Abstract
In the early hours of ischemic stroke, cerebral edema forms as Na, Cl, and water are secreted across the blood-brain barrier (BBB) and astrocytes swell. We have shown previously that ischemic factors, including hypoxia, aglycemia, and arginine vasopressin (AVP), stimulate BBB Na-K-Cl cotransporter (NKCC) and Na/H exchanger (NHE) activities and that inhibiting NKCC and/or NHE by intravenous bumetanide and/or HOE-642 reduces edema and infarct in a rat model of ischemic stroke. Estradiol also reduces edema and infarct in this model and abolishes ischemic factor stimulation of BBB NKCC and NHE. There is evidence that NKCC and NHE also participate in ischemia-induced swelling of astrocytes. However, little is known about estradiol effects on astrocyte cell volume. In this study, we evaluated the effects of AVP (100 nM), hypoxia (7.5% O2), aglycemia, hypoxia (2%)/aglycemia [oxygen glucose deprivation (OGD)], and estradiol (1–100 nM) on astrocyte cell volume using 3-O-methyl-d-[3H]glucose equilibration methods. We found that AVP, hypoxia, aglycemia, and OGD (30 min to 5 h) each significantly increased astrocyte cell volume, and that estradiol (30–180 min) abolished swelling induced by AVP or hypoxia, but not by aglycemia or OGD. Bumetanide and/or HOE-642 also abolished swelling induced by AVP but not aglycemia. Abundance of aquaporin-4, known to participate in ischemia-induced astrocyte swelling, was significantly reduced following 7-day but not 2- or 3-h estradiol exposures. Our findings suggest that hypoxia, aglycemia, and AVP each contribute to ischemia-induced astrocyte swelling, and that the edema-attenuating effects of estradiol include reduction of hypoxia- and AVP-induced astrocyte swelling and also reduction of aquaporin-4 abundance.
Keywords: astrocytes, cytotoxic edema, cerebral ischemia, aquaporin-4
edema formation is a major contributor to brain damage during ischemic stroke (24, 53). During the early hours of cerebral ischemia, edema forms by a process involving increased secretion of Na, Cl, and osmotically obliged water across the blood-brain barrier (BBB), from the blood into the brain (7–9, 25, 32, 56). At the same time, perivascular astrocytes rapidly swell as they take up ions and water entering the brain, forming cytotoxic edema (11, 31, 33). In the absence of reperfusion, the BBB begins to break down 3–4 h after the start of ischemia, allowing paracellular movement of solutes and water into the brain and development of vasogenic edema (31, 47, 56). Previous studies by our group have demonstrated that arginine vasopressin (AVP), hypoxia, and aglycemia all stimulate cerebral microvascular endothelial cell (CMEC) Na-K-Cl cotransport (NKCC) and Na/H exchange (NHE) activities (12, 21, 37, 48–50). Our studies have also shown that both NKCC and NHE reside predominantly in the luminal BBB membrane, and that inhibiting these transporters by intravenous administration of bumetanide or HOE-642, respectively, significantly attenuates edema and infarct in the rat permanent middle cerebral artery occlusion (MCAO) model of ischemic stroke (15, 16, 51). Furthermore, we have shown that CMEC exposed to moderate-to-severe hypoxia do not swell or exhibit significant increases in Na content until 3 or more hours after the onset of hypoxia, a finding consistent with increased NKCC and NHE activities contributing to BBB secretion of Na from blood into brain during the early hours of stroke rather than simple swelling of the endothelial cells (12, 21). However, CMEC swelling observed after 5-h exposure to hypoxia is abolished in the presence of bumetanide and HOE-642, suggesting that both NKCC and NHE participate in the gradual swelling of the cells that occurs with hypoxia (12). There is evidence that the rapid astrocyte swelling that occurs during cerebral ischemia also involves NKCC and NHE activities. Previous studies have shown that astrocyte NKCC activity is stimulated by oxygen glucose deprivation (OGD) (41), AVP (30), and also by high extracellular K+ concentration ([K+]o) (62), and that OGD (34) and hypoxia (10) stimulate astrocyte NHE activity. It has also been found that OGD, AVP, and high [K+]o can induce astrocyte swelling (17, 34, 39, 45).
Much evidence has been presented that estradiol is neuroprotective in stroke (19, 63, 65). Others and our laboratory have demonstrated that estradiol attenuates ischemia-induced cerebral edema (50, 57) and infarct (19, 50) following MCAO in the rat. Our laboratory has further shown that estradiol abolishes hypoxia- and AVP-induced stimulation of NKCC activity, reduces NKCC abundance (50), and also reduces AVP-stimulated NHE activity in BBB endothelial cells (37). Because estradiol can freely enter the brain, one would predict that it might also act on astrocyte NKCC and NHE, attenuating astrocyte swelling during ischemia. Little is known, however, about estradiol effects on astrocyte cell volume or astrocyte Na transporter activities.
A number of studies have provided evidence that the aquaporin-4 (AQP4) water channel, highly concentrated in perivascular astrocyte endfeet, also plays an essential role in swelling of astrocytes during ischemic stroke (1, 2, 4, 40), and that an increase in astrocyte AQP4 protein abundance is induced by MCAO (3, 52, 55). It has also been found that BBB disruption, as occurs after several hours of cerebral ischemia, causes an increase in astrocyte AQP4 mRNA and protein abundance in a manner that is attenuated by estradiol (64). This suggests the possibility that, like NKCC and NHE, AQP4 may be a target for the edema-reducing actions of estradiol.
The present study was conducted to determine whether astrocyte swelling induced by ischemic factors, including hypoxia, aglycemia, and AVP, is reduced by exposure of the cells to estradiol. We also evaluated the contributions of NKCC and NHE to astrocyte swelling induced by the ischemic factors. In addition, we evaluated the effects of estradiol on astrocyte AQP4 abundance. We report here that intracellular volume of cultured primary rat astrocytes is significantly increased within 30–60 min following exposure to AVP, hypoxia (7.5% O2), aglycemia, or combined hypoxia (2% O2)/aglycemia (OGD), and that estradiol (1–100 nM) significantly reduces AVP- and hypoxia-induced astrocyte swelling, but has no effect on aglycemia- or OGD-induced astrocyte swelling. We further report here that inhibition of NKCC and/or NHE in the astrocytes abolishes AVP- but not aglycemia-induced astrocyte swelling. Finally, we report that estradiol (100 nM, 7 days) significantly reduces AQP4 protein abundance.
MATERIALS AND METHODS
Astrocyte cell culture.
Astrocytes were isolated and cultured from the cortex of 1- to 2-day-old Sprague-Dawley rats using a modified version of the procedures previously described (46). This study followed the Animal Use and Care Guidelines issued by the National Institutes of Health using a protocol approved by the Animal Use and Care Committee at the University of California, Davis. Rats were decapitated, and the brains placed in ice-cold PBS supplemented with 1% antibiotic-antimycotic. The cerebral cortex was removed from each hemisphere of the brain, and the meninges were removed by microdissection. Cells from the cortex were isolated by passing the cortical hemisphere through an 80 μM Nitrex Nylon filter (Lab Pak, Depew, NY) and cultured in suspension in Dulbecco's modified Eagle's medium (DMEM) containing 10% fetal bovine serum (FBS) and 1% antibiotic-antimycotic agents. On day 9, the cultures were agitated to remove nonadherent oligodendrocytes. Adhering astrocytes were harvested by trypsinization and then grown in collagen-coated 24-well plates using DMEM containing 5 mM d-glucose, supplemented with 2 mM l-glutamine, 50 μg/ml gentamicin, and 10% FBS. Astrocyte cultures were incubated at 37°C with 5% CO2; growth medium was changed every other day. The noted experiments were performed using astrocytes that had been passaged two to four times.
