Abstract
Designer natriuretic peptides (NPs) are novel hybrid peptides that are engineered from the native NPs through addition, deletion, or substitution of amino acid(s) with a goal toward optimization of pharmacological actions while minimizing undesirable effects. In this article, selected peptides that were designed in our laboratory are reviewed, and future directions for research and development of designer NPs are discussed.
Keywords: natriuretic peptide, renal function, hybrid peptides
INTRODUCTION
Designer natriuretic peptides (NPs) are novel hybrid peptides that are engineered from the native NPs through addition, deletion, or substitution of amino acid(s) (AA) with a goal toward optimization of pharmacological actions while minimizing undesirable effects. These strategies are often based on the integration of key structural determinants from the native NPs in activating the second messenger, cyclic guanosine monophosphate (cGMP), via the NP receptors (NPRs), resulting in renal, hemodynamic, and humoral effects that are characteristic of the native NPs. These designer peptides may exhibit improved pharmacological profiles as compared with the native peptides and may also possess unique biological actions that are not found in the native NPs.1 In this article, we review 2 hybrid peptides that were designed in our laboratory in the past 15 years and discuss future directions for the research and development of novel designer NPs.
Native Natriuretic Peptides—The Backbone for Chimeric/Designer Peptides
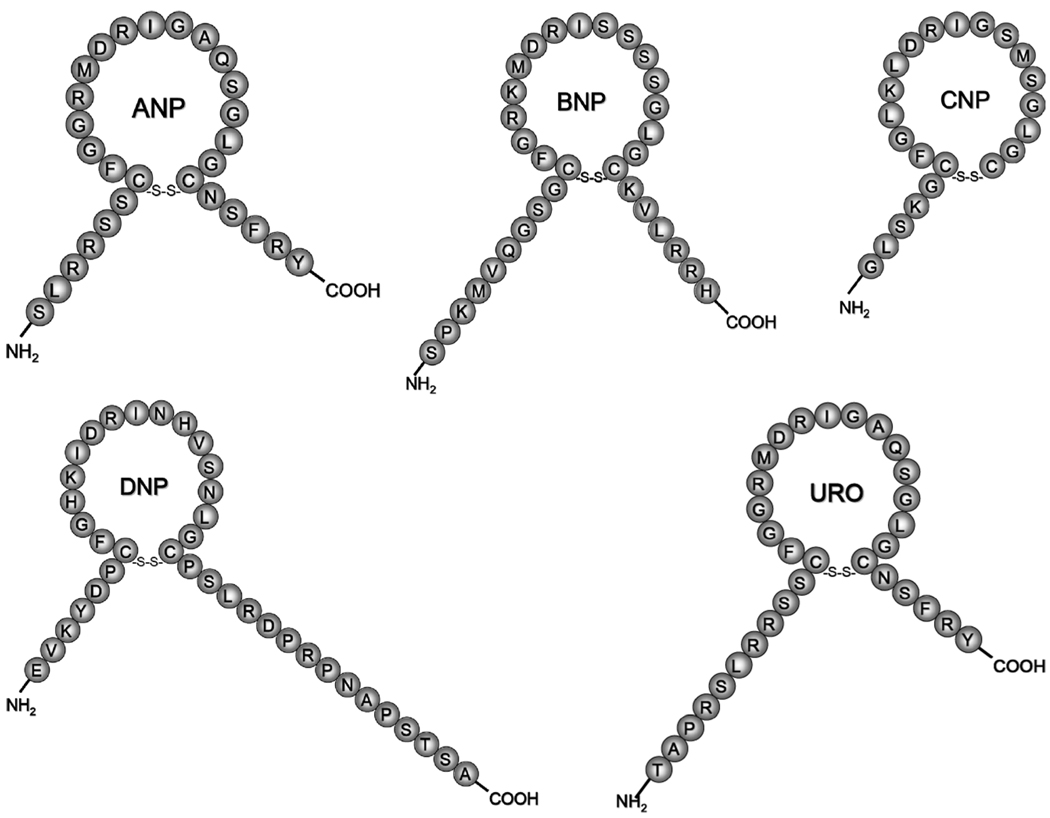
Four mammalian NPs, atrial natriuretic factor (or atrial NP, ANP),2 B-type NP (BNP),3 C-type NP (CNP),4 and urodilatin (URO),5 have been identified and characterized (Fig. 1). In addition, Dendroaspis NP, from the green mamba snake (Dendroaspis angusticeps),6 has been isolated and studied.7–9 Atrial NP, BNP, URO, and Dendroapsis NP (DNP) are ligands for NPR-A, whereas CNP is the ligand for NPR-B.6,10–13 Activation of NPR-A or NPR-B results in an increase in the second messenger, cGMP, which mediates favorable physiological responses in the cardiorenal axis and counteracts neurohumoral activation under pathophysiological conditions.13
FIGURE 1.
