Abstract
Background:
Folk medicine uses preparations of Arrabidaea chica (pariri) leaves to treat various liver pathologies. We evaluated the effects of the hydroethanolic extract of these leaves (CHEE) on an in vivo model of liver intoxication.
Materials and Methods:
Different groups of rats were treated orally for 7 days with CHEE at doses of 300, 500 or 600 mg/kg or silymarin at 35 mg/kg. The control group received only 0.5 ml of distilled water. After 7 days of treatment, both the groups received CCl4, and activities of glutamic pyruvic transaminase (GPT), glutamic oxaloacetic transaminase (GOT) and bilirubin level were assessed. The ability of CHEE to suppress hepatic injury triggered by CCl4 was evaluated based on suppression (%) of activities of GOT, GPT and bilirubin levels.
Results:
The chromatograms of the CHEE obtained at 330 and 400 nm show features of two main classes of secondary metabolites: quinones and flavonoids. The administration of 300, 500 or 600 mg/kg of CHEE resulted in the reduction of GPT levels by 85.34%, 88.59% and 93.72%, respectively. The suppression of GOT levels was 56.86%, 65.27% and 68.95%, respectively, and that of plasma bilirubin was 83.81%, 83.12% and 84.14%, respectively. These results demonstrate the protective character of CHEE and its ability to maintain the functional integrity of hepatic cells.
Conclusions:
The results obtained are possibly due to the presence of quinones and flavonoids in A. chica CHEE, both detected using high performance liquid chromatography.
Keywords: Arrabidaea chica, carbon tetrachloride, flavonoids, hepatic protection, quinones
INTRODUCTION
Arrabidaea chica (Humb. and Bonpl.) B. Verl. is a member of the Bignoniaceae family, which comprises around 120 genera and 800 species, and is distributed mainly in the tropical regions of South America and Africa.[1] In Brazil, plants of this family are widespread and found from the Amazon region to Rio Grande do Sul. They do not have a unique habitat and can be found in the Cerrado, Atlantic Forest, and the Amazon Region. In folk medicine, A. chica is known by different names, depending on the region; for example, crajiru in the State of Amazonas, pariri in Pará, and carajuru, punca-panga, and chica elsewhere.[1]
The leaves of this species have been popularly used as teas with astringent properties to treat diarrhea, anemia, leukemia, jaundice and albuminuria. This medicinal plant, when topically applied, is efficient in the treatment of dermatosis and other skin conditions, and it is also commonly used for washing wounds and ulcers.[2] Barbosa et al.[3] reported the in vitro activity of a vegetable dye from this species against Trypanosoma cruzi dermatophytes (Trichophyton mentagrophytes) and trypomastigotes.
When subjected to fermentation and manipulated as indigo, the leaves provide a dark red or brick red dye. Since ancient times, this dye has been used by Indians as body paint, ornamental paint, clothing and utensils, as well as for tattoos. The medicinal potential of this species is characterized by the popular use of its leaves and, given that its flowering is rare even in its natural habitat in the northern Brazilian states, its exchange for other species at the time of use is common.
According to Jorge et al.,[4] A. chica has traditionally been used as a healing agent for wounds. This property has been confirmed by in vitro and in vivo studies addressing the healing properties of leaf extracts of this plant. These studies showed that A. chica had a moderate antioxidant effect and stimulated fibroblast growth in a dose-dependent matter.
De Souza et al.[5] recorded the presence of a biologically active compound with hypotensive and anti-inflammatory activities. A study using A. chica leaves showed that carajurina, a 3-deoxy anthocyanidin, inhibits the transcription factor Nuclear Factor (NF)-κB, resulting in its anti-inflammatory activity. Several indigenous groups in the State of Amapα, Brazil, including Galibis, Kumarumã, Galibi Mariorno, Galibi Calin, Paliku, Karipuna and Wajãpi, use the tea made from A. chica leaves alone or in conjunction with other plant species as a hepatic detoxifying remedy for malaria.[6]
Based on folk medicine, in which the leaves of A. chica (pariri) are used to treat diseases wherein liver function is compromised, we evaluated the effect of a hydroalcoholic extract of this plant species in an in vivo model of liver intoxication.
MATERIALS AND METHODS
Acquisition of plant material
We obtained 8 kg of fresh plant material collected in the metropolitan region of Belém, Par(á, Brazil, in May 2009. A herbarium specimen of the plant material was prepared and identified by comparing it to another specimen from the Herbarium of the Museu Paraense Emilio Goeldi registered under number MG 123919.