Cell volume measurements.
Intracellular volume was determined by radioisotopic evaluation of intracellular water space using previously described methods (6, 12, 35). Confluent monolayers of astrocytes were placed in a hypoxia chamber (COY Laboratory Products, Grass Lake, MI) preset to 37°C, 5% CO2, and the desired oxygen level. The O2 levels were reduced by displacing air in the chamber with N2 (via O2 sensor/controller) and verified using a Corning or Oakton dissolved O2 meter (Corning, NY, and Vernon Hills, IL). Oxygen levels used ranged from 19% (for control, normoxia) down to 2% O2. Once in the hypoxia chamber, culture media was immediately replaced with medium containing DMEM, 5.5 mM d-glucose, 1 mM pyruvate, and 10 mM HEPES (DMEM HEPES) that was preequilibrated to the desired oxygen level. Experiments using glucose-free medium, contained DMEM with 10 mM HEPES (DMEM HEPES), but lacked both glucose and pyruvate. The osmolality of all assay media was 290 mosmol/kgH2O (as verified by osmometry). Astrocytes were exposed to the assay medium with the desired oxygen level for 30, 60, 90, 180, or 300 min. Ten minutes before the end of the exposure, 0.5 μCi/ml of [14C]sucrose (a marker for extracellular water space) or 3-O-methyl-d-[3H]glucose (a marker for total cellular water space, both intracellular and trapped extracellular) were added to the assay media. In some experiments, AVP, bumetanide, and/or HOE-642 were also present in the media, as indicated in the figure legends. In experiments evaluating the effects of estradiol, 0 (vehicle), 1, 10, or 100 nM 17β-estradiol were present in assay media. Assays were terminated by washing monolayers with ice-cold isotonic MgCl2. Cell-associated radioactivity was determined by liquid scintillation counting of 2% SDS cell extracts (Tri-Carb 2500 TR Liquid Scintillation Counter). Total protein in each well was measured using the Pierce BCA assay (Rockford, IL). Cell volume was calculated as total water space volume minus extracellular water space volume (expressed as μl/mg of protein). In initial experiments conducted to optimize these methods, cells were exposed to [14C]sucrose or 3-O-methyl-d-[3H]glucose over a time course of 1–30 min. It was determined that radioisotopic equilibrium was reached by 10 min (data not shown).
Gel electrophoresis and Western blot analysis.
Astrocyte cultures were exposed to 17β-estradiol (1, 10, or 100 nM) or vehicle and AVP, hypoxia (7.5% O2), or aglycemia acutely (2 or 3 h) or chronically (7 days). Cells were washed two times with ice-cold PBS containing protease inhibitors and then lysed, and protein was extracted with 1% SDS containing protease inhibitors. Total protein content of each lysate preparation was analyzed in triplicate using the Pierce BCA assay (Rockford, IL). Lysate samples were denatured in SDS reducing buffer containing dithiothreitol (Invitrogen NuPage, Carlsbad, CA) and heated to 70°C for 10 min and then used immediately for gel electrophoresis. Proteins samples were electrophoretically separated on 12% Tris-glycine gels (Lonza PAGEr Gold Precast, Walkersville, MD; Invitrogen XCell Sure Lock Mini-Cell, Carlsbad, CA), and the resolved proteins were electrophoretically transferred to polyvinylidene fluoride membranes using an Invitrogen XCell Blot Module. The polyvinylidene fluoride membranes were blocked with 7.5% milk solution and incubated with anti-AQP4, anti-NKCC1, or anti-NHE antibodies (1:1,000 dilutions). The membranes were washed three times and incubated with horseradish peroxidase-conjugated goat anti-rabbit IgG. The blots were washed, and bound secondary antibody was visualized using an enhanced chemiluminescence assay (VWR, West Chester, PA). Images of protein bands were captured using Fuji Film Imager/LAS 4000 (Stamford, CT). Protein abundance was determined using ImageQuant software (Molecular Dynamics, Sunnyvale, CA). For Western blot assays, β-actin was used as a loading control.
Materials.
DMEM, l-glutamine, and trypsin-EDTA were purchased from Invitrogen (Carlsbad, CA), and gentamicin was obtained from A.G. Scientific (San Diego, CA). Defined FBS was obtained from Hyclone (Logan, UT). 3-O-methyl-d-[3H]glucose was purchased from GE Healthcare (Piscataway, NJ), [14C]sucrose from Perkin Elmer (Waltham, MA), and 86Rb from Perkin Elmer (Welles, MA). HOE-642 was obtained from Aventis (Bridgewater, NJ). Bumetanide, methyl-d-glucose, [Arg8]-vasopressin, and 17β-estradiol were purchased from Sigma (St. Louis, MO). NKCC1 antibody (T4, rabbit polyclonal) was from University of Iowa Development Studies Hybridoma Bank (Iowa City, IA). NHE1 (4E9, rabbit polyclonal) and AQP4 (rabbit polyclonal) antibodies were purchased from Millipore (Billerica, MA). Goat anti-rabbit horseradish peroxidase-conjugated antibody was purchased from Invitrogen (Carlsbad, CA).
Statistical analysis.
Data are presented as means ± SE. In each experiment, three or four replicates were used for each condition. The sample sizes (n values) noted represent the number of separate experiments conducted. Data were analyzed for significance using paired Student’s t-test or analysis of variance as indicated. Differences with P values < 0.05 were considered statistically significant.
RESULTS
Hypoxia, aglycemia, and AVP all increase astrocyte intracellular volume.
Previous in vitro studies have provided evidence that AVP and OGD can induce astrocyte swelling (17, 34, 39, 45) and that astrocytes in vivo rapidly swell during cerebral ischemia (36, 43) (15 and 90 min, respectively). However, little is known about the contributions of individual ischemic factors, in particular, hypoxia, aglycemia, and AVP, to astrocyte swelling during ischemia. To determine the relative effects of these ischemic factors on astrocyte intracellular volume, we exposed cultured cortical primary rat astrocytes to control medium (glucose-containing normoxic DMEM HEPES, 19% O2) or to AVP (100 nM, in normoxic DMEM HEPES) or aglycemia (normoxic DMEM HEPES lacking glucose and pyruvate) for 30 min and assessed intracellular volume by 3-O-methyl-d-[3H]glucose equilibration, as described in materials and methods. We also tested the effects of exposing the cells to hypoxia (DMEM HEPES, 7.5% O2) for 60 min. As shown in Table 1, AVP, aglycemia, and 7.5% O2 caused significant increases in astrocyte intracellular volume of 12, 26, and 28%, respectively, after these acute exposures. The increases in volume found with 60-min exposure to 7.5% O2, a moderate level of hypoxia as found in the penumbra of ischemic brain, or to 30 min of aglycemia, were similar to that observed with 30-min exposure to the more severe condition of OGD (2% O2 in glucose-, pyruvate-free DMEM HEPES), as found in the ischemic core. Longer exposures to 7.5% O2 (5 h) or OGD (90 min and 3 h) did not result in further increases in astrocyte cell volume (as shown in Figs. 2 and 3).