Amino acid structures for atrial natriuretic peptide (ANP), B-type natriuretic peptide (BNP), C-type natriuretic peptide (CNP), Dendroaspis natriuretic peptide (DNP), and urodilatin (URO).
Vasonatrin
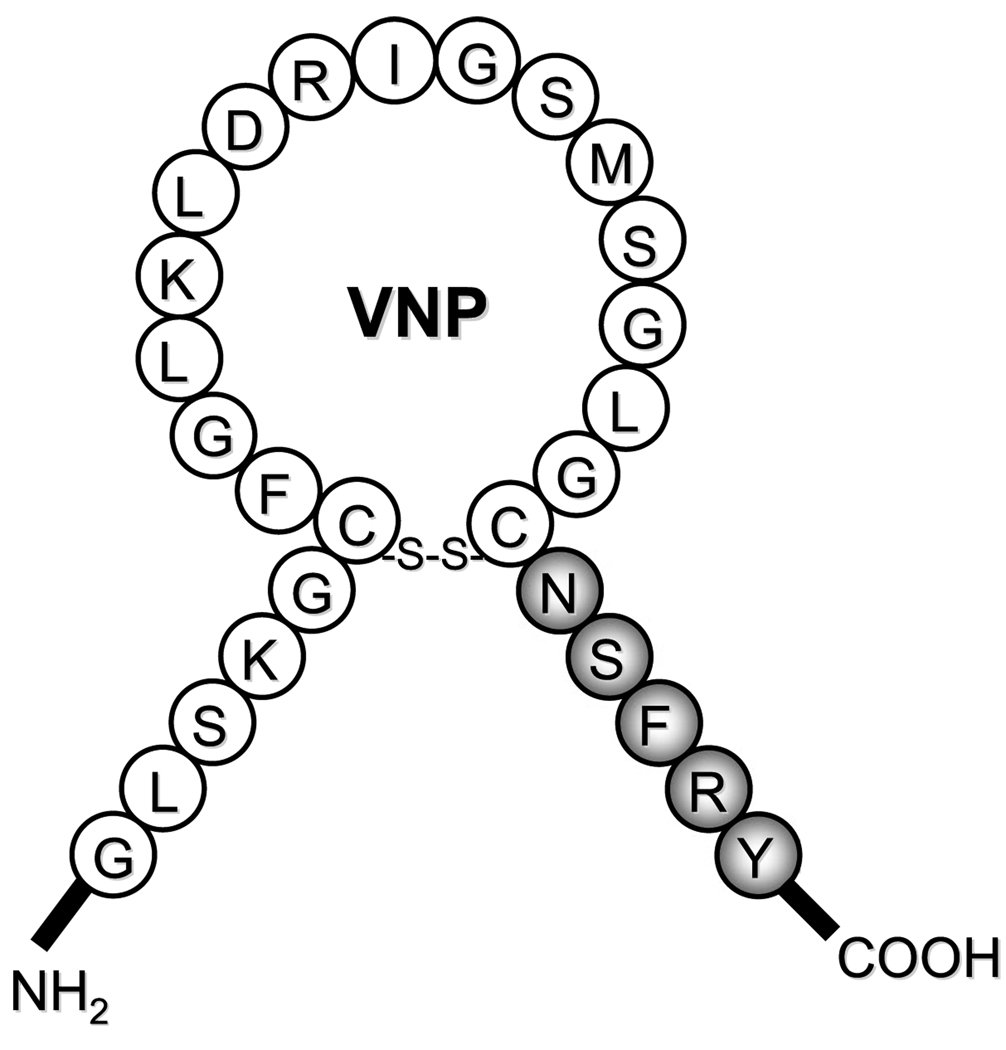
Vasonatrin NP (VNP) was a synthetic NP that was designed in the Burnett Laboratory.1 It was a 27-AA peptide, consisting of the full-length, 22-AA of human CNP and the 5-AA C-terminus of human ANP (Fig. 2).1 In 1993,Wei et al1 from our laboratory reported that VNP exerted venodilating actions like CNP and induced natriuresis like ANP but also possessed arterial vasodilating properties that were unique to VNP.1 The rank order of vasorelaxation was observed to be VNP > ANP ≥ CNP in systemic arteries and VNP > CNP > ANP in systemic veins.1 The vasorelaxant effect of VNP was independent of the endothelium.1,14 In rats, VNP 50 µg/kg intravenously administered (IV) bolus increased plasma cGMP, urinary cGMP excretion, urine flow, and urinary sodium excretion, as well as decreased mean arterial pressure (MAP) and right atrial pressure, with enhanced natriuresis, diuresis, and urinary cGMP excretion in response to VNP versus CNP, but were less than those with ANP.1 In autoradiographic studies, VNP was demonstrated to bind primarily to NPR-C in isolated rat and canine glomeruli and in canine femoral arteries.15 In rat glomerular membranes, binding of VNP to NPR-A was demonstrated by cross-linking studies.15 In isolated rat glomeruli, increase in cGMP production was induced by VNP (1 µM), suggesting NPR-A activation in the glomeruli.15
FIGURE 2.