Preparation of plant material
Intact leaves were washed with water and dipped in ethanol for microbiological stabilization. After drying, the material was dehydrated at 40°C in an oven with circulating air. The material was then crushed with a knife grinder and weighed. A final amount of 1250 g (15.63% of the fresh plant weight) was obtained.
Processing and analysis of the extract
The maceration of 1 kg of the plant drug for 8 days in 10 l of 70% (v/v) ethanol resulted, after filtration, in 8 l of A. chica extract. The average pH of the resulting extract, which was measured using a potentiometer calibrated at pH 4 and 7, was 5.6. The dye was concentrated under reduced pressure until complete evaporation of the ethanol. The water was removed by evaporation of the azeotrope formed with n-butanol and by lyophilization. The final yield of crude hydroalcoholic extract was 177.87 g (CHEE).
Phytochemical screening
The phytochemical screening of the secondary metabolites in the CHEE was performed according to the technique described by Barbosa et al.[3] We observed the presence of saponins, reducing sugars, organic acids, phenols, tannins, alkaloids (cross-reaction with quinone products), depsides, depsidones, steroids, triterpenoids, flavonoids, catechins, coumarin and benzoquinone derivatives as well as naphthoquinones and phenantraquinones.
CHEE fractionation
Forty-three grams of CHEE suspended in MeOH/H2O (8:2) was fractionated by liquid–liquid partition with increasing polarity solvents (4 × 200 ml), which resulted in hexane, chloroform, ethyl acetate and residual fractions. The phytochemical fraction approach showed a strong reaction for quinones in the chloroform fraction and for flavonoids in the methanol fraction. The extract and fractions were analyzed by thin-layer chromatography (TLC) on activated 10 cm × 10 cm normal phase chromatographic silica gel plates (Merck®). The fraction samples were prepared at a concentration of 5 mg/ml in their respective solvents in addition to the preparation of a 10 mg/ml crude extract fraction dissolved in MeOH/H2O (9:1). The chromatogram was eluted with hexane/acetone/methanol (67.5:27.5:5; v/v), developed with an aminoethanol diphenylborate-polyethilene glicol - NP/PEG solution and observed under ultraviolet light (365 nm). A band with Rf = 0.17 was observed in the hexane and ethyl acetate fractions.
Determination of the chromatographic profile by high performance liquid chromatography
Samples of CHEE diluted at either 10 mg/ml for the crude extract or 5 mg/ml for the other fractions were dissolved in high performance liquid chromatography (HPLC)-grade methanol, filtered through a 0.45-μm membrane and analyzed in a chromatography system (Merck-Hitachi, model D-7000) equipped with a detector with a diode arrangement and a LiChrospher® 100 HPLC Cartridge 250-3 RP-18 HPLC column. The samples were eluted with acetonitrile (A) and ultrapure water at pH 3 by the addition of trifluoroacet acid - TFA (B) in a gradient of 18:82 A:B at t = 0 min; 55:45 at t = 20 min; 0:100 at t = 50 min and 82:18 at t = 60 min. The column was maintained at 26°C (±1°C). Chromatograms were recorded at 254, 330, 400 and 450 nm, with monitoring at 254 nm.
Evaluation of the hepatoprotective activity of CHEE
Animals
Male Wistar rats weighing around 250 ± 5 g were obtained from the animal facilities of the Laboratório Central do Estado do Amapá – LACEN. The animals were kept in a temperature- and humidity-controlled environment in polyethylene boxes in groups of five, and they received food and water ad libitum. The project was approved by the Research Ethics Committee of the Universidade Federal do Amapâ, according to protocol number 004/2010.
Experimental procedure
Different animal groups (n = 5 per group) were treated orally for 7 days with CHEE (300, 500 or 600 mg/kg) or silymarin (35 mg/kg), and the control group was treated with 0.5 ml distilled water. On the 7th day, all the groups were given 1.5 ml/kg CCl4 orally. The animals were kept at room temperature with free access to water and food. Twenty-four hours after the treatment with CCl4, the animals were anesthetized with 45 mg/kg sodium pentobarbital (i.p.) and blood samples from the abdominal aorta were collected. Serum glutamic oxaloacetate transaminase (GOT), serum glutamic pyruvate transaminase (GPT) activities and bilirubin level of the blood samples obtained were determined according to the methods of Kondo et al.[7] The effect of CHEE in suppressing liver injury triggered by CCl4 was evaluated in terms of suppression (%) of the elevation of the activity of GOT, GPT and bilirubin. Suppression was calculated using the following equation:
Suppression (%) of GOT, GPT or bilirubin activity = {1 −(A − B)/(C − B)}× 100,
where A is the average GPT activity of the CHEE treated group, B is the average GPT, GOT or bilirubin activity of the normal group, and C is the average activity of GPT, GOT and bilirubin in the control group, which was treated with CCl4 only.