Table 1.
Effect of ischemic factors on astrocyte intracellular volume
| Condition | Intracellular Volume, μl/mg protein |
|---|---|
| Control | 3.70 ± 0.17 |
| AVP | 4.14 ± 0.24* |
| Hypoxia | 4.75 ± 0.72* |
| Aglycemia | 4.66 ± 0.12* |
| OGD | 4.73 ± 0.26* |
Values are means ± SE of at least 4 experiments. Cortical rat astrocytes grown on collagen-coated 24-well plates were exposed to control conditions (glucose-containing DMEM HEPES, normoxia 19% O2) or various ischemic conditions in a 37°C hypoxia chamber. Cells were exposed to either hypoxia (7.5% O2) for 60 min, or aglycemia (glucose-free DMEM HEPES), 100 nM arginine-vasopressin (AVP), or oxygen-glucose deprivation (OGD; 2% hypoxia and glucose free DMEM HEPES) for 30 min. Intracellular volume was assessed using 3-O-methyl-d-[3H]glucose equilibration, as described in materials and methods.
Significantly different from corresponding control condition, P < 0.05 by paired Student’s t-test.
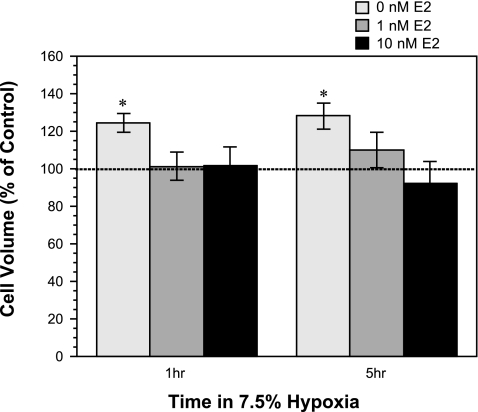
Fig. 2.
Effect of acute estradiol (E2) treatment on hypoxia-induced astrocyte swelling. Cultured rat astrocytes were exposed to 19% O2 for 1 h (control) or 7.5% O2 for 1 or 5 h in the presence of 0 (vehicle), 1, or 10 nM estradiol. Intracellular volume was assessed by 3-O-methyl-d-[3H]glucose equilibration, as described in materials and methods. Dashed line represents cell volume observed with normoxic conditions and 0 nM estradiol. Values are means ± SE of 6 experiments. *Significantly different from control by ANOVA, P < 0.05.
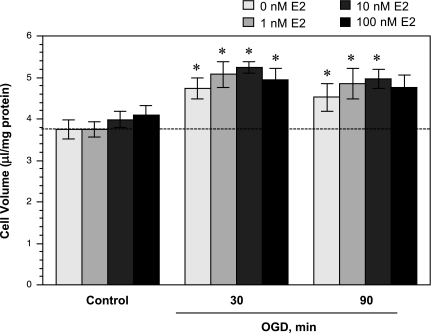
Fig. 3.
Effect of acute estradiol treatment on oxygen glucose deprivation (OGD)-induced astrocyte swelling. Cultured rat astrocytes were exposed to normoxia (control, with or without estradiol) for 30 min or to 2% O2 in glucose-free DMEM HEPES (OGD, with or without estradiol) for 30, 90, or 180 min. Cell volume was assessed as described in materials and methods. Values are means ± SE of 6 experiments. Dashed line represents cell volume observed with normoxic conditions and 0 nM estradiol. *Significantly different from matched control by paired Student’s t-test, P < 0.05.
Estradiol abolishes AVP- and hypoxia-induced but not aglycemia-induced astrocyte swelling.
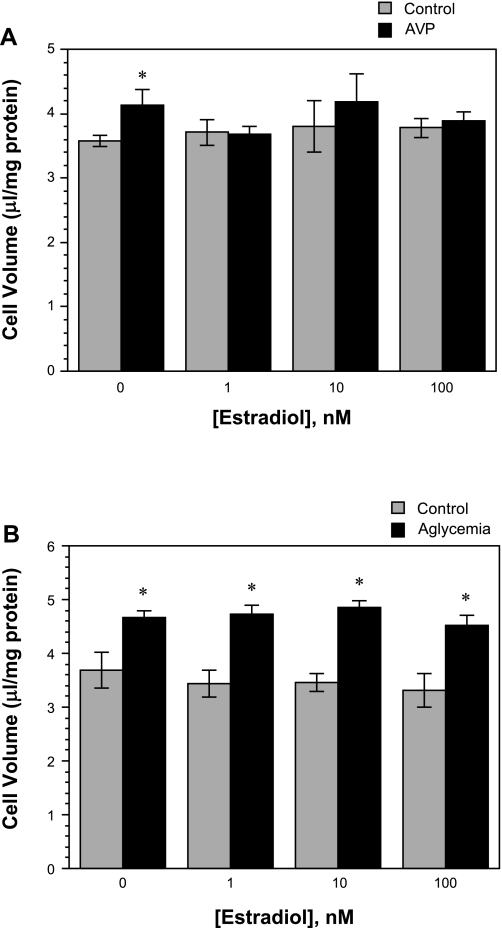
Estradiol is known to be neuroprotective (19, 27), and our laboratory's previous studies have demonstrated that estradiol reduces edema formation and infarct volume in an in vivo rat MCAO model of ischemic stroke (50). Estradiol also abolishes ischemia-induced stimulation of the BBB NKCC in vitro (50). However, the effect of estradiol on astrocyte swelling during ischemia has not been investigated. Thus, in the present study, we evaluated the effects of AVP, hypoxia, aglycemia, and also OGD on intracellular volume of astrocytes treated with 17β-estradiol. We found that the increase in astrocyte cell volume observed following exposure to AVP (100 nM, 30 min) was abolished in cells also exposed to estradiol (1, 10, or 100 nM, 30 min) (Fig. 1A). In contrast, the aglycemia-induced increase in astrocyte cell volume was not significantly reduced by exposure to estradiol, whether at 1, 10, or 100 nM (30 min) (Fig. 1B). When astrocytes were exposed to 7.5% O2 for either 60 min or 5 h, we found significant increases in intracellular volume that were abolished in the presence of 1 or 10 nM estradiol (Fig. 2). In these experiments, we also examined the effects of estradiol on astrocytes exposed to OGD. Figure 3 shows that estradiol, whether at 1, 10, or 100 nM, did not reduce astrocyte swelling occurring with a 30-min exposure to OGD. Similarly, estradiol was without effect on astrocyte swelling following 90-min exposure to OGD. Here again, significant volume increases were observed with OGD, whether estradiol was present at 0, 1, or 10 nM. While the increase in volume of cells exposed to OGD plus 100 nM estradiol for 90 min compared with control did not reach significance, neither was there a significant decrease in volume compared with 0 nM estradiol-treated cells. Astrocyte swelling induced by OGD (30, 60, or 90 min) was also not reduced by a chronic (7 day) exposure to estradiol (1–100 nM, n = 6, data not shown). While estradiol reduced astrocyte swelling observed in the presence of AVP and hypoxia, exposure of the cells to estradiol (1–100 nM) under control conditions (normoxic, glucose-containing medium) did not alter intracellular volume, whether the exposure time was 30 min (as shown in Fig. 1, A and B); 1.5, 3, or 5 h (n = 4); or 1, 3, or 7 days (n = 3) (data not shown).
Fig. 1.