Amino acid structures for vasonatrin (VNP).
In isolated rat pulmonary artery, abdominal aorta, and celiac vein, the vasorelaxant effects of VNP have been demonstrated by Feng et al.,16 with median effective concentrations of 16, 35, and 12 nmol/L, respectively. When compared with ANP and CNP, the rank orders of the vasorelaxant effects were VNP > ANP ≥ CNP for pulmonary artery, VNP > ANP > CNP for abdominal aorta, and VNP > CNP > ANP for celiac vein.16 The vasorelaxant effects of VNP were similar regardless of the presence or absence of the endothelium.16 In addition, this vasorelaxant effect has also been observed in human intramammary artery in a dose-dependent and endothelium-independent manner.17
In neonatal rat cardiac fibroblasts, VNP has been reported to attenuate hypoxia-induced, growth-promoting effects and increase in intracellular Ca2+.16,18 In pulmonary arterial smooth muscle cells, VNP inhibited proliferation to a greater extent than ANP and CNP.19
CD-NP
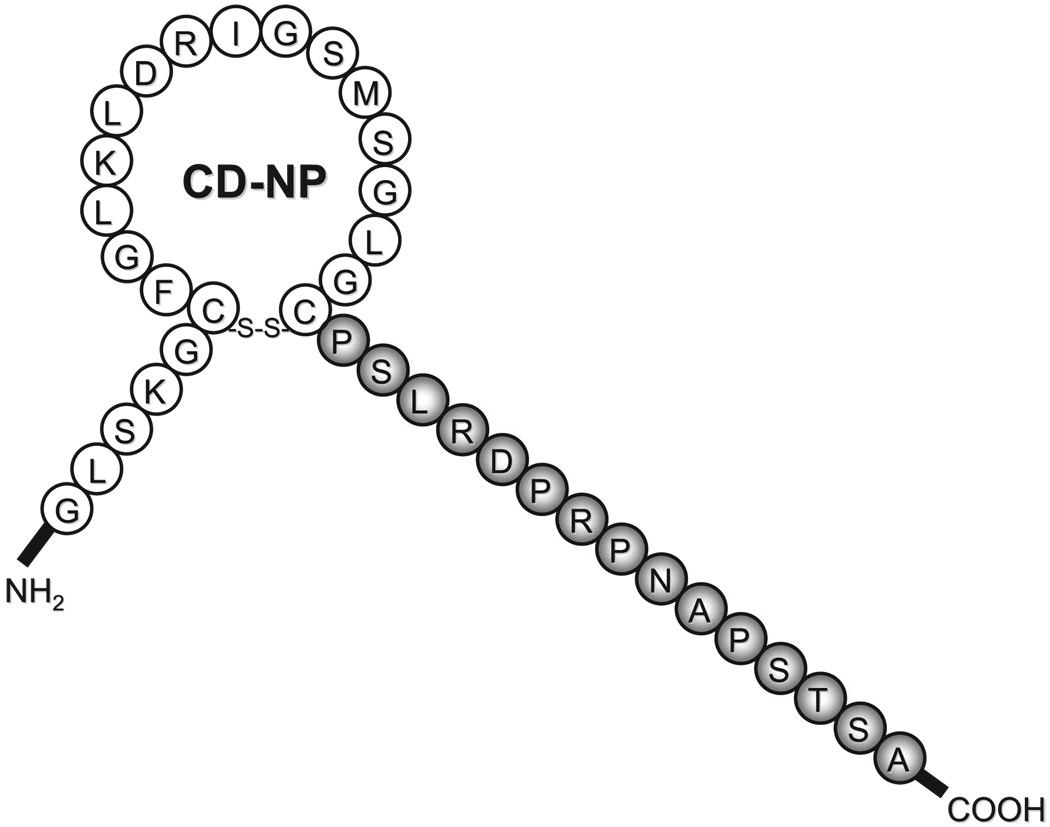
More recently, a novel chimeric NP, named CD-NP, was designed by Lisy et al in our laboratory.20 CD-NP consists of the full-length 22 AAs of human CNP and the 15-AA C-terminus of DNP (Fig. 3). The rationale for the design of CD-NP was to transform CNP into a CNP-like peptide with enhanced renal actions without inducing systemic hypotension, as the latter is a clinically important side effect with the use of recombinant ANP21 and BNP,22 and in the clinical development of URO.23,24 C-type NP, an NP of endothelial cell origin, was selected for this modular drug design, as CNP has been demonstrated to exert predominantly venodilating effects25 and thus is less likely to result in systemic hypotension.13 The avoidance of excessive hypotension is an important consideration in the design of novel peptides to preserve renal perfusion pressure and renal function. 26,27 In addition, CNP possesses antiproliferative effects,28 which is a highly desirable property for novel cardiovascular drugs against hypertrophy and remodeling. However, an important limitation of CNP is that it exerts only modest renal actions,29 whereas DNP is potently natriuretic and diuretic, but it induces significant hypotension.7,8 Thus, CD-NP was synthesized with the goal of combining the above complementary profiles of CNP and DNP into a single chimeric NP with renal-enhancing actions without inducing excessive hypotension.