Evaluation of hematological parameters after CHEE treatment
The acute toxicity of CHEE was evaluated by the analysis of hematological parameters performed on the 7th day, after the daily oral administration of 300, 500 or 600 mg/kg CHEE.[8]
For hematological analysis, the animals were anesthetized with 45 mg/kg sodium pentobarbital (i.p.), and blood samples were collected from the abdominal artery. The activities of plasma enzymes such as aspartate aminotransferase (AST – GOT) and alanine aminotransferase (ALT – GPT) were measured according to the kinetic method by Deutsche Gesellschaft fur Klinische Chemie and by the kinetic method described by Hausamen et al.[9] The analysis of erythrocytes, leukocytes and differentiated leukocyte numbers was done using an automatic blood cell counter, model D2. Hemoglobin was measured with a hemoglobinometer using the methods of cyanmethamoglobin and microhematocrit as described by Strumia et al.[10] The results obtained in the various tests were expressed as mean ± SEM of each experimental group.
Statistical analysis
Statistical analysis of the results was performed using the Mann–Whitney “U” test. For comparison of the measurements obtained in different experiments, we used the Student's “t” test (unpaired). Results with P levels greater than 0.05 were considered statistically significant.
RESULTS AND DISCUSSION
Determination of the HPLC chromatographic profile
The chromatograms obtained at 330 and 400 nm show features of two main classes of secondary metabolites: quinones and flavonoids. Specifically, on the 330 nm chromatogram, we observed significant peaks between 4 and 24 nm, corresponding to spectra characteristic of flavonoids [Figure 1]. We highlight the peak with retention time (Rt) = 8.67 min, purity = 0.9914, on the ultraviolet spectrum with two maximum absorbances, 275 and 324 nm [Figure 2], which, according to Harbone,[11] are characteristic of flavonols, which have a hydroxyl at the C3 position. Among the members of this metabolic class, previously described in this species are kaempferol, vicenin-2 and 4′-hydroxy-3,7-dimethoxyflavone.[3]
Figure 1.
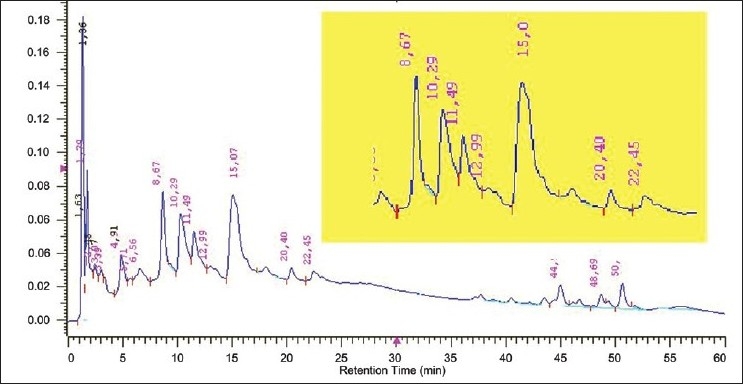
A. chica (CHEE) HPLC-DAD chromatogram at 330 nm. The highlighted inset shows the peaks registered between 4 and 24 min, especially with a retention time of 8.67 min. Lower intensity peaks were observed at retention times between 37 and 54 min
Figure 2.