Effect of acute estradiol treatment on arginine vasopressin (AVP)- and aglycemia-induced astrocyte swelling. A: cultured cortical rat astrocytes were exposed to 100 nM AVP or vehicle (control) in the presence of various concentrations of estradiol ([estradiol]) for 30 min under normoxic (19% O2) conditions. Cell volume was assessed using 3-O-methyl-d-[3H]glucose equilibration, as described in materials and methods. B: rat astrocytes were exposed to glucose-containing (control) or glucose-free (aglycemia) DMEM HEPES in the presence of various concentrations of estradiol for 30 min under normoxic (19% O2) conditions. Cell volume was assessed as described in materials and methods. Values are means ± SE of 4 and 6 experiments for A and B, respectively. *Significantly different from corresponding control condition by paired Student’s t-test, P < 0.05.
Bumetanide and HOE-642 attenuate astrocyte swelling induced by AVP but not by aglycemia.
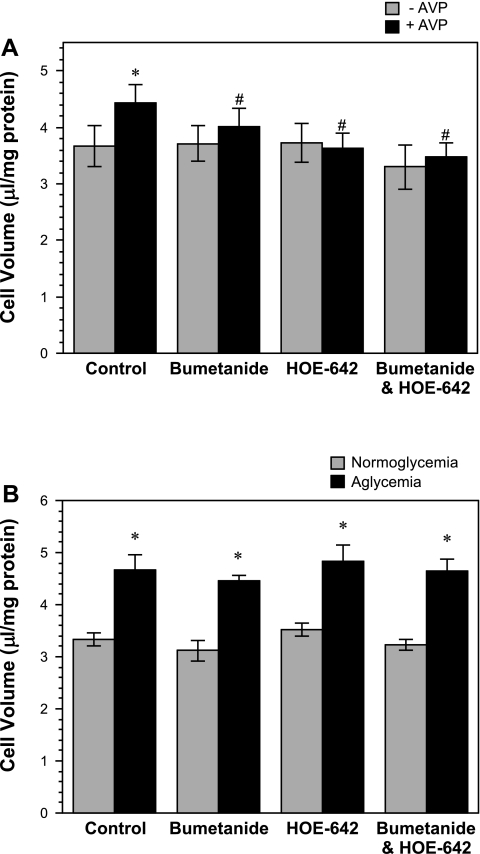
Previous studies have provided evidence that astrocyte NKCC and NHE activities can be stimulated by ischemic factors (10, 30, 34, 41). To investigate whether NKCC and/or NHE contribute to ischemic factor-induced astrocyte swelling, in the present study, we evaluated the effects of AVP and aglycemia on astrocyte intracellular volume in the presence of bumetanide and/or HOE-642, inhibitors of NKCC and NHE, respectively. As shown in Fig. 4A, we found that swelling of astrocytes induced by AVP (100 nM, 30 min) was abolished in the presence of bumetanide (10 μM) and/or HOE-642 (20 μM). In contrast, astrocyte swelling induced by aglycemia (30 min) was not affected by either bumetanide or HOE-642, alone or in combination (Fig. 4B).
Fig. 4.
Effects of bumetanide and HOE-642 on AVP- and aglycemia-induced astrocyte swelling. A: cultured rat astrocytes were exposed to 100 nM AVP (+AVP) or vehicle (−AVP) in the presence or absence of bumetanide (10 μM) and/or HOE-642 (20 μM) for 30 min under normoxic (19% O2) conditions. Cell volume was assessed as described in materials and methods. Values are means ± SE of 5 experiments. Significantly different from control *without AVP and #with 100 nM AVP: P < 0.05 by paired Student t-test. B: cultured rat astrocytes were exposed to glucose-containing (normoglycemia) or glucose-free DMEM HEPES (aglycemia) in the presence or absence of bumetanide (10 μM) and/or HOE-642 (20 μM) for 30 min under normoxic (19% O2) conditions. Cell volume was assessed as described in materials and methods. Values are means ± SE of 5 experiments. *Significantly different from corresponding normoglycemia condition, P < 0.05 by paired Student’s t-test.
Estradiol reduces AQP4 but not NKCC or NHE1 abundance in astrocytes.
A number of investigations have provided strong evidence for a role of perivascular astrocyte AQP4 in ischemia-induced cerebral edema formation (1–4, 52, 55). This, together with the observation that estradiol attenuates edema formation following permanent or transient MCAO (50, 57), suggests the possibility that the effects of estradiol on reducing astrocyte swelling during ischemia are mediated, at least in part, by changes in AQP4 abundance and/or permeability. In previous studies, our laboratory has shown that estradiol also reduces abundance of NKCC and NHE in BBB endothelial cells (14, 50). Thus it is possible that estradiol effects on astrocyte swelling could involve changes in astrocyte NKCC and/or NHE abundance, as well as changes in AQP4. To evaluate these possibilities, we began by examining the effects of acute and/or or chronic estradiol exposures on AQP4 abundance in the astrocytes. Rat cortical astrocytes were exposed to 0, 1, 10, or 100 nM estradiol for 2 or 3 h (acute exposure) or for 7 days (chronic exposure). Cell lysates were then examined for AQP4 abundance by Western blot and quantitative densitometric analysis, as described in materials and methods. When the astrocytes were exposed to estradiol for either 2 or 3 h, no statistically significant changes in AQP4 abundance relative to control (vehicle only, 0 nM estradiol) were detected for any of the estradiol concentrations tested (Fig. 5, A and B). Astrocyte AQP4 abundance was also not changed by estradiol in cells also exposed to AVP or aglycemia (n = 8 for both AVP and aglycemia, data not shown). However, exposing the astrocytes to estradiol (100 nM) for 7 days produced a significant reduction in abundance of AQP4 protein (Fig. 5, C and D). The estradiol-induced reduction of astrocyte AQP4 protein was observed only for 100 nM estradiol, with 1 and 10 nM estradiol 7-day exposures having no significant effect. In these studies, we also evaluated the effects of estradiol on abundance of NKCC and NHE in the astrocytes. For NHE experiments, we focused on NHE1, the isoform form previously reported to be present in astrocytes (34) and also the isoform our laboratory found previously to be reduced in BBB endothelial cells exposed to estradiol (14, 50). Here, we found that neither abundance of NKCC nor NHE1 was altered in the cells following 3-h exposure to hypoxia (7.5% O2), aglycemia, or AVP (100 nM) (n = 7–9 for NKCC and 7–8 for NHE1, data not shown). Furthermore, estradiol (100 nM, 3 h) was without effect on either NKCC or NHE1 abundance in astrocytes also exposed to 3 h of hypoxia, aglycemia, or AVP (n = 7–9 for NKCC and 7–8 for NHE1, data not shown).
Fig. 5.
Western blot analysis of aquaporin-4 (AQP4) protein expression in cells exposed to acute or chronic estradiol. A: cultured astrocytes were exposed to media containing 0 (vehicle), 1, 10, or 100 nM estradiol for 2 or 3 h. At the end of the incubation period, cells were lysed and prepared for gel electrophoresis and Western blotting, as described in materials and methods. Blots were probed with AQP4 antibody (Chemicon), and bands were detected by enhanced chemiluminescence. Data shown are representative Western blots for cells exposed to vehicle (control), 1, 10, or 100 nM estradiol for 2 or 3 h. B: Western blots generated as described above were subjected to densitometric analysis. Values are means ± SE of 3 experiments for each estradiol treatment. C: rat astrocytes were exposed to media containing 0 (vehicle), 1, 10, or 100 nM estradiol for 7 days, and then lysates were subjected to Western blotting, as described in materials and methods. Blots were probed with AQP4 antibody (Chemicon), and bands were detected by enhanced chemiluminescence. In the representative Western blot shown, double bands at ∼34 kDa, which represent the two-splice variants of AQP4, M1, and M2, can be seen (58). D: Western blots generated as described above were subjected to densitometric analysis. Values are means ± SE of 5 experiments for each estradiol treatment. *Significantly different from control without estradiol by ANOVA, P < 0.05.