FIGURE 3.
Fusion peptide of C-type natriuretic peptide and C-terminus of Dendroapsis natriuretic peptide (CD-NP).
In normal anesthetized dogs, CD-NP activates cGMP and exerts natriuretic, diuretic, and cardiac unloading and renin-angiotensin–suppressing actions with minimal effects on systemic blood pressure.30,31 When compared with conventional therapy (BNP) on an equimolar basis, Lisy et al.32 demonstrated that CD-NP 50 ng/kg per minute IV exerted significantly less effect on systemic blood pressure and was associated with a greater increase in glomerular filtration rate. When compared with an equimolar dose of CNP, Lee et al.31 demonstrated that CD-NP 50 ng/kg per minute IV elicited greater increases in plasma cGMP, urinary cGMP excretion, and net renal generation of cGMP, which was associated with enhanced natriuresis. Moreover, CD-NP, not CNP, enhanced glomerular filtration rate and suppressed plasma renin activity and angiotensin II.31 Taken together, these findings suggest that CD-NP represents a successful transformation of CNP to a CNP-like peptide with enhanced renal and neurohumoral actions.
In vitro, CD-NP stimulated cGMP production in cultured human cardiac fibroblasts to a greater extent than equimolar concentrations of BNP and DNP (10−6 mol/L).20 CD-NP also exerted antiproliferative actions in cultured human cardiac fibroblasts that were treated with CT-1 to induce cell proliferation. 20 Moreover, CD-NP (10−6 mol/L), when compared with an equimolar concentration of CNP, stimulated an 8-fold greater cGMP response, which was attenuated by an NPR-A antagonist, suggesting involvement of NPR-A in mediating the cGMP response.33 In comparison, the actions of CD-NP in human cardiac fibroblasts, in which NPR-B is expressed,34 would be consistent with NPR-B activation by CD-NP. Thus, CD-NP may represent a novel chimeric NP that targets both NPR-A and NPR-B.