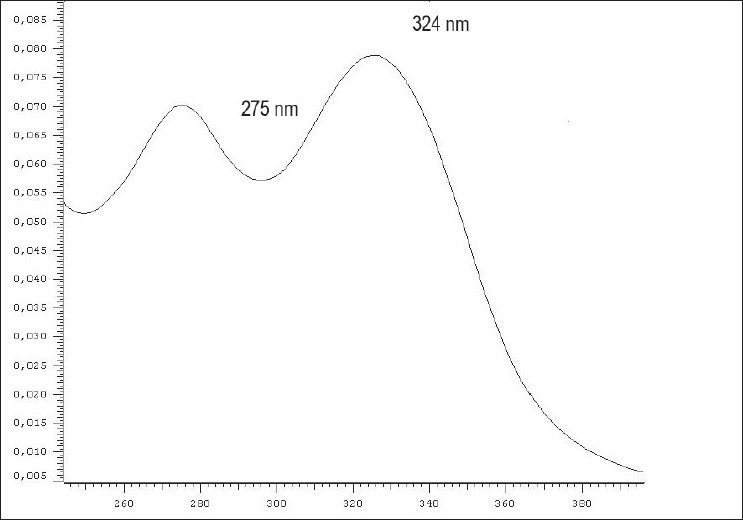
Spectra of the A. chica (CHEE) HPLC-DAD peak observed at 330 nm, with Rt = 8.67 min and a purity of 0.9914. We observed two maxima of absorption, one at 275 nm and another at 324 nm
On the chromatogram obtained at 400 nm [Figure 3], we observed peaks corresponding to spectra characteristic of quinones at retention times between 37 and 54 min, which were less intense at 330 nm. We highlight the peak with Rt = 50.64 min, purity = 0.9989 and four maximum absorbances: 222, 278, 324 and 404 nm [Figure 4].
Figure 3.
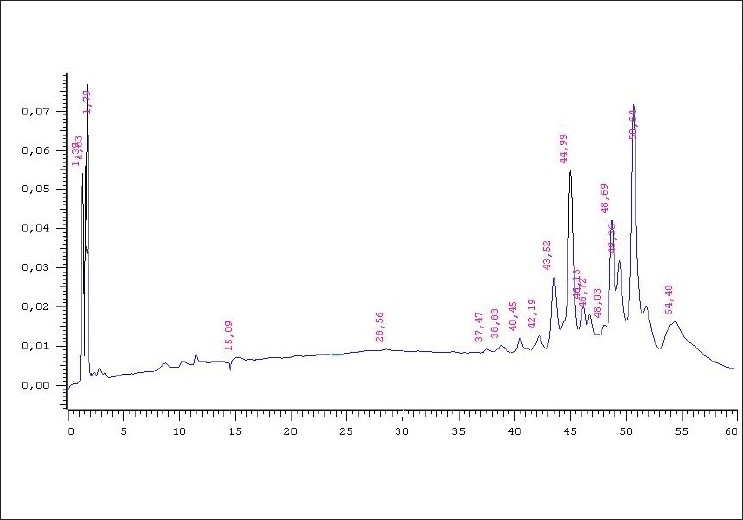
A. chica (CHEE) HPLC-DAD chromatogram at 400 nm. The peaks between the retention times of 37 and 54 min are noteworthy, especially the one indicated by the arrow with Rt = 50.64 min
Figure 4.
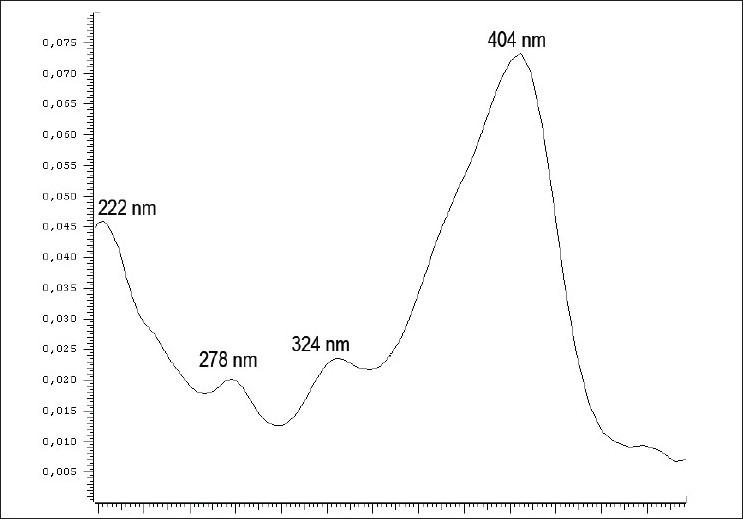
Spectra of the peak observed in the A. chica (CHEE) HPLCDAD chromatogram at 400 nm with Rt = 50.64 min, purity of 0.9989 with four absorption maxima: 222, 278, 324 and 404 nm
Analysis of the hepatoprotective activity of CHEE
The liver is a large, solid and highly complex organ with many functions, including nutrient storage, carbohydrate homeostasis maintenance, secretory and excretory functions, protein synthesis and certain vital metabolic functions unique to this organ. These functions include the metabolism of hormones (insulin, glucagon, thyroxine and glucocorticoids), lipid metabolism [cholesterol, triglycerides and high-density lipoproteins (HDL)], protein metabolism (amino acid degradation and the synthesis of plasma factors, proteins and urea) and detoxification of xenobiotics.[12]
Disorders of the liver are as complex as the organ itself. There are not many drugs within the realm of conventional medicine for the treatment of diseases such as hepatitis, cirrhosis, liver injury by toxins and biliary tract disorders. However, drugs of plant origin from Chinese indigenous and traditional medicine have been used to treat liver and biliary tract diseases. Pharmacological and clinical assays have demonstrated the beneficial effects of these drugs.[12] One of the most effective compounds used is silymarin, extracted from Silybum marianum (L.) Gaertn. (Compositae), which is used for all types of liver disorders, in particular to counteract severe toxicity from the ingestion of fatal substances. In our experiments, animals treated orally with 35 mg/kg silymarin showed 86% suppression of the injury caused by CCl4. This suppressive effect was also observed in the groups treated with A. chica CHEE. Specifically, administration of 300, 500 and 600 mg/kg CHEE for 7 days decreased GPT plasma levels by 85.34%, 88.59%, and 93.72%, respectively. GOT levels were decreased by 56.86%, 65.27% and 68.95%, respectively, and plasma bilirubin was lowered by 83.81%, 83.12% and 84.14%, respectively. These results demonstrate the protective ability of this compound in maintaining the functional integrity of hepatic cells [Table 1].
Table 1.
Biochemical parameters of animals (rats) treated orally for 7 days with 300, 500 or 600 mg/kg Arrabidaea chica (CHEE), 35 mg/kg silymarin or 0.5 ml water after administration of carbon tetrachloride