DISCUSSION
Previous studies have shown that estradiol is neuroprotective in ischemic stroke (19, 20, 23, 50, 59, 63, 65, 68, 69), reducing both cerebral edema (50, 57) and infarct (19, 20, 50, 65). Formation of ischemia-induced cerebral edema results, in part, from BBB endothelial cell transport of Na, Cl, and water from blood into brain (8, 9, 32, 51, 56) and swelling of astrocytes as they take up ions and water (11, 31, 33). While we have demonstrated previously that estradiol reduces ischemic factor stimulation of BBB ion transporters (37, 50), little is known about estradiol effects on astrocyte swelling during ischemia. Our present study demonstrates that estradiol reduces astrocyte swelling caused by hypoxia and AVP, but not that caused by aglycemia or OGD. Furthermore, we show here that AVP- but not aglycemia-induced swelling of astrocytes is attenuated by bumetanide and HOE-642, inhibitors NKCC and NHE, respectively. Finally, we present evidence that estradiol reduces AQP4 water channel abundance in astrocytes following a 7-day exposure. These studies support the hypothesis that the neuroprotective effects of estradiol include attenuation of edema-forming processes, in regions of mild to moderate hypoxic insult (penumbral region), occurring at both the BBB and astrocytes.
A number of studies have shown that astrocytes swell in the presence of cerebral ischemia. While there are many factors known to contribute to astrocyte swelling, including elevated brain interstitial [K] and glutamate (42, 54), the extent to which hypoxia, aglycemia, and AVP, three prominent factors present during ischemic stroke, each contribute astrocyte swelling has not been well understood. The results of our studies reveal that significant astrocyte swelling occurs within 30 min of exposing the cells to either AVP or aglycemia and within 60 min of exposure to moderate hypoxia (7.5% O2). The combination of severe hypoxia (2% O2) plus aglycemia (OGD) also causes significant swelling of the cells within 30 min, but does not produce greater swelling than any of the individual factors alone. Thus it appears that moderate hypoxia and AVP, both factors present in the ischemic penumbra (5), are by themselves sufficient to induce swelling of astrocytes, with the magnitude of swelling not significantly different than that found with aglycemia and severe hypoxia, conditions present in the ischemic core. Our finding that OGD increases intracellular volume of rat astrocytes is consistent with previous reports that OGD also induces swelling in mouse astrocytes (34, 41). It should be noted that those studies assessed cell volume indirectly, either by cross-sectional area using morphometric differential interference contrast microscopy or by changes in calcein fluorescence intensity. Our present study also reveals that AVP, released from extrahypothalamic neuronal processes during ischemia (18, 29, 38, 60), increases astrocyte volume within 30 min to a similar degree as that induced by hypoxia, aglycemia, or OGD. Our finding is consistent with a previous report that astrocytes swell in response to AVP, although in that study astrocytes were exposed to a significantly higher dose of AVP (1 μM), and the 3-O-methyl-d-[3H]glucose space was not corrected for trapped extracellular space (39) as in the present study. Previous studies have also demonstrated that an AVP V1 receptor antagonist reduces edema and infarct following MCAO in mice (44, 66). Collectively, these findings underscore the importance of cerebral AVP as a significant contributor to edema formation during ischemia.
Our studies demonstrate that astrocyte swelling observed following exposure to moderate hypoxia is abolished in the presence of estradiol at doses of 1 nM and greater, as is swelling induced by AVP. This effect occurs when the cells are exposed to estradiol acutely, i.e., concurrently with 30 or 60 min hypoxia/AVP exposure, suggesting that the underlying signaling pathways involved are nongenomic. Our studies also show that the same acute exposures to estradiol (1–100 nM) do not significantly reduce aglycemia- or OGD-induced astrocyte swelling. This suggests that acute estradiol is more likely to be effective in attenuating swelling in the ischemic penumbra, but not the ischemic core where severe hypoxia and aglycemia are present. Our findings also suggest that mechanisms underlying hypoxia and AVP-induced swelling differ from mechanisms responsible for aglycemia-induced astrocyte swelling. The reason for lack of estradiol effect on astrocyte swelling in the presence of aglycemia and OGD is not clear. However, it has been shown previously that, under aglycemic conditions, glycogen stores are mobilized to support neuronal activity (13), and that the resulting increase in lactate causes astroglial swelling (28, 61). This would suggest that estradiol is without effect on swelling caused by accumulation of lactate in the cells. Future studies are needed to identify mechanisms responsible for aglycemia-induced astrocyte swelling, including those using 1,4-dideoxy-1,4-imino-d-arabinitol, recently shown to be an effective inhibitor of glycogen phosphorylase activity in brain (67).
Our laboratory's previous studies have shown that ischemic factors stimulate NKCC and NHE activities in cultured bovine CMECs in a manner that is abolished by estradiol (37, 50). While activity of these CMEC Na transporters is increased within minutes of exposure to hypoxia, aglycemia, and/or AVP, the endothelial cells swell only in response to hypoxia (moderate or severe), not aglycemia or AVP, and, furthermore, significant swelling does not occur until 3–5 h. This is consistent with both NKCC and NHE participating in secretion of Na, Cl, and water across the BBB from blood into brain during ischemic stroke. However, we have also found that the CMEC swelling that occurs after 5 h of hypoxia is attenuated by bumetanide and HOE-642, suggesting that both Na transporters also participate in the eventual swelling of the BBB endothelial cells (12). In the present study, we report that bumetanide and HOE-642, alone and in combination, also attenuate the rapid swelling of astrocytes induced by AVP, but not swelling induced by aglycemia. Thus, like estradiol, the NKCC and NHE inhibitors appear to be effective in reducing swelling induced by conditions found in the ischemic penumbra, but not the core. This suggests that both NKCC and NHE of the astrocytes are targets for reducing astrocyte swelling in the ischemic penumbra. In previous studies of mouse astrocytes, it was reported that bumetanide or genetic ablation of NKCC reduced OGD-induced astrocyte swelling (41), as did HOE-642 or genetic ablation of the NHE1 isoform of NHE (34). It should be noted that swelling was assessed in the mouse studies by morphometry or calcein fluorescence methods vs. the radioisotopic equilibration determination of intracellular volume used in the present study. Whether bumetanide and HOE-642 might attenuate swelling induced by aglycemia in combination with severe hypoxia (OGD) or attenuate swelling after initiation of ischemic insult in our rat astrocytes remains to be determined. In previous studies, our laboratory has shown that NKCC activity of cultured rat primary astrocytes is reduced by a 5-min exposure to 10 nM estradiol (50). Whether estradiol attenuates ischemic factor-stimulated astrocyte NKCC and NHE activities, as it does in CMEC, will require further investigation. Furthermore, determining whether bumetanide and HOE-642 attenuation of ischemic factor-induced astrocyte swelling is solely due to inhibition of Na transporter activity or might also include a decrease in NKCC or NHE1 transporter protein abundance should also be addressed in future studies.