Following toxicology studies in rats and in dogs and subsequent regulatory approval, the first-in-human clinical trial on CD-NP was recently conducted.35 This first-in-human clinical trial consisted of 2 stages: an open-label sequential dose escalation study (stage 1) and a randomized, double-blind, placebo-controlled study (stage 2).35 In the first stage, 3 cohorts with a total of 12 subjects (4 subjects per cohort) were enrolled in the dose-escalation study. In stage 2, 10 subjects were randomized in the double-blind study, which evaluated the maximum tolerated dose (as determined in the first stage) of CD-NP versus placebo as a 4-hour continuous infusion. In the dose-escalation study, significant increases in plasma cGMP were observed when CD-NP was infused at 10, 17.5, and 25 ng/kg per minute IV.35 Symptomatic orthostatic hypotension was observed in 2 subjects at the latter dose. The maximum tolerated dose was subsequently determined to be 17.5 ng/kg per minute and confirmed.35 In the second stage, CD-NP significantly increased plasma cGMP, urinary cGMP excretion, and urinary sodium excretion, as compared with placebo.35 Urine flow increased significantly in the CD-NP group versus baseline, whereas no significant increases were observed in the placebo group.35 Glomerular filtration rate was preserved in both groups without a significant between-group difference in MAP, despite a slight decrease in MAP in the CD-NP group from baseline: (mean ± SEM) 85 ± 2 mm Hg to the end of infusion 82 ± 3 mm Hg (P < 0.05). No severe adverse events were reported. Overall, the favorable cGMP-activating, renal and hemodynamic effects of CD-NP that were observed in experimental studies30,31 were indeed demonstrated for the first time in humans. The in vivo renal mechanisms of action of CD-NP and the therapeutic potential of CD-NP in human heart failure and the acute coronary syndromes are being explored in multicenter clinical trials.
Future Directions
As highlighted in a recent review by Letts and Loscalzo,36 “chimeric molecules are single-chemical entities that possess at least 2 separate functions. In the design of new chimeric medicines, the 2 biologic actions are often designed to be synergistic37 and thereby complement each other in activating a specific target, such as a gene, a receptor or an enzyme.” Our work, to date, has demonstrated that novel designer NPs preserve favorable cardiorenal and neurohumoral effects of the native NPs while minimizing systemic hypotension, thus holding promise as a new generation of therapeutics for the prevention and treatment of various cardiorenal disease states. In particular, based on the favorable pharmacological profiles of the native NPs, it is anticipated that novel designer NPs may have potential applications in ischemic heart disease,38 post-infarction remodeling,28,39–41 peripheral vascular disease,42 chronic heart failure,43 hypertension,44,45 metabolic diseases,46,47 and cancer.48–50 As potential therapeutic applications of the NPs continue to expand from acute to chronic diseases, there is an increasing need for novel strategies to optimize chronic peptide delivery, which has become a reality.44,51 Moreover, futurework is needed to explore the therapeutic potential of combination therapy in augmenting the endogenous NP system and to identify the optimal strategies for incorporating biomarkers to guide therapy.
Acknowledgments
This study was supported by the National Institutes of Health (R01 HL36634, P01 HL76611, and R01 HL83231) and the Mayo Foundation (grants to J.C.B.), the Canadian Institutes of Health Research Clinical Research Initiative Fellowship Award (2006–07), a 2007 Heart Failure Society of America Research Fellowship Award, and the 2007 American Society for Clinical Pharmacology and Therapeutics Young Investigator Award to C.Y.W.L.
REFERENCES