Several studies have shown that the administration of CCl4 leads to the formation of trichloromethyl peroxide radicals, which are involved in liver damage.[13,14] The high levels of GPT, GOT and bilirubin in the serum of animals treated with only carbon tetrachloride (control group) was a result of the ability of this compound to cause hepatic dysfunction and promote the lesion of hepatic cells.[15] This was not observed in groups treated with A. chica CHEE.
Several factors are related to the induction of liver injury, such as endotoxemia which is commonly observed in patients with certain liver diseases and in animals with experimental liver injuries.[16] It has been shown that lipid peroxidation in the livers of rats treated with CCl4 increases with lesions[17,18] and that the levels of lipid peroxides in the hepatic microsomal fraction also increase with the progression of liver injury and decrease with treatment.[17]
Gonzáles Padróon et al.[19] demonstrated the possibility that the decrease of the hepatic microsomal glucose-6-phosphatase activity in rats intoxicated with CCl4 is maintained as a result of the effect of lipid peroxidation on the cellular components present in the phase of the G6 system. It has been also demonstrated that exposure of the hepatic microsome to ascorbic acid//Fe+2, the product of lipid peroxidation, contributes to the inactivation of the G6 phase.[20] Thus, for protection against the hepatic injury caused by CCl4, it is necessary that the drug used presents antiperoxidative action. Silymarin (flavonolignan), used here as a standard, acts by incorporating itself to the cell membrane, thus increasing membrane resistance against injury, probably by changing the membrane physical and chemical properties. This also protects physiological antioxidants (glutathione and superoxide dismutase), preventing their depletion and consequently preserving cell function and structure.[16] Animals treated with A. chica CHEE and subsequently subjected to CCl4 intoxication showed suppression of injury.
Anthocyanins, flavonoids, tannins, phytosterols and flavones have been identified in extracts prepared from A. chica leaves. Considering that several flavonoids such as quercetin, in addition to having antioxidant activity, can exert a variety of effects on liver metabolism, the presence of flavonoids in the extract from A. chica leaves suggests that this extract could also affect cellular energy metabolism.[2]
We observed the presence of 3-deoxy anthocyanidin, 6,3′-trihydroxy-5-4′-dimethoxy-flavylium, 6,7,3′,4′-tetrahydroxy-5-methoxy-flavylium and 6,7-dihydroxy-5,4′-dimethoxy-flavylium (carajurina) in the leaves of A. chica.[21] It has been shown that the following compounds are present in the carajurina pigment, which were previously identified and isolated from A. chica dry leaves: 3-deoxy anthocyanidin, 6,7,3′,4′-tetrahydroxy-5-methoxyflavylium and 6,7,4-trihydroxy-5-methoxyflavylium.[1] All of these compounds could possibly be involved in the protective liver ability of this plant species.
