- Topczewska JM, Topczewski J, Solnica-Krezel L, Hogan BL. Sequence and expression of zebrafish foxc1a and foxc1b, encoding conserved forkhead/winged helix transcription factors. Mech Dev. 2001;100:343–347. doi: 10.1016/s0925-4773(00)00534-7. [DOI] [PubMed] [Google Scholar]
- van Eeden FJ, Granato M, Schach U, Brand M, Furutani-Seiki M, Haffter P, Hammerschmidt M, Heisenberg CP, Jiang YJ, Kane DA, et al. Mutations affecting somite formation and patterning in the zebrafish, Danio rerio. Development. 1996;123:153–164. doi: 10.1242/dev.123.1.153. [DOI] [PubMed] [Google Scholar]
- Westlin J, Lardelli M. Three novel Notch genes in zebrafish: Implications for vertebrate Notch gene evolution and function. Dev Genes Evol. 1997;207:51–63. doi: 10.1007/s004270050091. [DOI] [PubMed] [Google Scholar]