There is good evidence that the rapid movement of water and electrolytes underlying formation of edema during cerebral ischemia involves not only BBB endothelial cell and astrocyte ion transporters, but also AQP4 water channels present in perivascular astrocyte endfeet (2–4, 40, 52, 55). In the present study, we have presented evidence that neuroprotection observed following chronic exposure to estradiol may include attenuation of astrocyte swelling during ischemia by reduction of AQP4 abundance in the astrocytes. While 7-day exposure to 100 nM estradiol decreases astrocyte AQP4 abundance, our studies show that acute estradiol exposure (2–3 h) does not appear to alter AQP4 abundance, regardless of dose (1–100 nM). These findings are consistent with chronic, but not acute, estradiol treatment-induced attenuation of edema formation during ischemia occurring, in part, by reduced astrocyte endfoot water permeability. Further investigation will be needed to fully evaluate the time course and dose effects of estradiol on astrocyte AQP4 abundance. Our previous studies demonstrated that both chronic (7-day) and acute (30 min) estradiol treatments significantly reduce edema and infarct in the rat MCAO model of stroke. This, together with the present findings, suggests that the edema-reducing effects of acute estradiol treatment do not involve changes in astrocyte AQP4 abundance. It should be noted that our findings do not rule out the possibility of acute estradiol exposure causing an in vivo AQP4 redistribution away from the endfeet into the astrocyte cell body, thus decreasing local water permeability at the BBB-astrocyte endfoot interface without decreasing total AQP4 abundance in the astrocytes, as has been demonstrated previously to occur in the ischemic core of mice 24 h following ischemia-reperfusion (22). Other studies have demonstrated erythropoietin, a known neuroprotectant, reduces glutamate-induced increases in AQP4 permeability (26). This suggests the additional possibility that acute estradiol could alter AQP4 permeability in the astrocytes, decreasing the rate of water uptake into the astrocytes, thereby reducing edema formation.
In summary, we demonstrate, for the first time, that acute treatments of estradiol effectively reduce astrocyte swelling induced by moderate hypoxia and by AVP, factors present in the ischemic penumbra, whereas they are without effect when aglycemia or OGD are present, conditions associated with the ischemic core. We also demonstrate that chronic treatments of estradiol reduce astrocyte AQP4 abundance. These findings suggest that estradiol, which readily penetrates into the brain and can, therefore, target astrocytes as well as BBB endothelial cells, deserves further attention as a therapeutic approach to reduce cerebral edema in the ischemic penumbra.
GRANTS
This work was supported by National Institutes of Health (NIH) Grants NINDS NS039953 (M. E. O'Donnell) and P01 AG17164 (P. M. Wise). This investigation was conducted, in part, in a facility constructed with support from Research Facilities Improvement Program Grant Number C06 RR17348-01 from the National Center for Research Resources, NIH.
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the author(s).
REFERENCES
- 1. Amiry-Moghaddam M, Frydenlund DS, Ottersen OP. Anchoring of aquaporin-4 in brain: molecular mechanisms and implications for the physiology and pathophysiology of water transport. Neuroscience 129: 999–1010, 2004 [DOI] [PubMed] [Google Scholar]
- 2. Amiry-Moghaddam M, Xue R, Haug FM, Neely JD, Bhardwaj A, Agre P, Adams ME, Froehner SC, Mori S, Ottersen OP. Alpha-syntrophin deletion removes the perivascular but not endothelial pool of aquaporin-4 at the blood-brain barrier and delays the development of brain edema in an experimental model of acute hyponatremia. FASEB J 18: 542–544, 2004 [DOI] [PubMed] [Google Scholar]
- 3. Badaut J, Ashwal S, Tone B, Regli L, Tian HR, Obenaus A. Temporal and regional evolution of aquaporin-4 expression and magnetic resonance imaging in a rat pup model of neonatal stroke. Pediatr Res 62: 248–254, 2007 [DOI] [PubMed] [Google Scholar]
- 4. Badaut J, Lasbennes F, Magistretti PJ, Regli L. Aquaporins in brain: distribution, physiology, and pathophysiology. J Cereb Blood Flow Metab 22: 367–378, 2002 [DOI] [PubMed] [Google Scholar]
- 5. Baringa M. Finding new drugs to treat stroke. Science 272: 646–666, 1996 [PubMed] [Google Scholar]
- 6. Bender AS, Schousboe A, Reichelt W, Norenberg MD. Ionic mechanisms in glutamate-induced astrocyte swelling: role of K+ influx. J Neurosci Res 52: 307–321, 1998 [DOI] [PubMed] [Google Scholar]
- 7. Betz AL. Alterations in cerebral endothelial cell function in ischemia. Adv Neurol 71: 301–313, 1996 [PubMed] [Google Scholar]
- 8. Betz AL, Iannotti F, Hoff JT. Brain edema: a classification based on blood-brain barrier integrity. Cerebrovasc Brain Metab Rev 1: 133–154, 1989 [PubMed] [Google Scholar]
- 9. Betz AL, Keep RF, Beer ME, Ren XD. Blood-brain barrier permeability and brain concentration of sodium, potassium, and chloride during focal ischemia. J Cereb Blood Flow Metab 14: 29–37, 1994 [DOI] [PubMed] [Google Scholar]
- 10. Bevensee MO, Boron WF. Effects of acute hypoxia on intracellular-pH regulation in astrocytes cultured from rat hippocampus. Brain Res 1193: 143–152, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Bourke RS, Kimelberg HK, Nelson LR, Barron KD, Auen EL, Popp AJ, Waldman JB. Biology of glial swelling in experimental brain edema. Adv Neurol 28: 99–109, 1980 [PubMed] [Google Scholar]
- 12. Brillault J, Lam TI, Rutkowsky JM, Foroutan S, O'Donnell ME. Hypoxia effects on cell volume and ion uptake of cerebral microvascular endothelial cells. Am J Physiol Cell Physiol 294: C88–C96, 2008 [DOI] [PubMed] [Google Scholar]
- 13. Brown AM, Ransom BR. Astrocyte glycogen and brain energy metabolism. Glia 55: 1263–1271, 2007 [DOI] [PubMed] [Google Scholar]
- 14. Chang E, O'Donnell ME, Barakat AI. Shear stress and 17beta-estradiol modulate cerebral microvascular endothelial Na-K-Cl cotransporter and Na/H exchanger protein levels. Am J Physiol Cell Physiol 294: C363–C371, 2008 [DOI] [PubMed] [Google Scholar]