Bilirubin is directly involved in liver function. It is generally considered that alterations of its levels can be an indication of liver compromise which is manifested by changes in skin coloration. We observed a significant difference between the bilirubin levels of the control group and the groups treated with CHEE, with the latter showing decreased plasma bilirubin levels, thus demonstrating its protective role in hepatic cells [Table 1].
Analysis of hematological parameters following treatment with CHEE
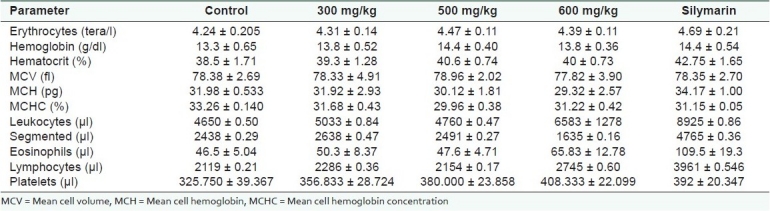
An important aspect to be considered regarding the action of new drugs is their effect on hematological parameters, which can be an indication of an adverse or serious toxic effect.[22] The different CHEE doses administered did not cause significant changes on the hematological parameters evaluated after 7 days of treatment when compared to the control group [Table 2]. These results demonstrate the absence of side effects of the acute use of CHEE with regard to the main hematological parameters.
Table 2.
Hematological parameters of male rats treated orally for 7 days with 300, 500 or 600 mg/kg Arrabidaea chica (CHEE), 35 mg/kg silymarin or 0.5 ml water
CONCLUSIONS
The results of this study show that hydroalcoholic preparations of A. chica leaves have a liver protective effect and that oral acute administration of this extract has no impact on the hematological profile of the animal species studied.
Acknowledgments
We would like to thank the Conselho Nacional de Desenvolvimento Cientνfico e Tecnológico – CNPq – Brazil, for the scholarship toward scientific initiation of the Universidade Federal do Amapá PIBIC program.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- 1.Devia B, Llabres G, Wouters J, Wouters J, Dupont L, Escribano-Bailon MT, et al. New 3-Deoxyanthocyanidins from Leaves of Arrabidaea chica. Phytochem Analysis. 2002;13:114–20. doi: 10.1002/pca.632. [DOI] [PubMed] [Google Scholar]
- 2.Takemura OS, Iinuma M, Tosa H, Miguel OG, Moreira EA, Nozawa Y. A flavone from leaves of Arrabidaea chica f. cuprea. Phytochemistry. 1995;38:1299–300. [Google Scholar]
- 3.Barbosa WL, Pinto LN, Quignard E, Vieira JM, Silva JO, Jr, Albuquerque S. Arrabidaea chica (HandB) Verlot: Phytochemical approach, antifungal and trypanocidal activities. Rev Bras Farmacogn. 2008;18:544–8. [Google Scholar]
- 4.Jorge MP, Madjarof C, Gois Ruiz AL, Fernandes AT, Ferreira Rodrigues RA, de Oliveira Sousa IM, et al. Evaluation of wound healing properties of Arrabidaea chica Verlot extract. J Ethnopharmacol. 2008;118:361–6. doi: 10.1016/j.jep.2008.04.024. [DOI] [PubMed] [Google Scholar]
- 5.de Souza AS, Pagadigorria CL, Ishii-Iwamoto EL, Bracht A, Cortez DA, Yamamoto NS. Effects of the Arrabidaea chica extract on energy metabolism in the rat liver. Pharm Biol. 2009;47:154–61. [Google Scholar]
- 6.da Mata ND. Participação da mulher Wajãpi no uso tradicional de plantas medicinais.Dissertação (Mestrado Integrado em Desenvolvimento Regional) - Universidade Federal do Amapá, 2009. 2009:112. [Google Scholar]
- 7.Kondo K, Takahashi M, Ohmizu H, Matsumoto M, Taguchi I, Iwasaki T. 2,2′-disubstituted biphenys: Synthesis and suppressive effect against carbon tetrachloride-induced liver injury. Chem Pharmac Bull. 1994;42:62–6. doi: 10.1248/cpb.42.62. [DOI] [PubMed] [Google Scholar]