- 1.Wei CM, Kim CH, Miller VM, et al. Vasonatrin peptide: a unique synthetic natriuretic and vasorelaxing peptide. J Clin Invest. 1993;92:2048–2052. doi: 10.1172/JCI116800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.de Bold AJ, Borenstein HB, Veress AT, et al. A rapid and potent natriuretic response to intravenous injection of atrial myocardial extract in rats. Life Sci. 1981;28:89–94. doi: 10.1016/0024-3205(81)90370-2. [DOI] [PubMed] [Google Scholar]
- 3.Sudoh T, Kangawa K, Minamino N, et al. A new natriuretic peptide in porcine brain. Nature. 1988;332:78–81. doi: 10.1038/332078a0. [DOI] [PubMed] [Google Scholar]
- 4.Sudoh T, Minamino N, Kangawa K, et al. C-type natriuretic peptide (CNP): a new member of natriuretic peptide family identified in porcine brain. Biochem Biophys Res Commun. 1990;168:863–870. doi: 10.1016/0006-291x(90)92401-k. [DOI] [PubMed] [Google Scholar]
- 5.Schulz-Knappe P, Forssmann K, Herbst F, et al. Isolation and structural analysis of “urodilatin,” a new peptide of the cardiodilatin-(ANP)-family, extracted from human urine. Klin Wochenschr. 1988;66:752–759. doi: 10.1007/BF01726570. [DOI] [PubMed] [Google Scholar]
- 6.Schweitz H, Vigne P, Moinier D, et al. A new member of the natriuretic peptide family is present in the venom of the green mamba (Dendroaspis angusticeps) J Biol Chem. 1992;267:13928–13932. [PubMed] [Google Scholar]
- 7.Lisy O, Jougasaki M, Heublein D, et al. Renal actions of synthetic Dendroaspis natriuretic peptide. Kidney Int. 1999;56:502–508. doi: 10.1046/j.1523-1755.1999.00573.x. [DOI] [PubMed] [Google Scholar]
- 8.Lisy O, Lainchbury JG, Leskinen H, et al. Therapeutic actions of a new synthetic vasoactive and natriuretic peptide, Dendroaspis natriuretic peptide, in experimental severe congestive heart failure. Hypertension. 2001;37:1089–1094. doi: 10.1161/01.hyp.37.4.1089. [DOI] [PubMed] [Google Scholar]
- 9.Best PJ, Burnett JC, Wilson SH, et al. Dendroaspis natriuretic peptide relaxes isolated human arteries and veins. Cardiovasc Res. 2002;55:375–384. doi: 10.1016/s0008-6363(02)00402-9. [DOI] [PubMed] [Google Scholar]
- 10.Koller KJ, Lowe D, Bennett G, et al. Selective activation of the B natriuretic peptide receptor by C-type natriuretic peptide (CNP) Science. 1991;252:120–123. doi: 10.1126/science.1672777. [DOI] [PubMed] [Google Scholar]
- 11.Suga S, Nakao K, Hosoda K, et al. Receptor selectivity of natriuretic peptide family, atrial natriuretic peptide, brain natriuretic peptide, and C-type natriuretic peptide. Endocrinology. 1992;130:229–239. doi: 10.1210/endo.130.1.1309330. [DOI] [PubMed] [Google Scholar]
- 12.Singh G, Kuc RE, Maguire JJ, et al. Novel snake venom ligand Dendroaspis natriuretic peptide is selective for natriuretic peptide receptor-A in human heart: downregulation of natriuretic peptide receptor-A in heart failure. Circ Res. 2006;99:183–190. doi: 10.1161/01.RES.0000232322.06633.d3. [DOI] [PubMed] [Google Scholar]
- 13.Lee CYW, Burnett JC., Jr Natriuretic peptides and therapeutic applications. Heart Fail Rev. 2007;12:131–142. doi: 10.1007/s10741-007-9016-3. [DOI] [PubMed] [Google Scholar]
- 14.Wei CM, Kim CH, Khraibi AA, et al. Atrial natriuretic peptide and C-type natriuretic peptide in spontaneously hypertensive rats and their vasorelaxing actions in vitro. Hypertension. 1994;23:903–907. doi: 10.1161/01.hyp.23.6.903. [DOI] [PubMed] [Google Scholar]
- 15.Woodard GE, Li X, Brown J, et al. Receptor subtypes for vasonatrin peptide in renal glomeruli and arteries [published correction appears in Regul Pept 2008;2147(2001–2003):2110] Regul Pept. 2005;129:183–189. doi: 10.1016/j.regpep.2005.02.006. [DOI] [PubMed] [Google Scholar]
- 16.Feng HS, Zhang Z, Zhu X, et al. Comparison of vasorelaxing actions of vasonatrin peptide, C-type natriuretic peptide and atrial natriuretic peptide (translated from Chinese) Sheng Li Xue Bao. 1999;51:515–520. [PubMed] [Google Scholar]
- 17.Yu J, Zhe X, Wei G, et al. Vasorelaxing role of vasonatrin peptide in human intramammary artery in vitro (translated from Chinese) Sheng Li Xue Bao. 2003;55:187–190. [PubMed] [Google Scholar]