- 15. Chen YJ, Anderson SE, O'Donnell ME. Bumetanide and HOE642 administered after initiation of middle cerebral artery occlusion effectively reduce rat brain Na uptake and infarct (Abstract). FASEB J 23: A614.–613., 2009 [Google Scholar]
- 16. Chen YJ, Lam TI, Anderson SE, Walton JH, O'Donnell ME. Blood-brain barrier Na-K-Cl cotransporter and Na/H exchanger: therapeutic targets for ischemia-induced brain Na uptake and edema formation (Abstract). J Cereb Blood Flow Metab 29: S489, 2009 [Google Scholar]
- 17. Del Bigio MR, Fedoroff S, Qualtiere LF. Morphology of astroglia in colony cultures following transient exposure to potassium ion, hypoosmolarity and vasopressin. J Neurocytol 21: 7–18, 1992 [DOI] [PubMed] [Google Scholar]
- 18. Dóczi T. Volume regulation of the brain tissue–a survey. Acta Neurochir (Wien) 121: 1–8, 1992 [DOI] [PubMed] [Google Scholar]
- 19. Dubal DB, Kashon ML, Pettigrew LC, Ren JM, Finklestein SP, Rau SW, Wise PM. Estradiol protects against ischemic injury. J Cereb Blood Flow Metab 18: 1253–1258, 1998 [DOI] [PubMed] [Google Scholar]
- 20. Dubal DB, Wise PM. Neuroprotective effects of estradiol in middle-aged female rats. Endocrinology 142: 43–48, 2001 [DOI] [PubMed] [Google Scholar]
- 21. Foroutan S, Brillault J, Forbush B, O'Donnell ME. Moderate-to-severe ischemic conditions increase activity and phosphorylation of the cerebral microvascular endothelial cell Na+-K+-Cl− cotransporter. Am J Physiol Cell Physiol 289: C1492–C1501, 2005 [DOI] [PubMed] [Google Scholar]
- 22. Frydenlund DS, Bhardwaj A, Otsuka T, Mylonakou MN, Yasumura T, Davidson KG, Zeynalov E, Skare O, Laake P, Haug FM, Rash JE, Agre P, Ottersen OP, Amiry-Moghaddam M. Temporary loss of perivascular aquaporin-4 in neocortex after transient middle cerebral artery occlusion in mice. Proc Natl Acad Sci U S A 103: 13532–13536, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Garcia-Segura LM, Azcoitia I, DonCarlos LL. Neuroprotection by estradiol. Prog Neurobiol 63: 29–60, 2001 [DOI] [PubMed] [Google Scholar]
- 24. Gartshore G, Patterson J, Macrae IM. Influence of ischemia and reperfusion on the course of brain tissue swelling and blood-brain barrier permeability in a rodent model of transient focal cerebral ischemia. Exp Neurol 147: 353–360, 1997 [DOI] [PubMed] [Google Scholar]
- 25. Gotoh O, Asano T, Koide T, Takakura K. Ischemic brain edema following occlusion of the middle cerebral artery in the rat. I. The time course of the brain water, sodium and potassium contents and blood-brain barrier permeability to 125I-albumin. Stroke 16: 101–109, 1985 [DOI] [PubMed] [Google Scholar]
- 26. Gunnarson E, Song Y, Kowalewski JM, Brismar H, Brines M, Cerami A, Andersson U, Zelenina M, Aperia A. Erythropoietin modulation of astrocyte water permeability as a component of neuroprotection. Proc Natl Acad Sci U S A 106: 1602–1607, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Hurn PD, Macrae IM. Estrogen as a neuroprotectant in stroke. J Cereb Blood Flow Metab 20: 631–652, 2000 [DOI] [PubMed] [Google Scholar]
- 28. Jakubovicz DE, Klip A. Lactic acid-induced swelling in C6 glial cells via Na+/H+ exchange. Brain Res 485: 215–224, 1989 [DOI] [PubMed] [Google Scholar]
- 29. Jójárt I, Joo F, Siklos L, Laszlo FA. Immunoelectronhistochemical evidence for innervation of brain microvessels by vasopressin-immunoreactive neurons in the rat. Neurosci Lett 51: 259–264, 1984 [DOI] [PubMed] [Google Scholar]
- 30. Katay L, Latzkovits L, Fonagy A, Janka Z, Lajtha A. Effects of arginine vasopressin and atriopeptin on chloride uptake in cultured astroglia. Neurochem Res 23: 831–836, 1998 [DOI] [PubMed] [Google Scholar]
- 31. Kimelberg HK. Astrocytic swelling in cerebral ischemia as a possible cause of injury and target for therapy. Glia 50: 389–397, 2005 [DOI] [PubMed] [Google Scholar]
- 32. Kimelberg HK. Current concepts of brain edema. Review of laboratory investigations. J Neurosurg 83: 1051–1059, 1995 [DOI] [PubMed] [Google Scholar]
- 33. Kimelberg HK, Barron KD, Bourke RS, Nelson LR, Cragoe EJ. Brain anti-cytoxic edema agents. Prog Clin Biol Res 361: 363–385, 1990 [PubMed] [Google Scholar]
- 34. Kintner DB, Su G, Lenart B, Ballard AJ, Meyer JW, Ng LL, Shull GE, Sun D. Increased tolerance to oxygen and glucose deprivation in astrocytes from Na+/H+ exchanger isoform 1 null mice. Am J Physiol Cell Physiol 287: C12–C21, 2004 [DOI] [PubMed] [Google Scholar]
- 35. Kletzien RF, Pariza MW, Becker JE, Potter VR. A method using 3-O-methyl-d-glucose and phloretin for the determination of intracellular water space of cells in monolayer culture. Anal Biochem 68: 537–544, 1975 [DOI] [PubMed] [Google Scholar]
- 36. Kuroiwa T, Miyasaka N, Fengyo Z, Yamada I, Nakane M, Nagaoka T, Tamura A, Ohno K. Experimental ischemic brain edema: morphological and magnetic resonance imaging findings. Neurosurg Focus 22: E11, 2007 [DOI] [PubMed] [Google Scholar]
- 37. Lam TI, Wise PM, O'Donnell ME. Cerebral microvascular endothelial cell Na/H exchange: evidence for the presence of NHE1 and NHE2 isoforms and regulation by arginine vasopressin. Am J Physiol Cell Physiol 297: C278–C289, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Landgraf R. Central release of vasopressin: stimuli, dynamics, consequences. Prog Brain Res 91: 29–39, 1992 [DOI] [PubMed] [Google Scholar]
- 39. Latzkovits L, Cserr HF, Park JT, Patlak CS, Pettigrew KD, Rimanoczy A. Effects of arginine vasopressin and atriopeptin on glial cell volume measured as 3-MG space. Am J Physiol Cell Physiol 264: C603–C608, 1993 [DOI] [PubMed] [Google Scholar]
- 40. Lehmann GL, Gradilone SA, Marinelli RA. Aquaporin water channels in central nervous system. Curr Neurovasc Res 1: 293–303, 2004 [DOI] [PubMed] [Google Scholar]
- 41. Lenart B, Kintner DB, Shull GE, Sun D. Na-K-Cl cotransporter-mediated intracellular Na+ accumulation affects Ca2+ signaling in astrocytes in an in vitro ischemic model. J Neurosci 24: 9585–9597, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Liang D, Bhatta S, Gerzanich V, Simard JM. Cytotoxic edema: mechanisms of pathological cell swelling. Neurosurg Focus 22: E2, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Liu KF, Li F, Tatlisumak T, Garcia JH, Sotak CH, Fisher M, Fenstermacher MD. Regional variations in the apparent diffusion coefficient and the intracellular distribution of water in rat brain during acute focal ischemia. Stroke 32: 1897–1905, 2001 [DOI] [PubMed] [Google Scholar]