- 8.Stevens KP, Gallo MA. Practical considerations in the conduct of chronic toxicity studies. In: Hayes AW, editor. Principles and Methods of toxicology. New York: Raven Press; 1982. pp. 53–77. [Google Scholar]
- 9.Hausamen TU, Helger R, Rick W, Gross W. Optimal conditions for the determination of serum alkaline phosphatase by a new kinetic method. Clin Chim Acta. 1967;15:241–5. [Google Scholar]
- 10.Strumia MM, Sample AB, Hart ED. An improved microhematocrit method. Am J Clin Pathol. 1954;24:1016–24. doi: 10.1093/ajcp/24.9.1016. [DOI] [PubMed] [Google Scholar]
- 11.Harborne JB. 3rd ed. London: Chapman and Hall; 1988. Phytochemical Methods: A guide to modern techniques of plant analysis; p. 302. [Google Scholar]
- 12.Williamson EM, Okpako DT, Evans FJ. London: John Wiley and Sons; 1996. The liver and biliary system: Selection, preparation and pharmacological evaluation of plant material; p. 228. [Google Scholar]
- 13.Upadhyay R, Pandey ND, Narvi SS, Verma A, Ahmed B. Antihepatotoxic effect of Feronia limonia fruit against carbon tetrachloride induced hepatic damage in albino rats. Chinese Med. 2010;1:18–22. [Google Scholar]
- 14.Cheeseman KH, Albano EF, Thomasi A, Slater T. Biochemical studies on the metabolic activation of halogenated alkanes. Environ Health Perspect. 1985;64:85–101. doi: 10.1289/ehp.856485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Singh K, Khanna AK, Chandan R. Hepatoprotective activity of ellagic acid against carbon tetrachloride induced hepatotoxicity in rats. Indian J Exp Biol. 1999;37:1025–6. [PubMed] [Google Scholar]
- 16.Leng-Peschlow E. Properties and medical use of flavonolignans (Silymarin) from Silybum marianum. Phytother Res. 1996;10:25–6. [Google Scholar]
- 17.Aguilar-Delfin I, López-Barrera F, Hernández-Muñoz R. Selective enhancement of lipid peroxidation in plasma membrane in two experimental models of liver regeneration, partial hepatectomy and acute CCl4 administration. Hepatology. 1996;24:657–62. doi: 10.1002/hep.510240331. [DOI] [PubMed] [Google Scholar]
- 18.Ohta Y, Nishida K, Sasaki E, Kongo M, Ishiguro I. Attenuation of disrupted hepatic active oxygen metabolism with the recovery of acute liver injury in rats intoxicated with carbon tetrachloride. Res Commun Mol Pathol. 1997;95:191–208. [PubMed] [Google Scholar]
- 19.González Padrón A, De Toranzo EC, Castro JA. Depression of liver microssomal glucose-6-phosphatase activity in carbon tetrachloride-poisoned rats: Potential synergistic effects of lipid peroxidation and of covalent binding of haloalkane-derived free radicals to cellular components in the process. Free Radical Biol Med. 1996;21:81–7. doi: 10.1016/0891-5849(95)02223-6. [DOI] [PubMed] [Google Scholar]
- 20.Ohyashiki T, Kamata K, Takeuchi M, Matsui K. Contribuition of peroxidation products to oxidative inactivation of rat liver microsomal glucose-6-phosphatase. J Biochem. 1995;118:508–14. doi: 10.1093/oxfordjournals.jbchem.a124937. [DOI] [PubMed] [Google Scholar]
- 21.Zorn B, Garcia-Piñeres AJ, Castro V, Murillo R, Mora G, Merfort I. 3-Desoxyanthocyanidins from Arrabidaea chica. Phytochemistry. 2001;56:831–5. doi: 10.1016/s0031-9422(01)00038-3. [DOI] [PubMed] [Google Scholar]
- 22.Acharya RN, Shukla VJ, Skandhan KP, Sharma PP, Ravishankar B. Toxicological evaluation of Cynoglossum glochidiatum Wall. Ex. Benth (Tejaraj): A folklore aphrodisiac medicinal plant. Ayu. 2009;30:175–80. [Google Scholar]