- 18.Lu SY, Zhu MZ, Guo HT, et al. Inhibition of moderate hypoxia-induced protein synthesis by vasonatrin peptide in cultured neonatal rat cardiomyocytes (translated from Chinese) Sheng Li Xue Bao. 2002;54:7–11. [PubMed] [Google Scholar]
- 19.Dong MQ, Zhu MZ, Yu J, et al. Comparison of inhibitory effects of three natriuretic peptides on the proliferation of pulmonary artery smooth muscle cells of rats (translated from Chinese) Sheng Li Xue Bao. 2000;52:252–254. [PubMed] [Google Scholar]
- 20.Lisy O, Huntley BK, McCormick DJ, et al. Design, synthesis, and actions of a novel chimeric natriuretic peptide: CD-NP. J Am Coll Cardiol. 2008;52:60–68. doi: 10.1016/j.jacc.2008.02.077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Suwa M, Seino Y, Nomachi Y, et al. Multicenter prospective investigation on efficacy and safety of carperitide for acute heart failure in the ’real world’ of therapy. Circ J. 2005;69:283–290. doi: 10.1253/circj.69.283. [DOI] [PubMed] [Google Scholar]
- 22.Publication Committee for the VMAC Investigators (Vasodilation in the Management of Acute CHF) Intravenous nesiritide vs nitroglycerin for treatment of decompensated congestive heart failure: a randomized controlled trial [published correction appears in JAMA 2002;1288:1577] JAMA. 2002;287:1531–1540. doi: 10.1001/jama.287.12.1531. [DOI] [PubMed] [Google Scholar]
- 23.Mitrovic V, et al. Haemodynamic and clinical effects of ularitide in decompensated heart failure. Eur Heart J. 2006;27:2823–2832. doi: 10.1093/eurheartj/ehl337. [DOI] [PubMed] [Google Scholar]
- 24.Dorner GT, Selenko N, Kral T, et al. Hemodynamic effects of continuous urodilatin infusion: a dose-finding study. Clin Pharmacol Ther. 1998;64:322–330. doi: 10.1016/S0009-9236(98)90181-3. [DOI] [PubMed] [Google Scholar]
- 25.Wei CM, Aarhus LL, Miller VM, et al. Action of C-type natriuretic peptide in isolated canine arteries and veins. Am J Physiol. 1993;264:H71–H73. doi: 10.1152/ajpheart.1993.264.1.H71. [DOI] [PubMed] [Google Scholar]
- 26.Burnett JC, Jr, Opgenorth TJ, Granger JP. The renal action of atrial natriuretic peptide during control of glomerular filtration. Kidney Int. 1986;30:16–19. doi: 10.1038/ki.1986.144. [DOI] [PubMed] [Google Scholar]
- 27.Burnett JC, Jr, Korinek J. The tumultuous journey of nesiritide: past, present, and future. Circ Heart Fail. 2008;1:6–8. doi: 10.1161/CIRCHEARTFAILURE.108.776294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Soeki T, Kishimoto I, Okumura H, et al. C-type natriuretic peptide, a novel antifibrotic and antihypertrophic agent, prevents cardiac remodeling after myocardial infarction. J Am Coll Cardiol. 2005;45:608–616. doi: 10.1016/j.jacc.2004.10.067. [DOI] [PubMed] [Google Scholar]
- 29.Clavell AL, Stingo AJ, Wei CM, et al. C-type natriuretic peptide: a selective cardiovascular peptide. Am J Physiol. 1993;264:R290–R295. doi: 10.1152/ajpregu.1993.264.2.R290. [DOI] [PubMed] [Google Scholar]
- 30.Lisy O, Burnett JC., Jr The design, synthesis and cardiorenal actions of a new chimeric natriuretic peptide CD-NP [abstract] J Am Coll Cardiol. 2003;41:312A. doi: 10.1016/j.jacc.2008.02.077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lee CY, Boerrigter G, Harty GJ, et al. Pharmacodynamic profile of a novel chimeric natriuretic peptide, CD-NP, as compared to C-type natriuretic peptide [abstract] Circulation. 2007;116 suppl II:II-550. [Google Scholar]
- 32.Lisy O, Huntley BK, McCormick DJ, et al. Design, synthesis and unique biological actions of CD-NP: a novel CNP-like chimeric natriuretic peptide [abstract] Circulation. 2006;114:II-440. [Google Scholar]
- 33.Lee CY, Sandberg SM, Chen HH, et al. Renal cyclic GMP stimulating actions of a novel chimeric natriuretic peptide CD-NP in isolated glomeruli: evidence for NPR-A activation [abstract] J Card Fail. 2008;14:S11. [Google Scholar]
- 34.Huntley BK, Burnett JJC. Anti-proliferative mechanisms of BNP in cultured human cardiac fibroblasts: role for the clearance receptor, p21 and cyclin D1 [abstract] J Card Fail. 2006;12:S43. [Google Scholar]