- 44. Liu X, Nakayama S, Amiry-Moghaddam M, Ottersen OP, Bhardwaj A. Arginine-vasopressin V1 but not V2 receptor antagonism modulated infarct volume brain water content, and aquaporin-4 expression following experimental stroke. Neurocrit Care 12: 124–131, 2010 [DOI] [PubMed] [Google Scholar]
- 45. MacVicar BA, Feighan D, Brown A, Ransom B. Intrinsic optical signals in the rat optic nerve: role for K(+) uptake via NKCC1 and swelling of astrocytes. Glia 37: 114–123, 2002 [DOI] [PubMed] [Google Scholar]
- 46. McCarthy KD, de Vellis J. Preparation of separate astroglial and oligodendroglial cell cultures from rat cerebral tissue. J Cell Biol 85: 890–902, 1980 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Menzies SA, Betz AL, Hoff JT. Contributions of ions and albumin to the formation and resolution of ischemic brain edema. J Neurosurg 78: 257–266, 1993 [DOI] [PubMed] [Google Scholar]
- 48. O'Donnell ME, Duong V, Suvatne J, Foroutan S, Johnson DM. Arginine vasopressin stimulation of cerebral microvascular endothelial cell Na-K-Cl cotransporter activity is V1 receptor and [Ca] dependent. Am J Physiol Cell Physiol 289: C283–C292, 2005 [DOI] [PubMed] [Google Scholar]
- 49. O'Donnell ME, Lam TI, Tran L, Anderson SE. The role of the blood-brain barrier Na-K-2Cl cotransporter in stroke. Adv Exp Med Biol 559: 67–75, 2004 [DOI] [PubMed] [Google Scholar]
- 50. O'Donnell ME, Lam TI, Tran LQ, Foroutan S, Anderson SE. Estradiol reduces activity of the blood-brain barrier Na-K-Cl cotransporter and decreases edema formation in permanent middle cerebral artery occlusion. J Cereb Blood Flow Metab 26: 1234–1249, 2006 [DOI] [PubMed] [Google Scholar]
- 51. O'Donnell ME, Tran L, Lam TI, Liu XB, Anderson SE. Bumetanide inhibition of the blood-brain barrier Na-K-Cl cotransporter reduces edema formation in the rat middle cerebral artery occlusion model of stroke. J Cereb Blood Flow Metab 24: 1046–1056, 2004 [DOI] [PubMed] [Google Scholar]
- 52. Okuno K, Taya K, Marmarou CR, Ozisik P, Fazzina G, Kleindienst A, Gulsen S, Marmarou A. The modulation of aquaporin-4 by using PKC-activator (phorbol myristate acetate) and V1a receptor antagonist (SR49059) following middle cerebral artery occlusion/reperfusion in the rat. Acta Neurochir Suppl (Wien) 102: 431–436, 2008 [DOI] [PubMed] [Google Scholar]
- 53. Park CK, Jun SS, Cho SH, Kang JK. Assessment of the relationship between ischemic damage and brain swelling in frozen brain slices. Acta Neurochir Suppl (Wien) 70: 17–19, 1997 [DOI] [PubMed] [Google Scholar]
- 54. Pasantes-Morales H, Cruz-Rangel S. Brain volume regulation: osmolytes and aquaporin perspectives. Neuroscience 168: 871–884, 2010 [DOI] [PubMed] [Google Scholar]
- 55. Ribeiro Md, Hirt L, Bogousslavsky J, Regli L, Badaut J. Time course of aquaporin expression after transient focal cerebral ischemia in mice. J Neurosci Res 83: 1231–1240, 2006 [DOI] [PubMed] [Google Scholar]
- 56. Schielke GP, Moises HC, Betz AL. Blood to brain sodium transport and interstitial fluid potassium concentration during early focal ischemia in the rat. J Cereb Blood Flow Metab 11: 466–471, 1991 [DOI] [PubMed] [Google Scholar]
- 57. Shi J, Bui JD, Yang SH, He Z, Lucas TH, Buckley DL, Blackband SJ, King MA, Day AL, Simpkins JW. Estrogens decrease reperfusion-associated cortical ischemic damage: an MRI analysis in a transient focal ischemia model. Stroke 32: 987–992, 2001 [DOI] [PubMed] [Google Scholar]
- 58. Silberstein C, Bouley R, Huang Y, Fang P, Pastor-Soler N, Brown D, Van Hoek AN. Membrane organization and function of M1 and M23 isoforms of aquaporin-4 in epithelial cells. Am J Physiol Renal Physiol 287: F501–F511, 2004 [DOI] [PubMed] [Google Scholar]
- 59. Simpkins JW, Rajakumar G, Zhang YQ, Simpkins CE, Greenwald D, Yu CJ, Bodor N, Day AL. Estrogens may reduce mortality and ischemic damage caused by middle cerebral artery occlusion in the female rat. J Neurosurg 87: 724–730, 1997 [DOI] [PubMed] [Google Scholar]
- 60. Sorensen PS, Gjerris A, Hammer M. Cerebrospinal fluid vasopressin in neurological and psychiatric disorders. J Neurol Neurosurg Psychiatry 48: 50–57, 1985 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Staub F, Peters J, Kempski O, Schneider GH, Schurer L, Baethmann A. Swelling of glial cells in lactacidosis and by glutamate: significance of Cl(-)-transport. Brain Res 610: 69–74, 1993 [DOI] [PubMed] [Google Scholar]
- 62. Su G, Kintner DB, Sun D. Contribution of Na+-K+-Cl− cotransporter to high-[K+]o-induced swelling and EAA release in astrocytes. Am J Physiol Cell Physiol 282: C1136–C1146, 2002 [DOI] [PubMed] [Google Scholar]
- 63. Suzuki S, Brown CM, Wise PM. Mechanisms of neuroprotection by estrogen. Endocrine 29: 209–215, 2006 [DOI] [PubMed] [Google Scholar]
- 64. Tomas-Camardiel M, Venero JL, Herrera AJ, De Pablos RM, Pintor-Toro JA, Machado A, Cano J. Blood-brain barrier disruption highly induces aquaporin-4 mRNA and protein in perivascular and parenchymal astrocytes: protective effect by estradiol treatment in ovariectomized animals. J Neurosci Res 80: 235–246, 2005 [DOI] [PubMed] [Google Scholar]
- 65. Toung TJ, Traystman RJ, Hurn PD. Estrogen-mediated neuroprotection after experimental stroke in male rats. Stroke 29: 1666–1670, 1998 [DOI] [PubMed] [Google Scholar]
- 66. Vakill A, Kataoka H, Plesnilla N. Role of arginine vasopressin V1 and V2 receptors for brain damage after transient focal ischemia. J Cereb Blood Flow Metab 25: 1012–1019, 2005 [DOI] [PubMed] [Google Scholar]
- 67. Walls AB, Sickmann HM, Brown A, Bouman SD, Ransom B, Schousboe A, Waagepetersen HS. Characterization of 1.4-dideoxy-1, 4-imino-D-arabinitol. (DAB) as an inhibitor of brain glycogen shunt activity. J Neurochem 105: 1462–1470, 2008 [DOI] [PubMed] [Google Scholar]
- 68. Wise PM, Dubal DB. Estradiol protects against ischemic brain injury in middle-aged rats. Biol Reprod 63: 982–985, 2000 [DOI] [PubMed] [Google Scholar]
- 69. Wise PM, Dubal DB, Wilson ME, Rau SW, Bottner M, Rosewell KL. Estradiol is a protective factor in the adult and aging brain: understanding of mechanisms derived from in vivo and in vitro studies. Brain Res Brain Res Rev 37: 313–319, 2001 [DOI] [PubMed] [Google Scholar]