- 35.Lee CYW, Chen H, Lieu H, et al. A phase I clinical trial on a novel chimeric natriuretic peptide, CD-NP [abstract] J Am Coll Cardiol. 2008;51:A324. [Google Scholar]
- 36.Letts G, Loscalzo J. Frontiers in nephrology: targeting inflammation using novel nitric oxide donors. J Am Soc Nephrol. 2007;18:2863–2869. doi: 10.1681/ASN.2007030321. [DOI] [PubMed] [Google Scholar]
- 37.Mills RM, LeJemtel T, Horton D, et al. Sustained hemodynamic effects of an infusion of nesiritide (human b-type natriuretic peptide) in heart failure: a randomized, double-blind, placebo-controlled clinical trial. J Am Coll Cardiol. 1999;34:155–162. doi: 10.1016/s0735-1097(99)00184-9. [DOI] [PubMed] [Google Scholar]
- 38.Kitakaze M, Asakura M, Kim J, et al. Human atrial natriuretic peptide and nicorandil as adjuncts to reperfusion treatment for acute myocardial infarction (J-WIND): two randomised trials. Lancet. 2007;370:1483–1493. doi: 10.1016/S0140-6736(07)61634-1. [DOI] [PubMed] [Google Scholar]
- 39.Hillock RJ, Frampton C, Yandle T, et al. B-type natriuretic peptide infusions in acute myocardial infarction. Heart. 2008;94:617–622. doi: 10.1136/hrt.2006.110239. [DOI] [PubMed] [Google Scholar]
- 40.Kasama S, Furuya M, Toyama T, et al. Effects of intravenous atrial natriuretic peptide on cardiac sympathetic nerve activity and left ventricular remodeling in patients with first anterior acute myocardial infarction. J Am Coll Cardiol. 2007;49:667–674. doi: 10.1016/j.jacc.2006.09.048. [DOI] [PubMed] [Google Scholar]
- 41.Chen H, Schirger J, Lerman A, et al. Intravenous BNP at the time of acute anterior myocardial infarction in humans improves left ventricular remodeling [abstract] Circulation. 2005 [Google Scholar]
- 42.Park K, Itoh H, Yamahara K, et al. Therapeutic potential of atrial natriuretic peptide administration on peripheral arterial diseases. Endocrinology. 2008;149:483–491. doi: 10.1210/en.2007-1094. [DOI] [PubMed] [Google Scholar]
- 43.Chen HH, Redfield MM, Nordstrom LJ, et al. Subcutaneous administration of the cardiac hormone BNP in symptomatic human heart failure. J Card Fail. 2004;10:115–119. doi: 10.1016/j.cardfail.2003.08.011. [DOI] [PubMed] [Google Scholar]
- 44.Cataliotti A, Shirger J, Martin F, et al. Oral human brain natriuretic peptide activates cyclic guanosine 3′,5′-monophosphate and decreases mean arterial pressure. Circulation. 2005;112:836–840. doi: 10.1161/CIRCULATIONAHA.105.538520. [DOI] [PubMed] [Google Scholar]
- 45.Cataliotti A, James K, Chen HH, et al. Chronic actions of a novel oral B-type natriuretic peptide conjugate in normal dogs and acute actions in angiotensin IIYmediated hypertension. Circulation. 2008;118:1729–1736. doi: 10.1161/CIRCULATIONAHA.107.759241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sengenes C, Berlan M, De Glisezinski I, et al. Natriuretic peptides: a new lipolytic pathway in human adipocytes. FASEB J. 2000;14:1345–1351. [PubMed] [Google Scholar]
- 47.Meirhaeghe A, Sandu M, McCarthy M, et al. Association between the T-381C polymorphism of the brain natriuretic peptide gene and risk of type 2 diabetes in human populations. Hum Mol Genet. 2007;16:1343–1350. doi: 10.1093/hmg/ddm084. [DOI] [PubMed] [Google Scholar]
- 48.Vesely BA, McAfee Q, Gower WR, Jr, et al. Four peptides decrease the number of human pancreatic adenocarcinoma cells. Eur J Clin Invest. 2003;33:998–1005. doi: 10.1046/j.1365-2362.2003.01262.x. [DOI] [PubMed] [Google Scholar]
- 49.Vesely DL. Atrial natriuretic peptides: anticancer agents. J Investig Med. 2005;53:360–365. doi: 10.2310/6650.2005.53708. [DOI] [PubMed] [Google Scholar]
- 50.Vesely DL, Song S, Sanchez-Ramos J, et al. Four peptide hormones decrease the number of human breast adenocarcinoma cells. Eur J Clin Invest. 2005;35:60–69. doi: 10.1111/j.1365-2362.2005.01444.x. [DOI] [PubMed] [Google Scholar]
- 51.Cataliotti A, Chen HH, James KD, et al. Oral brain natriuretic peptide: a novel strategy for chronic protein therapy for cardiovascular disease. Trends Cardiovasc Med. 2007;17:10–14. doi: 10.1016/j.tcm.2006.10.001. [DOI] [PubMed] [Google Scholar]